A Review of Recent Advances in Depression Techniques for Flotation Separation of Cu–Mo Sulfides in Porphyry Copper Deposits
Abstract
1. Introduction
2. Typical Process of Cu–Mo Sulfide Ores
3. Alternative Options for Selective Flotation of Cu–Mo Bulk Concentrates
3.1. Molybdenite Depression
3.1.1. Dextrin
3.1.2. Lignosulfonates
3.1.3. Carboxymethyl-Based Organic Compounds and Humic Acid
3.2. Depression of Cu Minerals
3.2.1. Inorganic Depressants
3.2.2. Organic Depressants
3.3. Oxidation Treatments for Depressing Cu Minerals
3.3.1. Ozone Oxidation
3.3.2. Plasma Oxidation
3.3.3. H2O2 Oxidation
3.3.4. Electrolysis Oxidation
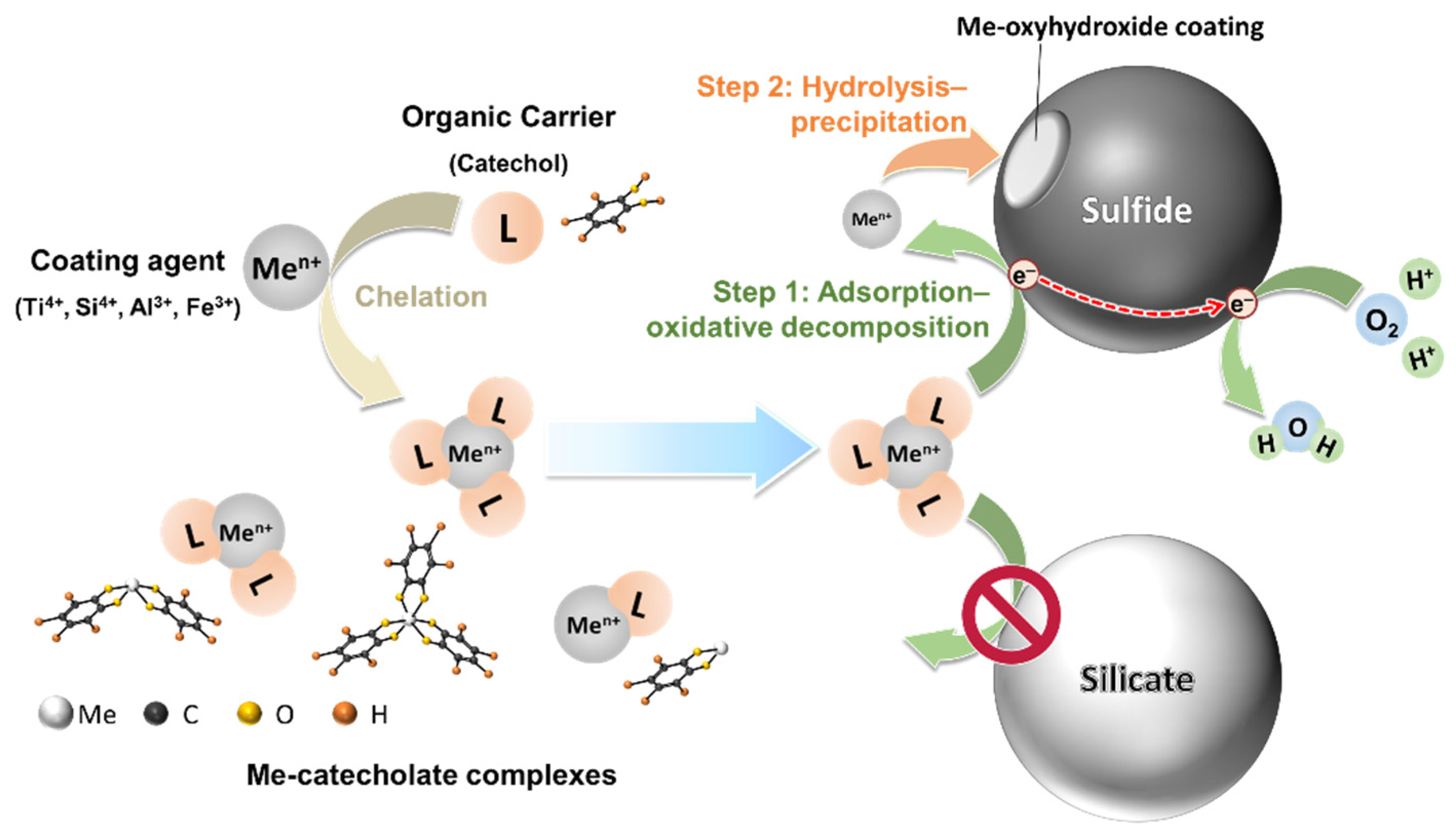
3.4. Microencapsulation Techniques for Depressing Cu Minerals
4. Summary
Author Contributions
Funding
Conflicts of Interest
References
- John, D.A.; Ayuso, R.A.; Barton, M.D.; Blakely, R.J.; Bodnar, R.J.; Dilles, J.H.; Gray, F.; Graybeal, F.T.; Mars, J.C.; McPhee, D.K.; et al. Porphyry Copper Deposit Model: Chapter B of Mineral Deposit Models for Resource Assessment; Scientific Investigations Report 2010–5070–B; US Geological Survey: Menlo Park, CA, USA, 2010; 169p. [Google Scholar]
- John, D.A.; Taylor, R.D. By-products of porphyry copper and molybdenum deposits. Rev. Econ. Geol. 2016, 18, 137–164. [Google Scholar]
- Lee, C.-T.A.; Tang, M. How to make porphyry copper deposits. Earth Planet. Sci. Lett. 2020, 529, 115868. [Google Scholar] [CrossRef]
- Seedorff, E.; Dilles, J.H.; Proffett, J.M., Jr.; Einaudi, M.T.; Zurcher, L.; Stavast, W.J.A.; Johnson, D.A.; Barton, M.D. Porphyry deposits: Characteristics and origin of hypogene features. Econ. Geol. 2005, 100, 251–298. [Google Scholar]
- Sinclair, W.D. Porphyry deposits. In Mineral Deposits of Canada: A Synthesis of Major Deposit-Types, District Metallogeny, the Evolution of Geological Provinces, and Exploration Methods, Geological Association of Canada, Mineral Deposits Division; Goodfellow, W.D., Ed.; Special Publication: London, UK, 2007; Volume 5, pp. 223–243. [Google Scholar]
- Bulatovic, S.M. 12–Flotation of Copper Sulfide Ores. In Handbook of Flotation Reagents; Bulatovic, S.M., Ed.; Elsevier: Amsterdam, The Netherlands, 2007; pp. 235–293. ISBN 978-0-444-53029-5. [Google Scholar]
- Amelunxen, P.; Schmitz, C.; Hill, L.; Goodweiler, N.; Andres, J. Molybdenum. In SME Mineral Processing & Extractive Metallurgy Handbook; Dunne, R.C., Komar Kawatra, S., Young, C.A., Eds.; Society for Mining, Metallurgy & Exploration (SME): Littleton, CO, USA, 2019; Volume 2, pp. 1891–1916. [Google Scholar]
- Liu, G.; Lu, Y.; Zhong, H.; Cao, Z.; Xu, Z. A novel approach for preferential flotation recovery of molybdenite from a porphyry copper-molybdenum ore. Miner. Eng. 2012, 36, 37–44. [Google Scholar] [CrossRef]
