Accelerating Recovery from Exercise-Induced Muscle Injuries in Triathletes: Considerations for Olympic Distance Races
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
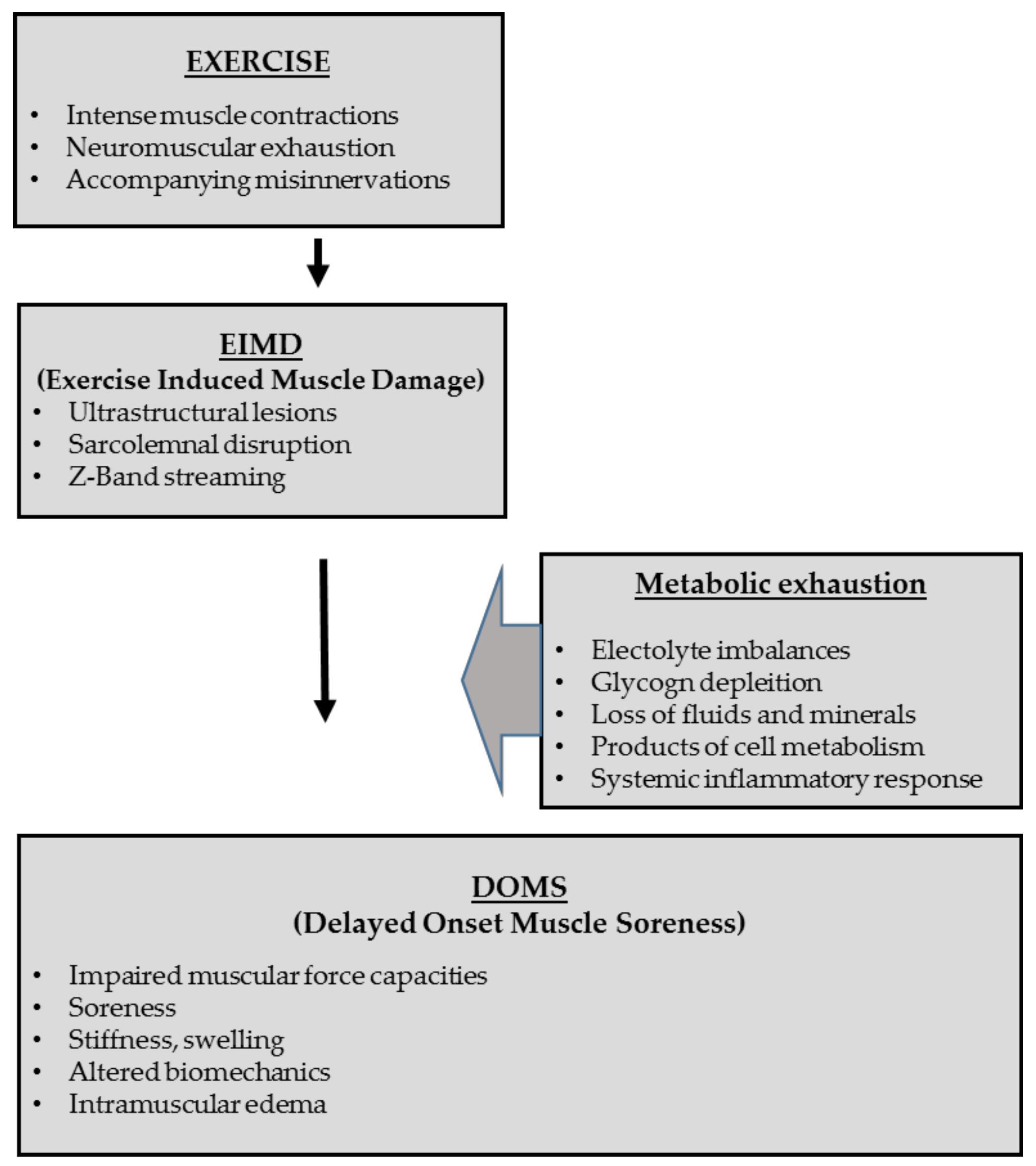
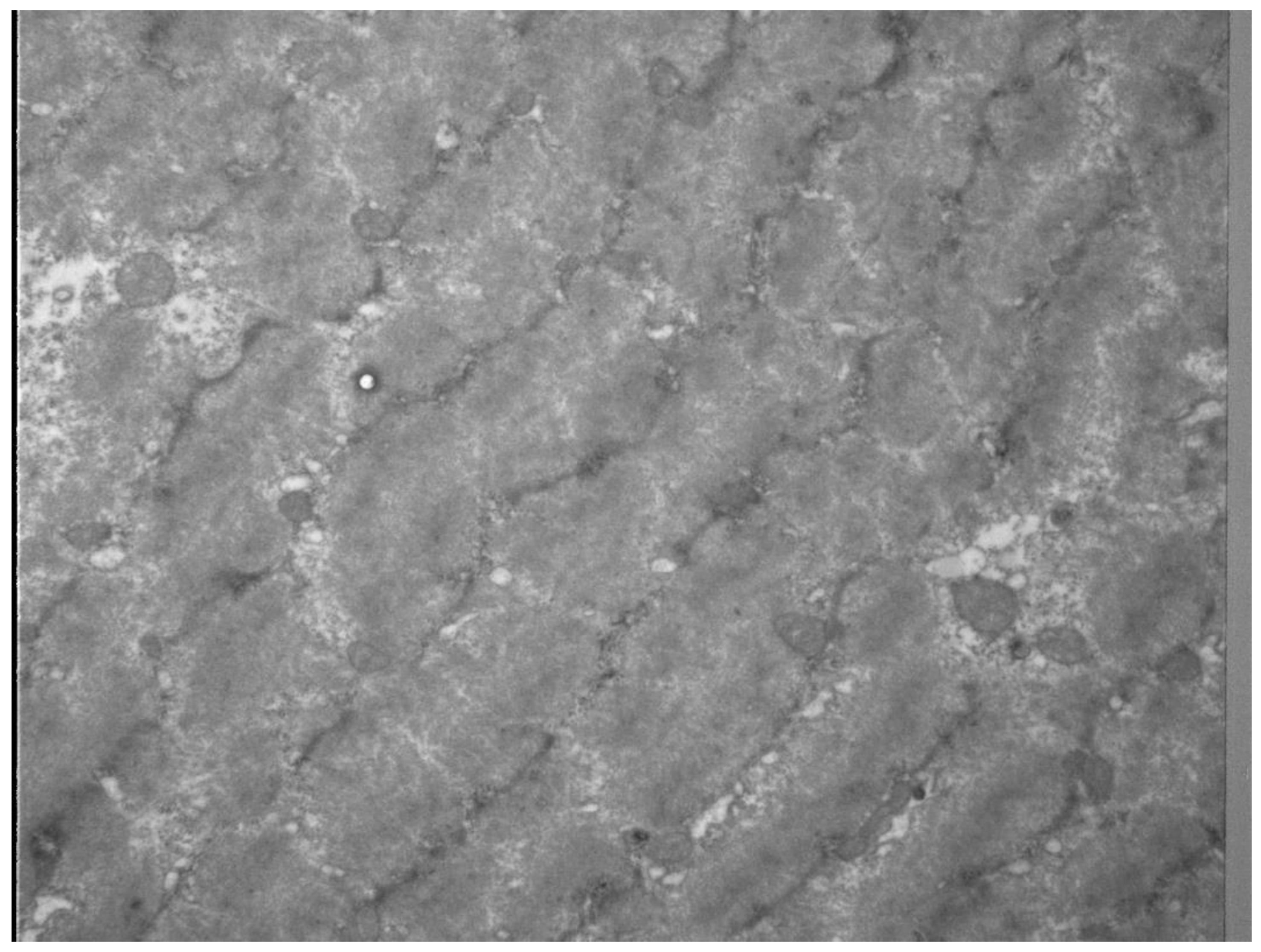
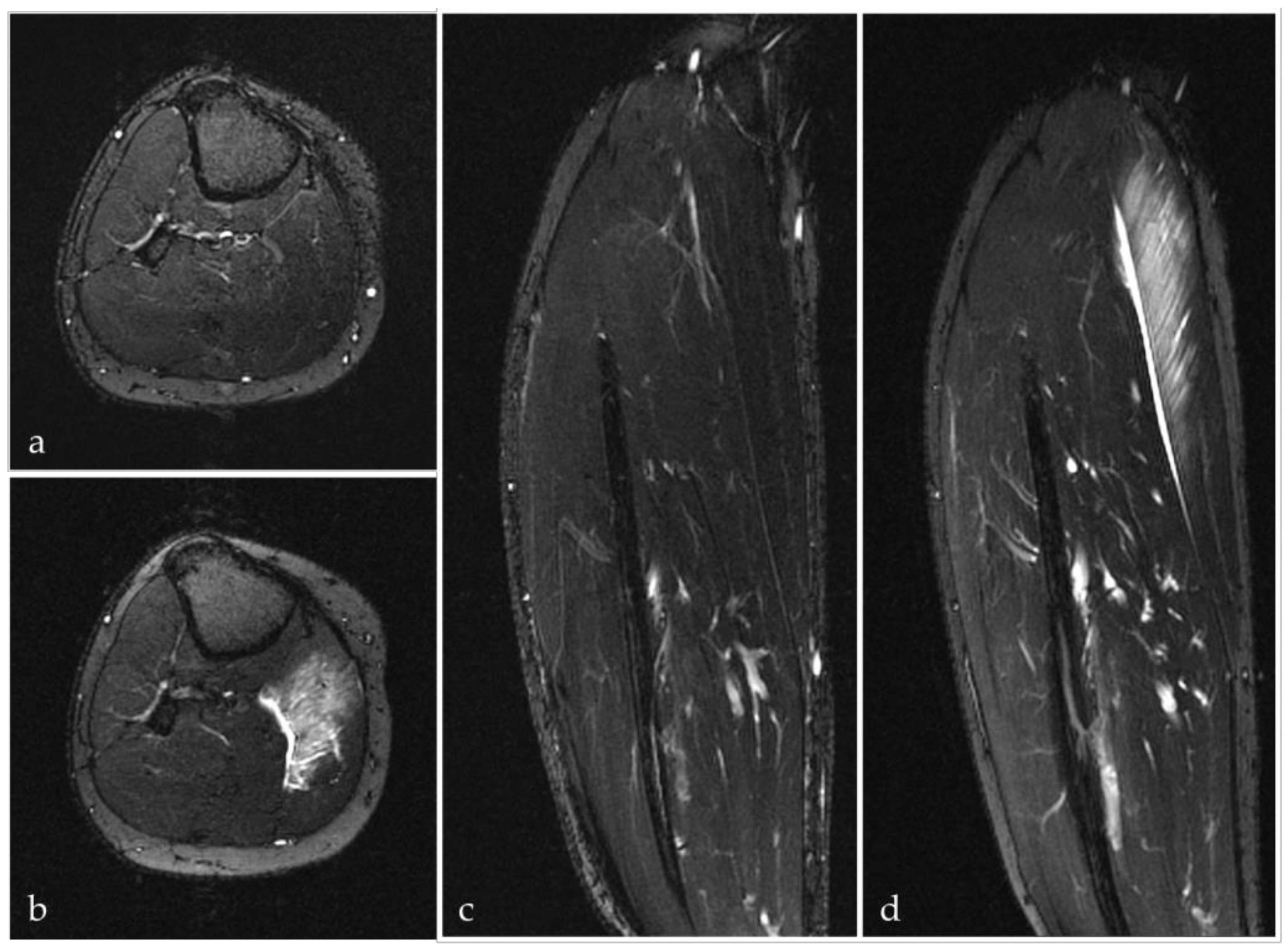
3.1. Mechanisms and Pathogenesis of Exercise-Induced Muscle Injures
3.1.1. Differentiation between Muscle Soreness, Exercise-Induced Muscle Damage (EIMD) and Delayed Onset Muscle Soreness (DOMS)
3.1.2. Inflammatory and Healing Responses
4. Treatments and Strategies to Target Exercise-Induced Muscle Injuries
4.1. The Role of Sleep
4.2. Compression Therapy
4.2.1. Compression Therapy during Exercise
4.2.2. Compression Therapy Post Exercise
4.2.3. Intermittent Compression Therapy
4.3. Thermal Therapy
4.3.1. Cold Water Immersion Therapy (CWI)
4.3.2. Heat Therapy
4.4. Active Regeneration
4.5. Nutrition
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kienstra, C.M.; Asken, T.R.; Garcia, J.D.; Lara, V.; Best, T.M. Triathlon Injuries: Transitioning from Prevalence to Prediction and Prevention. Curr. Sports Med. Rep. 2017, 16, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Olcina, G.; Timon, R.; Brazo-Sayavera, J.; Martinez-Guardado, I.; Marcos-Serrano, M.; Crespo, C. Changes in physiological and performance variables in non-professional triathletes after taking part in an Olympic distance triathlon. Res. Sports Med. (Print) 2018, 26, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Neidel, P.; Wolfram, P.; Hotfiel, T.; Engelhardt, M.; Koch, R.; Lee, G.; Zwingenberger, S. Cross-Sectional Investigation of Stress Fractures in German Elite Triathletes. Sports 2019, 7, 88. [Google Scholar] [CrossRef] [PubMed]
- International Triathlon Union. Available online: https://www.triathlon.org/ (accessed on 1 May 2019).
- Jeukendrup, A.E.; Jentjens, R.L.; Moseley, L. Nutritional considerations in triathlon. Sports Med. (Auckl. NZ) 2005, 35, 163–181. [Google Scholar] [CrossRef] [PubMed]
- Del Coso, J.; Gonzalez-Millan, C.; Salinero, J.J.; Abian-Vicen, J.; Soriano, L.; Garde, S.; Perez-Gonzalez, B. Muscle damage and its relationship with muscle fatigue during a half-iron triathlon. PLoS ONE 2012, 7, e43280. [Google Scholar] [CrossRef]
- Hiller, W.D. Dehydration and hyponatremia during triathlons. Med. Sci. Sports Exerc. 1989, 21, S219–S221. [Google Scholar]
- Speedy, D.B.; Noakes, T.D.; Kimber, N.E.; Rogers, I.R.; Thompson, J.M.; Boswell, D.R.; Ross, J.J.; Campbell, R.G.; Gallagher, P.G.; Kuttner, J.A. Fluid balance during and after an ironman triathlon. Clin. J. Sport Med. 2001, 11, 44–50. [Google Scholar] [CrossRef]
- Margaritis, I.; Tessier, F.; Verdera, F.; Bermon, S.; Marconnet, P. Muscle enzyme release does not predict muscle function impairment after triathlon. J. Sports Med. Phys. Fit. 1999, 39, 133–139. [Google Scholar]
- Galan, B.S.; Carvalho, F.G.; Santos, P.C.; Gobbi, R.B.; Kalva-Filho, C.A.; Papoti, M.; da Silva, A.S.; Freitas, E.C. Effects of taurine on markers of muscle damage, inflammatory response and physical performance in triathletes. J. Sports Med. Phys. Fit. 2018, 58, 1318–1324. [Google Scholar] [CrossRef]
- Fernandez-Revelles, A.B. Infographic. Correlation between phases and final result in Men’s triathlon competition at the Olympic Games in Sydney 2000. Br. J. Sports Med. 2018. [Google Scholar] [CrossRef]
