Solvent Extraction of Metal Ions from Synthetic Copper Leaching Solution Using R4NCy
Abstract
1. Introduction
2. Materials and Methods
Liquid/Liquid Extraction
3. Results and Discussion
3.1. Effect of the R4NCy Concentration in Metal Ions Extraction
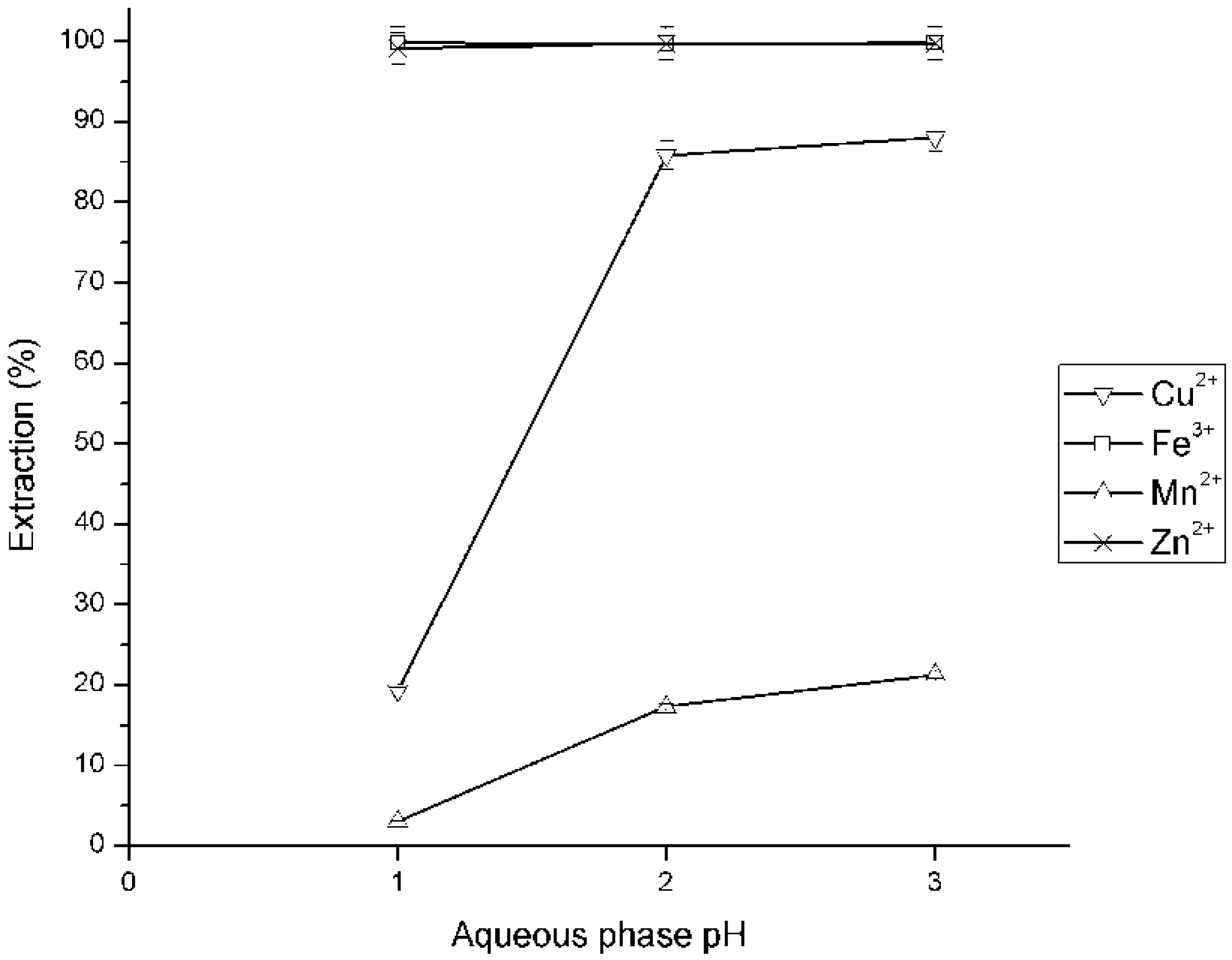
3.2. Effect of Initial pH in Metal Ions Extraction
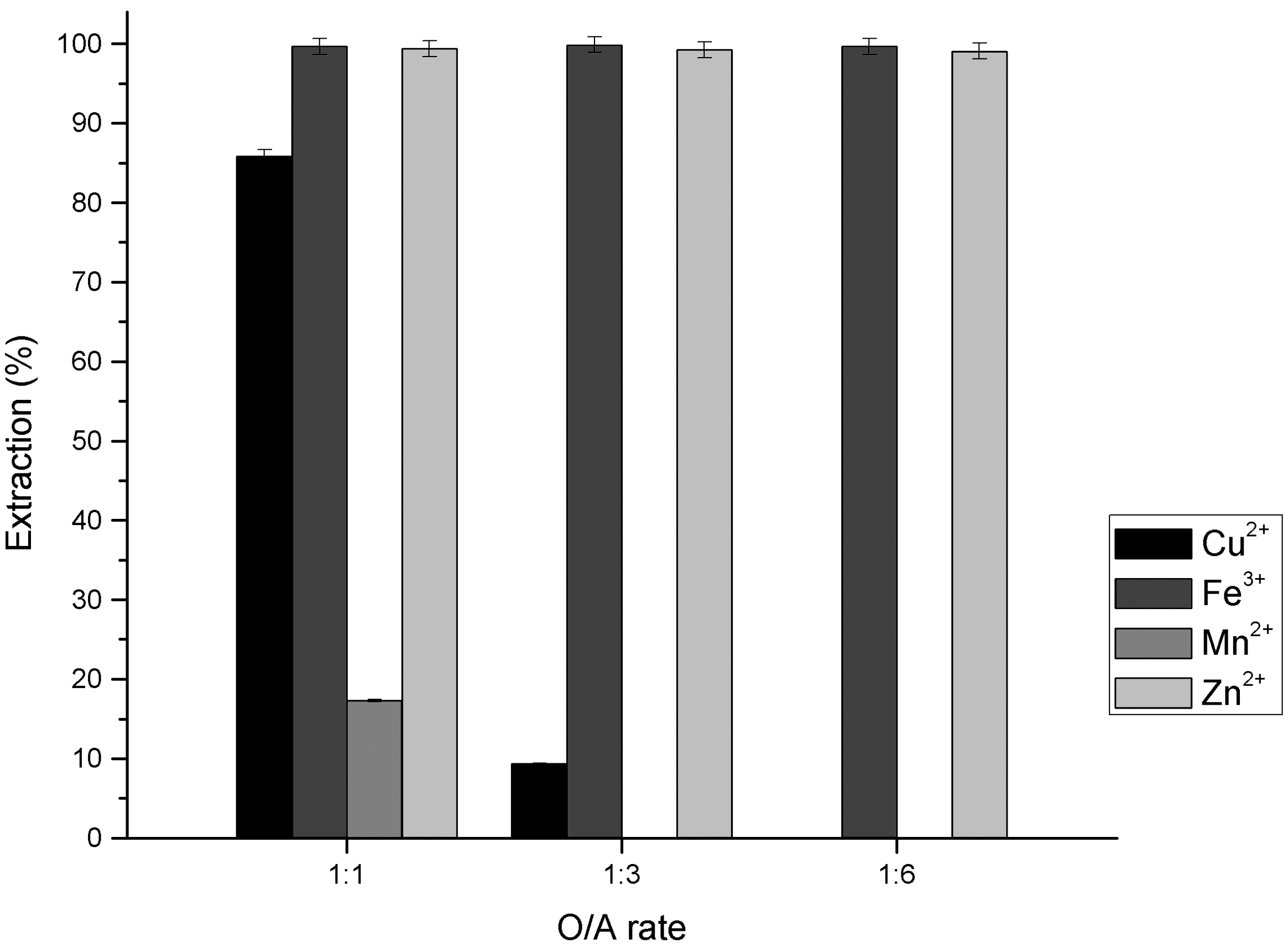
3.3. Effect of O/A Ratio in Metal Ions Extraction
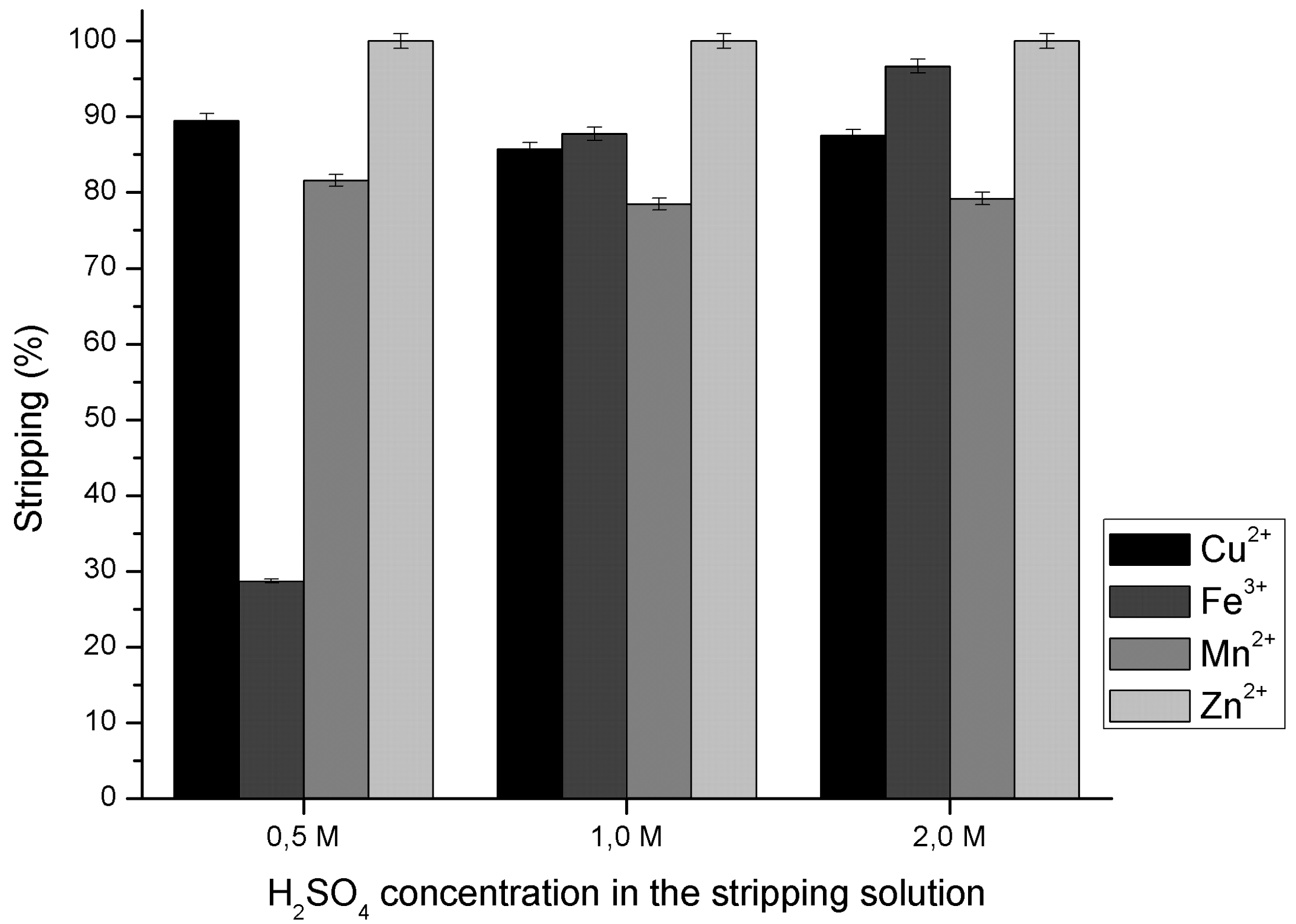
3.4. Effect of Sulfuric Acid in Metal Ions Stripping
3.5. Effect of O/A in Metal Ions Stripping
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ruiz, M.C.; González, I.; Rodriguez, V.; Padilla, R. Solvent Extraction of Copper from Sulfate–Chloride Solutions Using LIX 84-IC and LIX 860-IC. Miner. Processing Extr. Metall. Rev. 2021, 42, 1–8. [Google Scholar] [CrossRef]
- Mahmoudi, A.; Shakibania, S.; Rezaee, S.; Mokmeli, M. Effect of the Chloride Content of Seawater on the Copper Solvent Extraction Using Acorga M5774 and LIX 984N Extractants. Sep. Purif. Technol. 2020, 251, 117394. [Google Scholar] [CrossRef]
- Elizalde, M.P.; Rúa, M.S.; Menoyo, B.; Ocio, A. Solvent Extraction of Copper from Acidic Chloride Solutions with LIX 84. Hydrometallurgy 2019, 183, 213–220. [Google Scholar] [CrossRef]
- Zhang, W.; Xie, X.; Tong, X.; Du, Y.; Song, Q.; Feng, D. Study on the Effect and Mechanism of Impurity Aluminum on the Solvent Extraction of Rare Earth Elements (Nd, Pr, La) by P204-P350 in Chloride Solution. Minerals 2021, 11, 61. [Google Scholar] [CrossRef]
- Salinas, K.; Herreros, O.; Torres, C. Leaching of Primary Copper Sulfide Ore in Chloride-Ferrous Media. Minerals 2018, 8, 312. [Google Scholar] [CrossRef]
