A Review of SLMed Magnesium Alloys: Processing, Properties, Alloying Elements and Postprocessing
Abstract
1. Introduction
2. Formation and Energy Density of SLMed Magnesium Alloys
3. Properties of SLMed Magnesium Alloy
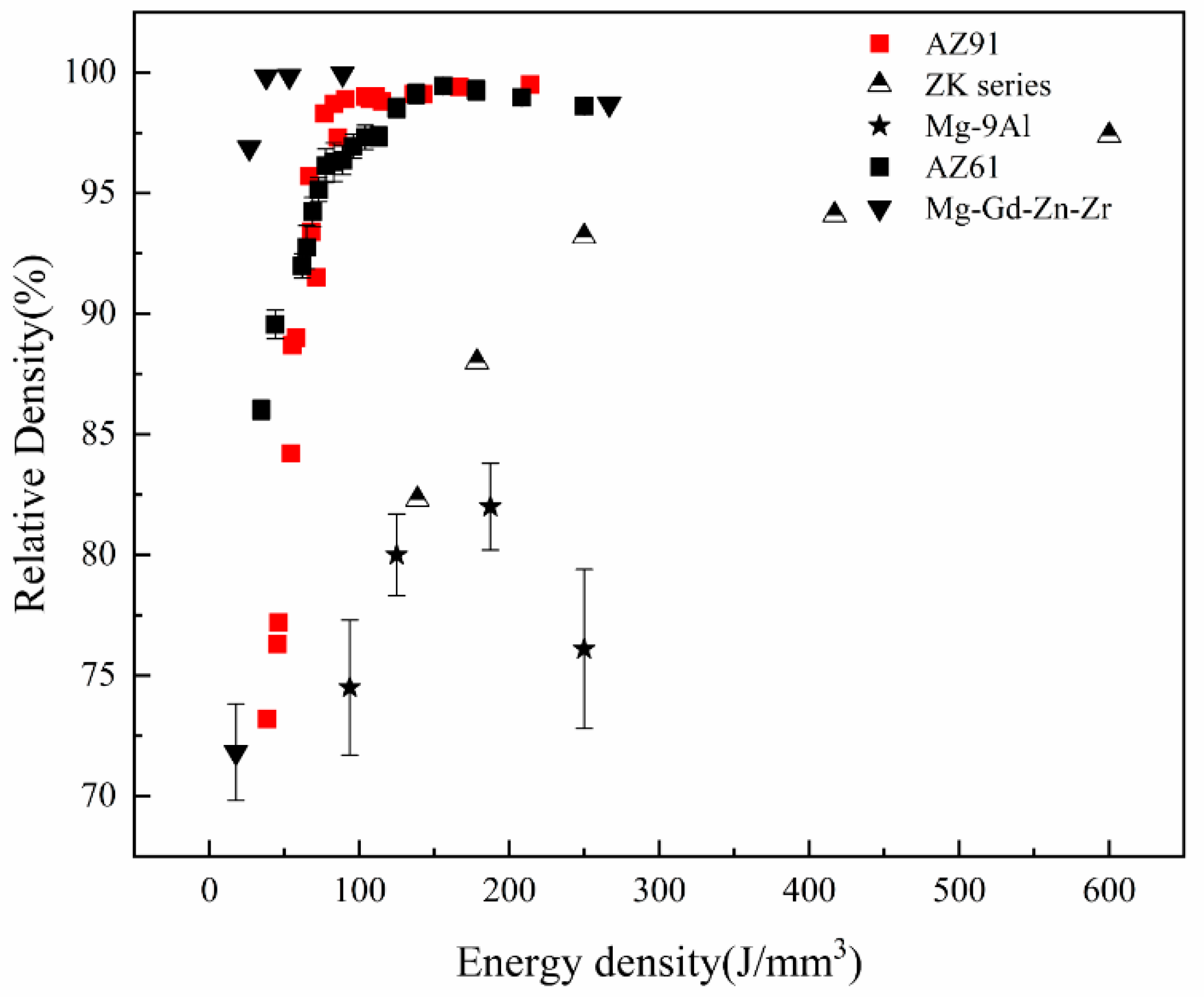
3.1. Relative Density of SLMed Magnesium Alloy
3.2. Microhardness of SLMed Magnesium Alloy
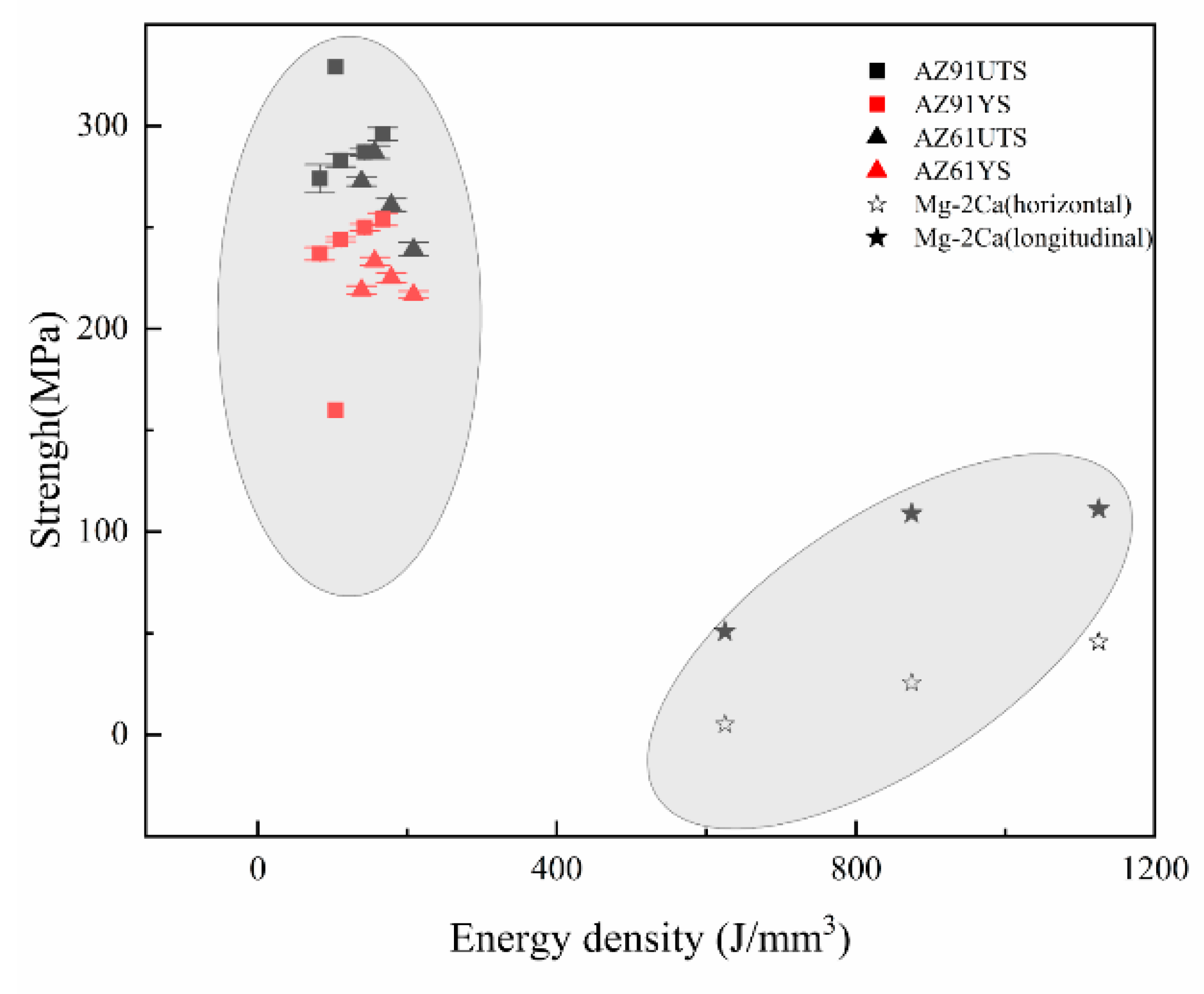
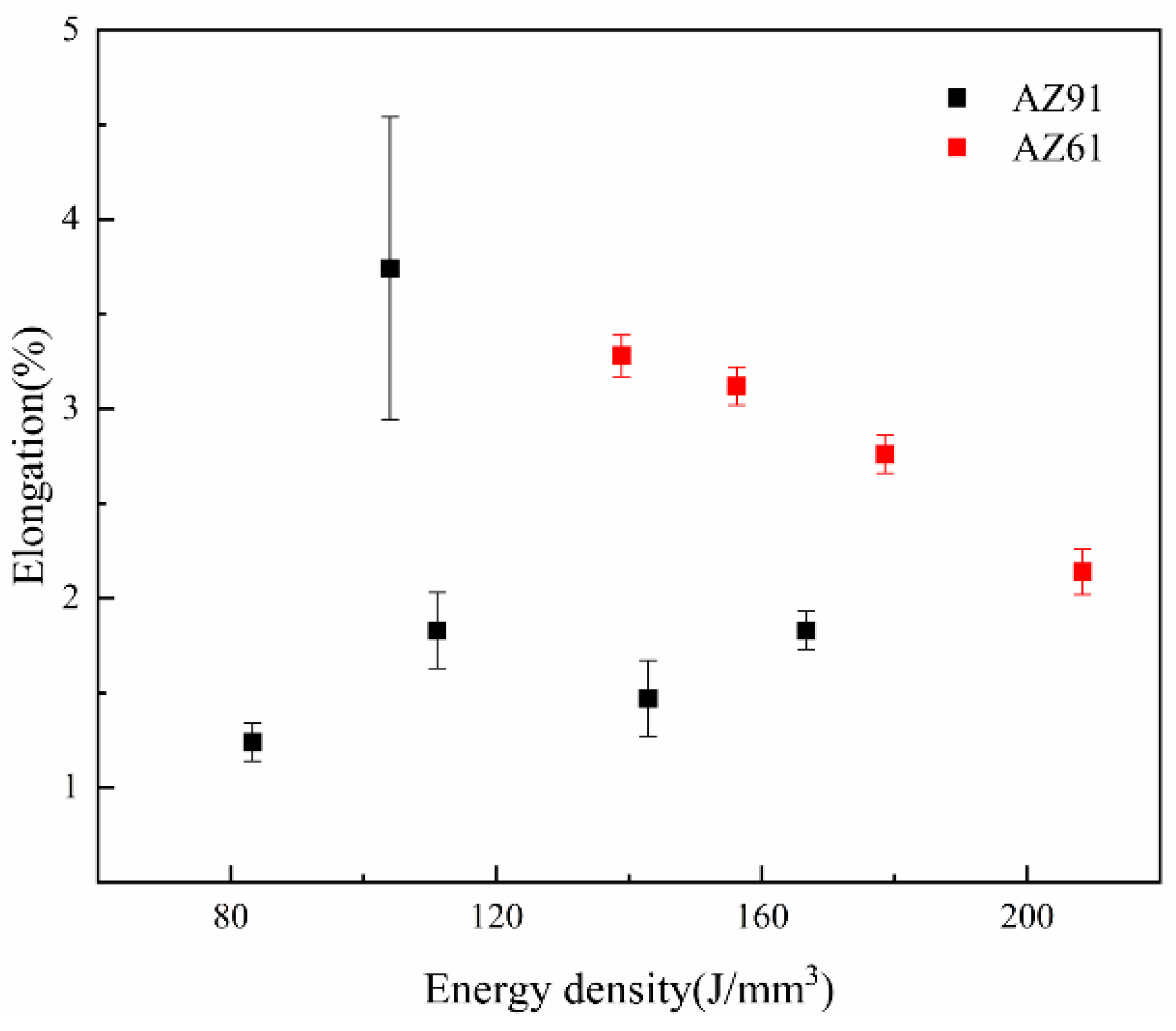
3.3. Mechanical Properties of SLMed Magnesium Alloy
3.4. Corrosion
3.5. Factors that Affect Performance Defects
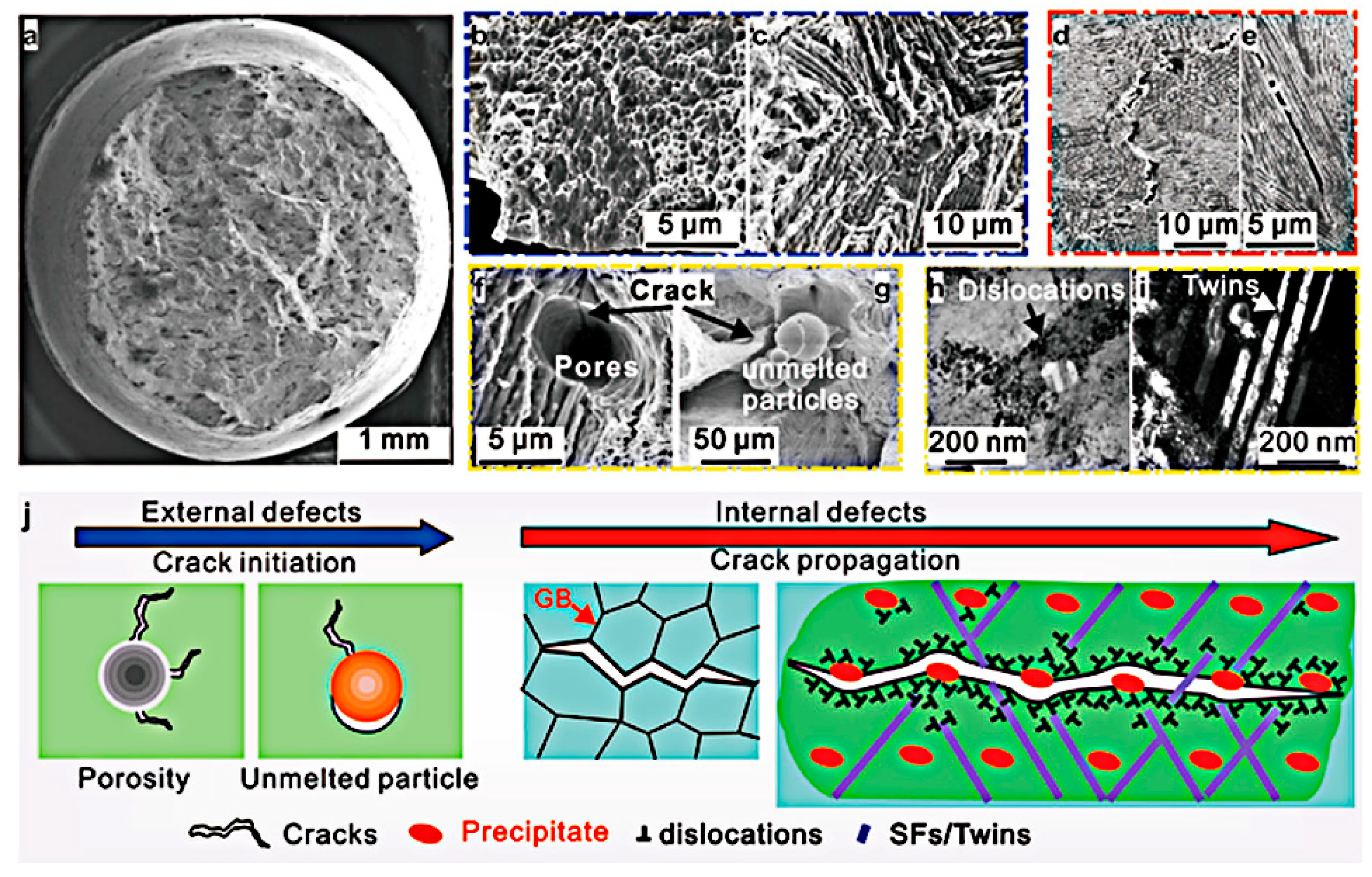
3.5.1. Cracking
3.5.2. Oxide Inclusions
3.5.3. Other Factors
4. Effect of Alloying Elements on the Properties of SLMed Magnesium Alloy
4.1. Regular Elements
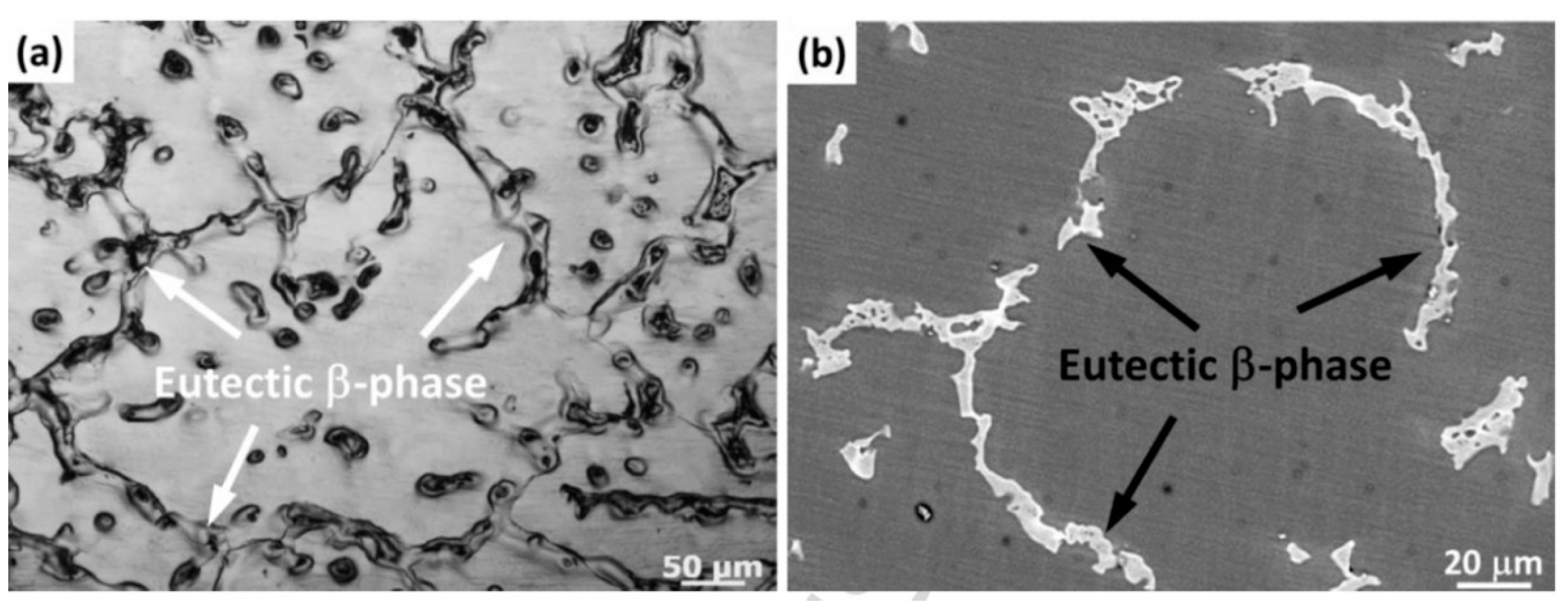
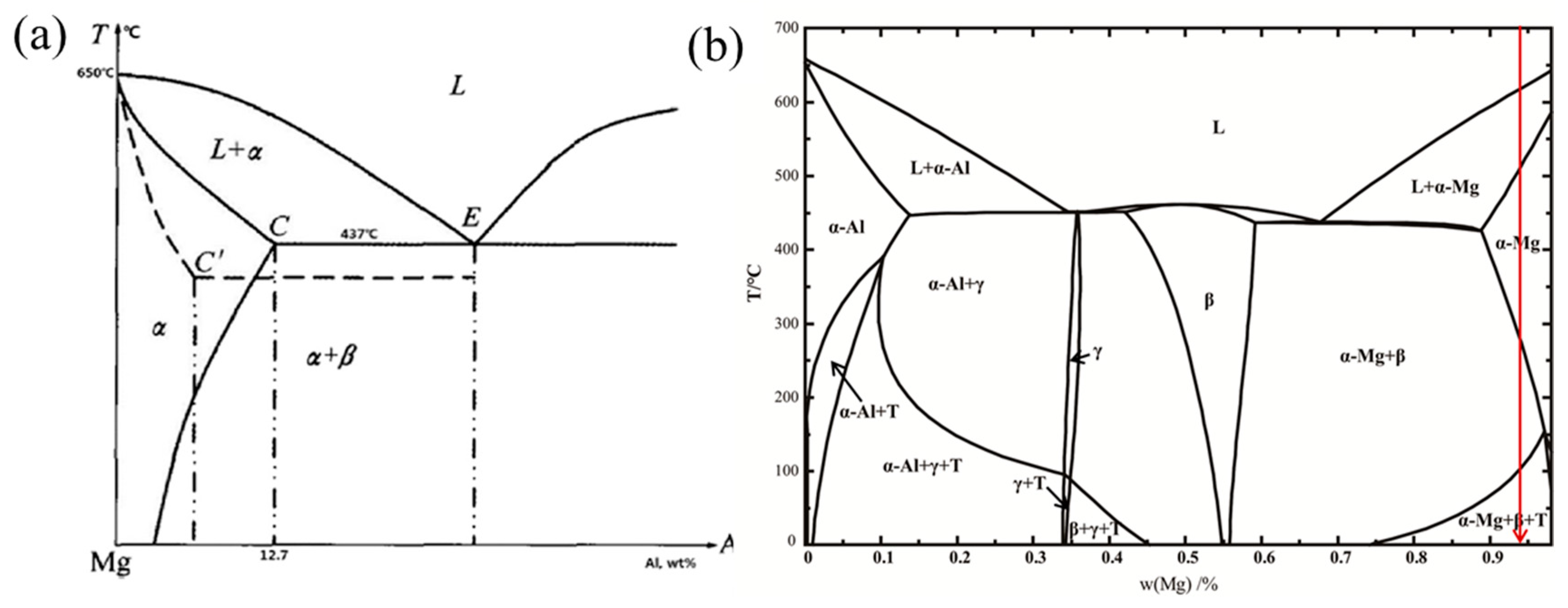
4.1.1. Alloying Element: Al
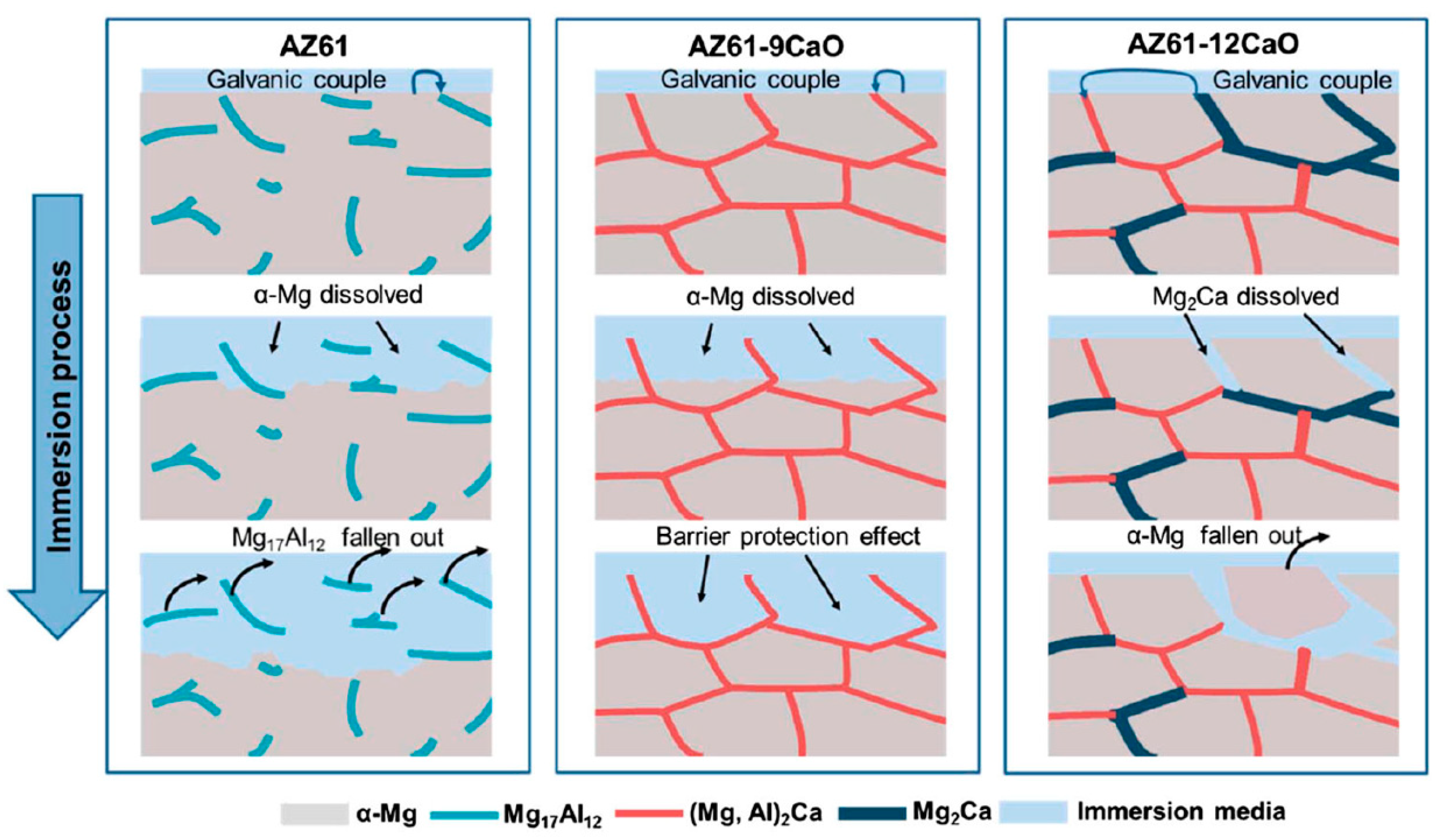
4.1.2. Alloying Element: Ca
4.1.3. Alloying Element: Sn
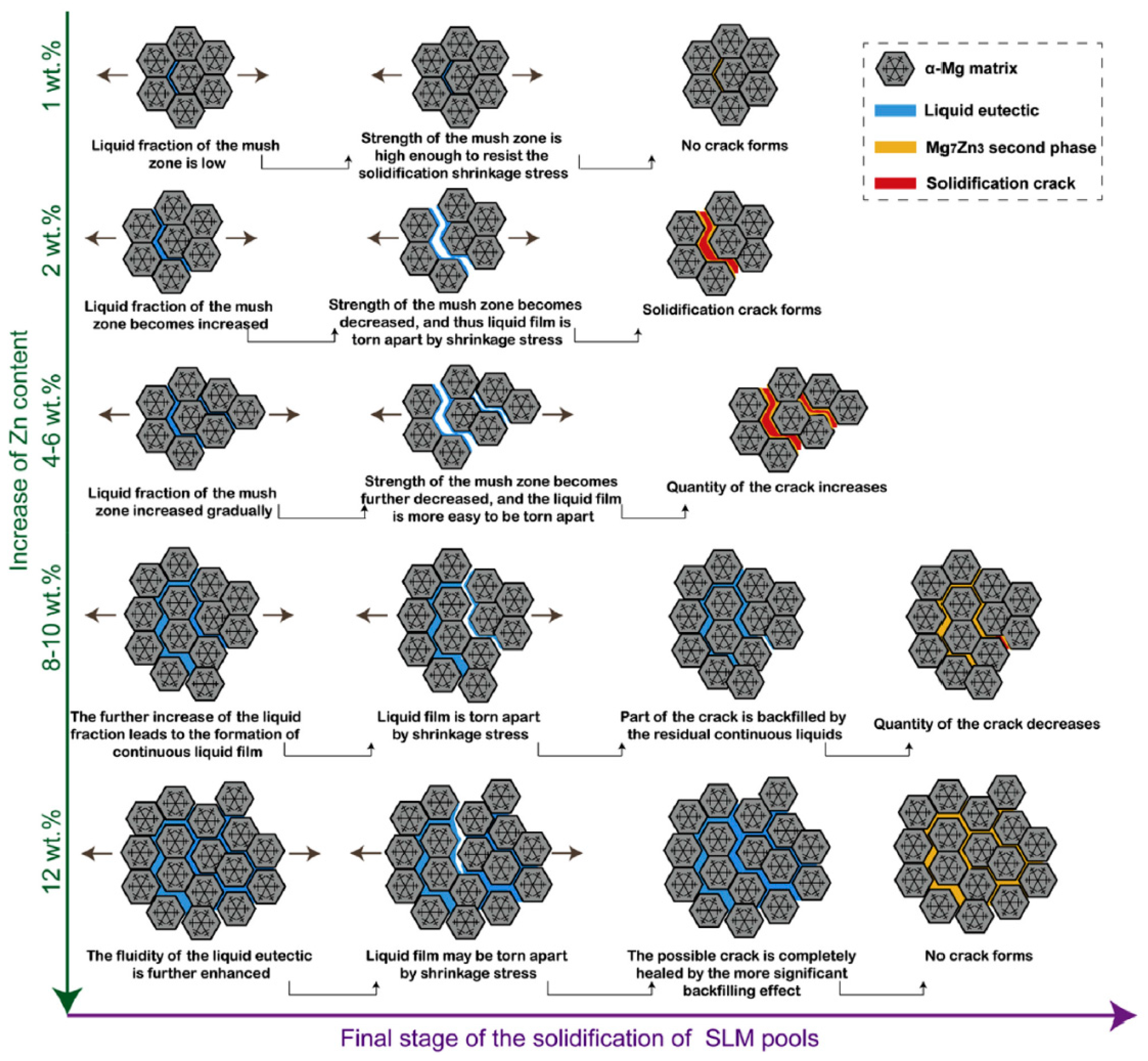
4.1.4. Alloying Element: Zn
4.1.5. Other Regular Elements
4.2. Rare Earth Elements