- Peterson, J.A.; Saran, M.S.; Wisnouskas, J.S. Differential Flotation Reagent for Molybdenum Separation. U.S. Patent No. 4,575,419, 11 March 1986. [Google Scholar]
- Yin, Z.; Sun, W.; Hu, Y.; Zhai, J.; Qingjun, G. Evaluation of the replacement of NaCN with depressant mixtures in the separation of copper-molybdenum sulphide ore by flotation. Sep. Purif. Technol. 2017, 173, 9–16. [Google Scholar] [CrossRef]
- Sutherland, K.L.; Wark, I.W. Principles of Flotation, 2nd ed.; Australasian Inst. Mining & Metallurgy: Melbourne, Australia, 1955. [Google Scholar]
- Mu, Y.; Peng, Y.; Lauten, R.A. The depression of pyrite in selective flotation by different reagent systems—A literature review. Miner. Eng. 2016, 96–97, 143–156. [Google Scholar] [CrossRef]
- Hirajima, T.; Miki, H.; Suyantara, G.P.W.; Matsuoka, H.; Elmahdy, A.M.; Sasaki, K.; Imaizumi, Y.; Kuroiwa, S. Selective flotation of chalcopyrite and molybdenite with H2O2 oxidation. Miner. Eng. 2017, 100, 83–92. [Google Scholar] [CrossRef]
- Napier-Munn, T.; Wills, B.A. Wills’ Mineral Processing Technology: An Introduction to the Practical Aspects of Ore Treatment and Mineral Recovery, 7th ed.; Butterworth-Heinemann: Oxford, UK, 2006. [Google Scholar]
- Zhao, Q.; Liu, W.; Wei, D.; Wang, W.; Cui, B.; Liu, W. Effect of copper ions on the flotation separation of chalcopyrite and molybdenite using sodium sulfide as a depressant. Miner. Eng. 2018, 115, 44–52. [Google Scholar] [CrossRef]
- Guo, B.; Peng, Y.; Espinosa-Gomez, R. Cyanide chemistry and its effect on mineral flotation. Miner. Eng. 2014, 66–68, 25–32. [Google Scholar] [CrossRef]
- Sinozaki, H.; Hara, R.; Mitsukuri, S. The vapour pressures of hydrogen cyanide. Bull. Chem. Soc. Jpn. 1926, 1, 59–61. [Google Scholar] [CrossRef]
- Stull, D.R. Vapor pressure of pure substances. Organic and inorganic compounds. Ind. Eng. Chem. 1947, 39, 517–540. [Google Scholar] [CrossRef]
- Milby, T.H.; Baselt, R.C. Hydrogen sulfide poisoning: Clarification of some controversial issues. Am. J. Ind. Med. 1999, 35, 192–195. [Google Scholar] [CrossRef]
- National Research Council. Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 2; The National Academies Press: Washington, DC, USA, 2002. [Google Scholar] [CrossRef]
- Johnston, A.; Meadows, D.G.; Cappuccitti, F. Copper mineral processing. In SME Mineral Processing & Extractive Metallurgy Handbook; Dunne, R.C., Komar Kawatra, S., Young, C.A., Eds.; Society for Mining, Metallurgy & Exploration (SME): Littleton, CO, USA, 2019; Volume 2, pp. 1615–1642. [Google Scholar]
