Kinetic Study on the Dehydration Behavior of Titanium Dioxide as a Denitration Catalyst Carrier
Abstract
:1. Introduction
2. Experiment
2.1. Raw Materials
2.2. Experimental Procedure
2.3. Kinetic Theory
3. Results and Discussion
3.1. TG Analysis
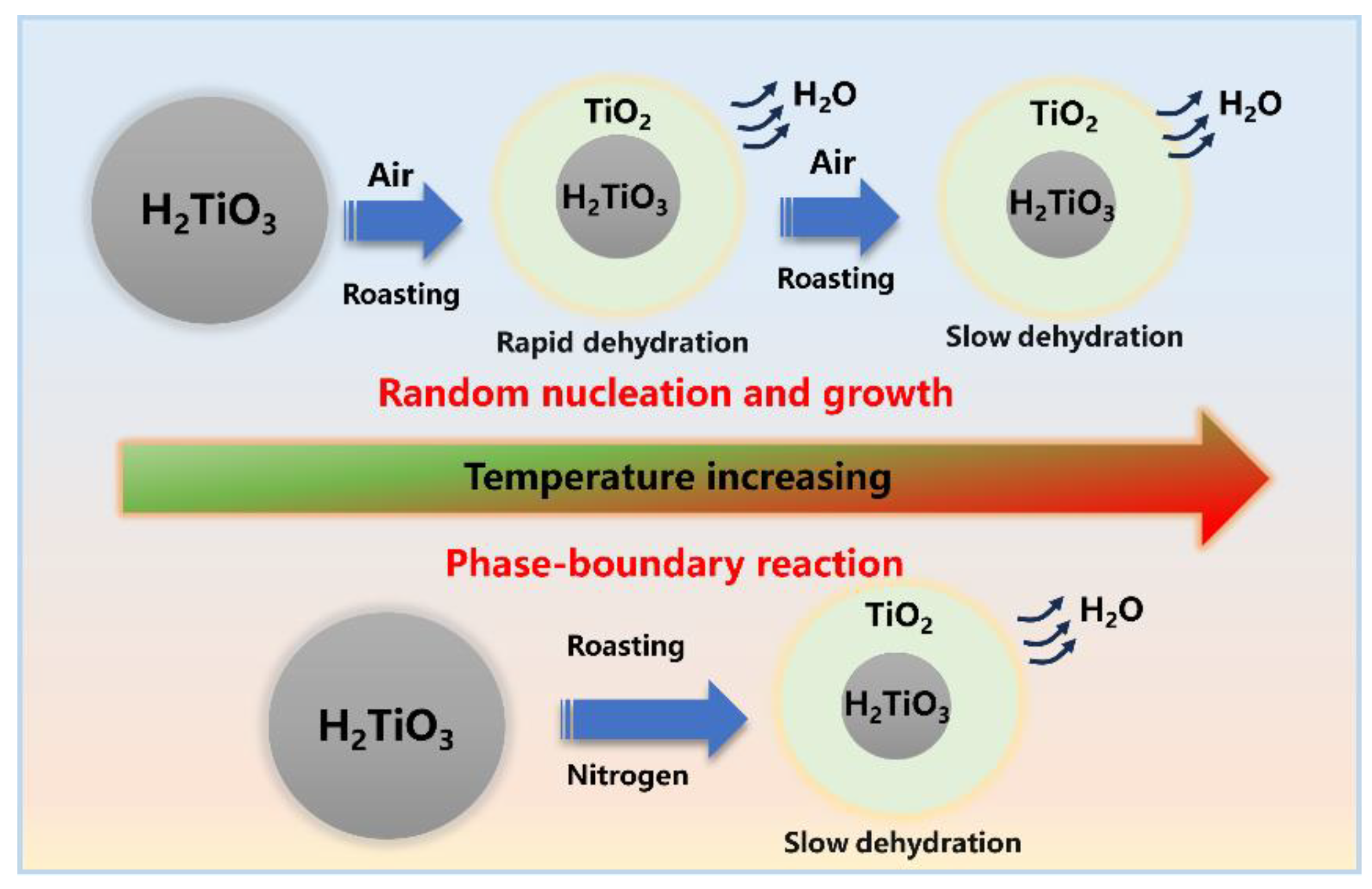
3.2. Dynamic Analysis of Dehydration Behavior
3.3. Kinetic Mechanism Analysis
3.4. Composition Anazlysis of Carrier Titanium Dioxide
4. Conclusions
- (1)
- The dehydration behavior of denitration catalyst carrier titanium dioxide is simple and belongs to the general decomposition reaction. The heating rate is positively correlated with dehydration speed such that the higher the rate of temperature increase, the more favorable the dehydration reaction is.
