Effects of Thermomechanical Processing on Hydrogen Embrittlement Properties of UltraHigh-Strength TRIP-Aided Bainitic Ferrite Steels
Abstract
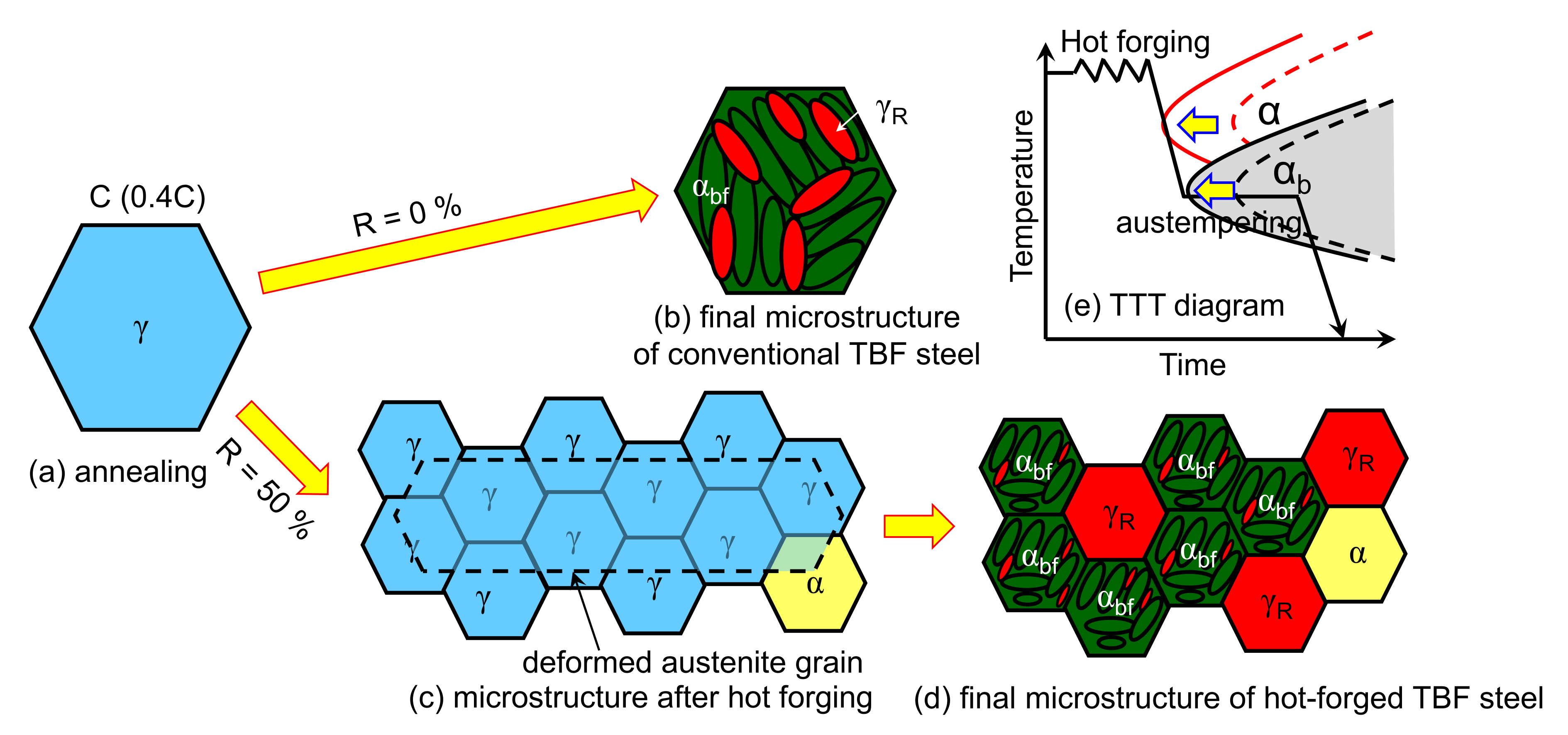
1. Introduction
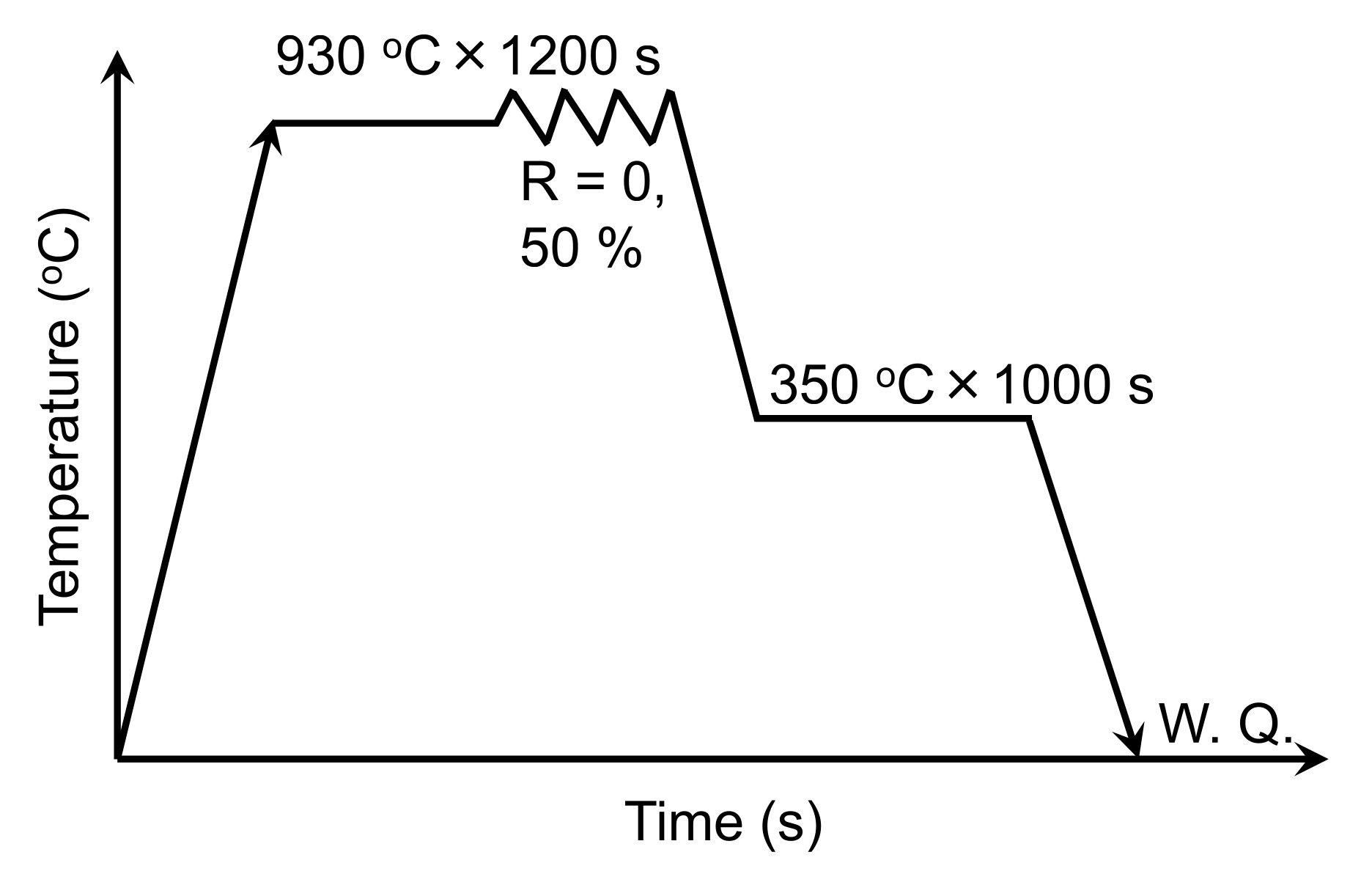
2. Materials and Methods
+ 0.0031Moγ + 0.0039Tiγ,
3. Results
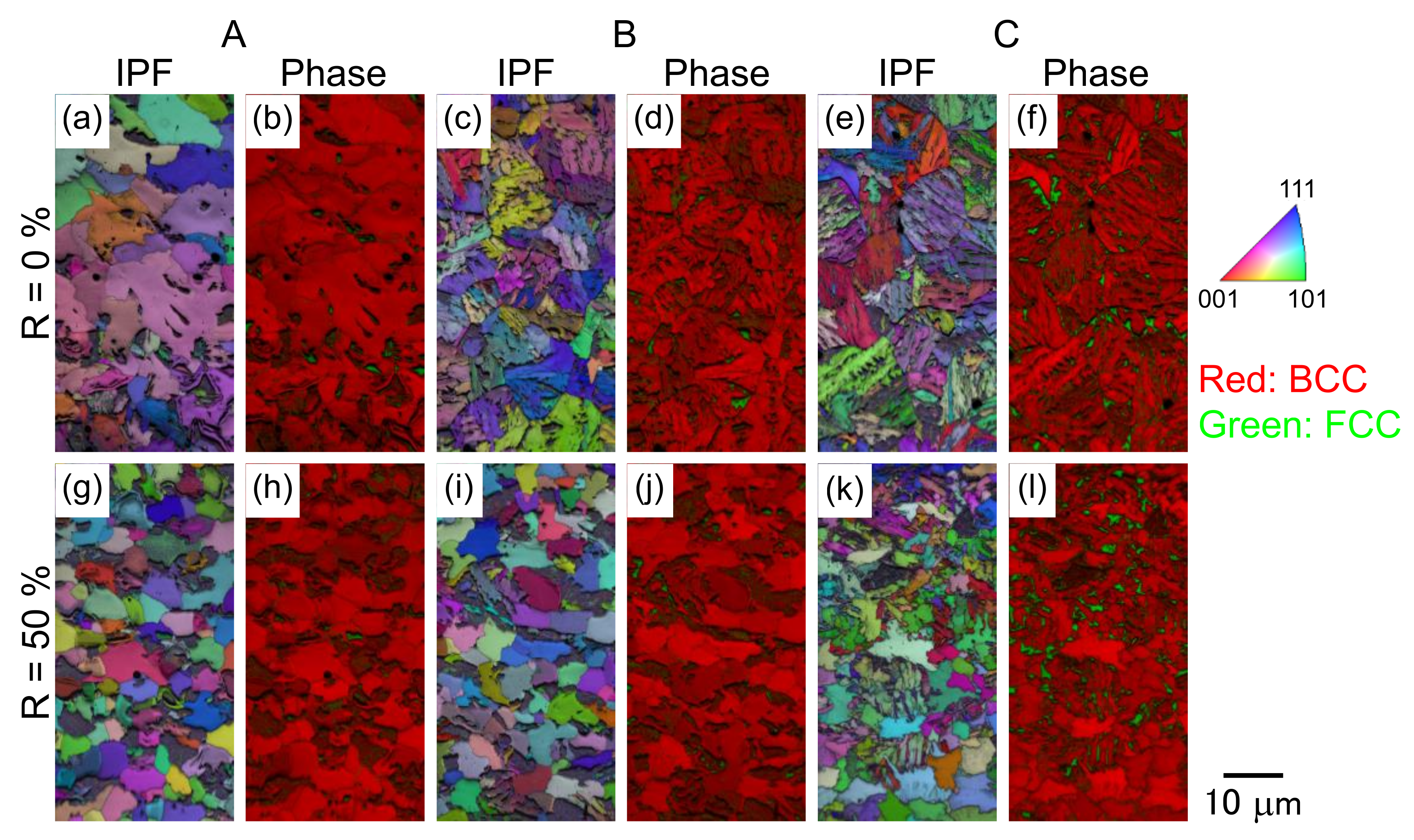
3.1. Microstructure and Tensile Properties
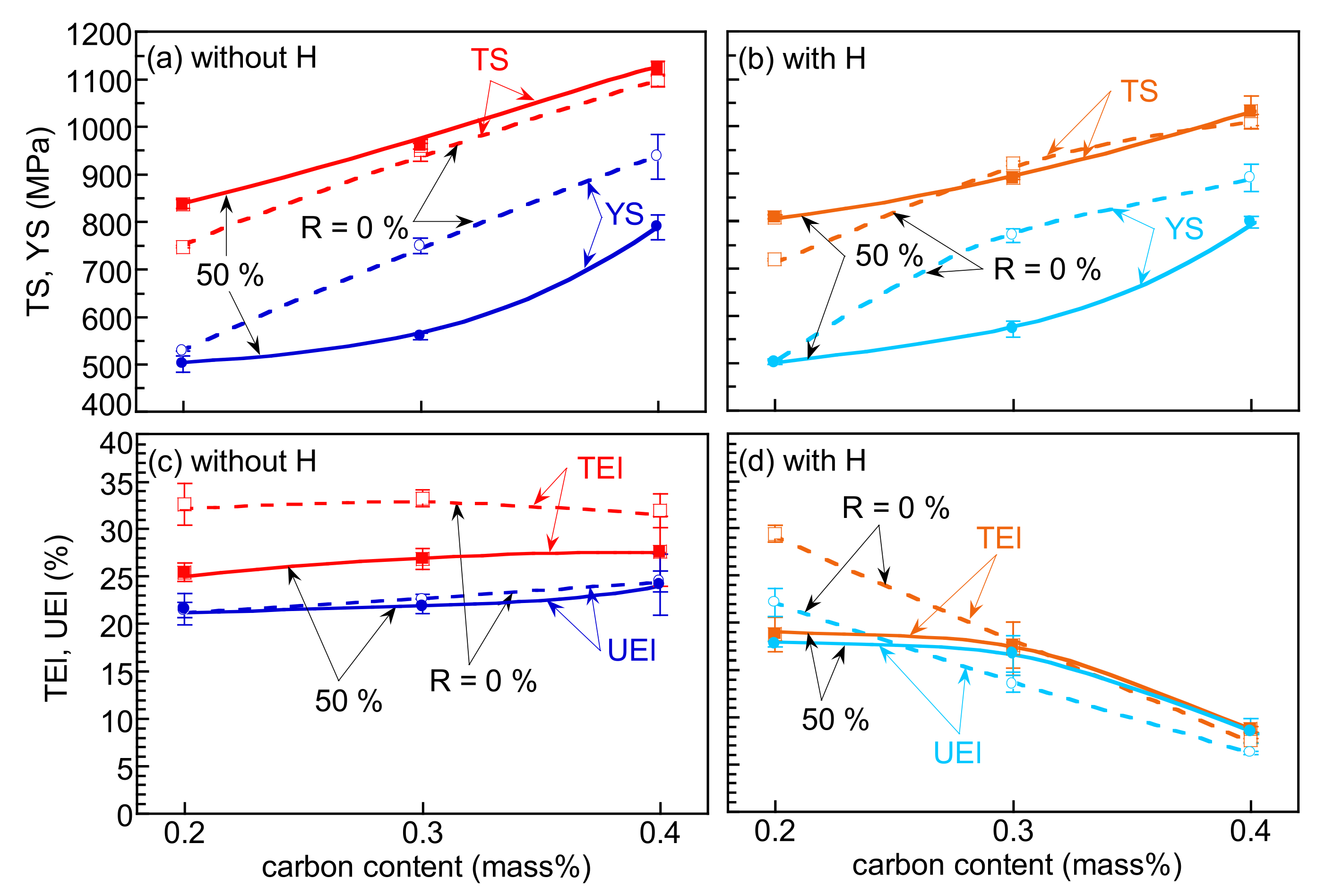
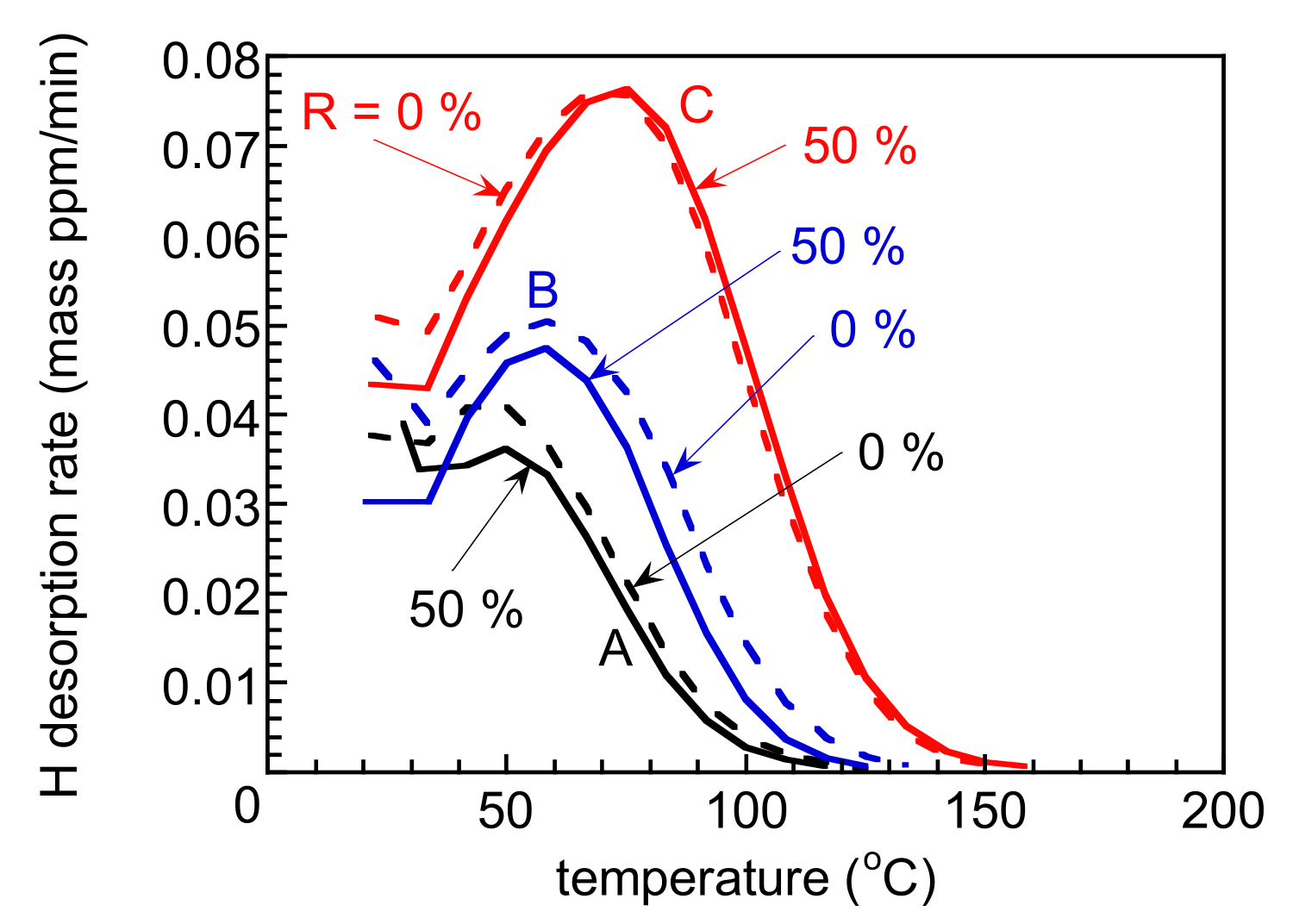
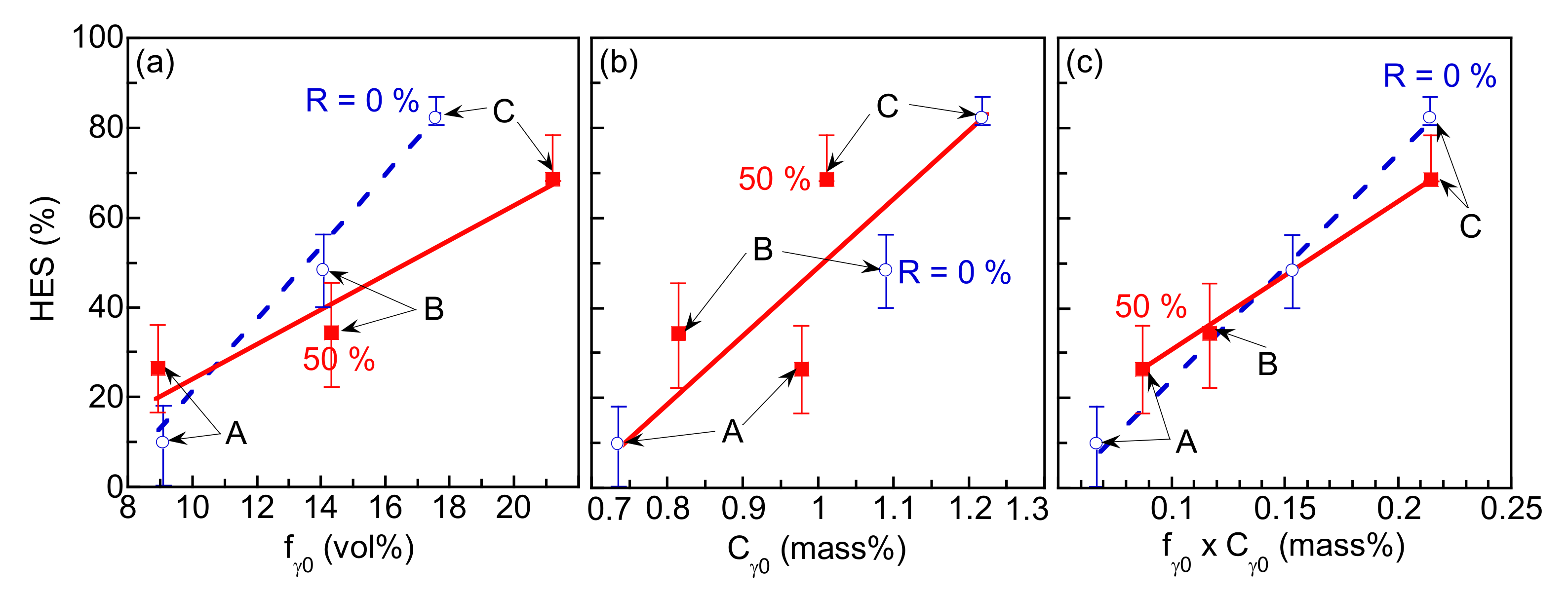
3.2. Hydrogen Embrittlement Properties Evaluated by Tensile Tests
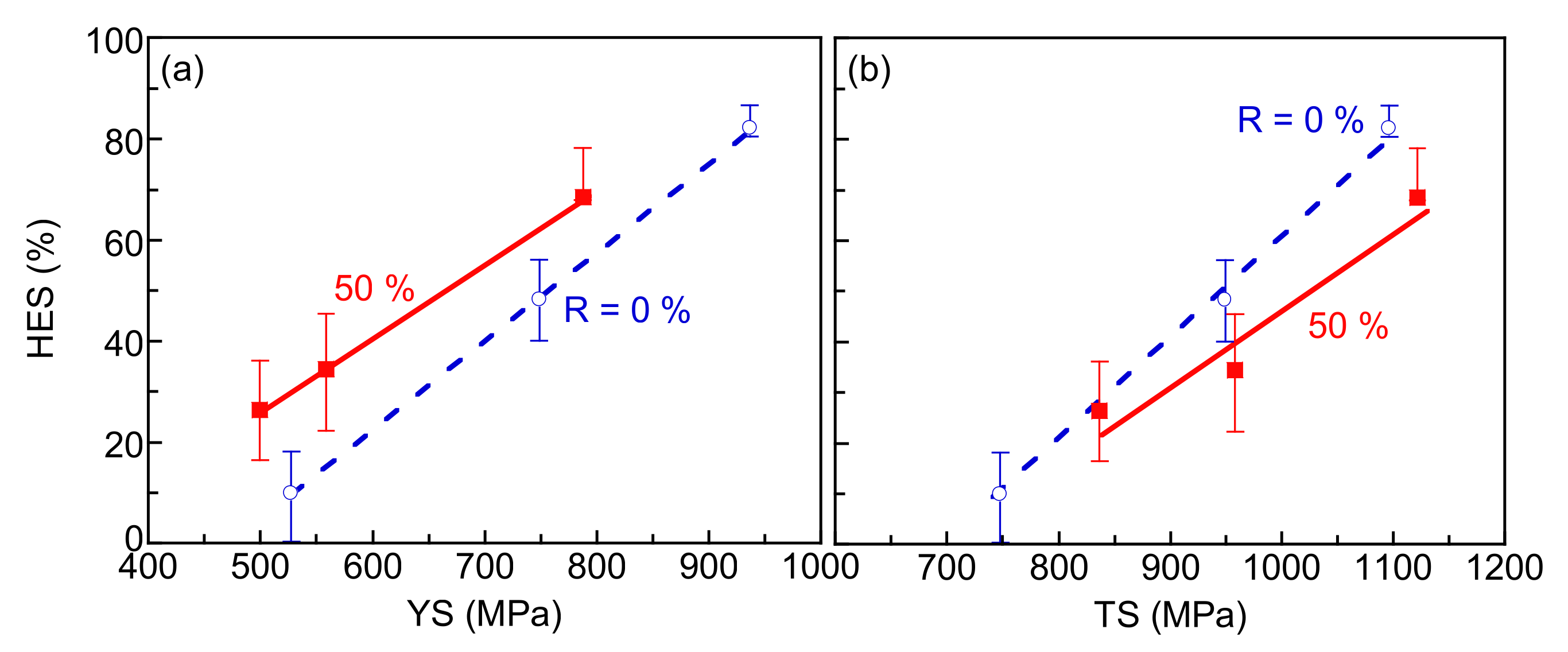
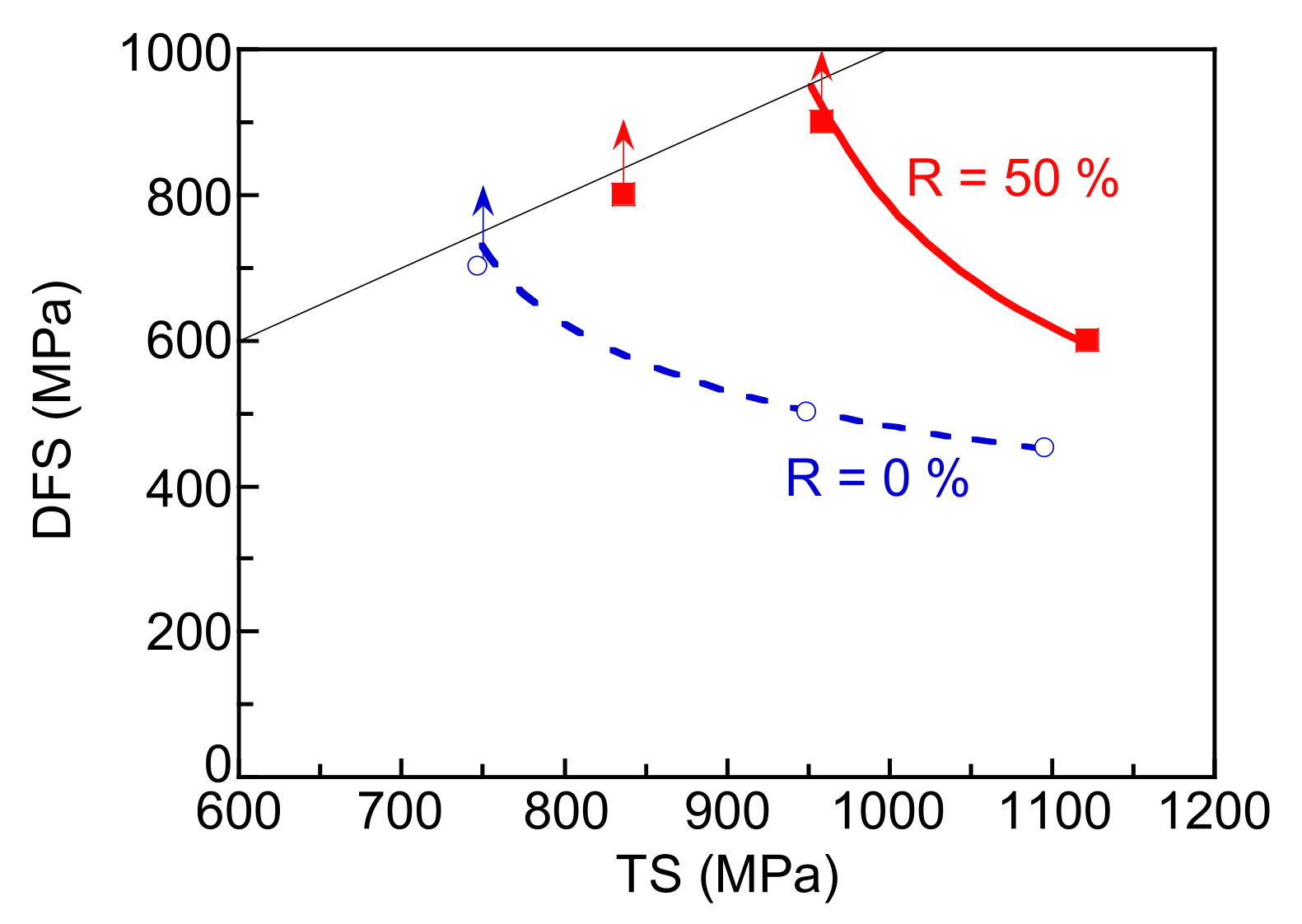
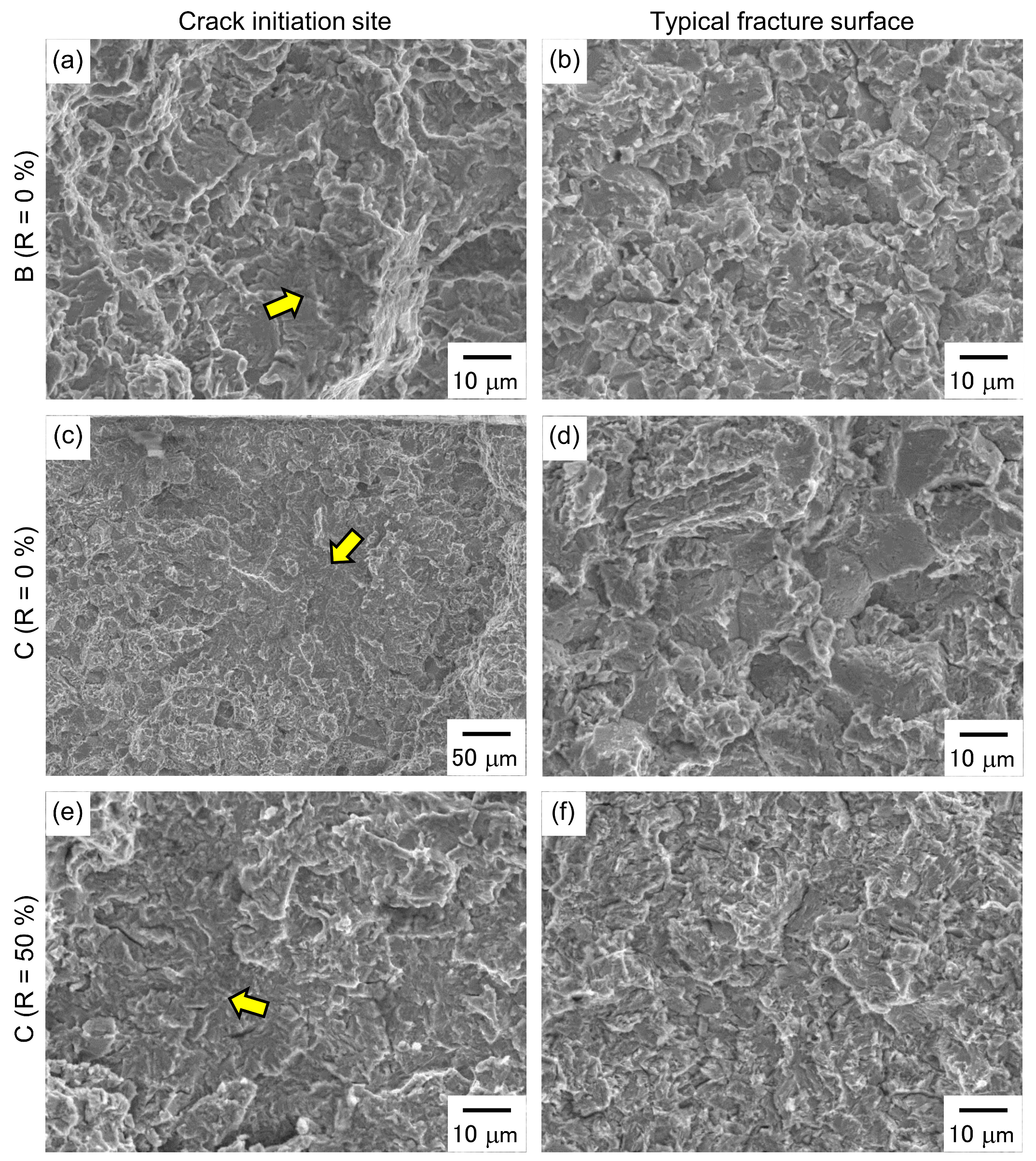
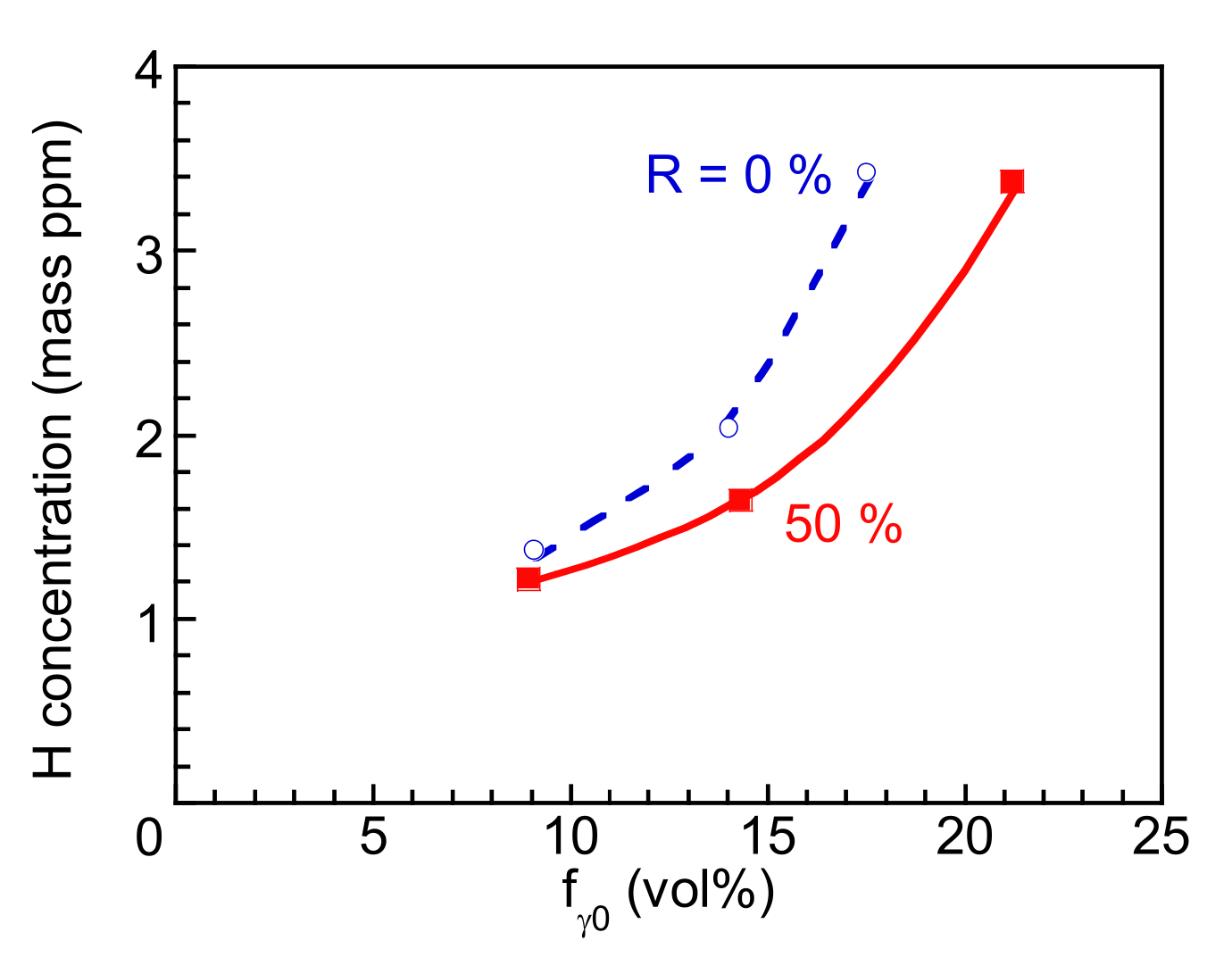
3.3. Hydrogen Embrittlement Properties Evaluated by Four-Point Bending Tests
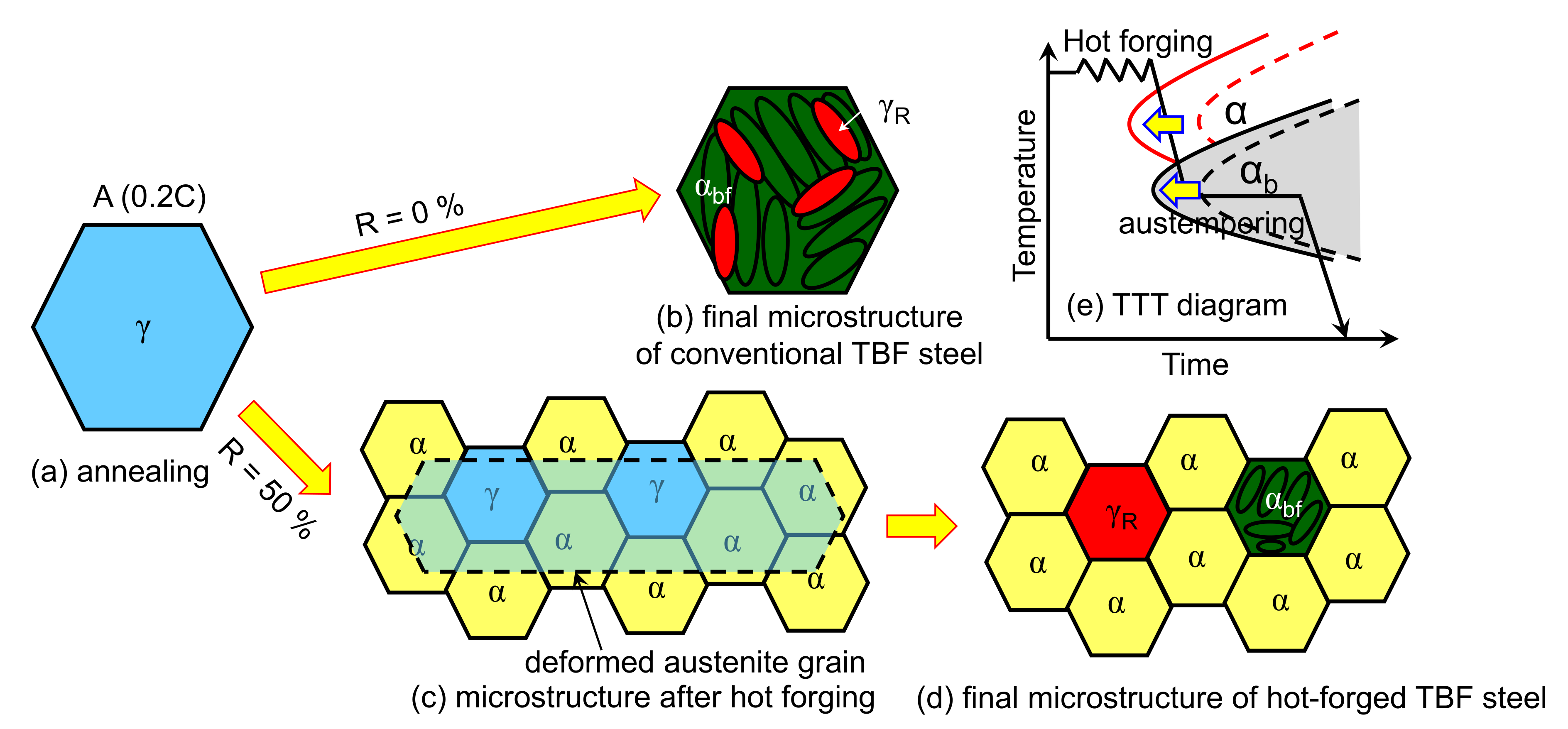
4. Discussion
4.1. Effects of Thermomechanical Processing on Microstructure Evolution and Mechanical Properties
4.2. Improvement of Hydrogen Embrittlement Properties by Thermomechanical Processing
4.3. Evaluation of Hydrogen Embrittlement Properties by Tensile Tests and Four-Point Bending Tests
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Matlock, D.K.; Speer, J.G.; De Moor, E.; Gibbs, P.J. Recent developments in advanced high strength sheet steels for automotive applications: An overview. JESTECH 2012, 15, 1–12. [Google Scholar]
- Heibel, S.; Dettinger, T.; Nester, W.; Clausmeyer, T.; Tekkaya, A.E. Damage mechanisms and mechanical properties of high-strength multiphase steels. Materials 2018, 11, 761. [Google Scholar] [CrossRef] [PubMed]
