3. Experimental Procedure
To be able to realise charging conditions as accurately as possible, especially for materials with high diffusion coefficients, an apparatus for pressurised hydrogen charging is presented, which is characterised by an agile thermal behaviour and short process times. Thereby, the hydrogen effusion after completion of the charging processes can be minimised.
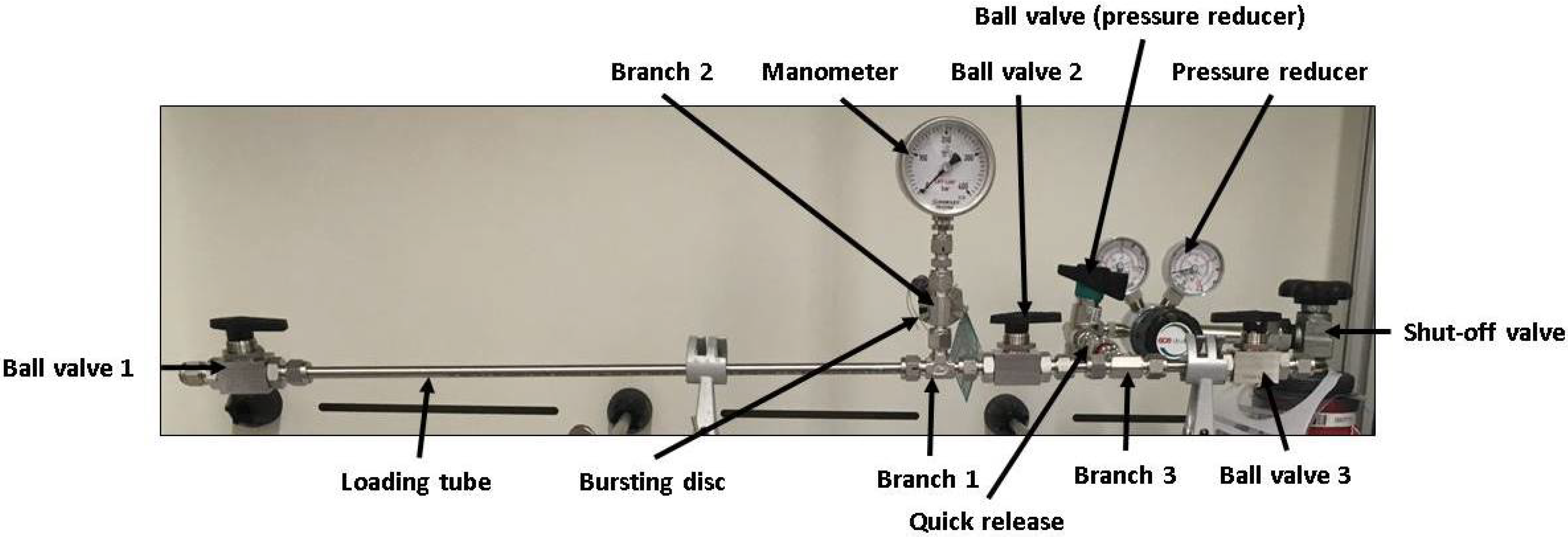
The used apparatus is shown in
Figure 2. The loading chamber is realised by a thin-walled stainless steel tube (wall-thickness 1 mm, material type 1.4435) exhibiting a small reactor volume. Ball valves are attached to the ends of the charging tube, which allow a sufficient sealing of the tube during the charging process, as well as enable a fast loading and unloading of the metallic samples. The samples used are 3 mm in diameter and 10 mm in length and can be transferred into the tubular autoclave through a ball valve (Ball Valve 1, see
Figure 2). The sample geometry ensures a fast loading and unloading of the sample as well as a fast hydrogen charging. Furthermore, a sample weight of approximately 0.5 g is sufficient for hydrogen contents to be reliably determined by application of TDS.
The current pressure in the charging tube is controlled via a pressure gauge and for safety reasons the device is equipped with a bursting disc. A hydrogen gas-bottle is used to apply pressure via a quick connector, which allows disconnecting the device from the gas-bottle during the runtime of the charging experiments. The heating of the charging chamber is realised by a heating sleeve, which is placed in a mid position around the charging tube. The specimens are positioned centrally between Ball Valve 1 and Ball Valve 2 in the loading tube. The autoclave is aligned as horizontally as possible, so that there is a linear contact surface between the outer surface of the specimens and the inner tube wall. Whether this contact surface has an influence on the loading kinetics is not investigated further.
