Tendon and Ligament Injuries in Elite Rugby: The Potential Genetic Influence
Abstract
1. Introduction
2. Tendon and Ligament Injury Incidence Rates and Severity in Rugby
2.1. Tendon and Ligament Injury Incidence Rates in Rugby
2.2. Tendon and Ligament Injury Severity and Burden in Rugby
3. Risk Factors for Injury in Rugby
4. Tendon and Ligament Pathologies
4.1. Tendinopathy
4.2. Tendon Rupture
4.3. Molecular Changes in Tendinopathy and Tendon Rupture
4.4. ACL Tear and Rupture
4.5. Molecular Characteristics of ACL Tear and Rupture
5. Genetics of Tendon and Ligaments
6. Identifying Candidate Genes
6.1. COL1A1 as a Candidate Gene
6.2. COL3A1 as a Candidate Gene
6.3. COL5A1 as a Candidate Gene
6.4. MIR608 as a Candidate Gene
6.5. MMP3 as a Candidate Gene
6.6. TIMP2 as a Candidate Gene
6.7. VEGFA as a Candidate Gene
6.8. Additional Candidate Genes of Interest
7. Future Directions/Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gabbett, T.J. Incidence of injury in junior and senior rugby league players. Sports Med. 2004, 34, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Hoskins, W.; Pollard, H.; Hough, K.; Tully, C. Injury in rugby league. J. Sci. Med. Sport. 2006, 9, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Brooks, J.H.M.; Kemp, S.P. Recent trends in rugby union. Clin. Sports Med. 2008, 27, 51–73. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, N. Injuries in professional rugby league: A three year prospective study of the South Sydney Professional Rugby League Football Club. Am. J. Sports Med. 1993, 21, 696–700. [Google Scholar] [CrossRef] [PubMed]
- Gabbett, T.J.; Jenkins, D.G.; Abernethy, B. Physical collisions and injury in professional rugby league match-play. J. Sci. Med. Sport 2011, 14, 210–215. [Google Scholar] [CrossRef]
- Brooks, J.H.M.; Fuller, C.W.; Kemp, S.P.T.; Reddin, D.B. Epidemiology of injuries in English professional rugby union: Part 1 match injuries. Br. J. Sports Med. 2005, 39, 757–766. [Google Scholar] [CrossRef]
- Fuller, C.W.; Sheerin, K.; Targett, S. Rugby World Cup 2011: International Rugby Board Injury Surveillance Study. Br. J. Sports Med. 2013, 47, 1184–1191. [Google Scholar] [CrossRef]
- Fuller, C.W.; Taylor, A.; Kemp, S.P.T.; Raftery, M. Rugby World Cup 2015: World Rugby injury surveillance study. Br. J. Sports Med. 2017, 51, 51–57. [Google Scholar] [CrossRef]
- Fuller, C.W.; Brooks, J.H.M.; Cancea, R.J.; Hall, J.; Kemp, S.P.T. Contact events in rugby union and their propensity to cause injury. Br. J. Sports Med. 2007, 41, 862–867. [Google Scholar] [CrossRef]
- Gabbett, T.J.; Hodgson, P. Incidence of injury in semi-professional rugby league players. Br. J. Sports Med. 2003, 37, 36–44. [Google Scholar] [CrossRef]
- Williams, S.; Trewartha, G.; Kemp, S.; Stokes, K. A Meta-Analysis of Injuries in Senior Men’s Professional Rugby Union. Sports Med. 2013, 43, 1043–1055. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.; Trewartha, G.; Kemp, S.P.T.; Brooks, J.H.M.; Fuller, C.W.; Taylor, A.E.; Cross, M.J.; Stokes, K.A. Time loss injuries compromise team success in Elite Rugby Union: A 7-year prospective study. Br. J. Sports Med. 2016, 50, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Best, J.P.; McIntosh, A.S.; Savage, T. Rugby World Cup 2003 injury surveillance project. Br. J. Sports Med. 2005, 39, 812–817. [Google Scholar] [CrossRef] [PubMed]
- Fuller, C.W.; Laborde, F.; Leather, R.J.; Molloy, M.G. International Rugby Board Rugby World Cup 2007 injury surveillance study. Br. J. Sports Med. 2008, 42, 452–459. [Google Scholar] [CrossRef] [PubMed]
- Kemp, S.; Brooks, J.; Cross, M.; Morrow, P.; Williams, S.; Anstiss, T.; Smith, A.; Taylor, A.; Trewartha, G.; Widdowson, J.; et al. England Professional Rugby Injury Surveillance Project 2013–2014 Season Report; Rugby Football Union: Twickenham, UK, 2015. [Google Scholar]
- Kemp, S.; Brooks, J.; Cross, M.; Morrow, P.; Williams, S.; Anstiss, T.; Smith, A.; Taylor, A.; Palmer, C.; Bryan, R.; et al. England Professional Rugby Injury Surveillance Project 2014–2015 Season Report; Rugby Football Union: Twickenham, UK, 2016. [Google Scholar]
- Haseler, C.M.; Carmont, M.R.; England, M. The epidemiology of injuries in English youth community rugby union. Br. J. Sports Med. 2010, 44, 1093–1099. [Google Scholar] [CrossRef]
- Roberts, S.P.; Trewartha, G.; England, M.; Shaddick, G.; A Stokes, K. Epidemiology of time-loss injuries in English community-level rugby union. BMJ Open 2013, 3, e003998. [Google Scholar] [CrossRef] [PubMed]
- Fuller, C.W.; Molloy, M.G.; Bagate, C.; Bahr, R.; Brooks, J.H.; Donson, H.; Kemp, S.P.; McCrory, P.; McIntosh, A.S.; Meeuwisse, W.H.; et al. Consensus Statement on Injury Definitions and Data Collection Procedures for Studies of Injuries in Rugby Union. Clin. J. Sport Med. 2007, 17, 177–181. [Google Scholar] [CrossRef]
- Fitzpatrick, A.C.; Naylor, A.S.; Myler, P.; Robertson, C. A three-year epidemiological prospective cohort study of rugby league match injuries from the European Super League. J. Sci. Med. Sport 2018, 21, 160–165. [Google Scholar] [CrossRef]
- Van Mechelen, W.; Hlobil, H.; Kemper, H.C. Incidence, severity, aetiology and prevention of sports injuries. A review of concepts. Sports Med. 1992, 14, 82. [Google Scholar] [CrossRef]
- Fuller, C.W.; Drawer, D. The Application of Risk Management in Sport. Sports Med. 2004, 34, 49–56. [Google Scholar] [CrossRef]
- Gissane, C.; Jennings, D.C.; Standing, P. Incidence of Injury in Rugby League Football. Physiotherapy 1993, 79, 305–310. [Google Scholar] [CrossRef]
- Seward, H.; Orchard, J.; Hazard, H.; Collinson, D. Football injuries in Australia at the élite level. Med. J. Aust. 1993, 159, 298–301. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, S.; Gissane, C.; Jennings, D. Injury in rugby league: A four year prospective survey. Br. J. Sports Med. 1996, 30, 331–334. [Google Scholar] [CrossRef] [PubMed]
- Gissane, C.; Jennings, D.; Kerr, K.; White, J. Injury Rates in Rugby League Football: Impact of Change in Playing Season. Am. J. Sports Med. 2003, 31, 954–958. [Google Scholar] [CrossRef] [PubMed]
- Gissane, C.; Marshall, S.W.; King, D.; Clark, T. The Incidence of Match and Training Injuries in Rugby League: A Pooled Data Analysis of Published Studies. Int. J. Sports Sci. Coach. 2014, 9, 417–432. [Google Scholar]
- Cross, M.; Williams, S.; Kemp, S.P.; Fuller, C.; Taylor, A.; Brooks, J.; Trewartha, G.; Stokes, K. Does the Reliability of Reporting in Injury Surveillance Studies Depend on Injury Definition? Orthop. J. Sports Med. 2018, 6, 2325967118760536. [Google Scholar] [CrossRef] [PubMed]
- Orchard, J.; Hoskins, W. For Debate: Consensus Injury Definitions in Team Sports Should Focus on Missed Playing Time. Clin. J. Sport Med. 2007, 17, 192–196. [Google Scholar] [CrossRef]
- Hodgson, L.; Gissane, C.; Gabbett, T.J.; A King, D. For Debate: Consensus Injury Definitions in Team Sports Should Focus on Encompassing all Injuries. Clin. J. Sport Med. 2007, 17, 188–191. [Google Scholar] [CrossRef]
- King, D.; Gabbett, T.; Gissane, C.; Hodgson, L. Epidemiological studies of injuries in rugby league: Suggestions for definitions, data collection and reporting methods. J. Sci. Med. Sport 2009, 12, 12–19. [Google Scholar] [CrossRef]
