Abstract
To overcome the scarcity and resource limitations of Ti metal, deoxidation of Ti scrap was conducted through electrolytic refining and chemical reaction with MgCl2 molten salt electrolysis. The oxygen concentration in Ti scraps was decreased by the electrochemical and chemical reactions generated by the applied voltages. The optimized conditions for the process were derived by controlling the conditions and parameters by decreasing the thermodynamic activity of the reactants. The correlation between the deoxidation efficiency and the behavior of the voltage and current was confirmed by setting the conditions of the electrolysis process in various voltage ranges. In addition, the correlation between the presence of impurities and the measured oxygen concentration was evaluated. The surface element analysis result indicated that the salt that was not removed contained a certain amount of oxygen. Thus, the removal efficiencies of impurities and particles by deriving various post-treatment process conditions were analyzed. The results confirmed that the most stable and efficient current was formed at a specific higher voltage. Moreover, the best deoxidation result was 2425 ppm, which was 50% lower than that of the initial Ti scrap.
1. Introduction
Commercially available Ti metals used in the manufacturing of products have high purity [1,2]. Among Ti metals, TiCl4 is usually collected by refining raw materials such as rutile and ilmenite and converted to high-purity titanium, called Ti sponge, using the Kroll process [3,4,5,6]. However, as the outer shell of the reaction chamber is used, TiCl4 can be contaminated by iron and alloy elements, generating off-grade Ti sponge with high oxygen and iron contents [7,8].
Referring to the Ti-O phase diagram, Ti reacts strongly with oxygen at 1473 K in air and with pure oxygen at 883 K to form Ti dioxide [9]. In water and air atmospheres at room temperature, the reaction is less intense because a passive oxide coating is formed, which protects the bulk metal from further oxidation [10]. Oxygen from the internal structure of Ti scrap is considered a factor that can cause defects. Many studies have already analyzed various grades of Ti samples using nano-compression tests and examined the resulting impact using advanced transmission electron microscopy techniques and quantum mechanical predictions of defect structures [11,12,13]. The results indicated the presence of interactions between oxygen and dislocations, which are crystalline defects. In addition, oxygen atoms hindered the formation of corkscrew-shaped dislocations found in Ti. Therefore, synthetically, as the oxygen solubility in Ti increases, the Ti becomes more difficult to bend and susceptible to cracking, which hinders the manufacturing of Ti-based products. To overcome this issue, various studies have focused on the removal of oxygen dissolved in Ti [14,15,16]. The two most common processes used for Ti refining and deoxidation are called Ono–Suzuki (OS) and Fray–Farthing–Chen (FFC) Cambridge processes. The OS process is one of the representative process methods for smelting titanium metal. This method produces Ca as a reducing agent from CaO by applying electrolysis and injecting powdery TiO2 into a molten salt containing metallic calcium to produce titanium powder by thermal reduction [17]. In general, oxides are difficult to dissolve in chloride liquid, but CaO is unique, as it is dissolved by about 20 mol% in molten CaCl2 [18]. Electrolysis and reduction of TiO2 can also be performed in a separate cell. The FFC Cambridge process (Fray–Farthing–Chen) is a direct electrolysis method using TiO2 as a raw material. It utilizes the abilities of TiO2 to become a semiconductor at high temperature and to dissolve large amounts of CaO that form from CaCl2 [19,20]. TiO2 sintered in a CaCl2 salt bath is used as a cathode with carbon as an anode to reduce TiO2 and proceed with deoxidation to electrolytically collect titanium with low oxygen concentration. Electrons flowing to the cathode extract oxygen as ions, which react with the carbon anode and are released as gas molecules. On the other hand, in the FFC method, extra effort and cost are required to sinter the cathode, and there is also a problem with the material of the reaction vessel, since the produced titanium is sintered due to a slow reaction over a long time. Both processes are process methods developed for Ti smelting. However, these processes have various problems. First, in the case of the FCC process, additional time and cost are incurred for precise plastic processing of the anode, and titanium is exposed to a long reaction, which causes a problem in cleaning the molten salt [21]. In addition, in the case of the OS process, strictly speaking, it has a mechanism closer to the calcium thermal reduction method than electrolysis. In addition, it is difficult to separate Ti powder produced from a large amount of molten salt containing a reducing agent. In comparison with this, in the case of molten salt electrolytic deoxidation, which consists of a smelting target as an electrode and proceeds with a difference in reactivity with the oxygen of the reducing agent, there is a wide range of process costs depending on the type of reducing agent, and it has the advantage of being usable for a long period [22]. Ca has a higher oxygen capacity than the Mg-reducing agent. It has been reported that only approximately 0.006 wt% of oxygen dissolves in Ti at 1000 °C, and the deoxidation limit in Ti in equilibrium with Ca-CaO is approximately 0.05 wt%. However, the cost of Ca is higher compared to Mg, and complete removal of the reaction product CaO is difficult [23,24]. In addition, calcium metal has chemical characteristics that cause it to explode when reacting excessively with moisture in the air. Most of the smelting processes are carried out in a high-vacuum argon or hydrogen atmosphere, but a difficult part is controlling the calcium metal reduced by a separate process, such as a target metal recovery and washing process [25,26,27,28]. Thus, an efficient Ti electrolytic deoxidation process using MgCl2 electrolyte was attempted, and the optimal efficiency was derived through various process conditions and variable control.
Theoretically, MgCl2 has a weaker deoxidation effect than CaCl2; thus, its deoxidation effect was maximized by increasing the reactivity of O and Mg dissolved in Ti by adding Mg metal, which is related to the MgO activity. The deoxidation process using MgCl2 molten salt is closely related to the activity of the MgO product produced by the chemical reaction of Mg and O, as follows [29]:
where O is the oxygen dissolved in Ti, R is the gas constant, T is the temperature, is the partial pressure of oxygen, is the Henrian activity coefficient of oxygen dissolved in Ti, and α-Mg and α-MgO are the activity coefficients of Mg and MgO, respectively. As shown in Equations (1)–(3), the mass percentage of oxygen dissolved in Ti metals decreases with the activity coefficient of MgO. Regarding the relationship between the activity of the reactant and oxygen, Okabe et al. proposed an Ellingham diagram of the oxygen concentration in Ti depending on the level of MgO activity [30,31]. Considering this diagram, when the activity of the MgO reaction product is reduced to 10−3, the oxygen concentration that Ti can contain is reduced because of a high Gibbs free energy value of up to several tens of parts per million. Therefore, the deoxidation efficiency can be increased by decreasing MgO activity. It is very difficult to create an environment with MgO as low as that reported in the literature. However, if the degree of electrochemical decomposition of continuously generated MgO is increased by inducing a smooth reaction between Mg and O, the deoxidation efficiency can be increased by lowering the MgO activity to a certain level. Thus, in this study, the deoxidation efficacy was experimentally verified by removing the O from Ti while reducing the MgO formed by the electrode reaction, and the conditions for the optimum removal efficiency of the molten salt after the process were derived.
