Influence of Mn in Balancing the Tensile and Electrical Conductivity Properties of Al-Mg-Si Alloy
Abstract
1. Introduction
2. Materials and Experiment Procedures
2.1. Sample Preparation
2.2. Microscopic Morphology Characterization
2.3. Mechanical Properties Test
2.4. Electrical Conductivity Measurement
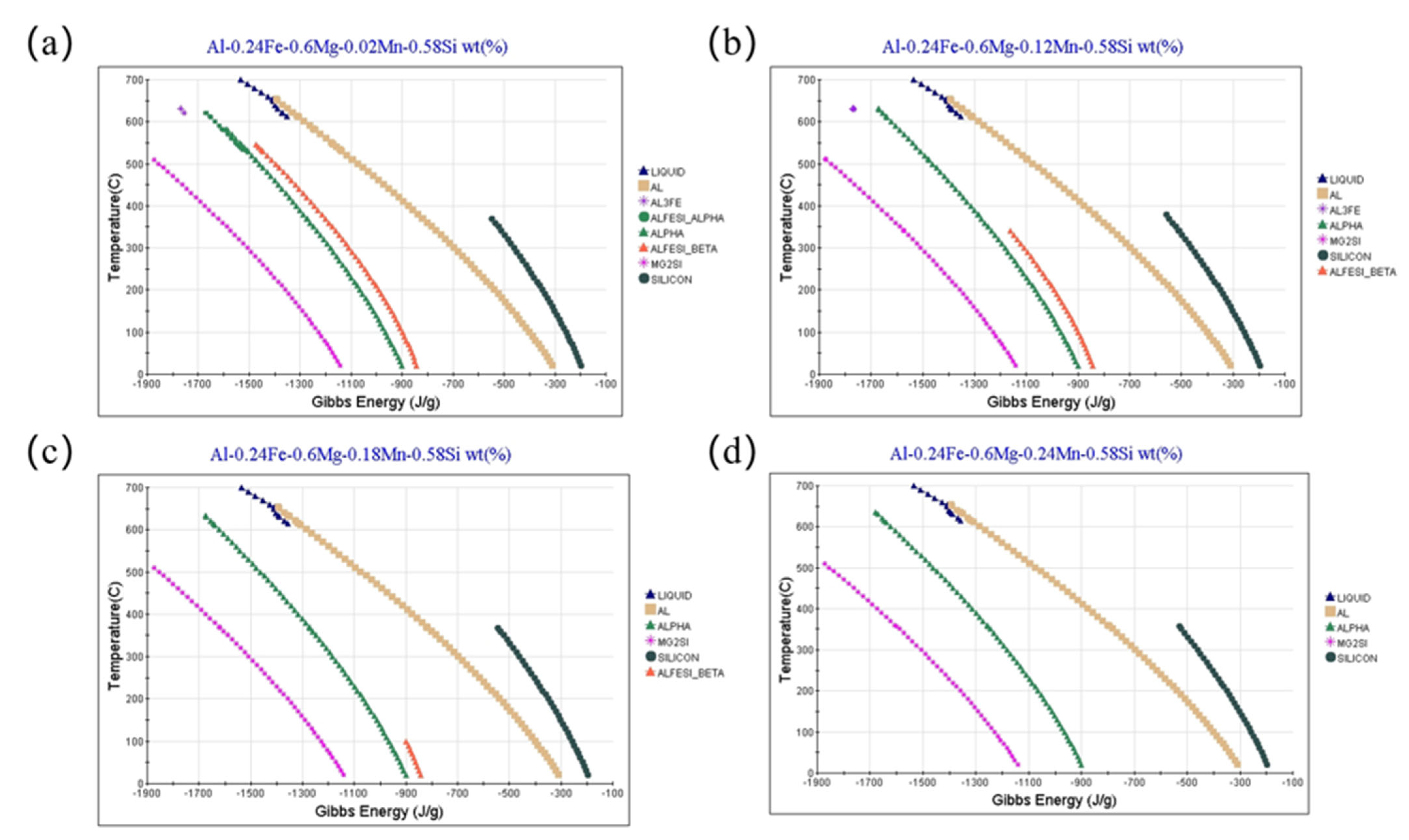
2.5. Thermodynamic Calculation
3. Results
3.1. Microstructure Evolution of As-Cast Alloys
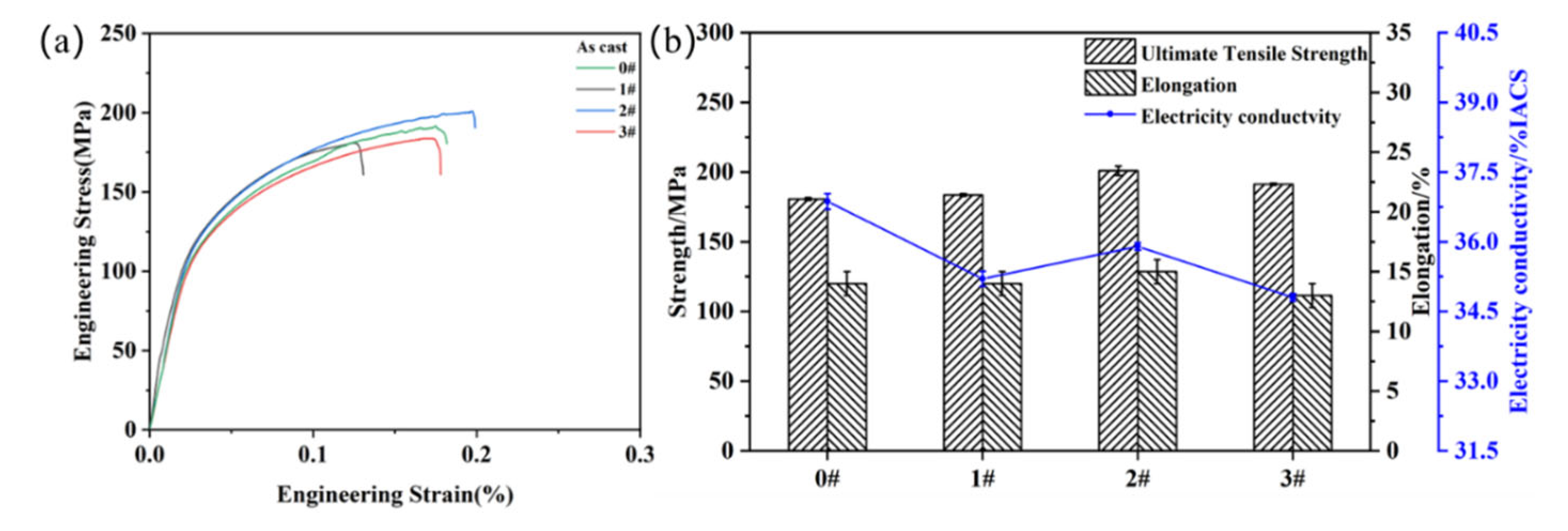
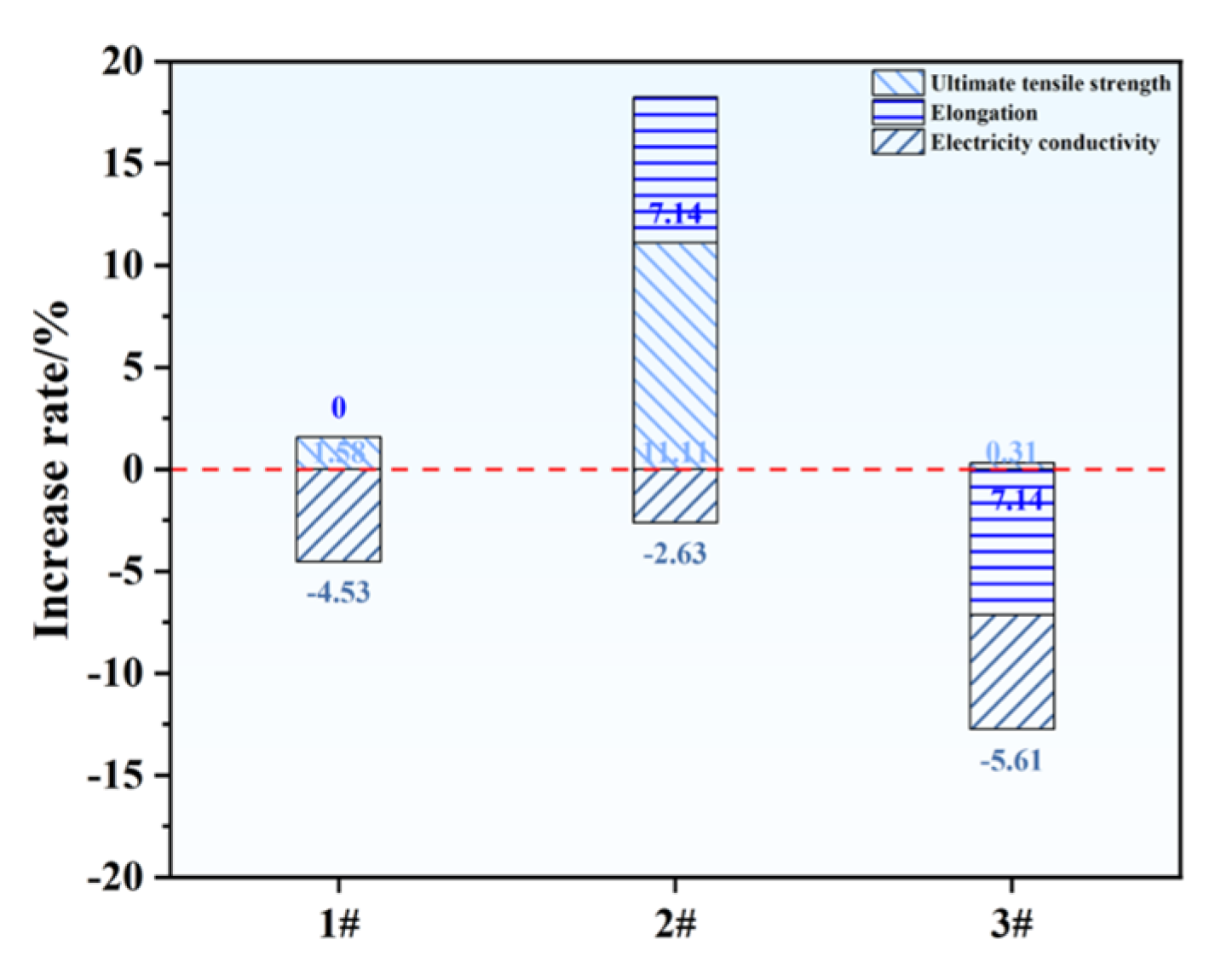
3.2. Properties of As-Cast Alloys
3.3. Microstructure Evolution of Hot-Extruded Alloys
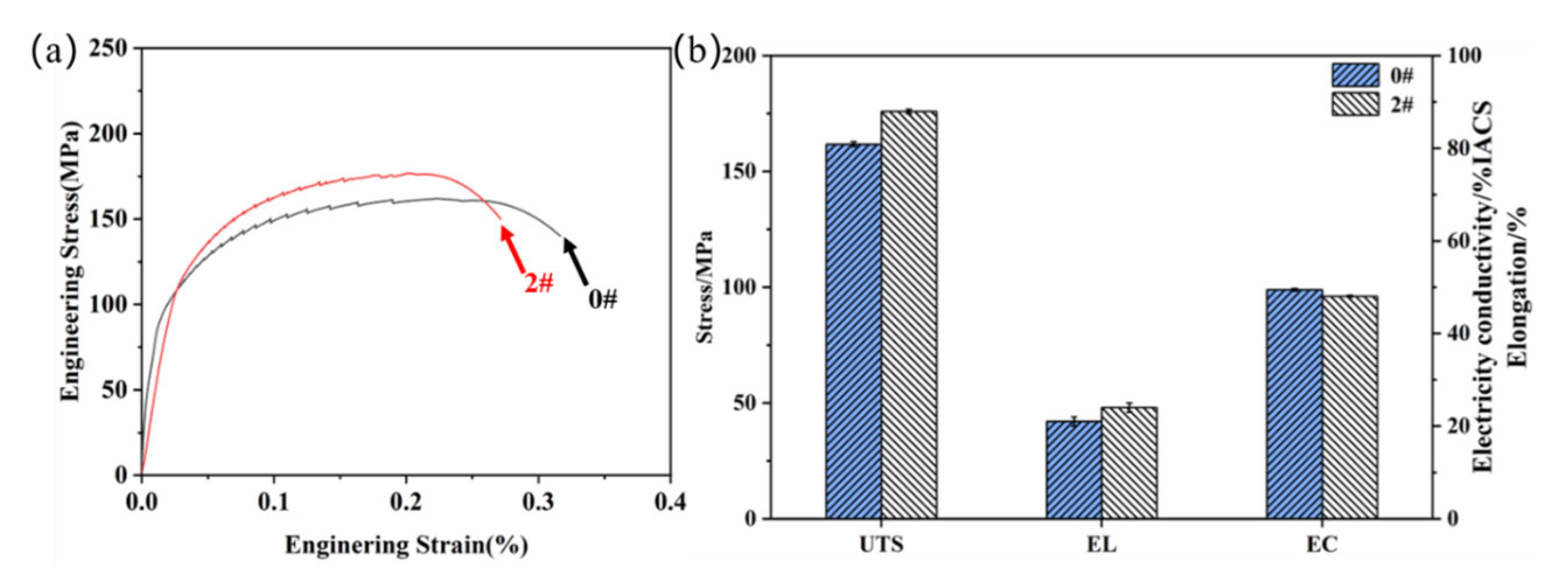
3.4. Properties of Hot-Extruded Alloys
4. Discussion
4.1. Formation Mechanism and Morphological Evolution of Fe-Rich Phase
4.2. Strengthening Mechanisms
4.3. Conductivity Mechanisms
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sauvage, X.; Bobruk, E.V.; Murashkin, M.Y.; Nasedkina, Y.; Enikeev, N.A.; Valiec, R.Z. Optimization of electrical conductivity and strength combination by structure design at the nanoscale in Al–Mg–Si alloys. Acta Mater. 2015, 98, 355–366. [Google Scholar] [CrossRef]
- Kalombo, R.B.; Martínez, J.M.G.; Ferreira, J.L.A.; da Silva, C.R.M.; Araújo, J.A. Comparative fatigue resistance of overhead conductors made of aluminium and aluminium alloy: Tests and analysis. Procedia Eng. 2015, 133, 223–232. [Google Scholar] [CrossRef]