- Hernlund, R.W. Extraction of molybdenite from copper flotation products. Q. J. Colo. Sch. Mines 1961, 56, 177–196. [Google Scholar]
- Blanco, M.; Coello, J.; Iturriaga, H.; Maspoch, S.; González Bañó, R. On-line monitoring of starch enzymatic hydrolysis by near-infrared spectroscopy. Analyst 2000, 125, 749–752. [Google Scholar] [CrossRef]
- Wie, J.M.; Fuerstenau, D.W. The effect of dextrin on surface properties and the flotation of molybdenite. Int. J. Miner. Process. 1974, 1, 17–32. [Google Scholar] [CrossRef]
- Beaussart, A.; Mierczynska-Vasilev, A.; Beattie, D.A. Adsorption of dextrin on hydrophobic minerals. Langmuir 2009, 25, 9913–9921. [Google Scholar] [CrossRef]
- Jorjani, E.; Barkhordari, H.R.; Tayebi Khorami, M.; Fazeli, A. Effects of aluminosilicate minerals on copper-molybdenum flotation from Sarcheshmeh porphyry ores. Miner. Eng. 2011, 24, 754–759. [Google Scholar] [CrossRef]
- Ansari, A.; Pawlik, M. Floatability of chalcopyrite and molybdenite in the presence of lignosulfonates. Part I. Adsorption studies. Miner. Eng. 2007, 20, 600–608. [Google Scholar] [CrossRef]
- Ansari, A.; Pawlik, M. Floatability of chalcopyrite and molybdenite in the presence of lignosulfonates. Part II. Hallimond tube flotation. Miner. Eng. 2007, 20, 609–616. [Google Scholar] [CrossRef]
- Buza, T.B.; Hiscox, T.O.; Kuhn, M.C. Use of lignin sulphonate as moly depressant boosts recovery at Twin Buttes. Eng. Min. J. 1975, 176, 87–91. [Google Scholar]
- Mathieu, G.; Bruce, R. Getting the talc out of molybdenite ores. Can. Min. J. 1974, 95, 75–77. [Google Scholar]
- Yuan, D.; Cadien, K.; Liu, Q.; Zeng, H. Separation of talc and molybdenite: Challenges and opportunities. Miner. Eng. 2019, 143, 105923. [Google Scholar] [CrossRef]
- Yuan, D.; Cadien, K.; Liu, Q.; Zeng, H. Flotation separation of Cu-Mo sulfides by O-Carboxymethyl chitosan. Miner. Eng. 2019, 134, 202–205. [Google Scholar] [CrossRef]
- Yuan, D.; Cadien, K.; Liu, Q.; Zeng, H. Adsorption characteristics and mechanisms of O-Carboxymethyl chitosan on chalcopyrite and molybdenite. J. Colloid Interface Sci. 2019, 552, 659–670. [Google Scholar] [CrossRef] [PubMed]
- Yuan, D.; Cadien, K.; Liu, Q.; Zeng, H. Selective separation of copper-molybdenum sulfides using humic acids. Miner. Eng. 2019, 133, 43–46. [Google Scholar] [CrossRef]
- Kor, M.; Korczyk, P.M.; Addai-Mensah, J.; Krasowska, M.; Beattie, D.A. Carboxymethylcellulose adsorption on molybdenite: The effect of electrolyte composition on adsorption, bubble–surface collisions, and flotation. Langmuir 2014, 30, 11975–11984. [Google Scholar] [CrossRef]