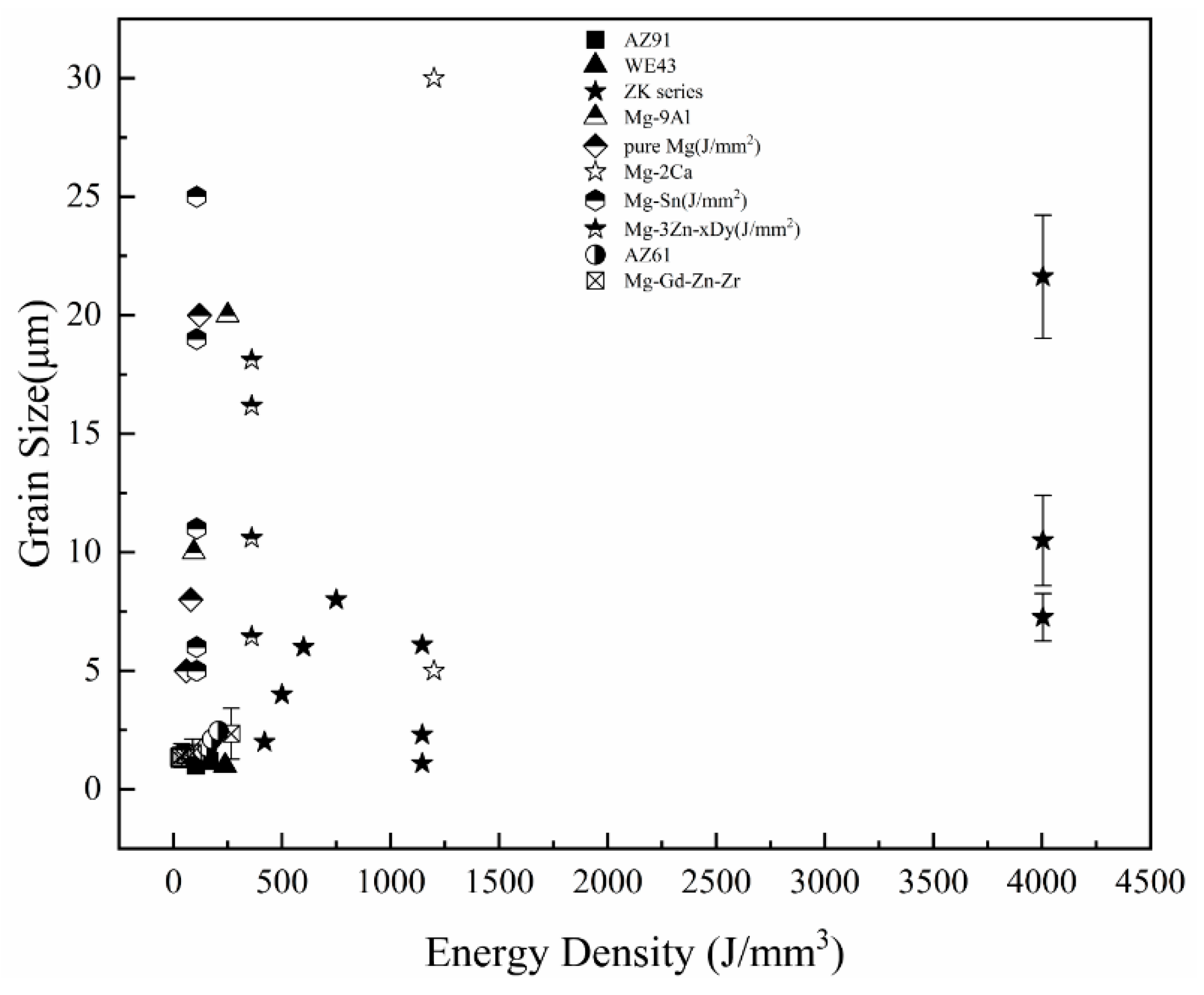
5. Microstructure of SLMed Magnesium Alloy
6. Effect of Heat Treatment on SLMed Magnesium Alloy
6.1. HIP
6.2. Heat Treatment
7. Outlook
8. Conclusions
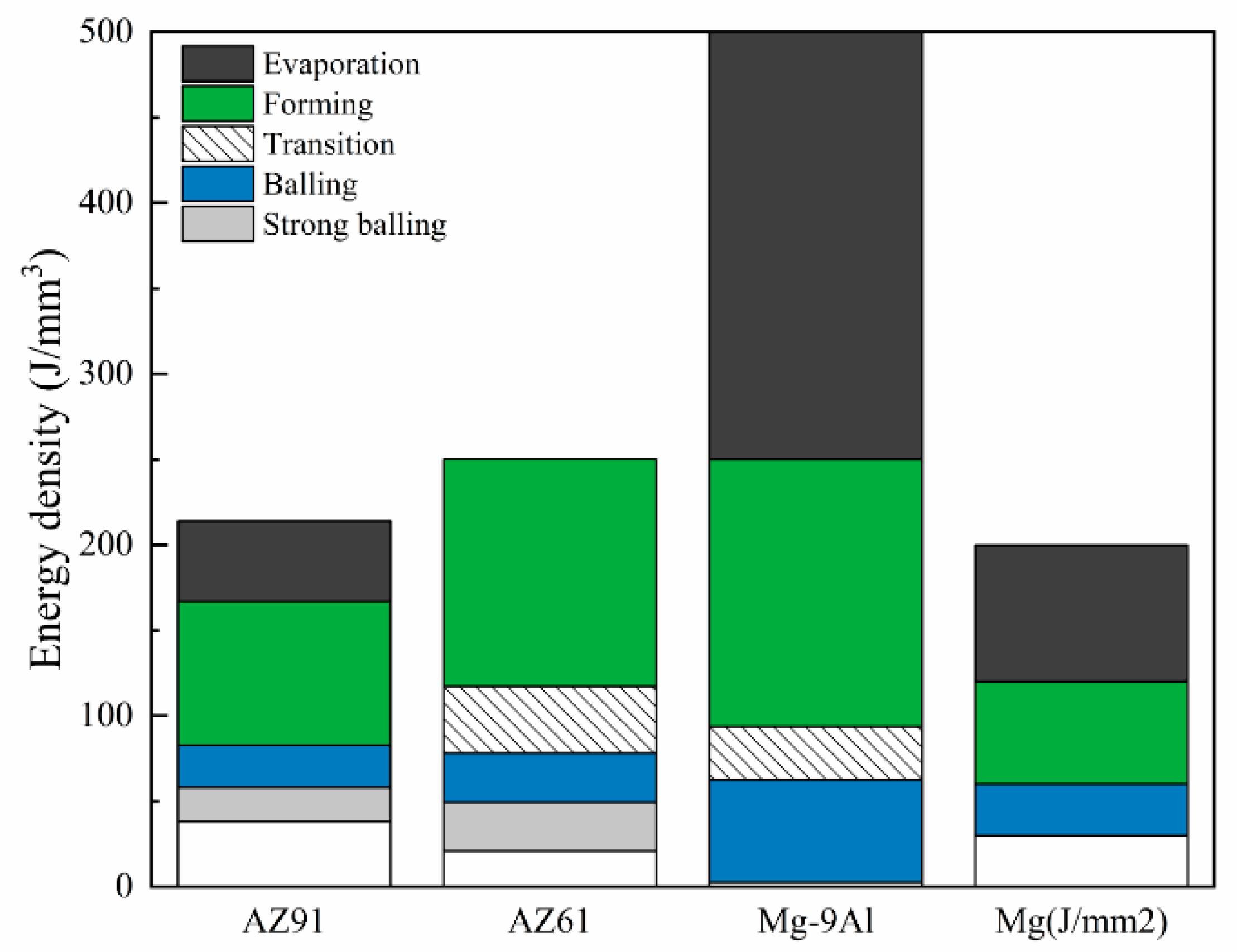
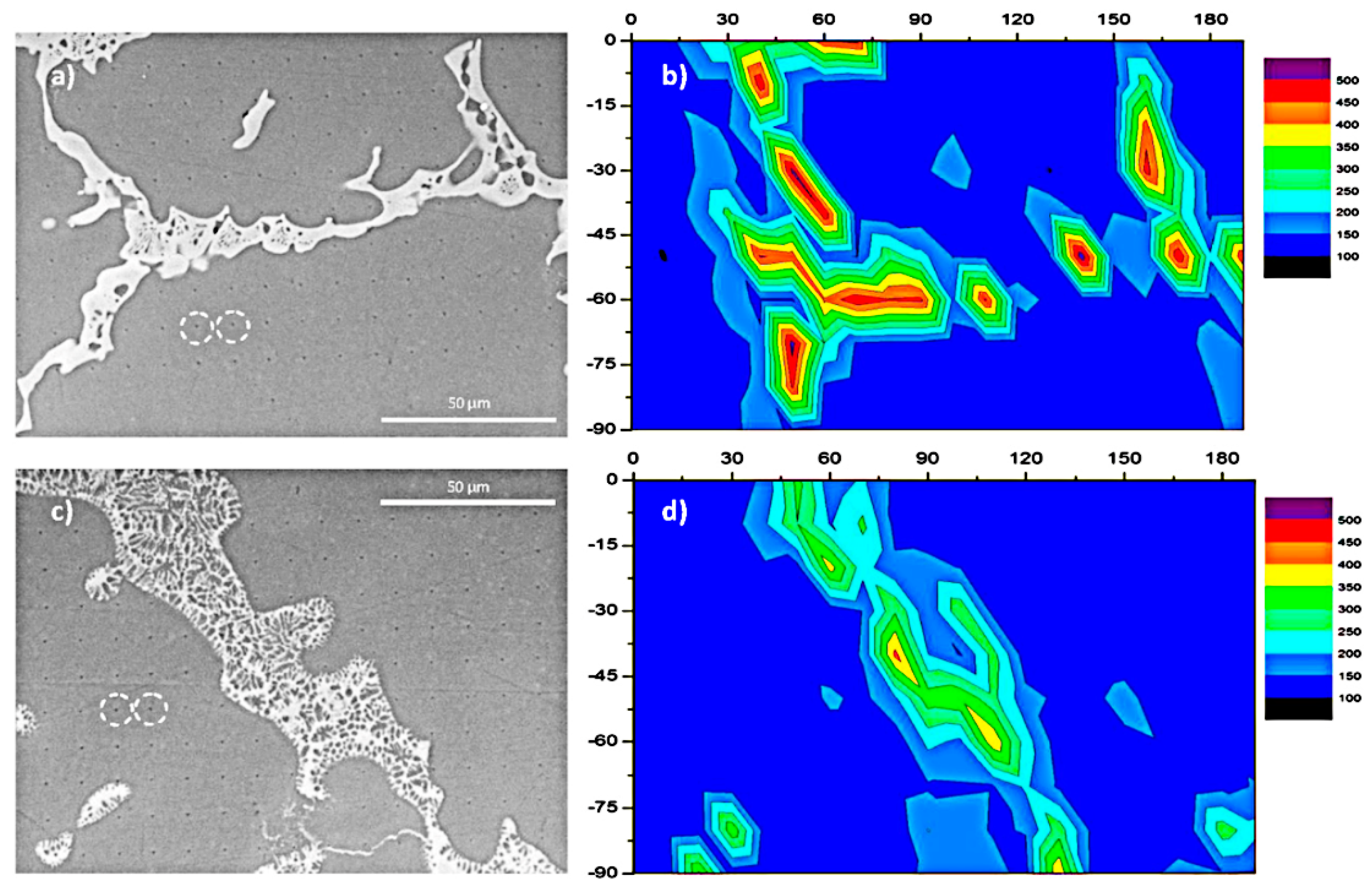
- With respect to immature process conditions and metallurgical defects, the influence of processing parameters (scanning speed and laser power) on the forming and performance of magnesium alloys cannot be ignored. The relative density of magnesium alloys is closely related to the processing parameters. If the scanning speed is too high or laser power is to low (energy density is too low), the powder cannot be fully melted, and the system is in a state of solid–liquid coexistence. In this state, the surface tension and viscosity of the liquid increase, which inhibits the liquid from flowing smoothly and causing the liquid to agglomerate into spheres and pores, thereby preventing the sample from becoming dense. If the energy density is too high, it will lead to the loss of alloying elements due to the evaporation of elements in the powder. On the other hand, under these conditions, the solute capture effect will be weakened, and the decrease in the solid solution content will cause the relative density of the SLMed sample to decrease. Therefore, the preparation of dense SLMed magnesium alloy requires the energy density to be controlled in a suitable range to enhance the solid solution strengthening and reduce the liquid surface tension, thereby eliminating the pores and spherical particles between the tracks. However, in the SLM process of magnesium alloys, it is difficult to remove the pores by only adjusting the process conditions. Post-treatment methods such as HIP are required to remove these defects. Microcracks are commonly attributed to solidification cracking and liquation cracking. The cause of solidification cracking is a residual thin film of liquid phase between the primary crystallized grains, and liquation cracking is caused by cyclic heat input, which occurs in multilayer welding and layered fabrication.