- Kozłowska, A.; Grzegorczyk, B.; Morawiec, M.; Grajcar, A. Explanation of the PLC effect in advanced high-strength medium-Mn steels. A review. Materials 2019, 12, 4175. [Google Scholar] [CrossRef] [PubMed]
- Zackay, V.F.; Parker, E.R.; Fahr, D.; Bush, R. The enhancement of ductility in high-strength steels. Trans. Am. Soc. Met. 1967, 60, 252–259. [Google Scholar]
- Branco, R.; Berto, F. Mechanical behavior of high-strength, low-alloy steels. Metals 2018, 8, 610. [Google Scholar] [CrossRef]
- Sugimoto, K.; Tsunezawa, M.; Hojo, T.; Ikeda, S. Ductility of 0.1-0.6C-1.5Si-1.5Mn ultra high-strength TRIP-aided sheet steels with bainitic ferrite matrix. ISIJ Int. 2004, 44, 1608–1614. [Google Scholar] [CrossRef]
- Sugimoto, K.; Hojo, T.; Srivastava, A.K. An overview of fatigue strength of case-hardening TRIP-aided martensitic steels. Metals 2018, 8, 355. [Google Scholar] [CrossRef]
- Sugimoto, K.; Mizuno, Y.; Natori, M.; Hojo, T. Effects of fine particle peening on fatigue strength of a TRIP-aided martensitic steel. Int. J. Fatigue 2017, 100, 206–214. [Google Scholar] [CrossRef]
- Hojo, T.; Kobayashi, J.; Sugimoto, K. Impact properties of low-alloy transformation-induced plasticity-steels with different matrix. Mater. Sci. Technol. 2016, 32, 1035–1042. [Google Scholar] [CrossRef]
- Caballero, F.G.; Roelofs, H.; Hasler, S.; Capdevila, C.; Chao, J.; Cornide, J.; Garcia-Mateo, C. Influence of bainite morphology on impact toughness of continuously cooled cementite free bainitic steels. Mater. Sci. Technol. 2013, 28, 95–102. [Google Scholar] [CrossRef]
- Kvackaj, T.; Bidulská, J.; Bidulský, R. Overview of HSS steel grades development and study of reheating condition fffects on austenite grain size changes. Materials 2021, 14, 1988. [Google Scholar] [CrossRef]
