The Dose Response for Sprint Interval Training Interventions May Affect the Time Course of Aerobic Training Adaptations
Abstract
1. Introduction
2. Material and Methods
2.1. Experimental Design
2.2. Participants
2.3. Control Period (BL1 and 2—Wk 1–6)
2.4. Training Intervention (Wk 7–12)
2.5. Training Cessation Period (Wk 13–15)
2.6. Testing Procedures
2.6.1. Baseline 1 (BL1)
2.6.2. BL2, 0POST, and 3POST
2.7. Data and Statistical Analysis
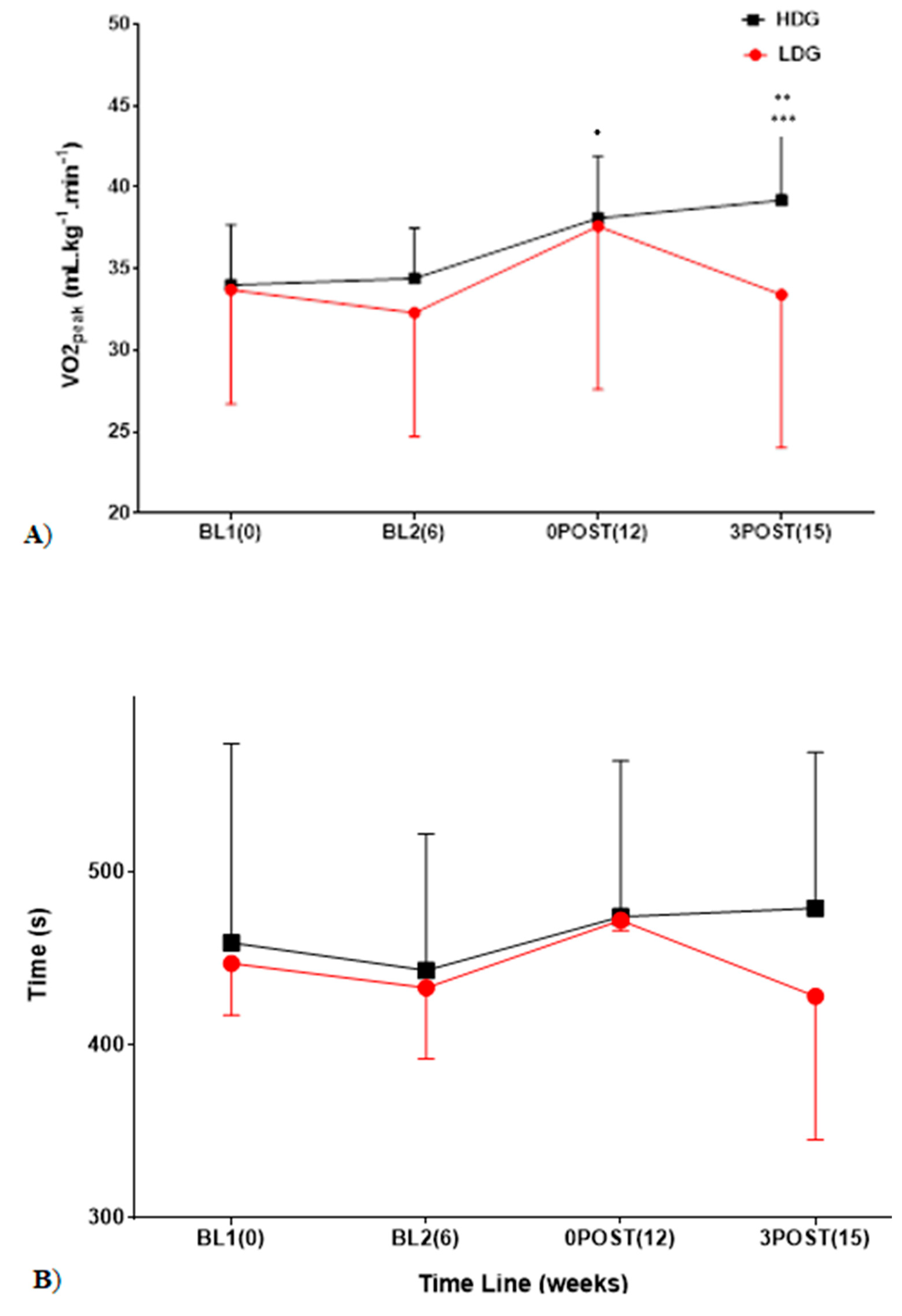
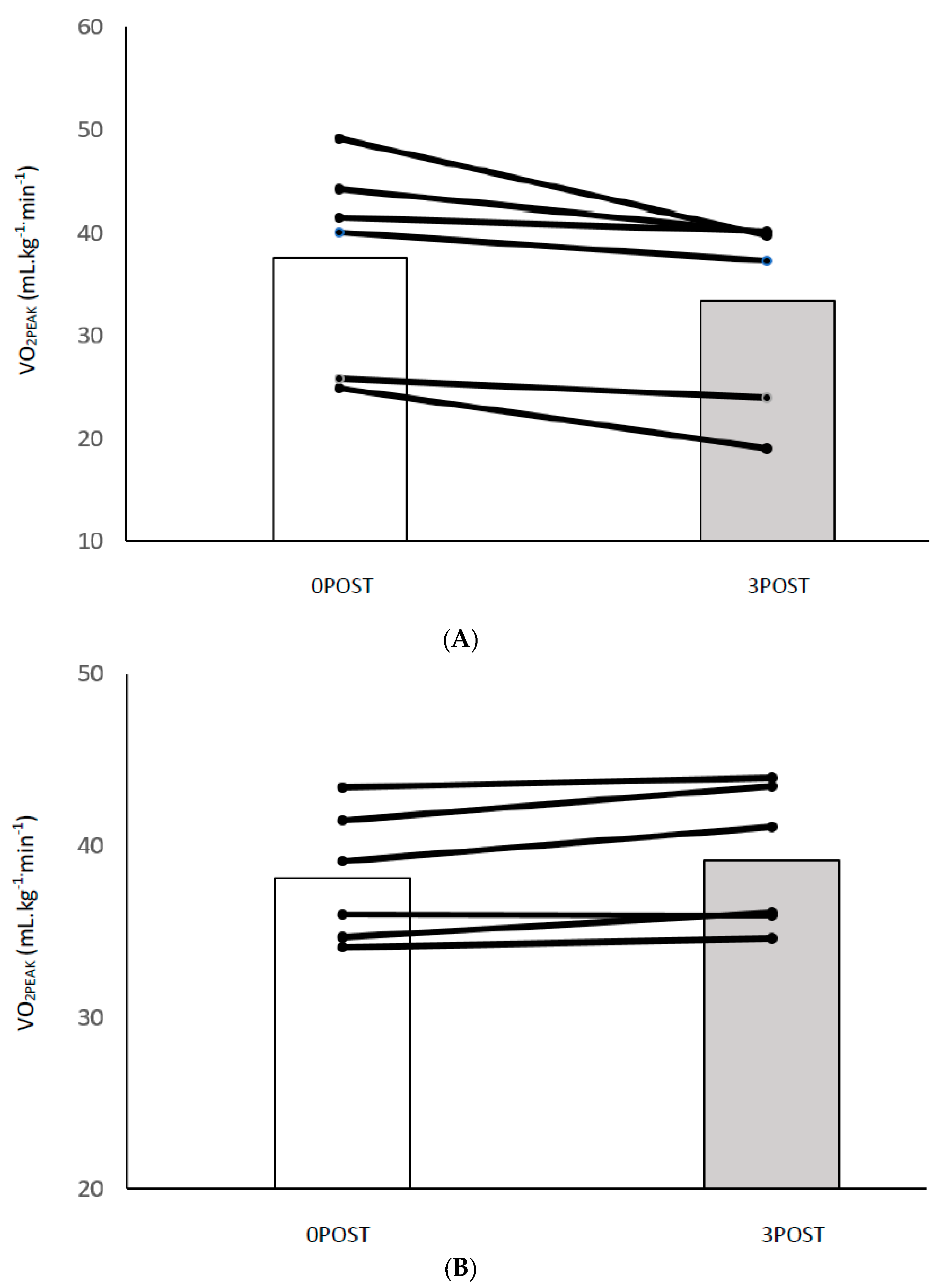
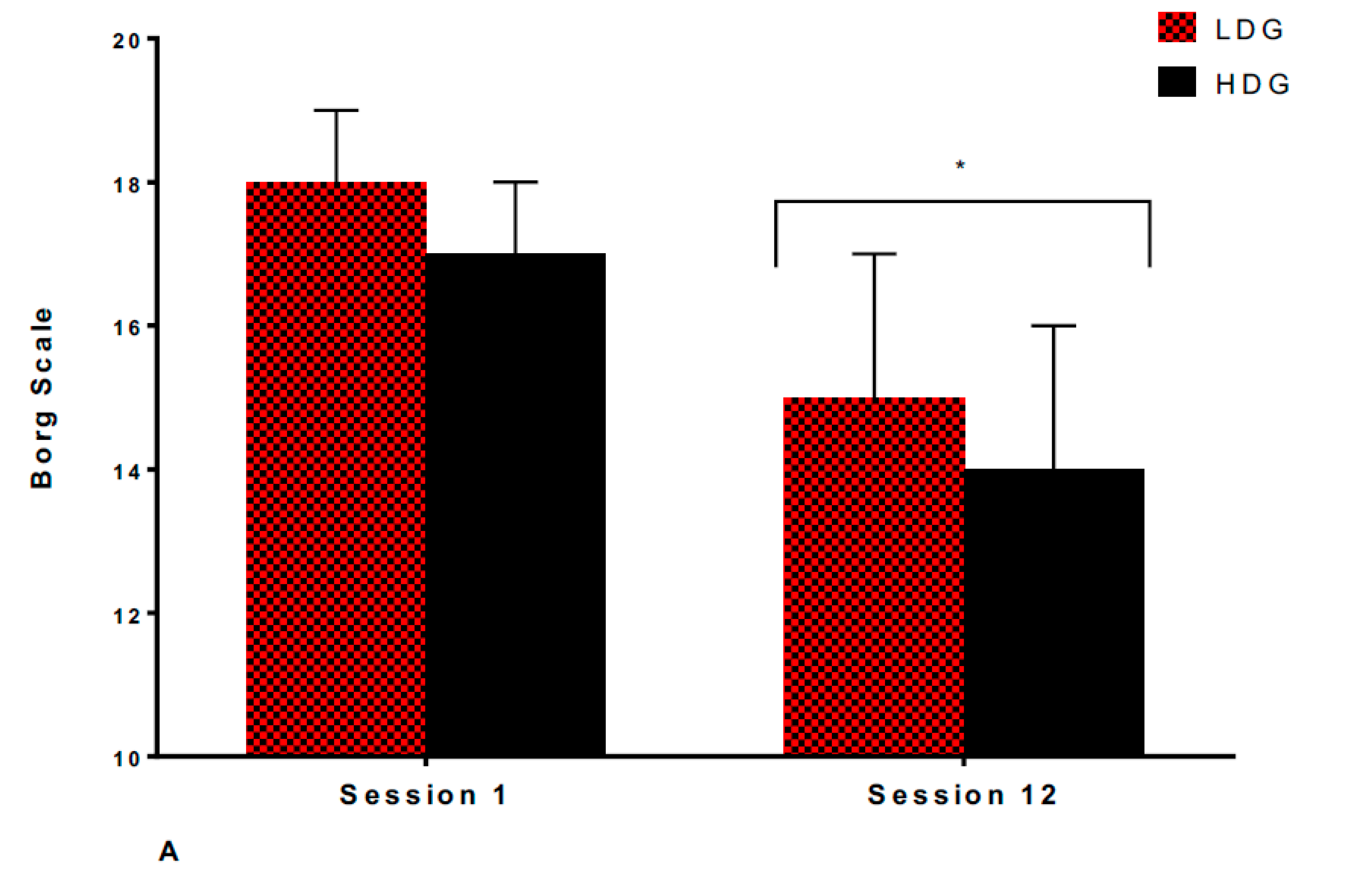
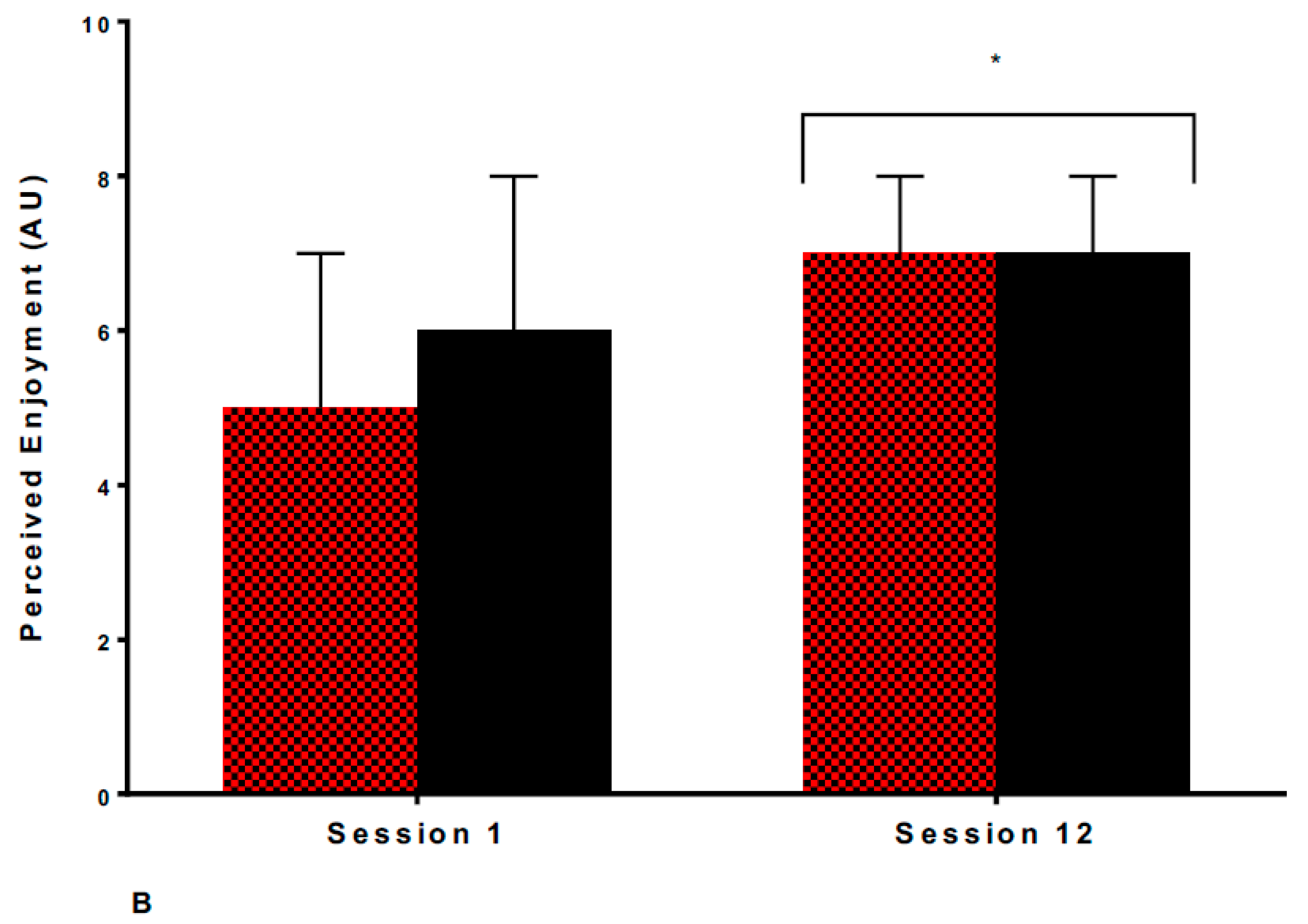
3. Results
3.1. Indices of Cardiorespiratory Fitness
3.2. Psychological Responses
3.3. Blood Lipid Profile
3.4. Anthropometric Indices
3.5. Blood Pressure
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hallal, P.C.; Andersen, L.B.; Bull, F.C.; Guthold, R.; Haskell, W.; Ekelund, U.; Lancet Physical Activity Series Working Group. Global physical activity levels: Surveillance progress, pitfalls, and prospects. Lancet 2012, 380, 247–257. [Google Scholar] [CrossRef]
- Trost, S.G.; Owen, N.; Bauman, A.E.; Sallis, J.F.; Brown, W. Correlates of adults’ participation in physical activity: Review and update. Med. Sci. Sports Exerc. 2002, 34, 1996–2001. [Google Scholar] [CrossRef]
- Metcalfe, R.S.; Babraj, J.A.; Fawkner, S.G.; Vollaard, N.B. Towards the minimal amount of exercise for improving metabolic health: Beneficial effects of reduced-exertion high-intensity interval training. Eur. J. Appl. Physiol. 2012, 112, 2767–2775. [Google Scholar] [CrossRef]