- (2)
- The dehydration reaction of denitration catalyst carrier titanium dioxide is related to the atmosphere. In an air atmosphere (oxygen-enriched atmosphere), dehydration occurred quickly, followed by slow dehydration until the reaction was fully realized. In an oxygen-free ambient atmosphere (nitrogen atmosphere), the reaction proceeded slowly.
- (3)
- Kinetic calculations of the dehydration behavior of denitration catalyst carrier titanium dioxide were carried out using the mode-free and mode function methods. The analysis revealed that the dehydration behavior of denitration catalyst carrier titanium dioxide under the air atmosphere conformed to the Avrami–Erofeev equation. Dehydration behavior was also affected by crystalline phase transformation and grain nucleation growth of denitration catalyst carrier titanium dioxide at higher-temperature environments. The chemical composition of denitration catalyst carrier TiO2 was characterized as TiO2·0.2H2O·0.06SO3.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gesenhues, U. Calcination of metatitanic acid to titanium dioxide white pigments. Chem. Eng. Technol. 2001, 24, 685–694. [Google Scholar] [CrossRef]
- Lee, H.J.; Shim, J.W.; Lee, J.J.; Lee, W.J. The Encapsulation of Natural Organic Dyes on TiO2 for Photochromism Control. Int. J. Mol. Sci. 2023, 24, 7860. [Google Scholar] [CrossRef] [PubMed]
- Kumarasinghe, K.; Rajapakse, R.M.G.; Tennakone, K.; Kumara, G.R.A. Stability and efficiency improvement of TiO2-based dye-sensitized solar cells by surface modification of MgO. J. Solid State Electrochem. 2023. [Google Scholar] [CrossRef]
- Li, G.J.; Huang, R.; Zhu, C.Y.; Jia, G.; Zhang, S.L.; Zhong, Q. Effect of oxygen vacancies and its quantity on photocatalytic oxidation performance of titanium dioxide for NO removal. Colloids Surf. A Physicochem. Eng. Asp. 2021, 614, 126156. [Google Scholar] [CrossRef]
- Wu, X.W.; Wu, D.J.; Liu, X.J. Optical investigation on sulfur-doping effects in titanium dioxide nanoparticles. Appl. Phys. A Mater. Sci. Process. 2009, 97, 243–248. [Google Scholar] [CrossRef]
- Man, Y.K.; Mu, L.Y.; Wang, Y.; Lin, S.H.; Rempel, G.L.; Pan, Q.M. Synthesis and Characterization of Rutile Titanium Dioxide/Polyacrylate Nanocomposites for Applications in Ultraviolet Light-Shielding Materials. Polym. Compos. 2015, 36, 8–16. [Google Scholar] [CrossRef]
