Dynamic of Mining Systems: Impact of Cl− Ion Concentration on Heap Copper Leaching Process at Industrial Scale
Abstract
1. Introduction
2. Materials and Methods
2.1. Heap Leaching
2.2. Modeling of Heap Leaching Process
- Process controlled by diffusion through the product layer: formation of a product layer around the material that resists diffusion of the oxidant to the surface of the material and slows down leaching.
- Chemically controlled process: the product layer is absent or its presence does not affect the free movement of the oxidant to the surface and the reaction between the surface and the reagent is much slower than the diffusion of the oxidant.
- Film Diffusion Process—Bulk leach solution resists movement of oxidant to the surface and this can slow leach kinetics.
3. Results and Discussions
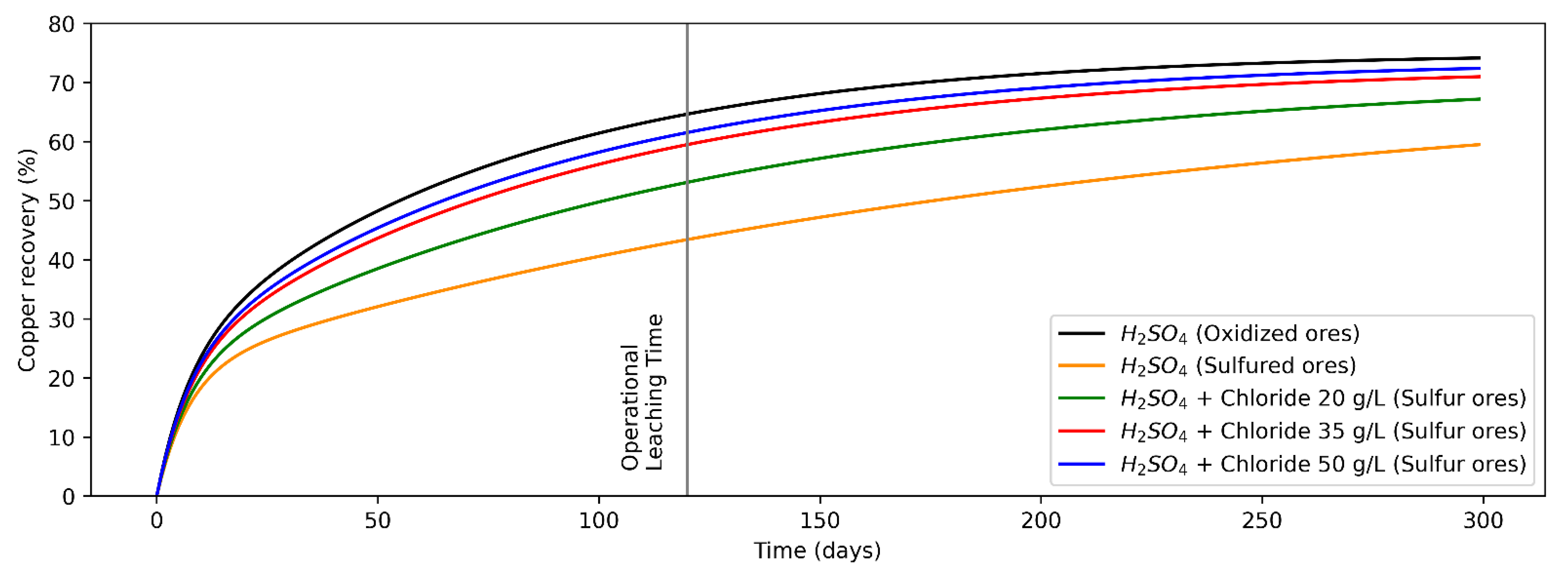
3.1. Adjustment of Analytical Models
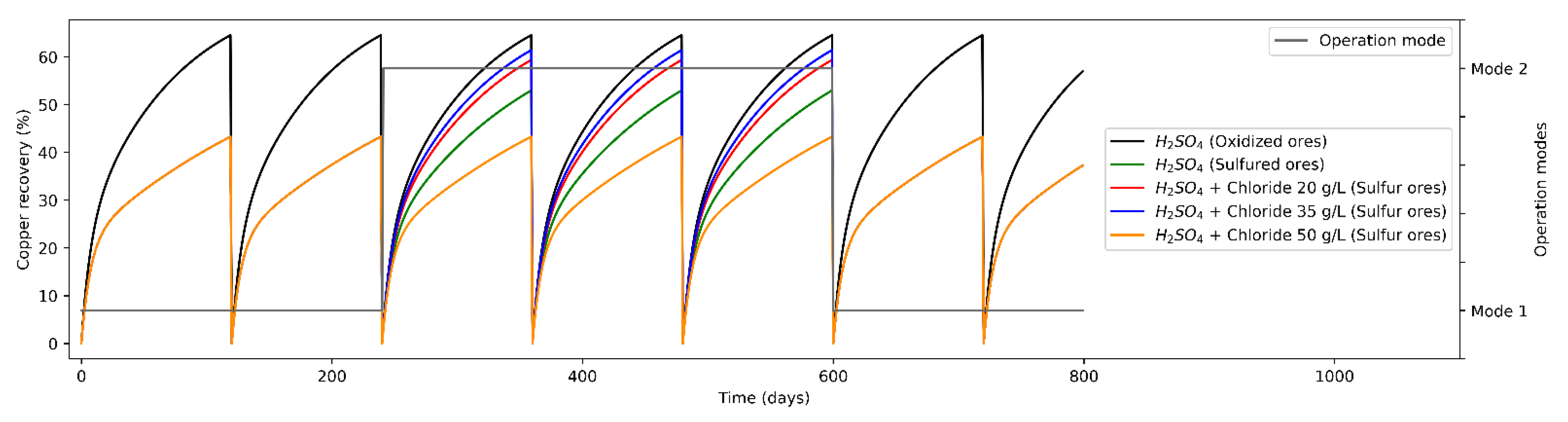
3.2. Scenarios Modeling
- Mode 1: Leaching of oxidized and secondary sulfides minerals only with H2SO4 as a leaching agent. Leaching of oxides and secondary sulfides with sulfuric acid (leaching of secondary sulfides with H2SO4 slows down mineral extraction from the rock, increasing the time required until one is marginal, or a smaller proportion of the valuable mineral is recovered, if considering the constant leaching time (how it usually works under operational conditions in the mining industry) [17]. Then, Mode 1 consists of two operation strategies:
- ◦
- Strategy 1: leaching of oxidized copper mineral using only H2SO4.
- ◦
- Strategy 2: leaching of sulfide copper mineral using only H2SO4.
- Mode 2: Leaching of oxidized minerals by H2SO4 and sulfide minerals at different levels of Cl− ions concentration (20, 35 and 50 g/L). Leaching of secondary sulfides with Cl− ions accelerates copper recovery from sulfide mineral [52,53,54,55]. Different chloride addition configurations are considered in order to determine the levels that improve mineral extraction.
- ◦
- Strategy 3: leaching of oxidized minerals with H2SO4 and sulfide minerals by adding chloride at a concentration of 20 g/L.
- ◦
- Strategy 4: leaching of oxidized minerals with H2SO4 and sulfide minerals by adding chloride at a concentration of 35 g/L.
- ◦
- Strategy 5: leaching of oxidized minerals with H2SO4 and sulfide minerals by adding chloride at a concentration of 50 g/L.
3.3. Uncertainty Analysis
3.3.1. Descriptive Statistics Base Case
3.3.2. Descriptive Statistics Proposed Case
3.3.3. Scenarios Comparison
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Flanagan, D.M. Copper. In Mineral Commodity Summaries 2022; U.S.G.S: Reston, VA, USA, 2022; pp. 54–55. ISBN 9780333227794. [Google Scholar]
- ICSG. The World Copper Factbook 2022; ICSG: Lisbon, Portugal, 2022.