- Prislupčák, P.; Kvačkaj, T.; Bidulská, J.; Záhumenský, P.; Homolová, V.; Zimovčák, P. Austenite-ferrite transformation temperatures of C Mn Al HSLA steel. Acta Metall. Slovaca 2021, 27, 207–209. [Google Scholar] [CrossRef]
- Zhao, J.; Jiang, Z. Thermomechanical processing of advanced high strength steels. Prog. Mater. Sci. 2018, 94, 174–242. [Google Scholar] [CrossRef]
- Sugimoto, K.; Hayakawa, A.; Hojo, T.; Hashimoto, S.; Ikeda, S. Grain refinement of high strength low alloy TRIP-aided ferrous steels by thermomechanical processing in alpha plus gamma region. Tetsu Hagane 2003, 89, 1233–1239. [Google Scholar] [CrossRef][Green Version]
- Sugimoto, K.; Itoh, M.; Hojo, T.; Hashimoto, S.; Ikeda, S.; Arai, G. Microstructure and mechanical properties of ausformed ultra high-strength TRIP-aided steels. Mater. Sci. Forum 2007, 539-543, 4309–4314. [Google Scholar] [CrossRef]
- Sugimoto, K.; Hojo, T.; Srivastava, A. Low and medium carbon advanced high-strength forging steels for automotive applications. Metals 2019, 9, 1263. [Google Scholar] [CrossRef]
- Sugimoto, K.; Sato, S.; Kobayashi, J.; Srivastava, A.K. Effects of Cr and Mo on mechanical properties of hot-forged medium carbon TRIP-aided bainitic ferrite steels. Metals 2019, 9, 1066. [Google Scholar] [CrossRef]
- Hojo, T.; Kobayashi, J.; Kochi, T.; Sugimoto, K. Effects of thermomechanical processing on microstructure and shear properties of 22SiMnCrMoB TRIP-aided martensitic steel. Iron Steel Technol. 2015, 10, 102–110. [Google Scholar]
- Hojo, T.; Kochi, T.; Sugimoto, K. Effects of warm working on microstructural and shear deformation properties of TRIP-aided martenitic steel. Mater. Sci. Forum 2017, 879, 2312–2317. [Google Scholar] [CrossRef]