- Heiss, R.; Freiwald, J.; Hoppe, M.W.; Lutter, C.; Forst, R.; Grim, C.; Poettgen, K.; Bloch, W.; Huttel, M.; Hotfiel, T. Advances in Delayed-Onset Muscle Soreness (DOMS): Part II: Treatment and Prevention. Sportverletz. Sportschaden 2019, 33, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Crowther, F.; Sealey, R.; Crowe, M.; Edwards, A.; Halson, S. Influence of recovery strategies upon performance and perceptions following fatiguing exercise: A randomized controlled trial. BMC Sports Sci. Med. Rehabil. 2017, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Hausswirth, C.; Mujika, I. Recovery for Performance in Sport; Human Kinetics: Champaign, IL, USA, 2013. [Google Scholar]
- Meyer, T.; Ferrauti, A.; Kellmann, M.; Pfeiffer, M. Regenerationsmanagement im Spitzensport. REGman-Ergebnisse und Handlungsempfehlungen; Sportverlag Strauß: Hellenthal, Germany, 2016. [Google Scholar]
- Hotfiel, T.; Seil, R.; Bily, W.; Bloch, W.; Gokeler, A.; Krifter, R.M.; Mayer, F.; Ueblacker, P.; Weisskopf, L.; Engelhardt, M. Nonoperative treatment of muscle injuries–recommendations from the GOTS expert meeting. J. Exp. Orthop. 2018, 5, 24. [Google Scholar] [CrossRef] [PubMed]
- Pearcey, G.E.; Bradbury-Squires, D.J.; Kawamoto, J.E.; Drinkwater, E.J.; Behm, D.G.; Button, D.C. Foam rolling for delayed-onset muscle soreness and recovery of dynamic performance measures. J. Athl. Train. 2015, 50, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Valle, X.; Til, L.; Drobnic, F.; Turmo, A.; Montoro, J.B.; Valero, O.; Artells, R. Compression garments to prevent delayed onset muscle soreness in soccer players. Muscles Ligaments Tendons J. 2013, 3, 295–302. [Google Scholar] [CrossRef]
- Kim, S.K.; Kim, M.C. The affect on delayed onset muscle soreness recovery for ultrasound with bee venom. J. Phys. Ther. Sci. 2014, 26, 1419–1421. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, S.; Morii, I.; Tsuchiya, Y.; Goto, K. Wearing Compression Garment after Endurance Exercise Promotes Recovery of Exercise Performance. Int. J. Sports Med. 2016, 37, 870–877. [Google Scholar] [CrossRef]
- Hotfiel, T.; Kellermann, M.; Swoboda, B.; Wildner, D.; Golditz, T.; Grim, C.; Raithel, M.; Uder, M.; Heiss, R. Application of Acoustic Radiation Force Impulse (ARFI) Elastography in Imaging of Delayed Onset Muscle Soreness (DOMS): A Comparative Analysis With 3T MRI. J. Sport Rehabil. 2017, 1–29. [Google Scholar] [CrossRef]
- Pollock, N.; James, S.L.J.; Lee, J.C.; Chakraverty, R. British athletics muscle injury classification: A new grading system. Br. J. Sports Med. 2014, 48. [Google Scholar] [CrossRef]
- Mueller-Wohlfahrt, H.W.; Haensel, L.; Mithoefer, K.; Ekstrand, J.; English, B.; McNally, S.; Orchard, J.; van Dijk, C.N.; Kerkhoffs, G.M.; Schamasch, P.; et al. Terminology and classification of muscle injuries in sport: The Munich consensus statement. Br. J. Sports Med. 2013, 47, 342–350. [Google Scholar] [CrossRef]
- Heiss, R.; Kellermann, M.; Swoboda, B.; Grim, C.; Lutter, C.; May, M.S.; Wuest, W.; Uder, M.; Nagel, A.M.; Hotfiel, T. Effect of Compression Garments on the Development of Delayed-Onset Muscle Soreness: A Multimodal Approach Using Contrast-Enhanced Ultrasound and Acoustic Radiation Force Impulse Elastography. J. Orthop. Sports Phys. Ther. 2018, 48, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.Y.; Jeong, J.G.; Lee, B.H. Evaluation of muscle damage using ultrasound imaging. J. Phys. Ther. Sci. 2015, 27, 531–534. [Google Scholar] [CrossRef] [PubMed]
- Kellermann, M.H.M.; Swoboda, B.; Gelse, K.; Freiwald, J.; Grim, C.; Nagel, A.; Uder, M.; Wildner, D.; Hotfiel, T. Intramuscular perfusion response in delayed onset muscle soreness (DOMS): A quantitative analysis with contrast-enhanced ultrasound (CEUS). Int. J. Sports Med. 2017, 38, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, G.; Mikkelsen, U.R.; Raastad, T.; Peake, J.M. Leucocytes, cytokines and satellite cells: What role do they play in muscle damage and regeneration following eccentric exercise? Exerc. Immunol. Rev. 2012, 18, 42–97. [Google Scholar] [PubMed]