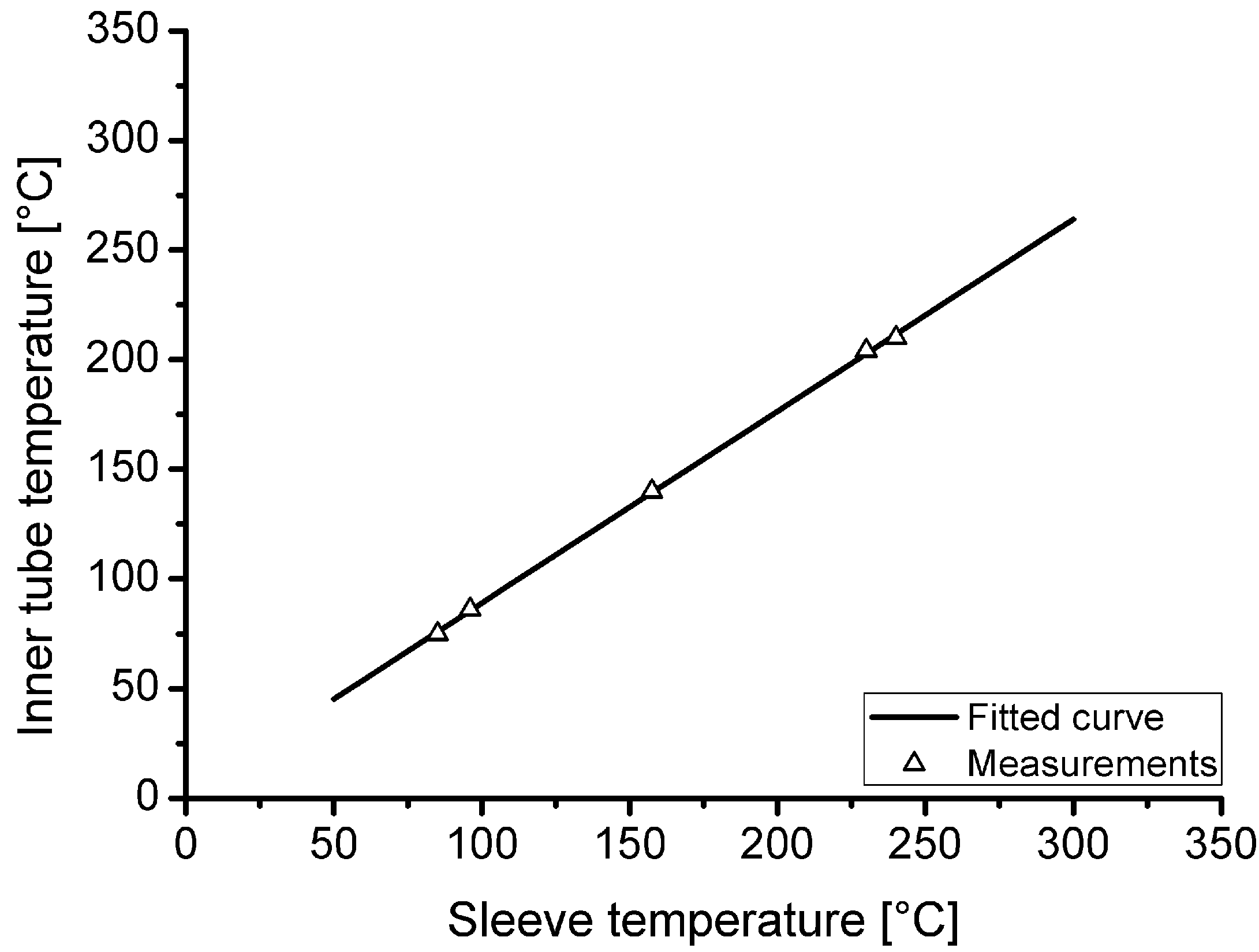
Because the temperature inside the loading tube is not measured during hydrogen charging, a thermal investigation was carried out in advance to evaluate the actual temperature as a function of the heating sleeve temperature. Furthermore, the heating and cooling behaviour as well as the temperature gradient in the tube were investigated.