2. Materials and Methods
2.1. Experimental Preparation
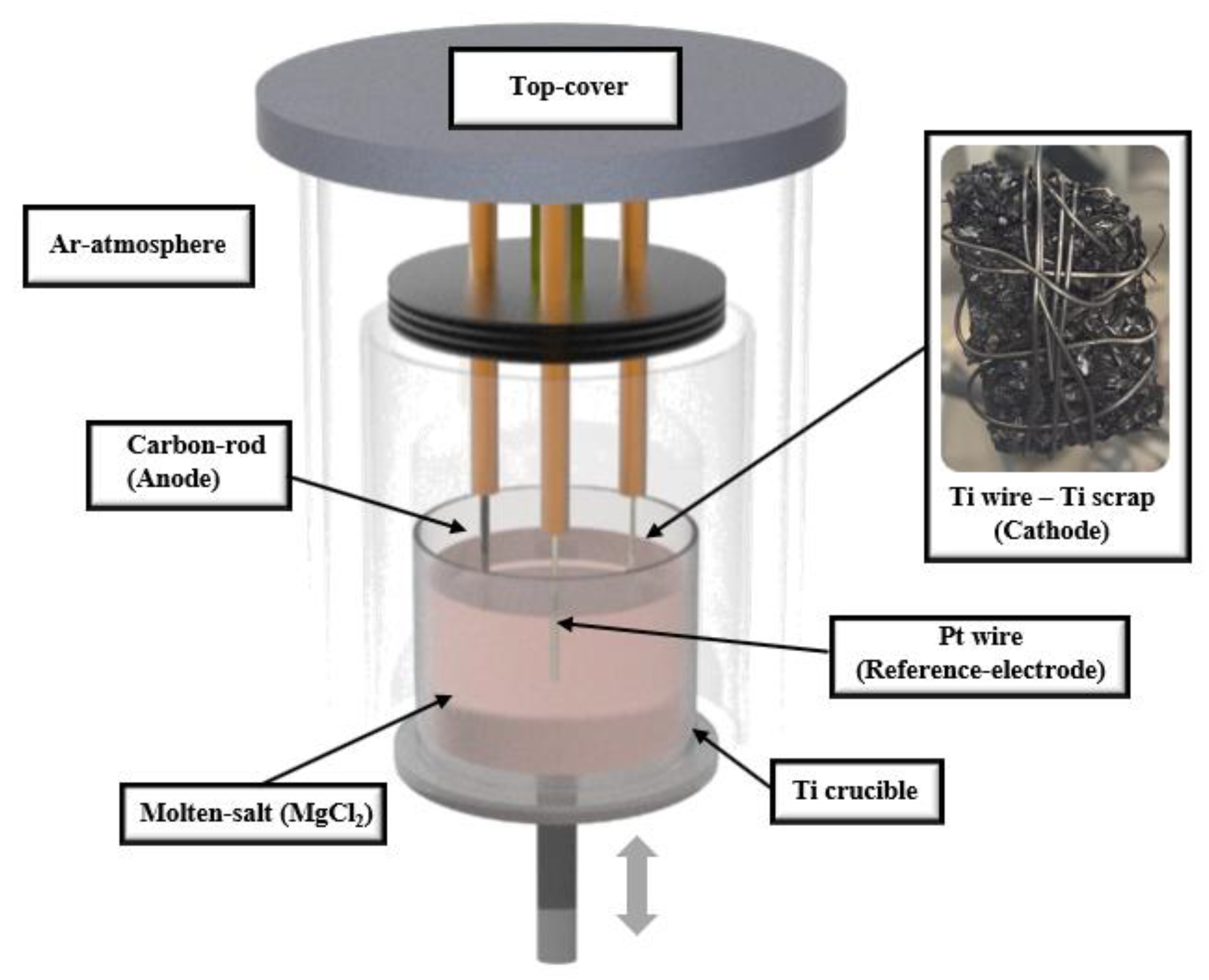
High-purity Ar gas was inserted into a glove box containing a lift furnace, as shown in Figure 1. Process materials were selected by considering the thermodynamic simulations from FactSage software to avoid unnecessary reactions of molten salt and materials on the equipment. A total of 4.5 kg of MgCl2 (99.9%, Kojundo Chemical Laboratory Co., Sakado, Japan) was placed in a crucible made of Ti, and the temperature was increased to 900 °C and maintained until the MgCl2 was completely melted. Ti wire (⌀ 10 mm × 200 mm), C rod (⌀ 15 mm × 220 mm), and Pt wire (⌀ 6 mm × 200 mm) were used as working, counter, and reference electrodes, respectively, which are non-reactive with the electrolyte and have chemical and electrochemical stability using thermodynamic software. To proceed with the deoxidation process, certain ranges of different voltages were applied to the cathode electrode, which was set up with target brittle Ti scraps that were tied to the Ti wire. After deoxidation treatment, the Ti scrap was washed with 1 M citric acid, 0.5–8 M HCl solution, and 12 M H2SO4 diluted solution to remove the MgO and residual chlorides by stirring on a magnetic stirrer at 150 rpm for 30 min, and the washed Ti scrap was then dried in a vacuum atmosphere.
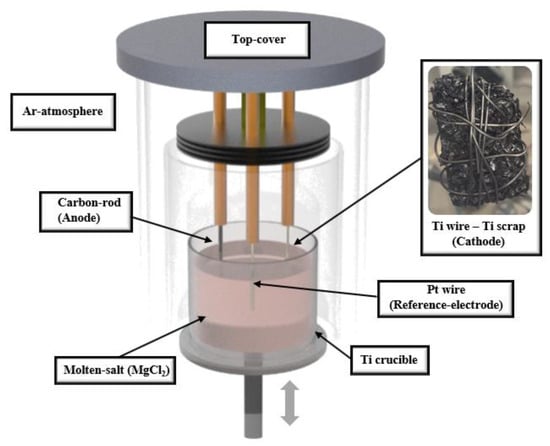
Figure 1.
Schematic diagram of device structure for electrolytic deoxidation process.
2.2. Characterization
Field-emission scanning electron microscopy (FE-SEM) and energy-dispersive X-ray spectroscopy (EDS) analyses were conducted to confirm the morphology and surface components of the scraps. To identify the observed impurities and specific surface states, X-ray diffraction analysis (XRD) and X-ray photoelectron spectroscopy (XPS) were conducted. To measure the oxygen content of titanium scrap for each process step, oxygen, nitrogen, and hydrogen contents were measured by using NOH 2000 (ELTRA Co., Haan, Germany) equipment. For the electrochemical analysis, cyclic voltammetry (CV) was performed before the voltage was applied to observe the electrochemical behavior of the electrolyte and added elements. Furthermore, to confirm the current formation during the deoxidation process, constant voltage analysis by chronoamperometry (CA) was performed.
3. Results and Discussion
3.1. Cyclic Voltammetry Results
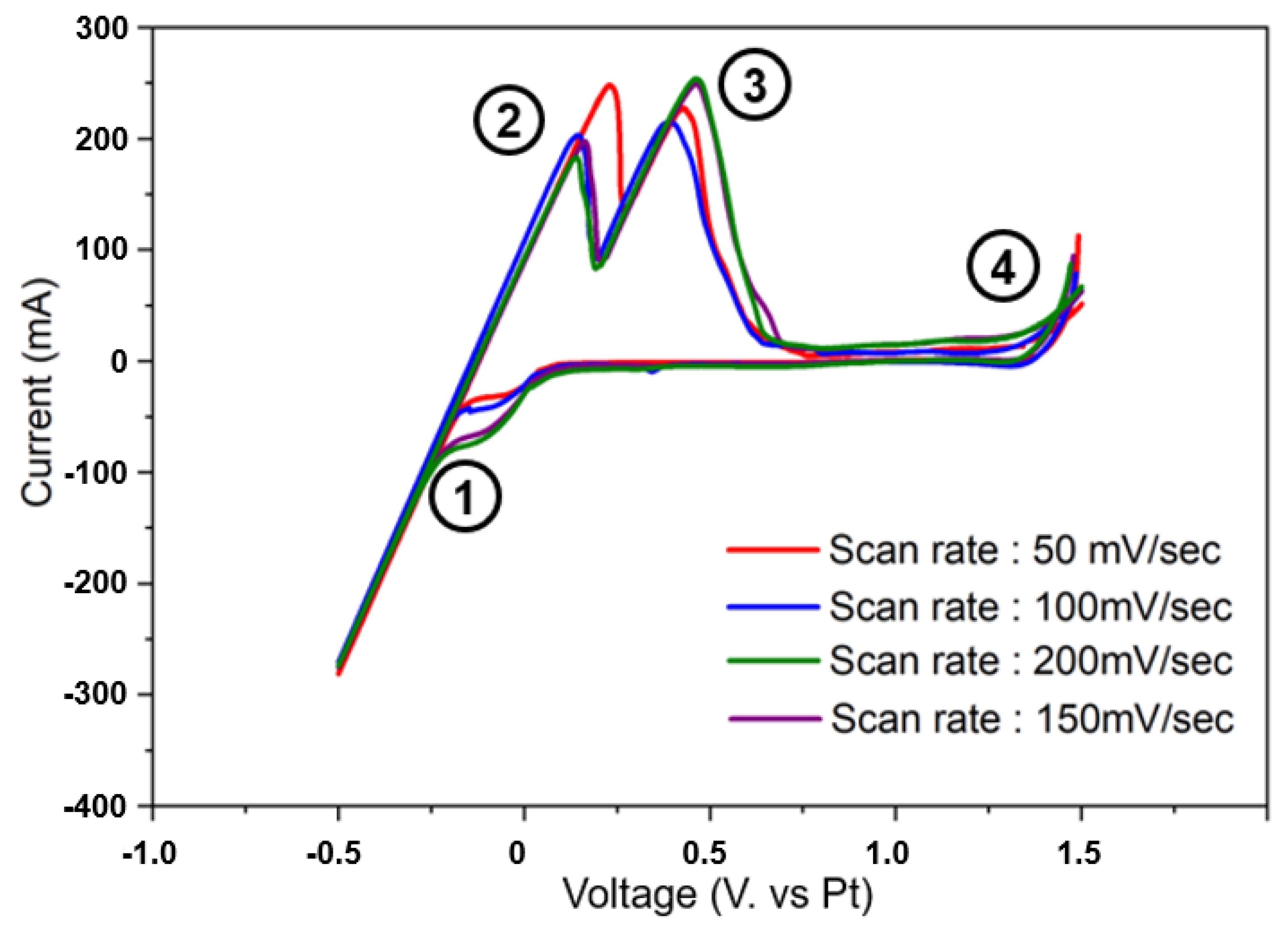
The Ti deoxidation reaction occurs by the following mechanism. Oxygen present in the titanium scrap cathode reacts with Mg ions in MgCl2 molten salt flux to form MgO, thereby removing oxygen from Ti. At this time, since a voltage is applied, the formed MgO that cannot be remelted due to limited MgO dissolution with molten MgCl2 flux is decomposed into Mg and oxygen ions by electrolysis. The divided Mg can participate in the deoxidation reaction again. Therefore, before applying a voltage to initiate the deoxidation process, CV analysis was performed to measure the appropriate cell potential between the electrodes and to confirm the behavior of electrolyte ions. CV analysis was conducted at a voltage range of 1.5 V to −0.5 V at 1103 K with a set of four scan rate ranges, as shown in Figure 2. As shown in Figure 2, some intensities indicate redox reactions of Mg, MgO, O, and Cl−. In the range of −0.5 to 0.0 V, in the CV curves marked numbers 1 and 2, various intensities indicate the reduction of the added Mg compounds, and in the range of −0.5 to 0.0 V, the CV curve marked number 1 