- Hotfiel, T.; Freiwald, J.; Hoppe, M.W.; Lutter, C.; Forst, R.; Grim, C.; Bloch, W.; Huttel, M.; Heiss, R. Advances in Delayed-Onset Muscle Soreness (DOMS): Part I: Pathogenesis and Diagnostics. Sportverletz. Sportschaden 2018, 32, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.; Nosaka, K.; Suzuki, K. Characterization of inflammatory responses to eccentric exercise in humans. Exerc. Immunol. Rev. 2005, 11, 64–85. [Google Scholar] [PubMed]
- Lewis, P.B.; Ruby, D.; Bush-Joseph, C.A. Muscle soreness and delayed-onset muscle soreness. Clin. Sports Med. 2012, 31, 255–262. [Google Scholar] [CrossRef]
- Douglas, J.; Pearson, S.; Ross, A.; McGuigan, M. Eccentric Exercise: Physiological Characteristics and Acute Responses. Sports Med. (Auckl. NZ) 2017, 47, 663–675. [Google Scholar] [CrossRef]
- Linari, M.; Lucii, L.; Reconditi, M.; Casoni, M.E.; Amenitsch, H.; Bernstorff, S.; Piazzesi, G.; Lombardi, V. A combined mechanical and X-ray diffraction study of stretch potentiation in single frog muscle fibres. J. Physiol. 2000, 526 Pt 3, 589–596. [Google Scholar] [CrossRef]
- Nishikawa, K.C.; Monroy, J.A.; Uyeno, T.E.; Yeo, S.H.; Pai, D.K.; Lindstedt, S.L. Is titin a ‘winding filament’? A new twist on muscle contraction. Proc. Biol. Sci. 2012, 279, 981–990. [Google Scholar] [CrossRef]
- Herzog, W. Mechanisms of enhanced force production in lengthening (eccentric) muscle contractions. J. Appl. Physiol. (Bethesda MD 1985) 2014, 116, 1407–1417. [Google Scholar] [CrossRef] [PubMed]
- Nicol, C.; Avela, J.; Komi, P.V. The stretch-shortening cycle: A model to study naturally occurring neuromuscular fatigue. Sports Med. (Auckl. NZ) 2006, 36, 977–999. [Google Scholar] [CrossRef] [PubMed]
- Tesch, P.A.; Fernandez-Gonzalo, R.; Lundberg, T.R. Clinical Applications of Iso-Inertial, Eccentric-Overload (YoYo) Resistance Exercise. Front. Physiol. 2017, 8, 241. [Google Scholar] [CrossRef] [PubMed]
- Pinho, R.A.; Silva, L.A.; Pinho, C.A.; Scheffer, D.L.; Souza, C.T.; Benetti, M.; Carvalho, T.; Dal-Pizzol, F. Oxidative stress and inflammatory parameters after an Ironman race. Clin. J. Sport Med. 2010, 20, 306–311. [Google Scholar] [CrossRef] [PubMed]
- Yanagisawa, O.; Sakuma, J.; Kawakami, Y.; Suzuki, K.; Fukubayashi, T. Effect of exercise-induced muscle damage on muscle hardness evaluated by ultrasound real-time tissue elastography. Springerplus 2015, 4, 308. [Google Scholar] [CrossRef] [PubMed]
- Nie, H.; Madeleine, P.; Arendt-Nielsen, L.; Graven-Nielsen, T. Temporal summation of pressure pain during muscle hyperalgesia evoked by nerve growth factor and eccentric contractions. Eur. J. Pain 2009, 13, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, J. A review of nutritional intervention on delayed onset muscle soreness. Part I. J. Exerc. Rehabil. 2014, 10, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Bush, J.A.; Wickham, R.B.; Denegar, C.R.; Gomez, A.L.; Gotshalk, L.A.; Duncan, N.D.; Volek, J.S.; Putukian, M.; Sebastianelli, W.J. Influence of compression therapy on symptoms following soft tissue injury from maximal eccentric exercise. J. Orthop. Sports Phys. Ther. 2001, 31, 282–290. [Google Scholar] [CrossRef]
- Ulbricht, A.; Gehlert, S.; Leciejewski, B.; Schiffer, T.; Bloch, W.; Hohfeld, J. Induction and adaptation of chaperone-assisted selective autophagy CASA in response to resistance exercise in human skeletal muscle. Autophagy 2015, 11, 538–546. [Google Scholar] [CrossRef]
- Deme, D.; Telekes, A. Prognostic importance of lactate dehydrogenase (LDH) in oncology. Orv. Hetil. 2017, 158, 1977–1988. [Google Scholar] [CrossRef]
