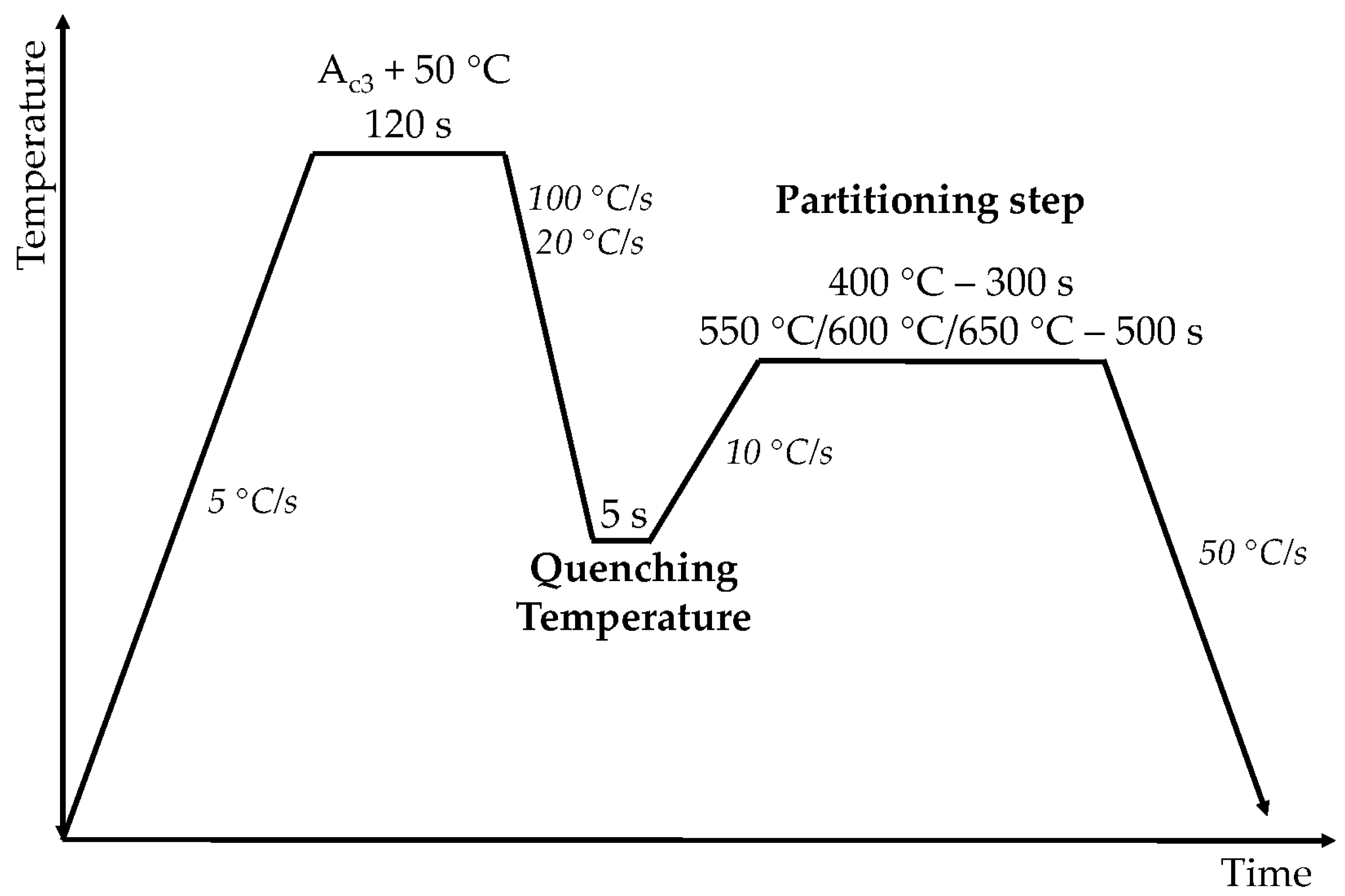
First, the final microstructures were characterized, and then, the results combined with dilatometry analysis allowed for the investigation of microstructure evolution at conventional and high PT cycles.