- Moore, I.S.; Ranson, C.; Mathema, P. Injury Risk in International Rugby Union: Three-Year Injury Surveillance of the Welsh National Team. Orthop. J. Sports Med. 2015, 3, 2325967115596194. [Google Scholar] [CrossRef]
- Brooks, J.H.M.; Fuller, C.; Kemp, S.; Reddin, D. A prospective study of injuries and training amongst the England 2003 Rugby World Cup squad. Br. J. Sports Med. 2005, 39, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Brooks, J.H.M.; Fuller, C.W.; Kemp, S.P.T.; Reddin, D.B. Epidemiology of injuries in English professional rugby union: Part 2 training Injuries. Br. J. Sports Med. 2005, 39, 767–775. [Google Scholar] [CrossRef] [PubMed]
- Fuller, C.W.; Raftery, M.; Readhead, C.; Targett, S.G.R.; Molloy, M.G. Impact of the International Rugby Board’s experimental law variations on the incidence and nature of match injuries in southern hemisphere professional rugby union. S. Afr. Med J. 2009, 99, 232. [Google Scholar] [PubMed]
- Fuller, C.W.; Clarke, L.; Molloy, M.G. Risk of injury associated with rugby union played on artificial turf. J. Sports Sci. 2010, 28, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Kemp, S.; Brooks, J.; West, S.; Cross, M.; Morrow, P.; Williams, S.; Anstiss, T.; Smith, A.; Taylor, A.; Palmer, C.; et al. English Professional Rugby Injury Surveillance Project 2015–2016 Season Report; Rugby Football Union: Twickenham, UK, 2017. [Google Scholar]
- Kemp, S.; West, S.; Brooks, J.; Cross, M.; Williams, S.; Anstiss, T.; Smith, A.; Bryan, R.; Hibbins-Butler, R.; O’Leary, B.; et al. English Professional Rugby Injury Surveillance Project 2016–2017 Season Report; Rugby Football Union: Twickenham, UK, 2018. [Google Scholar]
- Kemp, S.; West, S.; Brooks, J.; Cross, M.; Williams, S.; Anstiss, T.; Smith, A.; Bryan, R.; Henderson, L.; Locke, D.; et al. English Professional Rugby Injury Surveillance Project 2017–2018 Season Report; Rugby Football Union: Twickenham, UK, 2019. [Google Scholar]
- Gibbs, N. Common rugby league injuries. Recommendations for treatment and preventative measures. Sports Med. 1994, 18, 438–450. [Google Scholar] [CrossRef] [PubMed]
- Orchard, J. Missed time through injury and injury management at an NRL club. Sport Health 2004, 22, 11–19. [Google Scholar]
- Bathgate, A.; Best, J.P.; Craig, G.; Jamieson, M. A prospective study of injuries to elite Australian rugby union players. Br. J. Sports Med. 2002, 36, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Quarrie, K.L.; Hopkins, W.G. Tackle Injuries in Professional Rugby Union. Am. J. Sports Med. 2008, 36, 1705–1716. [Google Scholar] [CrossRef]
- Cross, M.J.; Tucker, R.; Raftery, M.; Hester, B.; Williams, S.; A Stokes, K.; Ranson, C.; Mathema, P.; Kemp, S. Tackling concussion in professional rugby union: A case–control study of tackle-based risk factors and recommendations for primary prevention. Br. J. Sports Med. 2017. [Google Scholar] [CrossRef]
- Gabbett, T.J.; Jenkins, D.G. Relationship between training load and injury in professional rugby league players. J. Sci. Med. Sport 2011, 14, 204–209. [Google Scholar] [CrossRef]
- Cross, M.J.; Williams, S.; Trewartha, G.; Kemp, S.P.; Stokes, K.A. The Influence of In-Season Training Loads on Injury Risk in Professional Rugby Union. Int. J. Sports Physiol. Perform. 2016, 11, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Windt, J.; Gabbett, T.J.; Ferris, D.; Khan, K.M. Training load–injury paradox: Is greater preseason participation associated with lower in-season injury risk in elite rugby league players? Br. J. Sports Med. 2016, 51, 645–650. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.; Hume, P.A.; Kara, S. A Review of Football Injuries on Third and Fourth Generation Artificial Turfs Compared with Natural Turf. Sports Med. 2011, 41, 903–923. [Google Scholar] [CrossRef] [PubMed]
- Fuller, C.W.; Ashton, T.; Brooks, J.H.M.; Cancea, R.J.; Hall, J.; Kemp, S.P.T. Injury risks associated with tackling in rugby union. Br. J. Sports Med. 2010, 44, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Gabbett, T.J.; Ullah, S.; Finch, C.F. Identifying risk factors for contact injury in professional rugby league players – Application of a frailty model for recurrent injury. J. Sci. Med. Sport 2012, 15, 496–504. [Google Scholar] [CrossRef]
- Quarrie, K.L.; Alsop, J.; Waller, A.; Bird, Y.; Marshall, S.; Chalmers, D. The New Zealand rugby injury and performance project. VI. A prospective cohort study of risk factors for injury in rugby union football. Br. J. Sports Med. 2001, 35, 157–166. [Google Scholar] [CrossRef]
- Cross, M.; Kemp, S.; Smith, A.; Trewartha, G.; Stokes, K. Professional Rugby Union players have a 60% greater risk of time loss injury after concussion: A 2-season prospective study of clinical outcomes. Br. J. Sports Med. 2016, 50, 926–931. [Google Scholar] [CrossRef]
- Rafferty, J.; Ranson, C.; Oatley, G.; Mostafa, M.; Mathema, P.; Crick, T.; Moore, I.S. On average, a professional rugby union player is more likely than not to sustain a concussion after 25 matches. Br. J. Sports Med. 2018. [Google Scholar] [CrossRef]
- Brooks, J.H. The Epidemiology of Injuries in Professional Rugby; University of Leicester: Leicester, UK, 2004. [Google Scholar]
- Riley, G. The pathogenesis of tendinopathy. A molecular perspective. Rheumatology 2004, 43, 131–142. [Google Scholar] [CrossRef]
- Griffin, L.Y.; Albohm, M.J.; Arendt, E.A.; Bahr, R.; Beynnon, B.D.; DeMaio, M.; Dick, R.W.; Engebretsen, L.; Garrett, W.E.; Hannafin, J.A.; et al. Understanding and Preventing Noncontact Anterior Cruciate Ligament Injuries: A Review of the Hunt Valley II Meeting, January 2005. Am. J. Sports Med. 2006, 34, 1512–1532. [Google Scholar] [CrossRef]
- Meeuwise, W.H. Assessing causation in sport injury: A multifactorial model. Clin. J. Sport Med. 1994, 4, 166–170. [Google Scholar] [CrossRef]
- September, A.V.; Mokone, G.G.; Schwellnus, M.P.; Collins, M. Genetic risk factors for Achilles tendon injuries. Int. Sports Med. J. 2006, 7, 201–215. [Google Scholar]
- Khan, K.M.; Scott, A. Mechanotherapy: How physical therapists’ prescription of exercise promotes tissue repair. Br. J. Sports Med. 2009, 43, 247–252. [Google Scholar] [CrossRef]
- Collins, M.; Raleigh, S. Genetic Risk Factors for Musculoskeletal Soft Tissue Injuries. Elite Young Athlete 2009, 54, 136–149. [Google Scholar]
- September, A.V.; Posthumus, M. Application of Genomics in the Prevention, Treatment and Management of Achilles Tendinopathy and Anterior Cruciate Ligament Ruptures. Recent Patents DNA Gene Seq. 2012, 6, 216–223. [Google Scholar] [CrossRef]
- Magnusson, S.P.; Langberg, H.; Kjaer, M. The pathogenesis of tendinopathy: Balancing the response to loading. Nat. Rev. Rheumatol. 2010, 6, 262–268. [Google Scholar] [CrossRef]
- Almekinders, L.C.; Temple, J.D. Etiology, diagnosis, and treatment of tendonitis: An analysis of the literature. Med. Sci. Sports Exerc. 1998, 30, 1183–1190. [Google Scholar] [CrossRef]
- Bisset, L.; Beller, E.; Jull, G.; Brooks, P.; Darnell, R.; Vicenzino, B. Mobilisation with movement and exercise, corticosteroid injection, or wait and see for tennis elbow: Randomised trial. BMJ 2006, 333, 939. [Google Scholar] [CrossRef]
