Structural and Corrosion Study of Uncoated and Zn-Cu Coated Magnesium-Based Alloy
Abstract
:1. Introduction
2. Experimental Section
3. Results and Discussion
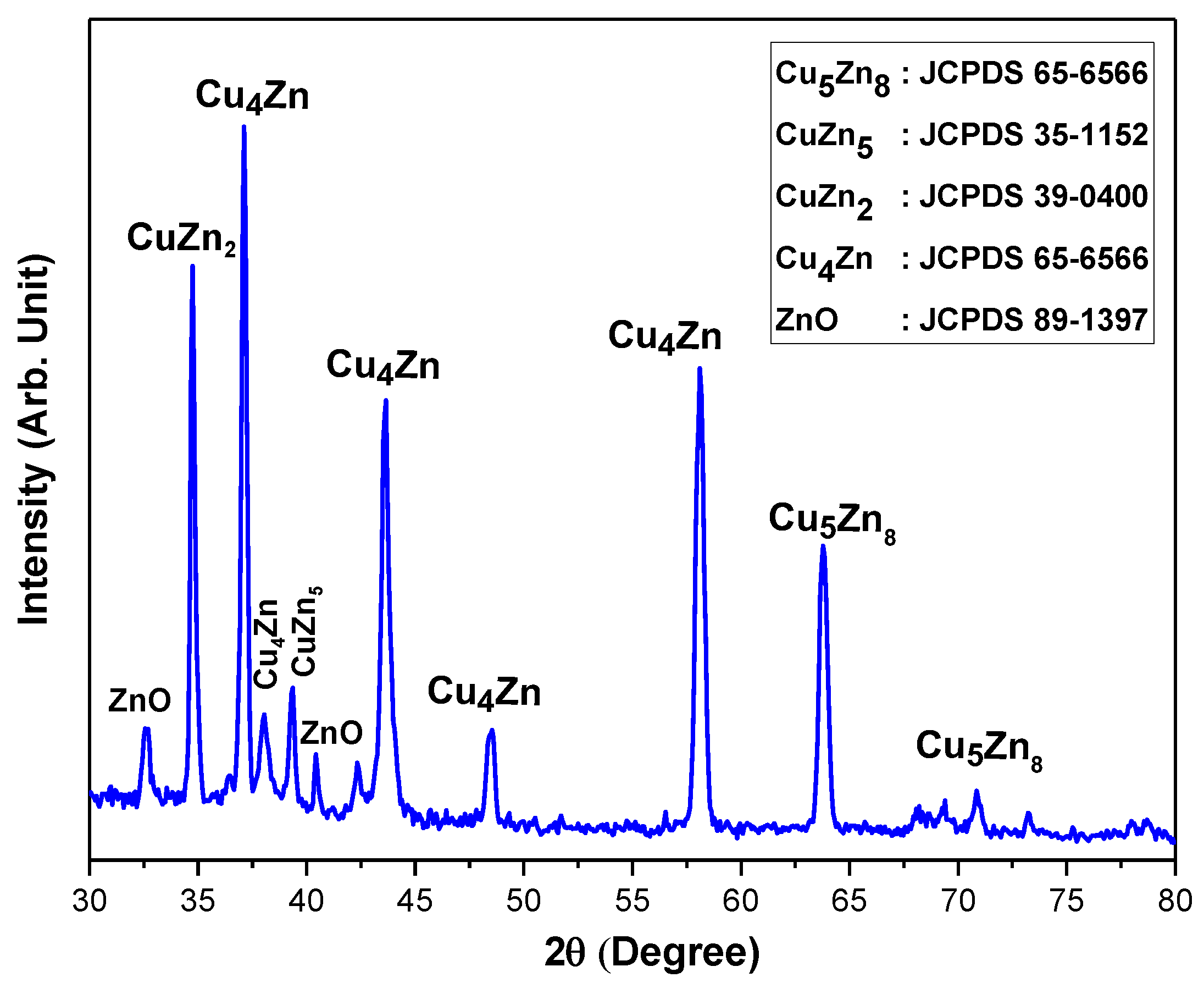
3.1. X-ray Diffraction Analysis
3.2. Scanning Electron Microscope Analysis
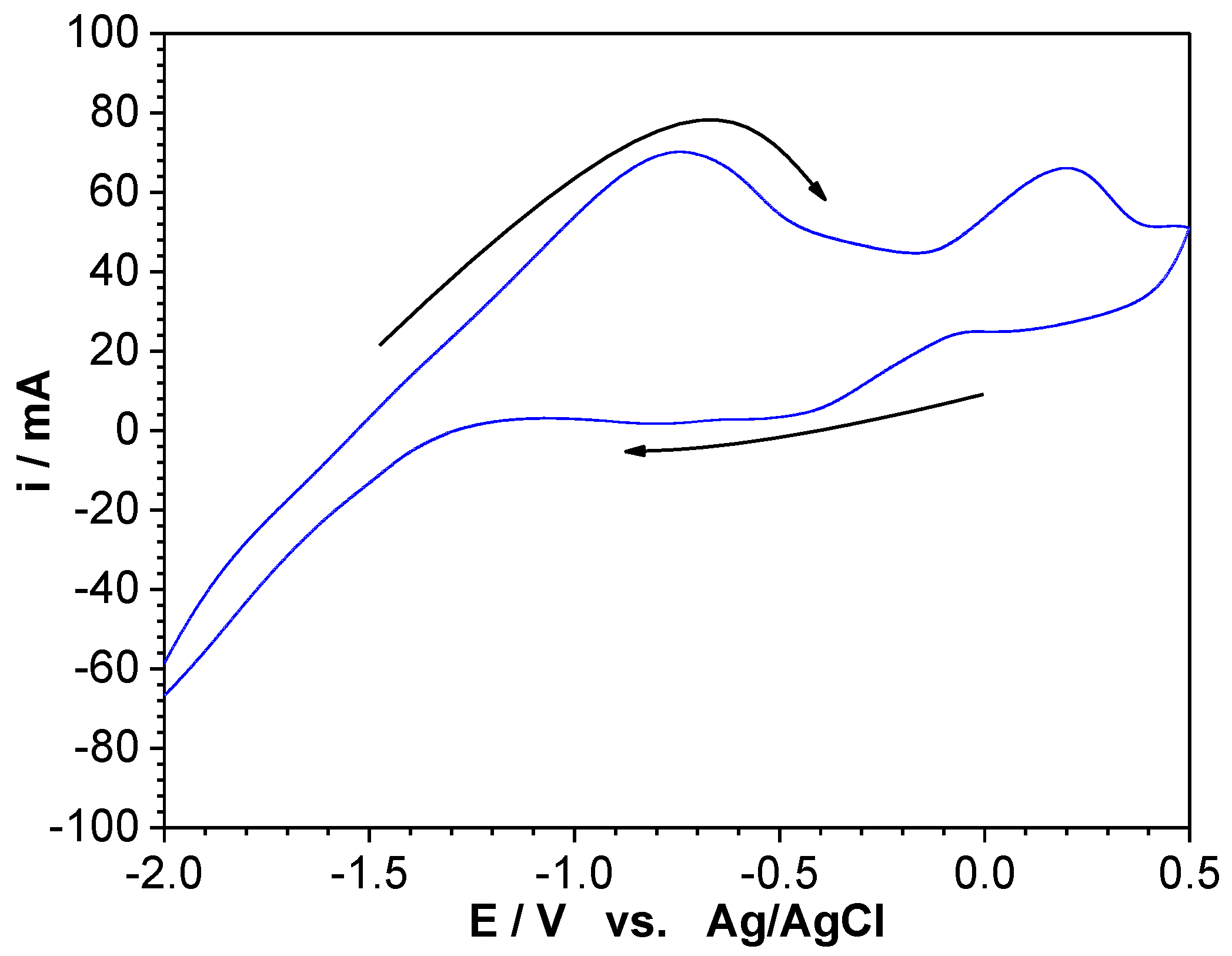
3.3. Cyclic Voltammetry Analysis
3.4. Linear Sweep Voltammetry Analyses of Uncoated and Zn-Cu Coated AZ63
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gupta, M.; Sharon, N.M.L. Magnesium, Magnesium Alloys, and Magnesium Composites; John Wiley & Sons: Somerset, NJ, USA, 2011; pp. 1–11. [Google Scholar]
- Forsmark, J.H.; Dowling, Z.; Gibson, K.; Mueller, C.; Godlewski, L.; Zindel, J.; Boileau, J. An investigation of the effects of cast skin on the mechanical properties of an AM60 die-cast magnesium alloy. SAE Int. J. Mater. Manuf. 2015, 8, 714–721. [Google Scholar] [CrossRef]
- Candan, S.; Unal, M.; Turkmen, M.; Koc, E.; Turen, Y.; Candan, E. Improvement of mechanical and corrosion properties of magnesium alloy by lead addition. Mater. Sci. Eng. A 2009, 501, 115–118. [Google Scholar] [CrossRef]
- Fu, B.; Qin, G.; Li, F.; Meng, X.; Zhang, J.; Wu, C. Friction stir welding process of dissimilar metals of 6061-T6 aluminum alloy to AZ31B magnesium alloy. J. Mater. Process.Technol. 2015, 218, 38–47. [Google Scholar] [CrossRef]
- Mishra, R.S.; Ma, Z. Friction stir welding and processing. Mater. Sci. Eng. R Rep. 2005, 50, 1–78. [Google Scholar] [CrossRef]
- Song, G.; Atrens, A.; Stjohn, D.; Nairn, J.; Li, Y. The electrochemical corrosion of pure magnesium in 1 N NaCl. Corros. Sci. 1997, 39, 855–875. [Google Scholar] [CrossRef]
- Song, G.L.; Atrens, A. Corrosion mechanisms of magnesium alloys. Adv. Eng. Mater. 1999, 1, 11–33. [Google Scholar] [CrossRef]