- Karabay, S. Modification of AA-6201 alloy for manufacturing of high conductivity and extra high conductivity wires with property of high tensile stress after artificial aging heat treatment for all-aluminium alloy conductors. Mater. Des. 2006, 27, 821–832. [Google Scholar] [CrossRef]
- Koch, S.; Antrekowitsch, H. Aluminum Alloys for Wire Harnesses in Automotive Engineering. Berg-Huttenmaenn. Monatsh. 2007, 152, 62–67. [Google Scholar]
- Zhao, Q.R.; Qian, Z.; Cui, X.L.; Wu, Y.Y.; Liu, X.F. Optimizing microstructures of dilute Al–Fe–Si alloys designed with enhanced electrical conductivity and tensile strength. J. Alloys Compd. 2015, 650, 768–776. [Google Scholar] [CrossRef]
- Hansen, V.; Hauback, B.; Sundberg, M.; Rømming, C.; Gjønnes, J. β-Al4.5FeSi: A combined synchrotron powder diffraction, electron diffraction, high-resolution electron microscopy and single-crystal X-ray diffraction study of a faulted structure. Acta Crystallogr. Sec. B Struct. Sci. 1998, 54, 351–357. [Google Scholar] [CrossRef]
- Liu, T.L.; Huang, G.R.; Sun, Y.M.; Zhang, L.; Huang, Z.W.; Wang, J.J.; Liu, C.Z. Effect of Mn and Fe on the formation of Fe- and Mn-rich intermetallics in Al–5Mg–Mn alloys solidified under near-rapid cooling. Materials 2016, 9, 88. [Google Scholar] [CrossRef]