- Li, X.; Ma, X.; Zhang, J.; Akiyama, E.; Wang, Y.; Song, X. Review of hydrogen embrittlement in metals: Hydrogen diffusion, hydrogen characterization, hydrogen embrittlement mechanism and prevention. Acta Metall. Sin. (Engl. Lett.) 2020, 33, 759–773. [Google Scholar] [CrossRef]
- Martínez, C.; Briones, F.; Villarroel, M.; Vera, R. Effect of atmospheric corrosion on the mechanical properties of SAE 1020 structural steel. Materials 2018, 11, 596. [Google Scholar] [CrossRef] [PubMed]
- Sojka, J.; Vodárek, V.; Schindler, I.; Ly, C.; Jérôme, M.; Váňová, P.; Ruscassier, N.; Wenglorzová, A. Effect of hydrogen on the properties and fracture characteristics of TRIP 800 steels. Corros. Sci. 2011, 53, 2575–2581. [Google Scholar] [CrossRef]
- Laureys, A.; Depover, T.; Petrov, R.; Verbeken, K. Characterization of hydrogen induced cracking in TRIP-assisted steels. Int. J. Hydrog. Energy 2015, 40, 16901–16912. [Google Scholar] [CrossRef]
- Hojo, T.; Kikuchi, R.; Waki, H.; Nishimura, F.; Ukai, Y.; Akiyama, E. Effect of strain rate on the hydrogen embrittlement property of ultra high-strength low alloy TRIP-aided steel. ISIJ Int. 2018, 58, 751–759. [Google Scholar] [CrossRef]
- Hojo, T.; Kumai, B.; Koyama, M.; Akiyama, E.; Waki, H.; Saitoh, H.; Shiro, A.; Yasuda, R.; Shobu, T.; Nagasaka, A. Hydrogen embrittlement resistance of pre-strained ultra-high-strength low alloy TRIP-aided steel. Int J. Fract. 2020, 224, 253–260. [Google Scholar] [CrossRef]
- Hojo, T.; Kobayashi, J.; Sugimoto, K.; Nagasaka, A.; Akiyama, E. Effects of alloying elements addition on delayed fracture properties of ultra high-strength TRIP-aided martensitic steels. Metals 2019, 10, 6. [Google Scholar] [CrossRef]