- Inoue, H.; Sugahara, K.; Yamamoto, A.; Tsubakino, H. Corrosion rate of magnesium and its alloys in buffered chloride solutions. Corros. Sci. 2002, 44, 603–610. [Google Scholar] [CrossRef]
- Liu, L.; Ren, D.; Liu, F. A review of dissimilar welding techniques for magnesium alloys to aluminum alloys. Materials 2014, 7, 3735–3757. [Google Scholar] [CrossRef]
- Commin, L.; Dumont, M.; Masse, J.E.; Barrallier, L. Friction stir welding of AZ31 magnesium alloy rolled sheets: Influence of processing parameters. Acta Mater. 2009, 57, 326–334. [Google Scholar] [CrossRef]
- Laser, T.; Nürnberg, M.R.; Janz, A.; Hartig, C.; Letzig, D.; Schmid-Fetzer, R.; Bormann, R. The influence of manganese on the microstructure and mechanical properties of AZ31 gravity die cast alloys. Acta Mater. 2006, 54, 3033–3041. [Google Scholar] [CrossRef]
- Song, G.; Atrens, A. Understanding magnesium corrosion—A framework for improved alloy performance. Adv. Eng. Mater. 2003, 5, 837–858. [Google Scholar] [CrossRef]
- Ambat, R.; Aung, N.N.; Zhou, W. Evaluation of microstructural effects on corrosion behaviour of AZ91D magnesium alloy. Corros. Sci. 2000, 42, 1433–1455. [Google Scholar] [CrossRef]
- Thomas, S.; Medhekar, N.; Frankel, G.; Birbilis, N. Corrosion mechanism and hydrogen evolution on Mg. Curr. Opin. Solid State Mater. Sci. 2015, 19, 85–94. [Google Scholar] [CrossRef]
- Ročňáková, I.; Montufar, E.B.; Horynová, M.; Zikmund, T.; Novotný, K.; Klakurková, L.; Čelko, L.; Song, G.-L.; Kaiser, J. Assessment of localized corrosion under simulated physiological conditions of magnesium samples with heterogeneous microstructure: Value of X-ray computed micro-tomography platform. Corros. Sci. 2016, 104, 187–196. [Google Scholar] [CrossRef]
- Song, G.; Atrens, A.; St. John, D.; Wu, X.; Nairn, J. The anodic dissolution of magnesium in chloride and sulphate solutions. Corros. Sci. 1997, 39, 1981–2004. [Google Scholar] [CrossRef]