- Mierczynska-Vasilev, A.; Beattie, D.A. Adsorption of tailored carboxymethyl cellulose polymers on talc and chalcopyrite: Correlation between coverage, wettability, and flotation. Miner. Eng. 2010, 23, 985–993. [Google Scholar] [CrossRef]
- Qui, X.; Yang, H.; Chen, G.; Luo, W. An Alternative Depressant of Chalcopyrite in Cu–Mo Differential Flotation and Its Interaction Mechanism. Minerals 2019, 9, 1. [Google Scholar]
- Bulatovic, S.M. Use of organic polymers in the flotation of polymetallic ores: A review. Miner. Eng. 1999, 12, 341–354. [Google Scholar] [CrossRef]
- Grano, S.; Ralston, J.; Smart, R.S.C. Influence of electrochemical environment on the flotation behavior of Mt. Isa copper and lead-zinc ore. Int. J. Miner. Process. 1990, 30, 69–97. [Google Scholar] [CrossRef]
- Heyes, G.W.; Trahar, W.J. The natural flotability of chalcopyrite. Int. J. Miner. Process. 1977, 4, 317–344. [Google Scholar] [CrossRef]
- Ralston, J. Eh and its consequences in sulphide mineral flotation. Miner. Eng. 1991, 4, 859–878. [Google Scholar] [CrossRef]
- Richardson, P.E.; Walker, G.W. The flotation of chalcocite, bornite, chalcopyrite, and pyrite in an electrochemical-flotation cell. In Proceedings of the XV International Mineral Processing Congress, Cannes, France, 2–9 June 1985; pp. 198–210. [Google Scholar]
- Guy, P.J.; Trahar, W.J. The effects of oxidation and mineral interaction on sulphide flotation. In Flotation of Sulphide Minerals; Forrsberg, K.S.E., Ed.; Elsevier: Amsterdam, The Netherlands, 1985; pp. 91–110. [Google Scholar]
- Miki, H.; Hirajima, T.; Muta, Y.; Suyantara, G.P.W.; Sasaki, K. Effect of sodium sulfite on floatability of chalcopyrite and molybdenite. Minerals 2018, 8, 172. [Google Scholar] [CrossRef]
- Grano, S.R.; Sollaart, M.; Skinner, W.; Prestidge, C.A.; Ralston, J. Surface modification in the chalcopyrite-sulphite ion system. I. collectorless flotation, XPS and dissolution study. Int. J. Miner. Process. 1997, 50, 1–26. [Google Scholar] [CrossRef]
- Castro, S.; Laskowski, J.S. Froth flotation in saline water. KONA Powder Part. J. 2011, 29, 4–15. [Google Scholar] [CrossRef]
- Castro, S. Challenges in flotation of Cu–Co sulfide ores in sea water. In The First International Symposium on Water in Mineral Processing; Drelich, J., Ed.; Society for Mining, Metallurgy & Exploration (SME): Littleton, CO, USA, 2012; pp. 29–40. [Google Scholar]
- Laskowski, J.S.; Castro, S.; Ramos, O. Effect of seawater main components on frothability in the flotation of Cu-Mo sulfide ore. Physicochem. Probl. Miner. Process. 2013, 50, 17–29. [Google Scholar]
- Suyantara, G.P.W.; Hirajima, T.; Miki, H.; Sasaki, K. Floatability of molybdenite and chalcopyrite in artificial seawater. Miner. Eng. 2018, 115, 117–130. [Google Scholar] [CrossRef]