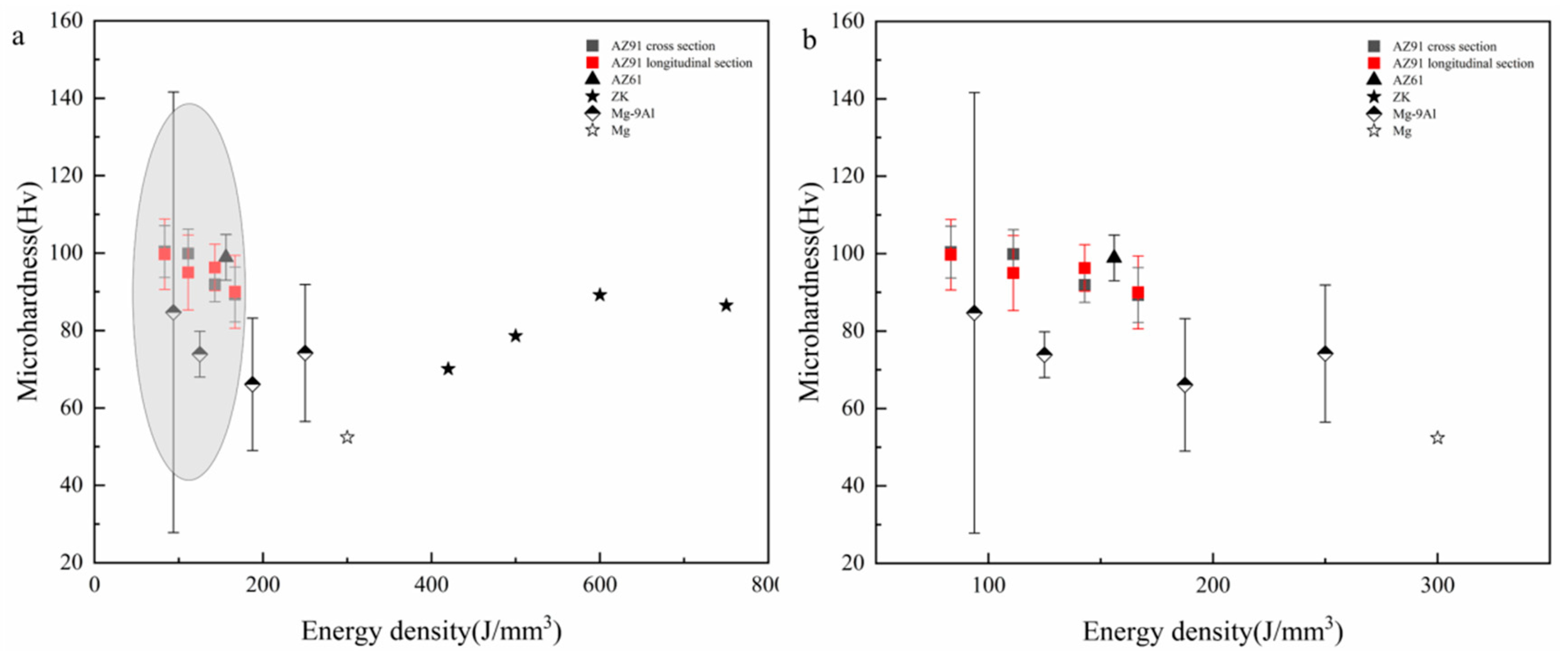
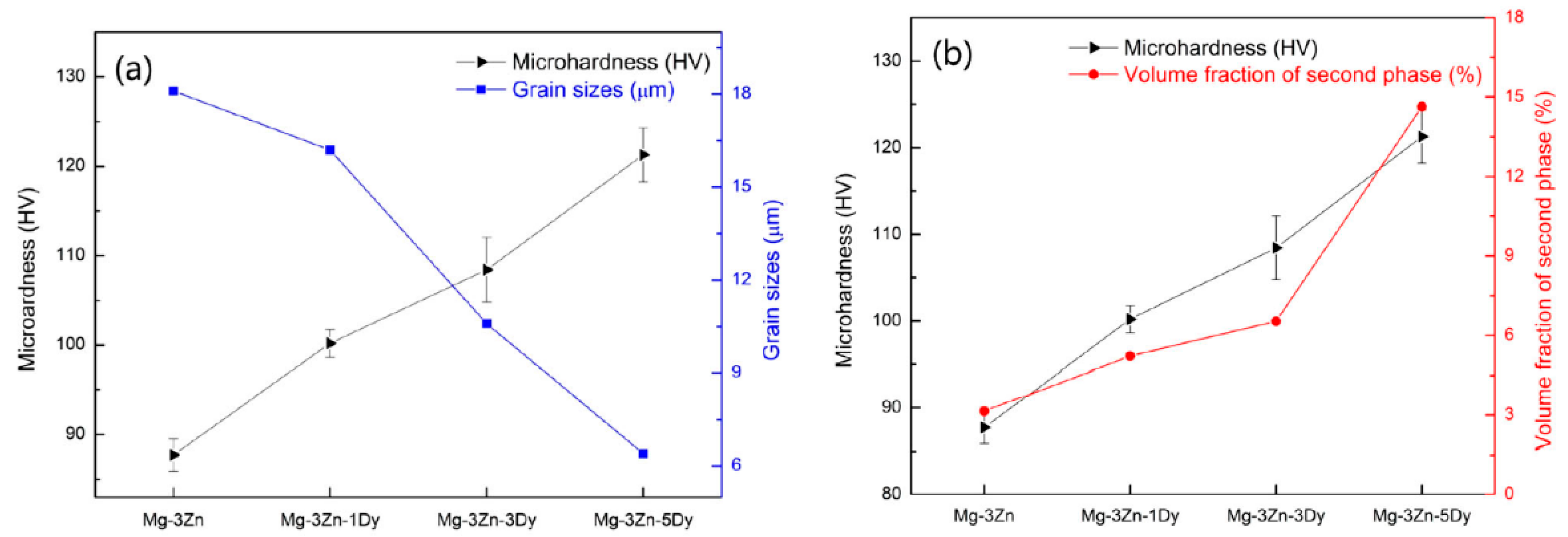
- The microhardness of the SLMed magnesium alloy is affected by the rapid solidification characteristics in the SLM process, which mainly affect the microstructure and the solid solution of the elements. On the one hand, the rapid solidification characteristics remarkably refine the microstructure of magnesium alloys, and the microhardness of SLMed magnesium alloys is notably higher than that of traditional as-cast magnesium alloys. On the other hand, the solid solution content of the alloying elements is different due to the differences in the solute trapping effects at different energy densities during the SLM process; thus, the strengthening effects are different. Moreover, the distribution and content of the second phase and the presence of defects will also affect the microhardness. Microhardness does not change monotonically with respect to the energy density. It is necessary to comprehensively consider the interaction between the solute capture effect at different molten pool temperatures and the solid solution of the elements under rapid solidification. Hence, while the microstructure is refined, the optimal solid solution is achieved, and the microhardness of the magnesium alloy is improved.
- The grain size of magnesium alloys is increased by increasing the energy input during the SLM process, and the mechanical properties are affected by the grain size and microstructure. In SLMed magnesium alloys, attention should be paid to the dual influence of the second phase on the material properties. The content of the second phase needs to be controlled to balance the strengthening of the second phase and its limitation on plasticity. The mechanical properties and corrosion resistance of SLMed magnesium alloy can be improved by introducing alloying elements. At present, there are several alloy systems under investigation, and more alloying elements, such as rare earth elements and Mn, need to be introduced to develop different SLMed magnesium alloy systems. The micropores and the second phase generated during the SLM process have certain restrictions on the application of the material. Adjusting the process conditions can reduce the harm of the pores and the second phase to a certain extent, but it cannot be completely avoided. Post-treatments, such as HIP and heat treatment, can be applied to help eliminate inherent porosity and improve the precipitation of the second phase. However, much remains to be done in this area.
- In view of the lack of processable materials, adding alloying elements and post-treatment is an effective way to improve SLMed magnesium alloy. These two methods have played an important role in improving the mechanical properties and corrosion resistance of SLMed magnesium alloy, especially the problem of poor plasticity of SLMed magnesium alloy. In the future, these two aspects need to be further studied to design magnesium alloy materials that are more suitable for SLM process applications.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xu, T.; Yang, Y.; Peng, X.; Song, J.; Pan, F. Overview of advancement and development trend on magnesium alloy. J. Magnes. Alloys 2019, 7, 536–544. [Google Scholar] [CrossRef]
- Liu, W.; Zhou, B.; Wu, G.; Zhang, L.; Peng, X.; Cao, L. High temperature mechanical behavior of low-pressure sand-cast Mg–Gd–Y–Zr magnesium alloy. J. Magnes. Alloys 2019, 7, 597–604. [Google Scholar] [CrossRef]
- Ali, Y.; Qiu, D.; Jiang, B.; Pan, F.; Zhang, M.-X. Current research progress in grain refinement of cast magnesium alloys: A review article. J. Alloys Compd. 2015, 619, 639–651. [Google Scholar] [CrossRef]
- Luo, K.; Zhang, L.; Wu, G.; Liu, W.; Ding, W. Effect of Y and Gd content on the microstructure and mechanical properties of Mg–Y–RE alloys. J. Magnes. Alloys 2019, 7, 345–354. [Google Scholar] [CrossRef]
- Yeganeh, M.; Mohammadi, N. Superhydrophobic surface of Mg alloys: A review. J. Magnes. Alloys 2018, 6, 59–70. [Google Scholar] [CrossRef]
- Sanders, P.G.; Keske, J.S.; Leong, K.H.; Kornecki, G. High power Nd:YAG and CO2 laser welding of magnesium. J. Laser Appl. 1999, 11, 96–103. [Google Scholar] [CrossRef]
- Zhou, Y.; Gui, Q.; Yu, W.; Liao, S.; He, Y.; Tao, X.; Yu, Y.; Wang, Y. Interfacial diffusion printing: An efficient manufacturing technique for artificial tubular grafts. ACS Biomater. Sci. Eng. 2019, 5, 6311–6318. [Google Scholar] [CrossRef]
- Du, J.; Lan, Z.; Zhang, H.; Lü, S.; Liu, H.; Guo, J. Catalytic enhanced hydrogen storage properties of Mg-based alloy by the addition of reduced graphene oxide supported V2O3 nanocomposite. J. Alloys Compd. 2019, 802, 660–667. [Google Scholar] [CrossRef]
- Ning, H.; Zhou, X.; Zhang, Z.; Zhou, W.; Guo, J. Ni catalytic effects for the enhanced hydrogenation properties of Mg17Al12(1 1 0) surface. Appl. Surf. Sci. 2019, 464, 644–650. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, Y.; Mai, S.; Wang, D.; Song, C. Investigation into spatter behavior during selective laser melting of AISI 316L stainless steel powder. Mater. Des. 2015, 87, 797–806. [Google Scholar] [CrossRef]
- Sing, S.L.; An, J.; Yeong, W.Y.; Wiria, F.E. Laser and electron-beam powder-bed additive manufacturing of metallic implants: A review on processes, materials and designs. J. Orthop. Res. 2015, 34, 369–385. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, J.; Yan, Q.; Zhang, L.; Wang, M.; Song, B.; Shi, Y. Amorphous alloy strengthened stainless steel manufactured by selective laser melting: Enhanced strength and improved corrosion resistance. Scr. Mater. 2018, 148, 20–23. [Google Scholar] [CrossRef]
- Gokuldoss, P.K.; Eckert, J.; Gokuldoss, P.K. Formation of metastable cellular microstructures in selective laser melted alloys. J. Alloys Compd. 2017, 707, 27–34. [Google Scholar]
- Kürnsteiner, P.; Wilms, M.B.; Weisheit, A.; Barriobero-Vila, P.; Jägle, E.; Raabe, D. Massive nanoprecipitation in an Fe-19Ni- X Al maraging steel triggered by the intrinsic heat treatment during laser metal deposition. Acta Mater. 2017, 129, 52–60. [Google Scholar] [CrossRef]
- Olakanmi, E.O.; Cochrane, R.; Dalgarno, K. A review on selective laser sintering/melting (SLS/SLM) of aluminium alloy powders: Processing, microstructure, and properties. Prog. Mater. Sci. 2015, 74, 401–477. [Google Scholar] [CrossRef]
- Manakari, V.; Parande, G.; Gupta, M. Selective laser melting of magnesium and magnesium alloy powders: A review. Metals 2016, 7, 2. [Google Scholar] [CrossRef]
- Cao, X.; Jahazi, M.; Immarigeon, J.; Wallace, W. A review of laser welding techniques for magnesium alloys. J. Mater. Process. Technol. 2006, 171, 188–204. [Google Scholar] [CrossRef]
- Jahangir, N.; Mamun, M.A.H.; Sealy, M.P. A review of additive manufacturing of magnesium alloys. In Proceedings of the 3rd International Conference on Mechanical Engineering (ICOME 2017), Birmingham, UK, 13–15 October 2017. [Google Scholar]
- Makarov, D.; Melzer, M.; Karnaushenko, D.; Schmidt, O.G. Review of selective laser melting: Materials and applications. Appl. Phys. Rev. 2016, 3, 011101. [Google Scholar] [CrossRef]
- Zhang, J.; Song, B.; Wei, Q.; Bourell, D.; Shi, Y. A review of selective laser melting of aluminum alloys: Processing, microstructure, property and developing trends. J. Mater. Sci. Technol. 2019, 35, 270–284. [Google Scholar] [CrossRef]
- Zhang, W.-N.; Wang, L.-Z.; Feng, Z.-X.; Chen, Y.-M. Research progress on selective laser melting (SLM) of magnesium alloys: A review. Optik 2020, 207, 163842. [Google Scholar] [CrossRef]
- Abe, F.; Santos, E.C.; Kitamura, Y.; Osakada, K.; Shiomi, M. Influence of forming conditions on the titanium model in rapid prototyping with the selective laser melting process. J. Mech. Eng. Sci. 2003, 217, 119–126. [Google Scholar] [CrossRef]
- Meier, H.; Haberland, C. Experimental studies on selective laser melting of metallic parts. Mater. Werkst. 2008, 39, 665–670. [Google Scholar] [CrossRef]
- Song, B.; Dong, S.; Zhang, B.; Liao, H.; Coddet, C. Effects of processing parameters on microstructure and mechanical property of selective laser melted Ti6Al4V. Mater. Des. 2012, 35, 120–125. [Google Scholar] [CrossRef]
- Lucas, J.; Meiners, W.; Vervoort, S.; Gayer, C.; Zumdick, N.A.; Zander, D. Selective laser melting of magnesium alloys. Eur. Cells Mater. 2015, 30, 1. [Google Scholar]
- Wei, K.; Gao, M.; Wang, Z.; Zeng, X. Effect of energy input on formability, microstructure and mechanical properties of selective laser melted AZ91D magnesium alloy. Mater. Sci. Eng. A 2014, 611, 212–222. [Google Scholar] [CrossRef]
- Wei, K.; Wang, Z.; Zeng, X. Influence of element vaporization on formability, composition, microstructure, and mechanical performance of the selective laser melted Mg–Zn–Zr components. Mater. Lett. 2015, 156, 187–190. [Google Scholar] [CrossRef]
- Liu, S.; Yang, W.; Shi, X.; Li, B.; Duan, S.; Guo, H.-J.; Guo, J. Influence of laser process parameters on the densification, microstructure, and mechanical properties of a selective Laser melted AZ61 magnesium alloy. J. Alloys Compd. 2019, 808, 151160. [Google Scholar] [CrossRef]
- He, C.; Bin, S.; Wu, P.; Gao, C.; Feng, P.; Shuai, C.; Liu, L.; Zhou, Y.; Zhao, M.; Yang, S.; et al. Microstructure evolution and biodegradation behavior of laser rapid solidified Mg–Al–Zn alloy. Metals 2017, 7, 105. [Google Scholar] [CrossRef]
- Shuai, C.; Shuai, C.; Wu, P.; Lin, X.; Liu, Y.; Zhou, Y.; Feng, P.; Liu, X.; Peng, S. Laser rapid solidification improves corrosion behavior of Mg-Zn-Zr Alloy. J. Alloys Compd. 2017, 691, 961–969. [Google Scholar] [CrossRef]
- Shuai, C.; He, C.; Feng, P.; Guo, W.; Gao, C.; Wu, P.; Yang, Y.; Bin, S. Biodegradation mechanisms of selective laser-melted Mg–xAl–Zn alloy: Grain size and intermetallic phase. Virtual Phys. Prototyp. 2017, 13, 59–69. [Google Scholar] [CrossRef]