3.2.1. Characterization of Microstructure after Q&P Heat Treatment
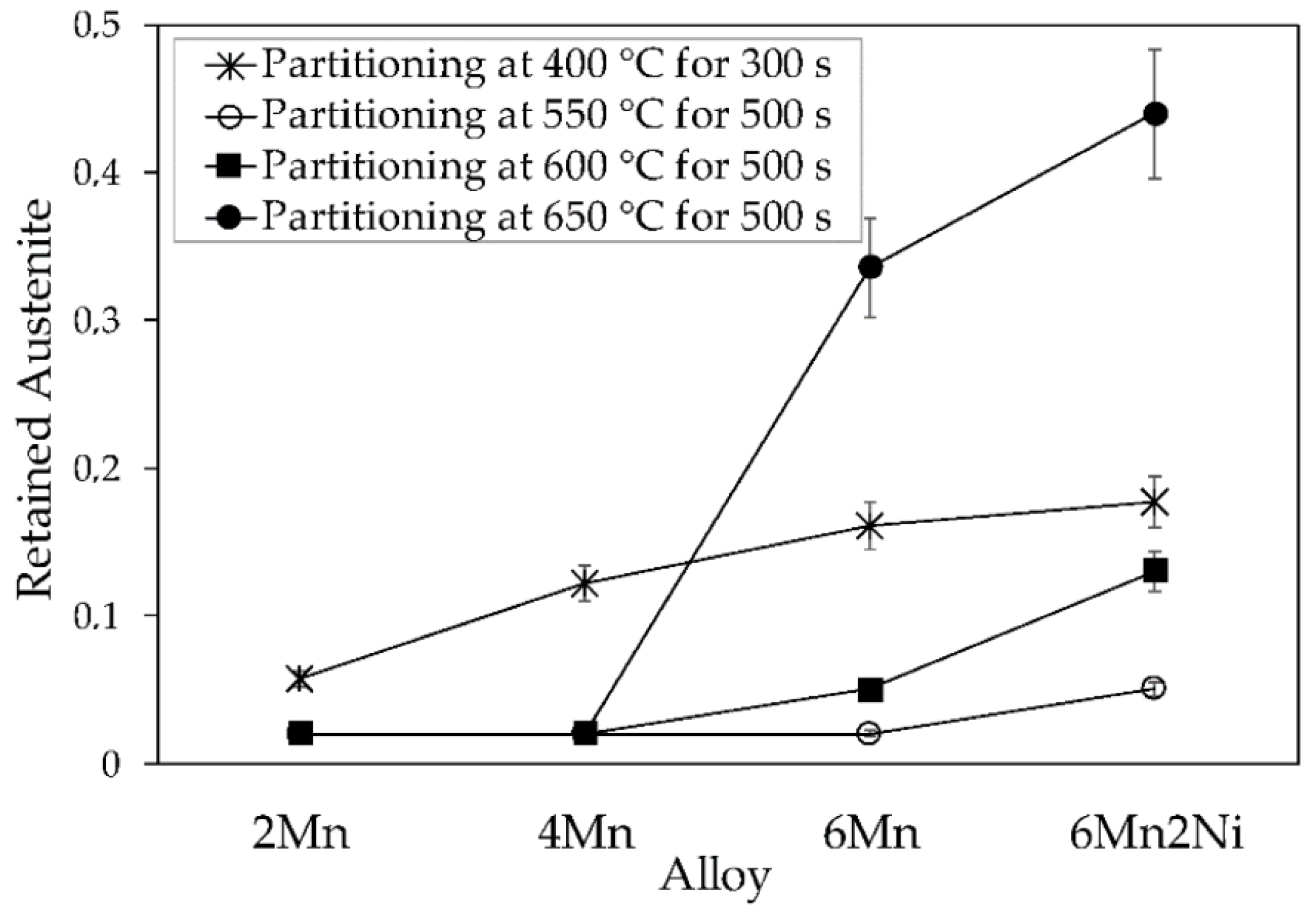
Figure 3 presents the volume fraction of retained austenite measured by XRD for all the alloys and Q&P cycles. In the case of the 400 °C partitioning temperature cycle (PT400), the figure shows an increasing volume fraction of retained austenite with Mn, confirming the stabilization role of this element. Furthermore, the addition of 1.6 wt.% Ni in alloy 6Mn2Ni resulted in a further increase in the volume fraction of retained austenite in the final microstructure. However, the effect of Ni in austenite stabilization was comparatively less significant than the effect of Mn.