- Kang, X.L.; Liu, S.H.; Dai, Z.D.; He, Y.P.; Song, X.Z.; Tan, Z.Q. Titanium Dioxide: From Engineering to Applications. Catalysts 2019, 9, 191. [Google Scholar] [CrossRef]
- Gopinath, K.P.; Madhav, N.V.; Krishnan, A.; Malolan, R.; Rangarajan, G. Present applications of titanium dioxide for the photocatalytic removal of pollutants from water: A review. J. Environ. Manag. 2020, 270, 110906. [Google Scholar] [CrossRef]
- Li Puma, G.L.; Bono, A.; Krishnaiah, D.; Collin, J.G. Preparation of titanium dioxide photocatalyst loaded onto activated carbon support using chemical vapor deposition: A review paper. J. Hazard. Mater. 2008, 157, 209–219. [Google Scholar] [CrossRef]
- Hu, J.G.; Tang, H.B.; Lin, X.D.; Luo, Z.K.; Cao, H.Q.; Li, Q.W.; Liu, Y.; Long, J.H.; Wang, P. Doped Titanium Dioxide Films Prepared by Pulsed Laser Deposition Method. Int. J. Photoenergy 2012, 2012, 758539. [Google Scholar] [CrossRef]
- Ragadhita, R.; Nandiyanto, A.B.D.; Maulana, A.C.; Oktiani, R.; Sukmafitri, A.; Machmud, A.; Surachman, E. Techo-economic analysis for the production of titanium dioxide nanoparticle produced by liquid-phase synthesis method. J. Eng. Sci. Technol. 2019, 14, 1639–1652. [Google Scholar]
- Zanardo, D.; Ghedini, E.; Menegazzo, F.; Cattaruzza, E.; Manzoli, M.; Cruciani, G.; Signoretto, M. Titanium Dioxide-Based Nanocomposites for Enhanced Gas-Phase Photodehydrogenation. Materials 2019, 12, 3093. [Google Scholar] [CrossRef] [PubMed]
- Masuda, Y.; Seo, W.S.; Koumoto, K. Selective deposition and micropatterning of titanium dioxide on self-assembled monolayers from a gas phase. Langmuir 2001, 17, 4876–4880. [Google Scholar] [CrossRef]
- Li, Y.T.; Yi, H.H.; Tang, X.L.; Liu, X.; Wang, Y.; Cui, B.C.; Zhao, S.Z. Study on the performance of simultaneous desulfurization and denitrification of Fe3O4-TiO2 composites. Chem. Eng. J. 2016, 304, 89–97. [Google Scholar] [CrossRef]
- Tian, C.X. Hydrothermal preparation of high purity TiO2 from industrial metatitanic acid by response surface methodology. Sci. Rep. 2022, 12, 20164. [Google Scholar] [CrossRef]
- Gao, Y.F.; Masuda, Y.; Seo, W.S.; Ohta, H.; Koumoto, K. TiO2 nanoparticles prepared using an aqueous peroxotitanate solutions. Ceram. Int. 2004, 30, 1365–1368. [Google Scholar] [CrossRef]