- Shakibania, S.; Mahmoudi, A.; Mokmeli, M.; Rashchi, F. The Effect of the Chloride Ion on Chemical Degradation of LIX 984N Extractant. Miner. Eng. 2020, 159, 106628. [Google Scholar] [CrossRef]
- Ruiz, M.C.; Risso, J.; Seguel, J.; Padilla, R. Solvent Extraction of Copper from Sulfate-Chloride Solutions Using Mixed and Modified Hydroxyoxime Extractants. Miner. Eng. 2020, 146, 106109. [Google Scholar] [CrossRef]
- Hernández, P.; Taboada, M.; Herreros, O.; Graber, T.; Ghorbani, Y. Leaching of Chalcopyrite in Acidified Nitrate Using Seawater-Based Media. Minerals 2018, 8, 238. [Google Scholar] [CrossRef]
- Araya, G.; Toro, N.; Castillo, J.; Guzmán, D.; Guzmán, A.; Hernández, P.; Jeldres, R.I.; Sepúlveda, R. Leaching of Oxide Copper Ores by Addition of Weak Acid from Copper Smelters. Metals 2020, 10, 627. [Google Scholar] [CrossRef]
- Velásquez-Yévenes, L.; Quezada-Reyes, V. Influence of Seawater and Discard Brine on the Dissolution of Copper Ore and Copper Concentrate. Hydrometallurgy 2018, 180, 88–95. [Google Scholar] [CrossRef]
- Li, G.; Wang, C.; Zhong, S.; Xie, H.; Chen, H. Identification and Regeneration of Degradation Products from Phenolic Hydroxyoxime-Based Extractant in Long-Term Copper Solvent Extraction Plant. Hydrometallurgy 2019, 183, 112–117. [Google Scholar] [CrossRef]
- Whewell, R.J.; Foakes, H.J.; Hughes, M.A. Degradation in Hydroxyoxime Solvent Extraction Systems. Hydrometallurgy 1981, 7, 7–26. [Google Scholar] [CrossRef]
- Sastre, A.M.; Szymanowski, J. Discussion of the Physicochemical Effects of Modifiers on the Extraction Properties of Hydroxyoximes. A Review. Solvent Extr. Ion Exch. 2004, 22, 737–759. [Google Scholar] [CrossRef]
- Xu, Z.; Zhou, T.; Zou, Q.; Wang, Y.; Wang, C.; Zhang, D.; Wang, F. Degradation of Hydroxyoxime Extractants by Nitration and Remedies with Inhibitor Reagent. Hydrometallurgy 2019, 183, 142–150. [Google Scholar] [CrossRef]