- Del Giudice, M.; Gangestad, S.W. Rethinking IL-6 and CRP: Why They Are More Than Inflammatory Biomarkers, and Why It Matters. Brain Behav. Immun. 2018. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S.S.H.; Teo, W.P.; Warmington, S.A. Effects of training and competition on the sleep of elite athletes: A systematic review and meta-analysis. Br. J. Sports Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Hausswirth, C.; Louis, J.; Aubry, A.; Bonnet, G.; Duffield, R.; Le Meur, Y. Evidence of disturbed sleep and increased illness in overreached endurance athletes. Med. Sci. Sports Exerc. 2014, 46, 1036–1045. [Google Scholar] [CrossRef] [PubMed]
- Halson, S.L. Sleep in elite athletes and nutritional interventions to enhance sleep. Sports Med. (Auckl. NZ) 2014, 44 (Suppl. 1), S13–S23. [Google Scholar] [CrossRef] [PubMed]
- Watson, A.M. Sleep and Athletic Performance. Curr. Sports Med. Rep. 2017, 16, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Kirschen, G.W.; Jones, J.J.; Hale, L. The Impact of Sleep Duration on Performance Among Competitive Athletes: A Systematic Literature Review. Clin. J. Sport Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Bonnar, D.; Bartel, K.; Kakoschke, N.; Lang, C. Sleep Interventions Designed to Improve Athletic Performance and Recovery: A Systematic Review of Current Approaches. Sports Med. (Auckl. NZ) 2018, 48, 683–703. [Google Scholar] [CrossRef]
- Thun, E.; Bjorvatn, B.; Flo, E.; Harris, A.; Pallesen, S. Sleep, circadian rhythms, and athletic performance. Sleep Med. Rev. 2015, 23, 1–9. [Google Scholar] [CrossRef]
- Milner, C.E.; Cote, K.A. Benefits of napping in healthy adults: Impact of nap length, time of day, age, and experience with napping. J. Sleep Res. 2009, 18, 272–281. [Google Scholar] [CrossRef]
- Waterhouse, J.; Atkinson, G.; Edwards, B.; Reilly, T. The role of a short post-lunch nap in improving cognitive, motor, and sprint performance in participants with partial sleep deprivation. J. Sports Sci. 2007, 25, 1557–1566. [Google Scholar] [CrossRef]
- Simpson, N.S.; Gibbs, E.L.; Matheson, G.O. Optimizing sleep to maximize performance: Implications and recommendations for elite athletes. Scand. J. Med. Sci. Sports 2017, 27, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.; Howatson, G.; van Someren, K.; Leeder, J.; Pedlar, C. Compression garments and recovery from exercise-induced muscle damage: A meta-analysis. Br. J. Sports Med. 2014, 48, 1340–1346. [Google Scholar] [CrossRef] [PubMed]
- Marques-Jimenez, D.; Calleja-Gonzalez, J.; Arratibel, I.; Delextrat, A.; Terrados, N. Are compression garments effective for the recovery of exercise-induced muscle damage? A systematic review with meta-analysis. Physiol. Behav. 2016, 153, 133–148. [Google Scholar] [CrossRef] [PubMed]
- Beliard, S.; Chauveau, M.; Moscatiello, T.; Cros, F.; Ecarnot, F.; Becker, F. Compression garments and exercise: No influence of pressure applied. J. Sports Sci. Med. 2015, 14, 75. [Google Scholar] [PubMed]
- da Silva, C.A.; Helal, L.; da Silva, R.P.; Belli, K.C.; Umpierre, D.; Stein, R. Association of Lower Limb Compression Garments During High-Intensity Exercise with Performance and Physiological Responses: A Systematic Review and Meta-analysis. Sports Med. 2018, 48, 1859–1873. [Google Scholar] [CrossRef] [PubMed]
- Venckunas, T.; Trinkunas, E.; Kamandulis, S.; Poderys, J.; Grunovas, A.; Brazaitis, M. Effect of lower body compression garments on hemodynamics in response to running session. Sci. World J. 2014, 2014, 353040. [Google Scholar] [CrossRef] [PubMed]
- Duffield, R.; Portus, M. Comparison of three types of full-body compression garments on throwing and repeat-sprint performance in cricket players. Br. J. Sports Med. 2007, 41, 409–414; discussion 414. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J.; Lee, J. Effect of compression garments on delayed-onset muscle soreness and blood inflammatory markers after eccentric exercise: A randomized controlled trial. J. Exerc. Rehabil. 2017, 13, 541–545. [Google Scholar] [CrossRef]