- Castro, S.; Rioseco, P.; Laskowski, J.S. Depression of molybdenite in sea water. In Proceedings of the XXVI International Mineral Processing Congress, New Delhi, India, 24–28 September 2012; pp. 737–752. [Google Scholar]
- Laskowski, J.S.; Castro, S. Flotation in concentrated electrolyte solutions. Int. J. Miner. Process. 2015, 144, 50–55. [Google Scholar] [CrossRef]
- Ramos, O.; Castro, S.; Laskowski, J.S. Copper-molybdenum ores flotation in sea water: Floatability and frothability. Miner. Eng. 2013, 53, 108–112. [Google Scholar] [CrossRef]
- Qui, Z.; Liu, G.; Liu, Q.; Zhong, H. Understanding the roles of high salinity in inhibiting the molybdenite flotation. Colloids Surf. A Physicochem. Eng. Asp. 2016, 509, 123–129. [Google Scholar]
- Hirajima, T.; Suyantara, G.P.W.; Ichikawa, O.; Elmahdy, A.M.; Miki, H.; Sasaki, K. Effect of Mg2+ and Ca2+ as divalent seawater cations on the floatability of molybdenite and chalcopyrite. Miner. Eng. 2016, 96–97, 83–93. [Google Scholar] [CrossRef]
- Nagaraj, D.R.; Farinato, R. Chemical factor effects in saline and hypersaline waters in the flotation of Cu and Cu-Mo ores. In Proceedings of the XXVII International Mineral Processing Congress, Santiago, Chile, 20–24 October 2014; pp. 20–24. [Google Scholar]
- Li, W.; Li, Y.; Wei, Z.; Xiao, Q.; Song, S. Fundamental Studies of SHMP in Reducing Negative Effects of Divalent Ions on Molybdenite Flotation. Minerals 2018, 8, 404. [Google Scholar] [CrossRef]
- Rebolledo, E.; Laskowski, J.S.; Gutierrez, L.; Castro, S. Use of dispersants in flotation of molybdenite in seawater. Miner. Eng. 2017, 100, 71–74. [Google Scholar] [CrossRef]
- Jeldres, R.I.; Arancibia-Bravo, M.P.; Reyes, A.; Aguirre, C.E.; Cortes, L.; Cisternas, L.A. The impact of seawater with calcium and magnesium removal for the flotation of copper-molybdenum sulphide ores. Miner. Eng. 2017, 109, 10–13. [Google Scholar] [CrossRef]
- Chen, J.; Lan, L.; Liao, X. Depression effect of pseudo glycolythiourea acid in flotation separation of copper–molybdenum. Trans. Nonferrous Met. Soc. China 2013, 23, 824–831. [Google Scholar] [CrossRef]
- Li, M.; Wei, D.; Shen, Y.; Liu, W.; Gao, S.; Liang, G. Selective depression effect in flotation separation of copper-molybdenum sulfides using 2,3-disulfanylbutanedioic acid. Trans. Nonferrous Met. Soc. China 2015, 25, 3126–3132. [Google Scholar] [CrossRef]
- Yin, Z.; Sun, X.; Hu, Y.; Guan, Q.; Zhang, C.; Gao, Y.; Zhai, J. Depressing behaviors and mechanism of disodium bis (carboxymethyl) trithiocarbonate on separation of chalcopyrite and molybdenite. Trans. Nonferrous Met. Soc. China 2017, 27, 883–890. [Google Scholar] [CrossRef]