- Gibala, M.J.; McGee, S.L. Metabolic adaptations to short-term high-intensity interval training: A little pain for a lot of gain? Exerc. Sport Sci. Rev. 2008, 36, 58–63. [Google Scholar] [CrossRef]
- Burgomaster, K.A.; Hughes, S.C.; Heigenhauser, G.J.; Bradwell, S.N.; Gibala, M.J. Six sessions of sprint interval training increases muscle oxidative potential and cycle endurance capacity in humans. J. Appl. Physiol. 1985, 98, 1985–1990. [Google Scholar] [CrossRef] [PubMed]
- Trapp, E.G.; Chisholm, D.J.; Freund, J.; Boutcher, S.H. The effects of high-intensity intermittent exercise training on fat loss and fasting insulin levels of young women. Int. J. Obes. (Lond.) 2008, 32, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Gillen, J.B.; Percival, M.E.; Skelly, L.E.; Martin, B.J.; Tan, R.B.; Tarnopolsky, M.A.; Gibala, M.J. Three minutes of all-out intermittent exercise per week increases skeletal muscle oxidative capacity and improves cardiometabolic health. PLoS ONE 2014, 9, e111489. [Google Scholar] [CrossRef] [PubMed]
- Lacewell, A.N.; Buck, T.M.; Romero, S.A.; Halliwill, J.R. Postexercise syncope: Wingate syncope test and effective countermeasure. Exp. Physiol. 2014, 99, 172–186. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, J.D.; Close, G.L.; MacLaren, D.P.M.; Gregson, W.; Drust, B.; Morton, J.P. High-intensity interval running is perceived to be more enjoyable than moderate-intensity continuous exercise: Implications for exercise adherence. J. Sports Sci. 2011, 29, 547–553. [Google Scholar] [CrossRef]