- Research & Markets. Global Copper Market: Analysis by Mined Copper Production, by Refined Copper Production, by Consumption, by First-Use, by End-Use, by Region Size and Trends with Impact of COVID-19 and Forecast up to 2027; Daedal-Research: Delhi, India, 2022. [Google Scholar]
- Consejo Minero. Cifras Actualizadas de La Minería; Consejo Minero: Santiago, Chile, 2019. [Google Scholar]
- Comisión Chilena del Cobre. Sulfuros Primarios: Desafíos y Oportunidades; Comisión Chilena del Cobre: Santiago, Chile, 2017. [Google Scholar]
- Nuorivaara, T.; Björkqvist, A.; Bacher, J.; Serna-Guerrero, R. Environmental Remediation of Sulfidic Tailings with Froth Flotation: Reducing the Consumption of Additional Resources by Optimization of Conditioning Parameters and Water Recycling. J. Environ. Manag. 2019, 236, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Postila, H.; Heiderscheidt, E.; Leiviskä, T. Removal of Metals from Mine Drainage Waters by in Situ Mineral Sorbent-Based Pilot Filter Systems. J. Environ. Manag. 2019, 236, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Altinkaya, P.; Mäkinen, J.; Kinnunen, P.; Kolehmainen, E.; Haapalainen, M.; Lundström, M. Effect of Biological Pretreatment on Metal Extraction from Flotation Tailings for Chloride Leaching. Min. Eng. 2018, 129, 47–53. [Google Scholar] [CrossRef]
- Hernández-Espriú, A.; Wolaver, B.; Arciniega-Esparza, S.; Scanlon, B.R.; Young, M.H.; Nicot, J.P.; Macías-Medrano, S.; Breña-Naranjo, J.A. A Screening Approach to Improve Water Management Practices in Undeveloped Shale Plays, with Application to the Transboundary Eagle Ford Formation in Northeast Mexico. J. Environ. Manag. 2019, 236, 146–162. [Google Scholar] [CrossRef] [PubMed]
- Toro, N.; Pérez, K.; Saldaña, M.; Jeldres, R.I.; Jeldres, M.; Cánovas, M. Dissolution of Pure Chalcopyrite with Manganese Nodules and Waste Water. J. Mater. Res. Technol. 2020, 9, 798–805. [Google Scholar] [CrossRef]
- Sosa, B.S.; Banda-Noriega, R.B.; Guerrero, E.M. Industrias de Fundición: Aspectos Ambientales e Indicadores de Condición Ambiental. Rev. Metal. 2013, 49, 5–19. [Google Scholar] [CrossRef]
- Cumpston, J.; Herding, R.; Lechtenberg, F.; Offermanns, C.; Thebelt, A.; Roh, K. Design of 24/7 Continuous Hydrogen Production System Employing the Solar-Powered Thermochemical S–I Cycle. Int. J. Hydrogen Energy 2020, 45, 24383–24396. [Google Scholar] [CrossRef]
- Gorensek, M.B.; Staser, J.A.; Stanford, T.G.; Weidner, J.W. A Thermodynamic Analysis of the SO2/H2SO4 System in SO2-Depolarized Electrolysis. Int. J. Hydrogen Energy 2009, 34, 6089–6095. [Google Scholar] [CrossRef]