- Zhang, B.; Liao, H.; Coddet, C. Effects of processing parameters on properties of selective laser melting Mg–9%Al powder mixture. Mater. Des. 2012, 34, 753–758. [Google Scholar] [CrossRef]
- Hu, N.; Wang, Y.; Zhang, D.; Hao, L.; Jiang, J.; Li, Z.; Chen, Y. Experimental investigation on selective laser melting of bulk net-shape pure magnesium. Mater. Manuf. Process. 2015, 30, 1298–1304. [Google Scholar] [CrossRef]
- Ng, C.C.; Savalani, M.M.; Man, H.; Gibson, I. Layer manufacturing of magnesium and its alloy structures for future applications. Virtual Phys. Prototyp. 2010, 5, 13–19. [Google Scholar] [CrossRef]
- Ng, C.C.; Savalani, M.; Lau, M.; Man, H. Microstructure and mechanical properties of selective laser melted magnesium. Appl. Surf. Sci. 2011, 257, 7447–7454. [Google Scholar] [CrossRef]
- Savalani, M.M.; Pizarro, J.M. Effect of preheat and layer thickness on selective laser melting (SLM) of magnesium. Rapid Prototyp. J. 2016, 22, 115–122. [Google Scholar] [CrossRef]
- Yang, Y.; Wu, P.; Lin, X.; Liu, Y.; Bian, H.; Zhou, Y.; Gao, C.; Shuai, C. System development, formability quality and microstructure evolution of selective laser-melted magnesium. Virtual Phys. Prototyp. 2016, 11, 173–181. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, M.; Chen, C. Effect of laser processing parameters on porosity, microstructure and mechanical properties of porous Mg-Ca alloys produced by laser additive manufacturing. Mater. Sci. Eng. A 2017, 703, 359–371. [Google Scholar] [CrossRef]
- Zhou, Y.; Wu, P.; Shuai, C.; Gao, D.; Feng, P.; Gao, C.; Wu, H.; Liu, Y.; Bian, H.; Shuai, C. The microstructure, mechanical properties and degradation behavior of laser-melted Mg Sn alloys. J. Alloys Compd. 2016, 687, 109–114. [Google Scholar] [CrossRef]
- Long, T.; Zhang, X.; Huang, Q.; Liu, L.; Liu, Y.; Ren, J.; Yin, Y.; Wu, D.; Wu, H. Novel Mg-based alloys by selective laser melting for biomedical applications: Microstructure evolution, microhardness and in vitro degradation behaviour. Virtual Phys. Prototyp. 2017, 13, 71–81. [Google Scholar] [CrossRef]
- Liu, S.; Guo, H.-J. Influence of hot isostatic pressing (HIP) on mechanical properties of magnesium alloy produced by selective laser melting (SLM). Mater. Lett. 2020, 265, 127463. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, C.; Liu, C.; Wang, S. Study on porous Mg-Zn-Zr ZK61 alloys produced by laser additive manufacturing. Metals 2018, 8, 635. [Google Scholar] [CrossRef]
- Wei, K.; Zeng, X.-Y.; Wang, Z.; Deng, J.; Liu, M.; Huang, G.; Yuan, X. Selective laser melting of Mg-Zn binary alloys: Effects of Zn content on densification behavior, microstructure, and mechanical property. Mater. Sci. Eng. A 2019, 756, 226–236. [Google Scholar] [CrossRef]
- Deng, Q.; Wu, Y.; Luo, Y.; Su, N.; Xue, X.; Chang, Z.; Wu, Q.; Xue, Y.; Peng, L. Fabrication of high-strength Mg-Gd-Zn-Zr alloy via selective laser melting. Mater. Charact. 2020, 165, 110377. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, J.; Pavanram, P.; Leeflang, M.; Fockaert, L.; Pouran, B.; Tümer, N.; Schröder, K.-U.; Mol, J.M.C.; Weinans, H.; et al. Additively manufactured biodegradable porous magnesium. Acta Biomater. 2018, 67, 378–392. [Google Scholar] [CrossRef]
- Shuai, C.; Liu, L.; Zhao, M.; Feng, P.; Shuai, C.; Guo, W.; Gao, C.; Yuan, F. Microstructure, biodegradation, antibacterial and mechanical properties of ZK60-Cu alloys prepared by selective Laser melting technique. J. Mater. Sci. Technol. 2018, 34, 1944–1952. [Google Scholar] [CrossRef]
- Li, R.; Liu, J.; Shi, Y.; Wang, L.; Jiang, W. Balling behavior of stainless steel and nickel powder during selective laser Melting process. Int. J. Adv. Manuf. Technol. 2011, 59, 1025–1035. [Google Scholar] [CrossRef]
- Tolochko, N.K.; Mozzharov, S.E.; Yadroitsev, I.A.; Laoui, T.; Froyen, L.; Titov, V.I.; Ignatiev, M.B. Balling processes during selective laser treatment of powders. Rapid Prototyp. J. 2004, 10, 78–87. [Google Scholar] [CrossRef]
- Gu, D.; Shen, Y. Balling phenomena in direct laser sintering of stainless steel powder: Metallurgical mechanisms and control methods. Mater. Des. 2009, 30, 2903–2910. [Google Scholar] [CrossRef]
- Aboulkhair, N.T.; Everitt, N.M.; Ashcroft, I.; Tuck, C.; Tuck, C. Reducing porosity in AlSi10Mg parts processed by selective laser melting. Addit. Manuf. 2014, 1, 77–86. [Google Scholar] [CrossRef]
- Gu, D.; Hagedorn, Y.-C.; Meiners, W.; Meng, G.; Batista, R.J.S.; Wissenbach, K.; Poprawe, R. Densification behavior, microstructure evolution, and wear performance of selective laser melting processed commercially pure titanium. Acta Mater. 2012, 60, 3849–3860. [Google Scholar] [CrossRef]
- Yadroitsev, I.; Gusarov, A.V.; Yadroitsava, I.; Smurov, I.Y. Single track formation in selective laser melting of metal powders. J. Mater. Process. Technol. 2010, 210, 1624–1631. [Google Scholar] [CrossRef]
- Khorasani, A.; Gibson, I.; Awan, U.S.; Ghaderi, A. The effect of SLM process parameters on density, hardness, tensile strength and surface quality of Ti-6Al-4V. Addit. Manuf. 2019, 25, 176–186. [Google Scholar] [CrossRef]
- Xu, W.; Lui, E.W.; Pateras, A.; Qian, M.; Brandt, M. In situ tailoring microstructure in additively manufactured Ti-6Al-4V for superior mechanical performance. Acta Mater. 2017, 125, 390–400. [Google Scholar] [CrossRef]
- Xia, M.; Gu, D.; Yu, G.; Dai, D.; Chen, H.; Shi, Q. Influence of hatch spacing on heat and mass transfer, thermodynamics and laser processability during additive manufacturing of inconel 718 alloy. Int. J. Mach. Tools Manuf. 2016, 109, 147–157. [Google Scholar] [CrossRef]
- Yadroitsev, I.; Bertrand, P.; Smurov, I.; Bertrand, P. Parametric analysis of the selective laser melting process. Appl. Surf. Sci. 2007, 253, 8064–8069. [Google Scholar] [CrossRef]
- Rashid, R.A.R.; Masood, S.; Ruan, D.; Palanisamy, S.; Brandt, M. Effect of scan strategy on density and metallurgical properties of 17-4PH parts printed by selective laser melting (SLM). J. Mater. Process. Technol. 2017, 249, 502–511. [Google Scholar] [CrossRef]
- Danilov, D.; Nestler, B. Phase-field modelling of solute trapping during rapid solidification of a Si–As alloy. Acta Mater. 2006, 54, 4659–4664. [Google Scholar] [CrossRef]
- Aziz, M.J.; Tsao, J.Y.; Thompson, M.; Peercy, P.S.; White, C.W. Solute trapping: Comparison of theory with experiment. Phys. Rev. Lett. 1986, 56, 2489–2492. [Google Scholar] [CrossRef]
- Risha, G.; Son, S.F.; Yetter, R.; Yang, V.; Tappan, B. Combustion of nano-aluminum and liquid water. Proc. Combust. Inst. 2007, 31, 2029–2036. [Google Scholar] [CrossRef]
- Chang, T.-C.; Wang, J.-Y.; Chu, C.-L.; Lee, S. Mechanical properties and microstructures of various Mg–Li alloys. Mater. Lett. 2006, 60, 3272–3276. [Google Scholar] [CrossRef]
- Taltavull, C.; Torres, B.; Lopez, A.J.; Rodrigo, P.; Otero, E.; Rams, J. Selective laser surface melting of a magnesium-aluminium alloy. Mater. Lett. 2012, 85, 98–101. [Google Scholar] [CrossRef]
- Suresh, C.H.; Koga, N. A consistent approach toward atomic radii. J. Phys. Chem. A 2001, 105, 5940–5944. [Google Scholar] [CrossRef]
- Wen, H.; Topping, T.D.; Isheim, D.; Seidman, D.N.; Lavernia, E.J. Strengthening mechanisms in a high-strength bulk nanostructured Cu–Zn–Al alloy processed via cryomilling and spark plasma sintering. Acta Mater. 2013, 61, 2769–2782. [Google Scholar] [CrossRef]
- Caceres, C.; Rovera, D. Solid solution strengthening in concentrated Mg–Al alloys. J. Light Met. 2001, 1, 151–156. [Google Scholar] [CrossRef]
- Yang, Y.; Peng, X.; Wen, H.; Zheng, B.; Zhou, Y.; Xie, W.; Lavernia, E. Influence of extrusion on the microstructure and mechanical behavior of Mg-9Li-3Al-XSr alloys. Met. Mater. Trans. A 2012, 44, 1101–1113. [Google Scholar] [CrossRef]
- Hakamada, M.; Furuta, T.; Chino, Y.; Chen, Y.; Kusuda, H.; Mabuchi, M. Life cycle inventory study on magnesium alloy substitution in vehicles. Energy 2007, 32, 1352–1360. [Google Scholar] [CrossRef]
- Kulekci, M.K. Magnesium and its alloys applications in automotive industry. Int. J. Adv. Manuf. Technol. 2007, 39, 851–865. [Google Scholar] [CrossRef]
- Teng, H.-T.; Li, T.-J.; Zhang, X.-L.; Zhang, Z.-T. Influence of sub-rapid solidification on microstructure and mechanical properties of AZ61A magnesium alloy. Trans. Nonferrous Met. Soc. China 2008, 18, s86–s90. [Google Scholar] [CrossRef]
- Casavola, C.; Campanelli, S.L.; Pappalettere, C. Preliminary investigation on distribution of residual stress generated by the selective Laser melting process. J. Strain Anal. Eng. Des. 2008, 44, 93–104. [Google Scholar] [CrossRef]
- Beausir, B.; Suwas, S.; Tóth, L.; Neale, K.W.; Fundenberger, J.-J. Analysis of texture Evolution in magnesium during equal channel angular extrusion. Acta Mater. 2008, 56, 200–214. [Google Scholar] [CrossRef]
- Teng, H.; Zhang, X.; Zhang, Z.; Li, T.; Cockcroft, S. Research on microstructures of sub-rapidly solidified AZ61 magnesium alloy. Mater. Charact. 2009, 60, 482–486. [Google Scholar] [CrossRef]
- Trevisan, F.; Calignano, F.; Lorusso, M.; Pakkanen, J.; Aversa, A.; Ambrosio, E.P.; Lombardi, M.; Fino, P.; Manfredi, D. On the selective laser melting (SLM) of the AlSi10Mg alloy: Process, microstructure, and mechanical properties. Materials 2017, 10, 76. [Google Scholar] [CrossRef]
- Xu, S.; Matsumoto, N.; Kamado, S.; Honma, T.; Kojima, Y. Effect of Mg17Al12 precipitates on the microstructural changes and mechanical properties of hot compressed AZ91 magnesium alloy. Mater. Sci. Eng. A 2009, 523, 47–52. [Google Scholar] [CrossRef]
- Lü, Y.; Wang, Q.; Ding, W.; Zeng, X.; Zhu, Y. Fracture behavior of AZ91 magnesium alloy. Mater. Lett. 2000, 44, 265–268. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, Q.; Zeng, X.; Ding, W.; Zhu, Y.; Lü, Y.Z.; Wang, Q.D.; Ding, W.J.; Zeng, X.Q.; Zhu, Y.P. Effects of silicon on microstructure, fluidity, mechanical properties, and fracture behaviour of Mg–6Al alloy. Mater. Sci. Technol. 2001, 17, 207–214. [Google Scholar]