- Song, G.; Atrens, A.; Wu, X.; Zhang, B. Corrosion behaviour of AZ21, AZ501 and AZ91 in sodium chloride. Corros. Sci. 1998, 40, 1769–1791. [Google Scholar] [CrossRef]
- Ambat, R.; Aung, N.N.; Zhou, W. Studies on the influence of chloride ion and pH on the corrosion and electrochemical behaviour of AZ91D magnesium alloy. J. Appl. Electrochem. 2000, 30, 865–874. [Google Scholar] [CrossRef]
- Altun, H.; Sen, S. Studies on the influence of chloride ion concentration and pH on the corrosion and electrochemical behaviour of AZ63 magnesium alloy. Mater. Des. 2004, 25, 637–643. [Google Scholar] [CrossRef]
- Gray, J.; Luan, B. Protective coatings on magnesium and its alloys—A critical review. J. Alloy. Compd. 2002, 336, 88–113. [Google Scholar] [CrossRef]
- Song, G.; Johannesson, B.; Hapugoda, S.; StJohn, D. Galvanic corrosion of magnesium alloy AZ91D in contact with an aluminium alloy, steel and zinc. Corros. Sci. 2004, 46, 955–977. [Google Scholar] [CrossRef]
- Lian, J.S.; Li, G.Y.; Niu, L.Y.; Gu, C.D.; Jiang, Z.H.; Jiang, Q. Electroless Ni–P deposition plus zinc phosphate coating on AZ91D magnesium alloy. Surf. Coat. Technol. 2006, 200, 5956–5962. [Google Scholar] [CrossRef]
- Ebrahimzadeh, M.; Gholami, M.; Momeni, M.; Kosari, A.; Moayed, M.; Davoodi, A. Theoretical and experimental investigations on corrosion control of 65Cu–35Zn brass in nitric acid by two thiophenol derivatives. Appl. Surf. Sci. 2015, 332, 384–392. [Google Scholar] [CrossRef]
- Jie, H.; Xu, Q.; Wei, L.; Min, Y. Etching and heating treatment combined approach for superhydrophobic surface on brass substrates and the consequent corrosion resistance. Corros. Sci. 2016, 102, 251–258. [Google Scholar] [CrossRef]
- Banerjee, T.; Allmand, A. Experiments on the electrodeposition of brass from cyanide solutions. Trans. Faraday Soc. 1948, 44, 819–833. [Google Scholar] [CrossRef]
