Recovery of Rubidium and Cesium Resources from Brine of Desalination through t-BAMBP Extraction
Abstract
1. Introduction
2. Material and Methods
2.1. Materials
2.2. Equipment
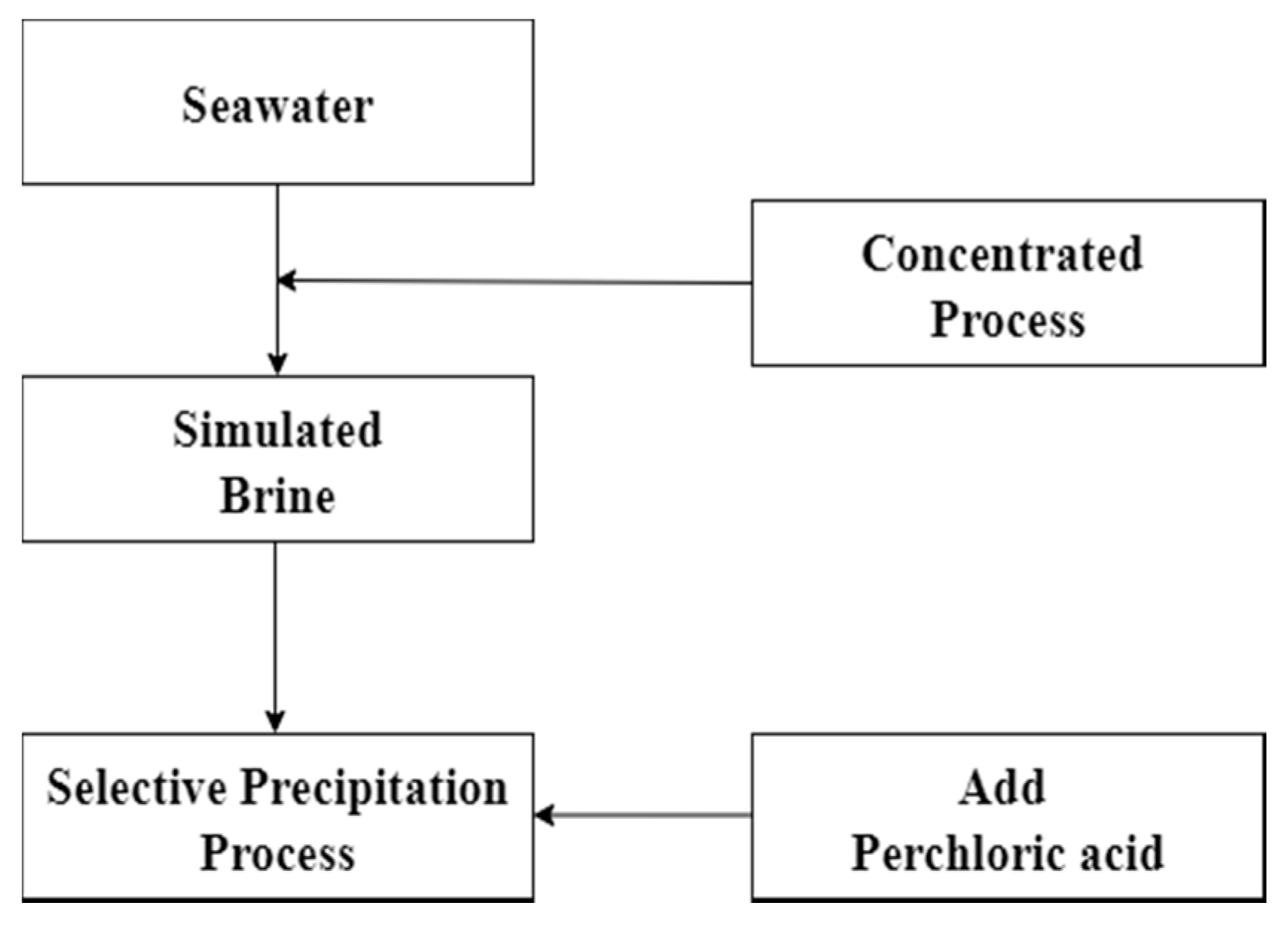
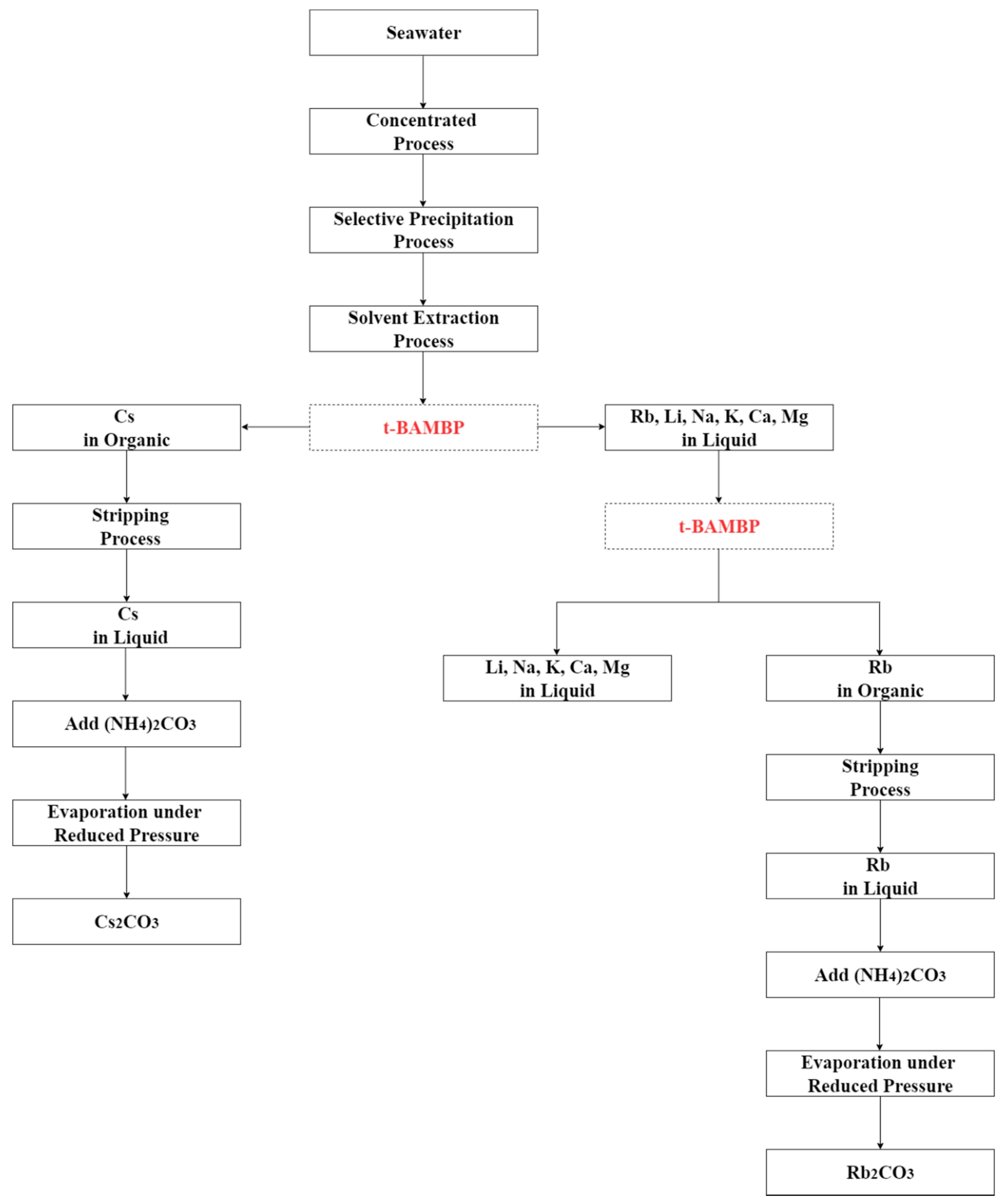