Figure 3 shows the correlation of the measured temperature inside the tube as function of the heating sleeve temperature. In the area of the innermost 50 mm of the heated tube section, a maximum temperature difference of 2.5 °C in air temperature was measured at steady state for a target loading temperature of 210 °C.
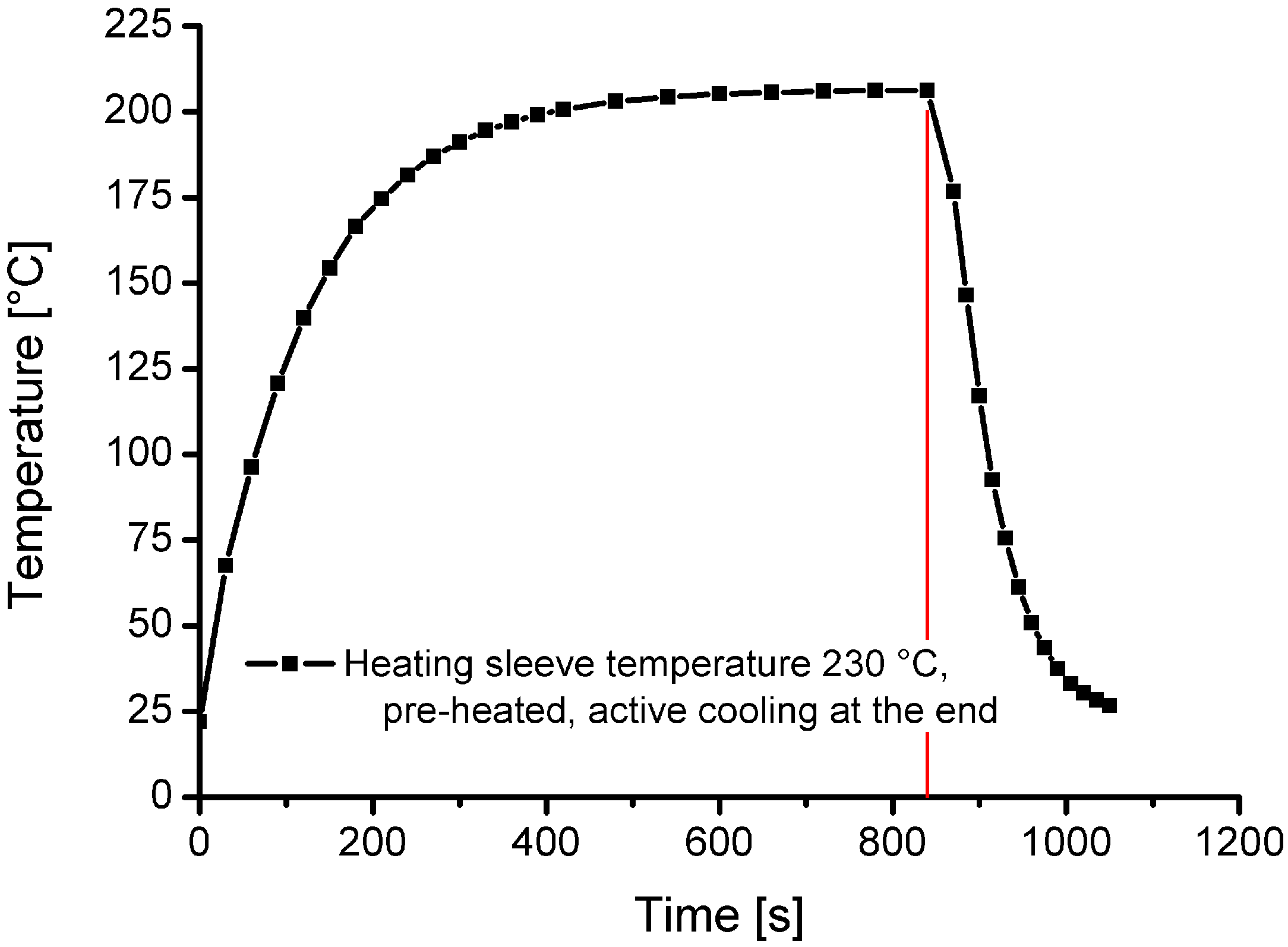
Measured cooling and heating curves for a charging temperature of 200 °C are displayed in
Figure 4. The presented curves and results were recorded in previous reference measurements, in an unpressurised state under ambient conditions (atmospheric pressure, room temperature and air).
