Corrosion Behavior of the Cu24Zn5Al Alloy in Sodium Sulfate Solution in the Presence of 1-Phenyl-5-mercaptotetrazole
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
- (A)
- Potentiodynamic polarization measurements (linear and cyclic voltammetry) were conducted in a 0.1 mol/dm3 sodium sulfate solution without and with the addition of various concentrations of the inhibitor (2.5 × 10−3 mol/dm3, 1 × 10−3 mol/dm3, 5 × 10−4 mol/dm3, 1 × 10−4 mol/dm3, 1 × 10−5 mol/dm3), as well as in 0.1 mol/dm3 sodium sulfate solutions containing various concentrations of chloride ions (5 × 10−2 mol/dm3, 1 × 10−2 mol/dm3, 1 × 10−3 mol/dm3, 1 × 10−4 mol/dm3).
- (B)
- The testing of the effect of the immersion time of the Cu24Zn5Al alloy in the 0.017 mol/dm3 PMT solution was conducted by allowing the Cu24Zn5Al electrode to stand in the solution for 15 min, 60 min, and 240 min, then rinsing with distilled water and immediately subjecting it to potentiodynamic polarization measurements (linear and cyclic voltammetry) in a 0.1 mol/dm3 sodium sulfate solution. After obtaining the optimal exposure time, the effect of chloride ions was examined by rinsing the Cu24Zn5Al electrode that was immersed for 60 min (optimal time) in the 0.017 mol/dm3 PMT solution with distilled water and immediately subjecting it to potentiodynamic polarization measurements (linear and cyclic voltammetry) in a 0.1 mol/dm3 sodium sulfate solution containing different concentrations of chloride ions (5 × 10−2 mol/dm3, 1 × 10−2 mol/dm3, 1 × 10−3 mol/dm3, 1 × 10−4 mol/dm3).
3. Results and Discussion
3.1. Electrochemical Measurements
- jcorr—corrosion current density in the sodium sulfate solution without inhibitor (μA/cm2);
- jcorr(inh)—corrosion current density in the sodium sulfate solution with PMT (μA/cm2).
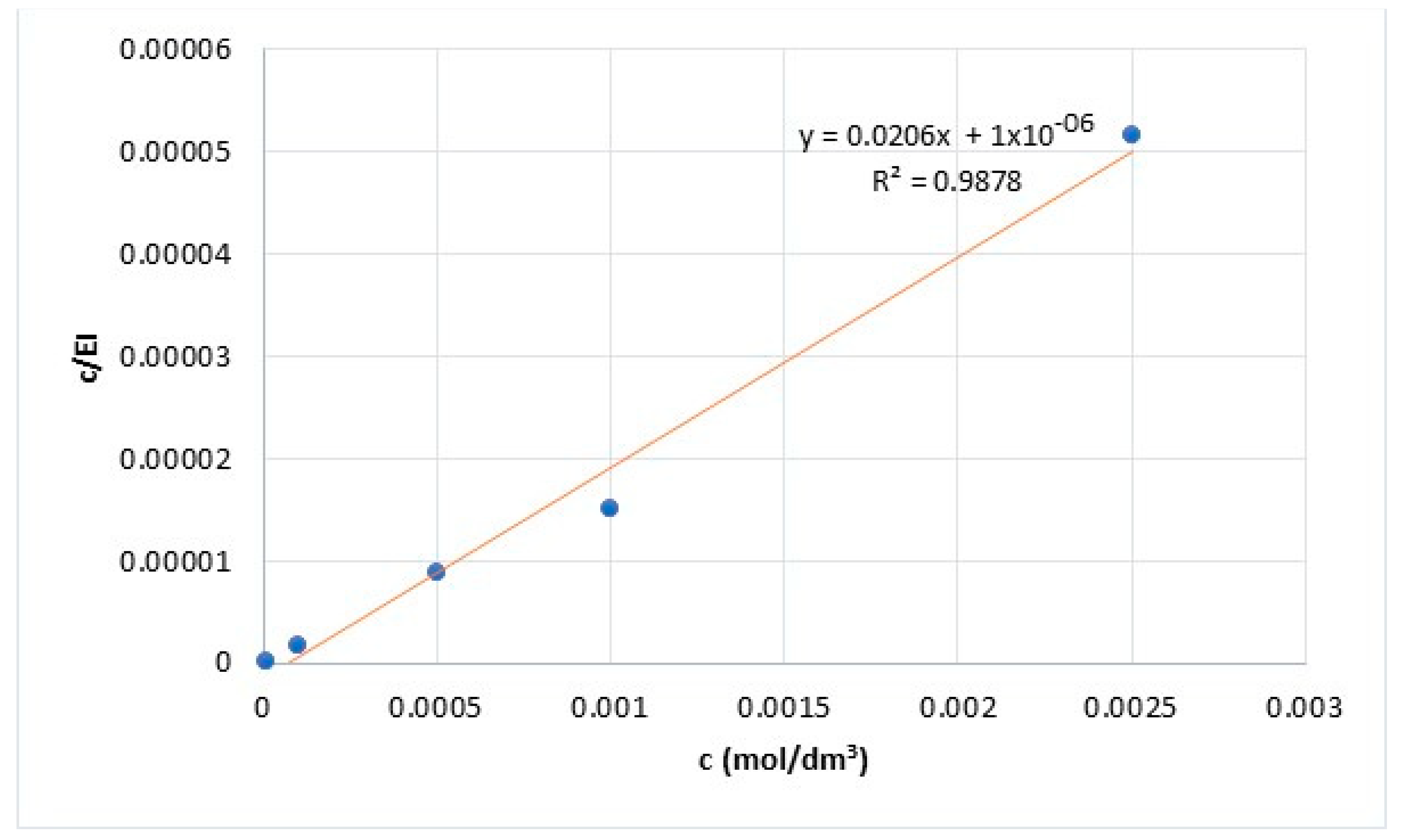
3.2. Adsorption Isotherm
- K—adsorptive equilibrium constant;
- ΔG—the Gibbs free energy of adsorption (J/mol);
- c—the concentration of PMT (mol/dm3);
- IE—the inhibition efficiency (%).