In the case of the 550 °C, 600 °C, and 650 °C partitioning temperature cycles (PT550, PT600, and PT650, respectively) in 2Mn and 4Mn alloys, the high partitioning temperature cycles were less effective in retaining austenite than the conventional partitioning done at 400 °C. In the 6Mn and 6Mn2Ni alloys, the results strongly depended on the PT condition. Thus, after partitioning at 550 °C, alloy 6Mn also presented a very low RA fraction, which was slightly raised by the addition of Ni. In comparison, the PT600 cycle resulted in a higher RA volume fraction in both alloys. However, in both PT conditions, the obtained austenite contents were lower than after the PT400 cycle. The results drastically changed with the application of the PT650 cycle, obtaining final austenite contents much higher than those obtained with the PT400 cycle, which were specifically 0.34 and 0.44 in the 6Mn and 6Mn2Ni alloys, respectively.
Given the high RA fraction obtained in alloy 6Mn2Ni after the PT650 cycle, the final microstructure of this alloy was observed in the SEM. The microstructure of alloy 2Mn was also observed for comparison, since, in the same condition, it presented a very low RA fraction. The SEM micrographs are presented in
Figure 4. In alloy 2Mn, the microstructure mainly consisted of tempered martensite containing carbides (
Figure 4a). Carbides presented a coarse size and the start of pearlite formation could be appreciated in some regions. In alloy 6Mn2Ni, the distribution and morphology of the phases indicated a high amount of RA (
Figure 4b). A bright thin lath type phase was observed, which was identified as retained austenite. The dark phase was identified as primary martensite (M1) where few carbides were observed and bigger islands with a smooth dark-grey aspect center and white edges were likely to be MA islands.
Additionally, EBSD phase maps were obtained in the 6Mn2Ni alloy partition-treated at 650 °C and 400 °C. EBSD images are shown in
Figure 5, where the red phase was identified as RA, green was martensite, and black indicates unidentified regions. As can be seen in
Figure 5a, EBSD maps also revealed a high volume fraction of retained austenite in the PT650 condition. Specifically, the volume fraction of retained austenite determined by EBSD was 0.45, which was in very good agreement with the value measured by XRD. The fraction and distribution of the retained austenite grains clearly indicated austenite reverse transformation during the partitioning stage. In the low PT condition (
Figure 5b), the volume fraction of RA determined by EBSD was 0.08, which was lower than the fraction measured by XRD. Austenite fractions determined by EBSD can be lower since the dark regions in the EBSD maps can be MA islands with retained austenite across the grain boundary. However, these regions are mostly recognized as unidentified regions. Additionally, the detection of nanometric film like retained austenite stabilized in between the martensitic laths is also difficult with EBSD.