- Sperline, R.P.; Song, Y.; Ma, E.; Freiser, H. Organic Constituents of Cruds in Cu Solvent Extraction Circuits.: II. Photochemical and Acid Hydrolytic Reactions of Alkaryl Hydroxyoxime Reagents. Hydrometallurgy 1998, 50, 23–38. [Google Scholar] [CrossRef]
- Barnard, K.R.; Turner, N.L. Hydroxyoxime Stability and Unusual Cobalt Loading Behaviour in the LIX 63-Versatic 10-Tributyl Phosphate Synergistic System under Synthetic Laterite Conditions. Hydrometallurgy 2011, 109, 29–36. [Google Scholar] [CrossRef]
- Krzyzanowska, E.; Olszanowski, A. Photostability of Hydroxyoxime Extractants of Metals 2. Photo-Isomerization and Photo-Degradation of 2-Hydroxy-5-Methylbenzophenone (Z)-oxime. Hydrometallurgy 1995, 37, 375–385. [Google Scholar] [CrossRef]
- Barnard, K.R.; Turner, N.L. The Effect of Temperature on Hydroxyoxime Stability in the LIX 63-Versatic 10-Tributyl Phosphate Synergistic Solvent Extraction System under Synthetic Nickel Laterite Conditions. Hydrometallurgy 2011, 109, 245–251. [Google Scholar] [CrossRef]
- Chen, M.; Li, S.; Jin, C.; Shao, M.; Huang, Z. Selective Recovery of Platinum by Combining a Novel Reusable Ionic Liquid with Electrodeposition. Sep. Purif. Technol. 2021, 259, 118204. [Google Scholar] [CrossRef]
- Diabate, P.D.; Dupont, L.; Boudesocque, S.; Mohamadou, A. Novel Task Specific Ionic Liquids to Remove Heavy Metals from Aqueous Effluents. Metals 2018, 8, 412. [Google Scholar] [CrossRef]
- Jing, X.; Wu, Z.; Zhao, D.; Li, S.; Kong, F.; Chu, Y. Environmentally Friendly Extraction and Recovery of Cobalt from Simulated Solution of Spent Ternary Lithium Batteries Using the Novel Ionic Liquids of [C8H17NH2][Cyanex 272]. ACS Sustain. Chem. Eng. 2021, 9, 2475–2485. [Google Scholar] [CrossRef]