- Dyson, D.J.; Holmes, B. Effect of alloying additions on the lattice parameter of austenite. J. Iron Steel Inst. 1970, 208, 469–474. [Google Scholar]
- Hojo, T.; Sugimoto, K.; Mukai, Y.; Ikeda, S. Effects of aluminum on delayed fracture properties of ultra high strength low alloy TRIP-aided steels. ISIJ Int. 2008, 48, 824–829. [Google Scholar] [CrossRef]
- Silveira, A.C.D.F.; Bevilaqua, W.L.; Dias, V.W.; De Castro, P.J.; Epp, J.; Rocha, A.D.S. Influence of hot forging parameters on a low carbon continuous cooling bainitic steel microstructure. Metals 2020, 10, 601. [Google Scholar] [CrossRef]
- Nürnberger, F.; Grydin, O.; Schaper, M.; Bach, F.W.; Koczurkiewicz, B.; Milenin, A. Microstructure transformations in tempering steels during continuous cooling from hot forging temperatures. Steel Res. Int. 2010, 81, 224–233. [Google Scholar] [CrossRef]
- Takaki, S.; Kawasaki, K.; Kimura, Y. Mechanical properties of ultra fine grained steels. J. Mater. Process. Technol. 2001, 117, 359–363. [Google Scholar] [CrossRef]
- Bergström, Y.; Hallén, H. Hall–petch relationships of iron and steel. Met. Sci. 2013, 17, 341–347. [Google Scholar] [CrossRef]
- Zan, N.; Ding, H.; Guo, X.; Tang, Z.; Bleck, W. Effects of grain size on hydrogen embrittlement in a Fe-22Mn-0.6C TWIP steel. Int. J. Hydrog. Energy 2015, 40, 10687–10696. [Google Scholar] [CrossRef]
- Koyama, M.; Ichii, K.; Tsuzaki, K. Grain refinement effect on hydrogen embrittlement resistance of an equiatomic cocrfemnni high-entropy alloy. Int. J. Hydrog. Energy 2019, 44, 17163–17167. [Google Scholar] [CrossRef]