- R—the universal gas constant;
- T—the thermodynamic temperature (293 K).
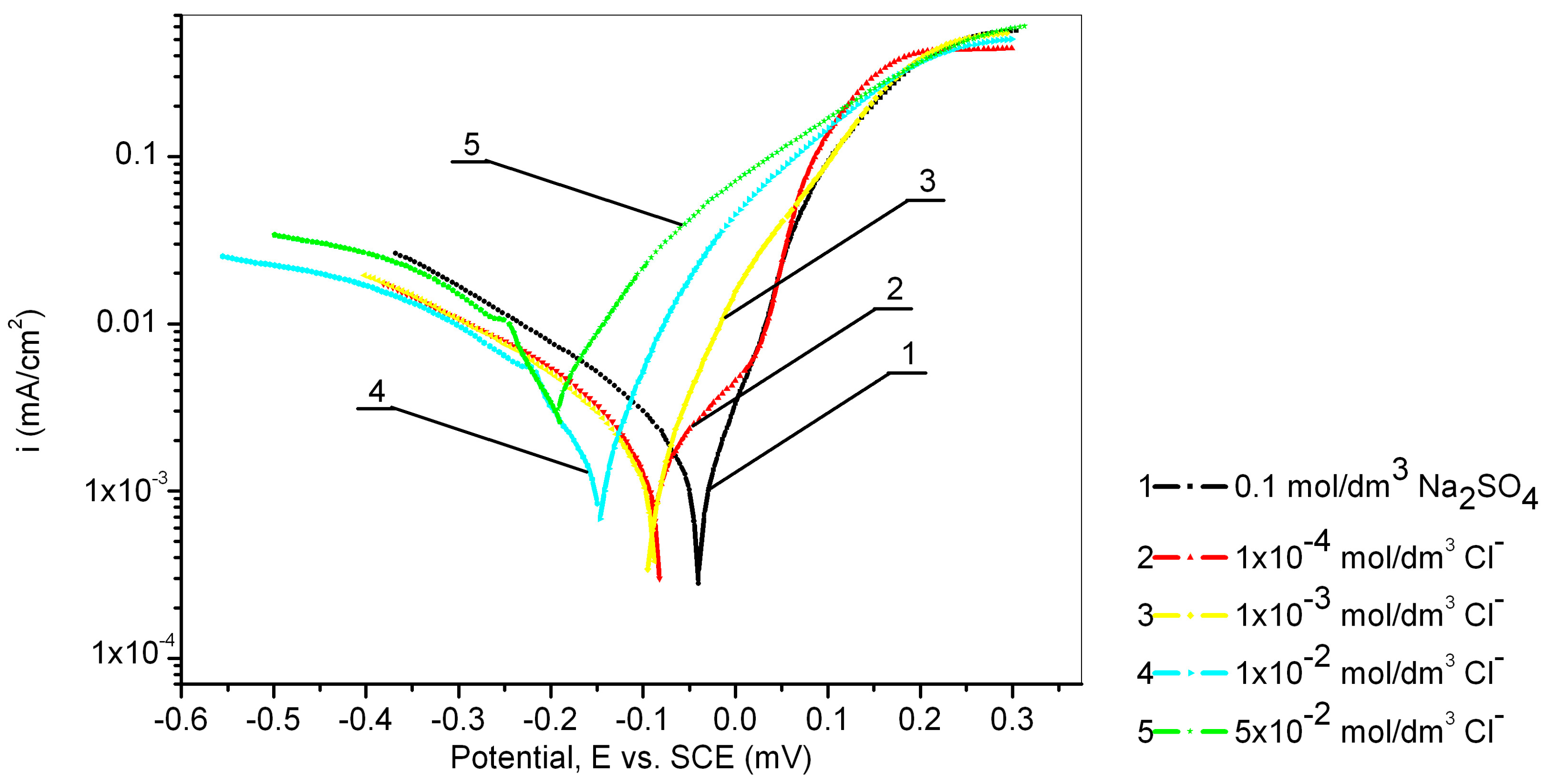
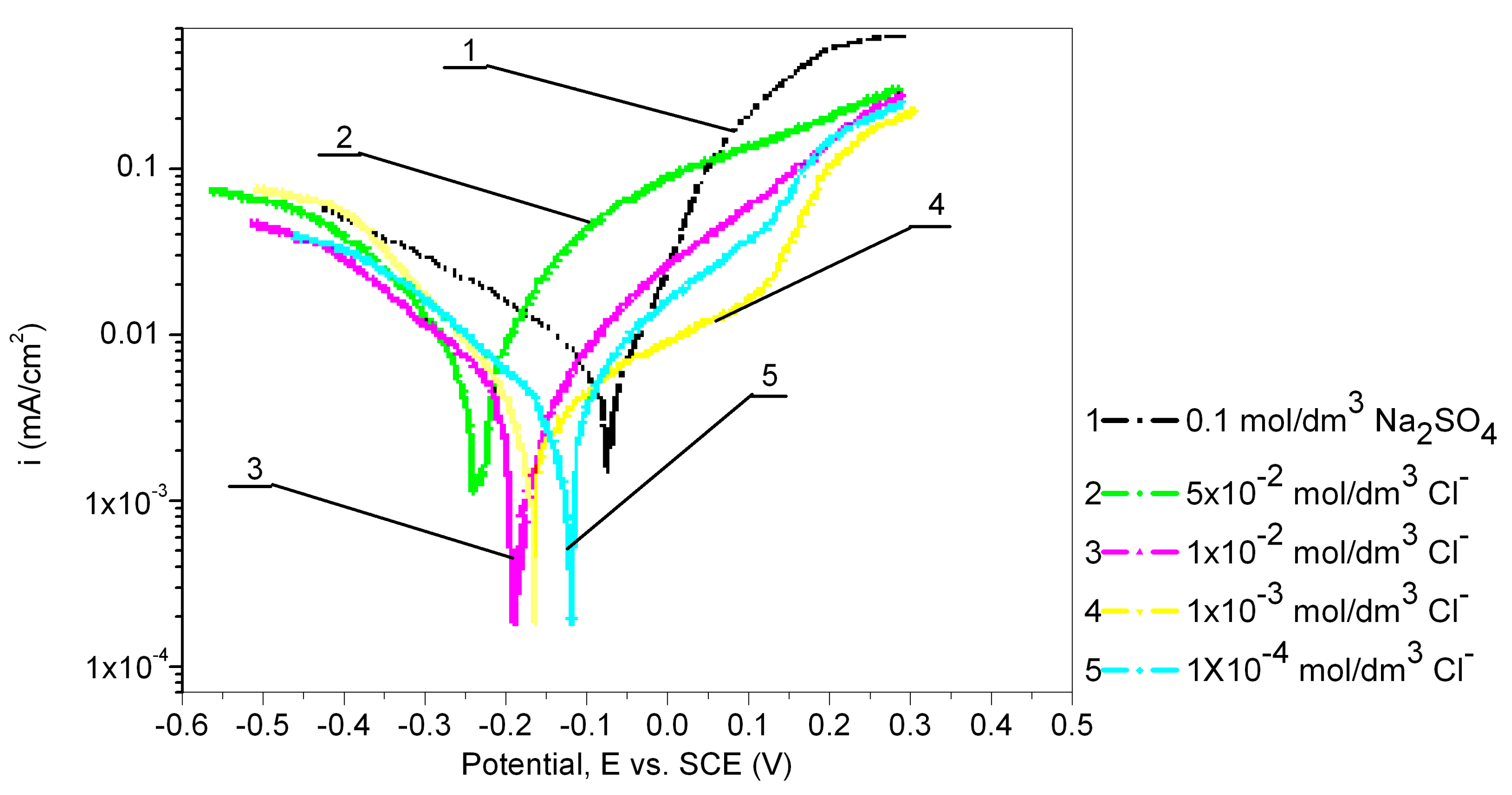
3.3. The Effect of Chloride Ion Concentration
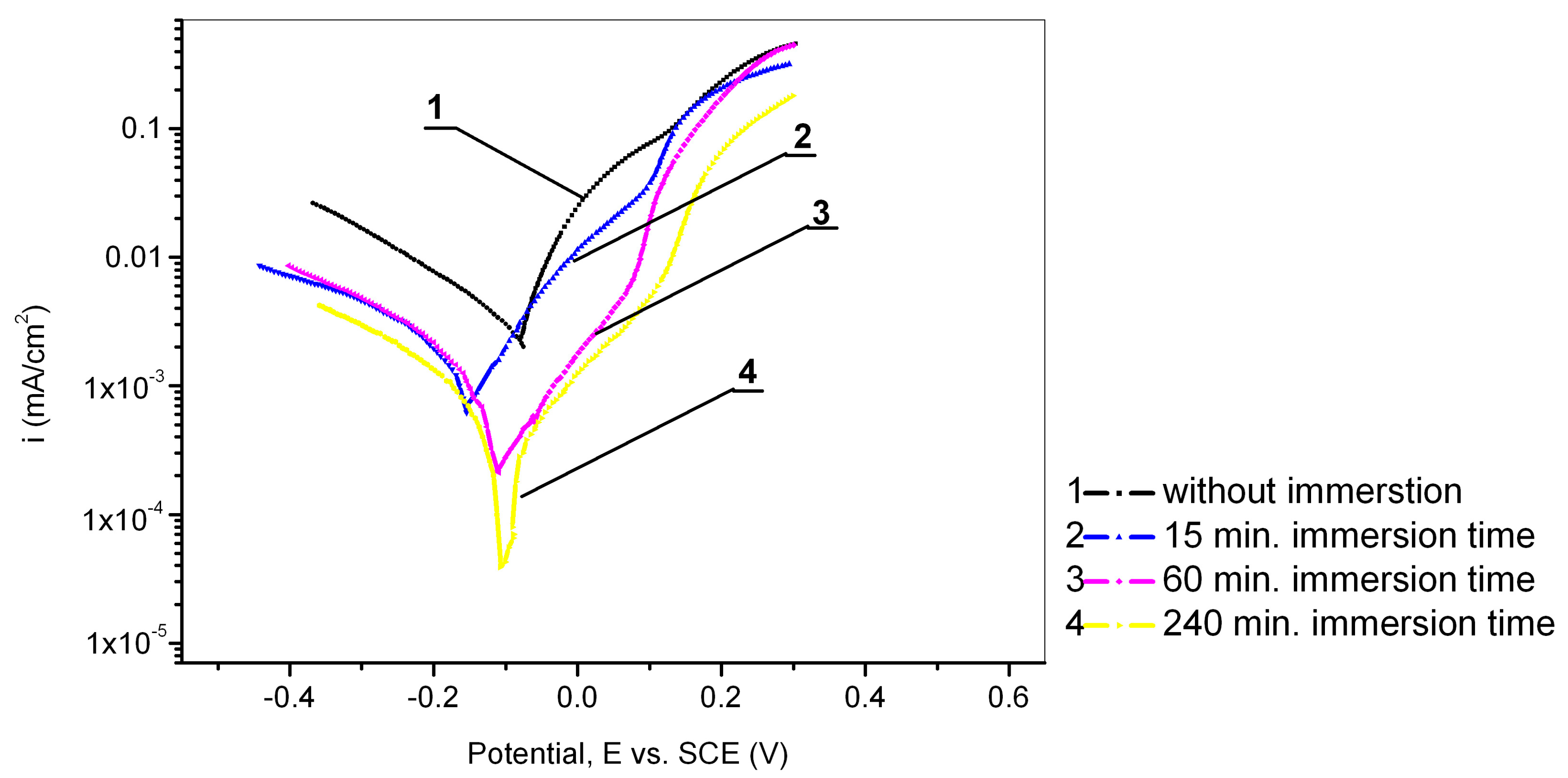
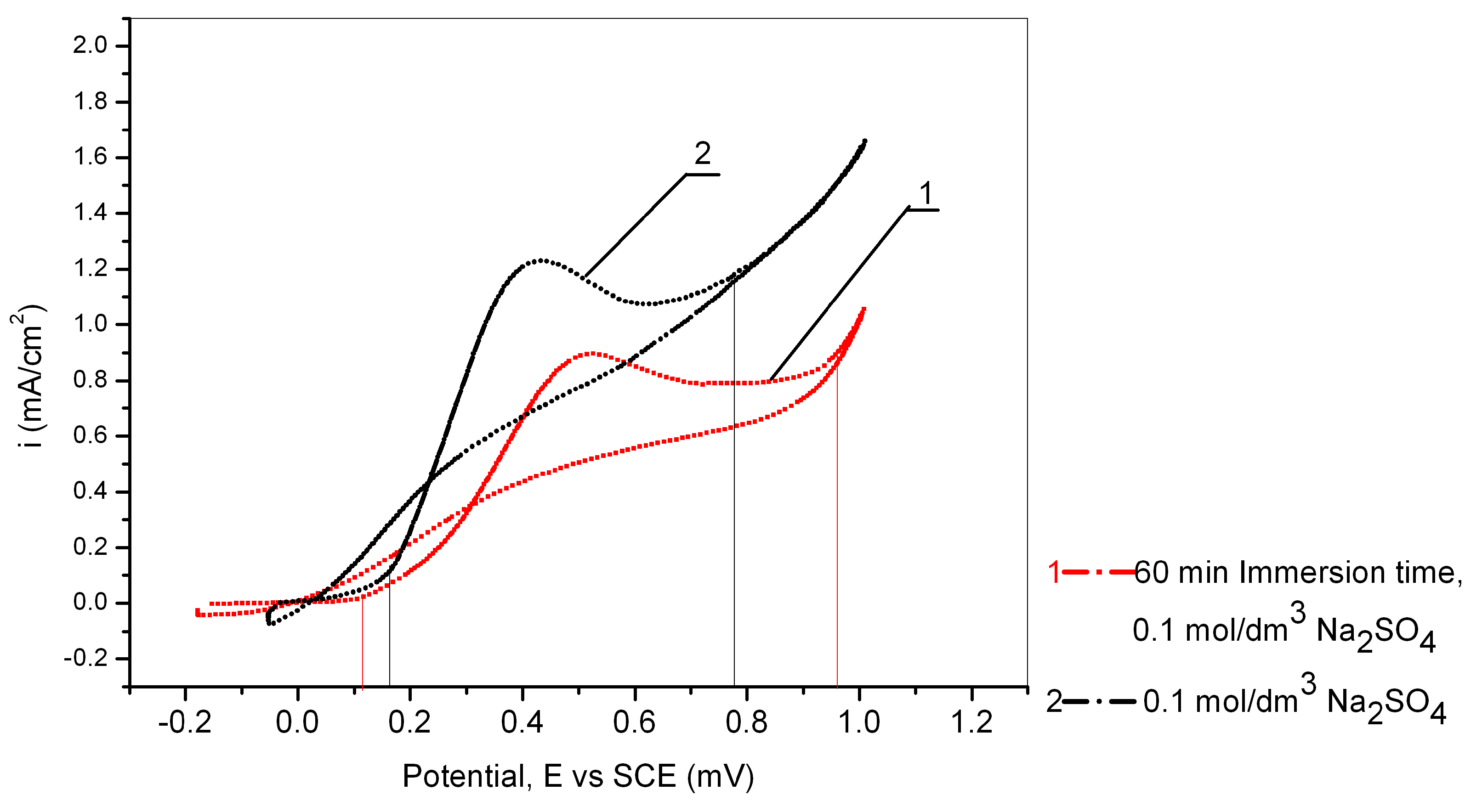
3.4. The Influence of the Immersion Time of Cu24Zn5Al Alloy in a Solution of 0.017 mol/dm3 PMT
- jcorr—corrosion current density obtained in sodium sulfate solution without pretreatment (μA/cm2);
- jcorr(immersion)—current density obtained in the sodium sulfate solution after pretreatment in the 0.017 mol/dm3 PMT solution (μA/cm2).
- —the area provided by integrated curve obtained in the sodium sulfate solution without pretreatment (mV mA/cm2);
- —the area provided by integrated curve obtained in the sodium sulfate solution after pretreatment in the 0.017 mol/dm3 PMT solution (μA/cm2).
- Sjcorr—the degree of corrosion current reduction (%);
- —corrosion current density obtained in sodium sulfate solution (μA/cm2);
- jcorr(immersion)—current density obtained in the sodium sulfate solution without and with the presence of different concentrations of chloride ions, after the immersion time of 60 min in a solution of 0.017 mol/dm3 PMT (μA/cm2).
- —the degree of corrosion current reduction (%);
- —corrosion current density obtained in sodium sulfatase solution with different concentrations of chloride ions (μA/cm2);
- —current density obtained in sodium sulfate solution with different concentrations of chloride ions, and after the immersion time of 60 min in 0.017 mol/dm3 PMT solution (μA/cm2).