- Vollard, N.B.J.; Metcalfe, R.S. Research into the Health Benefits of Sprint Interval Training Should Focus on Protocols with Fewer and Shorter Sprints. Sports Med. 2017, 47, 2443–2451. [Google Scholar] [CrossRef] [PubMed]
- Logan, G.R.; Harris, N.; Duncan, S.; Plank, L.D.; Merien, F.; Schofield, G. Low-Active Male Adolescents: A Dose Response to High-Intensity Interval Training. Med. Sci. Sports Exerc. 2016, 48, 481–490. [Google Scholar] [CrossRef]
- Kreher, J.B. Diagnosis and prevention of overtraining syndrome: An opinion on education strategies. Open Access J. Sports Med. 2016, 7, 115–122. [Google Scholar] [CrossRef]
- Steele, J.; Fisher, J.; Skivington, M.; Dunn, C.; Arnold, J.; Tew, G.; Batterham, A.M.; Nunan, D.; O’Driscoll, J.M.; Mann, S.; et al. A higher effort-based paradigm in physical activity and exercise for public health: Making the case for a greater emphasis on resistance training. BMC Public Health 2017, 17, 300. [Google Scholar] [CrossRef]
- Hooper, A.E.; Bryan, A.D.; Eaton, M. Menstrual cycle effects on perceived exertion and pain during exercise among sedentary women. J. Womens Health (Larchmt) 2011, 20, 439–446. [Google Scholar] [CrossRef]
- Jakeman, J.; Adamson, S.; Babraj, J. Extremely short duration high-intensity training substantially improves endurance performance in triathletes. Appl. Physiol. Nutr. Metab. 2012, 37, 976–981. [Google Scholar] [CrossRef]
- Little, J.P.; Gillen, J.B.; Percival, M.E.; Safdar, A.; Tarnopolsky, M.A.; Punthakee, Z.; Jung, M.E.; Gibala, M.J. Low-volume high-intensity interval training reduces hyperglycemia and increases muscle mitochondrial capacity in patients with type 2 diabetes. J. Appl. Physiol. 2011, 111, 1554–1560. [Google Scholar] [CrossRef]