- Wei, G.-T.; Yang, Z.; Chen, C.-J. Room Temperature Ionic Liquid as a Novel Medium for Liquid/Liquid Extraction of Metal Ions. Anal. Chim. Acta 2003, 488, 183–192. [Google Scholar] [CrossRef]
- Nguyen, V.N.H.; Le, M.N.; Lee, M.S. Comparison of Extraction Ability between a Mixture of Alamine 336/Aliquat 336 and D2EHPA and Ionic Liquid ALi-D2 from Weak Hydrochloric Acid Solution. Metals 2020, 10, 1678. [Google Scholar] [CrossRef]
- Castillo, J.; Coll, M.T.; Fortuny, A.; Navarro Donoso, P.; Sepúlveda, R.; Sastre, A.M. Cu(II) Extraction Using Quaternary Ammonium and Quaternary Phosphonium Based Ionic Liquid. Hydrometallurgy 2014, 141, 89–96. [Google Scholar] [CrossRef]
- Sobekova Foltova, S.; vander Hoogerstraete, T.; Banerjee, D.; Binnemans, K. Samarium/Cobalt Separation by Solvent Extraction with Undiluted Quaternary Ammonium Ionic Liquids. Sep. Purif. Technol. 2019, 210, 209–218. [Google Scholar] [CrossRef]
- Singh, R.; Mahandra, H.; Gupta, B. Recovery of Zinc and Cadmium from Spent Batteries Using Cyphos IL 102 via Solvent Extraction Route and Synthesis of Zn and Cd Oxide Nanoparticles. Waste Manag. 2017, 67, 240–252. [Google Scholar] [CrossRef]
- Prusty, S.; Pradhan, S.; Mishra, S. Ionic Liquid as an Emerging Alternative for the Separation and Recovery of Nd, Sm and Eu Using Solvent Extraction Technique-A Review. Sustain. Chem. Pharm. 2021, 21, 100434. [Google Scholar] [CrossRef]
- Zeng, Z.; Su, X.; Gao, Y.; Yu, G.; Ni, S.; Su, J.; Sun, X. Separation of Lutetium Using a Novel Bifunctional Ionic Liquid Based on Phosphonate Functionalization. Sep. Purif. Technol. 2021, 264, 118439. [Google Scholar] [CrossRef]
- Padhan, E.; Sarangi, K. Recovery of Nd and Pr from NdFeB Magnet Leachates with Bi-Functional Ionic Liquids Based on Aliquat 336 and Cyanex 272. Hydrometallurgy 2017, 167, 134–140. [Google Scholar] [CrossRef]