- Hojo, T.; Koyama, M.; Terao, N.; Tsuzaki, K.; Akiyama, E. Transformation-assisted hydrogen desorption during deformation in steels: Examples of α´- and ε-martensite. Int. J. Hydrog. Energy 2019, 44, 30472–30477. [Google Scholar] [CrossRef]
- Koyama, M.; Yamasaki, D.; Ikeda, A.; Hojo, T.; Akiyama, E.; Takai, K.; Tsuzaki, K. Detection of hydrogen effusion before, during, and after martensitic transformation: Example of multiphase transformation-induced plasticity steel. Int. J. Hydrog. Energy 2019, 44, 26028–26035. [Google Scholar] [CrossRef]
- Timokhina, I.B.; Hodgson, P.D.; Pereloma, E.V. Effect of microstructure on the stability of retained austenite in transformation-induced-plasticity steels. Metall. Mater. Trans. A 2004, 35, 2331–2341. [Google Scholar] [CrossRef]
- Zwaag, S.V.D.; Zhao, L.; Kruijver, S.O.; Sietsma, J. Thermal and mechanical stability of retained austenite in aluminum-containing multiphase TRIP steels. ISIJ Int. 2004, 42, 1565–1570. [Google Scholar] [CrossRef]
- Xiong, X.C.; Chen, B.; Huang, M.X.; Wang, J.F.; Wang, L. The effect of morphology on the stability of retained austenite in a quenched and partitioned steel. Scr. Mater. 2013, 68, 321–324. [Google Scholar] [CrossRef]
- Shen, Y.F.; Qiu, L.N.; Sun, X.; Zuo, L.; Liaw, P.K.; Raabe, D. Effects of retained austenite volume fraction, morphology, and carbon content on strength and ductility of nanostructured TRIP-assisted steels. Mater. Sci. Eng. A 2015, 636, 551–564. [Google Scholar] [CrossRef]
- Jacques, P.J.; Ladrie`Re, J.; Delannay, F. On the influence of interactions between phases on the mechanical stability of retained austenite in transformation-induced plasticity multiphase steels. Metall. Mater. Trans. A 2001, 32, 2759–2768. [Google Scholar] [CrossRef]