- Vagramyan, T.; Leach, J.; Moon, J. On the problems of electrodepositing brass from non-cyanide electrolytes. Electrochim. Acta 1979, 24, 231–236. [Google Scholar] [CrossRef]
- Schütte, K.; Meyer, H.; Gemel, C.; Barthel, J.; Fischer, R.A.; Janiak, C. Synthesis of Cu, Zn and Cu/Zn brass alloy nanoparticles from metal amidinate precursors in ionic liquids or propylene carbonate with relevance to methanol synthesis. Nanoscale 2014, 6, 3116–3126. [Google Scholar] [CrossRef] [PubMed]
- De Vreese, P.; Skoczylas, A.; Matthijs, E.; Fransaer, J.; Binnemans, K. Electrodeposition of copper–zinc alloys from an ionic liquid-like choline acetate electrolyte. Electrochim. Acta 2013, 108, 788–794. [Google Scholar] [CrossRef]
- Survila, A.; Mockus, Z.; Kanapeckaitė, S.; Stalnionis, G.; Juškėnas, R.; Jasulaitienė, V. Codeposition of zinc and copper in gluconate-sulfate solutions. J. Electrochem. Soc. 2013, 160, D428–D433. [Google Scholar] [CrossRef]
- De Almeida, M.R.H.; Barbano, E.P.; Zacarin, M.G.; de Brito, M.M.; Tulio, P.C.; Carlos, I.A. Electrodeposition of cuzn films from free-of-cyanide alkaline baths containing edta as complexing agent. Surf. Coat. Technol. 2016, 287, 103–112. [Google Scholar] [CrossRef]
- De Almeida, M.; Barbano, E.; de Carvalho, M.; Carlos, I.; Siqueira, J.; Barbosa, L. Electrodeposition of copper–zinc from an alkaline bath based on edta. Surf. Coat. Technol. 2011, 206, 95–102. [Google Scholar] [CrossRef]
- Juškėnas, R.; Karpavičienė, V.; Pakštas, V.; Selskis, A.; Kapočius, V. Electrochemical and xrd studies of Cu–Zn coatings electrodeposited in solution with d-mannitol. J. Electroanal. Chem. 2007, 602, 237–244. [Google Scholar] [CrossRef]
- Domínguez-Ríos, C.; Moreno, M.; Torres-Sánchez, R.; Antúnez, W.; Aguilar-Elguézabal, A.; González-Hernández, J. Effect of tartrate salt concentration on the morphological characteristics and composition of Cu-Zn electroless plating on zamak 5 zinc alloy. Surf. Coat. Technol. 2008, 202, 4848–4854. [Google Scholar] [CrossRef]