- Song, J.-M.; Lui, T.; Chang, H.; Chen, L. The influence of Al content and annealing on vibration fracture properties of wrought Mg–Al–Zn alloys. Scr. Mater. 2006, 54, 399–404. [Google Scholar] [CrossRef]
- Deng, J.; Lin, Y.; Li, S.-S.; Chen, J.; Ding, Y. Hot tensile deformation and fracture behaviors of AZ31 magnesium alloy. Mater. Des. 2013, 49, 209–219. [Google Scholar] [CrossRef]
- Du, X.; Zhang, E. Microstructure and mechanical behaviour of semi-solid die-casting AZ91D magnesium alloy. Mater. Lett. 2007, 61, 2333–2337. [Google Scholar] [CrossRef]
- Dini, H.; Andersson, N.-E.; Jarfors, A.E. Effect of Mg17Al12 fraction on mechanical properties of Mg-9%Al-1%Zn Cast alloy. Metals 2016, 6, 251. [Google Scholar] [CrossRef]
- Shamsaei, N.; Yadollahi, A.; Bian, L.; Thompson, S.M. An overview of direct laser deposition for additive manufacturing; part II: Mechanical behavior, process parameter optimization and control. Addit. Manuf. 2015, 8, 12–35. [Google Scholar] [CrossRef]
- Liu, G.; Zhang, J.; Xi, G.; Zuo, R.; Liu, S. Designing Mg alloys with high ductility: Reducing the strength discrepancies between soft deformation modes and hard deformation modes. Acta Mater. 2017, 141, 1–9. [Google Scholar] [CrossRef]
- Witte, F. The history of biodegradable magnesium implants: A review. Acta Biomater. 2010, 6, 1680–1692. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Hotta, M.; Mori, Y. Improved corrosion resistance of a high-strength Mg–Al–Mn–Ca magnesium alloy Made by rapid solidification powder metallurgy. Mater. Sci. Eng. A 2012, 544, 10–20. [Google Scholar] [CrossRef]
- Cai, S.; Lei, T.; Li, N.; Feng, F. Effects of Zn on microstructure, mechanical properties and corrosion behavior of Mg–Zn alloys. Mater. Sci. Eng. C 2012, 32, 2570–2577. [Google Scholar] [CrossRef]
- Nam, N.D.; Kim, W.C.; Kim, J.-G.; Shin, K.; Jung, H. Effect of mischmetal on the corrosion properties of Mg–5Al alloy. Corros. Sci. 2009, 51, 2942–2949. [Google Scholar] [CrossRef]
- Song, G.; Atrens, A.; Dargusch, M. Influence of microstructure on the corrosion of diecast AZ91D. Corros. Sci. 1998, 41, 249–273. [Google Scholar] [CrossRef]
- Ambat, R.; Aung, N.N.; Zhou, W. Evaluation of microstructural effects on corrosion behaviour of AZ91D magnesium alloy. Corros. Sci. 2000, 42, 1433–1455. [Google Scholar] [CrossRef]
- Song, G.L.; Atrens, A. Corrosion mechanisms of magnesium alloys. Adv. Eng. Mater. 1999, 1, 11–33. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, D.; Ma, C.; Guo, S. Improving mechanical properties and corrosion resistance of Mg-6Zn-Mn magnesium alloy by rapid solidification. Mater. Lett. 2013, 92, 45–48. [Google Scholar] [CrossRef]
- Daloz, D.; Steinmetz, P.; Michot, G. Corrosion behavior of rapidly solidified magnesium-aluminum-zinc alloys. Corros. Sci. 1997, 53, 944–954. [Google Scholar] [CrossRef]
- Izumi, S.; Yamasaki, M.; Kawamura, Y. Relation between corrosion behavior and microstructure of Mg–Zn–Y alloys prepared by rapid solidification at various cooling rates. Corros. Sci. 2009, 51, 395–402. [Google Scholar] [CrossRef]
- Wang, Z.; Xie, M.; Li, Y.; Zhang, W.; Yang, C.; Kollo, L.; Eckert, J.; Prashanth, K.G. Premature failure of an additively manufactured material. NPG Asia Mater. 2020, 12, 1–10. [Google Scholar] [CrossRef]
- Song, J.F.; Pan, F.; Jiang, B.; Atrens, A.; Zhang, M.-X.; Lu, Y. A review on hot tearing of magnesium alloys. J. Magnes. Alloys 2016, 4, 151–172. [Google Scholar] [CrossRef]
- Otani, Y.; Sasaki, S. Effects of the addition of silicon to 7075 aluminum alloy on microstructure, mechanical properties, and selective laser melting processability. Mater. Sci. Eng. A 2020, 777, 139079. [Google Scholar] [CrossRef]
- Li, L.; Li, R.; Yuan, T.; Chen, C.; Zhang, Z.; Li, X. Microstructures and tensile properties of a selective laser melted Al–Zn–Mg–Cu (Al7075) alloy by Si and Zr microalloying. Mater. Sci. Eng. A 2020, 787, 139492. [Google Scholar] [CrossRef]
- Kimura, T.; Nakamoto, T.; Mizuno, M.; Araki, H. Effect of silicon content on densification, mechanical and thermal properties of Al-XSi binary alloys fabricated using selective laser melting. Mater. Sci. Eng. A 2017, 682, 593–602. [Google Scholar] [CrossRef]
- Brandl, E.; Heckenberger, U.; Holzinger, V.; Buchbinder, D. Additive manufactured AlSi10Mg samples using selective laser melting (SLM): Microstructure, high cycle fatigue, and fracture behavior. Mater. Des. 2012, 34, 159–169. [Google Scholar] [CrossRef]
- Kumari, S.S.; Pillai, R.; Rajan, T.; Pai, B. Effects of individual and combined additions of Be, Mn, Ca and Sr on the solidification behaviour, structure and mechanical properties of Al–7Si–0.3Mg–0.8Fe alloy. Mater. Sci. Eng. A 2007, 460, 561–573. [Google Scholar] [CrossRef]
- Abderrazak, K.; Ben Salem, W.; Mhiri, H.; Lepalec, G.; Autric, M. Modelling of CO2 laser welding of magnesium alloys. Opt. Laser Technol. 2008, 40, 581–588. [Google Scholar] [CrossRef]
- Louvis, E.; Fox, P.; Sutcliffe, C.J. Selective laser melting of aluminium components. J. Mater. Process. Technol. 2011, 211, 275–284. [Google Scholar] [CrossRef]
- Dong, Z.; Kang, H.; Xie, Y.; Chi, C.; Peng, X. Effect of powder oxygen content on microstructure and mechanical properties of a laser additively-manufactured 12CrNi2 alloy steel. Mater. Lett. 2019, 236, 214–217. [Google Scholar] [CrossRef]
- Cao, L.; Zeng, W.; Xie, Y.; Liang, J.; Zhang, D. Effect of powder oxidation on interparticle boundaries and mechanical properties of bulk Al prepared by spark plasma sintering of Al powder. Mater. Sci. Eng. A 2018, 742, 305–308. [Google Scholar] [CrossRef]
- Rao, G.A.; Srinivas, M.; Sarma, D. Effect of oxygen content of powder on microstructure and mechanical properties of hot isostatically pressed superalloy inconel 718. Mater. Sci. Eng. A 2006, 435, 84–99. [Google Scholar] [CrossRef]
- Lei, Z.; Liu, X.; Wu, Y.; Wang, H.; Jiang, S.; Wang, S.; Hui, X.; Wu, Y.; Gault, B.; Kontis, P.; et al. Enhanced strength and ductility in a high-entropy alloy via ordered oxygen complexes. Nature 2018, 563, 546–550. [Google Scholar] [CrossRef]
- Das, S. Physical aspects of process control in selective laser Sintering of metals. Adv. Eng. Mater. 2003, 5, 701–711. [Google Scholar] [CrossRef]
- Fintová, S.; Kunz, L. Fatigue properties of magnesium alloy AZ91 processed by severe plastic deformation. J. Mech. Behav. Biomed. Mater. 2015, 42, 219–228. [Google Scholar] [CrossRef]
- Alaneme, K.K.; Okotete, E.A. Enhancing plastic deformability of Mg and its alloys—A review of traditional and nascent developments. J. Magnes. Alloys 2017, 5, 460–475. [Google Scholar] [CrossRef]
- Tahreen, N.; Chen, D.; Nouri, M.; Li, D.Y. Influence of aluminum content on twinning and texture development of Cast Mg–Al–Zn alloy during compression. J. Alloys Compd. 2015, 623, 15–23. [Google Scholar] [CrossRef]
- Zhou, W.R.; Zheng, Y.F.; Leeflang, M.A.; Zhou, J. Mechanical property, biocorrosion and in vitro biocompatibility evaluations of Mg–Li–(Al)–(RE) alloys for future cardiovascular stent application. Acta Biomater. 2013, 9, 8488–8498. [Google Scholar] [CrossRef]
- Kirkland, N.; Lespagnol, J.; Birbilis, N.; Staiger, M. A survey of bio-corrosion rates of magnesium alloys. Corros. Sci. 2010, 52, 287–291. [Google Scholar] [CrossRef]
- Zhang, B.; Hou, Y.; Wang, X.; Wang, Y.; Geng, L. Mechanical properties, degradation performance and cytotoxicity of Mg–Zn–Ca biomedical alloys with different compositions. Mater. Sci. Eng. C 2011, 31, 1667–1673. [Google Scholar] [CrossRef]
- Lai, H.; Li, J.; Li, J.; Zhang, Y.; Xu, Y. Effects of Sr on the microstructure, mechanical properties and corrosion behavior of Mg-2Zn-XSr alloys. J. Mater. Sci. Mater. Electron. 2018, 29, 87. [Google Scholar] [CrossRef] [PubMed]
- Homayun, B.; Afshar, A. Microstructure, mechanical properties, corrosion behavior and cytotoxicity of Mg–Zn–Al–Ca alloys as biodegradable materials. J. Alloys Compd. 2014, 607, 1–10. [Google Scholar] [CrossRef]
- Kirkland, N.; Birbilis, N.; Staiger, M. Assessing the corrosion of biodegradable magnesium implants: A critical review of current methodologies and their limitations. Acta Biomater. 2012, 8, 925–936. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.-C.; Liu, M.; Song, G.; Atrens, A. Influence of the β-phase morphology on the corrosion of the Mg alloy AZ91. Corros. Sci. 2008, 50, 1939–1953. [Google Scholar] [CrossRef]
- Lu, Y.; Bradshaw, A.; Chiu, Y.; Jones, I. The role of β1′ precipitates in the bio-corrosion performance of Mg–3Zn in simulated body fluid. J. Alloys Compd. 2014, 614, 345–352. [Google Scholar] [CrossRef]
- Shuai, C.; He, C.; Xu, L.; Li, Q.; Chen, T.; Yang, Y.; Peng, S. Wrapping effect of secondary phases on the grains: Increased corrosion resistance of Mg–Al alloys. Virtual Phys. Prototyp. 2018, 13, 292–300. [Google Scholar] [CrossRef]
- Li, Y.; Wen, C.; Mushahary, D.; Sravanthi, R.; Harishankar, N.; Pande, G.; Hodgson, P. Mg–Zr–Sr Alloys as biodegradable implant materials. Acta Biomater. 2012, 8, 3177–3188. [Google Scholar] [CrossRef]
- Niknejad, S.; Liu, L.; Lee, M.-Y.; Esmaeili, S.; Zhou, N.Y. Resistance spot welding of AZ series magnesium alloys: Effects of aluminum content on microstructure and mechanical properties. Mater. Sci. Eng. A 2014, 618, 323–334. [Google Scholar] [CrossRef]
- Zhou, M.; Morisada, Y.; Fujii, H. Effect of Ca addition on the microstructure and the mechanical properties of asymmetric double-sided friction stir welded AZ61 magnesium alloy. J. Magnes. Alloys 2020, 8, 91–102. [Google Scholar] [CrossRef]