- Pescatello, L. ACSM Guidelines for Exercise Testing and Prescription Philadelphia; Walters Kluwer/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2014. [Google Scholar]
- Bonsu, B.; Terblanche, E. The training and detraining effect of high-intensity interval training on post-exercise hypotension in young overweight/obese women. Eur. J. Appl. Physiol. 2016, 116, 77–84. [Google Scholar] [CrossRef]
- Drust, B.; Waterhouse, J.; Atkinson, G.; Edwards, B.; Reilly, T. Circadian rhythms in sports performance—An update. Chronobiol. Int. 2005, 22, 21–44. [Google Scholar] [CrossRef]
- Cohen, D. Statistical Power Analysis for the Behavioral Sciences; Routledge Academic: New York, NY, USA, 1988. [Google Scholar]
- Mujika, I.; Padilla, S. Cardiorespiratory and metabolic characteristics of detraining in humans. Med. Sci. Sports Exerc. 2001, 33, 413–421. [Google Scholar] [CrossRef]
- Tabata, I.; Nishimura, K.; Kouzaki, M.; Hirai, Y.; Ogita, F.; Miyachi, M.; Yamamoto, K. Effects of moderate-intensity endurance and high-intensity intermittent training on anaerobic capacity and VO2max. Med. Sci. Sports Exerc. 1996, 28, 1327–1330. [Google Scholar] [CrossRef]
- Gillen, J.B.; Martin, B.J.; MacInnis, M.J.; Skelly, L.E.; Tarnopolsky, M.A.; Gibala, M.J. Twelve Weeks of Sprint Interval Training Improves Indices of Cardiometabolic Health Similar to Traditional Endurance Training despite a Five-Fold Lower Exercise Volume and Time Commitment. PLoS ONE 2016, 11, e0154075. [Google Scholar] [CrossRef]