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Fateh, A.; Aliofkhazraei, M.; Rezvanian, A.R. Review of corrosive environments for copper and its corrosion inhibitors. Arab. J. Chem. 2020, 13, 481–544. [Google Scholar] [CrossRef]
- Liu, H.X.; Si, N.C.; Xu, G.F. Influence of process factors on shape memory effect of CuZnAl alloys. Trans. Nonferrous Met. Soc. China 2006, 16, 1402–1409. [Google Scholar] [CrossRef]
- Guerioune, M.; Amiour, Y.; Bounour, W.; Guellati, O.; Benaldjia, A.; Amara, A.; Chakri, N.E.; Ali-Rachedi, M.; Vrel, D. SHS of Shape Memory CuZnAl Alloys. Int. J. Self-Propagating High-Temp. Synth. 2008, 17, 41–48. [Google Scholar] [CrossRef]
- Jani, J.M.; Leary, M.; Subic, A.; Gibson, M.A. A review of shape memory alloy research, applications and opportunities. Mater. Des. 2014, 56, 1078–1113. [Google Scholar] [CrossRef]
- De Filippo, B.; Brotzu, A.; Natali, S. Corrosion Behavior of Cu-Zn-Al Shape Memory Alloy in Controlled Environments. AIP Conf. Proc. 2020, 2257, 020013. [Google Scholar] [CrossRef]
- Cubela, D. Shape Memory Alloys. Masinstvo 2002, 2, 83–92. [Google Scholar]
- Huang, W. Shape Memory Alloys and their Application to Actuators for Deployable Structures. Ph.D. Dissertation, Department of Engineering University of Cambridge, Cambridge, MA, USA, 1998. [Google Scholar]
- Subramanian, A. Fatigue Behavior of Copper Zinc Aluminum Shape Memory Alloys. Master’s Thesis, The Faculty of Graduate Studies, University of Manitoba, Winnipeg, MB, Canada, 1998. [Google Scholar]
- Agnihotri, R.; Bhardwaj, S. Synthesis and Characterization of Cuznal Based Shape Memory Alloys and to Optimize Behavior on Different Properties by Varying Weight Percentage. Int. J. Mater. Sci. Eng. 2016, 4, 229–234. [Google Scholar]
- Wang, Y.; Jing, T.; Peng, H.; He, W.; Yan, J.; Wang, S.; Li, N.; Wen, Y. Re-examination of martensitic stabilization in Cu-based shape memory alloys Part I. Identification of occurrence stage and degree of martensitic stabilization in CuZnAl alloys. J. Alloys Compd. 2022, 913, 165276. [Google Scholar] [CrossRef]
- Alaneme, K.K.; Okotete, E.A.; Oluwafemi, A.; Inyang, U. Assessment of the mechanical behaviour of thermally aged B and Fe modified CuZnAl shape memory alloys. Rev. De Metal. 2019, 55, 1–8. [Google Scholar] [CrossRef]
- Sampath, V. Improvement of Shape-Memory Characteristics and Mechanical Properties of Copper-Zinc-Aluminum Shape-Memory Alloy with Low Aluminum Content by Grain Refinement. Mater. Manuf. Process. 2006, 21, 789–795. [Google Scholar] [CrossRef]
- Sathish, S.; Mallik, U.S.; Raju, T.N. Corrosion Behavior of Cu-Zn-Ni Shape Memory Alloys. J. Miner. Mater. Charact. Eng. 2013, 1, 49–54. [Google Scholar] [CrossRef]
- Majed, R.A.; Elia, S.M.; Fadhel, Z.; Mohamed, D. Effect of Halogen Ions on the Corrosion of Brass in Na2SO4 Solution. Eng. Technol. J. 2010, 28, 1386–1395. [Google Scholar]
- Vrsalović, L.; Ivanić, I.; Čudina, D.; Lokas, L.; Kožuh, S.; Gojić, M. The Influence of Chloride Ion Concentration on the Corrosion Behavior of the Cualni Alloy. Tech. Bull. 2017, 11, 67–72. [Google Scholar]
- Qafsaou, W.; Blanc, C.; Peabeare, N.; Srhiri, A.; Mankowski, G. Study of different triazole derivative inhibitors to protect copper against pitting corrosion. J. Appl. Electrochem. 2000, 8, 959–966. [Google Scholar] [CrossRef]
- Lalitha, A.; Ramesh, S.; Rajeswari, S. Surface protection of copper in acid medium by azoles and surfactants. Electrochim. Acta 2005, 51, 47–55. [Google Scholar] [CrossRef]
- Gomma, G.K. Effect of azole compounds on corrosion of copper in acid medium. Mater. Chem. Phys. 1998, 56, 27–34. [Google Scholar] [CrossRef]
- Warraky, A.A.E. The effect of sulphide ions on the corrosion inhibition of copper in acidic chloride solutions. Anti-Corros. Methods Mater. 2003, 50, 40–46. [Google Scholar] [CrossRef]
- Ehteshamzade, M.; Shahrabi, T.; Hosseini, M.G. Inhibition of copper corrosion by self-assembled films of new Schiff bases and their modification with alkanethiols in aqueous medium. Appl. Surf. Sci. 2006, 252, 2949–2959. [Google Scholar] [CrossRef]
- Ma, H.; Chen, S.; Niu, L.; Zhao, S.; Li, S.; Li, D. Inhibition of copper corrosion by several Schiff bases in aerated halide solutions. Appl. Electrochem. 2002, 32, 65–72. [Google Scholar] [CrossRef]