- Li, M.; Wei, D.; Liu, Q.; Liu, W.; Zheng, J.; Sun, H. Flotation separation of copper-molybdenum sulfides using chitosan as a selective depressant. Miner. Eng. 2015, 83, 217–222. [Google Scholar] [CrossRef]
- Yin, Z.; Sun, W.; Hu, Y.; Zhang, C.; Guan, Q.; Zhang, C. Separation of molybdenite from chalcopyrite in the presence of novel depressant 4-amino-3-thioxo-3,4-dihydro-1,2,4-triazin-5(2H)-one. Minerals 2017, 7, 146. [Google Scholar]
- Yin, Z.; Sen, W.; Hu, Y.; Zhang, C.; Guan, Q.; Liu, R.; Chen, P.; Tian, M. Utilization of acetic acid-[(hydrazinylthioxomethyl)thio]-sodium as a novel selective depressant for chalcopyrite in the flotation separation of molybdenite. Sep. Purif. Technol. 2017, 179, 248–256. [Google Scholar] [CrossRef]
- Huang, P.; Cao, M.; Liu, Q. Using chitosan as a selective depressant in the differential flotation Cu-Pb sulfides. Int. J. Miner. Process. 2012, 106–109, 8–15. [Google Scholar] [CrossRef]
- Huang, P.; Cao, M.; Liu, Q. Adsorption of chitosan on chalcopyrite and galena from aqueous suspensions. Colloids Surf. A Physicochem. Eng. Asp. 2012, 409, 167–175. [Google Scholar] [CrossRef]
- Crini, G.; Badot, P.-M. Application of chitosan, a natural aminopolysaccharide, for dye removal from aqueous solutions by adsorption processes using batch studies: A review of recent literature. Prog. Polym. Sci. 2008, 33, 399–447. [Google Scholar] [CrossRef]
- Miller, J.D.; Ye, Y.; Jang, W.-H. Molybdenite Flotation from Copper Sulfide/Molybdenite Containing Materials by Ozone Conditioning. U.S. Patent No. 5,068,028, 26 November 1991. [Google Scholar]
- Ye, Y.; Jang, W.H.; Yalamanchili, M.R.; Miller, J.D. Molybdenite flotation from copper/molybdenum concentrates by ozone conditioning. Miner. Metall. Process. 1990, 7, 173–179. [Google Scholar] [CrossRef]
- Da Silva, L.M.; Jardim, W.F. Trends and strategies of ozone application in environmental problems. Quim. Nova 2006, 29, 310–317. [Google Scholar] [CrossRef]
- Kasprzyk-Hordern, B.; Ziółek, M.; Nawrocki, J. Catalytic ozonation and methods of enhancing molecular ozone reactions in water treatment. Appl. Catal. B Environ. 2003, 46, 639–669. [Google Scholar] [CrossRef]
- Huang, C.P.; Dong, C.; Tang, Z. Advanced chemical oxidation: Its present role and potential future in hazardous waste treatment. Waste Manag. 1993, 13, 361–377. [Google Scholar] [CrossRef]
- Castro, S.; Lopez-Valdivieso, A.; Laskowski, J.S. Review of the flotation of molybdenite. Part I: Surface properties and floatability. Int. J. Miner. Process. 2016, 148, 48–58. [Google Scholar] [CrossRef]