- Tang, H.-P.; Wang, Q.-D.; Lei, C.; Wang, K.; Ye, B.; Jiang, H.-Y.; Ding, W.-J. Effect of Cooling Rate on Microstructure and Mechanical Properties of Sand-Casted Al–5.0Mg–0.6Mn–0.25Ce Alloy. Acta Metall. Sin. (Engl. Lett.) 2019, 32, 1549–1564. [Google Scholar] [CrossRef]
- Cui, X.L.; Li, X.H.; Ye, H.; Cui, H.W.; Pan, Y.K.; Feng, R.; Wu, Y.Y.; Liu, X.F. Study on microstructure characterization, electrical conductivity and mechanical property improvement mechanisms of a novel Al-Si-Mg-Fe-Cu alloy. J. Alloys Compd. 2021, 885, 160959. [Google Scholar] [CrossRef]
- Marioara, C.D.; Andersen, S.J.; Jansen, J.; Zandbergen, H.W. Atomic model for GP-zones in a 6082 Al–Mg–Si system. Acta Mater. 2001, 49, 321–328. [Google Scholar] [CrossRef]
- Zhang, L.F.; Gao, J.W.; Damoah, L.N.W.; Robertson, D.G. Removal of iron from aluminum: A review. Miner. Process. Extr. Metall. Rev. 2012, 33, 99–157. [Google Scholar] [CrossRef]
- Deng, Y.; Yin, Z.; Zhao, K.; Duan, J.; He, Z. Effects of Sc and Zr microalloying additions on the microstructure and mechanical properties of new Al–Zn–Mg alloys. J. Alloys Compd. 2012, 530, 71–88. [Google Scholar] [CrossRef]
- Pramod, S.L.; Ravikiran; Prasada Rao, A.K.; Murty, B.S.; Bakshi, S.R. Effect of Sc addition and T6 aging treatment on the microstructure and mechanical properties of Al–4.2Mg–0.3Cu alloy. Mater. Sci. Eng. A 2016, 674, 438–450. [Google Scholar] [CrossRef]