- Sherif, E.M.; Park, S.M. Inhibition of copper corrosion in acidic pickling solutions by N-phenyl-1,4-phenylenediamine. Electrochim. Acta 2006, 51, 4665–4673. [Google Scholar] [CrossRef]
- Lisac, E.S.; Brnada, A.; Mance, A.D. Secondary amines as copper corrosion inhibitors in acid media. Corros. Sci. 2000, 42, 243–257. [Google Scholar] [CrossRef]
- Matos, J.B.; Pereira, L.P.; Agostinho, S.M.L.; Barcia, O.E.; Cordeiro, G.G.O.; D’Elia, E. Effect of cysteine on the anodic dissolution of copper in sulfuric acid medium. J. Electroanal. Chem. 2004, 570, 91–94. [Google Scholar] [CrossRef]
- Zhang, D.; Gao, L.; Zhou, G. Inhibition of copper corrosion in aerated hydrochloric acid solution by amino-acid Compounds. J. Appl. Electrochem. 2005, 35, 1081–1085. [Google Scholar] [CrossRef]
- Scendo, M. The effect of purine on the corrosion of copper in chloride solutions. Corros. Sci. 2007, 49, 373–390. [Google Scholar] [CrossRef]
- Subramanian, R.; Lakshminarayanan, V. Effect of adsorption of some azoles on copper passivation in alkaline medium. Corros. Sci. 2002, 44, 535–554. [Google Scholar] [CrossRef]
- Ye, X.R.; Xin, X.Q.; Zhu, J.J.; Xue, Z.L. Coordination compound films of 1-phenyl-5-mercaptotetrazole on copper surface. Appl. Surf. Sci. 1998, 135, 307–317. [Google Scholar] [CrossRef]
- Szocs, E.; Vastag, G.; Shaban, A.; Kalman, E. Electrochemical behavior of an inhibitor film formed on copper surface. Corros. Sci. 2005, 47, 893–908. [Google Scholar] [CrossRef]
- Szocs, E.; Vastag, G.; Shaban, A.; Konczos, G.; Kalman, E. Investigation of copper corrosion inhibition by STM and EQCM techniques. J. Appl. Electrochem. 1999, 29, 1339–1345. [Google Scholar] [CrossRef]
- Pi, J.; Chen, M.; Chen, T.; Wang, Q.; Cheng, S.; Fu, C. Corrosion inhibition effect of 1-phenyl-5-mercaptotetrazole on nickel-aluminum bronze in seawater: A combined experimental and theoretical study. Colloids Surf. A Physicochem. Eng. Asp. 2023, 666, 131354. [Google Scholar] [CrossRef]
- Altaf, F. Effect of Inhibitors on the Corrosion of Copper and its Alloys by Using Electrochemical Impedance Spectroscopy and Cyclic Voltammetry. Ph.D. Thesis, Department of Chemistry, Quaid-i-Azam University, Islamabad, Pakistan, 2013. [Google Scholar]
- Wang, J.L.; Wu, Y.G.; Liu, J. Effectiveness of Corrosion Inhibitors on Bronze and Cast Iron with Prefilming Treatment. Int. J. Electrochem. Sci. 2013, 8, 4631–4640. [Google Scholar] [CrossRef]
- Al-Otaibi, M.S.; Al-Mayouf, A.M.; Khan, M.; Mousa, A.A.; Al-Mazroa, S.A.; Alkhathlan, H.Z. Corrosion inhibitory action of some plant extracts on the corrosion of mild steel in acidic media. Arab. J. Chem. 2014, 7, 340–346. [Google Scholar] [CrossRef]
- Hrimla, M.; Bahsis, L.; Laamari, M.R.; Julve, M.; Stiriba, S.E. An Overview on the Performance of 1,2,3-Triazole Derivatives as Corrosion Inhibitors for Metal Surfaces. Int. J. Mol. Sci. 2022, 23, 16. [Google Scholar] [CrossRef] [PubMed]
- Mihit, M.; Laarej, K.; El Makarim, H.A.; Bazzi, L.; Salghi, R.; Hammouti, B. Study of the inhibition of the corrosion of copper and zinc in HNO3 solution by electrochemical technique and quantum chemical calculations. Arab. J. Chem. 2010, 3, 55–60. [Google Scholar] [CrossRef]
- Rao, B.V.A.; Reddy, M.N. Formation, characterization and corrosion protection efficiency of self-assembled 1-octadecyl-1H-imidazole films on copper for corrosion protection. Arab. J. Chem. 2017, 10, S3270–S3283. [Google Scholar] [CrossRef]
- Radovanović, M. Influence of Organic Inhibitors on Corrosion Behavior of Brass in Sodium Sulfate Solution. Ph.D. Thesis, University of Belgrade—Technical Faculty Bor, Bor, Serbia, 2012. [Google Scholar]
- Antonijevic, M.M.; Milic, S.M. Electrochemical behavior of Cu24Zn5Al alloy in alkaline medium in the presence of chloride ions and benzotriazole. Mater. Chem. Phys. 2009, 118, 385–391. [Google Scholar] [CrossRef]
- Huković, M.M.; Babić, R.; Paić, I. Copper corrosion at various pH values with and without the inhibitor. J. Appl. Electrochem. 2000, 30, 617–624. [Google Scholar] [CrossRef]