Figure 4 shows that the heating time from room temperature to 200 °C is approximately 480 s. The cooling time (with forced cooling) from 200 °C to less than 30 °C is around 180 s.
Hydrogen chargings are performed as follows. In one batch, three samples are introduced via a ball valve into the device and placed at a specific position using a magnet. The device is purged with hydrogen gas and then four subsequent pressure purgings are performed from 15 MPa to approximately 1 MPa. After the last pressure purging, the desired hydrogen pressure for the charging batch is adjusted. At this point, the (pre-heated) heating sleeve is applied and kept on the tube for the specified time. Once the desired holding time is reached, the cooling process is initiated. Therefore, the heating sleeve is removed and an axial fan is placed directly in front of the charging tube. After sufficient cooling to temperatures below 60 °C, the pressure is relieved from the device and the samples are unloaded. Directly after unloading, the samples are stored in a cryogenic container at approximately −156 °C, in which the hydrogen charged condition can be preserved. This procedure is repeated for three different pressures with three different long charging times each, so that nine batches are loaded with three samples each. The corresponding charging pressures and charging times are shown in
Table 3.
Hydrogen content measurements are performed with a TDS device as described in [
15], which was calibrated using titanium hydride. The samples are therefore separately taken from the cryogenic container, cleaned and defrosted in isopropanol to avoid ice formation on the sample surface. Afterwards, the samples are dried using a pressurised nitrogen flow and introduced into the TDS device. Once a sufficient vaccuum is achieved, the samples are heated from room temperature to 700 °C with a heating rate of 0.2 K/s. The emitted hydrogen is measured via a mass spectrometer and a desorption spectra is obtained as function of the temperature as described e.g., in [
15,
17]. To obtain the hydrogen contents the TDS spectra are integrated over temperature. Together with the calibration constant, determined with TiH
2, the hydrogen content can be determined (see [
15]). The time between the start of the measurement and the extraction from the cryo-container is usally less than 6 min.
With regard to the binding energy, however, it should be noted that temperature dependent desorption behaviour also depends on the sample geometry, heating rate as well as on the diffusion behaviour of the material. Therefore, the obtained curves are not readily comparable to other data sources [
18].
The established autoclave set-up was used to investigate the hydrogen uptake behaviour of the martensitic stainless steel 1.4418. Samples were exposed to hydrogen pressures of 1.1, 6.7 and 15 MPa at a charging temperature of 250 °C. The given pressures were achieved after heating from room temperature to 250 °C. The initially at room temperature adjusted pressures were 1, 6 and 13.5 MPa respectively. To capture the sorption behaviour dependent on the experimental conditions (temperature and pressure), different charging times at 250 °C peak temperature were scheduled.
4. Results and Discussion
The samples were charged using the procedure described above. The realised parameters are compiled in
Table 3. As charging temperature, 250 °C was chosen. The heating sleeve temperature was adjusted according to
Figure 3 to 284 °C.
Each charging batch included three samples. After charging with hydrogen under the given conditions in
Table 3, the samples were stored in a cryo-container for transportation and finally measured using thermal desorption spectroscopy.