- Saldaña, M.; Toro, N.; Castillo, J.; Hernández, P.; Navarra, A. Optimization of the Heap Leaching Process through Changes in Modes of Operation and Discrete Event Simulation. Minerals 2019, 9, 421. [Google Scholar] [CrossRef]
- Saldaña, M.; Gálvez, E.; Robles, P.; Castillo, J.; Toro, N. Copper Mineral Leaching Mathematical Models—A Review. Materials 2022, 15, 1757. [Google Scholar] [CrossRef]
- Saldaña, M.; Neira, P.; Gallegos, S.; Salinas-Rodríguez, E.; Pérez-Rey, I.; Toro, N. Mineral Leaching Modeling Through Machine Learning Algorithms—A Review. Front. Earth Sci. 2022, 10, 560. [Google Scholar] [CrossRef]
- Yévenes, L.V.; Miki, H.; Nicol, M. The Dissolution of Chalcopyrite in Chloride Solutions: Part 2: Effect of Various Parameters on the Rate. Hydrometallurgy 2010, 103, 80–85. [Google Scholar] [CrossRef]
- Velásquez-Yévenes, L.; Nicol, M.; Miki, H. The Dissolution of Chalcopyrite in Chloride Solutions Part 1. The Effect of Solution Potential. Hydrometallurgy 2010, 103, 108–113. [Google Scholar] [CrossRef]
- Nicol, M.; Miki, H.; Velásquez-Yévenes, L. The Dissolution of Chalcopyrite in Chloride Solutions Part 3. Mechanisms. Hydrometallurgy 2010, 103, 86–95. [Google Scholar] [CrossRef]
- Galleguillos, F.; Cáceres, L.; Maxwell, L.; Soliz, Á. Electrochemical Ion Pumping Device for Blue Energy Recovery: Mixing Entropy Battery. Appl. Sci. 2020, 10, 5537. [Google Scholar] [CrossRef]
- Hashemzadeh, M.; Dixon, D.G.; Liu, W. Modelling the Kinetics of Chalcocite Leaching in Acidified Cupric Chloride Media under Fully Controlled PH and Potential. Hydrometallurgy 2019, 189, 105114. [Google Scholar] [CrossRef]
- Hashemzadeh, M.; Dixon, D.G.; Liu, W. Modelling the Kinetics of Chalcocite Leaching in Acidified Ferric Chloride Media under Fully Controlled PH and Potential. Hydrometallurgy 2019, 186, 275–283. [Google Scholar] [CrossRef]
- Hashemzadeh, M.; Liu, W. Analysis of Iron and Copper Speciation and Activities in Chloride Leaching Solutions of High Ionic Strength. Hydrometallurgy 2020, 192, 105262. [Google Scholar] [CrossRef]
- Horányi, G. Investigation of the Specific Adsorption of HSO4−(SO42−) and Cl− Ions on Co and Fe by Radiotracer Technique in the Course of Corrosion of the Metals in Perchlorate Media. Corros. Sci 2004, 46, 1741–1749. [Google Scholar] [CrossRef]
- Torres, C.M.; Ghorbani, Y.; Hernández, P.C.; Justel, F.J.; Aravena, M.I.; Herreros, O.O. Cupric and Chloride Ions: Leaching of Chalcopyrite Concentrate with Low Chloride Concentration Media. Minerals 2019, 9, 639. [Google Scholar] [CrossRef]
- Choi, B.K.; Kang, D. Modeling and Simulation of Discrete-Event Systems; John Wiley & Sons: Hoboken, NJ, USA, 2013; ISBN 9781118386996. [Google Scholar]