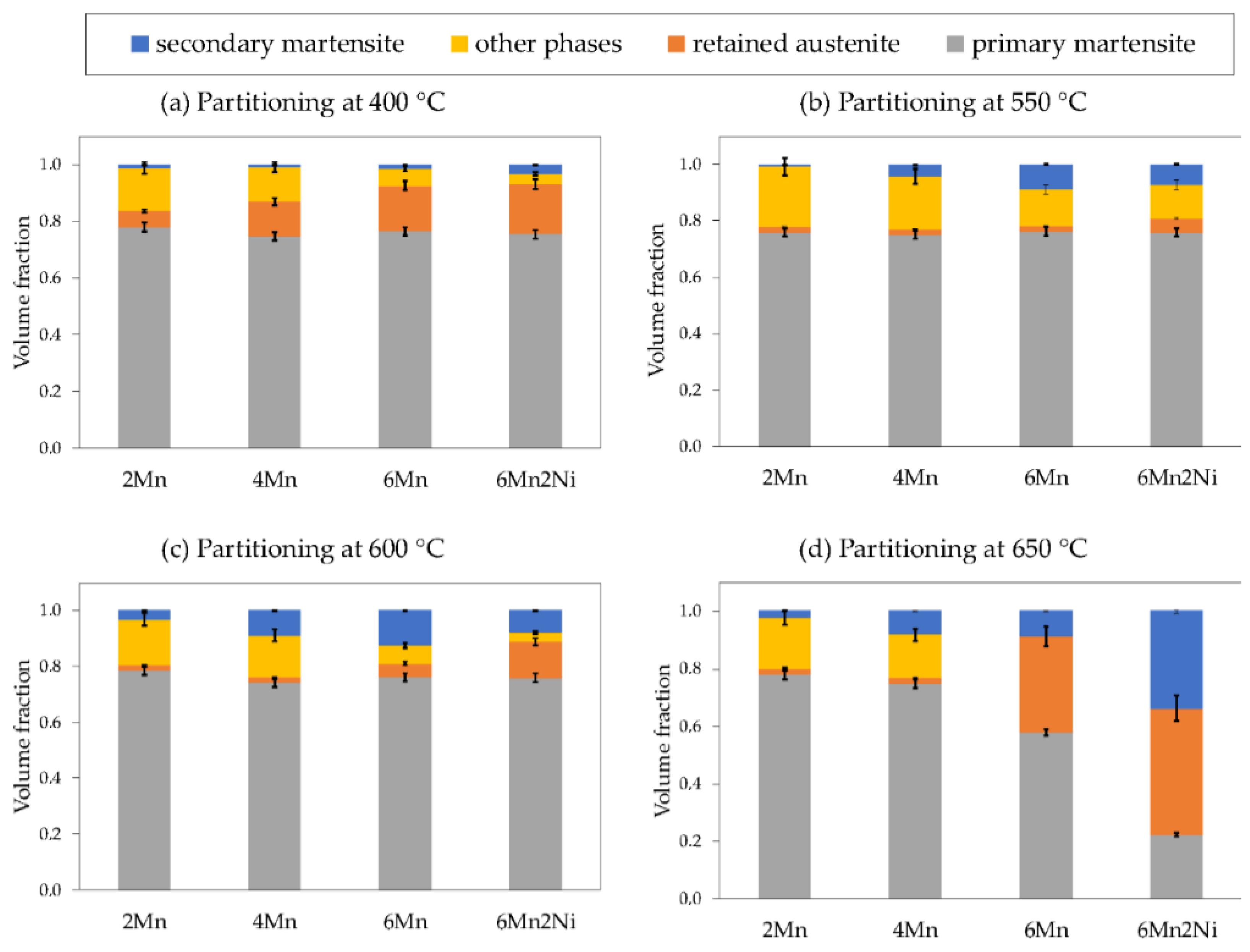
The volume fraction of phases existing after the application of Q&P treatments were estimated based on the dilatometry curves obtained in the simulation of Q&P cycles and the experimentally obtained RA results.
Figure 6 presents the volume fraction of phases obtained for all the alloys and conditions considered. The volume fraction of phases were estimated according to the following procedure. The primary martensite fraction was equal to that present at QT, which was targeted to be 0.75. The slight variations in the primary martensite fraction that can be seen in
Figure 6 are the consequence of considering the real QT achieved in the dilatometry experiments. Secondary martensite was obtained by comparing the change in length at the final cooling with the change in length given by the martensite transformation curve on the directly quenched sample. Retained austenite was experimentally measured by means of XRD measurements. Finally, the products formed by austenite decomposition in the partitioning stage were encompassed in the expression of “other phases”, which was calculated by phase fraction balance. In the conditions that ART occurred in the partitioning stage, RA and secondary martensite were obtained as described earlier, whereas primary martensite was calculated by balance. In this case, the reverted austenite fraction formed in the partitioning stage corresponded to the sum of retained austenite measured by XRD, plus the secondary martensite fraction, deducting the preliminary austenite fraction.