- Shabestari, S.G. The effect of iron and manganese on the formation of intermetallic compounds in aluminum–silicon alloys. J. Mater. Sci. Eng. A 2004, 383, 289–298. [Google Scholar] [CrossRef]
- Wang, E.R.; Hui, X.D.; Chen, G.L. Eutectic Al–Si–Cu–Fe–Mn alloys with enhanced mechanical properties at room and elevated temperature. Mater. Des. 2011, 32, 4333–4340. [Google Scholar] [CrossRef]
- Wang, G.D.; Tian, N.; He, C.S.; Zhao, G.; Zuo, L. Formation of Second-Phases in a Direct-Chill Casting Al-12Si-0.65Mg-xMn Alloy. Acta Metall. Sin. 2018, 54, 1059–1067. [Google Scholar] [CrossRef]
- Wang, X.F.; Guo, M.X.; Zhang, J.S.; Zhuang, L.Z. Effect of Zn addition on the microstructure and mechanical properties of Al–Mg–Si–Cu alloys. Mater. Sci. Eng. A 2016, 677, 522–533. [Google Scholar] [CrossRef]
- Sreeja Kumari, S.S.; Pillai, R.M.; Rajan, T.P.D.; Pai, B.C. Effects of individual and combined additions of Be, Mn, Ca and Sr on the solidification behaviour, structure and mechanical properties of Al–7Si–0.3Mg–0.8Fe alloy. Mater. Sci. Eng. A 2007, 460, 561–573. [Google Scholar] [CrossRef]
- Zheng, Y.; Bian, L.P.; Ji, H.L.; Liu, X.W.; Tian, F. Influence of Ca and Mn on Microstructure, Mechanical Properties, and Electrical Conductivity of As-Cast and Heat-Treated Al-Mg-Si Alloys. Rare Met. Mater. Eng. 2022, 51, 4010–4020. [Google Scholar]
- GB/T 228-2010; Metallic Materials—Tensile Testing at Ambient Temperature. Standardization Administration of China: Beijing, China, 2010.