- Yang, J.; Peng, J.; Nyberg, E.A.; Pan, F. Effect of Ca addition on the corrosion behavior of Mg–Al–Mn alloy. Appl. Surf. Sci. 2016, 369, 92–100. [Google Scholar] [CrossRef]
- Kondori, B.; Mahmudi, R. Effect of Ca additions on the microstructure, thermal stability and mechanical properties of a cast AM60 magnesium alloy. Mater. Sci. Eng. A 2010, 527, 2014–2021. [Google Scholar] [CrossRef]
- Yuesheng, C.; Zhigang, G.; Kangle, C.; Fang, D. Microstructure and mechanical properties of extruded Mg-Sm-Ca alloys. Rare Met. Mater. Eng. 2016, 45, 287–291. [Google Scholar] [CrossRef]
- Xu, S.; Matsumoto, N.; Yamamoto, K.; Kamado, S.; Honma, T.; Kojima, Y. High temperature tensile properties of As-Cast Mg–Al–Ca alloys. Mater. Sci. Eng. A 2009, 509, 105–110. [Google Scholar] [CrossRef]
- Aljarrah, M.; Medraj, M. Thermodynamic modelling of the Mg–Ca, Mg–Sr, Ca–Sr and Mg–Ca–Sr systems using the modified quasichemical model. Calphad 2008, 32, 240–251. [Google Scholar] [CrossRef]
- Zhang, L.; Deng, K.-K.; Nie, K.; Xu, F.-J.; Su, K.; Liang, W. Microstructures and mechanical properties of Mg–Al–Ca alloys affected by Ca/Al ratio. Mater. Sci. Eng. A 2015, 636, 279–288. [Google Scholar] [CrossRef]
- Oh-Ishi, K.; Watanabe, R.; Mendis, C.L.; Hono, K. Age-hardening response of Mg–0.3at.%Ca alloys with different Zn contents. Mater. Sci. Eng. A 2009, 526, 177–184. [Google Scholar] [CrossRef]
- Xu, S.; Oh-Ishi, K.; Kamado, S.; Uchida, F.; Homma, T.; Hono, K. High-strength extruded Mg–Al–Ca–Mn alloy. Scr. Mater. 2011, 65, 269–272. [Google Scholar] [CrossRef]
- Mendis, C.L.; Oh-Ishi, K.; Ohkubo, T.; Hono, K. Precipitation of prismatic plates in Mg–0.3Ca alloys with In additions. Scr. Mater. 2011, 64, 137–140. [Google Scholar] [CrossRef]
- Kondoh, K.; Fujita, J.; Umeda, J.; Imai, H.; Enami, K.; Ohara, M.; Igarashi, T. Thermo-dynamic analysis on solid-state reduction of CaO particles dispersed in Mg–Al alloy. Mater. Chem. Phys. 2011, 129, 631–640. [Google Scholar] [CrossRef]
- Nam, N.D.; Bian, M.; Forsyth, M.; Seter, M.; Tan, M.Y.; Shin, K. Effect of calcium oxide on the corrosion behaviour of AZ91 magnesium alloy. Corros. Sci. 2012, 64, 263–271. [Google Scholar] [CrossRef]
- Baek, S.-M.; Kang, J.S.; Shin, H.-J.; Yim, C.D.; You, B.S.; Ha, H.-Y.; Park, S.S. Role of alloyed Y in improving the corrosion resistance of extruded Mg–Al–Ca-based alloy. Corros. Sci. 2017, 118, 227–232. [Google Scholar] [CrossRef]
- Zeng, R.; Qi, W.-C.; Cui, H.-Z.; Zhang, F.; Zeng, R.-C.; Han, E.-H. In vitro corrosion of as-extruded Mg–Ca alloys—The influence of Ca concentration. Corros. Sci. 2015, 96, 23–31. [Google Scholar] [CrossRef]
- Zhu, G.; Wang, L.; Zhou, H.; Wang, J.; Shen, Y.; Tu, P.; Zhu, H.; Liu, W.; Jin, P.; Zeng, X. Improving ductility of a Mg alloy via non-basal slip induced by Ca addition. Int. J. Plast. 2019, 120, 164–179. [Google Scholar] [CrossRef]
- Stanford, N.; Barnett, M. Solute strengthening of prismatic slip, basal slip and twinning in Mg and Mg–Zn binary alloys. Int. J. Plast. 2013, 47, 165–181. [Google Scholar] [CrossRef]
- Khosravani, A.; Fullwood, D.T.; Adams, B.L.; Rampton, T.M.; Miles, M.P.; Mishra, R.K. Nucleation and propagation of {101 2} twins in AZ31 magnesium alloy. Acta Mater. 2015, 100, 202–214. [Google Scholar] [CrossRef]
- Pan, H.; Yang, C.; Yang, Y.; Dai, Y.; Zhou, D.; Chai, L.; Huang, Q.; Yang, Q.; Liu, S.; Ren, Y.; et al. Ultra-fine Grain Size and Exceptionally High Strength in Dilute Mg–Ca Alloys Achieved by Conventional One-Step extrusion. Mater. Lett. 2019, 237, 65–68. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, Y.; Geng, L.; Lu, C. Effects of calcium on texture and mechanical properties of hot-extruded Mg–Zn–Ca alloys. Mater. Sci. Eng. A 2012, 539, 56–60. [Google Scholar] [CrossRef]
- Zeng, Z.; Bian, M.; Xu, S.; Davies, C.H.J.; Birbilis, N.; Nie, J. Effects of dilute additions of Zn and Ca on ductility of magnesium alloy sheet. Mater. Sci. Eng. A 2016, 674, 459–471. [Google Scholar] [CrossRef]
- Sandlöbes, S.; Friák, M.; Korte-Kerzel, S.; Pei, Z.; Neugebauer, J.; Raabe, D. A Rare-earth free magnesium alloy with improved intrinsic ductility. Sci. Rep. 2017, 7, 10458. [Google Scholar] [CrossRef]
- Kim, K.-H.; Hwang, J.H.; Jang, H.-S.; Jeon, J.B.; Kim, N.J.; Lee, B.-J. Dislocation binding as an origin for the improvement of room temperature ductility in Mg alloys. Mater. Sci. Eng. A 2018, 715, 266–275. [Google Scholar] [CrossRef]
- Yasi, J.A.; Hector, L.G.; Trinkle, D., Jr. Prediction of thermal cross-slip stress in magnesium alloys from a geometric interaction model. Acta Mater. 2012, 60, 2350–2358. [Google Scholar] [CrossRef]
- Wang, Q.; Li, C.; Zou, Y.; Wang, H.; Yi, T.; Huang, C. A highly selective fluorescence sensor for Tin (Sn4+) and its application in imaging live cells. Org. Biomol. Chem. 2012, 10, 6740. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Pan, F.; Zhao, S.; Pan, H.; Song, K.; Tang, A. Preparation and characterization of as-extruded Mg–Sn alloys for orthopedic applications. Mater. Des. 2015, 70, 60–67. [Google Scholar] [CrossRef]
- Poddar, P.; Kamaraj, A.; Murugesan, A.; Bagui, S.; Sahoo, K.L. Microstructural features of Mg-8%Sn alloy and its correlation with mechanical properties. J. Magnes. Alloys 2017, 5, 348–354. [Google Scholar] [CrossRef]
- Tapiero, H.; Tew, K.D. Trace elements in human physiology and pathology: Zinc and metallothioneins. Biomed. Pharmacother. 2003, 57, 399–411. [Google Scholar] [CrossRef]
- Yan, Y.; Cao, H.; Kang, Y.; Yu, K.; Xiao, T.; Luo, J.; Deng, Y.; Fang, H.; Xiong, H.; Dai, Y. Effects of Zn concentration and heat treatment on the microstructure, mechanical properties and corrosion behavior of As-extruded Mg-Zn alloys produced by powder metallurgy. J. Alloys Compd. 2017, 693, 1277–1289. [Google Scholar] [CrossRef]
- Boehlert, C.J.; Knittel, K. The microstructure, tensile properties, and creep behavior of Mg–Zn alloys containing 0–4.4wt.% Zn. Mater. Sci. Eng. A 2006, 417, 315–321. [Google Scholar] [CrossRef]
- Xie, G.; Ma, Z.; Geng, L. Effect of microstructural evolution on mechanical properties of friction stir welded ZK60 alloy. Mater. Sci. Eng. A 2008, 486, 49–55. [Google Scholar] [CrossRef]
- Zhu, S.; Liu, Z.; Qu, R.; Wang, L.; Li, Q.; Guan, S. Effect of rare earth and Mn elements on the corrosion behavior of extruded AZ61 system in 3.5 Wt% NaCl solution and salt spray test. J. Magnes. Alloys 2013, 1, 249–255. [Google Scholar] [CrossRef]
- Wu, Z.; Ahmad, R.; Yin, B.; Sandlöbes, S.; Curtin, W. Mechanistic origin and prediction of enhanced ductility in magnesium alloys. Science 2018, 359, 447–452. [Google Scholar] [CrossRef]
- Zhu, T.; Fu, P.; Peng, L.; Hu, X.; Zhu, S.; Ding, W. Effects of Mn addition on the microstructure and mechanical properties of cast Mg–9Al–2Sn (wt.%) alloy. J. Magnes. Alloys 2014, 2, 27–35. [Google Scholar] [CrossRef]
- Hort, N.; Huang, Y.; Fechner, D.; Störmer, M.; Blawert, C.; Witte, F.; Vogt, C.; Drücker, H.; Willumeit, R.; Kainer, K.U. Magnesium alloys as implant materials—Principles of property design for Mg–RE alloys. Acta Biomater. 2010, 6, 1714–1725. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, S.; Leng, Z.; Li, M.; Meng, J.; Wu, R. Microstructures and mechanical properties of heat-resistant HPDC Mg–4Al-based alloys containing cheap misch metal. Mater. Sci. Eng. A 2011, 528, 2670–2677. [Google Scholar] [CrossRef]
- Nie, J.; Muddle, B. Characterisation of strengthening precipitate phases in a Mg–Y–Nd alloy. Acta Mater. 2000, 48, 1691–1703. [Google Scholar] [CrossRef]
- Tao, W.; Zhang, M.; Zhongyi, N.; Bin, L. Influence of rare earth elements on microstructure and mechanical properties of Mg-Li alloys. J. Rare Earths 2006, 24, 797–800. [Google Scholar] [CrossRef]
- Fu, L.; Le, Q.; Hu, W.; Zhang, J.; Wang, J. Strengths and ductility enhanced by micro-alloying Sm/La/Ca to Mg–0.5Zn–0.2Mn alloy. J. Mater. Res. Technol. 2020, 9, 6834–6849. [Google Scholar] [CrossRef]
- Yamasaki, M.; Anan, T.; Yoshimoto, S.; Kawamura, Y. Mechanical properties of warm-extruded Mg–Zn–Gd alloy with coherent 14H long periodic stacking ordered structure precipitate. Scr. Mater. 2005, 53, 799–803. [Google Scholar] [CrossRef]
- Itoi, T.; Seimiya, T.; Kawamura, Y.; Hirohashi, M. Long period stacking structures observed in Mg97Zn1Y2 alloy. Scr. Mater. 2004, 51, 107–111. [Google Scholar] [CrossRef]
- Ku, C.-H.; Pioletti, D.P.; Browne, M.; Gregson, P.J. Effect of different Ti–6Al–4V surface treatments on osteoblasts behaviour. Biomaterials 2002, 23, 1447–1454. [Google Scholar]
- El-Rahman, S.S.A. Neuropathology of aluminum toxicity in rats (glutamate and GABA impairment). Pharmacol. Res. 2003, 47, 189–194. [Google Scholar] [CrossRef]
- Jiang, M.; Yan, H.; Chen, R. Microstructure, texture and mechanical properties in an As-Cast AZ61 Mg alloy during multi-directional impact forging and subsequent heat treatment. Mater. Des. 2015, 87, 891–900. [Google Scholar] [CrossRef]
- Dahle, A.K.; Lee, Y.C.; Nave, M.D.; Schaffer, P.L.; StJohn, D. Development of the As-cast microstructure in magnesium–aluminium alloys. J. Light Met. 2001, 1, 61–72. [Google Scholar] [CrossRef]
- Wen, Z.; Wu, C.; Dai, C.; Yang, F. Corrosion behaviors of Mg and its alloys with different Al contents in a modified simulated body fluid. J. Alloys Compd. 2009, 488, 392–399. [Google Scholar] [CrossRef]
- Chen, J.; Wei, J.; Yan, H.; Su, B.; Pan, X. Effects of cooling rate and pressure on microstructure and mechanical properties of sub-rapidly solidified Mg–Zn–Sn–Al–Ca alloy. Mater. Des. 2013, 45, 300–307. [Google Scholar] [CrossRef]
- Cai, J.; Ma, G.; Liu, Z.; Zhang, H.; Xu, M. Influence of rapid solidification on the microstructure of AZ91HP alloy. J. Alloys Compd. 2006, 422, 92–96. [Google Scholar] [CrossRef]
- Zhao, Y.-C.; Zhao, M.-C.; Xu, R.; Liu, L.; Tao, J.-X.; Gao, C.; Shuai, C.; Atrens, A. Formation and characteristic corrosion behavior of alternately lamellar arranged α and β in As-cast AZ91 Mg alloy. J. Alloys Compd. 2019, 770, 549–558. [Google Scholar] [CrossRef]