As can be seen in
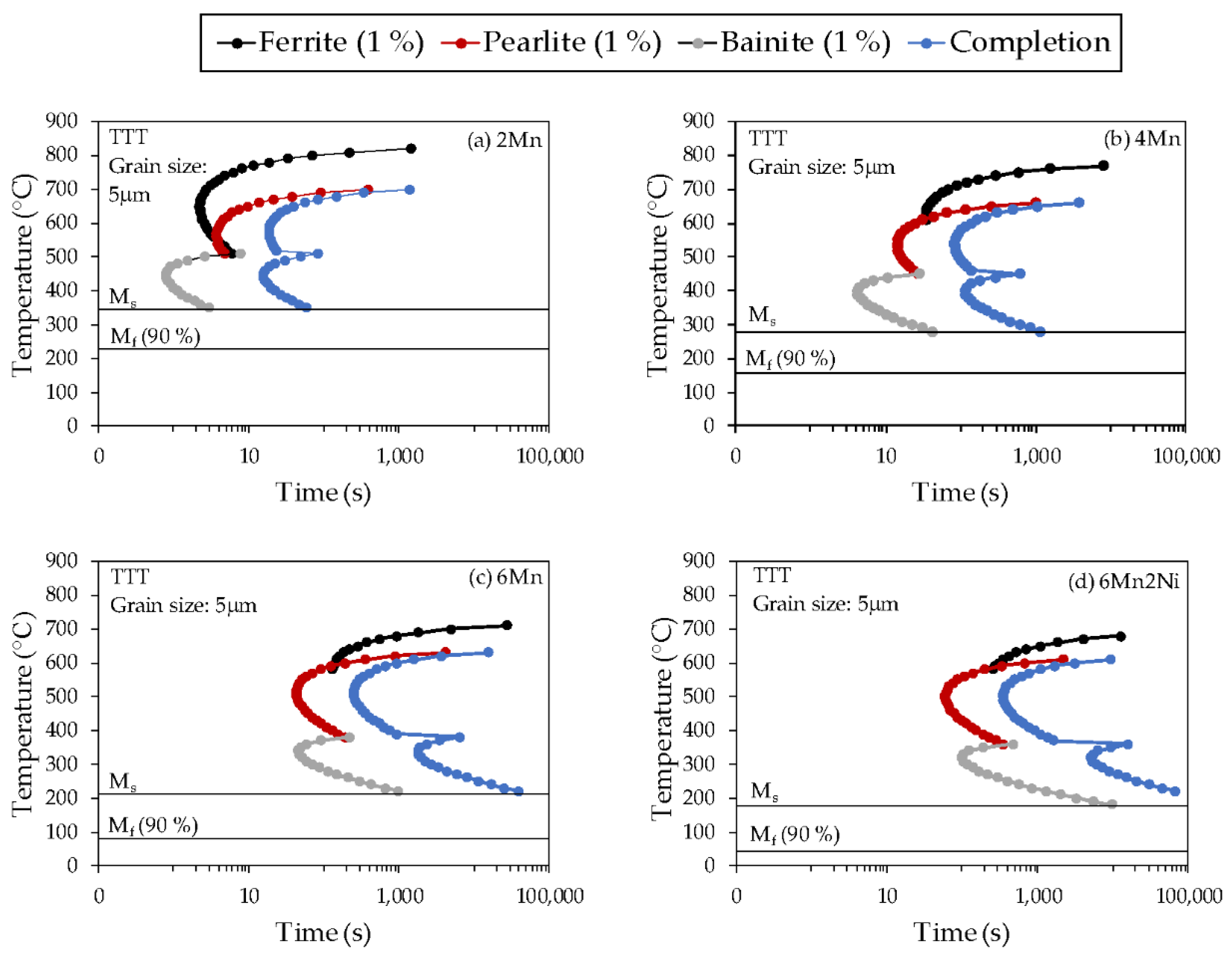
Figure 6, the volume fraction of austenite decomposition products was quite significant in many of the alloys and conditions. In order to aid in the analysis of the products formed in the partitioning stage, time-temperature-transformation (TTT) diagrams were obtained for each alloy using JMatPro-v9.1 software.