- Claves, S.R.; Elias, D.L.; Misiolek, W.Z. Analysis of the Intermetallic Phase Transformation Occurring during Homogenization of 6xxx Aluminum Alloys. Mater. Sci. Forum. 2002, 396, 667–674. [Google Scholar] [CrossRef]
- Donnadieu, P.; Lapasset, G.; Sanders, T.H. Manganese-induced ordering in the α-(Al-Mn-Fe-Si) approximant phase. Philos. Mag. Lett. 1994, 70, 319–326. [Google Scholar] [CrossRef]
- Liu, X.; Jia, H.L.; Wang, C.; Wu, X.; Zha, M.; Wang, H.Y. Enhancing mechanical properties of twin-roll cast Al–Mg–Si–Fe alloys by regulating Fe-bearing phases and macro-segregation. Mater. Sci. Eng. A 2022, 831, 142256. [Google Scholar] [CrossRef]
- Závodská, D.; Tillová, E.; Švecová, I.; Kuchariková, L. Secondary cast Al-alloys with higher content of iron. Mater. Today Proc. 2018, 5, 26680–26686. [Google Scholar] [CrossRef]
- Que, Z.P.; Mendis, C.L. Formation of θ-Al13Fe4 and the multi-step phase transformations to α-Al8Fe2Si, β-Al5FeSi and δ-Al4FeSi2 in Al–20Si-0.7Fe alloy. Intermetallics 2020, 127, 106960. [Google Scholar] [CrossRef]
- Chang, X.S.; Chen, G.; Sun, W.; Zhang, H.M.; Chu, G.N.; Zhang, X.; Han, F.; Zhang, W.C.; Du, Z.M. Microstructures, mechanical properties and solidification mechanism of a hot tearing sensitive aluminum alloy asymmetric part fabricated by squeeze casting. J. Alloys Compd. 2021, 886, 161254. [Google Scholar] [CrossRef]
- Pedersen, K.O.; Westermann, I.; Furu, T.; Børvik, T.; Hopperstad, O.S. Influence of microstructure on work-hardening and ductile fracture of aluminium alloys. Mater. Des. 2015, 70, 31–44. [Google Scholar] [CrossRef]
- Mikolajczak, P.; Ratke, L. Interplay between melt flow and the 3D distribution and morphology of Fe-rich phases in AlSi alloys. Metall. Mater. Trans. A 2015, 46, 1312–1327. [Google Scholar] [CrossRef]
- Li, Z.G.; Zhang, Z.J.; Zhou, G.W.; Zhao, P.; Jia, Z.H.; Poole, W.J. The effect of Mg and Si content on the microstructure, texture and bendability of Al–Mg–Si alloys. Mater. Sci. Eng. A 2021, 814, 141199. [Google Scholar] [CrossRef]
- Chen, H.L.; Chen, Q.; Bratberg, J.; Engström, A. Development and applications of the TCAL aluminum alloy database. Today Proc. 2015, 2, 4939–4948. [Google Scholar] [CrossRef]