- Bhadeshia, H.K.D.H. Prevention of hydrogen embrittlement in steels. ISIJ Int. 2016, 56, 24–36. [Google Scholar] [CrossRef]
- Momotani, Y.; Shibata, A.; Terada, D.; Tsuji, N. Effect of strain rate on hydrogen embrittlement in low-carbon martensitic steel. Int. J. Hydrog. Energy 2017, 42, 3371–3379. [Google Scholar] [CrossRef]
- Nagao, A.; Hayashi, K.; Oi, K.; Mitao, S. Effect of uniform distribution of fine cementite on hydrogen embrittlement of low carbon martensitic steel plates. ISIJ Int. 2012, 52, 213–221. [Google Scholar] [CrossRef]
- Takagi, S.; Hagihara, Y.; Hojo, T.; Urushihara, W.; Kawasaki, K. Comparison of hydrogen embrittlement resistance of high strength steel sheets evaluated by several methods. ISIJ Int. 2016, 56, 685–692. [Google Scholar] [CrossRef]
- Hirth, J.P.; Carnahan, B. Hydrogen adsorption at dislocations and cracks in Fe. Acta Metall. 1978, 26, 1795–1803. [Google Scholar] [CrossRef]



| Steels | C | Si | Mn | P | S | Al | Nb | Ti | B | O | N |
|---|---|---|---|---|---|---|---|---|---|---|---|
| A | 0.20 | 1.52 | 1.50 | 0.004 | 0.0021 | 0.039 | 0.05 | 0.02 | 0.0018 | 0.001 | 0.0011 |
| B | 0.28 | 1.51 | 1.52 | <0.005 | 0.0011 | 0.041 | 0.051 | 0.02 | 0.0016 | 0.001 | 0.0012 |
| C | 0.42 | 1.50 | 1.51 | <0.005 | 0.0009 | 0.043 | 0.05 | 0.02 | 0.0018 | 0.0019 | 0.0035 |
| Steels | R | TS | YS | YR | TEl | UEl | RA | TS × TEl | HES | fγ0 | Cγ0 | d |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | 0 50 | 748 837 | 528 500 | 0.71 0.60 | 32.6 25.4 | 21.4 21.5 | 39.9 36.2 | 24.4 21.3 | 9.8 26.4 | 9.1 8.9 | 0.74 0.98 | 10.7 7.4 |
| B | 0 50 | 950 959 | 749 559 | 0.79 0.58 | 33.2 26.8 | 22.5 21.8 | 38.7 32.0 | 31.5 25.7 | 48.2 34.3 | 14.1 14.3 | 1.09 0.82 | 12.8 7.0 |