- Trenell, M.I.; Rooney, K.B.; Sue, C.M.; Thomspon, C.H. Compression Garments and Recovery from Eccentric Exercise: A (31)P-MRS Study. J. Sports Sci. Med. 2006, 5, 106–114. [Google Scholar]
- Bringard, A.; Perrey, S.; Belluye, N. Aerobic energy cost and sensation responses during submaximal running exercise--positive effects of wearing compression tights. Int. J. Sports Med. 2006, 27, 373–378. [Google Scholar] [CrossRef]
- Ali, A.; Caine, M.P.; Snow, B.G. Graduated compression stockings: Physiological and perceptual responses during and after exercise. J. Sports Sci. 2007, 25, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Sperlich, B.; Born, D.-P.; Kaskinoro, K.; Kalliokoski, K.K.; Laaksonen, M.S. Squeezing the muscle: Compression clothing and muscle metabolism during recovery from high intensity exercise. PLoS ONE 2013, 8, e60923. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; von Stengel, S.; Kockritz, C.; Mayhew, J.; Wassermann, A.; Zapf, J. Effect of compression stockings on running performance in men runners. J. Strength Cond. Res. 2009, 23, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Hamlin, M.J.; Mitchell, C.J.; Ward, F.D.; Draper, N.; Shearman, J.P.; Kimber, N.E. Effect of compression garments on short-term recovery of repeated sprint and 3-km running performance in rugby union players. J. Strength Cond. Res. 2012, 26, 2975–2982. [Google Scholar] [CrossRef]
- Chatard, J.C.; Atlaoui, D.; Farjanel, J.; Louisy, F.; Rastel, D.; Guezennec, C.Y. Elastic stockings, performance and leg pain recovery in 63-year-old sportsmen. Eur. J. Appl. Physiol. 2004, 93, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.S.; Borges, A.R.; Beck, D.T. Peripheral conduit and resistance artery function are improved following a single, 1-h bout of peristaltic pulse external pneumatic compression. Eur. J. Appl. Physiol. 2015, 115, 2019–2029. [Google Scholar] [CrossRef] [PubMed]
- Keck, N.A.; Cuddy, J.S.; Hailes, W.S.; Dumke, C.L.; Ruby, B.C. Effects of commercially available pneumatic compression on muscle glycogen recovery after exercise. J. Strength Cond. Res. 2015, 29, 379–385. [Google Scholar] [CrossRef]
- Haun, C.T.; Roberts, M.D.; Romero, M.A.; Osburn, S.C.; Mobley, C.B.; Anderson, R.G.; Goodlett, M.D.; Pascoe, D.D.; Martin, J.S. Does external pneumatic compression treatment between bouts of overreaching resistance training sessions exert differential effects on molecular signaling and performance-related variables compared to passive recovery? An exploratory study. PLoS ONE 2017, 12, e0180429. [Google Scholar] [CrossRef]
- Machado, A.F.; Ferreira, P.H.; Micheletti, J.K.; de Almeida, A.C.; Lemes, I.R.; Vanderlei, F.M.; Netto Junior, J.; Pastre, C.M. Can Water Temperature and Immersion Time Influence the Effect of Cold Water Immersion on Muscle Soreness? A Systematic Review and Meta-Analysis. Sports Med. (Auckl. NZ) 2016, 46, 503–514. [Google Scholar] [CrossRef]
- Hohenauer, E.; Taeymans, J.; Baeyens, J.P.; Clarys, P.; Clijsen, R. The Effect of Post-Exercise Cryotherapy on Recovery Characteristics: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0139028. [Google Scholar] [CrossRef]
- Leeder, J.; Gissane, C.; van Someren, K.; Gregson, W.; Howatson, G. Cold water immersion and recovery from strenuous exercise: A meta-analysis. Br. J. Sports Med. 2012, 46, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Stocks, J.M.; Patterson, M.J.; Hyde, D.E.; Jenkins, A.B.; Mittleman, K.D.; Taylor, N.A. Effects of immersion water temperature on whole-body fluid distribution in humans. Acta Physiol. Scand. 2004, 182, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Wilcock, I.M.; Cronin, J.B.; Hing, W.A. Physiological response to water immersion: A method for sport recovery? Sports Med. 2006, 36, 747–765. [Google Scholar] [CrossRef] [PubMed]