- Kuijpers, N.C.W.; Kool, W.H.; Koenis, P.T.G.; Nilsen, K.E.; Todd, I.; van der Zwaag, S. Assessment of different techniques for quantification of α-Al(FeMn)Si and β-Al5FeSi intermetallics in AA 6xxx alloys. Mater. Charact. 2002, 49, 409–420. [Google Scholar] [CrossRef]
- Poole, W.J.; Wang, X.; Embury, J.D.; Lioyd, D.J. The effect of manganese on the microstructure and tensile response of an Al-Mg-Si alloy. Mater. Sci. Eng. A 2019, 755, 307–317. [Google Scholar] [CrossRef]
- Que, Z.P.; Wang, Y.; Fan, Z.Y. Formation of the Fe-containing intermetallic compounds during solidification of Al-5Mg-2Si-0.7 Mn-1.1 Fe alloy. Metall. Mater. Trans. A 2018, 49, 2173–2181. [Google Scholar] [CrossRef]
- Bradley, A.J.; Thewlis, J. The crystal structure of α-manganese. Proc. R. Soc. A 1927, 115, 456–471. [Google Scholar]
- Taylor, R.M.; Jones, R.M. Constitution and magnetic properties of iron-rich iron-aluminum alloys. J. Phys. Chem. Solids 1958, 6, 16–37. [Google Scholar] [CrossRef]
- Chen, Q.; Sundman, B. Modeling of thermodynamic properties for Bcc, Fcc, liquid, and amorphous iron. J. Phase Equilibria 2001, 22, 631–644. [Google Scholar] [CrossRef]
- Lodgaard, L.; Ryum, N. Precipitation of dispersoids containing Mn and/or Cr in Al–Mg–Si alloys. Mater. Sci. Eng. A 2000, 283, 144–152. [Google Scholar] [CrossRef]
- Jiang, S.Y.; Wang, R.H. Grain size-dependent Mg/Si ratio effect on the microstructure and mechanical/electrical properties of Al-Mg-Si-Sc alloys. J. Mater. Sci. Technol. 2019, 35, 1354–1363. [Google Scholar] [CrossRef]
- Moghanaki, S.K.; Kazeminezhad, M. Modeling of the mutual effect of dynamic precipitation and dislocation density in age hardenable aluminum alloys. J. Alloys Compd. 2016, 683, 527–532. [Google Scholar] [CrossRef]
- McElfresh, C.; Cui, Y.N.; Dudarev, S.L.; Po, G. Discrete stochastic model of point defect-dislocation interaction for simulating dislocation climb. Int. J. Plast. 2021, 136, 102848. [Google Scholar] [CrossRef]