- Tran, T.T.; Azra, N.; Iqbal, M.; Lee, M.S. Synthesis of Succinimide Based Ionic Liquids and Comparison of Extraction Behavior of Co(II) and Ni(II) with Bi-Functional Ionic Liquids Synthesized by Aliquat336 and Organophosphorus Acids. Sep. Purif. Technol. 2020, 238, 116496. [Google Scholar] [CrossRef]
- Wang, W.; Yang, H.; Cui, H.; Zhang, D.; Liu, Y.; Chen, J. Application of Bifunctional Ionic Liquid Extractants [A336][CA-12] and [A336][CA-100] to the Lanthanum Extraction and Separation from Rare Earths in the Chloride Medium. Ind. Eng. Chem. Res. 2011, 50, 7534–7541. [Google Scholar] [CrossRef]
- Yang, H.L.; Wang, W.; Cui, H.M.; Chen, J. Extraction Mechanism of Rare Earths with Bifuncional Ionic Liquids (Bif-ILs) [A336][CA-12]/[A336][CA-100] in Nitrate Medium. Fenxi Huaxue/ Chin. J. Anal. Chem. 2011, 39, 1561–1566. [Google Scholar] [CrossRef]
- Ismail, N.A.; Hisyam, A.; Shariff, S. Separation of Sm-Eu-Gd Mixed Solutions Using Bifunctional Ionic Liquid [A336][P204]. AIP Conf. Proc. 2019, 2124, 020053. [Google Scholar] [CrossRef]
- Alves-Lima, D.F.; Rodrigues, C.F.; Pinheiro, C.T.; Gando-Ferreira, L.M.; Quina, M.J.; Ferreira, A.G. Highly Selective Solvent Extraction of Zn(II) and Cr(III) with Trioctylmethylammonium Chloride Ionic Liquid. Can. J. Chem. Eng. 2022, 100, 131–142. [Google Scholar] [CrossRef]
- Swain, S.S.; Nayak, B.; Devi, N.; Das, S.; Swain, N. Liquid-Liquid Extraction of Cadmium(II) from Sulfate Medium Using Phosphonium and Ammonium Based Ionic Liquids Diluted in Kerosene. Hydrometallurgy 2016, 162, 63–70. [Google Scholar] [CrossRef]
- Zhu, Z.; Tulpatowicz, K.; Pranolo, Y.; Cheng, C.Y. Solvent Extraction of Molybdenum and Vanadium from Sulphate Solutions with Cyphos IL 101. Hydrometallurgy 2015, 154, 72–77. [Google Scholar] [CrossRef]