- Mery, N.; Emery, X.; Cáceres, A.; Ribeiro, D.; Cunha, E. Geostatistical Modeling of the Geological Uncertainty in an Iron Ore Deposit. Ore Geol. Rev. 2017, 88, 336–351. [Google Scholar] [CrossRef]
- Toro, N.; Moraga, C.; Torres, D.; Saldaña, M.; Pérez, K.; Gálvez, E. Leaching Chalcocite in Chloride Media—A Review. Minerals 2021, 11, 1197. [Google Scholar] [CrossRef]
- Zhao, S.; Peng, Y. The Oxidation of Copper Sulfide Minerals during Grinding and Their Interactions with Clay Particles. Powder Technol. 2012, 230, 112–117. [Google Scholar] [CrossRef]
- Pérez, K.; Toro, N.; Saldaña, M.; Salinas-Rodríguez, E.; Robles, P.; Torres, D.; Jeldres, R.I. Statistical Study for Leaching of Covellite in a Chloride Media. Metals 2020, 10, 477. [Google Scholar] [CrossRef]
- Schlesinger, M.; King, M.; Sole, K.; Davenport, W. Extractive Metallurgy of Copper, 5th ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2011; ISBN 9780080967899. [Google Scholar]
- Miki, H.; Nicol, M.; Velásquez-Yévenes, L. The Kinetics of Dissolution of Synthetic Covellite, Chalcocite and Digenite in Dilute Chloride Solutions at Ambient Temperatures. Hydrometallurgy 2011, 105, 321–327. [Google Scholar] [CrossRef]
- Mellado, M.E.; Cisternas, L.A.; Gálvez, E.D. An Analytical Model Approach to Heap Leaching. Hydrometallurgy 2009, 95, 33–38. [Google Scholar] [CrossRef]
- Mellado, M.E.; Gálvez, E.D.; Cisternas, L.A. Stochastic Analysis of Heap Leaching Process via Analytical Models. Min. Eng. 2012, 33, 93–98. [Google Scholar] [CrossRef]
- Yaghobi Moghaddam, M.; Shafaei Tonkaboni, S.Z.; Noaparast, M.; Doulati Ardejani, F. A Mathematical Model to Simulate Heap (Bio)-Leaching Process: An Exact Conceptual Model, Homotopy Theory and Comparative Insights with Conventional Methods. Int. J. Model. Simul. Sci. Comput. 2017, 8, 1750018. [Google Scholar] [CrossRef]
- Saldaña, M.; González, J.; Jeldres, R.; Villegas, Á.; Castillo, J.; Quezada, G.; Toro, N. A Stochastic Model Approach for Copper Heap Leaching through Bayesian Networks. Metals 2019, 9, 1198. [Google Scholar] [CrossRef]
- Dixon, D.G.; Hendrix, J.L. A Mathematical Model for Heap Leaching of One or More Solid Reactants from Porous Ore Pellets. Metall. Trans. B 1993, 24, 1087–1102. [Google Scholar] [CrossRef]
- Dixon, D.G.; Hendrix, J.L. A General Model for Leaching of One or More Solid Reactants from Porous Ore Particles. Metall. Trans. B 1993, 24, 157–169. [Google Scholar] [CrossRef]
- Remy, N.; Boucher, A.; Wu, J. Applied Geostatistics with SGeMS: A User’s Guide; Cambridge University Press: Cambridge, UK, 2009; Volume 9780521514, ISBN 9781139150019. [Google Scholar]