- Hirajima, T.; Mori, M.; Ichikawa, O.; Sasaki, K.; Miki, H.; Farahat, M.; Sawada, M. Selective flotation of chalcopyrite and molybdenite with plasma. Miner. Eng. 2014, 66–68, 102–111. [Google Scholar] [CrossRef]
- Suyantara, G.P.W.; Hirajima, T.; Miki, H.; Sasaki, K.; Yamane, M.; Takida, E.; Kuroiwa, S.; Imaizumi, Y. Selective flotation of chalcopyrite and molybdenite using H2O2 oxidation method with the addition of ferrous sulfate. Miner. Eng. 2018, 122, 312–326. [Google Scholar] [CrossRef]
- Lin, S.-S.; Gurol, M.D. Catalytic decomposition of hydrogen peroxide on iron oxide: Kinetics, mechanism, and implications. Environ. Sci. Technol. 1998, 32, 1417–1423. [Google Scholar] [CrossRef]
- Li, X.; Huang, Y.; Li, C.; Shen, J.; Deng, Y. Degradation of pCNB by Fenton like process using α-FeOOH. Chem. Eng. J. 2015, 260, 28–36. [Google Scholar] [CrossRef]
- Miki, H.; Matsuoka, H.; Hirajima, T.; Suyantara, G.P.W.; Sasaki, K. Electrolysis oxidation of chalcopyrite and molybdenite for selective flotation. Mater. Trans. 2017, 58, 761–767. [Google Scholar] [CrossRef]
- Jha, R.K.T.; Satur, J.; Hiroyoshi, N.; Ito, M.; Tsunekawa, M. Carrier-microencapsulation using Si-catechol complex for suppressing pyrite floatability. Miner. Eng. 2008, 21, 889–893. [Google Scholar] [CrossRef]
- Li, X.; Hiroyoshi, N.; Tabelin, C.B.; Naruwa, K.; Harada, C.; Ito, M. Suppressive effects of ferric-catecholate complexes on pyrite oxidation. Chemosphere 2019, 214, 70–78. [Google Scholar] [CrossRef]
- Park, I.; Tabelin, C.B.; Magaribuchi, K.; Seno, K.; Ito, M.; Hiroyoshi, N. Suppression of the release of arsenic from arsenopyrite by carrier-microencapsulation using Ti-catechol complex. J. Hazard. Mater. 2018, 344, 322–332. [Google Scholar] [CrossRef]
- Park, I.; Tabelin, C.B.; Seno, K.; Jeon, S.; Ito, M.; Hiroyoshi, N. Simultaneous suppression of acid mine drainage and arsenic release by carrier-microencapsulation using aluminum-catecholate complexes. Chemosphere 2018, 205, 414–425. [Google Scholar] [CrossRef] [PubMed]
- Park, I.; Tabelin, C.B.; Jeon, S.; Li, X.; Seno, K.; Ito, M.; Hiroyoshi, N. A review of recent strategies for acid mine drainage prevention and mine tailings recycling. Chemosphere 2019, 219, 588–606. [Google Scholar] [CrossRef]
- Park, I.; Tabelin, C.B.; Seno, K.; Jeon, S.; Inano, H.; Ito, M.; Hiroyoshi, N. Carrier-microencapsulation of arsenopyrite using Al-catecholate complex: Nature of oxidation products, effects on anodic and cathodic reactions, and coating stability under simulated weathering conditions. Heliyon 2020, 6, e03189. [Google Scholar] [CrossRef]

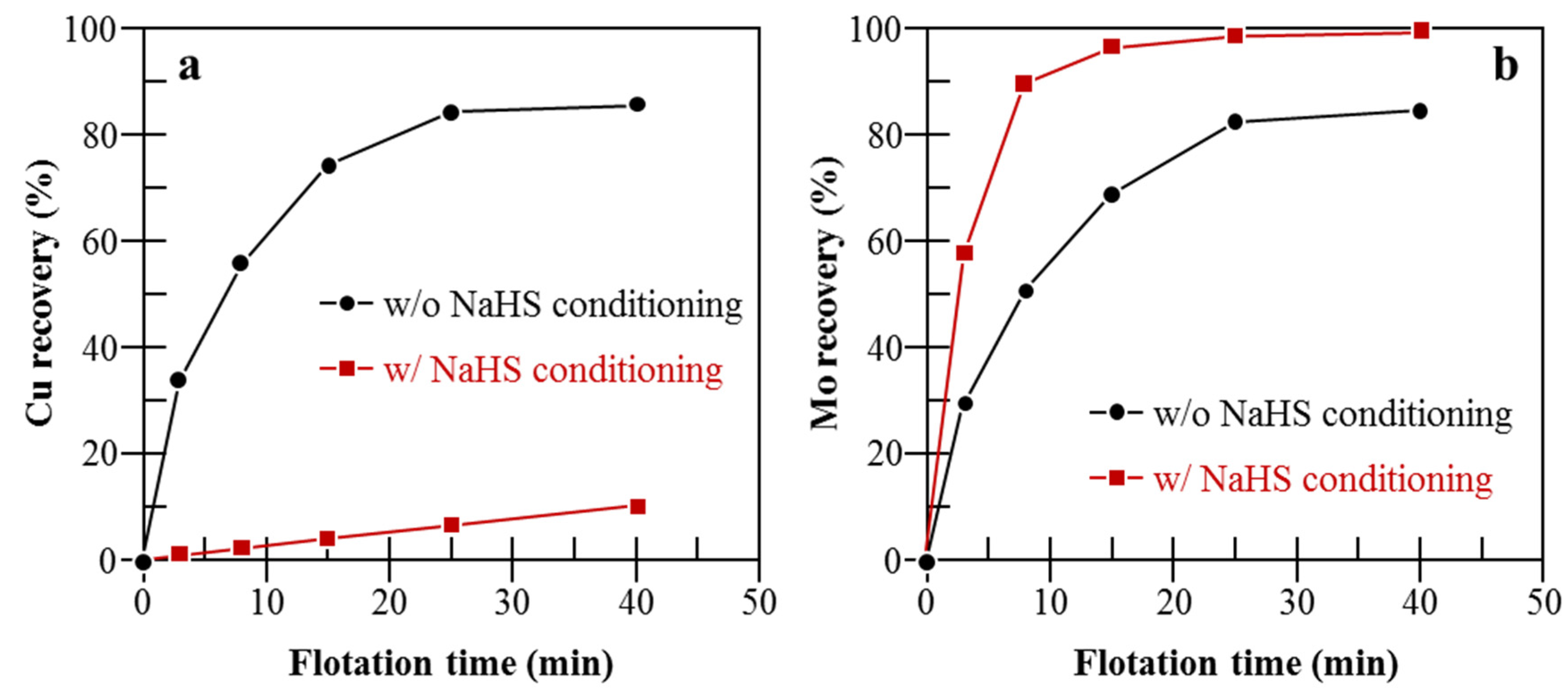
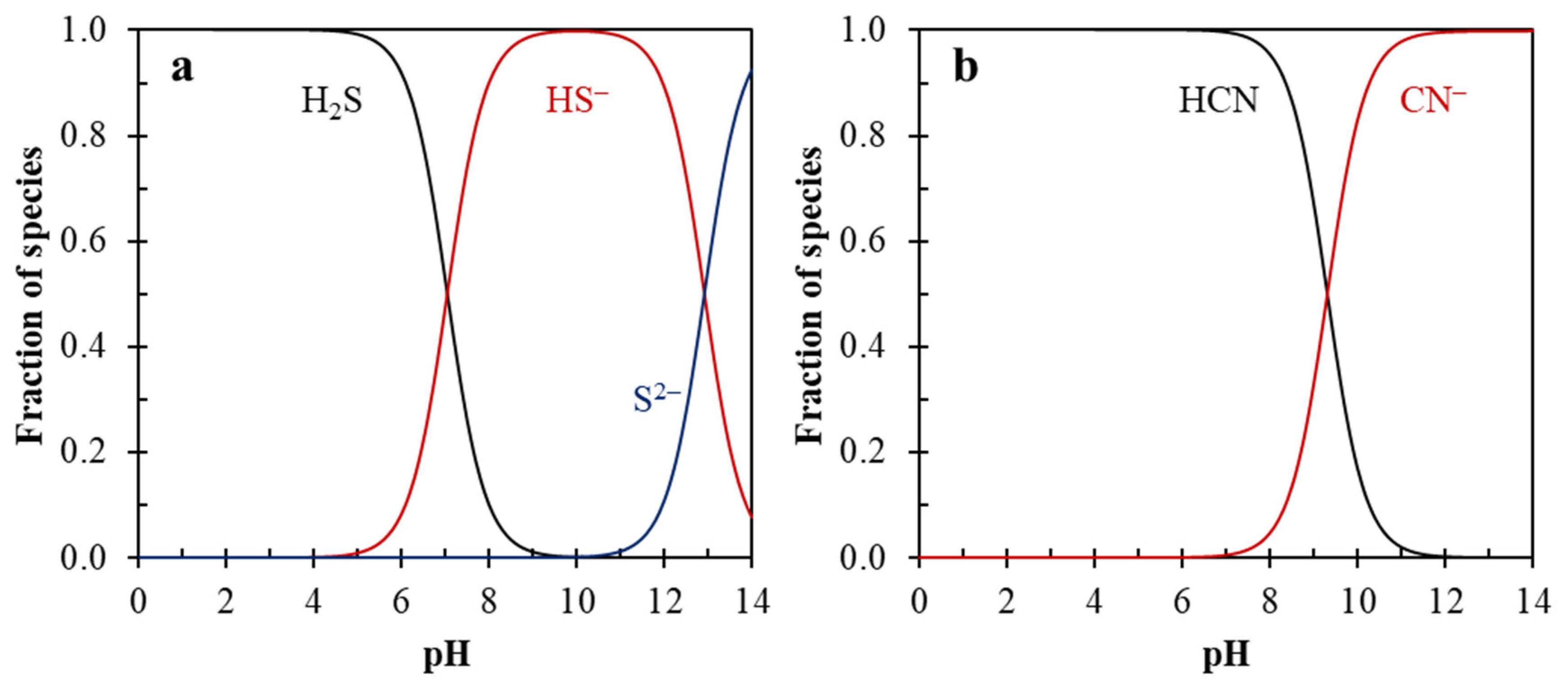
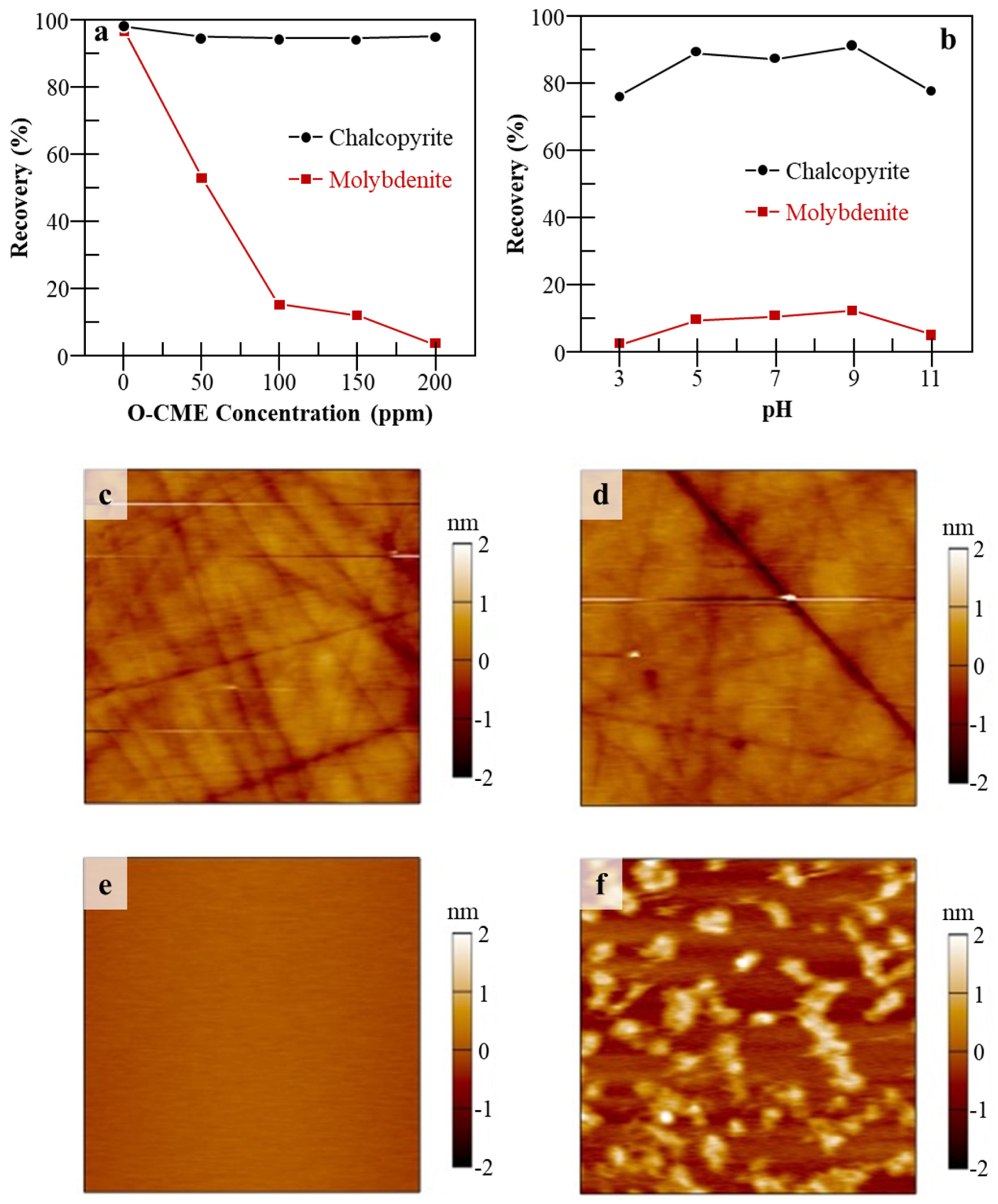


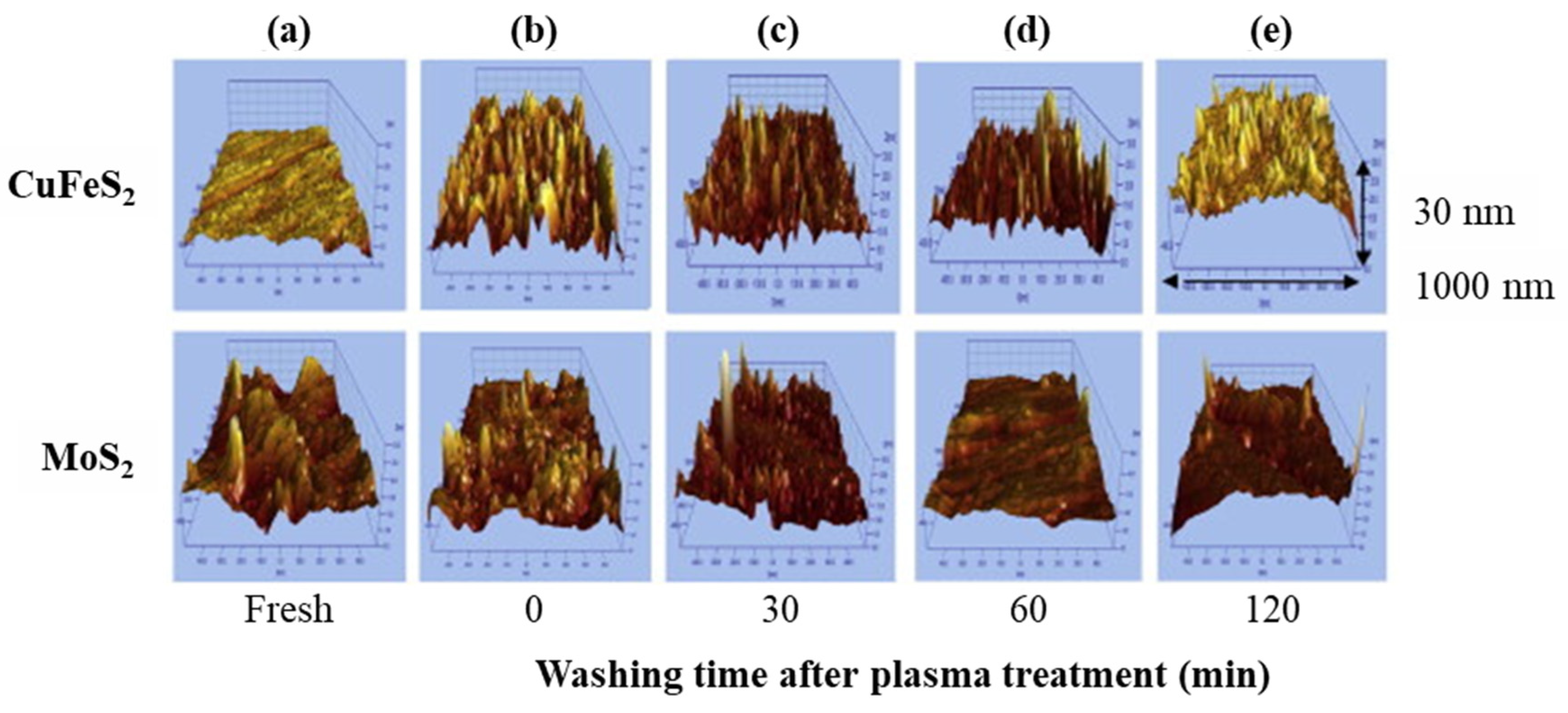