- Wang, Y.; Li, J.; Wang, L.N.; Xue, T.Y.; Qi, T. Preparation of Rutile Titanium Dioxide White Pigment via Doping and Calcination of Metatitanic Acid Obtained by the NaOH Molten Salt Method. Ind. Eng. Chem. Res. 2010, 49, 7693–7696. [Google Scholar] [CrossRef]
- Song, Y.W.; Wang, H.R.; Wang, R.; Zhou, J.C. Novel approach for high-efficiency recovery of titanium dioxide, hydrochloric acid, and organic solvents from titanium white waste acid. J. Clean. Prod. 2021, 315, 128105. [Google Scholar] [CrossRef]
- Zhang, W.; Ou, C.R.; Yuan, Z.G. Precipitation and growth behaviour of metatitanic acid particles from titanium sulfate solution. Powder Technol. 2017, 315, 31–36. [Google Scholar] [CrossRef]
- Lu, R.F.; Liu, C.; Wu, J.C.; Sun, W.; Sun, Q.; Dong, L.C. Process optimization of the extra-adding seeded hydrolysis of TiOSO4 to H2TiO3 by using the unenriched solution for the manufacture of TiO2 pigment. J. Cryst. Growth 2021, 572, 126268. [Google Scholar] [CrossRef]
- Chen, K.; Yan, X.H.; Wu, P.S.; Wang, Z.N.; Wu, B.; Lin, F.R. Effect of sulfate on crystal phase transition and crystal growth of titanium dioxide in metatitanic acid calcination. Phase Transit. 2021, 94, 353–365. [Google Scholar] [CrossRef]
- Tian, C.X. Orthogonal interactions and synergistic effects of salt treatment and calcination on rutile titanium dioxide pigment preparation. Mater. Chem. Phys. 2020, 249, 123125. [Google Scholar] [CrossRef]
- Wang, X.J.; Hu, Z.H.; Chen, Y.J.; Liu, Y.F.; Wen, Z.B.; Zhao, G.H. High Performance Supported Photocatalyst of Nano-TiO2/Activated Carbon from Metatitanic Acid. Acta Chim. Sin. 2008, 66, 2445–2450. [Google Scholar]
- Rui, B.; Liu, B.G.; Ting, Z.; Wu, B.J.; Dong, E.H.; Chao, Y.W. Rutile TiO2 Production: Optimization of Microwave Calcination of Metatitanic Acid Using Response Surface Methodology. Chem. Eng. Technol. 2022, 45, 1826–1834. [Google Scholar] [CrossRef]
- Wu, X.P.; Liu, Y. Phase change and crystal growth of TiO2 in metatitanic acid. Ceram. Int. 2023, 49, 4607–4613. [Google Scholar] [CrossRef]
- Lu, R.F.; Liu, C.; Wu, J.C.; Wu, Y.X.; Zhang, Q.; Sun, Q. Investigation on the structure evolution of rutile TiO2 during calcination of mixed-salt-treated metatitanic acid. J. Cryst. Growth 2023, 602, 126985. [Google Scholar] [CrossRef]