- Cieszynska, A.; Wisniewski, M. Extraction of Palladium(II) from Chloride Solutions with Cyphos IL 101/Toluene Mixtures as Novel Extractant. Sep. Purif. Technol. 2010, 73, 202–207. [Google Scholar] [CrossRef]
- Cui, L.; Cheng, F.; Zhou, J. Behaviors and Mechanism of Iron Extraction from Chloride Solutions Using Undiluted Cyphos IL 101. Ind. Eng. Chem. Res. 2015, 54, 7534–7542. [Google Scholar] [CrossRef]
- Zhu, Z.; Yoko, P.; Cheng, C.Y. Recovery of Cobalt and Manganese from Nickel Laterite Leach Solutions Containing Chloride by Solvent Extraction Using Cyphos IL 101. Hydrometallurgy 2017, 169, 213–218. [Google Scholar] [CrossRef]
- Pospiech, B. Studies on Extraction and Permeation of Cadmium(II) Using Cyphos IL 104 as Selective Extractant and Ion Carrier. Hydrometallurgy 2015, 154, 88–94. [Google Scholar] [CrossRef]
- Mahandra, H.; Singh, R.; Gupta, B. Liquid-Liquid Extraction Studies on Zn(II) and Cd(II) Using Phosphonium Ionic Liquid (Cyphos IL 104) and Recovery of Zinc from Zinc Plating Mud. Sep. Purif. Technol. 2017, 177, 281–292. [Google Scholar] [CrossRef]
- Rybka, P.; Regel-Rosocka, M. Nickel(II) and Cobalt(II) Extraction from Chloride Solutions with Quaternary Phosphonium Salts. Sep. Sci. Technol. 2012, 47, 1296–1302. [Google Scholar] [CrossRef]
- Fortuny, A.; Coll, M.T.; Sastre, A.M. Boron Removal from Seawater by Supported Liquid Membranes Using Cyphos IL 104 as a Carrier. Procedia Eng. 2012, 44, 807–808. [Google Scholar] [CrossRef][Green Version]
- Tran, T.T.; Lee, M.S. Interactions Between Ionic Liquid (ALiCY) and TBP and Their Use in Hydrometallurgy for Extracting Co(II) and Ni(II). Korean J. Met. Mater. 2020, 58, 423–432. [Google Scholar] [CrossRef]