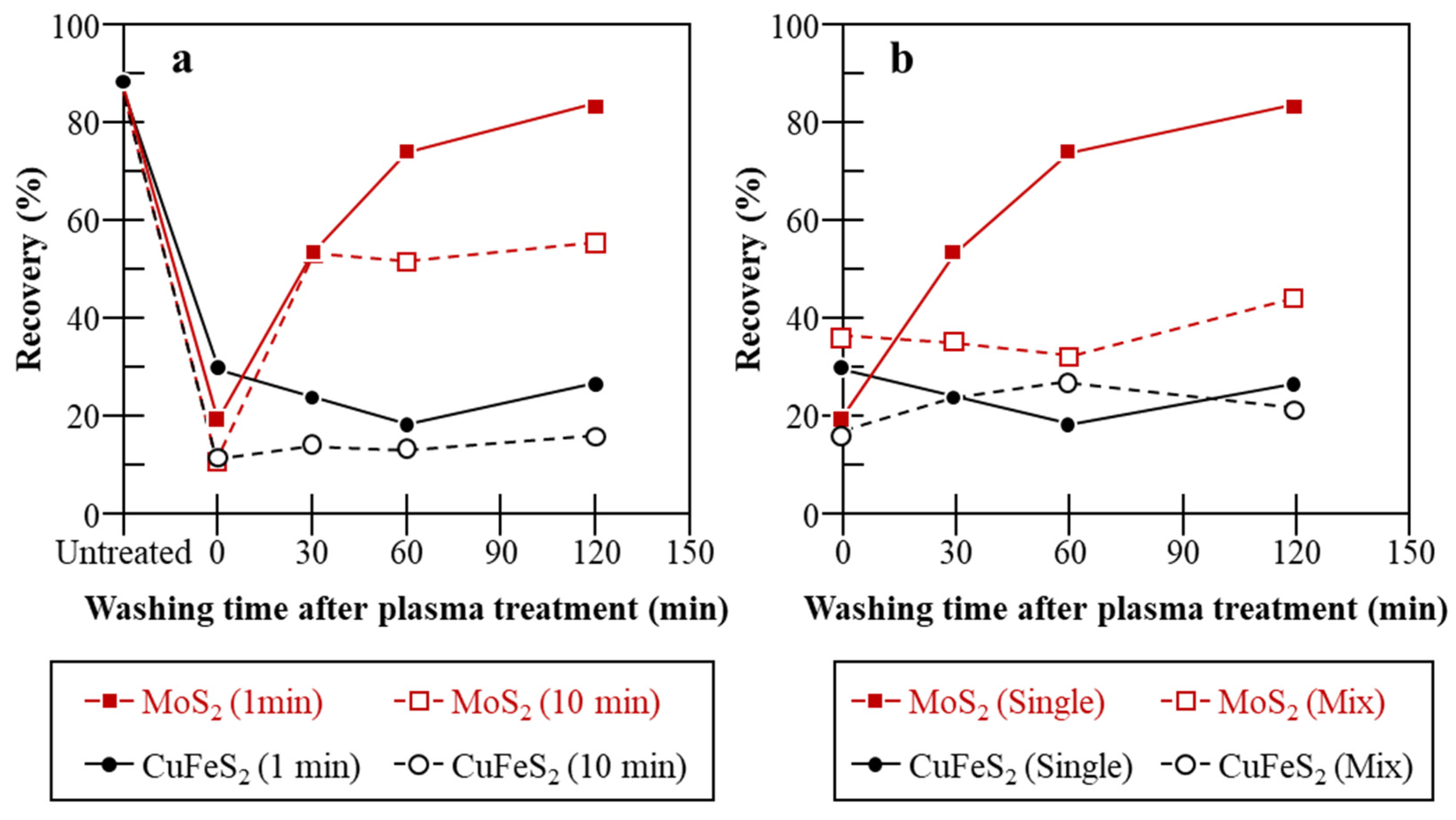
| Mo Depressant | Feed | Results | Features |
|---|---|---|---|
| Dextrin [22,23,24,25,26] | Natural MoS2 [24], Cu ore (0.7–3.03% Cu, 0.011–0.14% Mo) [26]. | ||
| Lignosulfonate [27,28,29,30,31] | Natural CuFeS2 and MoS2 [28]. |
|
|
| O-carboxymethyl chitosan (O-CMC) [32,33] | Natural CuFeS2 and MoS2 [32]. |
|
|
| Humic acid (HA) [34] | Natural CuFeS2 and MoS2 [34]. |
|
|
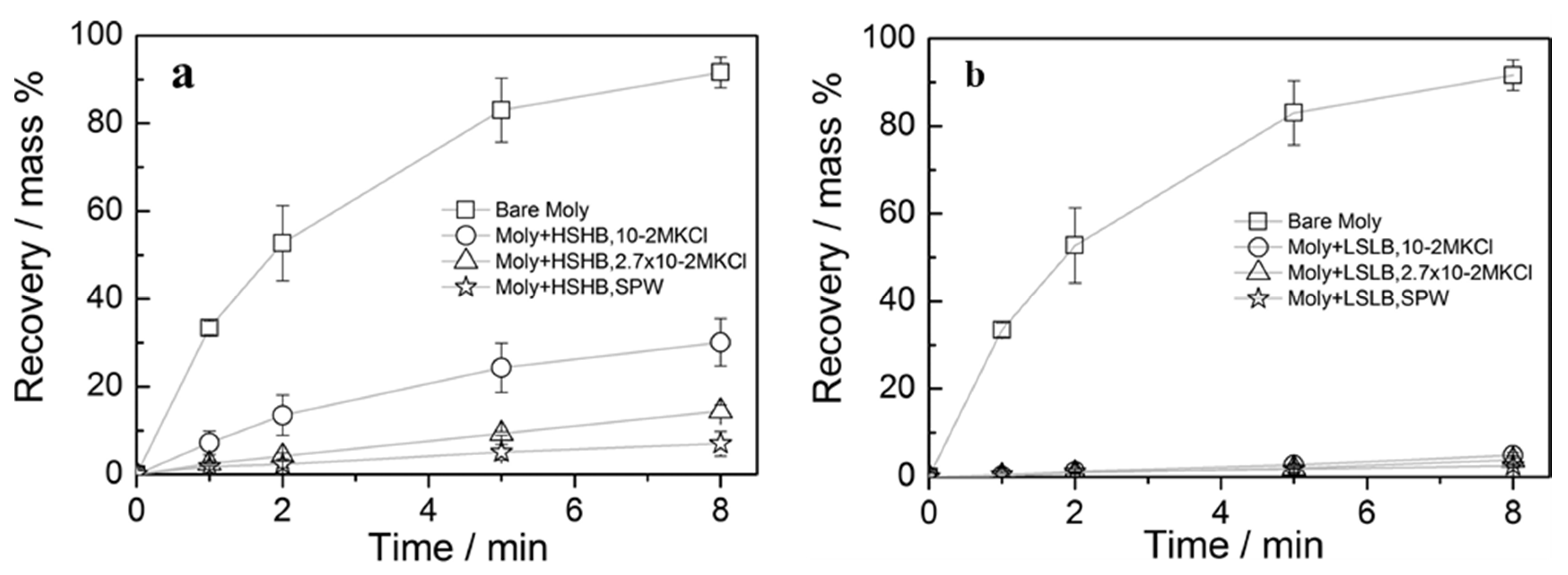
| Carboxymethylcellulose (CMC) [35,36,37] | Natural MoS2 [35]. |
|
|
| Electrolyte | HSHB | LSLB | ||
|---|---|---|---|---|
| Thickness (nm) | Γ (%) | Thickness (nm) | Γ (%) | |
| 10−2 M KCl | 1.1 ± 0.2 | 23 | 2.6 ± 0.2 | 100 |
| 2.76 × 10−2 M KCl | 1.6 ± 0.6 | 95 | 4.0 ± 0.4 | 100 |
| SPW * | 2.7 ± 0.5 | 100 | 6.3 ± 0.4 | 100 |
| Cu Depressant | Feed | Results | Features |
|---|---|---|---|
| Na2SO3 [39,40,41,42,43,44,45] | Natural CuFeS2 and MoS2 [44]. |
|
|
| Seawater [46,47,48,49,50,51,52,53,54,55,56,57,58] | Natural CuFeS2 and MoS2 [49]. |
|
|
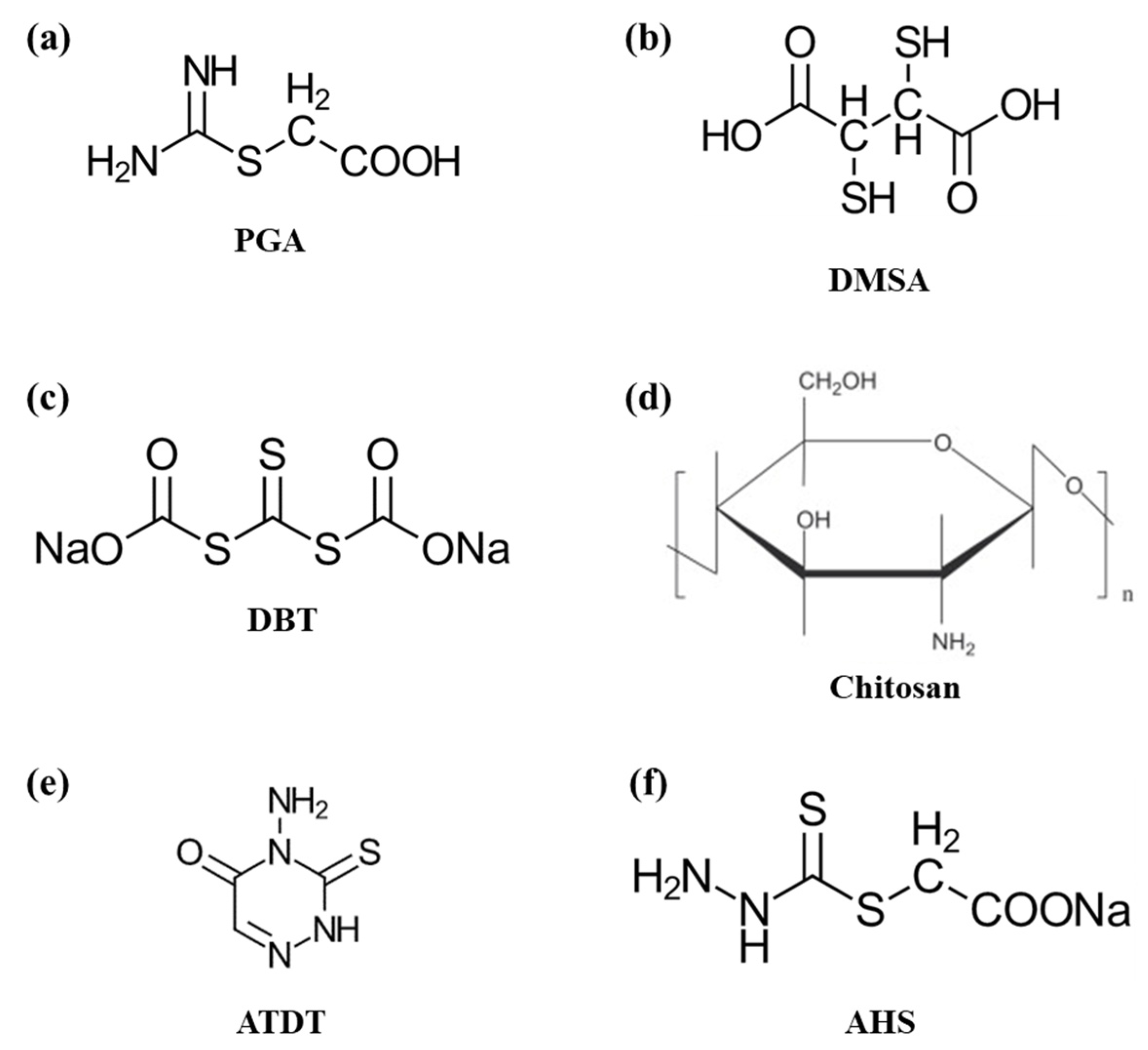
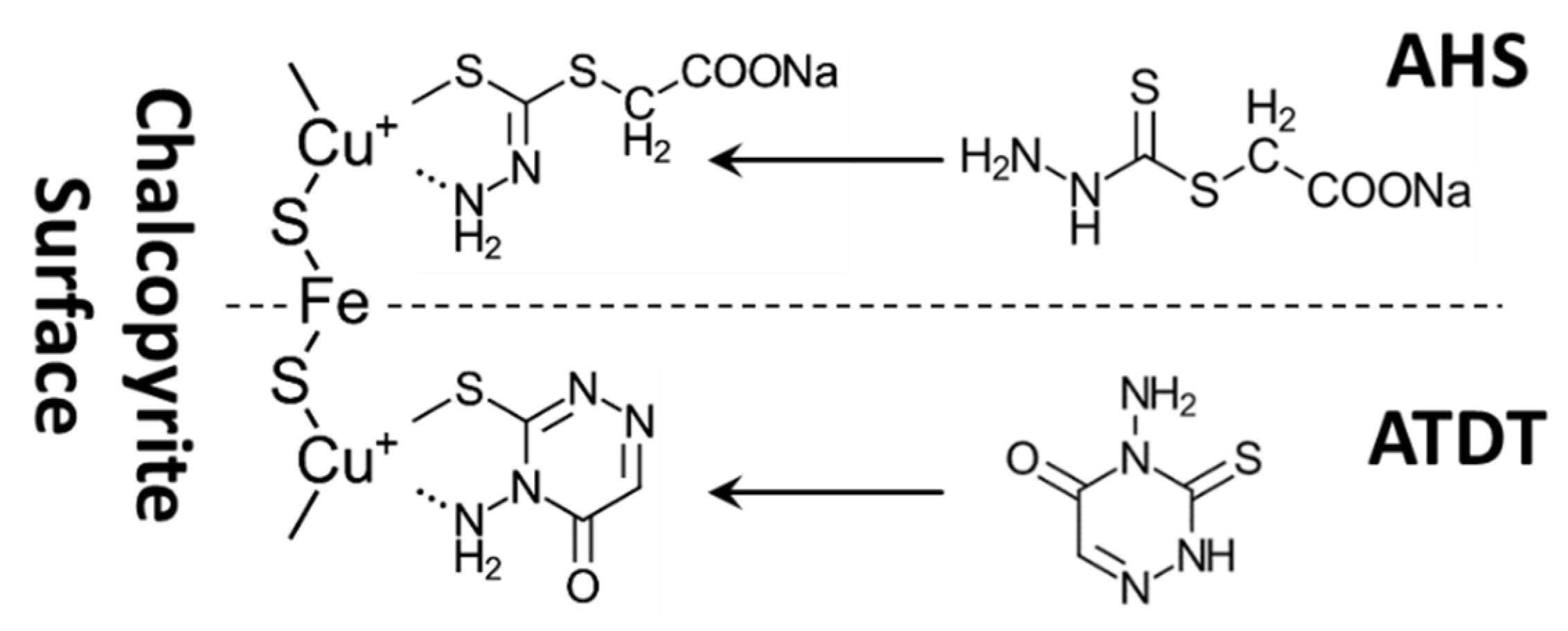
| Organic depressants (e.g., PGA [59], DMSA [60], DBT [61], chitosan [62,63,64,65], ATDT [66], and AHS [67]) | Bulk Cu–Mo conc. [59], Cu–Mo ore [60], Cu–Mo rough conc. [61], natural CuFeS2 and MoS2 [62], CuFeS2 and MoS2 purified by flotation [66,67]. |
|
|
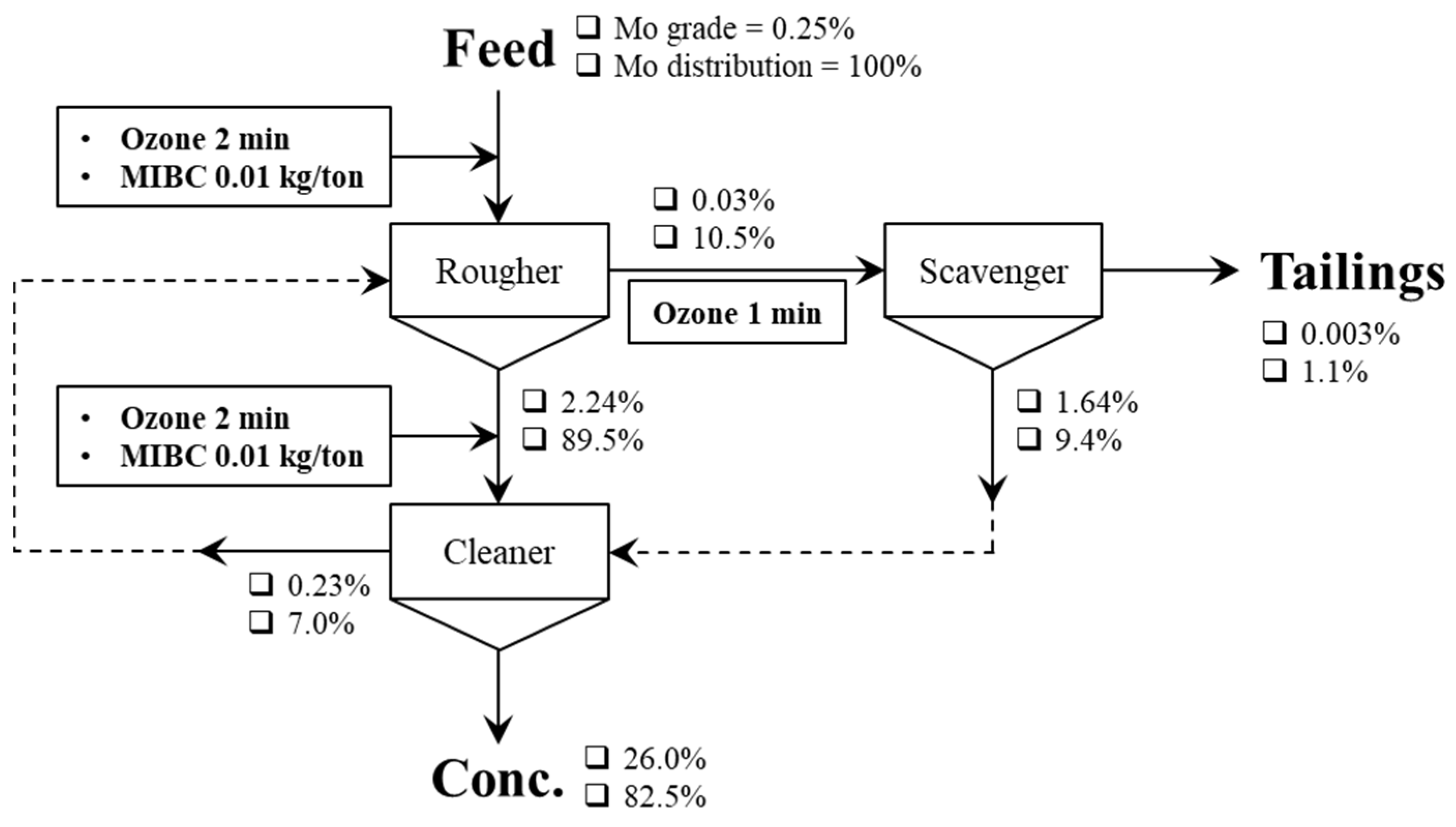
| Oxidation treatments (e.g., ozone (O3) [68,69,70,71,72,73], plasma [74], H2O2 [13,75,76,77], and electrolysis [78]). | Bulk Cu–Mo conc. [69], natural CuFeS2 and MoS2 [13,74,75], mineral electrodes [78]. |
|
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, I.; Hong, S.; Jeon, S.; Ito, M.; Hiroyoshi, N. A Review of Recent Advances in Depression Techniques for Flotation Separation of Cu–Mo Sulfides in Porphyry Copper Deposits. Metals 2020, 10, 1269. https://doi.org/10.3390/met10091269
Park I, Hong S, Jeon S, Ito M, Hiroyoshi N. A Review of Recent Advances in Depression Techniques for Flotation Separation of Cu–Mo Sulfides in Porphyry Copper Deposits. Metals. 2020; 10(9):1269. https://doi.org/10.3390/met10091269
Chicago/Turabian StylePark, Ilhwan, Seunggwan Hong, Sanghee Jeon, Mayumi Ito, and Naoki Hiroyoshi. 2020. "A Review of Recent Advances in Depression Techniques for Flotation Separation of Cu–Mo Sulfides in Porphyry Copper Deposits" Metals 10, no. 9: 1269. https://doi.org/10.3390/met10091269
APA StylePark, I., Hong, S., Jeon, S., Ito, M., & Hiroyoshi, N. (2020). A Review of Recent Advances in Depression Techniques for Flotation Separation of Cu–Mo Sulfides in Porphyry Copper Deposits. Metals, 10(9), 1269. https://doi.org/10.3390/met10091269