- Cobley, J.N.; Bartlett, J.D.; Kayani, A.; Murray, S.W.; Louhelainen, J.; Donovan, T.; Waldron, S.; Gregson, W.; Burniston, J.G.; Morton, J.P.; et al. PGC-1alpha transcriptional response and mitochondrial adaptation to acute exercise is maintained in skeletal muscle of sedentary elderly males. Biogerontology 2012, 13, 621–631. [Google Scholar] [CrossRef]
- Mujika, I.; Padilla, S. Detraining: Loss of training-induced physiological and performance adaptations. Part I: Short term insufficient training stimulus. Sports Med. 2000, 30, 79–87. [Google Scholar] [CrossRef]
- Marrier, B.; Robineau, J.; Piscione, J.; Lacome, M.; Peeters, A.; Hausswirth, C.; Morin, J.B.; Le Meur, Y. Supercompensation Kinetics of Physical Qualities During a Taper in Team-Sport Athletes. Int. J. Sports Physiol. Perform. 2017, 12, 1163–1169. [Google Scholar] [CrossRef]
- Thomas, L.; Busso, T. A theoretical study of taper characteristics to optimize performance. Med. Sci. Sports Exerc. 2005, 37, 1615–1621. [Google Scholar] [CrossRef]
- Mujika, I. Intense training: The key to optimal performance before and during the taper. Scand. J. Med. Sci. Sports 2010, 20 (Suppl. 2), 24–31. [Google Scholar] [CrossRef]
- Schoenfeld, B.; Dawes, J.M.S. High-Intensity Interval Training: Applications for General Fitness Training. Strength Cond. J. 2009, 31, 44–46. [Google Scholar] [CrossRef]