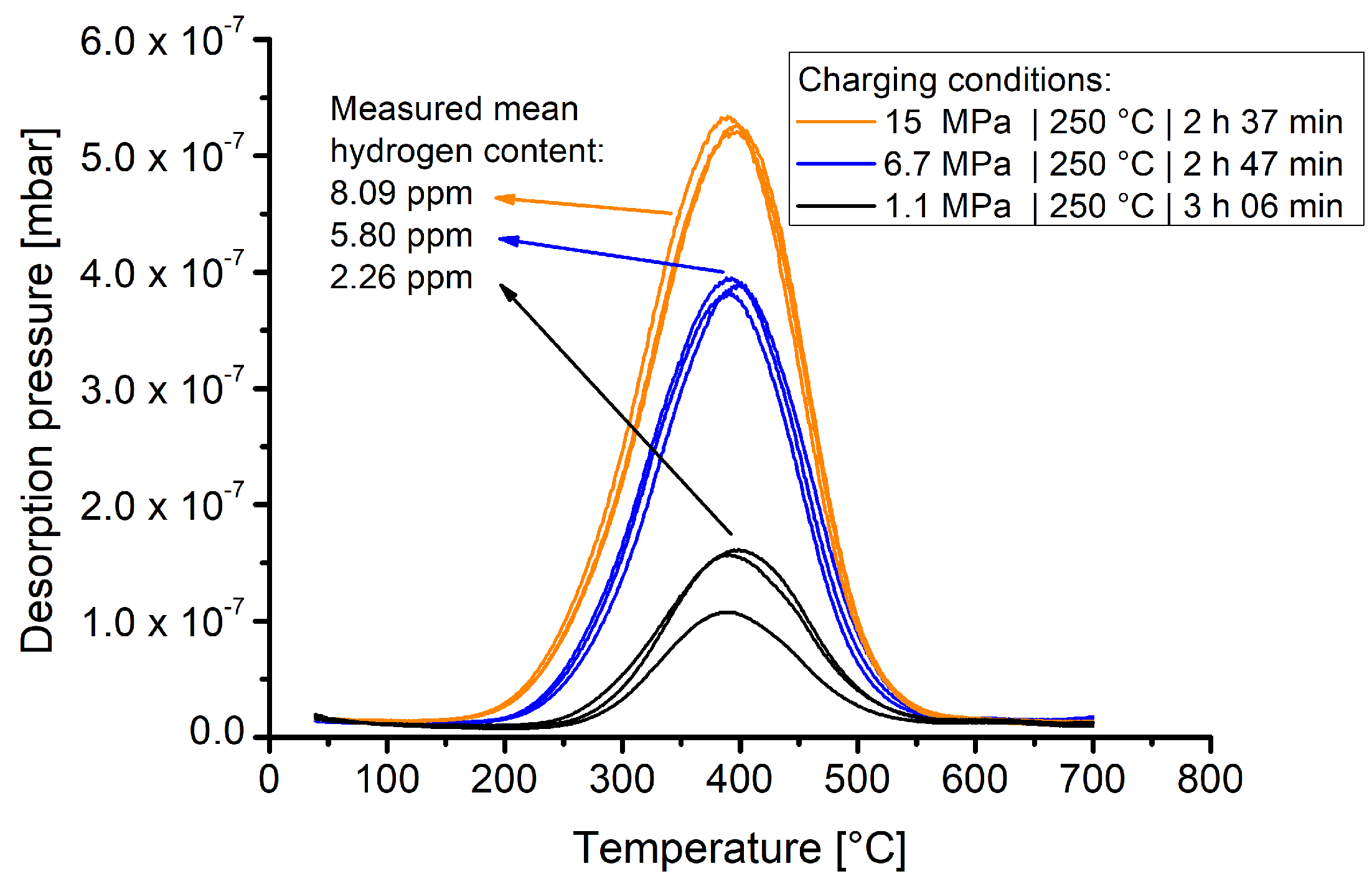
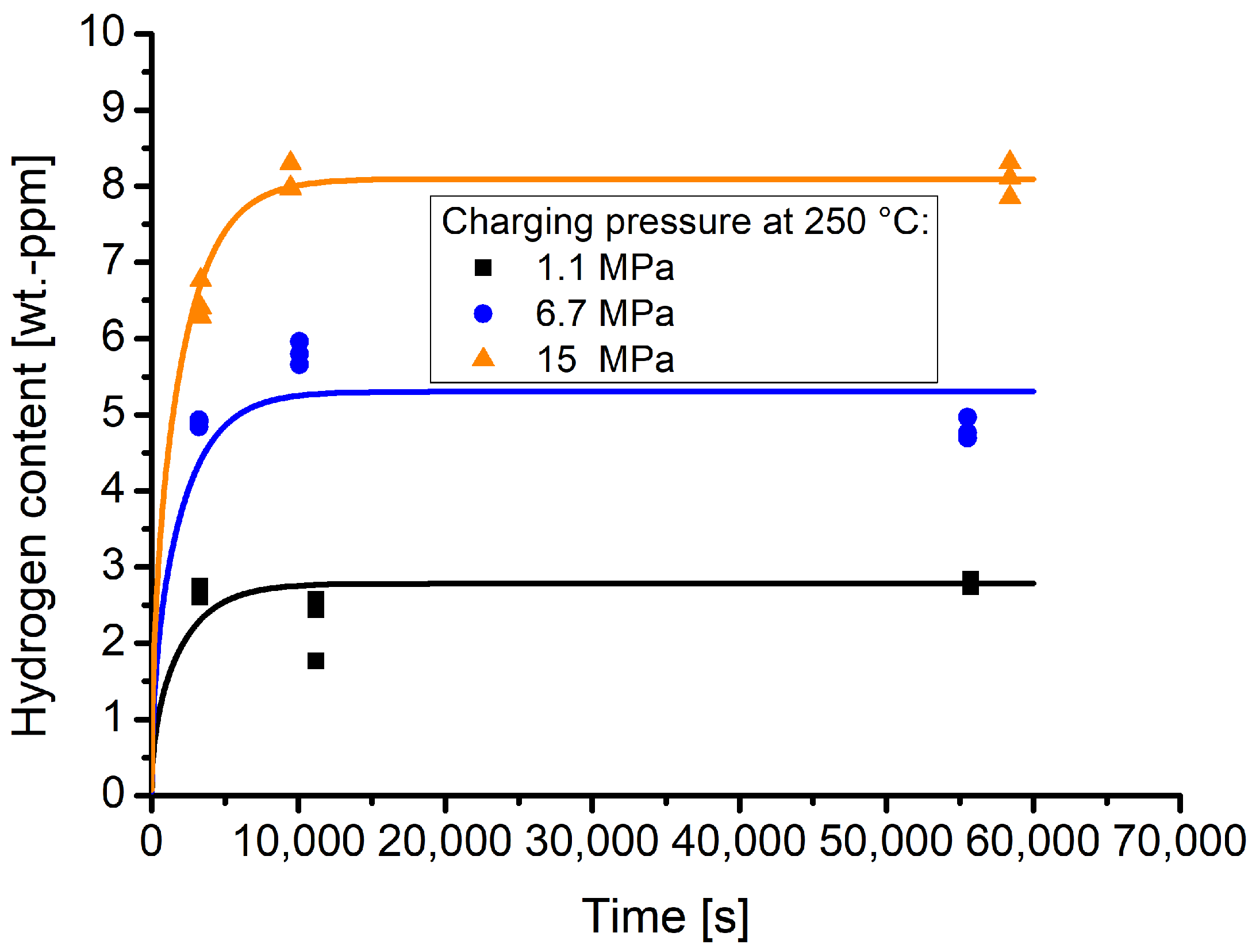
Figure 5 shows the TDS spectra of samples that were exposed for slightly more than 2.5 h to varying hydrogen pressures at 250 °C.
The results show that material 1.4418 exhibits a markedly pressure dependent hydrogen solubility. The pressure dependent hydrogen contents are clearly differentiable and the experimental curves of one batch show good reproducibility.
Figure 6 shows the time and pressure dependent hydrogen contents that were obtained by the TDS measurements. The lines indicate the respective sorption curves. The sorption behaviour is dependent on the hydrogen diffusion coefficient. The latter was determined using the experimental data points. Therefore, the pressure dependent measurements were normalised to the respective hydrogen equilibrium contents (see
Table 3), thus creating a kind of master curve. To obtain the hydrogen diffusion coefficient, a least squares fit was performed to fit Equation (
1) to the data.
where
is the hydrogen content at time
t,
is the equilibrium hydrogen content,
is the
nth zero of the Bessel function of 1 type and 0 order,
a is the radius and
D is the hydrogen diffusion coefficient [
19].
The hydrogen diffusion coefficient determined with this procedure is 1.64 × 10−10 m2/s.
It can be seen that, for the highest pressures of 15 MPa, the time dependent hydrogen contents are well along the sorption curve, while for the pressures 6.7 and 1.1 MPa deviations from the sorption curves are apparent. In the case of the values obtained for a pressure of 6.7 MPa and for a time of approximately 2.5 h, the measured hydrogen content is higher than that for longer holding times. In the case of 1.1 MPa, the hydrogen contents are lower at approximately 3 h holding than those obtained for approximately 1 h holding time. The deviations might be attributed to effects that arise due to the fact that the measurements were performed with certain time intervals in between. It cannot be completely rule out that the calibration constant of the TDS device experiences a drift. Furthermore, other effects that arise due to the storage within the cryo-container might also influence the hydrogen contents that are maintained within the samples. Nevertheless, due to the measured hydrogen contents at 250 °C with different pressures, the experiments are well suited to describe the hydrogen sorption behaviour of material 1.4418.