- Lv, J.X.; Zheng, J.H.; Yardley, V.A.; Shi, Z.S.; Lin, J.G. A review of microstructural evolution and modelling of aluminium alloys under hot forming conditions. Metals 2020, 10, 1516. [Google Scholar] [CrossRef]
- Wang, Y.X.; Zhao, H.; Chen, X.R.; Gault, B.; Brechet, Y.; Hutchinson, C. The effect of shearable clusters and precipitates on dynamic recovery of Al alloys. Acta Mater. 2024, 265, 119643. [Google Scholar] [CrossRef]
- Wei, T.; Wang, Y.D.; Tang, Z.H.; Xiao, S.F. The constitutive modeling and processing map of homogenized Al-Mg-Si-Cu-Zn alloy. Mater. Today Commun. 2021, 27, 102471. [Google Scholar]
- Tong, M.M.; Jiang, F.; Wang, H.L.; Jiang, J.Y.; Wu, M.J.; Tang, Z.Q. The evolutions of flow stress and microstructure of Al-Mg-Mn-Sc-Zr alloy at elevated temperatures. Mater. Charact. 2021, 182, 111560. [Google Scholar] [CrossRef]
- Hou, J.P.; Li, R.; Wang, Q.; Yu, H.Y.; Zhang, Z.J.L.; Chen, Q.Y.; Ma, H.; Wu, X.M.; Li, X.W.; Zhang, Z.F. Three principles for preparing Al wire with high strength and high electrical conductivity. J. Mater. Sci. Technol. 2019, 35, 742–751. [Google Scholar] [CrossRef]
- Raeisinia, B.; Poole, W.J.; Lloyd, D.J. Examination of precipitation in the aluminum alloy AA6111 using electrical resistivity measurements. J. Mater. Sci. Eng. A 2006, 420, 245–249. [Google Scholar] [CrossRef]
- Ma, K.K.; Wen, H.M.; Hu, T.; Toppng, T.D.; Isheim, D.; Seidman, D.N.; Lavernia, E.J. Mechanical behavior and strengthening mechanisms in ultrafine grain precipitation-strengthened aluminum alloy. Acta Mater. 2014, 62, 141–155. [Google Scholar] [CrossRef]
- Orlova, T.S.; Mavlyutov, A.M.; Bondarenko, A.S.; Kasatkin, I.A.; Murashkin, M.Y.; Valiev, R.Z. Influence of grain boundary state on electrical resistivity of ultrafine grained aluminium. Philos. Mag. 2016, 96, 2429–2444. [Google Scholar] [CrossRef]
- Guan, R.; Shen, Y.F.; Zhao, Z.Y.; Wang, X. A high-strength, ductile Al-0.35 Sc-0.2 Zr alloy with good electrical conductivity strengthened by coherent nanosized-precipitates. J. Mater. Sci. Technol. 2017, 33, 215–223. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, L.J.; Niu, G.D.; Mao, J. Conductive Al alloys: The contradiction between strength and electrical conductivity. Adv. Eng. Mater. 2021, 23, 2001249. [Google Scholar] [CrossRef]