- Tradowsky, U.; White, J.; Ward, R.; Read, N.; Reimers, W.; Attallah, M.M. Selective laser melting of AlSi10Mg: Influence of post-processing on the microstructural and tensile properties development. Mater. Des. 2016, 105, 212–222. [Google Scholar] [CrossRef]
- Zhao, X.; Li, S.; Zhang, M.; Liu, Y.; Sercombe, T.B.; Wang, S.; Hao, Y.; Yang, R.; Murr, L.E. Comparison of the microstructures and mechanical properties of Ti–6Al–4V fabricated by selective laser melting and electron beam melting. Mater. Des. 2016, 95, 21–31. [Google Scholar] [CrossRef]
- Yan, X.; Lupoi, R.; Wu, H.; Ma, W.; Liu, M.; O’Donnell, G.; Yin, S. Effect of hot isostatic pressing (HIP) treatment on the compressive properties of Ti6Al4V lattice structure fabricated by selective laser melting. Mater. Lett. 2019, 255, 126537. [Google Scholar] [CrossRef]
- Kaplanskii, Y.; Sentyurina, Z.A.; Loginov, P.; Levashov, E.; Korotitskiy, A.; Travyanov, A.Y.; Petrovskii, P. Microstructure and mechanical properties of the (Fe,Ni)Al-based alloy produced by SLM and HIP of spherical composite powder. Mater. Sci. Eng. A 2019, 743, 567–580. [Google Scholar] [CrossRef]
- Spierings, A.; Dawson, K.; Dumitraschkewitz, P.; Pogatscher, S.; Wegener, K. Microstructure characterization of SLM-processed Al-Mg-Sc-Zr alloy in the heat treated and HIPed condition. Addit. Manuf. 2018, 20, 173–181. [Google Scholar] [CrossRef]
- Feng, H.; Liu, S.; Du, Y.; Lei, T.; Zeng, R.; Yuan, T. Effect of the second phases on corrosion behavior of the Mg-Al-Zn alloys. J. Alloys Compd. 2017, 695, 2330–2338. [Google Scholar] [CrossRef]
- Song, G.-L.; Bowles, A.L.; StJohn, D.H. Corrosion resistance of aged die cast magnesium alloy AZ91D. Mater. Sci. Eng. A 2004, 366, 74–86. [Google Scholar] [CrossRef]
- Dumpala, S.C. Influence of heat treatment on the machinability and corrosion behavior of AZ91 Mg alloy. J. Magnes. Alloy 2018, 6, 52–58. [Google Scholar]
- Wang, Y.; Liu, G.; Fan, Z. Microstructural Evolution of Rheo-Diecast AZ91D Magnesium Alloy During Heat Treatment. Acta Mater. 2006, 54, 689–699. [Google Scholar] [CrossRef]
- Zhao, D.; Wang, Z.; Zuo, M.; Geng, H. Effects of heat treatment on microstructure and mechanical properties of extruded AZ80 magnesium alloy. Mater. Des. 2014, 56, 589–593. [Google Scholar] [CrossRef]
- Jia, H.; Feng, X.; Yang, Y. Influence of solution treatment on microstructure, mechanical and corrosion properties of Mg-4Zn alloy. J. Magnes. Alloys 2015, 3, 247–252. [Google Scholar] [CrossRef][Green Version]
- Zhang, L. State of the art in the control of inclusions in tire cord steels—A review. Steel Res. Int. 2006, 77, 158–169. [Google Scholar] [CrossRef]
- Park, J.H.; Kang, Y. Inclusions in stainless steels—A review. Steel Res. Int. 2017, 88, 1700130. [Google Scholar] [CrossRef]
- Shi, C.; Chen, X.-C.; Guo, H.-J.; Zhu, Z.-J.; Sun, X.-L. Control of MgO·Al2O3 spinel inclusions during protective gas electroslag remelting of die steel. Met. Mater. Trans. A 2012, 44, 378–389. [Google Scholar] [CrossRef]
- Shi, C.; Zheng, D.; Guo, B.; Li, J.; Jiang, F. Evolution of oxide–sulfide complex inclusions and its correlation with steel cleanliness during electroslag rapid remelting (ESRR) of tool steel. Met. Mater. Trans. A 2018, 49, 3390–3402. [Google Scholar] [CrossRef]
- Yan, M.; Xu, W.; Dargusch, M.; Tang, H.P.; Brandt, M.; Qian, M. Review of effect of oxygen on room temperature ductility of titanium and titanium alloys. Powder Met. 2014, 57, 251–257. [Google Scholar] [CrossRef]





| Types | Powder Shape | Partical Size (μm) | Processing | Relative Density (%) | Ultimate Tensile Strength (UTS) (MPa) | Yield Strength (YS) (MPa) | Elongation (EL) (%) | Microhardness (Hv) | Laser Power (W) | Scanning Speed (mm/s) | Energy Density (J·mm−3) | Refs. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AZ91 | irregular | 25–63 | SLM | >99 | 329 ± 6 | 264 ± 1 | 3.7 ± 0.8 | — | 100 | 800 | 104 | [25] |
| WE43 | irregular | 25–63 | SLM | >99 | 307 ± 6 | 302 ± 3 | 11.9 ± 1 | — | 200 | 700 | 238 | [25] |
| AZ91D | spherical | 59 | SLM | 99.52 | 296 ± 2 | 254 ± 4 | 1.8 ± 0.2 | 100 | 200 | 333 | 167 | [26] |
| 60 | spherical | 30 | SLM | 94.05 | - | - | - | 78 ± 10 | 200 | 300 | 416 | [27] |
| AZ61 | spherical | 48 | SLM | 99.4 ± 0.3 | 287 ± 3 | 233 ± 2 | 3.1 ± 0.1 | — | 150 | 400 | 156 | [28] |
| AZ61 | spherical | 70 | SLM | 99 ± 5 | - | - | - | 93 ± 4 | 80 | 3 | 9609.6 | [29] |
| ZK60 | spherical | 50 | SLM | 97.4 ± 2 | - | - | - | 89.2 ± 5 | 50 | 8 | 600 | [30] |
| ZK30-xAl | spherical | ZK30: 45–74; Al: 5–15 | SLM | - | - | - | - | 75.7 ± 6 | 80 | 3 | 4004 | [31] |
| Mg-9Al | Mg: irregular Al: spherical | Mg: 42 Al: 17 | SLM | 82 ± 3 | - | - | - | 80 ± 7 | 120 | 300 | 93.75 | [32] |
| Mg | spherical | 25.8, 43.32 | SLM | 96.13 | - | - | - | 52.4 | 90 | 10 | 300 | [33] |
| Mg | irregular | 75–150 | SLM | - | - | - | - | - | — | [34] | ||
| Mg | spherical | 5–45 | SLM | - | - | - | - | - | 20 | 20 | 0.99J/mm2 | [34] |
| Mg | spherical | 5–45 | SLM | - | - | - | - | 0.87 ± 0.13 GPa | 13–26 | 10–200 | 1.27 × 109 J/mm2 (CW) | [35] |
| Mg | spherical | 5–45 | SLM | - | - | - | - | 0.95 ± 0.08 GPa | 13–26 | 10–200 | 1.13 × 1012 J/mm2 (PW) | [35] |
| Mg | spherical | 10–45 | SLM | - | - | - | - | 0.72 ± 0.07 GPa | 18 | 1 | 118.2 J/mm2 | [36] |
| Mg | spherical | 1–10 | SLM | — | - | - | - | 48.3 | 50 | 5 | 100 J/mm2 | [37] |
| Mg-2Ca | spherical | 100–200 | LAM | - | 111.19 | - | - | 68 | 100 | 10 | 1200 | [38] |
| Mg-xSn | spherical | Mg: ~10 Sn: ~1 | SLM | - | - | - | - | 65.7 | 60 | 11 | 107.4 J/mm2 | [39] |
| Mg-3Zn-xDy | Mg-3Zn spherical Dy: irregular | Mg-3Zn: ~150 Dy: ~130 | SLM | - | - | - | - | 121.3 ± 3 | 20 | 3 | 360.4 J/mm2 | [40] |
| AZ61 | spherical | 48 | SLM + HIP | >99 | 230 ± 2 | 120 ± 1 | - | 98.9 ± 5.9 | - | - | 156 | [41] |
| ZK61-xZn | spherical | 50 | LAM | 69.1 ± 1 | - | - | - | 106.8 ± 2 | 90 | 10 | 1146 | [42] |
| Mg-Zn | spherical | Mg: ~50 Zn: ~31 | SLM | 99.35 ± 0.2 | 148 ± 5 | - | 11 ± 0.6 | 50 ± 1 | 180 | 700 | 183.7 | [43] |
| Mg-Gd-Zn-Zr | spherical | 44 | SLM | 99.95 (53.3 J/mm3) | 332 ± 5 | 325 ± 5 | 4 ± 0.2 | - | 80 | 300 | 88.9 | [44] |
| Types | Powder Shape | Particle Size (μm) | Processing | Biodegradation Rate | Refs. |
|---|---|---|---|---|---|
| AZ61 | spherical | 70 | SLM | 12.26 mg/cm−2 | [29] |
| ZK60 | spherical | 50 | SLM | 0.006 mL cm−2 h−1 | [30] |
| ZK30-xAl | spherical | ZK30: 45–74; Al: 5–15 | SLM | 0.17 ± 0.02 mg cm−2 day−1 | [31] |
| Mg | irregular | 75–150 | SLM | Fail | [34] |
| WE43 | spherical | 25–60 | SLM | 0.17 mL/cm2 | [45] |
| ZK60-Cu | spherical | ZK30: 50; Cu: 80 nm | SLM | Close to 1.01 mm y−1 | [46] |
| Surface Feature | Alloy | Scanning Speed (mm/s) | Energy Density (J/mm3) | Unmelted Powder | Pores | Balling |
|---|---|---|---|---|---|---|
| Strong Balling zone | AZ61 | 1000–1800 | 35–45 | Lots of | Lots of, ~14%, >100 µm, connected to each other, a network | Spherical: ~100 μm Ellipsoidal: 100–300 μm |
| AZ91 | 1000 | 38–58 | Lots of | Lots of, ~10% | Spherical: ~100 μm Ellipsoidal: ~300 μm | |
| Mg-9Al | - | - | - | - | - | |
| Balling zone | AZ61 | 800–950 | 63–78 | Less | Less, ~4%, <50 μm, | Spherical: <50 μm |
| AZ91 | 833 | 66–77 | Lots of | Lots of, ~4% | Spherical: ~100 μm | |
| Mg-9Al | 15–20 W: 160–1000 | 3–47 | - | Poor bond neck | Powder stacking | |
| Transition zone | AZ61 | 550–750 | 83–114 | None | None | Less/scanning tracks |
| AZ91 | - | - | - | - | - | |
| Mg-9Al | 10 W: 40 15–30 W: 80 | 63–94 | - | Loose metal structure | - | |
| Forming zone | AZ61 | 250–500 | 125–250 | None | None | None |
| AZ91 | 333–667 | 83–167 | None | None | None/flocculent depositions, scanning tracks | |
| Mg-9Al | 10 W: 10 15 W: 20–40 20 W: 40 | 94–250 | None | None | - | |
| Evaporation zone | AZ61 | - | - | - | - | - |
| AZ91 | ~166 | 214–429 | Fail | Fail | Fail | |
| Mg-9Al | 90–110 W: 0.01–1 | ~2750 | Fail | Fail | Fail |
| Alloy | Energy Density (J/mm3) | Grain Size (μm) |
|---|---|---|
| AZ91 [26] | 104–167 | 1–1.2 |
| WE43 [25] | 238 | 1 |
| ZK60 [30] | 420–750 | 2–8 |
| ZK30-xAl [31] | 4004 Al(wt.%): 0–7 | 21.6 ± 2.6–7.3 ± 1.0 |
| ZK61-xZn [42] | 1146 Zn(wt.%): 5–30 | 1.1–6.1 |
| Mg-9Al [32] | 94–250 | 10–20 |
| Mg [37] | 60–120 | 5–20 |
| Mg-2Ca [38] | 1200 | 5 |
| Mg-xSn [39] | 107 Sn(wt.%): 0–7 | 5–25 |
| Mg-3Zn-xDy [40] | 360 Dy(wt.%): 0–5 | 6.4–18.1 |
| AZ61 [41] | 138–208 | 1.6–2.5 |
| Mg-Gd-Zn-Zr [44] | 27–267 | 1.3 ± 0.4–2.3 ± 1.0 |
| Alloy | Laser Power (W) | Scanning Speed (mm/s) | Energy Density (J/mm3) | UTS (MPa) | YS (MPa) | EL (%) |
|---|---|---|---|---|---|---|
| AZ91 [26] | 200 | 333–667 | 83–167 | 274 ± 7–296 ± 3 | 227 ± 3–254 ± 4 | 1.2 ± 0.1–1.8 ± 0.2 |
| AZ91 [25] | 100 | 800 | 104 | 329 | 160 | 1.8 ± 0.2 |
| AZ61 [28] | 150 | 300–450 | 138–208 | 239 ± 3–296 ± 2 | 217 ± 3–233 ± 2 | 3.1 ± 0.1 |
| Mg-2Ca [38] | 50–100 | 10 | 625–1125 | 5-46 (UCS horizontal) 51-111 (UCS longitudinal) | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, S.; Guo, H. A Review of SLMed Magnesium Alloys: Processing, Properties, Alloying Elements and Postprocessing. Metals 2020, 10, 1073. https://doi.org/10.3390/met10081073
Liu S, Guo H. A Review of SLMed Magnesium Alloys: Processing, Properties, Alloying Elements and Postprocessing. Metals. 2020; 10(8):1073. https://doi.org/10.3390/met10081073
Chicago/Turabian StyleLiu, Shuai, and Hanjie Guo. 2020. "A Review of SLMed Magnesium Alloys: Processing, Properties, Alloying Elements and Postprocessing" Metals 10, no. 8: 1073. https://doi.org/10.3390/met10081073
APA StyleLiu, S., & Guo, H. (2020). A Review of SLMed Magnesium Alloys: Processing, Properties, Alloying Elements and Postprocessing. Metals, 10(8), 1073. https://doi.org/10.3390/met10081073