- Proudfoot, C.J.; Garry, E.M.; Cottrell, D.F.; Rosie, R.; Anderson, H.; Robertson, D.C.; Fleetwood-Walker, S.M.; Mitchell, R. Analgesia mediated by the TRPM8 cold receptor in chronic neuropathic pain. Curr. Biol. 2006, 16, 1591–1605. [Google Scholar] [CrossRef] [PubMed]
- Swenson, C.; Sward, L.; Karlsson, J. Cryotherapy in sports medicine. Scand. J. Med. Sci. Sports 1996, 6, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Ihsan, M.; Watson, G.; Abbiss, C.R. What are the physiological mechanisms for post-exercise cold water immersion in the recovery from prolonged endurance and intermittent exercise? Sports Med. 2016, 46, 1095–1109. [Google Scholar] [CrossRef] [PubMed]
- Merrick, M.A.; Rankin, J.M.; Andres, F.A.; Hinman, C.L. A preliminary examination of cryotherapy and secondary injury in skeletal muscle. Med. Sci. Sports Exerc. 1999, 31, 1516–1521. [Google Scholar] [CrossRef] [PubMed]
- Blackwood, S.J.; Hanya, E.; Katz, A. Effect of post-exercise temperature elevation on post-exercise glycogen metabolism of isolated mouse soleus muscle. J. Appl. Physiol. (1985) 2019. [Google Scholar] [CrossRef] [PubMed]
- Saga, N.; Katamoto, S.; Naito, H. Effect of heat preconditioning by microwave hyperthermia on human skeletal muscle after eccentric exercise. J. Sports Sci. Med. 2008, 7, 176–183. [Google Scholar] [PubMed]
- Cernych, M.; Baranauskiene, N.; Vitkauskiene, A.; Satas, A.; Brazaitis, M. Accelerated muscle contractility and decreased muscle steadiness following sauna recovery do not induce greater neuromuscular fatigability during sustained submaximal contractions. Hum. Mov. Sci. 2019, 63, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Halvorson, G.A. Therapeutic Heat and Cold for Athletic Injuries. Phys. Sportsmed. 1990, 18, 87–94. [Google Scholar] [CrossRef]
- Lohman, E.B., 3rd; Bains, G.S.; Lohman, T.; DeLeon, M.; Petrofsky, J.S. A comparison of the effect of a variety of thermal and vibratory modalities on skin temperature and blood flow in healthy volunteers. Med. Sci. Monit. 2011, 17, MT72–81. [Google Scholar] [CrossRef] [PubMed]
- Goto, K.; Oda, H.; Kondo, H.; Igaki, M.; Suzuki, A.; Tsuchiya, S.; Murase, T.; Hase, T.; Fujiya, H.; Matsumoto, I.; et al. Responses of muscle mass, strength and gene transcripts to long-term heat stress in healthy human subjects. Eur. J. Appl. Physiol. 2011, 111, 17–27. [Google Scholar] [CrossRef]
- Kim, K.; Kuang, S.; Song, Q.; Gavin, T.P.; Roseguini, B.T. Impact of heat therapy on recovery following eccentric exercise in humans. J. Appl. Physiol. (1985) 2019. [Google Scholar] [CrossRef]
- Frier, B.C.; Locke, M. Heat stress inhibits skeletal muscle hypertrophy. Cell Stress Chaperones 2007, 12, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Freiwald, J. Optimales Dehnen: Sport-Prävention-Rehabilitation; Spitta Verlag: Balingen, Germany, 2013. [Google Scholar]
- Cheung, K.; Hume, P.A.; Maxwell, L. Delayed Onset Muscle Soreness. Sports Med. 2003, 33, 145–164. [Google Scholar] [CrossRef]
- Weber, M.D.; Servedio, F.J.; Woodall, W.R. The effects of three modalities on delayed onset muscle soreness. J. Orthop. Sports Phys. Ther. 1994, 20, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Gulick, D.T.; Kimura, I.F.; Sitler, M.; Paolone, A.; Kelly IV, J.D. Various treatment techniques on signs and symptoms of delayed onset muscle soreness. J. Athl. Train. 1996, 31, 145. [Google Scholar]