Figure 7 presents the TTT diagrams calculated for an initial austenite grain size of 5 µm. Although not fully representative of the conditions existing at PT, these diagrams contributed to the understanding of the phenomenon occurring in the partitioning step. TTT diagrams showed that Mn addition shifted phase transformation curves to a lower temperature range and longer times. The addition of Ni mainly resulted in longer phase transformation times. The comparison between the TTT diagrams and experimental results is detailed in
Section 3.2.2.
3.2.2. Effect of Mn and Ni Alloying Elements on Microstructure Evolution
As mentioned earlier, the microstructure evolution during the Q&P cycle was discussed based on the dilatometry curves and volume fraction of phases determined in the final microstructure. Dilatometry curves obtained in the simulation of the different Q&P cycles are presented in
Figure 8 for the four alloys. Dilatometry curves start at QT and represent the heating to PT, partitioning stage, and final cooling. The results obtained at the conventional partitioning condition of 400 °C and 300 s are discussed first for a later comparison with the behavior observed when the partitioning temperature was raised to the range of 550 °C to 650 °C.
● Partitioning temperature – 400 °C
Dilatometry curves corresponding to the 400 °C partitioning temperature cycles are presented in
Figure 8a for the four alloys. Continuous expansion was observed in the partitioning stage in the four alloys, as indicated in the figure by means of ascending discontinuous arrows. The measured expansion decreased as the alloying content increased. Considering the expansion as ΔL/L
0 × 100, where ΔL is the change in length of the specimen during the partitioning and L
0 is the initial length, 0.100%, 0.028%, 0.018%, and 0.017% values were obtained for the 2Mn, 4Mn, 6Mn, and 6Mn2Ni alloys, respectively. Santofimia et al. reported that volume expansion during the partitioning stage could be related with the decomposition of austenite to ferrite, bainite, or isothermal martensite transformation [
6]. Carbon partitioning from supersaturated martensite to austenite can also cause volume expansion [
12,
17]. However, comparatively, austenite decomposition to bainite gives rise to a more pronounced expansion [
6].
In the case of the lower alloyed 2Mn steel, the volume expansion was more evident and was likely to be related to austenite decomposition. However, in the 4Mn, 6Mn, and 6Mn2Ni alloys, it was not clear whether the observed volume expansion was only due to carbon partitioning or if austenite decomposition also contributed. According to the estimated volume fraction of phases (
Figure 6a), the fraction of austenite decomposition products decreased with the addition of Mn and Ni. However, a significant fraction was obtained in alloys 4Mn, 6Mn, and 6Mn2Ni. Hence, it is likely that apart from carbon partitioning, austenite decomposition also contributed to the observed expansion in these alloys.
Possible austenite decomposition products were analyzed based on TTT diagrams (
Figure 7). At 400 °C, austenite is predicted to decompose into bainite in alloys 2Mn and 4Mn and in pearlite in alloys 6Mn and 6Mn2Ni. However, the theoretical times for the start of pearlite phase transformation are significantly longer, which is in agreement with the lower fraction of austenite decomposition products estimated in alloys 6Mn and 6Mn2Ni.
Finally, the amount of secondary martensite formed in the four alloys was very low, obtaining the highest amount, the 0.03 volume fraction, in alloy 6Mn2Ni. As can be seen in the dilatometry curves, secondary martensite formation was not evident in the final cooling. This means that the austenite existing at the end of the partitioning stage was stable enough to be mostly untransformed upon final cooling.
● Partitioning temperature – 550 °C, 600 °C, 650 °C
In
Figure 8b–d, the dilatometry curves are shown for partitioning temperatures of 550 °C, 600 °C, and 650 °C, respectively. Within the graphs, the discontinuous ascending or descending arrows indicate the expansive or contractive behavior of the samples during the partitioning stage. It can be seen that the results strongly depend on the alloying content. The lower Mn containing alloys, 2Mn and 4Mn, showed dilatation, whereas in the 6Mn and 6Mn2Ni alloys, continuous contraction was observed in all the cases.