- Komarkova, B.; Motlochova, M.; Slovak, V.; Ecorchard, P.; Bavol, D.; Subrt, J.; Bezdicka, P. Effect of amines on (peroxo)titanates: Characterization and thermal decomposition. J. Therm. Anal. Calorim. 2022, 147, 5009–5022. [Google Scholar] [CrossRef]
- Huang, L.; Chen, Y.C.; Liu, G.; Li, S.N.; Liu, Y.; Gao, X. Non-isothermal pyrolysis characteristics of giant reed (Arundo donax L.) using thermogravimetric analysis. Energy 2015, 87, 31–40. [Google Scholar] [CrossRef]
- Li, P.; Yu, Q.B.; Qin, Q.; Lei, W. Kinetics of CO2/Coal Gasification in Molten Blast Furnace Slag. Ind. Eng. Chem. Res. 2012, 51, 15872–15883. [Google Scholar] [CrossRef]
- Li, P.; Yu, Q.B.; Xie, H.Q.; Qin, Q.; Wang, K. CO2 Gasification Rate Analysis of Datong Coal Using Slag Granules as Heat Carrier for Heat Recovery from Blast Furnace Slag by Using a Chemical Reaction. Energy Fuels 2013, 27, 4810–4817. [Google Scholar] [CrossRef]

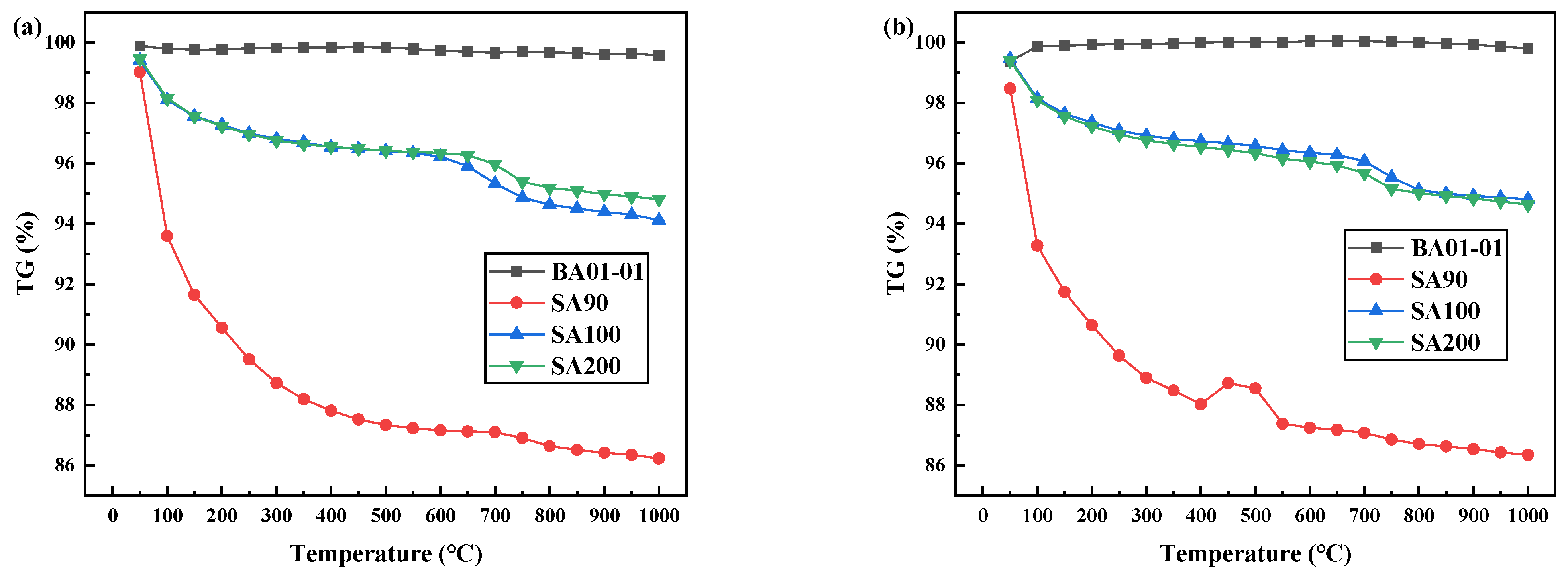


| TiO2 | WO3 | SiO2 | SO3 | |
|---|---|---|---|---|
| SA90 | 98.0 | – | – | 1.5~2.0 |
| SA100 | 95.0 | 3.5~4.0 | – | 1.0~1.5 |
| SA200 | 95.5 | – | 3.5~4.0 | 0.5~1.0 |
| α | Air Atmosphere | Nitrogen Atmosphere | ||||||
|---|---|---|---|---|---|---|---|---|
| 5 °C/min | 10 °C/min | 15 °C/min | 20 °C/min | 5 °C/min | 10 °C/min | 15 °C/min | 20 °C/min | |
| 0.05 | 66.02 | 72.88 | 73.95 | 78.48 | 66.68 | 73.67 | 74.38 | 73.42 |
| 0.10 | 116.32 | 123.51 | 124.44 | 128.92 | 117.48 | 124.27 | 124.95 | 123.91 |
| 0.20 | 166.75 | 174.20 | 175.08 | 179.52 | 168.05 | 174.85 | 175.46 | 174.50 |
| 0.25 | 217.17 | 224.65 | 225.46 | 229.89 | 218.45 | 255.26 | 225.91 | 224.90 |
| 0.30 | 267.43 | 274.91 | 275.64 | 280.05 | 268.67 | 275.46 | 276.06 | 275.14 |
| 0.35 | 317.51 | 325.00 | 325.67 | 330.17 | 318.73 | 325.51 | 326.08 | 325.20 |
| 0.40 | 367.47 | 374.86 | 375.69 | 380.11 | 368.68 | 375.45 | 376.09 | 375.27 |
| 0.45 | 417.34 | 424.76 | 425.53 | 429.95 | 418.53 | 425.32 | 425.95 | 425.17 |
| 0.50 | 467.07 | 474.61 | 475.36 | 479.78 | 468.33 | 475.17 | 475.76 | 475.01 |
| 0.55 | 516.87 | 524.46 | 525.19 | 529.62 | 518.11 | 525.02 | 525.57 | 524.86 |
| 0.60 | 566.69 | 574.34 | 575.01 | 579.54 | 567.86 | 574.86 | 575.49 | 574.71 |
| 0.65 | 616.47 | 624.20 | 624.85 | 629.38 | 617.63 | 624.72 | 625.32 | 624.55 |
| 0.70 | 666.17 | 674.09 | 674.80 | 679.22 | 667.39 | 674.58 | 675.14 | 674.41 |