suggests the formation of current by Mg ion reduction. In addition, according to the reference, the time point at which Mg is decomposed occurs in the −0.25 section, where the peak falls dramatically. As the voltage increases to −0.5 V, in this part, it can be seen that the Mg ions generated by dissolution in the MgCl2 salt flux are decomposed by excessive voltage, and the oxidation peaks occur at 0.0 V for Mg+ ions and at 0.5 V for oxygen, as shown by the curves marked numbers 3 and 4. As the chloride is decomposed from 1.5 V, as confirmed in the curve marked number 5, the optimized voltage potential can be considered to be in the range of −1.0 to 1.5 V. Despite the voltage potential obtained from the CV analysis, the theoretical conditions for applying voltages were considered over ranges of measured potential due to attempts to form high currents. Because the Mg ions are reduced by the applied voltages at the cathode electrode, higher currents were expected to result in more efficient deoxidation. The resulting O species, present mainly as O2− dissolved in molten MgCl2, reacted at the C anode to form COx gas, which was removed from the molten salt system.
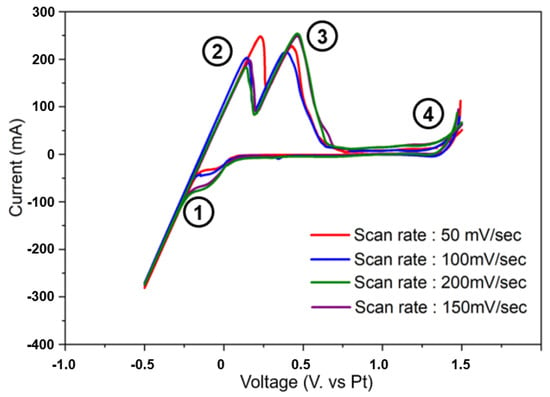
Figure 2.
Cyclic voltammetry analysis results for cell potential measurement according to redox reaction behavior analysis of electrolyte ions of constructed electrodes.
3.2. Chronoamperometry and Cell Potential
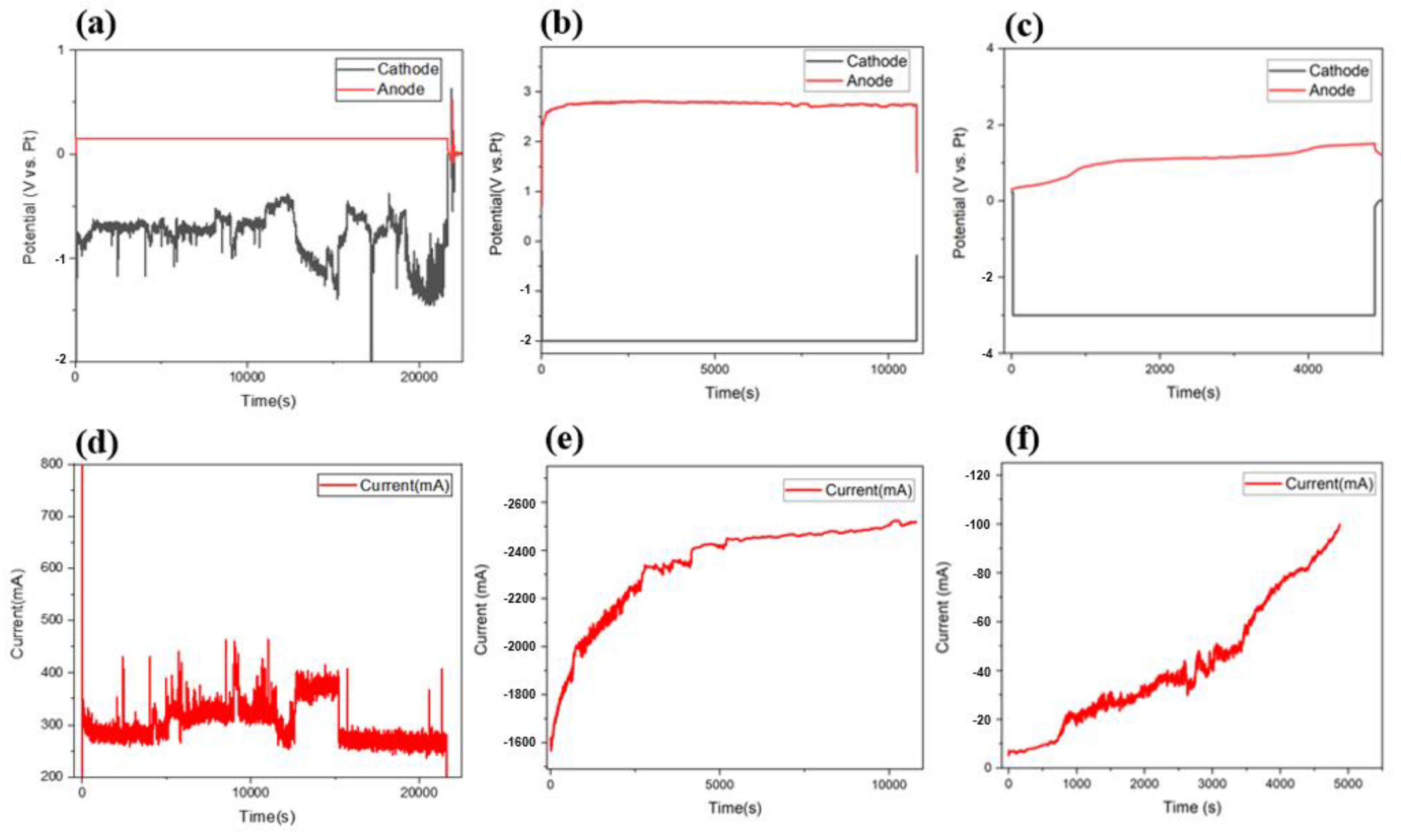
The behavioral changes in current and voltage between the Ti scrap cathode and C rod anode were analyzed by CA under potentiostat conditions, and the results are shown in Figure 3a–f. By applying −2 V and −3 V, different currents were formed compared to that when applying −0.2 V, and the results were in agreement with the voltage potential measured in the CV analysis. In the sample of the −0.2 V condition (Figure 3a,d), the currents measured were in the range of 200–500 mA, and the cell potential was 1.4 V during the deoxidation process. The potential of the counter electrode changed while the potential of the other electrode remained constant, and the current formation changed in proportion to the changing potential. As the process continued, peaks consistently appeared, indicating that the deoxidation process continuously occurred. The formation of a direct current to the MgCl2 flux is affected by several parameters, such as resistance from the electrode structure, specific resistance value according to electrolyte composition, and unidentified impurities in flux. The most significant parameter was the magnitude of the applied voltage. It is well known that, according to Ohm’s law, voltage is determined by current and resistance, and therefore, current is largely formed by large voltage and low resistance. As mentioned, the principle of directly removing oxygen in this process is based on the formation of oxides by chemical reactions between reduced Mg ions and oxygen inside Ti. Subsequently, the formed MgO was partially remelted to Mg2+ ions at a concentration of 2.9 mol %. In this part, to maximize the MgO redissolution process and prevent excessive vaporization of MgCl2, the process was performed at a temperature higher than the MgCl2 melting point. The MgO that did not redissolve was decomposed into Mg and O2− by electrolysis, and the formed Mg and Mg ions participated in the deoxidation process. The ionized oxygen atoms reacted with the carbon rod of the anode, after which they evaporated into the gas component of COx and were finally removed.