2.3. Experimental Procedures
2.3.1. Chemical Precipitation
2.3.2. Solvent Extraction–Extraction Process
2.3.3. Solvent Extraction–Stripping Process
3. Results and Discussion
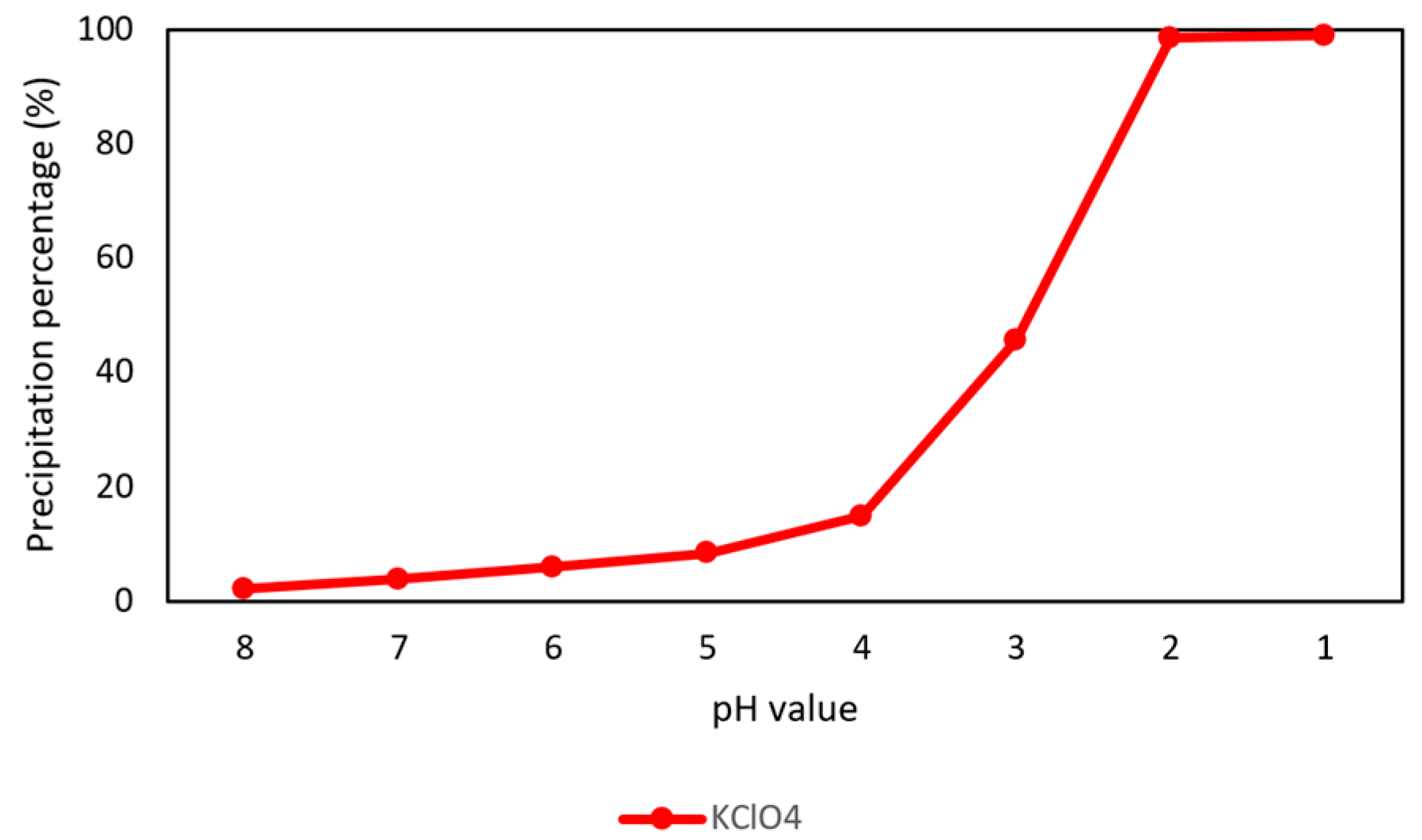
3.1. Removal of Potassium
3.2. First Stage of Solvent Extraction for Cs