| 0.75 | 715.98 | 723.97 | 724.65 | 729.08 | 717.19 | 724.43 | 725.09 | 724.28 |
| 0.75 | 765.81 | 773.74 | 774.53 | 778.93 | 766.97 | 774.29 | 774.92 | 774.08 |
| 0.80 | 815.65 | 823.62 | 824.40 | 828.83 | 816.76 | 824.08 | 824.71 | 823.81 |
| 0.85 | 865.38 | 873.37 | 874.11 | 878.56 | 866.53 | 873.73 | 874.38 | 873.50 |
| No. | Integral Form | Differential Form |
|---|---|---|
| 12 | ||
| 18 | ||
| 25 | α | 1 |
| No. | Heating Rate (°C/min) | Air Atmosphere | Nitrogen Atmosphere | ||||
|---|---|---|---|---|---|---|---|
| E/(kJ.mol−1) | lnA/s−1 | R2 | E/(kJ.mol−1) | lnA/s−1 | R2 | ||
| 12 | 5 | 14.16 | 25.51 | 0.9764 | 15.57 | 26.44 | 0.9826 |
| 10 | 15.12 | 26.41 | 0.9813 | 14.45 | 26.17 | 0.9365 | |
| 15 | 15.16 | 36.81 | 0.9804 | 13.91 | 26.66 | 0.9362 | |
| 20 | 15.17 | 27.06 | 0.9816 | 14.28 | 26.92 | 0.9441 | |
| 18 | 5 | 34.05 | 29.49 | 0.9122 | 32.59 | 28.01 | 0.9632 |
| 10 | 34.17 | 29.68 | 0.9269 | 31.47 | 28.88 | 0.9517 | |
| 15 | 34.36 | 30.15 | 0.9194 | 30.96 | 29.55 | 0.9508 | |
| 20 | 36.32 | 31.20 | 0.9327 | 31.29 | 29.84 | 0.9514 | |
| 25 | 5 | 20.03 | 26.19 | 0.9362 | 20.09 | 26.23 | 0.9745 |
| 10 | 20.55 | 26.89 | 0.9509 | 19.06 | 26.17 | 0.9532 | |
| 15 | 20.62 | 27.29 | 0.9454 | 18.45 | 26.41 | 0.9527 | |
| 20 | 21.21 | 27.81 | 0.9502 | 19.21 | 27.46 | 0.9787 | |
| Dehydration (wt. %) | Desulfurization (wt. %) | |
|---|---|---|
| SA90 | 12.32 | 1.63 |
| SA100 | 3.55 | 2.3 |
| SA200 | 3.56 | 1.74 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Qiu, G.; Lv, X.; Liu, Y. Kinetic Study on the Dehydration Behavior of Titanium Dioxide as a Denitration Catalyst Carrier. Metals 2023, 13, 1486. https://doi.org/10.3390/met13081486
Li H, Qiu G, Lv X, Liu Y. Kinetic Study on the Dehydration Behavior of Titanium Dioxide as a Denitration Catalyst Carrier. Metals. 2023; 13(8):1486. https://doi.org/10.3390/met13081486
Chicago/Turabian StyleLi, Huaquan, Guibao Qiu, Xuewei Lv, and Yongjie Liu. 2023. "Kinetic Study on the Dehydration Behavior of Titanium Dioxide as a Denitration Catalyst Carrier" Metals 13, no. 8: 1486. https://doi.org/10.3390/met13081486
APA StyleLi, H., Qiu, G., Lv, X., & Liu, Y. (2023). Kinetic Study on the Dehydration Behavior of Titanium Dioxide as a Denitration Catalyst Carrier. Metals, 13(8), 1486. https://doi.org/10.3390/met13081486