- Hashimoto, T.; Nobuhara, K.; Hamada, T. Pathologic Evidence of Degeneration as a Primary Cause of Rotator Cuff Tear. Clin. Orthop. Relat. Res. 2003, 415, 111–120. [Google Scholar] [CrossRef]
- Maffulli, N.; Wong, J.; Almekinders, L.C. Types and epidemiology of tendinopathy. Clin. Sports Med. 2003, 22, 675–692. [Google Scholar] [CrossRef]
- Abate, M.; Silbernagel, K.G.; Siljeholm, C.; Di Iorio, A.; De Amicis, D.; Salini, V.; Werner, S.; Paganelli, R. Pathogenesis of tendinopathies: Inflammation or degeneration? Arthritis Res. Ther. 2009, 11, 235. [Google Scholar] [CrossRef] [PubMed]
- Morrey, M.E.; Dean, B.J.F.; Carr, A.J.; Morrey, B.F. Tendinopathy: Same Disease Different Results—Why? Oper. Tech. Orthop. 2013, 23, 39–49. [Google Scholar] [CrossRef]
- Pajala, A.; Melkko, J.; Leppilahti, J.; Ohtonen, P.; Soini, Y.; Risteli, J. Tenascin-C and type I and III collagen expression in total Achilles tendon rupture. An immunohistochemical study. Histol. Histopathol. 2009, 24, 1207. [Google Scholar] [PubMed]
- Riley, G.P.; Harrall, R.L.; Constant, C.R.; Chard, M.D.; E Cawston, T.; Hazleman, B.L.; Riley, G. Tendon degeneration and chronic shoulder pain: Changes in the collagen composition of the human rotator cuff tendons in rotator cuff tendinitis. Ann. Rheum. Dis. 1994, 53, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves-Neto, J.; Witzel, S.; Teodoro, W.; Carvalho-Junior, A.; Fernandes, T.; Yoshinari, H. Changes in collagen matrix composition in human posterior tibial tendon dysfunction. Jt. Bone Spine 2002, 69, 189–194. [Google Scholar] [CrossRef]
- Eriksen, H.A.; Pajala, A.; Leppilahti, J.; Risteli, J. Increased content of type III collagen at the rupture site of human Achilles tendon. J. Orthop. Res. 2002, 20, 1352–1357. [Google Scholar] [CrossRef]
- Lapiere, C.M.; Nusgens, B.; Piérard, G.E. Interaction between Collagen Type I and Type III in Conditioning Bundles Organization. Connect. Tissue Res. 1977, 5, 21–29. [Google Scholar] [CrossRef]
- Magnusson, S.; Qvortrup, K.; Larsen, J.; Rosager, S.; Hanson, P.; Aagaard, P.; Krogsgaard, M.; Kjaer, M. Collagen fibril size and crimp morphology in ruptured and intact Achilles tendons. Matrix Boil. 2002, 21, 369–377. [Google Scholar] [CrossRef]
- Nourissat, G. Use of autologous growth factors in aging tendon and chronic tendinopathy. Front. Biosci. 2013, 5, 911–921. [Google Scholar] [CrossRef]
- Magnan, B.; Bondi, M.; Pierantoni, S.; Samaila, E. The pathogenesis of Achilles tendinopathy: A systematic review. Foot Ankle Surg. 2014, 20, 154–159. [Google Scholar] [CrossRef]
- Ribbans, W.J.; Collins, M. Pathology of the tendo Achillis Do our genes contribute? Bone Jt. J. 2013, 95B, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Kvist, M. Achilles Tendon Injuries in Athletes. Sports Med. 1994, 18, 173–201. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.C. Mechanobiology of tendon. J. Biomech. 2006, 39, 1563–1582. [Google Scholar] [CrossRef] [PubMed]
- Selvanetti, A.; Cipolla, M.; Puddu, G. Overuse tendon injuries: Basic science and classification. Oper. Tech. Sports Med. 1997, 5, 110–117. [Google Scholar] [CrossRef]
- Nourissat, G.; Berenbaum, F.; Duprez, D. Tendon injury: From biology to tendon repair. Nat. Rev. Rheumatol. 2015, 11, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Maffulli, N. Biology of tendon injury: Healing, modeling and remodeling. J. Musculoskelet. Neuronal Interact. 2006, 6, 181. [Google Scholar] [PubMed]
- Thornton, G.M.; A Hart, D. The interface of mechanical loading and biological variables as they pertain to the development of tendinosis. J. Musculoskelet. Neuronal Interact. 2011, 11, 94. [Google Scholar] [PubMed]
- Kaux, J.F.; Forthomme, B.; Goff, C.L.; Crielaard, J.M.; Croisier, J.L. Current opinions on tendinopathy. J. Sports Sci Med. 2011, 10, 238–253. [Google Scholar]
- Voleti, P.B.; Buckley, M.R.; Soslowsky, L.J. Tendon Healing: Repair and Regeneration. Annu. Rev. Biomed. Eng. 2012, 14, 47–71. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.C.; Guo, Q.; Li, B. Tendon biomechanics and mechanobiology—A minireview of basic concepts and recent advancements. J. Hand Ther. 2012, 25, 133. [Google Scholar] [CrossRef]
- Docheva, D.; Müller, S.A.; Majewski, M.; Evans, C.H. Biologics for tendon repair. Adv. Drug Deliv. Rev. 2015, 84, 222–239. [Google Scholar] [CrossRef] [PubMed]
- Leadbetter, W.B. Cell-matrix response in tendon injury. Clin. Sports Med. 1992, 11, 533. [Google Scholar] [PubMed]
- David, L.; Grood, E.S.; Noyes, F.R.; Zernicke, R.E. Biomechanics of ligaments and tendons. Exerc. Sport Sci. Rev. 1978, 6, 125. [Google Scholar]
- Longo, U.G.; Ronga, M.; Maffulli, N. Acute Ruptures of the Achilles Tendon. Sports Med. Arthrosc. Rev. 2009, 17, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Axibal, D.P.; Anderson, J.G. Multiple Tendon Ruptures of Unknown Etiology. Foot Ankle Spec. 2013, 6, 380–383. [Google Scholar] [CrossRef] [PubMed]
- Hess, G.W. Achilles tendon rupture: A review of etiology, population, anatomy, risk factors, and injury prevention. Foot Ankle Spec. 2010, 3, 29. [Google Scholar] [CrossRef] [PubMed]
- Corps, A.N.; Robinson, A.H.N.; Movin, T.; Costa, M.L.; Hazleman, B.L.; Riley, G.P. Increased expression of aggrecan and biglycan mRNA in Achilles tendinopathy. Rheumatology 2006, 45, 291–294. [Google Scholar] [CrossRef]
- Karousou, E.; Ronga, M.; Vigetti, D.; Passi, A.; Maffulli, N. Collagens, Proteoglycans, MMP-2, MMP-9 and TIMPs in Human Achilles Tendon Rupture. Clin. Orthop. Relat. Res. 2008, 466, 1577–1582. [Google Scholar] [CrossRef]
- Pufe, T.; Petersen, W.; Tillmann, B.; Mentlein, R. The angiogenic peptide vascular endothelial growth factor is expressed in foetal and ruptured tendons. Virchows Archiv. 2001, 439, 579–585. [Google Scholar] [CrossRef]
- Jones, G.C.; Corps, A.N.; Pennington, C.J.; Clark, I.M.; Edwards, D.R.; Bradley, M.M.; Hazleman, B.L.; Riley, G.P. Expression profiling of metalloproteinases and tissue inhibitors of metalloproteinases in normal and degenerate human Achilles tendon. Arthritis Rheum. 2006, 54, 832–842. [Google Scholar] [CrossRef]
- Xu, Y.l.; Murrell, G.A.C. The Basic Science of Tendinopathy. Clin. Orthop. Relat. Res. 2008, 466, 1528–1538. [Google Scholar] [CrossRef]
- Hakim, A.J.; Cherkas, L.F.; Spector, T.D.; MacGregor, A.J. Genetic associations between frozen shoulder and tennis elbow: A female twin study. Rheumatology 2003, 42, 739. [Google Scholar] [PubMed]
- Corry, I. Injuries of the sporting knee. Br. J. Sports Med. 2000, 34, 395. [Google Scholar] [CrossRef] [PubMed]
- Majewski, M.; Susanne, H.; Klaus, S. Epidemiology of athletic knee injuries: A 10-year study. Knee 2006, 13, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Von Porat, A.; Roos, E.M.; Roos, H. High prevalence of osteoarthritis 14 years after an anterior cruciate ligament tear in male soccer players: A study of radiographic and patient relevant outcomes. Ann. Rheum. Dise. 2004, 63, 269–273. [Google Scholar] [CrossRef]
- Nebelung, W.; Wuschech, H. Thirty-five Years of Follow-up of Anterior Cruciate Ligament—Deficient Knees in High-Level Athletes. Arthrosc. J. Arthrosc. Relat. Surg. 2005, 21, 696–702. [Google Scholar] [CrossRef] [PubMed]