- Hasson, S.M.; Williams, J.; Signorile, J. Fatigue-induced changes in myoelectric signal characteristics and perceived exertion. Can. J. Sport Sci. 1989, 14, 99–102. [Google Scholar]
- Vanderthommen, M.; Makrof, S.; Demoulin, C. Comparison of active and electrostimulated recovery strategies after fatiguing exercise. J. Sports Sci. Med. 2010, 9, 164–169. [Google Scholar]
- Gupta, S.; Goswami, A.; Sadhukhan, A.K.; Mathur, D.N. Comparative study of lactate removal in short term massage of extremities, active recovery and a passive recovery period after supramaximal exercise sessions. Int. J. Sports Med. 1996, 17, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Bangsbo, J.; Graham, T.; Johansen, L.; Saltin, B. Muscle lactate metabolism in recovery from intense exhaustive exercise: Impact of light exercise. J. Appl. Physiol. (1985) 1994, 77, 1890–1895. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Feng, B.; Chen, K.; Andersen, L.L.; Page, P.; Wang, Y. The Efficacy of Dynamic Contract-Relax Stretching on Delayed-Onset Muscle Soreness Among Healthy Individuals: A Randomized Clinical Trial. Clin. J. Sport Med. 2018, 28, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Torres, R.; Ribeiro, F.; Alberto Duarte, J.; Cabri, J.M. Evidence of the physiotherapeutic interventions used currently after exercise-induced muscle damage: Systematic review and meta-analysis. Phys. Ther. Sport 2012, 13, 101–114. [Google Scholar] [CrossRef]
- Macdonald, G.Z.; Button, D.C.; Drinkwater, E.J.; Behm, D.G. Foam rolling as a recovery tool after an intense bout of physical activity. Med. Sci. Sports Exerc. 2014, 46, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Jay, K.; Sundstrup, E.; Sondergaard, S.D.; Behm, D.; Brandt, M.; Saervoll, C.A.; Jakobsen, M.D.; Andersen, L.L. Specific and cross over effects of massage for muscle soreness: Randomized controlled trial. Int. J. Sports Phys. Ther. 2014, 9, 82–91. [Google Scholar]
- Fleckenstein, J.; Wilke, J.; Vogt, L.; Banzer, W. Preventive and Regenerative Foam Rolling are Equally Effective in Reducing Fatigue-Related Impairments of Muscle Function following Exercise. J. Sports Sci. Med. 2017, 16, 474–479. [Google Scholar] [PubMed]
- Kalen, A.; Perez-Ferreiros, A.; Barcala-Furelos, R.; Fernandez-Mendez, M.; Padron-Cabo, A.; Prieto, J.A.; Rios-Ave, A.; Abelairas-Gomez, C. How can lifeguards recover better? A cross-over study comparing resting, running, and foam rolling. Am. J. Emerg. Med. 2017, 35, 1887–1891. [Google Scholar] [CrossRef] [PubMed]
- Freiwald, J.; Baumgart, C.; Kühnemann, M.; Hoppe, M.W. Foam-Rolling in sport and therapy–Potential benefits and risks. Sports Orthop. Traumatol. 2016, 32, 258–266. [Google Scholar] [CrossRef]
- Freiwald, J.; Baumgart, C.; Kühnemann, M.; Hoppe, M.W. Foam-Rolling in sport and therapy – Potential benefits and risks. Sports Orthopaedics and Traumatology 2016, 32, 267–275. [Google Scholar] [CrossRef]
- Doering, T.M.; Reaburn, P.R.; Phillips, S.M.; Jenkins, D.G. Postexercise Dietary Protein Strategies to Maximize Skeletal Muscle Repair and Remodeling in Masters Endurance Athletes: A Review. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Harper, A.E.; Miller, R.H.; Block, K.P. Branched-chain amino acid metabolism. Annu. Rev. Nutr. 1984, 4, 409–454. [Google Scholar] [CrossRef] [PubMed]
- Foure, A.; Bendahan, D. Is Branched-Chain Amino Acids Supplementation an Efficient Nutritional Strategy to Alleviate Skeletal Muscle Damage? A Systematic Review. Nutrients 2017, 9, 1047. [Google Scholar] [CrossRef]
- Salinas-Garcia, M.E.; Martinez-Sanz, J.M.; Urdampilleta, A.; Mielgo-Ayuso, J.; Norte Navarro, A.; Ortiz-Moncada, R. Effects of branched amino acids in endurance sports: A review. Nutr. Hosp. 2014, 31, 577–589. [Google Scholar] [CrossRef] [PubMed]