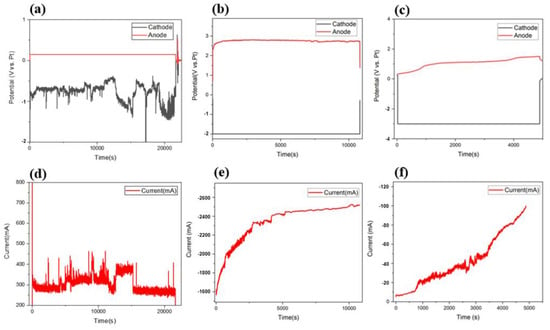
Figure 3.
CA analysis results for formed electrode potential and currents during the deoxidation process under different applied voltage conditions. −0.2 V (a,d); −2 V (b,e); −3 V (c,f).
Therefore, the electrochemical reaction between the additives and the process materials usually occurs more efficiently when the density and degree of the current formed during the electrochemically equivalent process are higher. Therefore, a higher current may form when a high voltage is applied because the applied voltage is the only directly controllable variable among the various variables mentioned. However, if the applied voltage exceeds the voltage limit that can be accommodated by the structured electrode device, the process may not proceed, as shown in Figure 3f. Therefore, by finding an appropriate voltage range in various process voltage ranges according to the current formation law, it was confirmed that the high current formation efficiency observed in the deoxidation process was obtained under a relatively high-voltage condition. Figure 3b,e show the shape of the current formed when a voltage of −2 V was applied to the cathode. For a total process time of 3 h, a current of approximately −1600 mA was initially formed, and the current value increased over time. In addition, it was confirmed that a current of −2500 mA continuously formed after approximately 80 min. Thus, the current formed was higher than that formed for a voltage of −0.2 V, and an average current of 2.34 A formed after approximately 3 h. In addition, the voltage of the relatively formed anode of the cathode to which the voltage was applied during the process was measured to be 2.81–2.69 V, and the total cell potential under this condition was measured to be a maximum of 4.81 V. Figure 3c,f, show the behavior of the formed current when a higher voltage of −3 V was applied to the cathode. As the process progressed, the current continuously formed and increased; the voltage application was interrupted at 85 min, after reaching the limit value. The range of the formed current was also confirmed to be lower than those for the conditions of −0.2 V and −2 V, and voltage cut-off due to overvoltage occurred. Thus, it can be concluded that this condition exceeded the voltage range that could be accommodated by the configured deoxidation electrode device. Therefore, it can be said that the optimum voltage condition for the electrolytic deoxidation process is within 1.5–2.0 V, considering the potential of 1.5 V measured in the CV analysis and the current behavior results confirmed in the CA analysis. The behavior of the current formation according to the applied voltage range was confirmed, and the resulting deoxidation process efficiency was confirmed through a component analysis of the Ti scrap.
3.3. Deoxidation Efficiency
The Ti scrap before the deoxidation process contained an average concentration of 5580 ppm of oxygen, 1982 ppm of nitrogen, and 14,200 ppm of carbon.
In the deoxidation process conducted by applying a voltage of −0.2 V, a maximum oxygen reduction efficiency of 2726 ppm was observed, but most of the other oxygen measurements were higher than that, so the final average oxygen content was measured to be 4298 ppm, which is approximately 22.9% lower compared to the initial concentration, as shown in Table 1. Furthermore, after repeating the process several times, the average result was over 4500 ppm under this voltage condition. Compared with the CA analysis based on this result, in the case of the highest deoxidation efficiency, for example, at −2 V, the anode voltage changed significantly over a certain period, and the resulting current formation changed significantly compared to other conditions. Therefore, it was confirmed that the deoxidation efficiency was maximized when the current was high, whereas the deoxidation efficiency was low when the change in current was negligible or when the current was low. Therefore, in the case of Ti scrap deoxidation conducted under the −2 V condition, an average of 2674 ppm of oxygen was measured, which indicates the removal of nearly 51% of oxygen compared to the initial concentration. In addition, in the case of Ti scrap with an applied voltage of −3 V, the average was 2782 ppm, which is 50% lower than the initial concentration. The Ti scrap under the −3 V condition showed lower current efficiency than that under the −2 V condition during the process, but the deoxidation efficiency was similar. In addition, the deoxidation efficiency was satisfactory, although a lower current was formed compared to that when a low voltage was applied. This is considered to be because the current continuously increased over time despite the occurrence of a low-current shape due to overvoltage.

Table 1.
Component analysis of Ti scrap after being subjected to deoxidation under various conditions.
3.4. Removal Process of Residual Salts
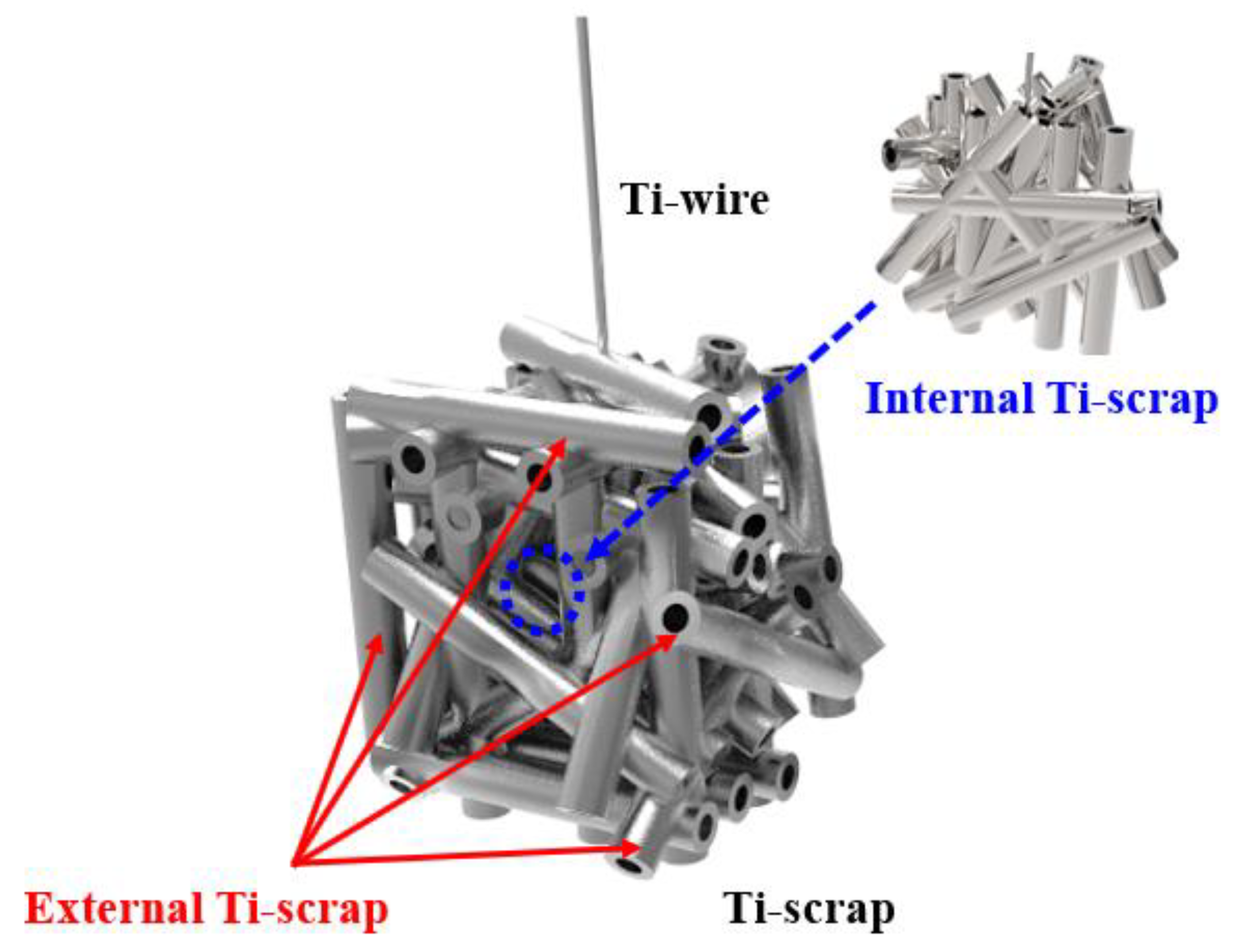
All Ti scraps were subjected to a separate pulverization operation after the deoxidation process to separate the scrap existing inside and outside the green compact, as shown in Figure 4. Then, component analysis was performed.