- Cao, F.; Wang, W.; Wei, D.; Liu, W. Separation of Tungsten and Molybdenum with Solvent Extraction Using Functionalized Ionic Liquid Tricaprylmethylammonium Bis(2,4,4-Trimethylpentyl)Phosphinate. Int. J. Miner. Metall. Mater. 2021, 28, 1769–1776. [Google Scholar] [CrossRef]
- Castillo, J.; Toro, N.; Hernández, P.; Navarro, P.; Vargas, C.; Gálvez, E.; Sepúlveda, R. Extraction of Cu(II), Fe(III), Zn(II), and Mn(II) from Aqueous Solutions with Ionic Liquid R4NCy. Metals 2021, 11, 1585. [Google Scholar] [CrossRef]
- Regel-Rosocka, M.; Nowak, Ł.; Wiśniewski, M. Removal of Zinc(II) and Iron Ions from Chloride Solutions with Phosphonium Ionic Liquids. Sep. Purif. Technol. 2012, 97, 158–163. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, J.; Liu, S.; Hu, F.; Liu, M.; Jin, W.; Hu, J. Extraction Separation of Copper and Cobalt Dependent on Intermolecular Interaction between Cyanex302 and Cyphos IL101. Sep. Purif. Technol. 2020, 240, 116625. [Google Scholar] [CrossRef]
- Janiszewska, M.; Markiewicz, A.; Regel-Rosocka, M. Hydrometallurgical Separation of Co(II) from Ni(II) from Model and Real Waste Solutions. J. Clean. Prod. 2019, 228, 746–754. [Google Scholar] [CrossRef]
| Metal | Concentration (mg/L) |
|---|---|
| Cu(II) | 3360 |
| Fe(III) | 1570 |
| Mn(II) | 1000 |
| Zn(II) | 110 |
| Metal | IL Concentration (M) | E(%) | D | βCu(II) | βFe(III) | βMn(II) | βZn(II) |
|---|---|---|---|---|---|---|---|
| Cu(II) | 0.10 | 0.00 | 0.0 | - | 113,755.00 | 702.70 | 373.20 |
| 0.27 | 37.93 | 0.61 | - | 884.20 | 0.80 | 275.30 | |
| 0.54 | 85.82 | 6.05 | - | 55.30 | 0.1. | 60.40 | |
| Fe(III) | 0.10 | 99.70 | 336.63 | 0.00 | - | 0.00 | 0.00 |
| 0.27 | 99.82 | 540.38 | 0.00 | - | 0.00 | 0.30 | |
| 0.54 | 99.70 | 336.63 | 0.02 | - | 0.00 | 1.10 | |
| Mn(II) | 0.10 | 17.30 | 0.21 | 0.00 | 1609.20 | - | 0.50 |
| 0.27 | 32.50 | 0.48 | 1.27 | 1122.30 | - | 349.40 | |