- Upadhyay, S.P.; Askari-Nasab, H. Simulation and Optimization Approach for Uncertainty-Based Short-Term Planning in Open Pit Mines. Int. J. Min. Sci. Technol. 2018, 28, 153–166. [Google Scholar] [CrossRef]
- Deck, C.; Guzmán, J.I.; Hinrichsen, C.; Lichtin, C.; Rademacher, M.; Minera, E.; Cancino, C.R.; Henríquez, J.; Jara, E.; Morales, G.; et al. Flexible Stochastic Planning: The Ultimate Frontier. In Proceedings of the GeoMin Mine Planning 2013, Santiago, Chile, 24–26 July 2013. [Google Scholar]
- Dimitrakopoulos, R.G.; Sabour, S.A.A. Evaluating Mine Plans under Uncertainty: Can the Real Options Make a Difference? Resour. Policy 2007, 32, 116–125. [Google Scholar] [CrossRef]
- Navarra, A.; Grammatikopoulos, T.; Waters, K. Incorporation of Geometallurgical Modelling into Long-Term Production Planning. Min. Eng. 2018, 120, 118–126. [Google Scholar] [CrossRef]
- Levenspiel, O. Chemical Reaction Engineering, 3rd ed.; Anderson, W., Ed.; John Wiley & Sons, Inc.: New York, NY, USA, 1999; ISBN 047125424X. [Google Scholar]
- Madsen, B.W.; Wadsworth, M.E.; Groves, R.D. Application of a mixed kinetics model to the leaching of low grade copper sulfide ores. Trans. Soc. Min. Eng. AIME 1975, 258, 69–74. [Google Scholar]
- Braun, R.L.; Lewis, A.E.; Wadsworth, M.E. In-Place Leaching Of Primary Sulfide Ores: Laboratory Leaching Data and Kinetics Model. Met. Trans. 1974, 5, 1717–1726. [Google Scholar] [CrossRef]
- Dixon, D.G.; Hendrix, J.L. Theoretical Basis for Variable Order Assumption in the Kinetics of Leaching of Discrete Grains. AIChE J. 1993, 39, 904–907. [Google Scholar] [CrossRef]
- Botz, M.; Marsden, J. Heap Leach Production Modeling: A Spreadsheet-Based Technique. Min. Met. Explor. 2019, 36, 1041–1052. [Google Scholar] [CrossRef]
- McBride, D.; Gebhardt, J.; Croft, N.; Cross, M. Heap Leaching: Modelling and Forecasting Using CFD Technology. Minerals 2018, 8, 9. [Google Scholar] [CrossRef]
- Mellado, M.; Cisternas, L.; Lucay, F.; Gálvez, E.; Sepúlveda, F. A Posteriori Analysis of Analytical Models for Heap Leaching Using Uncertainty and Global Sensitivity Analyses. Minerals 2018, 8, 44. [Google Scholar] [CrossRef]
- Toro, N.; Briceño, W.; Pérez, K.; Cánovas, M.; Trigueros, E.; Sepúlveda, R.; Hernández, P. Leaching of Pure Chalcocite in a Chloride Media Using Sea Water and Waste Water. Metals 2019, 9, 780. [Google Scholar] [CrossRef]
- Helle, S.; Jerez, O.; Kelm, U.; Pincheira, M.; Varela, B. The Influence of Rock Characteristics on Acid Leach Extraction and Re-Extraction of Cu-Oxide and Sulfide Minerals. Min. Eng. 2010, 23, 45–50. [Google Scholar] [CrossRef]