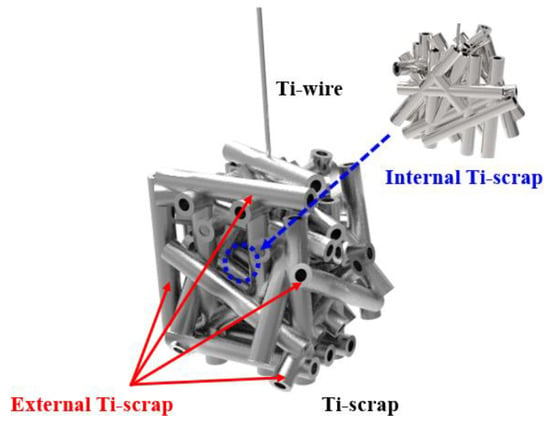
Figure 4.
Method of separation for deoxidated Ti scrap analysis.
The internal Ti scrap showed a higher deoxidation efficiency than the external Ti scrap. This result could be due to various factors. When the Ti scrap is separated from the molten salt electrolyte after the deoxidation process, a negligible amount can be instantaneously oxidized by oxygen in the equipment. Furthermore, the MgO formed by reaction with oxygen and impurities, such as iron and aluminum, in the scrap manufacturing process may be dissolved and remain in the solid solution, which was confirmed by EDS surface mapping analysis, as shown in Figure 5. More precisely, it was confirmed that many unidentified impurity particles and salts existed on the surface, and more impurities were present in the case of the external Ti scrap. In the analysis using EDS mapping and spectral points, it was confirmed that the impurity particles containing Mg, O, Fe, and Cl also contained a significant amount of oxygen.
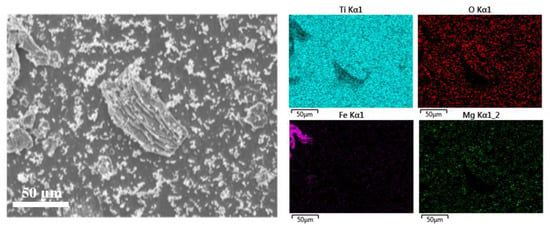
Figure 5.
Surface analysis result of external Ti scrap using FE-SEM and EDS mapping analysis.
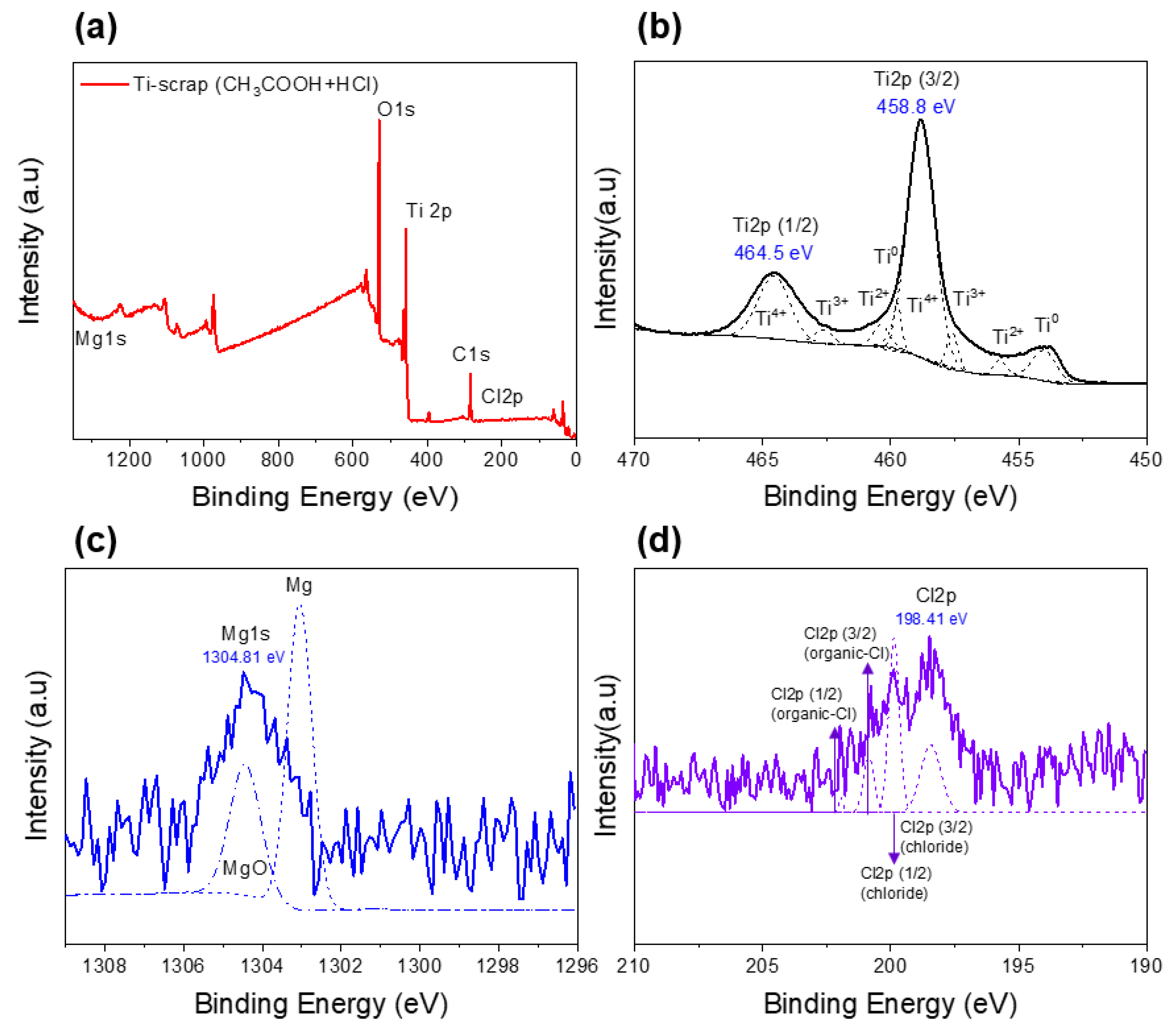
Therefore, for more detailed analysis, X-ray photoelectron spectroscopy and X-ray diffraction analysis were performed. Figure 6a shows the full surface XPS spectra of the Ti scrap that proceeded with the conventional process, and spectra of the designated elements states are observed in Figure 6b–d. A high level of oxygen content was confirmed in the sample because titanium naturally forms a thin oxide layer on the surface as a passivation film to prevent oxidation [32].