- Quatman, C.E.; Kiapour, A.; Myer, G.D.; Ford, K.R.; Demetropoulos, C.K.; Goel, V.K.; Hewett, T.E. Cartilage Pressure Distributions Provide a Footprint to Define Female Anterior Cruciate Ligament Injury Mechanisms. Am. J. Sports Med. 2011, 39, 1706–1713. [Google Scholar] [CrossRef]
- Chu, C.R.; Beynnon, B.D.; Buckwalter, J.A.; Garrett, J.W.E.; Katz, J.N.; Rodeo, S.A.; Spindler, K.P.; Stanton, R.A. Closing the gap between bench and bedside research for early arthritis therapies (EARTH): Report from the AOSSM/NIH U-13 Post-Joint Injury Osteoarthritis Conference II. Am. J. Sports Med. 2011, 39, 1569–1578. [Google Scholar] [CrossRef]
- Levine, J.W.; Kiapour, A.M.; Quatman, C.E.; Wordeman, S.C.; Goel, V.K.; Hewett, T.E.; Demetropoulos, C.K. Clinically Relevant Injury Patterns After an Anterior Cruciate Ligament Injury Provide Insight Into Injury Mechanisms. Am. J. sports Med. 2013, 41, 385–395. [Google Scholar] [CrossRef]
- Dallalana, R.J.; Brooks, J.H.M.; Kemp, S.P.T.; Williams, A.M. The Epidemiology of Knee Injuries in English Professional Rugby Union. Am. J. Sports Med. 2007, 35, 818–830. [Google Scholar] [CrossRef]
- Montgomery, C.; Blackburn, J.; Withers, D.; Tierney, G.; Moran, C.; Simms, C. Mechanisms of ACL injury in professional rugby union: A systematic video analysis of 36 cases. Br. J. Sports Med. 2016, 52, 994–1001. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.C.; Vacek, P.; Johnson, R.J.; Slauterbeck, J.R.; Hashemi, J.; Shultz, S.; Beynnon, B.D. Risk Factors for Anterior Cruciate Ligament Injury: A Review of the Literature—Part 1: Neuromuscular and Anatomic Risk. Sports Health A Multidiscip. Approach. 2012, 4, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.C.; Vacek, P.; Johnson, R.J.; Slauterbeck, J.R.; Hashemi, J.; Shultz, S.; Beynnon, B.D. Risk Factors for Anterior Cruciate Ligament Injury: A Review of the Literature—Part 2: Hormonal, Genetic, Cognitive Function, Previous Injury, and Extrinsic Risk Factors. Sports Health A Multidiscip. Approach 2012, 4, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Alentorn-Geli, E.; Myer, G.D.; Silvers, H.J.; Samitier, G.; Romero, D.; Lázaro-Haro, C.; Cugat, R. Prevention of non-contact anterior cruciate ligament injuries in soccer players. Part 1: Mechanisms of injury and underlying risk factors. Knee Surgery Sports Traumatol. Arthrosc. 2009, 17, 705–729. [Google Scholar] [CrossRef] [PubMed]
- Abramowitch, S.D.; Saw, M.S.S.C.; Fenwick, J.A.; Woo, S.L.-Y.; Debski, R.E.; Zeminski, J. Injury and Repair of Ligaments and Tendons. Annu. Rev. Biomed. Eng. 2000, 2, 83–118. [Google Scholar]
- Provenzano, P.P.; Heisey, D.; Hayashi, K.; Lakes, R.; Vanderby, R. Subfailure damage in ligament: A structural and cellular evaluation. J. Appl. Physiol. 2002, 92, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Harner, C.D.; Paulos, L.E.; Greenwald, A.E.; Rosenberg, T.D.; Cooley, V.C. Detailed Analysis of Patients with Bilateral Anterior Cruciate Ligament Injuries. Am. J. Sports Med. 1994, 22, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Flynn, R.K.; Pedersen, C.L.; Birmingham, T.B.; Kirkley, A.; Jackowski, D.; Fowler, P.J. The familial predisposition toward tearing the anterior cruciate ligament: A case control study. Am. J. Sports Med. 2005, 33, 23. [Google Scholar] [CrossRef]
- Khoschnau, S.; Melhus, H.; Jacobson, A.; Rahme, H.; Bengtsson, H.; Ribom, E.; Grundberg, E.; Mallmin, H.; Michaëlsson, K. Type I collagen alpha1 Sp1 polymorphism and the risk of cruciate ligament ruptures or shoulder dislocations. Am. J. Sports Med. 2008, 36, 2432. [Google Scholar] [CrossRef]
- Posthumus, M.; September, A.V.; Keegan, M.; O’Cuinneagain, D.; Van Der Merwe, W.; Schwellnus, M.; Collins, M. Genetic risk factors for anterior cruciate ligament ruptures: COL1A1 gene variant. Br. J. Sports Med. 2009, 43, 352–356. [Google Scholar] [CrossRef]
- O’Cuinneagain, D.; Posthumus, M.; September, A.V.; Collins, M.; Van Der Merwe, W.; Schwellnus, M.P. The COL5A1 Gene Is Associated With Increased Risk of Anterior Cruciate Ligament Ruptures in Female Participants. Am. J. Sports Med. 2009, 37, 2234–2240. [Google Scholar]
- Posthumus, M.; September, A.V.; O’Cuinneagain, D.; van der Merwe, W.; Schwellnus, M.P.; Collins, M. The association between the COL12A1 gene and anterior cruciate ligament ruptures. Br. J. Sports Med. 2010, 44, 1160–1165. [Google Scholar] [CrossRef] [PubMed]
- Malila, S.; Yuktanandana, P.; Saowaprut, S.; Jiamjarasrangsi, W.; Honsawek, S. Association between matrix metalloproteinase-3 polymorphism and anterior cruciate ligament ruptures. Genet. Mol. Res. 2011, 10, 4158–4165. [Google Scholar] [CrossRef] [PubMed]
- Posthumus, M.; Collins, M.; van der Merwe, L.; O’Cuinneagain, D.; van der Merwe, W.; Ribbans, W.J.; Schwellnus, M.P.; Raleigh, S.M. Matrix metalloproteinase genes on chromosome 11q22 and the risk of anterior cruciate ligament (ACL) rupture. Scand. J. Med. Sci. Sports 2012, 22, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Stępien-Słodkowska, M.; Ficek, K.; Eider, J.; Leońska-Duniec, A.; Maciejewska-Karłowska, A.; Sawczuk, M.; Zarębska, A.; Jastrzębski, Z.; Grenda, A.; Kotarska, K.; et al. The +1245g/t polymorphisms in the collagen type I alpha 1 (col1a1) gene in polish skiers with anterior cruciate ligament injury. Biol. Sport 2013, 30, 57. [Google Scholar] [CrossRef] [PubMed]
- Ficek, K.; Cięszczyk, P.; Kaczmarczyk, M.; Maciejewska-Karłowska, A.; Sawczuk, M.; Cholewiński, J.; Leońska-Duniec, A.; Stepien-Slodkowska, M.; Zarebska, A.; Stepto, N.K.; et al. Gene variants within the COL1A1 gene are associated with reduced anterior cruciate ligament injury in professional soccer players. J. Sci. Med. Sport 2013, 16, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Ficek, K.; Stepien-Slodkowska, M.; Kaczmarczyk, M.; Maciejewska-Karlowska, A.; Sawczuk, M.; Cholewinski, J.; Leonska-Duniec, A.; Zarebska, A.; Cieszczyk, P.; Zmijewski, P.; et al. Does the A9285G Polymorphism in Collagen Type XII α1 Gene Associate with the Risk of Anterior Cruciate Ligament Ruptures? Balk. J. Med Genet. 2014, 17, 41–46. [Google Scholar]
- Mannion, S.; Mtintsilana, A.; Posthumus, M.; Van Der Merwe, W.; Hobbs, H.; Collins, M.; September, A.V. Genes encoding proteoglycans are associated with the risk of anterior cruciate ligament ruptures. Br. J. Sports Med. 2014, 48, 1640–1646. [Google Scholar] [CrossRef]
- Rahim, M.; Gibbon, A.; Hobbs, H.; van der Merwe, W.; Posthumus, M.; Collins, M.; September, A.V. The association of genes involved in the angiogenesis-associated signaling pathway with risk of anterior cruciate ligament rupture: Angiogenesis and acl rupture risk. J. Orthop. Res. 2014, 32, 1612–1618. [Google Scholar] [CrossRef]
- Johnson, J.S.; Morscher, M.A.; Jones, K.C.; Moen, S.M.; Klonk, C.J.; Jacquet, R.; Landis, W.J. Gene expression differences between ruptured anterior cruciate ligaments in young male and female subjects. J. Bone Jt. Surg. 2015, 97, 71. [Google Scholar] [CrossRef]