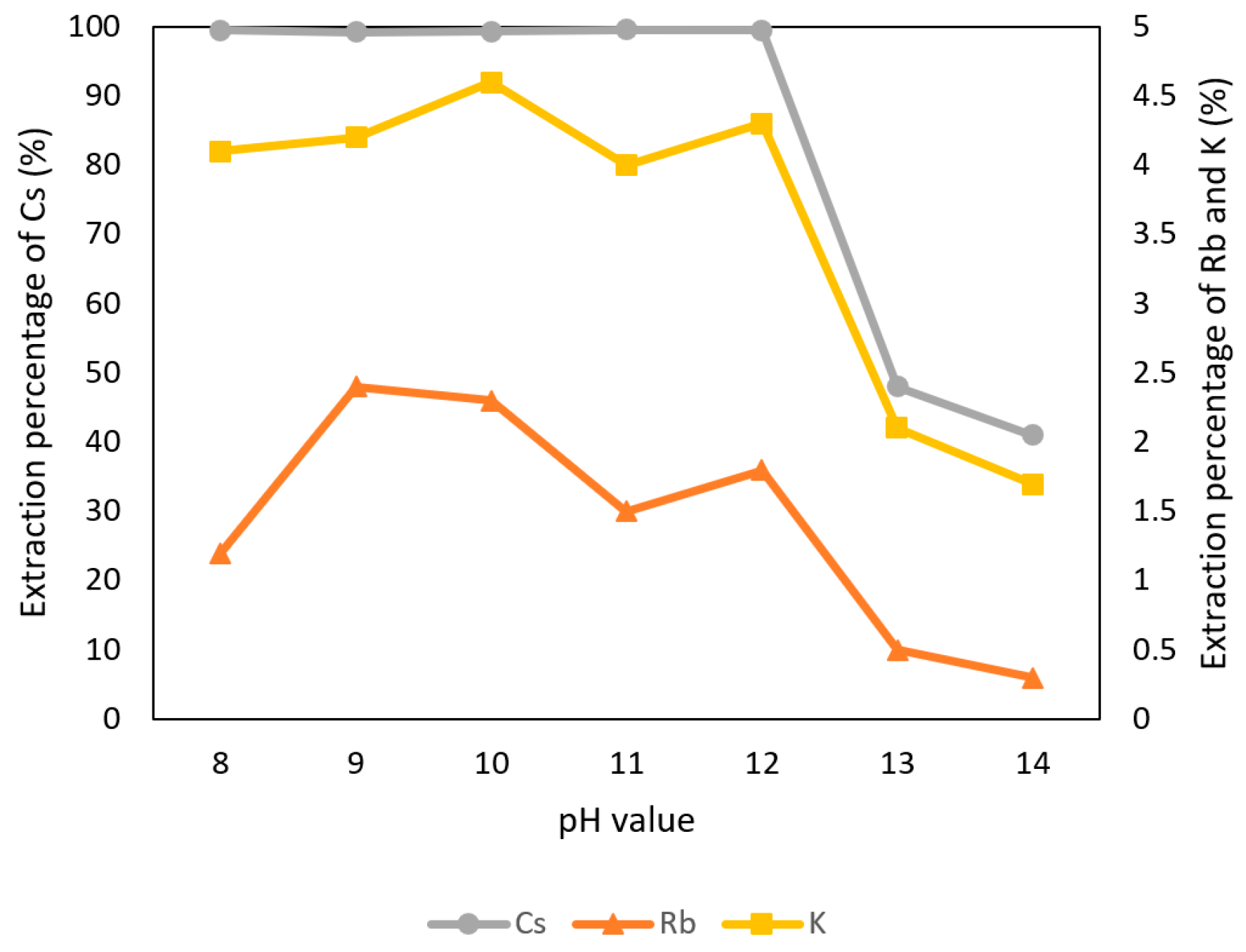
3.2.1. Effect of pH Value of the Aqueous Phase
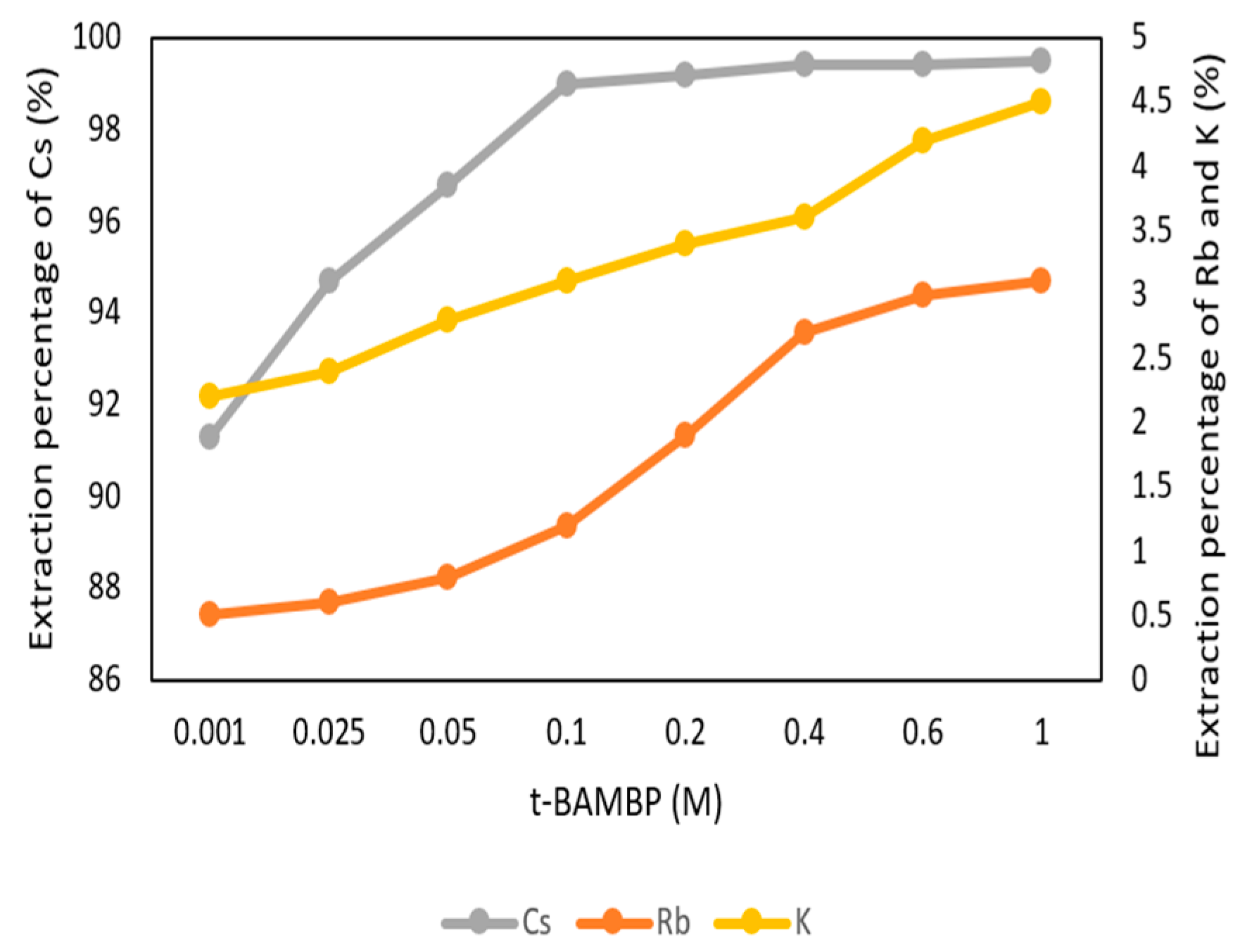
3.2.2. Effect of t-BAMBP Concentration
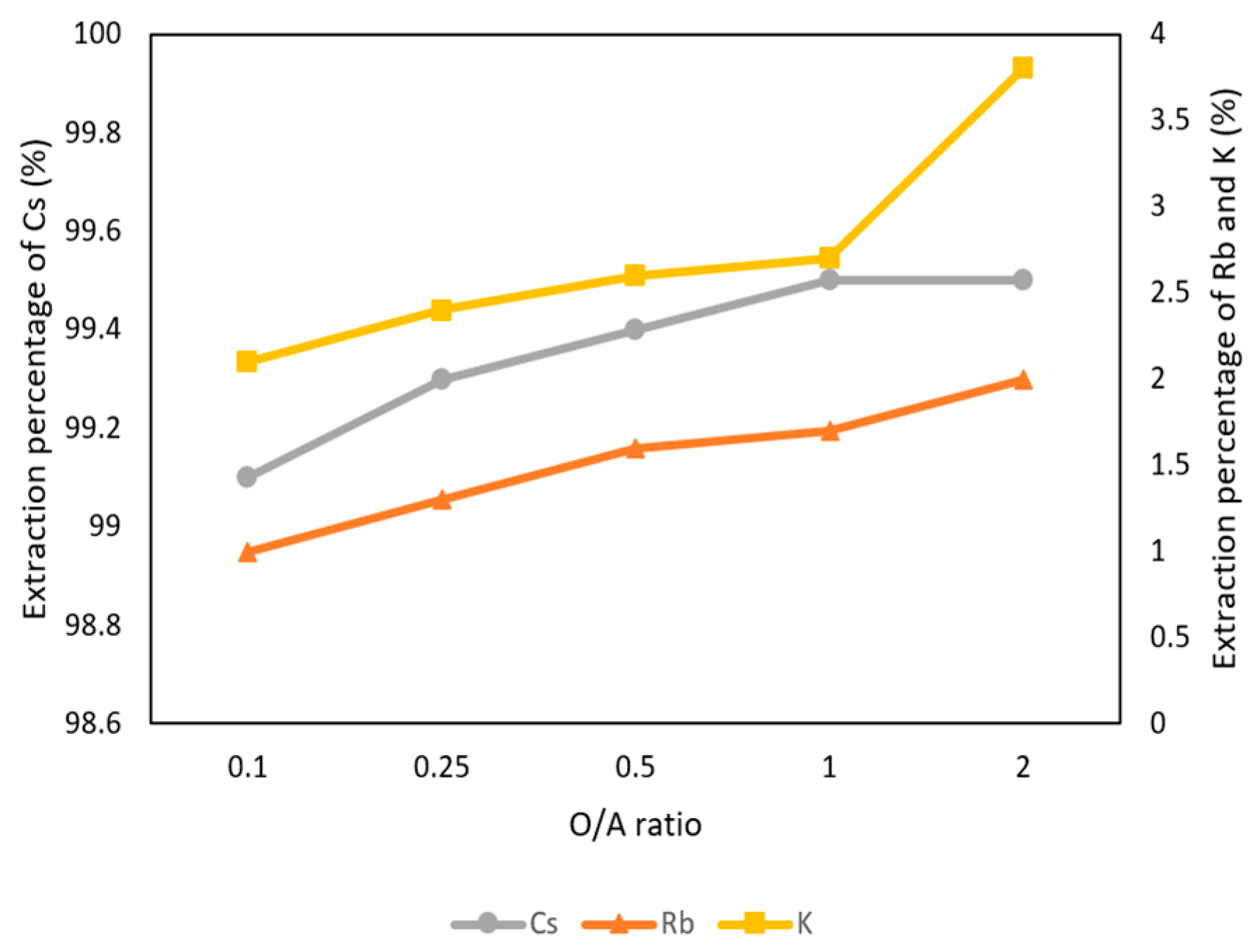
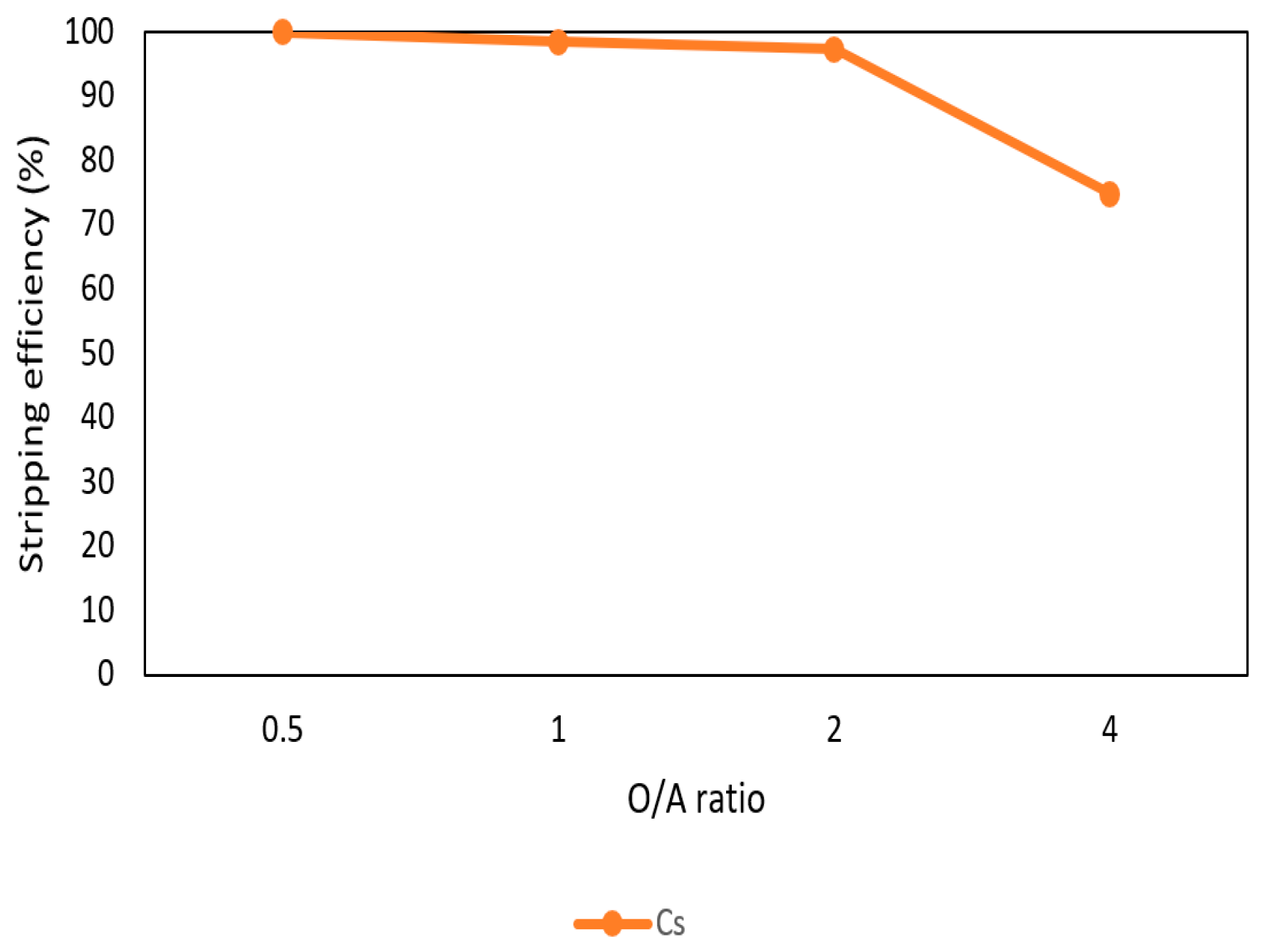
3.2.3. Effect of O/A Ratio