- Ibrahim, M.A.; Bakdash, R.S. New cyanide-free ammonia bath for brass alloy coatings on steel substrate by electrodeposition. Int. J. Electrochem. Sci. 2015, 10, 9666–9677. [Google Scholar]
- Johannsen, K.; Page, D.; Roy, S. A systematic investigation of current efficiency during brass deposition from a pyrophosphate electrolyte using RDE, RCE, and QCM. Electrochim. Acta 2000, 45, 3691–3702. [Google Scholar] [CrossRef]
- De Almeida, M.; Barbano, E.; de Carvalho, M.; Tulio, P.; Carlos, I. Copper–zinc electrodeposition in alkaline-sorbitol medium: Electrochemical studies and structural, morphological and chemical composition characterization. Appl. Surf. Sci. 2015, 333, 13–22. [Google Scholar] [CrossRef]
- Kumar, M.P.; Nidhi, M.; Srivastava, C. Electrochemical exfoliation of graphite to produce graphene using tetrasodium pyrophosphate. RSC Adv. 2015, 5, 24846–24852. [Google Scholar] [CrossRef]
- Sylla, D.; Savall, C.; Gadouleau, M.; Rebere, C.; Creus, J.; Refait, P. Electrodeposition of Zn–Mn alloys on steel using an alkaline pyrophosphate-based electrolytic bath. Surf. Coat. Technol. 2005, 200, 2137–2145. [Google Scholar] [CrossRef]
- Therese, G.H.A.; Kamath, P.V. Electrochemical synthesis of metal oxides and hydroxides. Chem. Mater. 2000, 12, 1195–1204. [Google Scholar] [CrossRef]
- Konno, H.; Nagayama, M. Mechanism of electrodeposition of copper from cupric pyrophosphate solutions. Electrochim. Acta 1977, 22, 353–358. [Google Scholar] [CrossRef]
- Kravtsov, V.; Kondratiev, V. Kinetics and mechanism of pyrophosphate metal complexes electroreduction. Electrochim. Acta 1991, 36, 427–434. [Google Scholar] [CrossRef]
- Narkevicius, A.; Bucinskiene, D.; Samuleviciene, M.; Ramanauskas, R. Corrosion behaviour of Zn coatings electrodeposited from alkaline and acid solutions. Trans. Inst. Met. Finish. 2003, 81, 93–97. [Google Scholar]