| 0.54 | 47.10 | 0.89 | 6.79 | 375.70 | - | 410.70 | |
| Zn(II) | 0.10 | 10.00 | 0.11 | 0.00 | 3029.70 | 1.9 | - |
| 0.27 | 99.41 | 168.23 | 0.00 | 3.2 | 0.00 | - | |
| 0.54 | 99.73 | 365.67 | 0.02 | 0.9 | 0.00 | - |
| Metal | O/A Rate | Metal Concentration in Loaded IL (mg/L) | S(%) | Metal Concentration in Aqueous (mg/L) |
|---|---|---|---|---|
| Cu(II) | 1:1 | 85.7 | 2497.0 | |
| 3:1 | 2912.3 | 73.1 | 6391.0 | |
| 6:1 | 43.3 | 7569.0 | ||
| Fe(III) | 1:1 | 28.7 | 447.3 | |
| 3:1 | 1560.0 | 0.6 | 28.1 | |
| 6:1 | 0.5 | 42.4 | ||
| Mn(II) | 1:1 | 81.6 | 185.0 | |
| 3:1 | 226.7 | 63.3 | 430.4 | |
| 6:1 | 41.9 | 570.0 | ||
| Zn(II) | 1:1 | 100.0 | 110.0 | |
| 3:1 | 110.0 | 21.3 | 70.2 | |
| 6:1 | 2.4 | 15.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sepúlveda, R.; Toro, N.; Hernández, P.; Navarro, P.; Vargas, C.; Gálvez, E.; Castillo, J. Solvent Extraction of Metal Ions from Synthetic Copper Leaching Solution Using R4NCy. Metals 2022, 12, 1053. https://doi.org/10.3390/met12061053
Sepúlveda R, Toro N, Hernández P, Navarro P, Vargas C, Gálvez E, Castillo J. Solvent Extraction of Metal Ions from Synthetic Copper Leaching Solution Using R4NCy. Metals. 2022; 12(6):1053. https://doi.org/10.3390/met12061053
Chicago/Turabian StyleSepúlveda, Rossana, Norman Toro, Pía Hernández, Patricio Navarro, Cristian Vargas, Edelmira Gálvez, and Jonathan Castillo. 2022. "Solvent Extraction of Metal Ions from Synthetic Copper Leaching Solution Using R4NCy" Metals 12, no. 6: 1053. https://doi.org/10.3390/met12061053
APA StyleSepúlveda, R., Toro, N., Hernández, P., Navarro, P., Vargas, C., Gálvez, E., & Castillo, J. (2022). Solvent Extraction of Metal Ions from Synthetic Copper Leaching Solution Using R4NCy. Metals, 12(6), 1053. https://doi.org/10.3390/met12061053