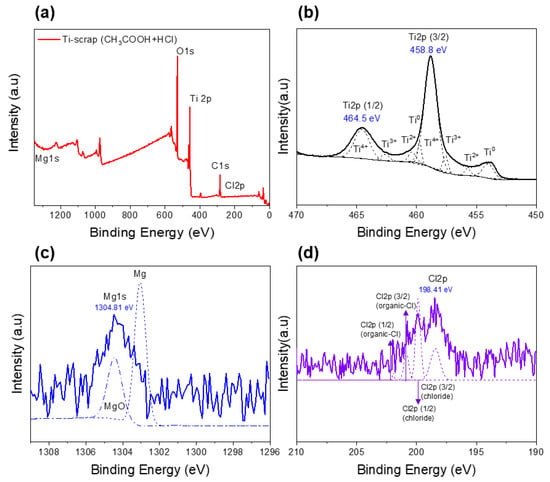
Figure 6.
XPS results of Ti scrap with deoxidation and conventional washing: full spectrum (a) and designated elements (b–d).
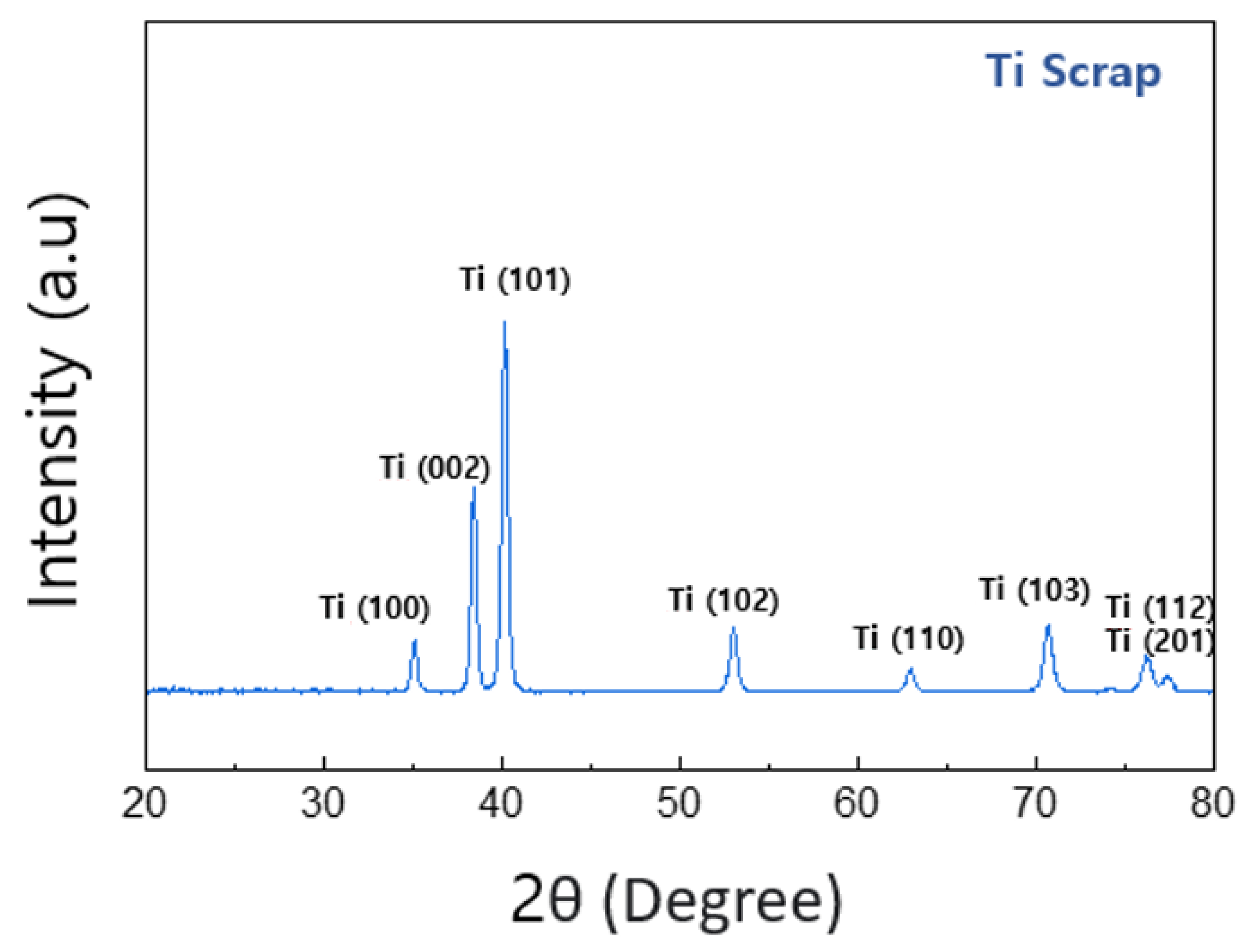
Thus, in the spectral results of the Ti element, the Ti2p (3/2) point exhibits a binding energy of about 458.8 eV, showing the characteristic of titanium oxide rather than pure Ti. XRD analysis was performed for more detailed and clearer identification, and the results confirmed that this Ti scrap had a clear titanium phase rather than titanium oxide, as shown in Figure 7. This is considered to be the result of the XPS characteristics due to the formation of the aforementioned titanium film and the penetration of relatively thin electrons compared with XRD analysis [33]. Subsequently, it was confirmed by the results in Figure 6c that this sample contained a small amount of MgO, as expected from the EDS analysis. The binding energy of Mg1s was confirmed to be Mg oxide (1304.5 eV) rather than Mg metal (1303 eV) [34], which indicates that the impurity particles, which were observed to contain both O and Mg elements in EDS mapping analysis, are more likely to be MgO.
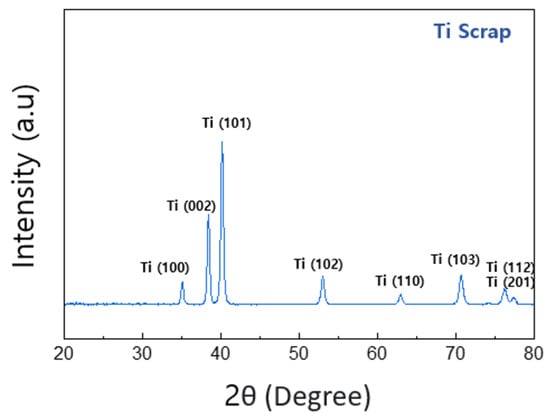
Figure 7.
Result of XRD of the deoxidated Ti scrap.
Therefore, in the remaining MgO, if the impurity element and the formed reaction products such as MgCl2 and MgO are removed through a subsequent leaching process, the dissolved oxygen will be removed as well. There are many related studies in the literature on the leaching process operation for the removal of MgO and MgCl2 [35,36,37,38]. The most representative solution is an acidic solution such as HCl, H2SO4, and HNO3, as it does not react with the Ti metal while reacting with Mg-based compounds and other impurities according to the following equations:
Therefore, we attempted to confirm the impurity removal efficiency using the four types of solvents mentioned above by performing FE-SEM analysis, and the results are shown in Figure 8.

Figure 8.
Subsequent treatment results from various solutions to remove impurities remaining on the Ti surface.