- Hatle, H.; Stobakk, P.K.; Molmen, H.E.; Brønstad, E.; Tjønna, A.E.; Steinshamn, S.; Skogvoll, E.; Wisløff, U.; Ingul, C.B.; Rognmo, Ø. Effect of 24 sessions of high-intensity aerobic interval training carried out at either high or moderate frequency, a randomized trial. PLoS ONE 2014, 9, e88375. [Google Scholar] [CrossRef]
- Rakobowchuk, M.; Tanguay, S.; Burgomaster, K.A.; Howarth, K.R.; Gibala, M.J.; MacDonald, M.J. Sprint interval and traditional endurance training induce similar improvements in peripheral arterial stiffness and flow-mediated dilation in healthy humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 295, R236–R242. [Google Scholar] [CrossRef]
- Whyte, L.J.; Gill, J.M.; Cathcart, A.J. Effect of 2 weeks of sprint interval training on health-related outcomes in sedentary overweight/obese men. Metabolism 2010, 59, 1421–1428. [Google Scholar] [CrossRef]
- Adamson, S.B.; Lorimer, R.; Cobley, J.N.; Babraj, J.A. Extremely short-duration high-intensity training substantially improves the physical function and self-reported health status of elderly adults. J. Am. Geriatr. Soc. 2014, 62, 1380–1381. [Google Scholar] [CrossRef]
- Cardoso, C.G., Jr.; Gomides, R.S.; Queiroz, A.C.; Pinto, L.G.; da Silveira Lobo, F.; Tinucci, T.; Mion, D., Jr.; de Moraes Forjaz, C.L. Acute and chronic effects of aerobic and resistance exercise on ambulatory blood pressure. Clinics (Sao Paulo) 2010, 65, 317–325. [Google Scholar] [CrossRef]
- Cornelissen, V.A.; Smart, N.A. Exercise training for blood pressure: A systematic review and meta-analysis. J. Am. Heart Assoc. 2013, 2, e004473. [Google Scholar] [CrossRef]