- Carlos, I.A.; de Almeida, M.R.H. Study of the influence of the polyalcohol sorbitol on the electrodeposition of copper–zinc films from a non-cyanide bath. J. Electroanal. Chem. 2004, 562, 153–159. [Google Scholar] [CrossRef]
- El Meguid, E.A.; Awad, N.K. Electrochemical pitting corrosion behaviour of α-brass in LiBr containing solutions. Corros. Sci. 2009, 51, 1134–1139. [Google Scholar] [CrossRef]
- Haciibrahimoglu, M.Y.; Yavuz, A.; Oztas, M.; Bedir, M. Electrochemical and structural study of zinc-rich brass deposited from pyrophosphate electroyte onto the carbon steel. Dig. J. Nanomater. Biostruct. 2016, 11, 251–262. [Google Scholar]
- Ballesteros, J.; Torres-Martínez, L.; Juárez-Ramírez, I.; Trejo, G.; Meas, Y. Study of the electrochemical co-reduction of Cu2+ and Zn2+ ions from an alkaline non-cyanide solution containing glycine. J. Electroanal. Chem. 2014, 727, 104–112. [Google Scholar] [CrossRef]
- Zeng, R.-C.; Zhang, J.; Huang, W.-J.; Dietzel, W.; Kainer, K.; Blawert, C.; Wei, K. Review of studies on corrosion of magnesium alloys. Trans. Nonferr. Met. Soc. China 2006, 16, s763–s771. [Google Scholar] [CrossRef]
- Pardo, A.; Merino, M.C.; Coy, A.E.; Arrabal, R.; Viejo, F.; Matykina, E. Corrosion behaviour of magnesium/aluminium alloys in 3.5 wt. % NaCl. Corros. Sci. 2008, 50, 823–834. [Google Scholar] [CrossRef]
- Stern, M.; Geary, A.L. Electrochemical polarization I. A theoretical analysis of the shape of polarization curves. J. Electrochem. Soc. 1957, 104, 56–63. [Google Scholar] [CrossRef]
- Liang, J.; Srinivasan, P.B.; Blawert, C.; Dietzel, W. Comparison of electrochemical corrosion behaviour of MgO and ZrO2 coatings on AM50 magnesium alloy formed by plasma electrolytic oxidation. Corros. Sci. 2009, 51, 2483–2492. [Google Scholar] [CrossRef]