- Jones, D.A.; Paul, A.J.P. Acid Leaching Behavior of Sulfide and Oxide Minerals Determined by Electrochemical Polarization Measurements. Min. Eng. 1995, 8, 511–521. [Google Scholar] [CrossRef]
- Cheng, C.Y.; Lawson, F. The Kinetics of Leaching Chalcocite in Acidic Oxygenated Sulphate-Chloride Solutions. Hydrometallurgy 1991, 27, 249–268. [Google Scholar] [CrossRef]
- Ruiz, M.C.; Honores, S.; Padilla, R. Leaching Kinetics of Digenite Concentrate in Oxygenated Chloride Media at Ambient Pressure. Metall. Mater. Trans. B Process Metall. Mater. Process. Sci. 1998, 29, 961–969. [Google Scholar] [CrossRef]
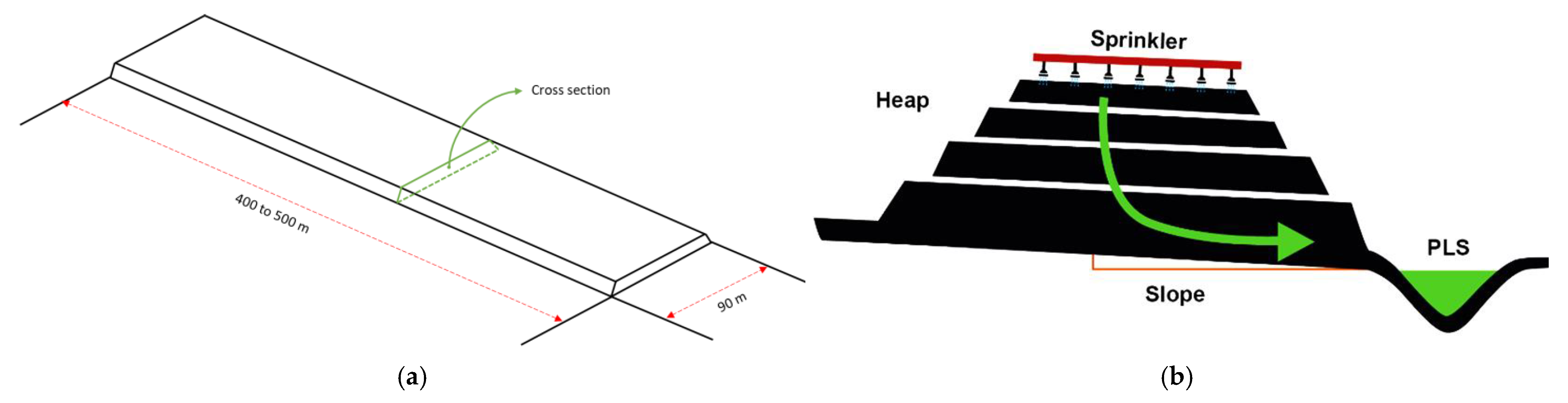

| Variable/Level | Low | Medium | High |
|---|---|---|---|
| (cm) | 600 | 800 | 900 |
| () | 9 | 28 | 57 |
| (mm) | 1.0 | 2.5 | 3.5 |
| (days) | 90 | 105 | 120 |
| Equation | MAD | MSE | p Value (Residuals Normality) |
|---|---|---|---|
| (2) | 0.136464313 | 0.041459923 | <0.01 |
| (3) | 0.041547655 | 0.003484736 | <0.01 |
| (4) | 0.028614690 | 0.002489411 | <0.01 |
| (5) | 0.136871445 | 0.042610117 | <0.01 |
| (6) | 0.120720494 | 0.031967063 | <0.01 |
| Ores/Stats | Mean | Standard Deviation | Normality (p Valor) |
|---|---|---|---|
| Oxides | 64.655 | 1.367 | <0.005 |
| Sulfide | 43.416 | 1.784 | 0.149 |
| Cl Conc./Stats | Mean | Standard Deviation | Normality (p Value) |
|---|---|---|---|
| Cl 20 g/L | 53.107 | 1.693 | 0.125 |
| Cl 35 g/L | 59.489 | 1.704 | 0.058 |
| Cl 50 g/L | 60.935 | 1.601 | 0.010 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saldaña, M.; Gálvez, E.; Gallegos, S.; Madrid, F.M.G.; Robles, P.; Toro, N. Dynamic of Mining Systems: Impact of Cl− Ion Concentration on Heap Copper Leaching Process at Industrial Scale. Metals 2023, 13, 465. https://doi.org/10.3390/met13030465
Saldaña M, Gálvez E, Gallegos S, Madrid FMG, Robles P, Toro N. Dynamic of Mining Systems: Impact of Cl− Ion Concentration on Heap Copper Leaching Process at Industrial Scale. Metals. 2023; 13(3):465. https://doi.org/10.3390/met13030465
Chicago/Turabian StyleSaldaña, Manuel, Edelmira Gálvez, Sandra Gallegos, Felipe M. Galleguillos Madrid, Pedro Robles, and Norman Toro. 2023. "Dynamic of Mining Systems: Impact of Cl− Ion Concentration on Heap Copper Leaching Process at Industrial Scale" Metals 13, no. 3: 465. https://doi.org/10.3390/met13030465
APA StyleSaldaña, M., Gálvez, E., Gallegos, S., Madrid, F. M. G., Robles, P., & Toro, N. (2023). Dynamic of Mining Systems: Impact of Cl− Ion Concentration on Heap Copper Leaching Process at Industrial Scale. Metals, 13(3), 465. https://doi.org/10.3390/met13030465