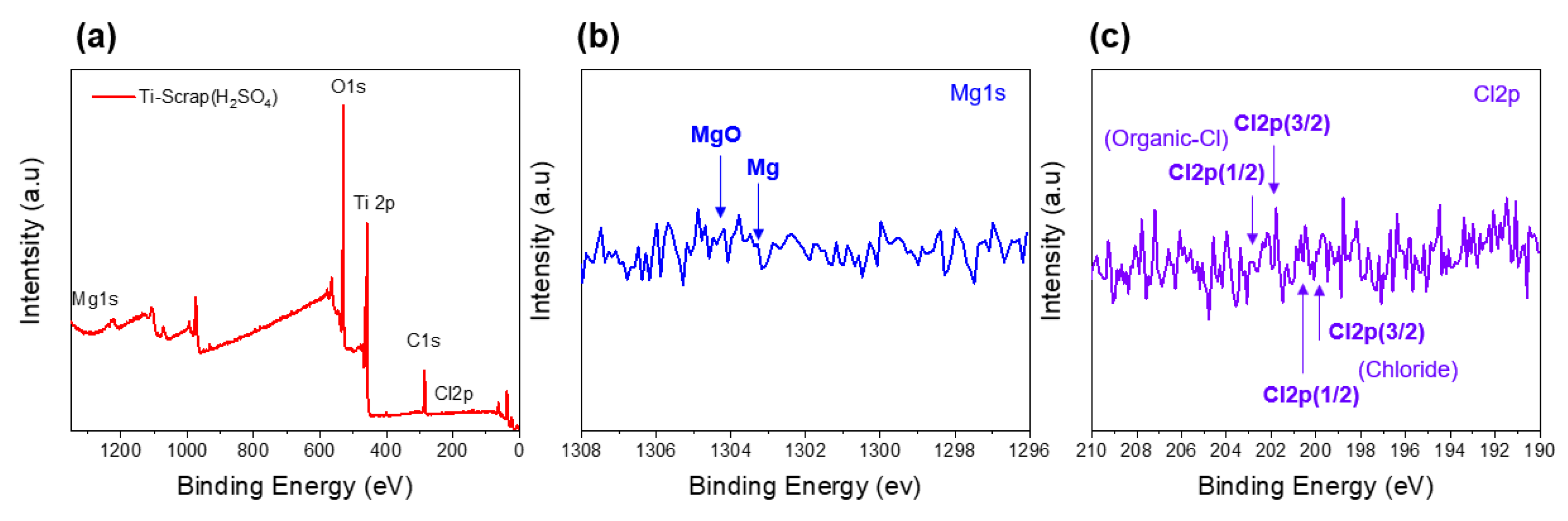
In the removal of the generated MgO and other impurities, it is well known that increasing the temperature during the reaction process further increases the efficiency. However, because the oxygen solubility of Ti metal also changes with temperature, the processes were conducted at 25 °C. The remainder of the Ti scrap that proceeded with other solutions had cleaner surface results compared to that of the citric acid sample. Among them, H2SO4 exhibited the most impressive removal efficiency for both internal and external Ti scrap, reaching a Ti atomic concentration of 96.25%, whereas the other samples reached 71.25–86.00% in general. The results of this improved cleaning process are also shown in the XPS results. By comparing Figure 9 with Figure 6, the disappearance of the binding energy peaks that represented MgO and Cl indicates that an efficient cleaning process was carried out with the H2SO4 solution.
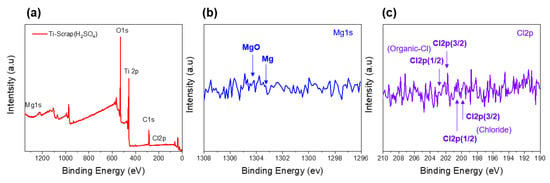
Figure 9.
XPS analysis of Ti scrap by proceeding with H2SO4 solution of full spectrum (a), magnesium (b), and chlorine (c).
The relationship between the final oxygen concentration and the observed impurities was investigated by analyzing and comparing the oxygen and nitrogen concentrations. The oxygen content obtained using NO/C analysis is shown in Table 2 for the various solution conditions used.

Table 2.
Ti scrap component analysis after being subjected to deoxidation with various leaching solutions.
Overall, it was confirmed that the oxygen content in the H2SO4 condition, which showed the highest removal efficiency in the surface analysis after the impurity removal process, was the lowest. As another feature, the measured oxygen content range showed a high error range in the previous samples; nevertheless, a homogeneous oxygen content result with a low error range was derived for a sample with high impurity removal. As shown in Figure 10, the final Ti scrap that underwent deoxidation and leaching processes is cleaner and has a metallic color compared to the previous one. Despite the results of the impurity removal process, the scrap of the part existing outside the compressed Ti scrap still showed lower deoxidation efficiency than that of the internal scrap. This result indicates that the impurity particles contained a certain amount of oxygen; however, the difference in the deoxidation efficiency of the external and internal scraps was not the main reason. Therefore, a small amount of oxygen in the Ar atmosphere or oxygen dissolved in Ti could have had an effect during the removal from the molten salt immediately after the process [39]. As a result, it is presumed that the outer part of the Ti scrap, which was more exposed to high temperature and external atmosphere, absorbed and reacted with the residual oxygen again.

Figure 10.
(a) Ti scrap before the electrolytic deoxidation process; (b) external and (c) internal Ti scraps after the process.
4. Conclusions
Using the MgCl2 molten salt electrolysis deoxidation process, the concentration of oxygen in Ti scrap, which was initially as high as 5000 ppm, decreased by 50%. During the process, CV analysis was performed to observe the chemical behavior of electrolytes and elements under the set conditions, and CA analysis was performed simultaneously with voltage application to investigate the correlation of current formation according to the applied voltage range. In addition, the presence of impurities was confirmed by conducting surface and component analysis immediately after the deoxidation process, and the relationship between the efficiency and oxygen content according to the removal process with different types of solutions was confirmed. As a result, as the voltage was strongly applied, the formed current increased, and the deoxidation efficiency was increased by the strong electrochemical reaction generated. Moreover, the oxygen content was determined to be constant and low in the efficient impurity removal process using H2SO4 solution.
Author Contributions
N.K.: investigation, data curation, writing—original draft, validation. J.-S.B.: Investigation. H.C.K.: investigation, data curation. S.G.H.: investigation. S.J.O.: writing—review and editing. S.-h.C.: investigation, data curation, writing—review and editing, S.-J.S.: resources, supervision, project administration, writing—review and editing. K.-T.P.: resources, supervision, project administration, writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data available on request.