- Benzbiria, N.; Zertoubi, M.; Azzi, M. Oxygen reduction reaction kinetics on pure copper in neutral sodium sulfate solution. SN Appl. Sci. 2020, 2, 2101. [Google Scholar] [CrossRef]
- Gardic, V.; Gupta, V.; Antonijevic, M. Corrosion behaviour of Cu24Zn5Al alloy in a sodium tetraborate solution in the presence of 1-phenyl-5-mercaptotetrazole. Indian J. Chem. Technol. 2014, 21, 350–358. [Google Scholar]
- Kozlica, D.K.; Kokalj, A.; Milosev, I. Corrosion inhibition of copper and aluminium by 2-mercaptobenzimidazole and octylphosphonic acid—Surface pre-treatment and method of film preparation. Electrochim. Acta 2022, 431, 141154. [Google Scholar] [CrossRef]
- Mihit, M.; ElIssami, S.; Bouklah, M.; Bazzi, L.; Hammouti, B.; AitAddi, E.; Salghi, R.; Kertit, S. The inhibited effect of some tetrazolic compounds towards the corrosion of brass in nitric acid solution. Appl. Surf. Sci. 2006, 252, 2389–2395. [Google Scholar] [CrossRef]
- Kozlica, D.K.; Ekar, J.; Kovač, J.; Milošev, I. Roles of Chloride Ions in the Formation of Corrosion Protective Films on Copper. J. Electrochem. Soc. 2021, 168, 031504. [Google Scholar] [CrossRef]
- Kozlica, D.K.; Kokalj, A.; Milosev, I. Synergistic effect of 2-mercaptobenzimidazole and octylphosphonic acid as corrosion inhibitors for copper and aluminium—An electrochemical, XPS, FTIR and DFT study. Corros. Sci. 2021, 182, 109082. [Google Scholar] [CrossRef]
- Kokalj, A. On the use of the Langmuir and other adsorption isotherms in corrosion inhibition. Corros. Sci. 2023, 2017, 111112. [Google Scholar] [CrossRef]
- Yu, P.; Liao, D.M.; Luo, Y.B.; Chen, Z.G. Studies of Benzotriazole and Tolytriazole as Inhibitors for Copper Corrosion in Deionized Water. Corrosion 2003, 59, 314–318. [Google Scholar] [CrossRef]
- Wang, X.; Li, W.; Tan, B.; Wang, F.; Du, H.; Liu, R.; Han, X.; Zhang, S. Corrosion inhibition effect of benzimidazole and two derivatives on copper in alkaline environments: Experimental and theoretical analyses. J. Mol. Liq. 2023, 390, 122985. [Google Scholar] [CrossRef]
- Yin, M.; Li, Z.; Xiao, Z.; Pang, Y.; Li, Y.; Shen, Z. Corrosion behavior of Cu-Al-Mn-Zn-Zr shape memory alloy in NaCl solution. Trans. Nonferrous Met. Soc. China 2021, 31, 1012–1022. [Google Scholar] [CrossRef]
- Nady, H.M.; El-Rabiei, M.M.; Badawy, W.A. Stability of Some Copper Ternary Alloys in Chloride Solutions Polluted by Sulfide Ions. Chem. Process Eng. Res. 2014, 20, 35–44. [Google Scholar]
- Feng, Y.; Teo, W.K.; Siow, K.S.; Tag, K.L.; Hsieh, A.K. The Corrosion Behaviour of Copper in Neutral Tap Water. Part I: Corrosion Mechanisms. Corros. Sci. 1996, 38, 369–385. [Google Scholar] [CrossRef]
- Gudić, S.; Vrsalović, L.; Radeljić, A.; Oguzie, E.E.; Ivanić, I.; Kožuh, S.; Gojić, M. Comparison of Corrosion Behavior of Copper and Copper Alloys in Aqueous Chloride Solution. Chem. Ind. Chem. Eng. Q. 2021, 27, 383–394. [Google Scholar] [CrossRef]
- Nady, K.; El-Rabiei, M.M.; El-Hafez, G.M.A.; Tribo, J.B. Electrochemical Stability of Cu–10Al–10Zn, Cu–10Al–10Ni, and Cu–10Ni–10Zn Ternary Alloys in Simulated Physiological Solutions. J. Bio Tribo-Corros. 2016, 2, 28. [Google Scholar] [CrossRef]
- Shaik, M.A.; Syed, K.H.; Golla, B.R. Electrochemical behavior of mechanically alloyed hard Cu-Al alloys in marine environment. Corros. Sci. 2019, 153, 249–257. [Google Scholar] [CrossRef]
- Brotzu, A.; Filippo, B.D.; Natali, S.; Zortea, L. Corrosion behavior of Shape Memory Alloy in NaCl environment and deformation recovery maintenance in Cu-Zn-Al system. Frat. Ed Integrità Strutt. 2022, 62, 64–74. [Google Scholar] [CrossRef]
- Ding, Y.; Zhao, R.; Qin, Z.; Wu, Z.; Wang, L.; Liu, L.; Lu, W. Evolution of the Corrosion Product Film on Nickel-Aluminum Bronze and Its Corrosion Behavior in 3.5 wt % NaCl Solution. Materials 2019, 12, 209. [Google Scholar] [CrossRef] [PubMed]
- Al-Mobarak, N.A.; Khaled, K.F.; Hamed, M.N.H.; Abdel-Azim, K.M. Employing electrochemical frequency modulation for studying corrosion and corrosion inhibition of copper in sodium chloride solutions. Arab. J. Chem. 2011, 4, 185–193. [Google Scholar] [CrossRef]
- Khiatia, Z.; Othmanc, A.A.; Morenoa, M.S.; Bernarda, M.C.; Joireta, S.; Suttera, E.M.M.; Viviera, V. Corrosioninhibitionofcopperinneutralchloridemediabyanovelderivative of 1,2,4-triazole. Corros. Sci. 2011, 53, 3092–3099. [Google Scholar] [CrossRef]