- O’Connell, K.; Knight, H.; Ficek, K.; Leonska-Duniec, A.; Maciejewska-Karlowska, A.; Sawczuk, M.; Stepien-Slodkowska, M.; O’Cuinneagain, D.; van der Merwe, W.; Posthumus, M.; et al. Interactions between collagen gene variants and risk of anterior cruciate ligament rupture. Eur. J. Sport Sci. 2015, 15, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Stępień-Słodkowska, M.; Ficek, K.; Kaczmarczyk, M.; Maciejewska-Karłowska, A.; Sawczuk, M.; Leońska-Duniec, A.; Stępiński, M.; Ziętek, P.; Król, P.; Chudecka, M.; et al. The Variants within the COL5A1 Gene are Associated with Reduced Risk of Anterior Cruciate Ligament Injury in Skiers. J. Hum. Kinet. 2015, 45, 103–111. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stępień-Słodkowska, M.; Ficek, K.; Maciejewska-Karłowska, A.; Sawczuk, M.; Ziętek, P.; Król, P.; Zmijewski, P.; Pokrywka, A.; Cięszczyk, P. Overrepresentation of the COL3A1 AA genotype in Polish skiers with anterior cruciate ligament injury. Boil. Sport 2015, 32, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Mokone, G.G.; Gajjar, M.; September, A.V.; Schwellnus, M.P.; Greenberg, J.; Noakes, T.D.; Collins, M. The Guanine-Thymine Dinucleotide Repeat Polymorphism within the Tenascin-C Gene is Associated with Achilles Tendon Injuries. Am. J. Sports Med. 2005, 33, 1016–1021. [Google Scholar] [CrossRef] [PubMed]
- Mokone, G.G.; Schwellnus, M.P.; Noakes, T.D.; Collins, M.; Schwellnus, M. The COL5A1 gene and Achilles tendon pathology. Scand. J. Med. Sci. Sports 2006, 16, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Raleigh, S.M.; Van Der Merwe, L.; Ribbans, W.J.; Smith, R.K.W.; Schwellnus, M.P.; Collins, M. Variants within the MMP3 gene are associated with Achilles tendinopathy: Possible interaction with the COL5A1 gene. Br. J. Sports Med. 2009, 43, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Posthumus, M.; Collins, M.; Cook, J.; Handley, C.J.; Ribbans, W.J.; Smith, R.K.W.; Schwellnus, M.P.; Raleigh, S.M. Components of the transforming growth factor-β family and the pathogenesis of human achilles tendon pathology-a genetic association study. Rheumatology 2010, 49, 2090–2097. [Google Scholar] [CrossRef] [PubMed]
- Nell, E.-M.; Van Der Merwe, L.; Cook, J.; Handley, C.J.; Collins, M.; September, A.V. The apoptosis pathway and the genetic predisposition to Achilles tendinopathy. J. Orthop. Res. 2012, 30, 1719–1724. [Google Scholar] [CrossRef] [PubMed]
- Abrahams, Y.; Laguette, M.-J.; Prince, S.; Collins, M. Polymorphisms within the COL5A1 3′-UTR That Alters mRNA Structure and the MIR608 Gene are Associated with Achilles Tendinopathy. Ann. Hum. Genet. 2013, 77, 204–214. [Google Scholar] [CrossRef]
- Saunders, C.J.; van der Merwe, L.; Posthumus, M.; Cook, J.; Handley, C.J.; Collins, M.; September, A.V. Investigation of variants within the COL27A1 and TNC genes and Achilles tendinopathy in two populations. J. Orthop. Res. 2013, 31, 632–637. [Google Scholar] [CrossRef] [PubMed]
- El Khoury, L.; Posthumus, M.; Collins, M.; Handley, C.J.; Cook, J.; Raleigh, S.M. Polymorphic variation within the ADAMTS2, ADAMTS14, ADAMTS5, ADAM12 and TIMP2 genes and the risk of Achilles tendon pathology: A genetic association study. J. Sci. Med. Sport 2013, 16, 493–498. [Google Scholar] [CrossRef] [PubMed]
- El Khoury, L.; Posthumus, M.; Collins, M.; Ribbans, W.; Raleigh, S. 82 TheCOL5A1Gene and Risk of Achilles Tendon Pathology in a British Cohort. Br. J. Sports Med. 2014, 48, A54. [Google Scholar] [CrossRef]
- El Khoury, L.; Posthumus, M.; Collins, M.; van der Merwe, W.; Handley, C.; Cook, J.; Raleigh, S.M. ELN and FBN2 Gene Variants as Risk Factors for Two Sports-related Musculoskeletal Injuries. Int. J. Sports Med. 2015, 36, 333–337. [Google Scholar] [CrossRef] [PubMed]
- El Khoury, L.; Ribbans, W.J.; Raleigh, S.M. MMP3 and TIMP2 gene variants as predisposing factors for Achilles tendon pathologies: Attempted replication study in a British case–control cohort. Meta Gene 2016, 9, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Tranchevent, L.-C.; Ardeshirdavani, A.; ElShal, S.; Alcaide, D.; Aerts, J.; Auboeuf, D.; Moreau, Y. Candidate gene prioritization with Endeavour. Nucleic Acids Res. 2016, 44, W117–W121. [Google Scholar] [CrossRef] [PubMed]
- Heffernan, S.M.; Stebbings, G.K.; Kilduff, L.P.; Erskine, R.M.; Day, S.H.; Morse, C.I.; McPhee, J.S.; Cook, C.J.; Vance, B.; Ribbans, W.J.; et al. Fat mass and obesity associated (FTO) gene influences skeletal muscle phenotypes in non-resistance trained males and elite rugby playing position. BMC Genet. 2017, 18, 4. [Google Scholar] [CrossRef] [PubMed]
- Tabor, H.K.; Risch, N.J.; Myers, R.M. Candidate-gene approaches for studying complex genetic traits: Practical considerations. Nat. Rev. Genet. 2002, 3, 391–397. [Google Scholar] [CrossRef]
- Drysdale, C.M.; McGraw, D.W.; Stack, C.B.; Stephens, J.C.; Judson, R.S.; Nandabalan, K.; Arnold, K.; Ruano, G.; Liggett, S.B. Complex promoter and coding region β2-adrenergic receptor haplotypes alter receptor expression and predict in vivo responsiveness. Proc. Natl. Acad. Sci. USA 2000, 97, 10483–10488. [Google Scholar] [CrossRef]
- Newton-Cheh, C.; Hirschhorn, J.N. Genetic association studies of complex traits: Design and analysis issues. Mutat. Res. Mol. Mech. Mutagen. 2005, 573, 54–69. [Google Scholar] [CrossRef]
- I Thompson, J.; Czernuszka, J.T. The effect of two types of cross-linking on some mechanical properties of collagen. Bio-Med. Mater. Eng. 1995, 5, 37. [Google Scholar]
- Posthumus, M.; September, A.V.; Schwellnus, M.P.; Collins, M. Investigation of the Sp1-binding site polymorphism within the COL1A1 gene in participants with Achilles tendon injuries and controls. J. Sci. Med. Sport 2009, 12, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Erduran, M.; Altinisik, J.; Meric, G.; Ates, O.; Ulusal, A.E.; Akseki, D. Is Sp1 binding site polymorphism within COL1A1 gene associated with tennis elbow? Gene 2014, 537, 308–311. [Google Scholar] [CrossRef] [PubMed]
- Gelse, K. Collagens—Structure, function, and biosynthesis. Adv. Drug Deliv. Rev. 2003, 55, 1531–1546. [Google Scholar] [CrossRef] [PubMed]
- Banos, C.C.; Thomas, A.H.; Kuo, C.K. Collagen fibrillogenesis in tendon development: Current models and regulation of fibril assembly. Birth Defects Res. Part C Embryo Today Rev. 2008, 84, 228–244. [Google Scholar] [CrossRef] [PubMed]
- von der Mark, K. Localization of collagen types in tissues. Int. Rev. Connect. Tissue Res. 1981, 9, 265. [Google Scholar] [PubMed]
- Frank, C.B. Ligament structure, physiology and function. J. Musculoskelet. Neuronal Interact. 2004, 4, 199. [Google Scholar]
- Liu, X.; Wu, H.; Byrne, M.; Krane, S.; Jaenisch, R. Type III collagen is crucial for collagen I fibrillogenesis and for normal cardiovascular development. Proc. Natl. Acad. Sci. USA 1997, 94, 1852–1856. [Google Scholar] [CrossRef]
- Minamitani, T.; Ikuta, T.; Saito, Y.; Takebe, G.; Sato, M.; Sawa, H.; Nishimura, T.; Nakamura, F.; Takahashi, K.; Ariga, H.; et al. Modulation of collagen fibrillogenesis by tenascin-X and type VI collagen. Exp. Cell Res. 2004, 298, 305–315. [Google Scholar] [CrossRef]
- Kim, S.K.; Roos, T.R.; Kleimeyer, J.P.; Ahmed, M.A.; Goodlin, G.T.; Fredericson, M.; Ioannidis, J.P.A.; Avins, A.L.; Dragoo, J.L. Genome-wide association screens for Achilles tendon and ACL tears and tendinopathy. PLoS ONE 2017, 12, e0170422. [Google Scholar] [CrossRef]
- Gillies, A.R.; Lieber, R.L. Structure and Function of the Skeletal Muscle Extracellular Matrix. Muscle Nerve 2011, 44, 318–331. [Google Scholar] [CrossRef]