- Sorensen, C.D.; Nelson, T.W. Friction stir welding of ferrous and nickel alloys. Frict. Stir Weld. Process. 2007. [Google Scholar] [CrossRef]
- Lee, W.-B.; Jung, S.-B. The joint properties of copper by friction stir welding. Mater. Lett. 2004, 58, 1041–1046. [Google Scholar] [CrossRef]
- Feliu, S.; Samaniego, A.; Barranco, V.; El-Hadad, A.A.; Llorente, I.; Adeva, P. The effect of low temperature heat treatment on surface chemistry and corrosion resistance of commercial magnesium alloys AZ31 and AZ61 in 0.6 M NaCl solution. Corros. Sci. 2014, 80, 461–472. [Google Scholar] [CrossRef]

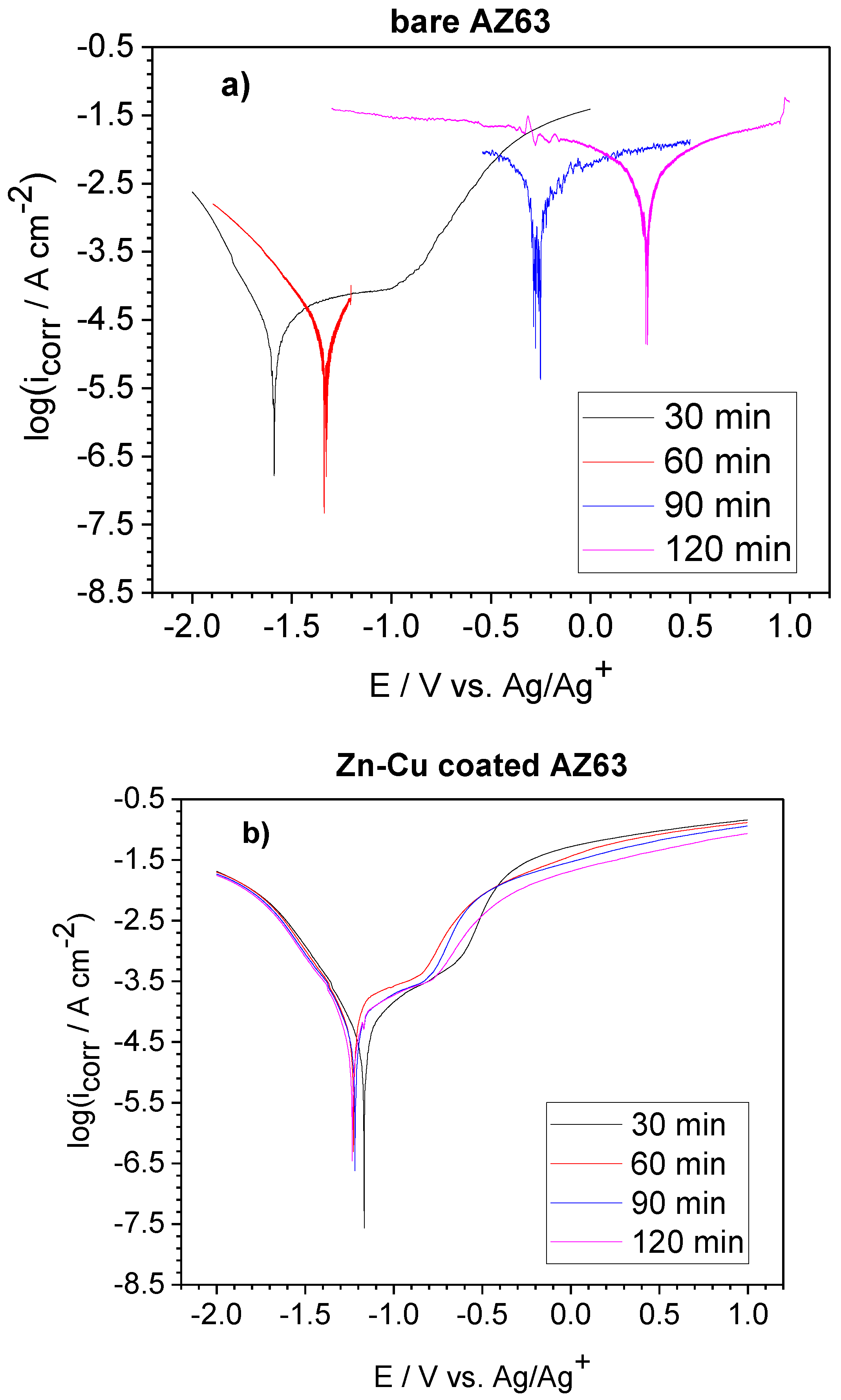
| Duration in 3 wt % NaCl | Uncoated AZ63 Alloy | Zn-Cu Coated AZ63 Alloy | ||||
|---|---|---|---|---|---|---|
| Ecorr (V) | icorr (µA·cm−2) | Rp (Ω·cm−2) | Ecorr (V) | icorr (µA·cm−2) | Rp (Ω·cm−2) | |
| 30 min | −1.61 | 19.1 | 4336 | −1.17 | 26.3 | 1791 |
| 60 min | −1.31 | 26.9 | 1957 | −1.23 | 70.8 | 1082 |
| 90 min | −0.23 | 2754 | 17.8 | −1.22 | 46.8 | 1664 |
| 120 min | +0.29 | 4677 | 18.5 | −1.23 | 49.0 | 1663 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hacıibrahimoğlu, M.Y.; Bedir, M.; Yavuz, A. Structural and Corrosion Study of Uncoated and Zn-Cu Coated Magnesium-Based Alloy. Metals 2016, 6, 322. https://doi.org/10.3390/met6120322
Hacıibrahimoğlu MY, Bedir M, Yavuz A. Structural and Corrosion Study of Uncoated and Zn-Cu Coated Magnesium-Based Alloy. Metals. 2016; 6(12):322. https://doi.org/10.3390/met6120322
Chicago/Turabian StyleHacıibrahimoğlu, Mehmet Yakup, Metin Bedir, and Abdulcabbar Yavuz. 2016. "Structural and Corrosion Study of Uncoated and Zn-Cu Coated Magnesium-Based Alloy" Metals 6, no. 12: 322. https://doi.org/10.3390/met6120322
APA StyleHacıibrahimoğlu, M. Y., Bedir, M., & Yavuz, A. (2016). Structural and Corrosion Study of Uncoated and Zn-Cu Coated Magnesium-Based Alloy. Metals, 6(12), 322. https://doi.org/10.3390/met6120322