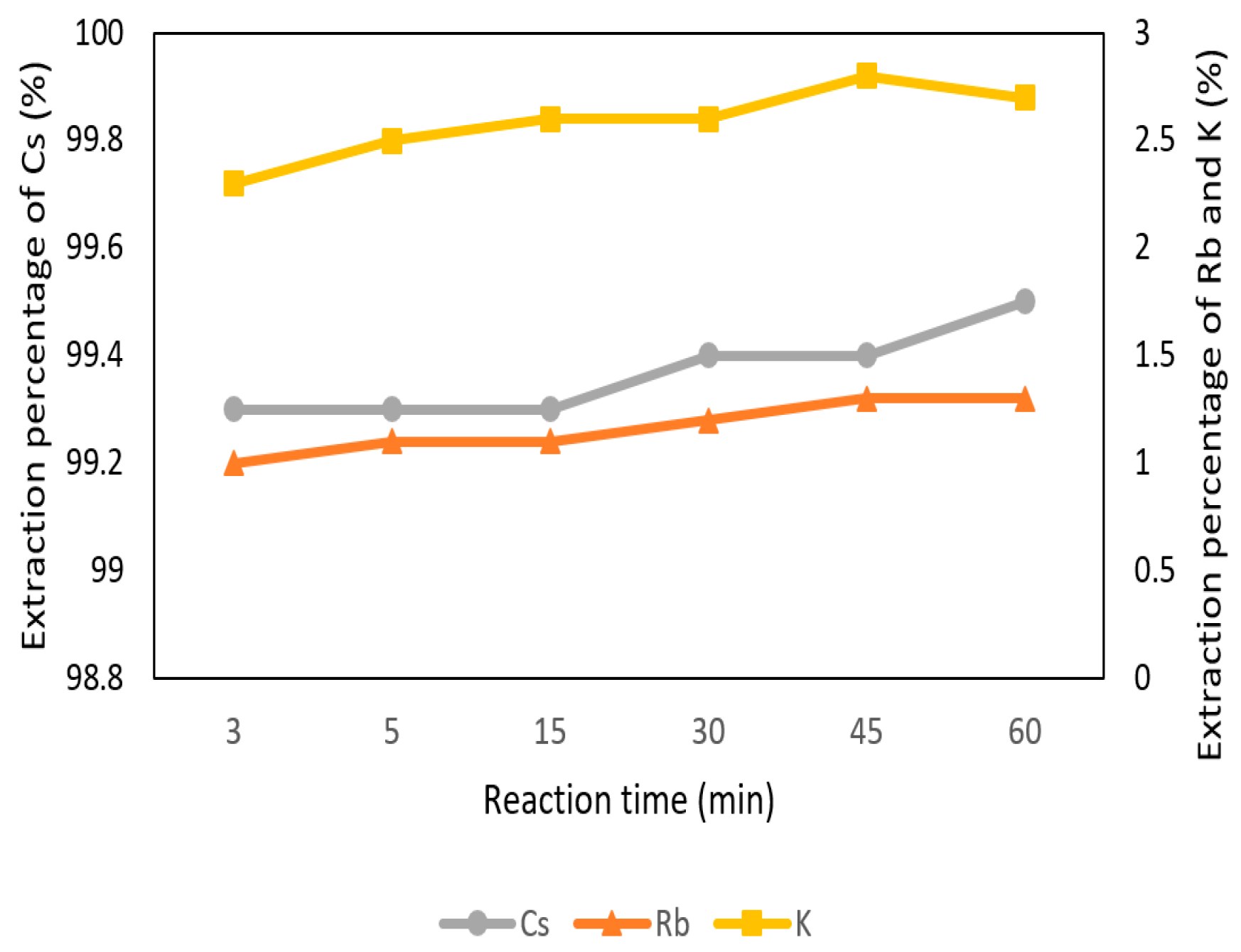
3.2.4. Effect of Reaction Time
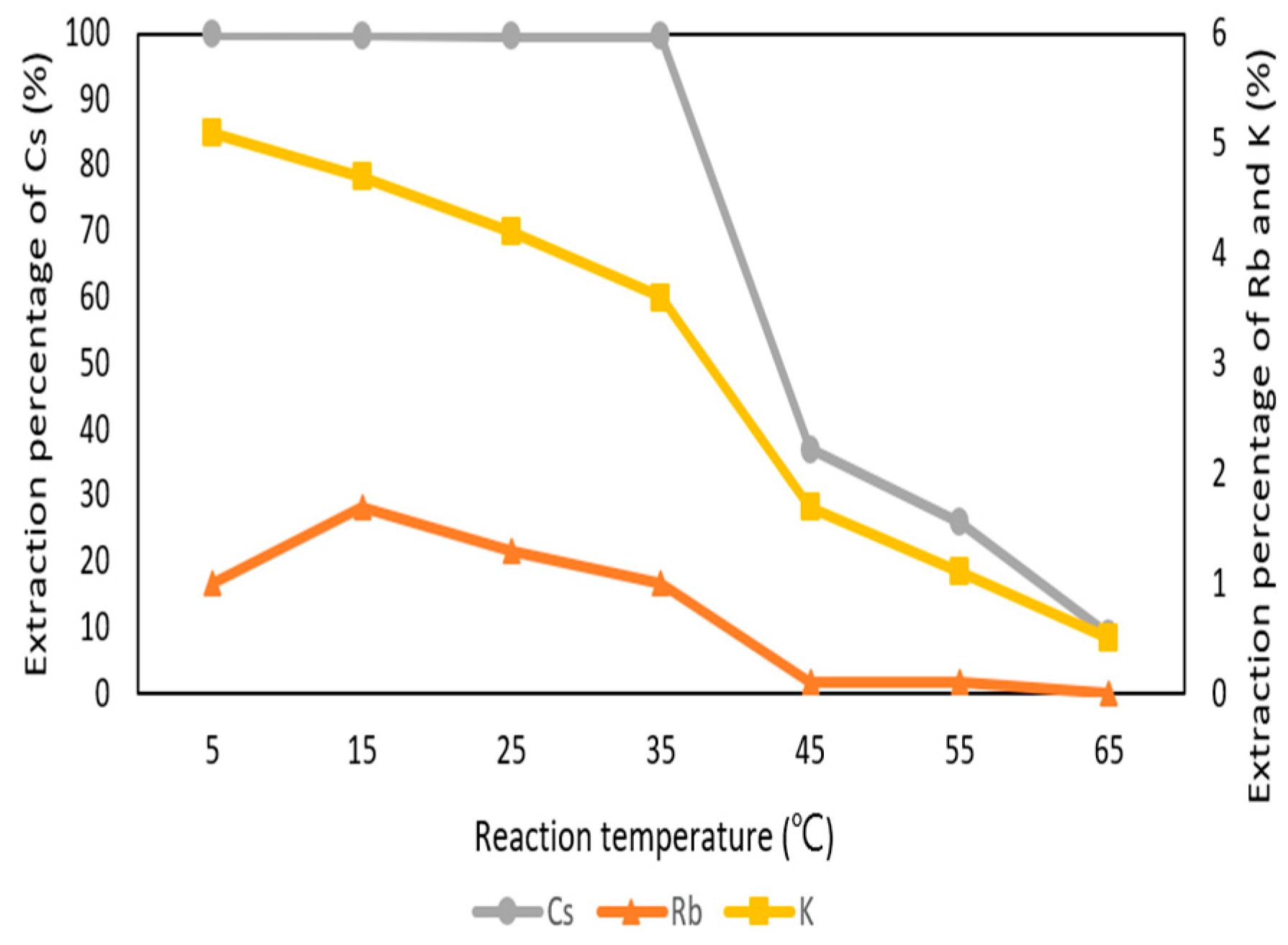
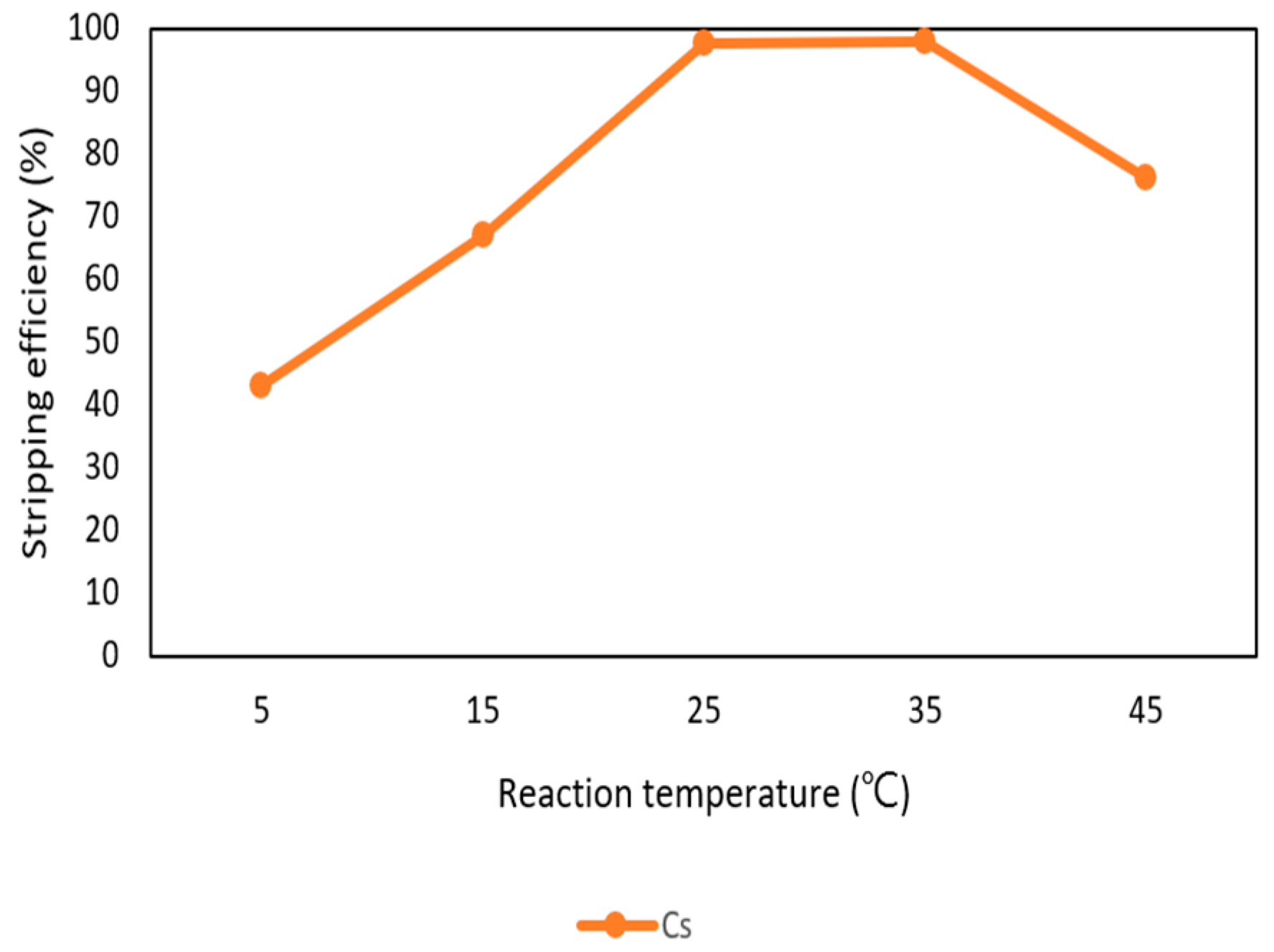
3.2.5. Effect of Reaction Temperature
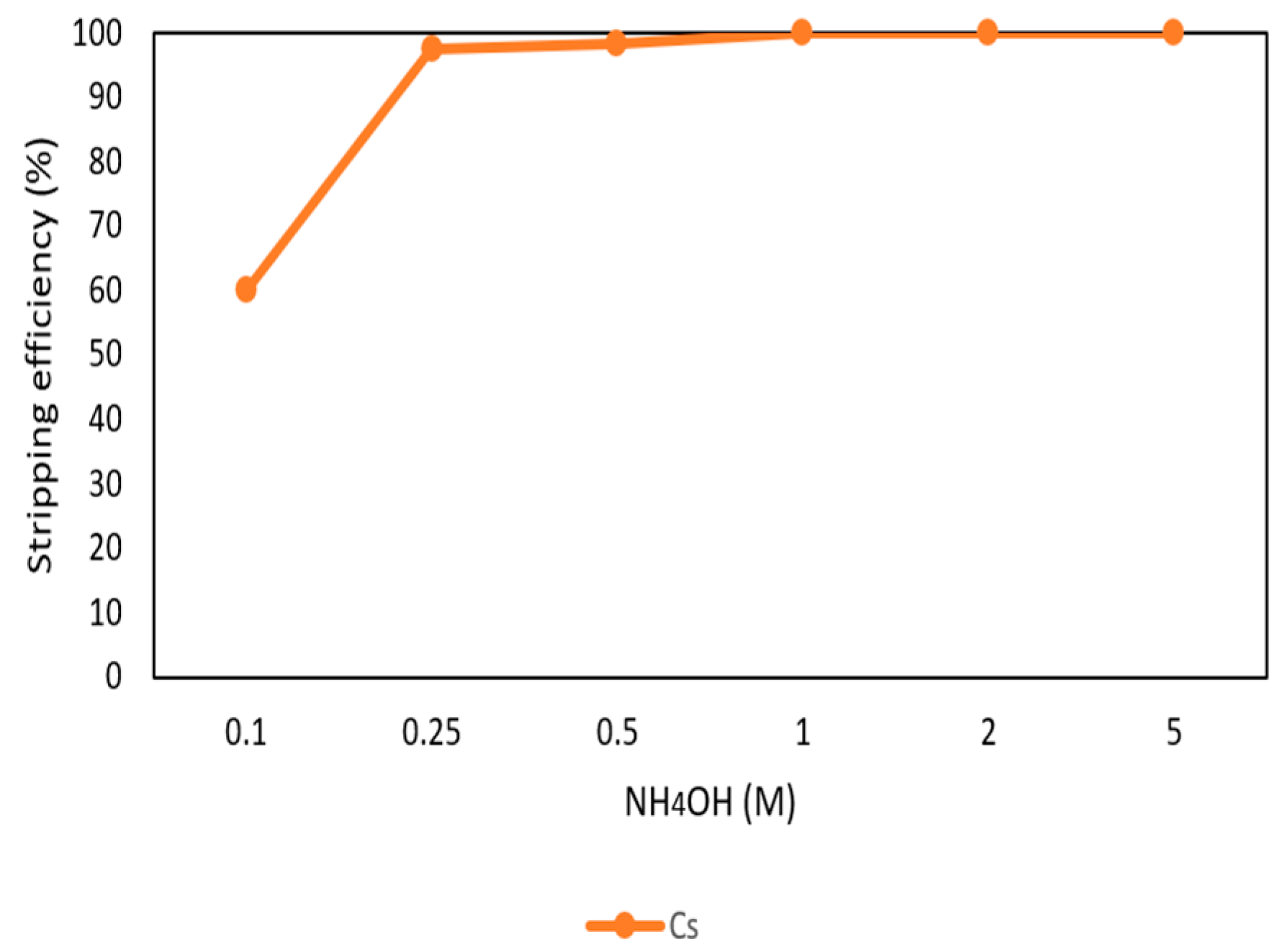