- Roulet, M.; Ruggiero, F.; Karsenty, G.; LeGuellec, D. A comprehensive study of the spatial and temporal expression of the col5a1 gene in mouse embryos: A clue for understanding collagen V function in developing connective tissues. Cell Tissue Res. 2007, 327, 323–332. [Google Scholar] [CrossRef] [PubMed]
- September, A.V.; Cook, J.; Handley, C.J.; Van Der Merwe, L.; Schwellnus, M.P.; Collins, M. Variants within the COL5A1 gene are associated with Achilles tendinopathy in two populations. Br. J. Sports Med. 2009, 43, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Altinisik, J.; Meric, G.; Erduran, M.; Ates, O.; Ulusal, A.E.; Akseki, D. The BstUI and DpnII Variants of the COL5A1 Gene Are Associated With Tennis Elbow. Am. J. Sports Med. 2015, 43, 1784–1789. [Google Scholar] [CrossRef] [PubMed]
- Heffernan, S.M.; Kilduff, L.P.; Day, S.H.; Pitsiladis, Y.P.; Williams, A.G. Genomics in rugby union: A review and future prospects. Eur. J. Sport Sci. 2015, 15, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Heffernan, S.M.; Kilduff, L.P.; Erskine, R.M.; Day, S.H.; Stebbings, G.K.; Cook, C.J.; Raleigh, S.M.; Bennett, M.A.; Wang, G.; Collins, M.; et al. COL5A1 gene variants previously associated with reduced soft tissue injury risk are associated with elite athlete status in rugby. BMC Genom. 2017, 18, 820. [Google Scholar] [CrossRef] [PubMed]
- Posthumus, M.; September, A.; O’Cuinneagain, D.; Van Der Merwe, W.; Schwellnus, M.; Collins, M. The COL5A1 Gene Is Associated With Increased Risk Of Anterior Cruciate Ligament Rupture In Female Participants: 611. Med. Sci. Sports Exerc. 2009, 41, 2234–2240. [Google Scholar] [CrossRef]
- Brown, K.L.; Seale, K.B.; El Khoury, L.Y.; Posthumus, M.; Ribbans, W.J.; Raleigh, S.M.; Collins, M.; September, A.V. Polymorphisms within the COL5A1 gene and regulators of the extracellular matrix modify the risk of Achilles tendon pathology in a British case-control study. J. Sports Sci. 2017, 35, 1475–1483. [Google Scholar] [CrossRef]
- Lau, N.C.; Lai, E.C. Diverse roles for RNA in gene regulation. Genome Biol. 2005, 6, 315. [Google Scholar] [CrossRef]
- Matzke, M.A.; Birchler, J.A. RNAi-mediated pathways in the nucleus. Nat. Rev. Genet. 2005, 6, 24–35. [Google Scholar] [CrossRef]
- Kim, J.; Bartel, D.P. Allelic imbalance sequencing reveals that single-nucleotide polymorphisms frequently alter microRNA-directed repression. Nat. Biotechnol. 2009, 27, 472–477. [Google Scholar] [CrossRef]
- Foster, B.P. Genetic Variation, Protein Composition and Potential Influences on Tendon Properties in Humans. Open Sports Med. J. 2012, 6, 8–21. [Google Scholar] [CrossRef]
- Gibbon, A.; Hobbs, H.; Van Der Merwe, W.; Raleigh, S.M.; Cook, J.; Handley, C.J.; Posthumus, M.; Collins, M.; September, A.V. The MMP3 gene in musculoskeletal soft tissue injury risk profiling: A study in two independent sample groups. J. Sports Sci. 2016, 35, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Visse, R.; Nagase, H. Matrix Metalloproteinases and Tissue Inhibitors of Metalloproteinases: Structure, Function, and Biochemistry. J. Am. Heart Assoc. 2003, 92, 827–839. [Google Scholar] [CrossRef] [PubMed]
- Petersen, W.; Pufe, T.; Zantop, T.; Tillmann, B.; Tsokos, M.; Mentlein, R. Expression of VEGFR-1 and VEGFR-2 in Degenerative Achilles Tendons. Clin. Orthop. Relat. Res. 2004, 420, 286–291. [Google Scholar] [CrossRef]
- Ferrara, N. Molecular and biological properties of vascular endothelial growth factor. J. Mol. Med. 1999, 77, 527–543. [Google Scholar] [CrossRef] [PubMed]
- Neufeld, G.; Cohen, T.; Gengrinovitch, S.; Poltorak, Z. Vascular endothelial growth factor (VEGF) and its receptors. FASEB J. 1999, 13, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Egginton, S. Invited review: Activity-induced angiogenesis. Pflüg. Archiv-Eur. J. Physiol. 2009, 457, 963–977. [Google Scholar] [CrossRef]
- Rahim, M.; El Khoury, L.Y.; Raleigh, S.M.; Ribbans, W.J.; Posthumus, M.; Collins, M.; September, A.V. Human Genetic Variation, Sport and Exercise Medicine, and Achilles Tendinopathy: Role for Angiogenesis-Associated Genes. OMICS J. Integr. Boil. 2016, 20, 520–527. [Google Scholar] [CrossRef]
- Rahim, M.; Hobbs, H.; Van Der Merwe, W.; Posthumus, M.; Collins, M.; September, A.V. Investigation of angiogenesis genes with anterior cruciate ligament rupture risk in a South African population. J. Sports Sci. 2017, 36, 551–557. [Google Scholar] [CrossRef]
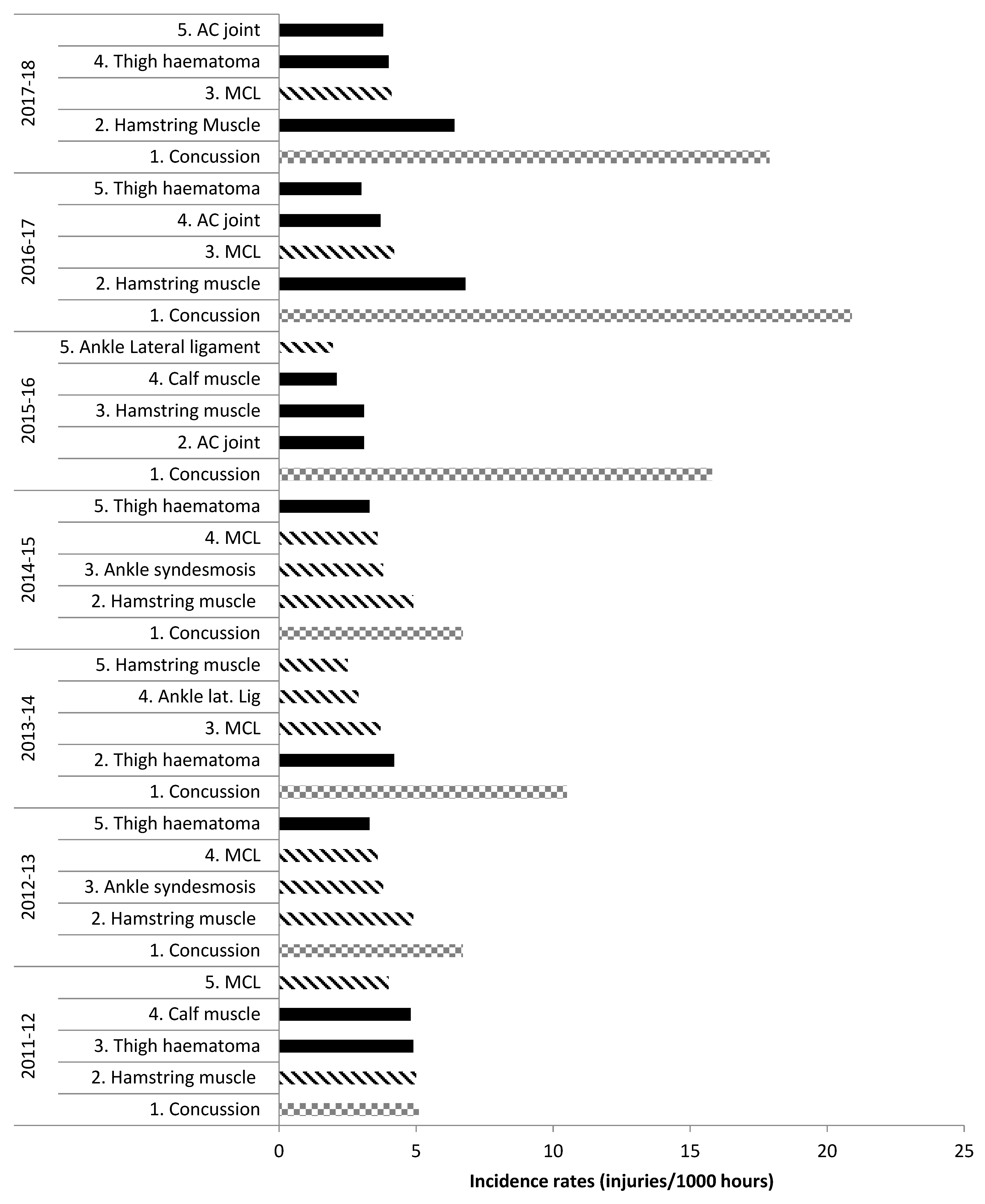
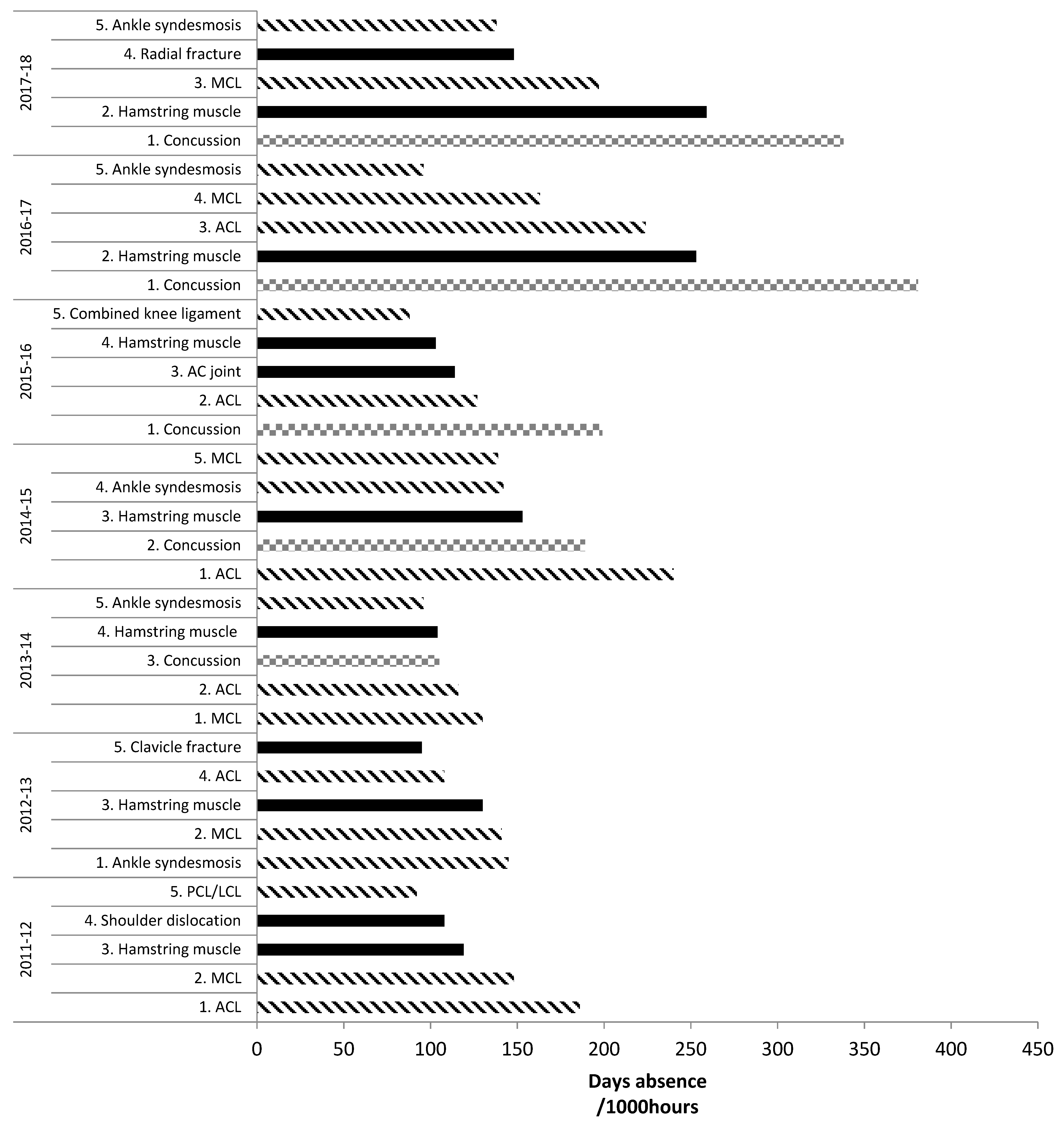
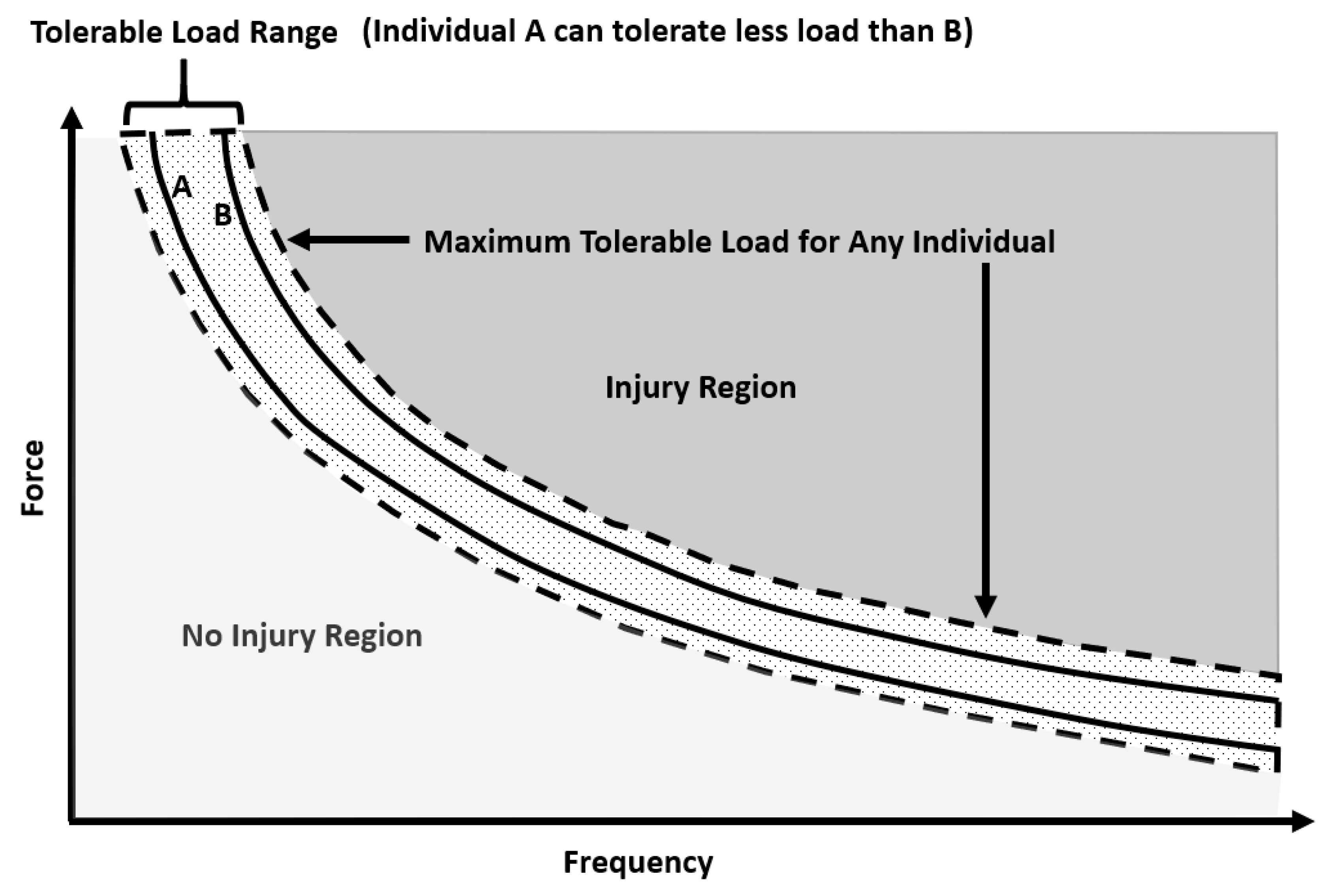
| Study | Level | Injury Type | Match Injuries Incidence (Injuries/1000 Player Hours (95% CI)) | |||
|---|---|---|---|---|---|---|
| Main Group | Sub-Group | Forwards | Backs | All | ||
| Brooks et al. [6] 1,2 | English Premiership clubs 4 | Muscle/Tendon | Strain/Tear/Rupture | 14 * | 20 * | 17 * |
| Ligament/Joint (non-bone) | Sprain/Rupture | 13 * | 15 * | 14 * | ||
| Fuller et al. [14] | International | Muscle/Tendon | Muscle Rupture/Tear/Strain | 18 (12–29) | 27 (18–40) | 22 (17–30) |
| Tendon rupture/Tendinopathy | 0 | 1 (0.2–8) | 0.5 (0.1–4) | |||
| Ligament/Joint (non-bone) | Sprain/Rupture | 25 (17–37) | 23 (15–36) | 25 (18–33) | ||
| Fuller et al. [7] | International | Muscle/Tendon | Muscle rupture/Tear/Strain | 20 (13–30) | 20 (13–32) | 20 (14–27) |
| Tendon rupture/Tendinopathy | 1 (0.1–7) | 5 (2–12) | 3 (1–6) | |||
| Ligament/Joint (non-bone) | Sprain/rupture | 22 (14–33) | 18 (11–29) | 20 (14–27) | ||
| Moore et al. [32] | International | Muscle/Tendon | Muscle strain/rupture | - | - | 34 (23–49) |
| Tendinopathy/rupture | - | - | 9 (4–18) | |||
| Ligament/Joint (non-bone) | Sprain/rupture | - | - | 43 (31–61) | ||
| Fuller et al. [8] | International | Muscle/Tendon | - | 34 | 39 | - |
| Ligament/Joint (non-bone) | - | 25 | 34 | |||
| Williams et al. [11] 3 | English Premiership clubs 4, Super 14 clubs 4, Vodacom cup 2008 clubs 4 and International | Muscle/Tendon | - | - | - | 40 (21–76) |
| Ligament/Joint (non-bone) | - | - | - | 34 (18–65) | ||
| Study | Level | Injury Type | Match Injuries Severity (Days Absence (95% CI)) | |||
|---|---|---|---|---|---|---|
| Main Group | Sub-Group | Forwards | Backs | All | ||
| Brooks et al. [6] 1,2 | English Premiership clubs 5 | Muscle/Tendon | Strain/Tear/Rupture | 20 # | 16 # | 17 # |
| Ligament/Joint (non-bone) | Sprain/Rupture | 22 # | 26 # | 24 # | ||
| Fuller et al. [14] | International | Muscle/Tendon | Muscle Rupture/Tear/Strain | 17 (10–25) | 21 (9–33) | 20 (12–27) |
| Tendon Rupture/Tendinopathy | 0 | 4 * | 4 * | |||
| Ligament/Joint (non-bone) | Sprain/Rupture | 14 (8–20) | 18 (9–27) | 16 (11–21) | ||
| Fuller et al. [7] | International | Muscle/Tendon | Muscle Rupture/Tear/Strain | 15 (8–23) | 27 (16–38) | 21 (14–28) |
| Tendon Rupture/Tendinopathy | 4 * | 36 (0–92) | 29 (0–75) | |||
| Ligament/Joint (non-bone) | Sprain/Rupture | 38 (8–68) | 42 (12–72) | 39 (18–61) | ||
| Fuller et al. [8] 2,3 | International | Ligament/Joint (non-bone) | Knee Ligament | - | - | 1507 |
| Achilles Tendon | - | - | 188 * | |||
| Williams et al. [11] 4 | English Premiership Clubs 5 and International | Muscle/Tendon | - | - | - | 15 (5–24) |
| Ligament/Joint (non-bone) | - | - | - | 29 (19–39) | ||
| Candidate Protein | Candidate Protein Abbreviation | Candidate Gene | Candidate Gene Abbreviations |