Acknowledgments
This work was supported by the Technology Innovation Program (or Industrial Strategic technology development program: 20010047) funded By the Ministry of Trade, industry & Energy (MI, Korea), and also supported by the Technology Innovation Program (or Industrial Strategic technology development program: 20010585) funded By the Ministry of Trade, industry & Energy (MI, Korea).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kaur, M.; Singh, K. Review on titanium and titanium based alloys as biomaterials for orthopaedic applications. Mater. Sci. Eng. C 2019, 102, 844–862. [Google Scholar] [CrossRef]
- Sai, P.V.; Madhukar, S.; Reddy, M.K.P.; Rao, N.R. Review on Machinability Aspects of Titanium Grade-2. Int. J. Sci. Eng. 2017, 2, 270–275. [Google Scholar]
- Gerdemann, S.J. Titanium process technologies. Adv. Mater. Process. 2001, 159, 41. [Google Scholar]
- Wartman, F.S.; Baker, D.H.; Nettle, J.R.; Homme, V.E. Some observations on the Kroll process for titanium. J. Electrochem. Soc. 1954, 101, 507. [Google Scholar] [CrossRef]
- Sohn, H.S. Production Technology of Titanium by Kroll Process. J. Korea Org. Resour. 2020, 29, 3–14. [Google Scholar]
- Nakamura, K.; Iida, T.; Nakamura, N.; Araike, T. Titanium sponge production method by Kroll process at OTC. Mater. Trans. 2017, 58, 319–321. [Google Scholar] [CrossRef] [Green Version]
- Zhuo, S.; Kaihua, L.; Liang, L.; Xiaozhe, C. Inflow process of Fe, Ni, and Cr impurities in Ti sponge during kroll process. Metall. Res. Technol. 2020, 117, 101. [Google Scholar] [CrossRef]
- Zheng, H.; Ito, H.; Okabe, T.H. Production of titanium powder by the calciothermic reduction of titanium concentrates or ore using the preform reduction process. Mater. Trans. 2007, 48, 2244. [Google Scholar] [CrossRef] [Green Version]
- Uwanyuze, S.; Kanyo, J.; Myrick, S.; Schafföner, S. A review on alpha case formation and modeling of mass transfer during investment casting of titanium alloys. J. Alloys Compd. 2021, 865, 158558. [Google Scholar] [CrossRef]
- Yan, M.; Xu, W.; Dargusch, M.S.; Tang, H.P.; Brandt, M.; Qian, M. Review of effect of oxygen on room temperature ductility of titanium and titanium alloys. Power Metall. 2014, 57, 251–257. [Google Scholar] [CrossRef]
- Nayak, S.K.; Hung, C.J.; Sharma, V.; Alpay, S.P.; Dongare, A.M.; Brindley, W.J.; Hebert, R.J. Insight into point defects and impurities in titanium from first principles. NPJ Comput. Mater. 2018, 4, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Pan, X.; Yang, M.Q.; Fu, X.; Zhang, N.; Xu, Y.J. Defective TiO2 with oxygen vacancies: Synthesis, properties and photocatalytic applications. Nanoscale 2013, 5, 3601–3614. [Google Scholar] [CrossRef]
- Taninouchi, Y.K.; Hamanaka, Y.; Okabe, T.H. Electrochemical deoxidation of titanium and its alloy using molten magnesium chloride. Metall. Mater. Trans. B 2016, 47, 3394–3404. [Google Scholar] [CrossRef]
- Okabe, T.H.; Nakamura, M.; Oishi, T.; Ono, K. Electrochemical deoxidation of titanium. Metall. Mater. Trans. B 1993, 24, 449–455. [Google Scholar] [CrossRef]
- Su, Y.; Wang, L.; Luo, L.; Jiang, X.; Guo, J.; Fu, H. Deoxidation of titanium alloy using hydrogen. Int. J. Hydrogen Energy 2009, 34, 8958–8963. [Google Scholar] [CrossRef]
- Zheng, C.; Ouchi, T.; Iizuka, A.; Taninouchi, Y.K.; Okabe, T.H. Deoxidation of Titanium Using Mg as Deoxidant in MgCl2-YCl3 Flux. Metall. Mater. Trans. B 2019, 50, 622–631. [Google Scholar] [CrossRef]
- Suzuki, R.O.; Natsui, S.; Kikuchi, T. OS process: Calciothermic reduction of TiO2 via CaO electrolysis in molten CaCl2. In Extractive Metallurgy of Titanium: Conventional and Recent Advances in Extraction and Production of Titanium Metal; Elsevier: Amsterdam, The Netherlands, 2019; pp. 287–313. [Google Scholar]
- Wang, S.; Zhang, F.; Liu, X.; Zhang, L. CaO solubility and activity coefficient in molten salts CaCl2–x (x = 0, NaCl, KCl, SrCl2, BaCl2 and LiCl). Thermochim. Acta 2008, 470, 105–107. [Google Scholar] [CrossRef]
- Schwandt, C.; Doughty, G.R.; Fray, D.J. The FFC-Cambridge process for titanium metal winning. Key Eng. Mater. 2010, 436, 3–25. [Google Scholar] [CrossRef] [Green Version]
- Hu, D.; Dolganov, A.; Ma, M.; Bhattacharya, B.; Bishop, M.T.; Chen, G.Z. Development of the Fray-Farthing-Chen Cambridge process: Towards the sustainable production of titanium and its alloys. JOM 2018, 70, 129–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sohn, H.S.; Jung, J.Y. Current Status of Titanium Smelting Technology. Resour. Recycl. 2016, 25, 68–79. [Google Scholar]
- Suzuki, R.O. Direct reduction processes for titanium oxide in molten salt. JOM 2007, 59, 68–71. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, R.O.; Aizawa, M.; Ono, K. Calcium-deoxidation of niobium and titanium in Ca-saturated CaCl2 molten salt. J. Alloys Compd. 1999, 288, 173–182. [Google Scholar] [CrossRef]
- Kong, L.; Ouchi, T.; Okabe, T.H. Direct Deoxidation of Ti by Mg in MgCl2–HoCl3 Flux. Mater. Trans. 2019, 60, 2059–2068. [Google Scholar] [CrossRef] [Green Version]
- Jaffe, L.F. Calcium explosions as triggers of development. Ann. N. Y. Acad. Sci. 1980, 339, 86–101. [Google Scholar] [CrossRef] [PubMed]
- Lesbani, A.; Tamba, P.; Mohadi, R.; Riyanti, F. Preparation of calcium oxide from Achatina fulica as catalyst for production of biodiesel from waste cooking oil. Indones. J. Chem. 2013, 13, 176–180. [Google Scholar] [CrossRef]
- Vrana, L.M. Calcium and Calcium Alloys. Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley and Sons: Hoboken, NJ, USA, 2000; p. 1226. [Google Scholar]
- Oh, J.M.; Roh, K.M.; Lee, B.K.; Suh, C.Y.; Kim, W.; Kwon, H.; Lim, J.W. Preparation of low oxygen content alloy powder from Ti binary alloy scrap by hydrogenation–dehydrogenation and deoxidation process. J. Alloys Compd. 2014, 593, 61–66. [Google Scholar] [CrossRef]
- Waseda, Y.; Isshiki, M. (Eds.) Purification Process and Characterization of Ultra-High Purity Metals: Application of Basic Science to Metallurgical Processing; Springer Science & Business Media: Berlin, Germany, 2012; pp. 332–408. [Google Scholar]
- Kong, L.; Ouchi, T.; Zheng, C.; Okabe, T.H. Electrochemical deoxidation of titanium scrap in MgCl2-HoCl3 system. J. Electrochem. Soc. 2019, 166, E429. [Google Scholar] [CrossRef]
- Okabe, T.H.; Hamanaka, Y.; Taninouchi, Y.K. Direct oxygen removal technique for recycling titanium using molten MgCl2 salt. Faraday Discuss. 2016, 190, 109–126. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.; Li, J.; Lv, K.; Zhang, W.; Ding, X.; Yang, G.; Jiang, X. Surface thermal oxidation on titanium implants to enhance osteogenic activity and in vivo osseointegration. Sci. Rep. 2016, 6, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Abbate, M.; Goedkoop, J.B.; De Groot, F.M.F.; Grioni, M.; Fuggle, J.C.; Hofmann, S.; Petersen, H.; Sacchi, M. Probing depth of soft x-ray absorption spectroscopy measured in total-electron-yield mode. Surf. Interface Anal. 1992, 18, 65–69. [Google Scholar] [CrossRef]
- Wan, Y.; Samundsett, C.; Bullock, J.; Hettick, M.; Allen, T.; Yan, D.; Peng, J.; Wu, Y.; Cui, J.; Javey, A.; et al. Conductive and stable magnesium oxide electron-selective contacts for efficient silicon solar cells. Adv. Energy Mater. 2017, 7, 1601863. [Google Scholar] [CrossRef]
- Fedoročková, A.; Raschman, P. Effects of pH and acid anions on the dissolution kinetics of MgO. Chem. Eng. Sci. 2008, 143, 265–272. [Google Scholar] [CrossRef]
- Cuadros-Lugo, E.; Lardizabal-Gutiérrez, D.; Carreño-Gallardo, C.; Estrada-Guel, I.; Herrera-Ramirez, J.; López, C. Removal of MgO impurity crystals by mechanical milling exfoliation of graphene obtained by CO2 atmosphere synthesis method. Microstruct. 2021, 27, 1762–1764. [Google Scholar] [CrossRef]
- Choi, S.H.; Sim, J.J.; Lim, J.H.; Seo, S.J.; Kim, D.W.; Hyun, S.K.; Park, K.T. Removal of Mg and MgO by-products through magnesiothermic reduction of Ti powder in self-propagating high-temperature synthesis. Metals 2019, 9, 169. [Google Scholar] [CrossRef] [Green Version]
- Mubarok, M.Z.; Yudiarto, A. Synthesis of Magnesium Oxide from Ferronickel Smelting Slag through Hydrochloric Acid Leaching-Precipitation and Calcination. In Energy Technology; Springer: Cham, Switzerland, 2017; pp. 247–258. [Google Scholar]
- Nersisyan, H.; Woo, H.Y.; Ri, V.; Thanh-Nam, H.; Moon, F.; MacDonald, A.; Lee, J.H. Hf metal powder synthesis via a chemically activated combustion-reduction process. Mater. Chem. Phys. 2021, 263, 124417. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).