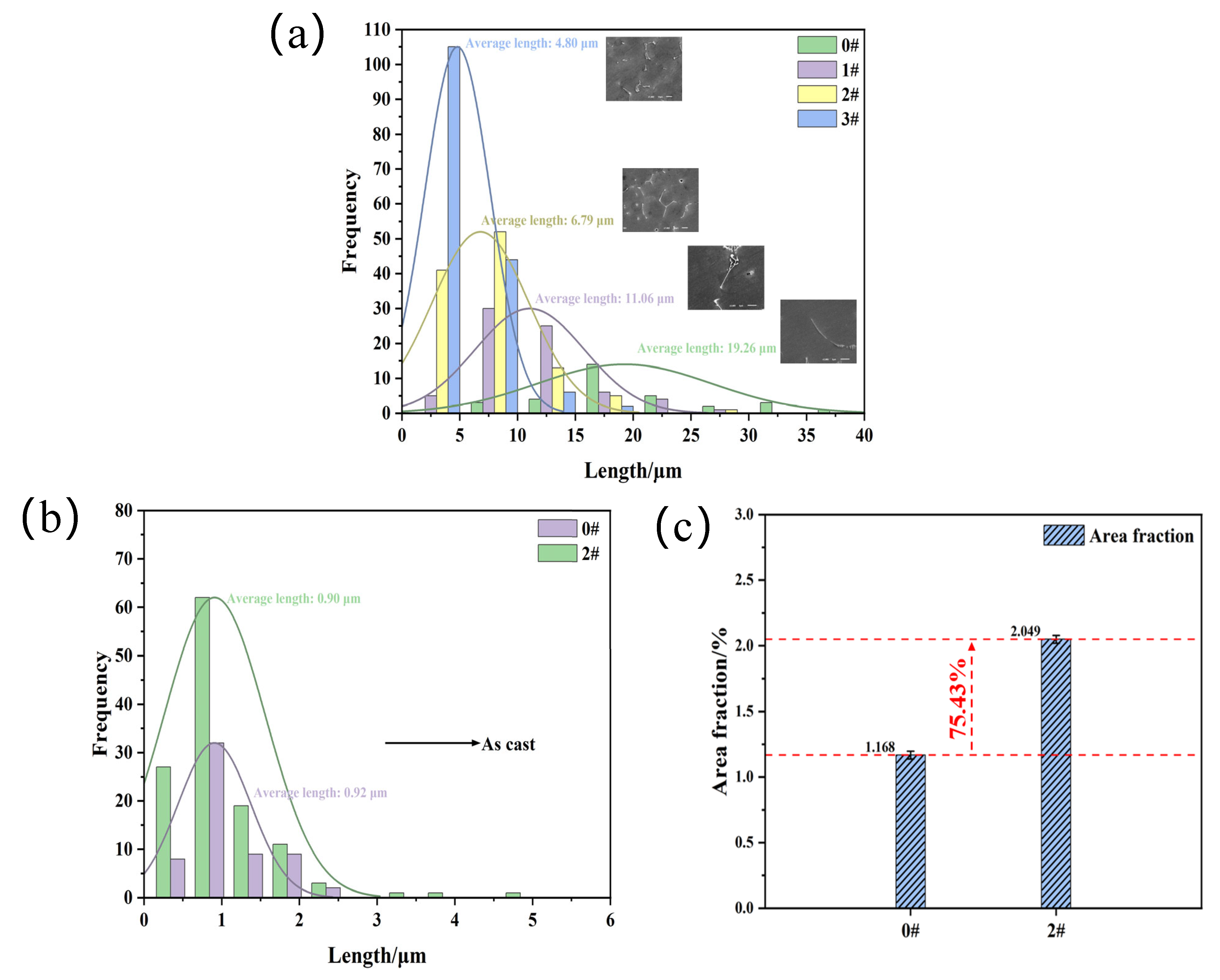

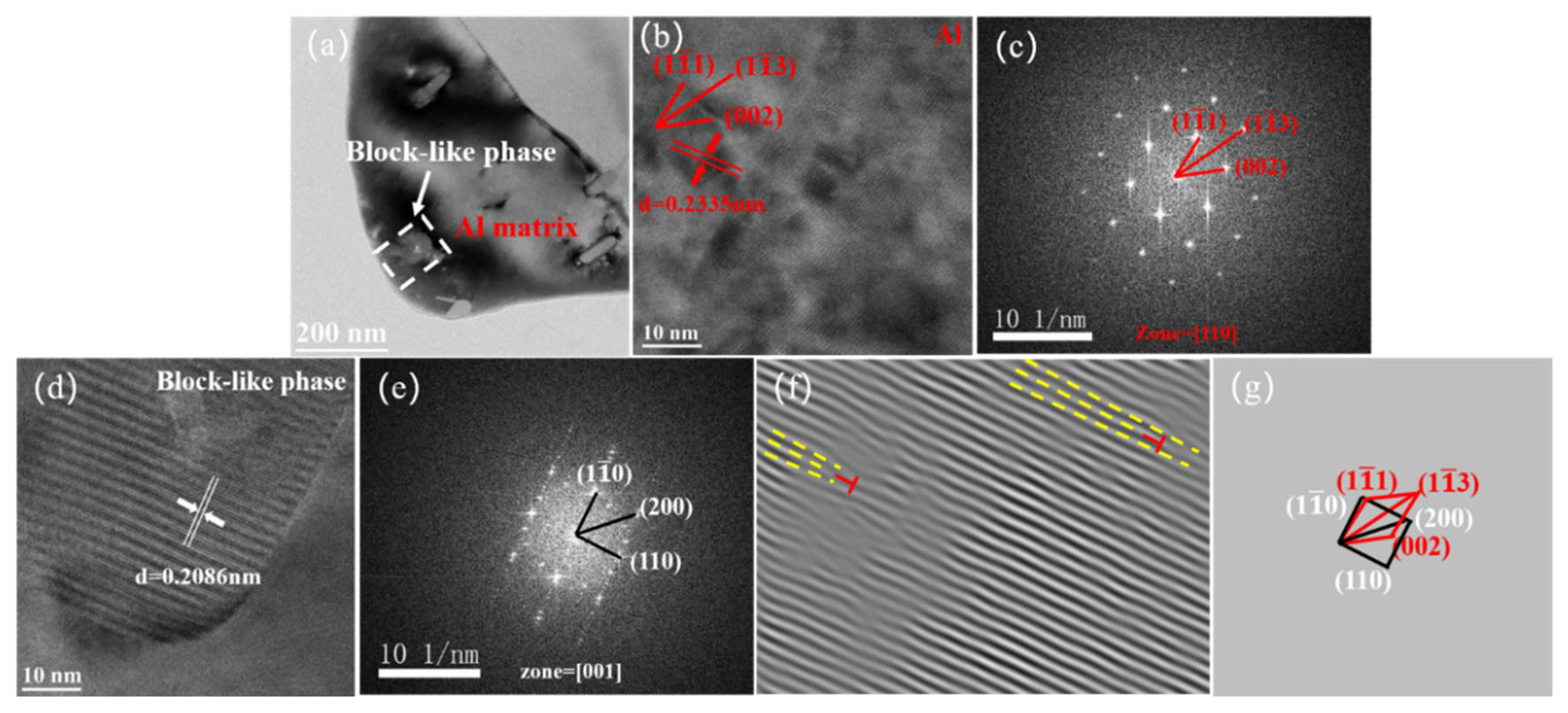


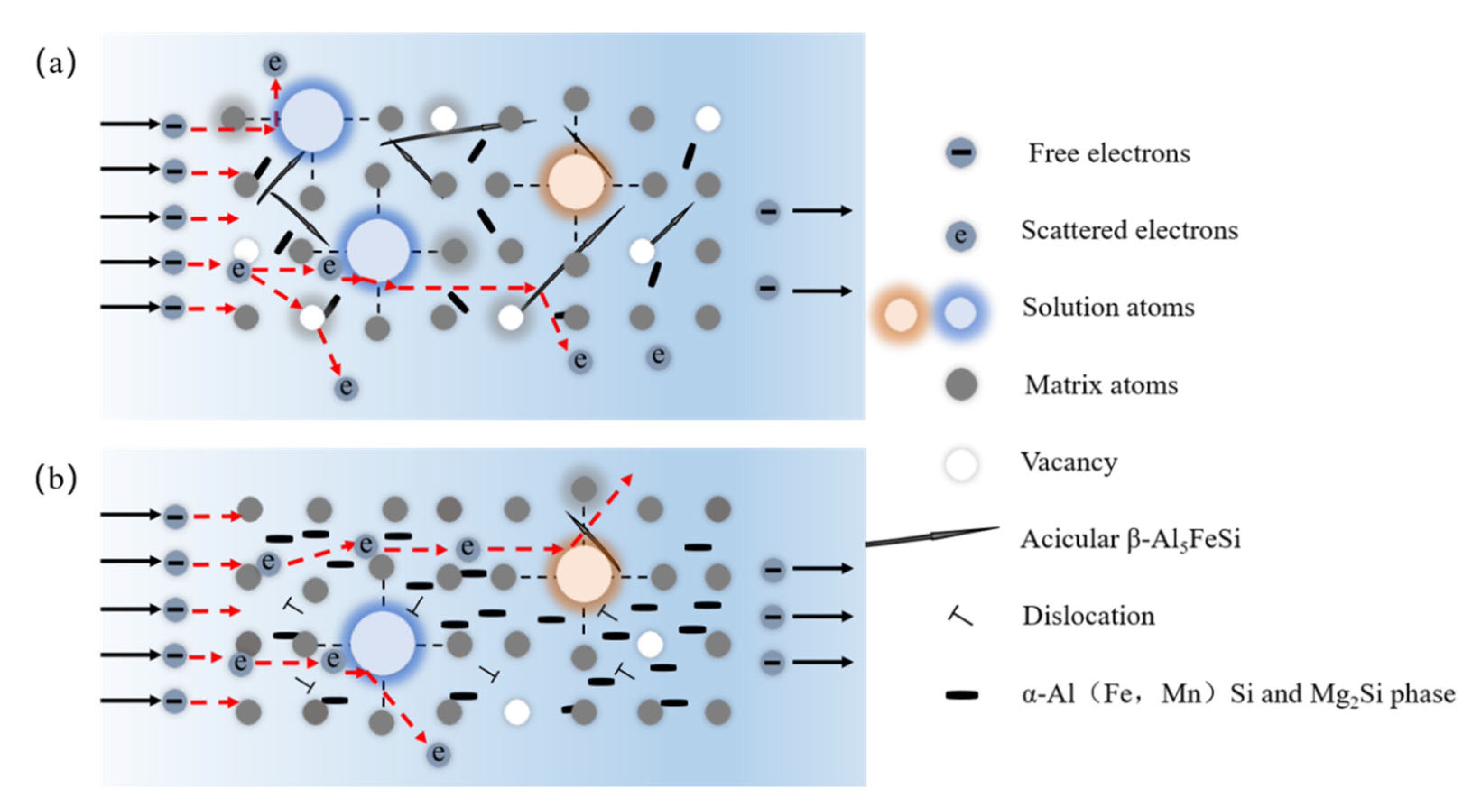
| Mg | Si | Fe | Mn | Ti | Cr | Others | Al | |
|---|---|---|---|---|---|---|---|---|
| 0# | 0.6 | 0.58 | 0.24 | 0.02 | 0.02 | 0.01 | ≤0.10 | Bal |
| 1# | 0.6 | 0.58 | 0.24 | 0.12 | 0.02 | 0.01 | ≤0.10 | Bal |
| 2# | 0.6 | 0.58 | 0.24 | 0.18 | 0.02 | 0.01 | ≤0.10 | Bal |
| 3# | 0.6 | 0.58 | 0.24 | 0.24 | 0.02 | 0.01 | ≤0.10 | Bal |
| Element | ||||||
|---|---|---|---|---|---|---|
| Mg | Al | Si | Mn | Fe | (Fe, Mn)/Si | |
| 1 | 0.68 | 96.94 | 1.13 | 0.13 | 1.13 | 1.12 |
| 2 | 0.48 | 94.91 | 2.14 | 0.33 | 2.04 | 1.11 |
| 3 | 0.59 | 90.44 | 3.65 | 1.28 | 4.04 | 1.46 |
| 4 | 0.53 | 92.30 | 2.62 | 1.09 | 3.05 | 1.58 |
| 5 | 1.58 | 80.36 | 6.59 | 3.99 | 7.47 | 1.74 |
| Alloy | 0# | 1# | 2# | 3# |
|---|---|---|---|---|
| Gibbs Energy/(J/g) | −1472.17 | −1161.56 | −897.74 | - |
| Temperature/°C | 545.85 | 341.24 | 99.72 | - |
| Element | Mg | Si | Mn | Fe |
|---|---|---|---|---|
| Solid solubility | 14.9 | 1.65 | 1.82 | 0.042 |
| 0.54 | 1.02 | 2.94 | 2.56 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, J.; Wang, J.; Ding, J.; Wang, Y.; Qi, W. Influence of Mn in Balancing the Tensile and Electrical Conductivity Properties of Al-Mg-Si Alloy. Metals 2025, 15, 923. https://doi.org/10.3390/met15080923
He J, Wang J, Ding J, Wang Y, Qi W. Influence of Mn in Balancing the Tensile and Electrical Conductivity Properties of Al-Mg-Si Alloy. Metals. 2025; 15(8):923. https://doi.org/10.3390/met15080923
Chicago/Turabian StyleHe, Jiaxing, Jiangbo Wang, Jian Ding, Yao Wang, and Wenshu Qi. 2025. "Influence of Mn in Balancing the Tensile and Electrical Conductivity Properties of Al-Mg-Si Alloy" Metals 15, no. 8: 923. https://doi.org/10.3390/met15080923
APA StyleHe, J., Wang, J., Ding, J., Wang, Y., & Qi, W. (2025). Influence of Mn in Balancing the Tensile and Electrical Conductivity Properties of Al-Mg-Si Alloy. Metals, 15(8), 923. https://doi.org/10.3390/met15080923