- Fisher, G.; Brown, A.W.; Bohan Brown, M.M.; Alcorn, A.; Noles, C.; Winwood, L.; Resuehr, H.; George, B.; Jeansonne, M.M.; Allison, D.B. High Intensity Interval- vs. Moderate Intensity- Training for Improving Cardiometabolic Health in Overweight or Obese Males: A Randomized Controlled Trial. PLoS ONE 2015, 10, e0138853. [Google Scholar] [CrossRef]
- Nybo, L.; Sundstrup, E.; Jakobsen, M.D.; Mohr, M.; Hornstrup, T.; Simonsen, L.; Bülow, J.; Randers, M.B.; Nielsen, J.J.; Aagaard, P.; et al. High-intensity training versus traditional exercise interventions for promoting health. Med. Sci. Sports Exerc. 2010, 42, 1951–1958. [Google Scholar] [CrossRef]
- Boutcher, S.H. High-intensity intermittent exercise and fat loss. J. Obes. 2011, 2011, 868305. [Google Scholar] [CrossRef]
- Townsend, L.K.; Islam, H.; Dunn, E.; Eys, M.; Robertson-Wilson, J.; Hazell, T.J. Modified sprint interval training protocols. Part II. Psychological responses. Appl. Physiol. Nutr. Metab. 2017, 42, 347–353. [Google Scholar] [CrossRef]
- Coyle, E. Very intense exercise-training is extremely potent and time efficient: A reminder. J. Appl. Physiol. 2005, 98, 1983–1984. [Google Scholar] [CrossRef][Green Version]
| Group | BL1 | BL2 | 0POST | 3POST | Time | Group × Time | |
|---|---|---|---|---|---|---|---|
| Body Mass (kg) | LDG | 95.4 ± 27.8 | 96.0 ± 26.5 | 96.0 ± 26.1 | 96.0 ± 26.9 | p = 0.987 | p = 0.622 |
| HDG | 86.4 ± 7.0 | 85.8 ± 7.4 | 85.8 ± 6.5 | 86.1 ± 7.5 | |||
| Body Fat (%) | LDG | 27.6 ± 7.8 | 27.4 ± 7.6 | 27.3 ± 7.5 | 27.8 ± 7.3 | p = 0.195 | p = 0.463 |
| HDG | 24.6 ± 2.5 | 24.8 ± 2.8 | 23.8 ± 2.6 | 24.8 ± 2.7 | |||