- Hurley, C.F.; Hatfield, D.L.; Riebe, D.A. The effect of caffeine ingestion on delayed onset muscle soreness. J. Strength Cond. Res. 2013, 27, 3101–3109. [Google Scholar] [CrossRef]
- Jouris, K.B.; McDaniel, J.L.; Weiss, E.P. The Effect of Omega-3 Fatty Acid Supplementation on the Inflammatory Response to eccentric strength exercise. J. Sports Sci. Med. 2011, 10, 432–438. [Google Scholar]
- Su, Q.S.; Tian, Y.; Zhang, J.G.; Zhang, H. Effects of allicin supplementation on plasma markers of exercise-induced muscle damage, IL-6 and antioxidant capacity. Eur. J. Appl. Physiol. 2008, 103, 275–283. [Google Scholar] [CrossRef]

| Obtain adequate total sleep duration | Strategy 1: Track sleep for 2 weeks using a self-report sleep diary. Gradually increase sleep duration by 15 min every few nights, until athlete feels well rested and alert during the day. Consider increasing nighttime sleep by 30–60 min/night; this is particularly important if average sleep duration is <7 h/night | Strategy 2: Consider implementing regular naps, beginning on weekends or off-days if needed. Allow adequate time to return to full alertness after daytime naps |
| Maintain healthy sleep habits | Strategy 1: Develop a good sleep environment: the ideal room is cool, dark, and comfortable. Avoid having/using electronics or personal devices in bedroom | Strategy 2: Avoid alerting factors in the evening. Reduce ambient light exposure in late evening hours if possible, limit electronic device use at least 1 h prior to bedtime, allow for a 30–60 min relaxing wind-down period before bed. Ideally, consume no caffeine after lunch; limit alcohol use in late evening |
| Minimizing impact of travel | Strategy 1: Factor-in time needed to adjust to new time zone; as a rule of thumb, the body can adjust to 1 hr of time zone difference each day. Consider starting to shift body clock prior to departure or during flight; personalized travel planners (available online) may be helpful | Strategy 2: Reduce impact of non-jet lag travel effects: dehydration, acoustic stress, low physical activity, changes in food/drinking patterns |
| Intervention | Practical Implications | |
|---|---|---|
| Compression |
|
|
| ||
| ||
| Thermal therapy |
|
|
| ||
| ||
| Active regeneration |
|
|
| ||
| ||
| Oral medications and nutrition |
|
|
| Improving sleep |
|
|
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hotfiel, T.; Mayer, I.; Huettel, M.; Hoppe, M.W.; Engelhardt, M.; Lutter, C.; Pöttgen, K.; Heiss, R.; Kastner, T.; Grim, C. Accelerating Recovery from Exercise-Induced Muscle Injuries in Triathletes: Considerations for Olympic Distance Races. Sports 2019, 7, 143. https://doi.org/10.3390/sports7060143
Hotfiel T, Mayer I, Huettel M, Hoppe MW, Engelhardt M, Lutter C, Pöttgen K, Heiss R, Kastner T, Grim C. Accelerating Recovery from Exercise-Induced Muscle Injuries in Triathletes: Considerations for Olympic Distance Races. Sports. 2019; 7(6):143. https://doi.org/10.3390/sports7060143
Chicago/Turabian StyleHotfiel, Thilo, Isabel Mayer, Moritz Huettel, Matthias Wilhelm Hoppe, Martin Engelhardt, Christoph Lutter, Klaus Pöttgen, Rafael Heiss, Tom Kastner, and Casper Grim. 2019. "Accelerating Recovery from Exercise-Induced Muscle Injuries in Triathletes: Considerations for Olympic Distance Races" Sports 7, no. 6: 143. https://doi.org/10.3390/sports7060143
APA StyleHotfiel, T., Mayer, I., Huettel, M., Hoppe, M. W., Engelhardt, M., Lutter, C., Pöttgen, K., Heiss, R., Kastner, T., & Grim, C. (2019). Accelerating Recovery from Exercise-Induced Muscle Injuries in Triathletes: Considerations for Olympic Distance Races. Sports, 7(6), 143. https://doi.org/10.3390/sports7060143