3.2.6. Stripping of Cs from the Organic Phase through Ammonia
3.3. Second Stage of Solvent Extraction for Rb
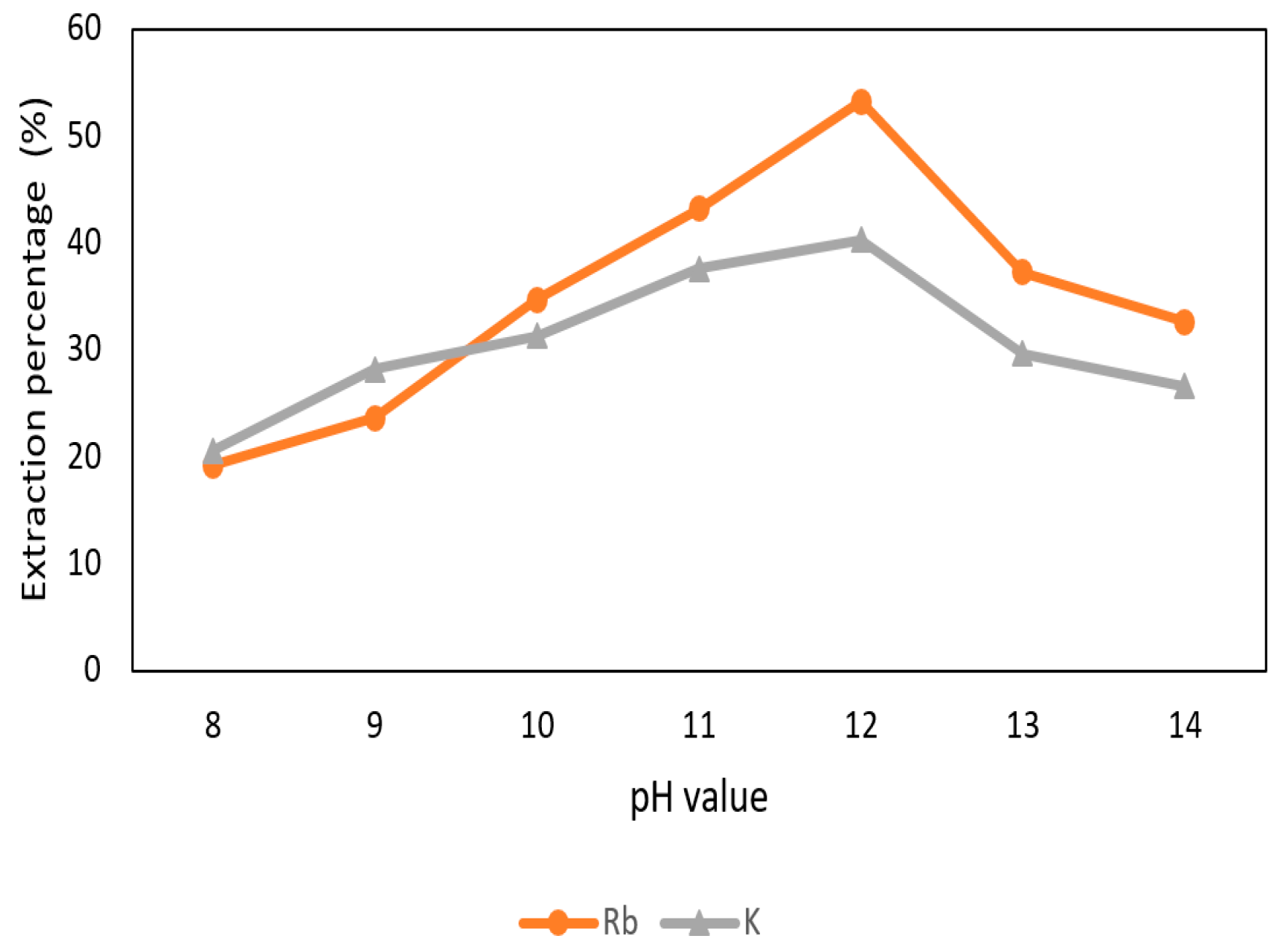
3.3.1. Effect of pH Value of the Aqueous Phase
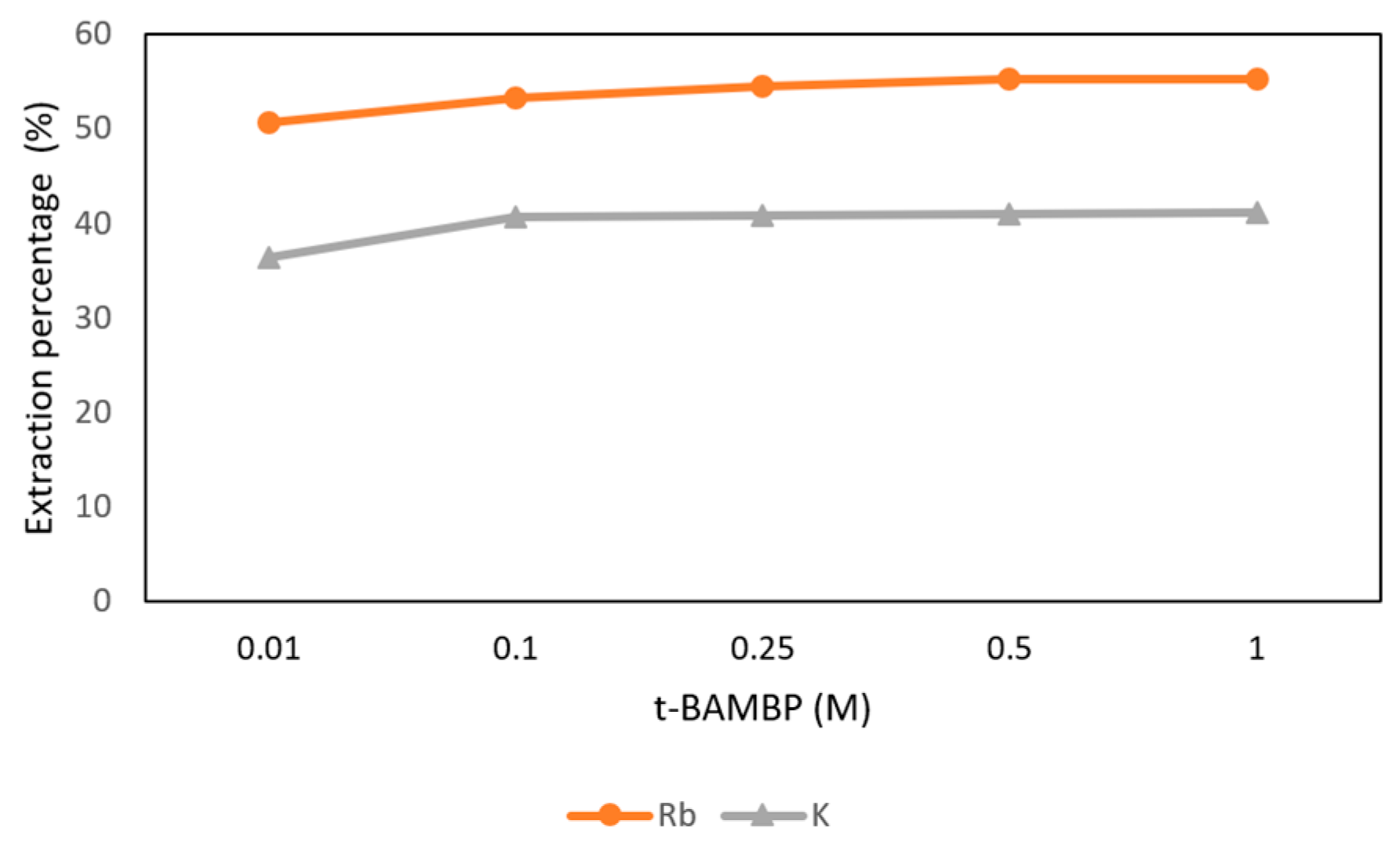
3.3.2. Effect of t-BAMBP Concentration
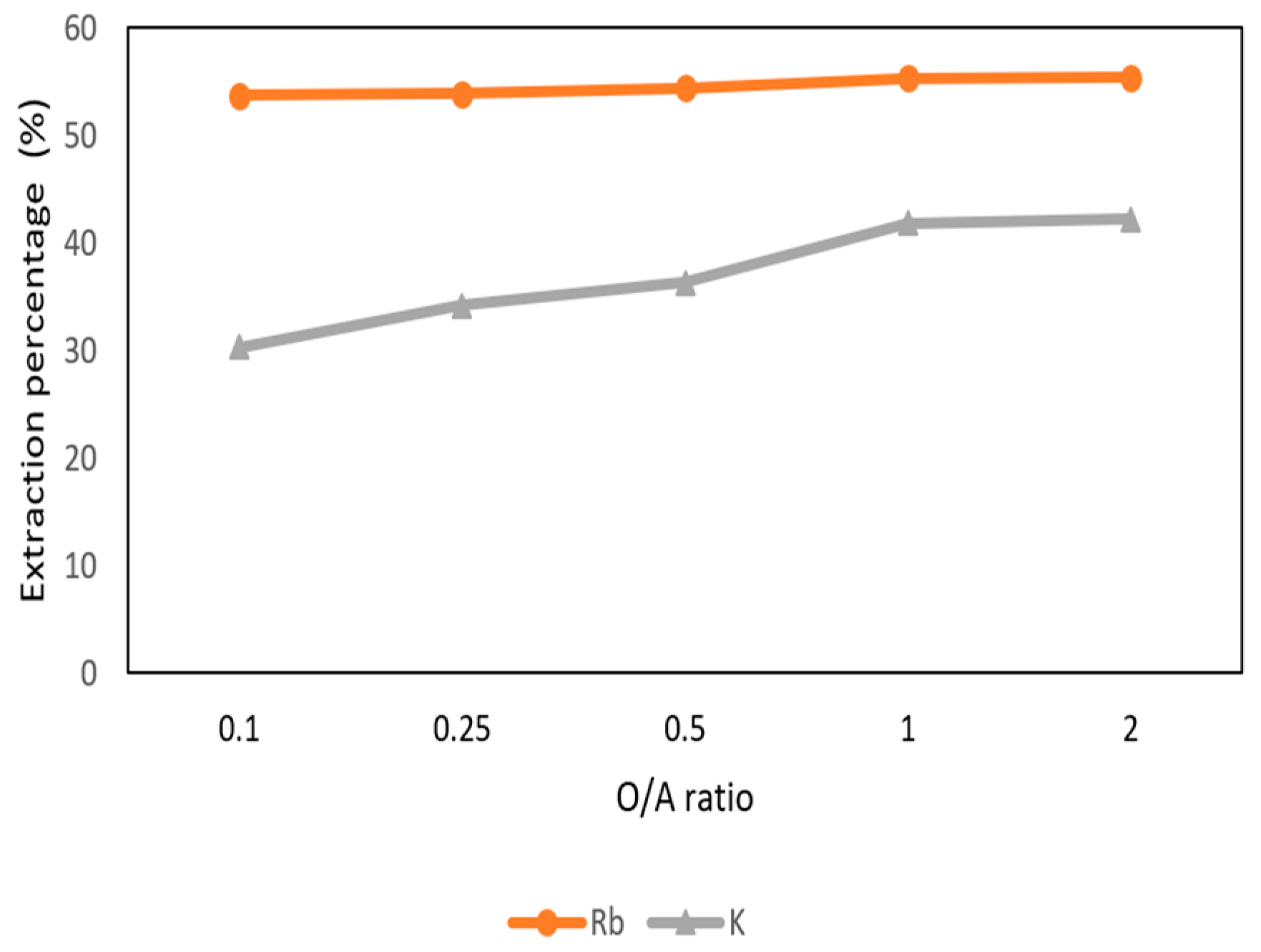