| Muscle Mass (kg) | LDG | 69.0 ± 7.4 | 69.0 ± 7.2 | 69.0 ± 7.0 | 68.6 ± 6.9 | p = 0.260 | p = 0.468 |
| HDG | 71.7 ± 2.4 | 71.5 ± 2.7 | 72.4 ± 2.5 | 71.7 ± 2.5 | |||
| Trunk Fat (%) | LDG | 31.1 ± 7.5 | 31.3 ± 7.6 | 31.4 ± 7.4 | 30.0 ± 6.7 | p = 0.584 | p = 0.125 |
| HDG | 27.3 ± 3.2 | 27.5 ± 3.4 | 26.3 ± 3.1 | 27.2 ± 1.9 | |||
| Right Leg Girth (cm) | LDG | 61 ± 8.0 | 62 ± 6.0 | 65 ± 6.0 * | 64 ± 6 | p < 0.001 | p = 0.158 |
| HDG | 58 ± 2.0 | 60 ± 3.0 | 60 ± 3.0 | 60 ± 3.0 | |||
| Right Leg Muscle (%) | LDG | 11.5 ± 1.9 | 11.5 ± 1.9 | 11.6 ± 1.9 | 11.4 ± 1.9 | p = 0.540 | p = 0.137 |
| HDG | 10.8 ± 0.9 | 10.6 ± 0.9 | 10.6 ± 0.8 | 10.6 ± 1.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
O’Connor, D.; Malone, J.K. The Dose Response for Sprint Interval Training Interventions May Affect the Time Course of Aerobic Training Adaptations. Sports 2019, 7, 85. https://doi.org/10.3390/sports7040085
O’Connor D, Malone JK. The Dose Response for Sprint Interval Training Interventions May Affect the Time Course of Aerobic Training Adaptations. Sports. 2019; 7(4):85. https://doi.org/10.3390/sports7040085
Chicago/Turabian StyleO’Connor, Dominic, and John K. Malone. 2019. "The Dose Response for Sprint Interval Training Interventions May Affect the Time Course of Aerobic Training Adaptations" Sports 7, no. 4: 85. https://doi.org/10.3390/sports7040085
APA StyleO’Connor, D., & Malone, J. K. (2019). The Dose Response for Sprint Interval Training Interventions May Affect the Time Course of Aerobic Training Adaptations. Sports, 7(4), 85. https://doi.org/10.3390/sports7040085