Additionally, a volume change was observed in the final cooling in alloys 4Mn, 6Mn, and 6Mn2Ni, at the three high PTs, which denoted the formation of secondary martensite. This means that austenite, at the end of partitioning, was not stable enough to remain untransformed in the final cooling. In alloy 2Mn, this volume change was not detected, indicating that secondary martensite formation was not significant. Additionally, alloy 2Mn presented a non-linear dilatation during the heating from QT to PT, as can be observed in
Figure 8b–d. For the remaining alloys, a linear expansion was registered during the heating. The non-linear expansion is likely to indicate the occurrence of a phase transformation in this alloy during the heating to PT. For the 550 °C partitioning temperature cycle, only the onset of the transformation was observed at around 393 °C. In the Q&P cycles with 600 °C and 650 °C as partitioning temperatures, the start and end of the transformation were estimated as 379 °C/552 °C and 358 °C/583 °C, respectively. These temperatures are marked with arrows in
Figure 8b–d. Based on the previously presented SEM micrograph (
Figure 4a), the observed expansion could be related with pearlite formation. Furthermore, since secondary martensite formation was not evident in the final cooling in the 2Mn alloy and the volume fraction of austenite was rather low in the final microstructure, it is likely that austenite was mostly decomposed during the heating to PT and holding at PT.
In the PT550 cycle, a higher Mn content did not result in more austenite in the final microstructure. According to the estimated volume fraction of phases (
Figure 6b), the increase in Mn gave rise to a lower decomposition of austenite in the partitioning stage as the volume fraction of other phases diminished. However, the undecomposed austenite seemed to not be sufficiently stable and transformed into secondary martensite in the final cooling, resulting in a higher volume fraction of secondary martensite in the alloy with a higher Mn content. The addition of Ni in the 6Mn2Ni alloy seemed to be slightly favorable for austenite stabilization.
According to the TTT diagrams, isothermal holding at 550 °C decomposes austenite into pearlite in the four alloys. The formation of pearlite clearly reduces the carbon available for austenite stabilization. This is in agreement with the results found by Ayenampudi et al. [
12], who found that strong competitive reactions such as pearlite formation occurred at 500 °C partitioning, decreasing the final austenite content. However, pearlite formation would result in volume expansion [
12], which was only observed in alloys 2Mn and 4Mn. In contrast, the 6Mn and 6Mn2Ni alloys gave rise to a slight contraction in the partitioning stage. Kannan et al. [
18] and Onink et al. [
19] showed that austenite films saturated with carbon tended to decompose into carbon-depleted austenite and cementite, and this phenomenon gave rise to contraction. Therefore, it seems that in alloys 2Mn and 4Mn, the main reaction leading to the decomposition of austenite in the partitioning was pearlite formation, and in 6Mn and 6Mn2Ni alloys, due to the higher Mn and Ni contents, pearlite formation was retarded and the main competitive reaction occurring during the partitioning was austenite decomposition into carbon depleted austenite and cementite. Both reactions resulted in a lower carbon available for austenite stabilization.
In the PT600 cycle, the volume fractions of phases were very similar in alloys 2Mn and 4Mn to those determined after the PT550 cycle (
Figure 6b,c). The most significant difference was that lower fraction of other phases and an increase in the formation of secondary martensite were obtained in the PT600 cycle. This behavior denoted that the amount of austenite decomposed into pearlite was lower at 600 °C, which is in agreement with the theoretical TTT diagrams that place the nose of pearlite closer to 550 °C. However, the fraction of austenite was not stable enough and secondary martensite formed in the final cooling. The further addition of Mn and particularly Ni was more effective in retarding the competitive reactions occurring during the partitioning stage, which resulted in more retained austenite in the final microstructure. Nevertheless, at both 550 °C and at 600 °C, the occurrence of austenite decomposition reactions strongly competed with carbon partitioning as well as with the potential austenite stabilization by Mn and Ni, which is in agreement with the work published in [
12].