- Yanga, J.; Zenga, y.; Zhua, M.; Liub, L.; Mengc, Y.; Chena, Y.; Maod, S. Study on effect of chloride and temperature on corrosion behavior of CoCrFeMnNi high entropy alloy. Int. J. Electrochem. Sci. 2023, 18, 100132. [Google Scholar] [CrossRef]


| Cu | Zn | Al | Fe | Other |
|---|---|---|---|---|
| 70.91 | 24.00 | 5.02 | 0.05 | 0.02 |
| Parameters | ||||||
|---|---|---|---|---|---|---|
| Inhibitor Concentration | OCP | Ecorr | jcorr | βc | βa | IE |
| Unit | ||||||
| mol/dm3 PMT | V vs. SCE | V vs. SCE | μA/cm2 | V/dec | V/dec | % |
| / | −0.030 | −0.040 | 4.7 | −0.292 | 0.148 | / |
| 1 × 10−5 | −0.040 | −0.054 | 2.1 | −0.238 | 0.087 | 54.9 |
| 1 × 10−4 | −0.035 | −0.054 | 1.9 | −0.379 | 0.084 | 58.4 |
| 5 × 10−4 | −0.022 | −0.020 | 2.0 | −0.302 | 0.120 | 56.4 |
| 1 × 10−3 | 0.012 | 0.018 | 1.6 | −0.299 | 0.091 | 66.1 |
| 2.5 × 10−3 | 0.010 | 0.011 | 2.4 | −0.440 | 0.110 | 48.5 |
| Parameters | |||||
|---|---|---|---|---|---|
| Concentration of Chloride Ions in 0.1 mol/dm3 Na2SO4 | OCP | Ecorr | jcorr | βc | βa |
| Unit | |||||
| mol/dm3 Cl− | V vs. SCE | V vs. SCE | μA/cm2 | V/dec | V/dec |
| / | −0.030 | −0.040 | 4.7 | −0.292 | 0.148 |
| 1 × 10−4 | −0.198 | −0.082 | 2.9 | −0.388 | 0.108 |
| 1 × 10−3 | −0.144 | −0.090 | 3.5 | −0.413 | 0.130 |
| 1 × 10−2 | −0.094 | −0.144 | 4.5 | −0.432 | 0.137 |
| 5 × 10−2 | −0.077 | −0.200 | 11.8 | −0.552 | 0.255 |
| Parameters | ||||||
|---|---|---|---|---|---|---|
| Immersion Time of Electrode in 0.017 mol/dm3 PMT Solution | OCP | Ecorr | jcorr | βc | βa | IE |
| Unit | ||||||
| min | V vs. SCE | V vs. SCE | μA/cm2 | V/dec | V/dec | % |
| 0 | −0.030 | −0.040 | 4.7 | −0.292 | 0.148 | / |
| 15 min | −0.035 | −0.157 | 1.9 | −0.390 | 0.196 | 59.6 |
| 60 min | −0.045 | −0.113 | 1.3 | −0.341 | 0.144 | 72.3 |
| 240 min | −0.035 | −0.105 | 0.9 | −0.344 | 0.159 | 80.8 |
| Parameters | |||
|---|---|---|---|
| Immersion Time of Electrode in 0.017 mol/dm3 PMT Solution | Range of Potential | Obtained Area by Integrating | IE |
| min | V vs. SCE | mVmA/cm2 | % |
| 0 | 0–1 | 0.911 | / |
| 60 min | 0–1 | 0.576 | 36.77 |
| 0 | 0–0.3 | 0.069 | / |
| 60 min | 0–0.3 | 0.027 | 60.87 |
| Parameters | |||||||
|---|---|---|---|---|---|---|---|
| 0.1 mol/dm3 Na2SO4 Containing Various Concentrations of Cl− | OCP | Ecorr | jcorr | βc | βa | IE | |
| Unit | |||||||
| mol/dm3 Cl− | V vs. SCE | V vs. SCE | μA/cm2 | V/dec | V/dec | % | % |
| Without immersion in 0.017 mol/dm3 PMT | |||||||
| / | −0.030 | −0.040 | 4.7 | −0.292 | 0.148 | / | / |
| After 60 min immersion time in 0.017 mol/dm3 PMT | |||||||
| / | −0.045 | −0.113 | 1.3 | −0.341 | 0.144 | 72.3 | / |
| 1 × 10−4 | −0.082 | −0.128 | 2.7 | −0.234 | 0.116 | 42.6 | 6.90 |
| 1 × 10−3 | −0.076 | −0.181 | 3.3 | −0.207 | 0.249 | 29.8 | 7.14 |
| 1 × 10−2 | −0.077 | −0.198 | 4.1 | −0.255 | 0.268 | 12.8 | 7.87 |
| 5 × 10−2 | −0.073 | −0.248 | 9.8 | −0.275 | 0.219 | n/a | 16.95 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gardić, V.; Tasić, Ž.Z.; Petrović Mihajlović, M.B.; Radovanović, M.B.; Antonijević, M.M. Corrosion Behavior of the Cu24Zn5Al Alloy in Sodium Sulfate Solution in the Presence of 1-Phenyl-5-mercaptotetrazole. Metals 2023, 13, 1863. https://doi.org/10.3390/met13111863
Gardić V, Tasić ŽZ, Petrović Mihajlović MB, Radovanović MB, Antonijević MM. Corrosion Behavior of the Cu24Zn5Al Alloy in Sodium Sulfate Solution in the Presence of 1-Phenyl-5-mercaptotetrazole. Metals. 2023; 13(11):1863. https://doi.org/10.3390/met13111863
Chicago/Turabian StyleGardić, Vojka, Žaklina Z. Tasić, Marija B. Petrović Mihajlović, Milan B. Radovanović, and Milan M. Antonijević. 2023. "Corrosion Behavior of the Cu24Zn5Al Alloy in Sodium Sulfate Solution in the Presence of 1-Phenyl-5-mercaptotetrazole" Metals 13, no. 11: 1863. https://doi.org/10.3390/met13111863
APA StyleGardić, V., Tasić, Ž. Z., Petrović Mihajlović, M. B., Radovanović, M. B., & Antonijević, M. M. (2023). Corrosion Behavior of the Cu24Zn5Al Alloy in Sodium Sulfate Solution in the Presence of 1-Phenyl-5-mercaptotetrazole. Metals, 13(11), 1863. https://doi.org/10.3390/met13111863