|---|---|---|---|
| Type I collagen | Col I | Collagen type I alpha I | COL1A1 |
| Type III collagen | Col III | Collagen type III alpha I | COL3A1 |
| Type V collagen | Col V | Collagen type V alpha I | COL5A1 |
| N/A Non-coding RNA | N/A | MicroRNA 608 | MIR608 |
| Matrix metalloproteinase-3 | MMP3 | Matrix metalloproteinase-3 | MMP3 |
| Tissue inhibitors of metalloproteinases-2 | TIMP2 | Tissue inhibitors of metalloproteinases-2 | TIMP2 |
| Vascular endothelial growth factor A | VEGFA | Vascular endothelial growth factor A | VEGFA |
| Nidogen 1 | NID1 | Nidogen 1 | NID1 |
| Collagen beta(1-O) galactosyltransferase 1 | COLGALT1 | Collagen beta(1-O) galactosyltransferase 1 | COLGALT1 |
| Study | Phenotype | Target Population | Participants | Findings |
|---|---|---|---|---|
| Khoschnau et al. [115] | Cruciate ligament ruptures, shoulder dislocations | Sweden | No ethnicity reported. 233 cruciate ligament injury participants, 126 shoulder dislocation participants, 325 female controls | Individuals with TT genotype had a reduced risk of injury for cruciate ligament ruptures and shoulder dislocations compared to GG carriers. |
| Posthumus et al. [116] | ACL injuries | SA | Caucasian, 117 ACL rupture participants, 130 controls | TT genotype underrepresented in ACL injury group compared to controls. |
| Posthumus et al. [147] | Achilles tendinopathy, Achilles tendon ruptures | SA | Caucasian, 85 Achilles tendinopathy participants, 41 participants with partial or complete ruptures, 126 controls | No differences in genotypes. |
| Ficek et al. [122] | ACL injuries | Poland | Caucasian, 91 professional football players with ACL rupture—all non-contact, 143 apparently healthy professional soccer players as controls | No differences in genotypes. There was an overrepresentation of G–T haplotypes (1997G+1245T) in controls suggesting, carriers may have reduced risk of injury. |
| Stępien-Słodkowska et al. [121] | ACL injuries | Poland | No ethnicity reported, 138 male recreational skiers with ACL rupture, 183 apparently healthy male skiers as controls | Carriers of the GG genotype were at lower risk of ACL injury than carriers of the TT. genotype. |
| Erduran et al. [148] | Tennis elbow | Turkey | No ethnicity reported, 103 with tennis elbow, 103 apparently healthy controls | No differences in genotypes. |
| Study | Phenotype | Target Population | Participants | Findings |
|---|---|---|---|---|
| Stephien-Slodkowska et al. [129] | ACL rupture | Poland | No ethnicity reported, 138 male recreational skiers with ACL ruptures, 183 male apparently healthy skiers | The AA genotype was overrepresented in the ACL group compared to controls. |
| O’Connell et al. [127] | ACL rupture | SA/ Poland | Caucasian. 333 participants with ACL rupture (242 SA and 91 Poland), 378 apparently healthy controls (235 SA and 143 Poland). | No differences in genotype frequency distributions between the SA ACL group and the SA control group. However, the AA genotype was overrepresented in the Polish ACL group compared to Polish controls. No allele associations for any of the groups. |
| Kim et al. [155] | ACL rupture | Caucasian, Latin-American, East Asian, African, South-East Asian. 5148 Achilles tendon injury participants, 97,831 apparently healthy controls, 598 ACL rupture participants, 98,744 apparently healthy controls, | No associations after Benjamini–Hochberg correction for testing multiple hypotheses. |
| Study | Phenotype | Target Population | Participants | Findings |
|---|---|---|---|---|
| Mokone et al. [131] | Achilles tendon pathology, Achilles tendinopathy, Achilles tendon rupture | SA | Caucasian, 111 participants with current or past history of Achilles tendon pathology, including 72 chronic tendinopathy participants, 39 Achilles tendon rupture participants | The frequency of the A2 (C) allele was higher in the controls compared to the Achilles tendon pathology group. An even stronger protective role was seen for the A2 (C) allele in in controls compared to the chronic tendinopathy patients. |
| September et al. [158] | Achilles tendinopathy | SA/Australia | Caucasian, 83 Australian and 93 SA tendinopathy patients, 210 Australian, and 132 SA controls | Individuals with CC genotype in both populations (Australian/SA) had a reduced risk of developing Achilles tendinopathy compared to any other genotypes. |
| Posthumus et al. [162] | ACL injuries | SA | Caucasian, 129 ACL rupture participants, 216 physically active controls with no history of ACL injury | The CC genotype was underrepresented in the female ACL rupture group, but not in the male. |
| Stepien–Slodkowska et al. [128] | ACL injuries | Poland | No ethnicity reported, 138 male recreational skiers with ACL ruptures, 183 apparently healthy male recreational skiers without any reported history of ligament or tendon injury. | No differences in genotype distribution between groups. Higher frequency of rs12722 C-T and rs13946 C-T polymorphisms haplotype in controls suggests reduced risk of ACL injury. |
| Altinisik et al. [159] | Tennis elbow | Turkey | No ethnicity reported, 152 tennis elbow patients, 195 healthy controls. | Individuals with the A2 (C) allele were underrepresented in patient group. Individuals with A1 allele (T) have an increased risk of developing tennis elbow. |
| Brown et al. [163] | Achilles tendinopathy, Achilles tendon rupture | UK | Caucasian, 87 Achilles tendinopathy participants, 25 Achilles tendon rupture participants, 130 asymptomatic controls | No independent differences found between groups. Three inferred allele combinations from rs12722, rs3196378, and rs71746744 were identified as risk modifiers. The T–C–D combination was associated with increased risk of Achilles tendon pathology and rupture, the C–A–I combination was associated with increased risk of Achilles tendon pathology, tendinopathy and rupture, the C–C–D combination was associated with decreased risk of Achilles tendon pathology and rupture. |
| Kim et al. [155] | Achilles tendinopathy, Achilles tendon rupture | Caucasian, Latin-American, East Asian, African, South-East Asian, 5148 Achilles tendon injury participants, 97,831 apparently healthy controls, 598 ACL rupture participants, 98,744 apparently healthy controls | No associations after Benjamini–Hochberg correction for testing multiple hypotheses. |
| Study | Phenotype | Target Population | Participants | Findings |
|---|---|---|---|---|
| Abrahams et al. [135] | Achilles tendinopathy | SA/Australia | Caucasian, 160 chronic Achilles tendinopathy participants, 342 apparently healthy controls | The CC genotype frequency of rs4919510 was overrepresented compared to the CG and GG genotypes. The combined rs4919510 CC genotype and COL5A1 rs3196378 CA genotype was overrepresented in the tendon group compared to controls. Furthermore, the rs4919510 CC genotype and the COL5A1 rs3196378 A allele was overrepresented in the tendon group compared to controls. |