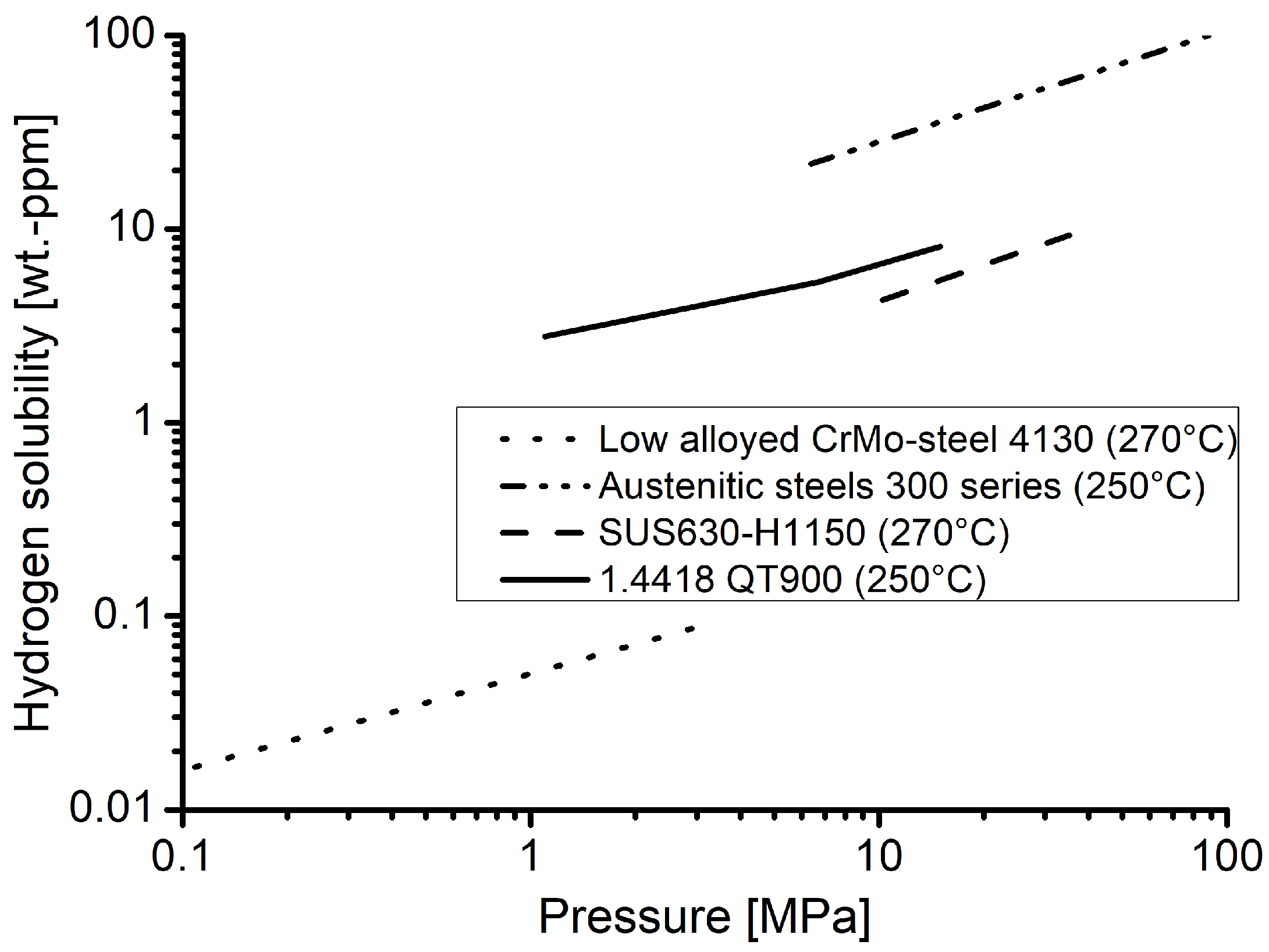
Furthermore, the hydrogen contents measured in material 1.4418 can be classified well with data for other materials from the literature [
20,
21], as displayed in
Figure 7. The values obtained for the nickel martensitic stainless steel are significantly higher than for a quenched and tempered low alloyed CrMo-steel, but still below values reported for austentic steels of the 300 series. The material SUS630 is a nickel martensitic precipitation hardened alloy. Due to its chemical composition, it can be assumed that the hydrogen content soluble in this material should be comparable to the one in material 1.4418 of the underlying investigation. As shown in
Figure 7, this is the case and the values are plausible in comparison to the other material classes as well as in agreement with expectations.