In the PT650 cycle, the results of the volume fraction of phases were, as before, very similar in the 2Mn and 4Mn alloys (
Figure 6d). However, partitioning at 650 °C on alloy 6Mn led to 0.34 retained austenite in the final microstructure. As described earlier, in this alloy and PT condition, volume contraction was observed in the partitioning stage. In this case, the high austenite volume fraction measured by XRD and EBSD and the distribution and morphology of austenite observed in the SEM and EBSD images pointed to the ART phenomenon as an explanation for the measured contraction.
The onset of ART was calculated by means of dilatometry tests, as described earlier. In
Table 2, it can be seen that the ART phenomenon started at a temperature lower than the A
c1 temperature determined in the first heating stage of the Q&P cycle. The difference between both temperatures was between 35 °C and 52 °C considering the four alloys and was related to the presence of pre-existing austenite, which likely reduced the nucleation step of the transformation.
The temperature for the start of ART was determined to be 660 °C in alloy 6Mn considering a continuous heating from QT. However, stopping the heating at 650 °C and keeping it there for 500 s resulted in the formation of a 0.19 volume fraction of reverted austenite. Thus, the austenite fraction at the end of the partitioning, which represented the accumulation of pre-existing austenite (0.24) and the reverted austenite contents, was 0.43. The latter volume fraction was below the equilibrium fraction at 650 °C predicted by ThermoCalc (v. 2018b), which was 0.55. Given that 0.09 of secondary martensite appeared in the final cooling, the finally retained austenite volume fraction was 0.34, which was considered a high volume fraction.
Therefore, an important finding in this work was that ART in Q&P routes can stabilize a high amount of austenite at room temperature in medium Mn steels. Additionally, it should be emphasized that, in comparison with the intercritical annealing of medium Mn steels, the treatment applied in the present work requires shorter partitioning times to stabilize a high amount of austenite. It is likely that the pre-existing austenite reduces the nucleation time for ART and overall results in lower annealing times needed for obtaining high austenite volume fractions. This finding can represent an important advantage in terms of industrialization.
The occurrence of ART in Q&P heat treatments was previously analyzed by Ding et al. [
16] in what they called the quenching-austenite reversion treatment. They reported the formation of new austenite by reheating the initial primary martensite + pre-existed austenite microstructure in the intercritical region in a Fe-0.2C-8Mn-2Al steel alloy. Considering a pre-existing austenite fraction of 0.20 and 0.30 and a partitioning done at 660 °C for one hour, the final RA measured by XRD was around 0.44 and 0.35, respectively. Here, for an initial austenite of 0.25, the final austenite was slightly lower at 0.34, which could be explained by the lower Mn addition in alloy 6Mn.
Finally, the addition of the 1.6 wt.% Ni in alloy 6Mn2Ni resulted in a further increase in the final fraction of austenite, obtaining a 0.44 volume fraction of RA after the application of the PT650 cycle. The addition of Ni resulted in a stronger contraction in the partitioning stage, as can be seen in
Figure 8d. In this case, austenite formation in the continuous heating from QT was determined to start at 640 °C and the estimated austenite volume fraction at the end of the partitioning stage was 0.78, which corresponded to the pre-existing austenite (0.25) and new austenite formed in the partitioning (0.53). As this alloy presented a lower temperature for the start of ART, the driving force for austenite reversion was higher, which resulted in a very high austenite volume fraction. This value was indeed higher than the equilibrium fraction predicted by ThermoCalc at 650 °C, 0.70, which was likely due to the error in the estimation of the austenite fraction at the end of the partitioning, which was assumed to be 12%. Nevertheless, both numbers were of the same order and it can be said that the volume fraction at the end of the partitioning was very close to the equilibrium fraction predicted for alloy 6Mn2Ni. In alloy 6Mn2Ni, a very significant amount of secondary martensite, 0.34, was formed in the final cooling.