3.3.3. Effect of O/A Ratio
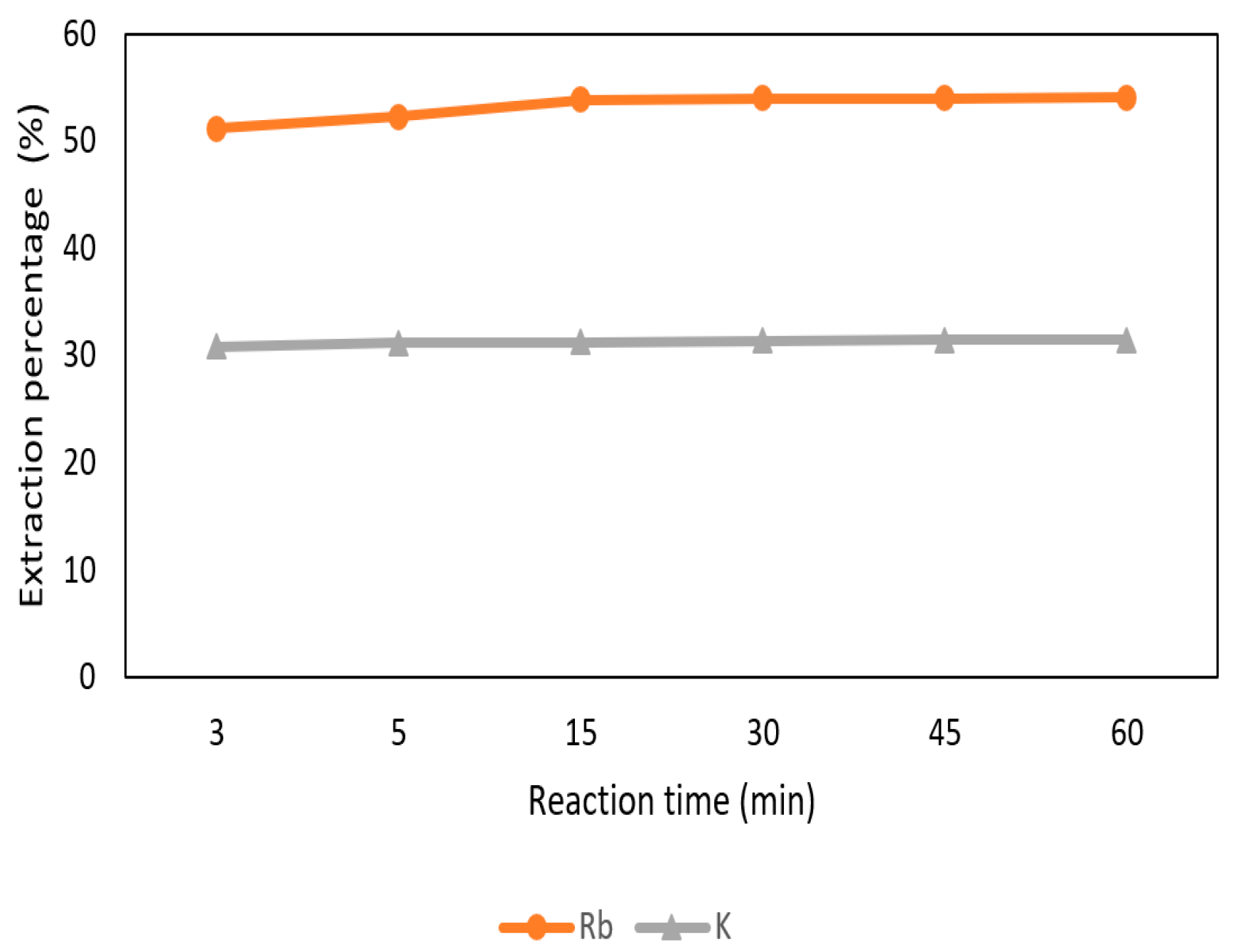
3.3.4. Effect of Reaction Time
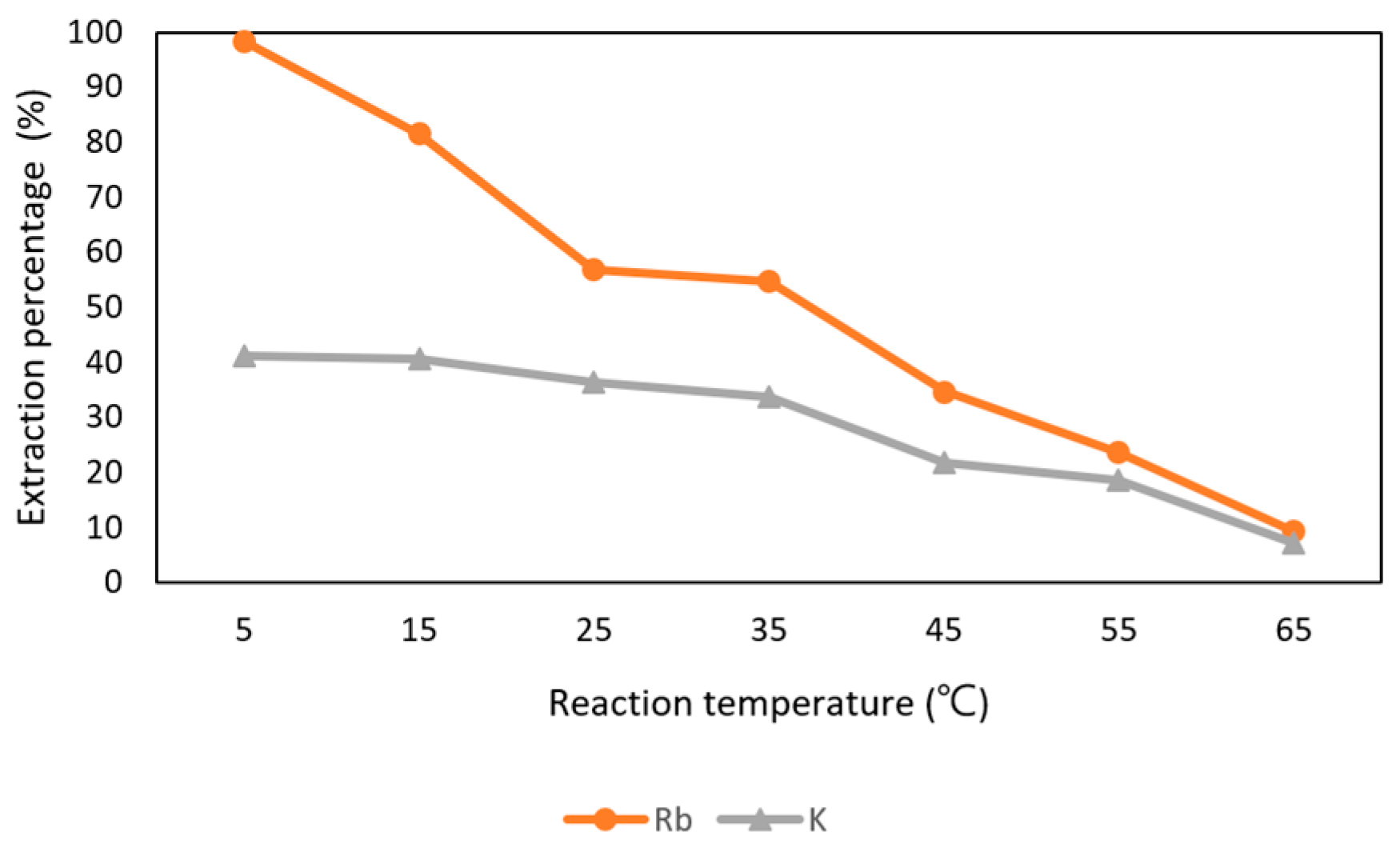
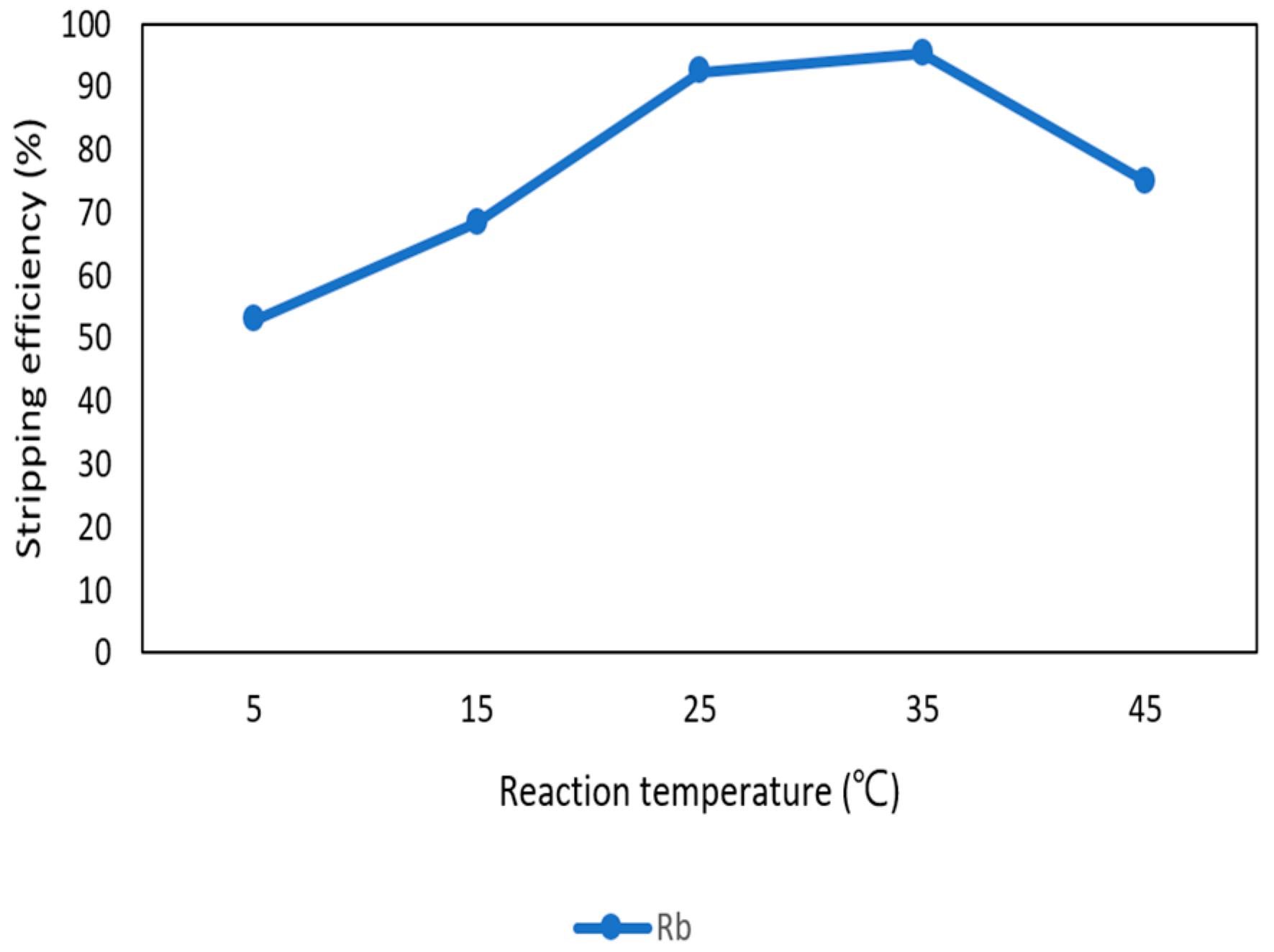
3.3.5. Effect of Reaction Temperature
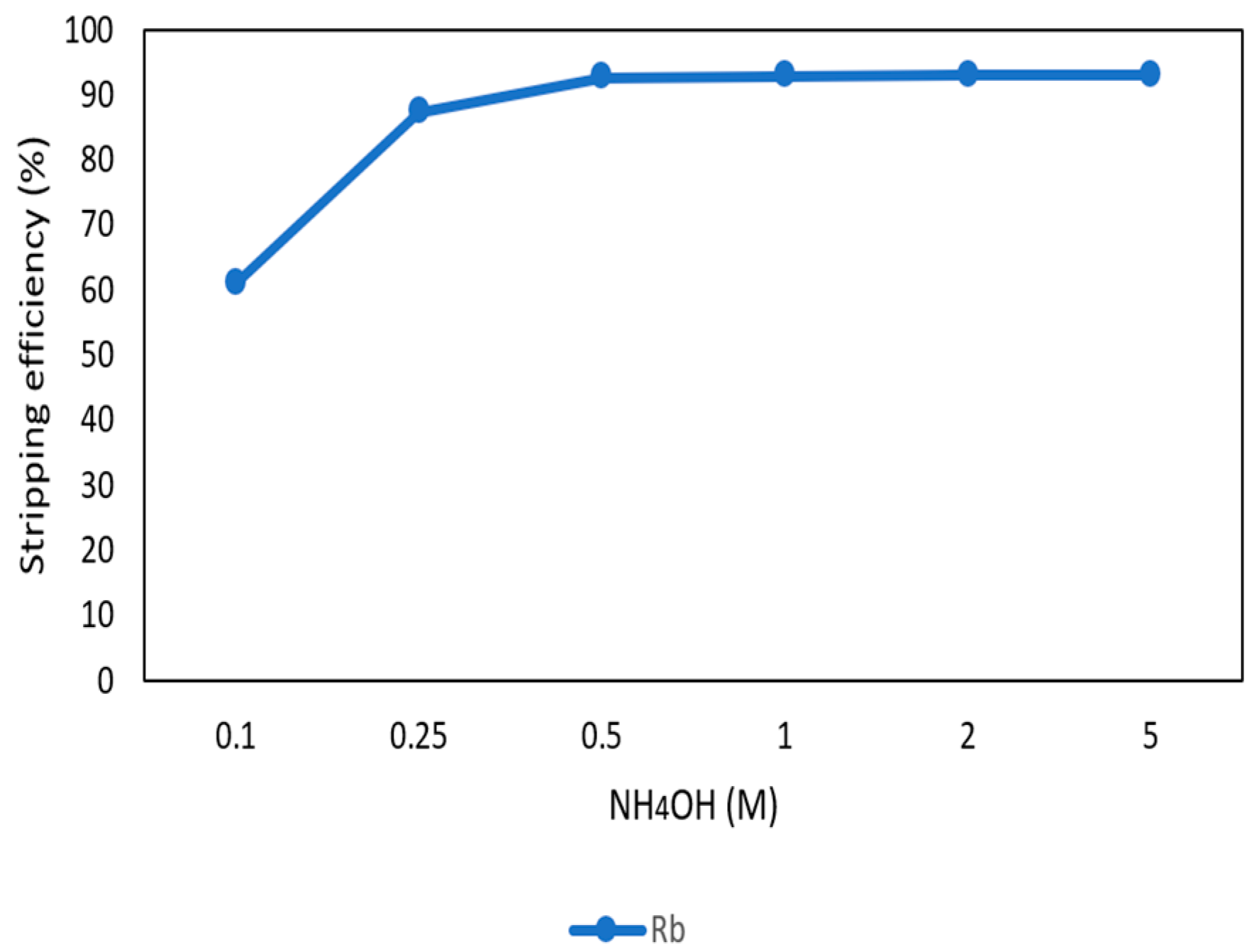
3.3.6. Stripping of Rb from the Organic Phase through Ammonia
3.4. Production of Rubidium Carbonate and Cesium Carbonate
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- The United Nations World Water Development Report 2012; UNESCO: Paris, France, 2012.