The values determined for the hydrogen diffusion coefficient can be classified similarly with regard to the data from literature.
Figure 8 shows the diffusion coefficients of material 1.4418 at room temperature [
10] and at 250 °C from the underlying investigation, together with scatterbands for austenitic as well as ferritic and martensitic steels [
12] and for selected alloys.
The steel 1.4418 exhibits a diffusion coefficient which is at the lower end of the scatterband for the ferritic/martensitic steels. This is true for the coefficient determined with an electrochemical double-cell as well as for the coefficient determined at 250 °C with gaseous hydrogen. In agreement with the hydrogen solubilities for the different alloy classes, the diffusion coefficient can also be classified to be between the ferritic/martensitic steels and the austenitic steels.
The investigated hydrogen solubilities and the calculated hydrogen diffusion furthermore are in support of the materials remarkable hydrogen compatibility in 1 MPa hydrogen atmosphere. This is at least in part explainable due to the fact that mechanisms describing hydrogen environment embrittlement as, e.g., the hydrogen-enhanced-localised-plasticity mechanism (HELP) [
4] are dependent on the materials hydrogen diffusivity.
The underlying paper presents a method to study the temperature and pressure dependent hydrogen solubility of materials. Advantageous are the uncomplicated handling of the autoclave set-up as well as its agile thermal behaviour including the possibility to rapidly load and unload samples. The short handling times reduce the hydrogen egress from samples that might occur when dealing with larger autoclave devices. Hydrogen egress might only occur during the cooling phase of the autoclave and shortly before the TDS measurements are started due to the fact that the samples need to be defrosted and a certain pressure needs to be achieved within the measurement apparatus.
It was further shown that the hydrogen content in material 1.4418 is high compared to low alloyed steels. In agreement with that, the hydrogen diffusivity is comparably low, thus supporting the good hydrogen compatibility under consideration of the relatively high material strength. Hydrogen compatibility can often be correlated with the diffusion and solubility behaviour of a material, due to the fact that mechanisms discussed in the literature, e.g., the HELP mechanism [
4], are dependent on the materials hydrogen diffusivity.
For material 1.4418, a clear pressure dependency of the hydrogen solubility was observed. The batch measurements showed a good reproducibility and the overall behaviour showed the suitability of the approach to study the sorption behaviour as well as the hydrogen equilibrium contents.
The equilibrium hydrogen contents as well as the evaluated diffusion coefficient can be classified well to data from the literature. In comparison to other material classes as well as to the material behaviour at room temperature, it has to be mentioned, however, that the calculation of the diffusion behaviour at 250 °C is based on the assumption that no surface reactions (e.g., adsorption, dissociation or recombination) are rate limiting but the only determining factor describing the sorption behaviour is the bulk diffusion of the material.
At lower temperatures, hydrogen reactions with the surface such adsorption, dissociation and absorption might become rate limiting, with a respective effect on the hydrogen sorption behaviour. These effects can be investigated by comparing to diffusion coefficients obtained by measurements at high temperature as well as by electrochemical measurements at lower temperatures.
The methodology allows studying the solubilities and diffusivities of materials with significantly reduced times where hydrogen egress might occur especially in comparison to commonly larger autoclave setups. Furthermore, the diffusion behaviour can be studied based on the sorption behaviour. Although surface effects cannot be separated, the temperature, pressure and time dependent sorption behaviour can be quantified, which is very valuable, e.g., when it comes to a hydrogen pre-conditioning prior to mechanical (static or dynamic) testing.