| C | 0 50 | 1097 1122 | 937 788 | 0.86 0.71 | 31.9 27.6 | 24.4 24.1 | 44.7 32.2 | 35.0 31.0 | 85.3 68.5 | 17.6 21.1 | 1.22 1.01 | 16.3 7.3 |
| Steel | R (%) | HC (Mass ppm) |
|---|---|---|
| A | 0 50 | 1.36 1.21 |
| B | 0 50 | 2.03 1.64 |
| C | 0 50 | 3.41 3.38 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hojo, T.; Zhou, Y.; Kobayashi, J.; Sugimoto, K.-i.; Takemoto, Y.; Nagasaka, A.; Koyama, M.; Ajito, S.; Akiyama, E. Effects of Thermomechanical Processing on Hydrogen Embrittlement Properties of UltraHigh-Strength TRIP-Aided Bainitic Ferrite Steels. Metals 2022, 12, 269. https://doi.org/10.3390/met12020269
Hojo T, Zhou Y, Kobayashi J, Sugimoto K-i, Takemoto Y, Nagasaka A, Koyama M, Ajito S, Akiyama E. Effects of Thermomechanical Processing on Hydrogen Embrittlement Properties of UltraHigh-Strength TRIP-Aided Bainitic Ferrite Steels. Metals. 2022; 12(2):269. https://doi.org/10.3390/met12020269
Chicago/Turabian StyleHojo, Tomohiko, Yutao Zhou, Junya Kobayashi, Koh-ichi Sugimoto, Yoshito Takemoto, Akihiko Nagasaka, Motomichi Koyama, Saya Ajito, and Eiji Akiyama. 2022. "Effects of Thermomechanical Processing on Hydrogen Embrittlement Properties of UltraHigh-Strength TRIP-Aided Bainitic Ferrite Steels" Metals 12, no. 2: 269. https://doi.org/10.3390/met12020269
APA StyleHojo, T., Zhou, Y., Kobayashi, J., Sugimoto, K.-i., Takemoto, Y., Nagasaka, A., Koyama, M., Ajito, S., & Akiyama, E. (2022). Effects of Thermomechanical Processing on Hydrogen Embrittlement Properties of UltraHigh-Strength TRIP-Aided Bainitic Ferrite Steels. Metals, 12(2), 269. https://doi.org/10.3390/met12020269