- Wangnick, K. 2004 IDA Worldwide Desalting Plants Inventory; Wangnick Consulting: Gnarrenburg, Germany, 2004. [Google Scholar]
- Kumar, A.; Phillips, K.; Thiel, G.; Schröder, U.; Lienhard, J. Direct electrosynthesis of sodium hydroxide and hydrochloric acid from brine streams. Nat. Catal. 2019, 2, 106–113. [Google Scholar] [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2015; U.S. Geological Survey: Reston, VA, USA, 2015; p. 196. [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2016; U.S. Geological Survey: Reston, VA, USA, 2016; p. 202. [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2017; U.S. Geological Survey: Reston, VA, USA, 2017; p. 202. [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2018; U.S. Geological Survey: Reston, VA, USA, 2018; p. 200. [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2019; U.S. Geological Survey: Reston, VA, USA, 2019; p. 200. [CrossRef]
- Li, Z.; Wakai, R.; Walker, T. Parametric modulation of an atomic magnetometer. Appl. Phys. Lett. 2006, 89, 134105. [Google Scholar] [CrossRef] [PubMed]
- Yen, C.; Yano, Y.; Budinger, T.; Friedland, R.; Derenzo, S.; Huesman, R. Brain tumor evaluation using pet and Rb-82. Clin. Nucl. Med. 1981, 6, 448. [Google Scholar] [CrossRef]
- Groeger, S.; Pazgalev, A.; Weis, A. Comparison of discharge lamp and laser pumped cesium magnetometers. Appl. Phys. B 2005, 80, 645–654. [Google Scholar] [CrossRef]
- Downs, J. Drilling and Completing Difficult HP/HT Wells With the Aid of Cesium Formate Brines-A Performance Review. In Proceedings of the IADC/SPE Drilling Conference, Miami, FL, USA, 21–23 February 2006. [Google Scholar] [CrossRef]
- Jiayong, C. Handbook of Hydrometallurgy; Metallurgical Industry Press Co., Ltd: Beijing, China, 2005. [Google Scholar]
- Paschalis, C.; Jenner, F.; Lee, C. Effects of Rubidium Chloride on the Course of Manic-Depressive Illness. J. R. Soc. Med. 1978, 71, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Malek-Ahmadi, P.; Williams, J. Rubidium in psychiatry: Research implications. Pharmacol. Biochem. Behav. 1984, 21, 49–50. [Google Scholar] [CrossRef]
- Canavese, C.; DeCostanzi, E.; Branciforte, L.; Caropreso, A.; Nonnato, A.; Sabbioni, E. Depression in dialysis patients: Rubidium supplementation before other drugs and encouragement? Kidney Int. 2001, 60, 1201. [Google Scholar] [CrossRef] [PubMed]
- Lake, J. Textbook of Integrative Mental Health Care; Thieme New York: New York, NY, USA, 2007. [Google Scholar]
- O’Neil, M.J. The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals; Whitehouse Station, N.J.: Merck: Readington, NJ, USA, 2001. [Google Scholar]
- Li, Z.; Pranolo, Y.; Zhu, Z.; Cheng, C. Solvent extraction of cesium and rubidium from brine solutions using 4-tert-butyl-2-(α-methylbenzyl)-phenol. Hydrometallurgy 2017, 171, 1–7. [Google Scholar] [CrossRef]
- Liu, S.; Liu, H.; Huang, Y.; Yang, W. Solvent extraction of rubidium and cesium from salt lake brine with t-BAMBP–kerosene solution. Trans. Nonferrous Met. Soc. China 2015, 25, 329–334. [Google Scholar] [CrossRef]
- Wang, J.; Che, D.; Qin, W. Extraction of rubidium by t-BAMBP in cyclohexane. Chin. J. Chem. Eng. 2015, 23, 1110–1113. [Google Scholar] [CrossRef]
- Xing, P.; Wang, G.; Wang, C.; Ma, B.; Chen, Y. Separation of rubidium from potassium in rubidium ore liquor by solvent extraction with t-BAMBP. Miner. Eng. 2018, 121, 158–163. [Google Scholar] [CrossRef]


| Element | Li | Na | K | Ca | Mg | Rb | Cs |
|---|---|---|---|---|---|---|---|
| Concentration (mg/L) | 182 | 50,130 | 5914 | 702 | 13,980 | 7.1 | 43.6 |
| Elements | Li | Na | K | Ca | Mg | Rb | Cs |
|---|---|---|---|---|---|---|---|
| Concentration (mg/L) | 167 | 491,80 | 91 | 622 | 135,70 | 6.94 | 42.14 |
| Extractant and Stripping Agent | pH Value of Aqueous Phase | Concentration (M) | O/A Ratio | Reaction Time (min) | Reaction Temperature (°C) |
|---|---|---|---|---|---|
| t-BAMBP | 8.0 | 0.1 | 0.1 | 3 | 35 |
| Ammonia | - | 1 | 2 | - | 25 |
| Extractant and Stripping Agent | pH Value of Aqueous Phase | Concentration (M) | O/A Ratio | Reaction Time (min) | Reaction Temperature (°C) |
|---|---|---|---|---|---|
| t-BAMBP | 12.0 | 0.5 | 0.1 | 15 | 5 |
| Ammonia | - | 0.5 | 2 | - | 35 |
| Element | Li | Na | K | Ca | Mg | Rb | Cs |
|---|---|---|---|---|---|---|---|
| Concentration (mg/L) | N.D. | 8.13 | 10.33 | N.D. | N.D. | 1.39 | 1635 |
| Element | Li | Na | K | Ca | Mg | Rb | Cs |
|---|---|---|---|---|---|---|---|
| Concentration (mg/L) | N.D. | 0.02 | 6.02 | N.D. | N.D. | 293.68 | N.D. |
| Compounds | Li | Na | K | Ca | Mg | Rb | Cs |
|---|---|---|---|---|---|---|---|
| Rb2CO3 | N.D. | 0.1% | 1.9% | N.D. | N.D. | 98.0% | N.D. |
| Cs2CO3 | N.D. | 0.4% | 0.5% | N.D. | N.D. | 0.2% | 98.9% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, W.-S.; Lee, C.-H.; Chung, Y.-F.; Tien, K.-W.; Chen, Y.-J.; Chen, Y.-A. Recovery of Rubidium and Cesium Resources from Brine of Desalination through t-BAMBP Extraction. Metals 2020, 10, 607. https://doi.org/10.3390/met10050607
Chen W-S, Lee C-H, Chung Y-F, Tien K-W, Chen Y-J, Chen Y-A. Recovery of Rubidium and Cesium Resources from Brine of Desalination through t-BAMBP Extraction. Metals. 2020; 10(5):607. https://doi.org/10.3390/met10050607
Chicago/Turabian StyleChen, Wei-Sheng, Cheng-Han Lee, Yi-Fan Chung, Ko-Wei Tien, Yen-Jung Chen, and Yu-An Chen. 2020. "Recovery of Rubidium and Cesium Resources from Brine of Desalination through t-BAMBP Extraction" Metals 10, no. 5: 607. https://doi.org/10.3390/met10050607
APA StyleChen, W.-S., Lee, C.-H., Chung, Y.-F., Tien, K.-W., Chen, Y.-J., & Chen, Y.-A. (2020). Recovery of Rubidium and Cesium Resources from Brine of Desalination through t-BAMBP Extraction. Metals, 10(5), 607. https://doi.org/10.3390/met10050607