| Brown et al. [163] | Achilles tendinopathy and Achilles tendon rupture | UK | Caucasian, 112 Achilles tendon pathology participants (87 chronic Achilles tendinopathy and 25 Achilles tendon rupture, 130 apparently healthy controls | No differences in genotype frequency or allele frequency distributions between Achilles tendinopathy and controls. However, the CG genotype of rs4919510 was associated with decreased risk of rupture compared to controls. When inferred allele combinations were analyzed for rs4919510 and COL5A1 rs3196378, and no associations found with risk of Achilles tendinopathy. |
| Kim et al. [155] | Achilles tendinopathy, Achilles tendon rupture | Caucasian, Latin-American, East Asian, African, South-East Asian, 5148 Achilles tendon injury participants, 97,831 apparently healthy controls, 598 ACL rupture participants, 98,744 apparently healthy controls | Moderate–weak evidence of replication (p = 5.1 × 10−3) for Achilles tendinopathy or rupture, but no replication with ACL rupture, after Benjamini–Hochberg correction for testing multiple hypotheses. |
| Study | Phenotype | Target Population | Participants | Findings |
|---|---|---|---|---|
| Raleigh et al. [132] | Achilles tendinopathy and rupture | SA | Caucasian, 114 Achilles tendon pathology patients including 75 with Achilles tendinopathy and 39 with partial or complete rupture, 98 controls | Independent associations between the GG genotype of rs679620, the CC genotype of rs591058, and the AA genotype of rs650108 and Achilles tendinopathy. The ATG haplotype (rs679620, rs591058, and rs650108) was under-represented in the tendinopathy compared to controls. No associations between MMP3 variants and Achilles tendon rupture. |
| Posthumus et al. [120] | ACL rupture | SA | Caucasian, 129 ACL rupture patients, 216 apparently healthy controls | No independent associations for rs679620 compared to controls. Haplotypes T-1G-A-A and C-2G-G-G (MMP10 rs485055, MMP1 rs1799750, MMP3 rs679620, and MMP12 rs2276109) were different between control and ACL groups and controls and non-contact subgroup, respectively. |
| El Khoury et al. [140] | Achilles tendinopathy and rupture | UK | Caucasian. 118 Achilles tendon pathology patients including 93 with Achilles tendinopathy and 25 participants with partial or complete rupture, 131 asymptomatic controls | rs679620 GG genotype overrepresented in Achilles tendon rupture group compared to controls. No association with Achilles tendinopathy. |
| Gibbon et al. [168] | Achilles tendinopathy ACL rupture | Australia/SA | White Caucasian, 160 Achilles tendinopathy patients, 195 apparently healthy controls, 234 ACL rupture patients, 232 apparently healthy controls | No independent differences for rs679620, rs591058 and 650108 between Achilles tendinopathy and controls or between ACL rupture and controls. Haplotype 6a-G-C-G (rs3205058, rs679620, rs591058 and rs650108) overrepresented in the control group compared to the Achilles tendinopathy group when only Australian samples analyzed. No genotype or allele frequency differences from inferred haplotypes for ACL injury. |
| Kim et al. [155] | Achilles tendinopathy, Achilles tendon rupture | Caucasian, Latin-American, East Asian, African, South-East Asian. 5148 Achilles tendon injury participants. 97,831 apparently healthy controls, 598 ACL rupture participants, 98,744 apparently healthy controls | No associations after Benjamini–Hochberg correction for testing multiple hypotheses. |
| Study | Phenotype | Target Population | Participants | Findings |
|---|---|---|---|---|
| El Khoury et al. [137] | Achilles tendinopathy and rupture | SA/Australia | Caucasian, 173 Achilles tendon pathology participants of which 134 with Achilles tendinopathy and 39 with partial or complete rupture, 248 asymptomatic controls | Association between TIMP2 rs4789932 and Achilles tendinopathy. The CC variant was overrepresented within controls, while the CT variant was overrepresented within the combined Achilles tendon pathology group. No differences between the rupture group and controls. |
| El Khoury et al. [140] | Achilles tendinopathy and rupture | UK | Caucasian, 118 Achilles tendon pathology participants of which 93 had chronic Achilles tendinopathy and 25 participants with Achilles tendon rupture, 131 asymptomatic controls | Difference in genotype frequency between male Achilles tendon pathology compared to male controls. Further, difference between male ruptures compared to controls. The CT genotype was associated with lower risk of Achilles tendon pathology. |
| Kim et al. [155] | Achilles tendinopathy, Achilles tendon rupture | Caucasian, Latin-American, East Asian, African, South-East Asian 5148 Achilles tendon injury participants, 97,831 apparently healthy controls 598 ACL rupture participants, 98,744 apparently healthy controls. | No associations after Benjamini–Hochberg correction for testing multiple hypotheses. |
| Study | Phenotype | Target Population | Participants | Findings |
|---|---|---|---|---|
| Rahim et al. [125] | ACL rupture | SA | Caucasian. 227 ACL rupture participants, 227 apparently healthy controls with no history of ACL injury | The CC genotype of rs699947 was overrepresented in participants with non-contact ACL ruptures compared to controls. The rs1570360 GA genotype was overrepresented within controls. The A-A-G haplotype (rs699947, rs1570360 and 2010963) was overrepresented in the control group compared to the non-contact ACL group. |
| Rahim et al. [174] | Achilles tendinopathy | SA/UK | Caucasian, 195 chronic Achilles tendinopathy participants (87 from SA, 108 UK), 250 asymptomatic controls (120 SA, 130 from UK) | The CC genotype of rs699947 was overrepresented in the SA control group compared to the SA tendinopathy group. No other independent frequency differences found. The VEGFA A-G-G inferred haplotype (rs699947, rs1570360, and rs2010963) was associated with increased risk of tendinopathy in the SA group and the SA and UK combined group. |
| Rahim et al. [175] | ACL rupture | SA | SA colored ethnic group (unique to Western Cape of SA), 98 ACL rupture participants, 100 physically active asymptomatic controls with no history of tendon or ligament injury | No differences in genotype or allele frequency data for any of the VEGFA polymorphisms studied. Further, no associations found from inferred haplotype analysis. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brazier, J.; Antrobus, M.; Stebbings, G.K.; Day, S.H.; Heffernan, S.M.; Cross, M.J.; Williams, A.G. Tendon and Ligament Injuries in Elite Rugby: The Potential Genetic Influence. Sports 2019, 7, 138. https://doi.org/10.3390/sports7060138
Brazier J, Antrobus M, Stebbings GK, Day SH, Heffernan SM, Cross MJ, Williams AG. Tendon and Ligament Injuries in Elite Rugby: The Potential Genetic Influence. Sports. 2019; 7(6):138. https://doi.org/10.3390/sports7060138
Chicago/Turabian StyleBrazier, Jon, Mark Antrobus, Georgina K. Stebbings, Stephen H. Day, Shane M. Heffernan, Matthew J. Cross, and Alun G. Williams. 2019. "Tendon and Ligament Injuries in Elite Rugby: The Potential Genetic Influence" Sports 7, no. 6: 138. https://doi.org/10.3390/sports7060138
APA StyleBrazier, J., Antrobus, M., Stebbings, G. K., Day, S. H., Heffernan, S. M., Cross, M. J., & Williams, A. G. (2019). Tendon and Ligament Injuries in Elite Rugby: The Potential Genetic Influence. Sports, 7(6), 138. https://doi.org/10.3390/sports7060138







