Reporting Characteristics in Sports Nutrition
Abstract
1. Introduction
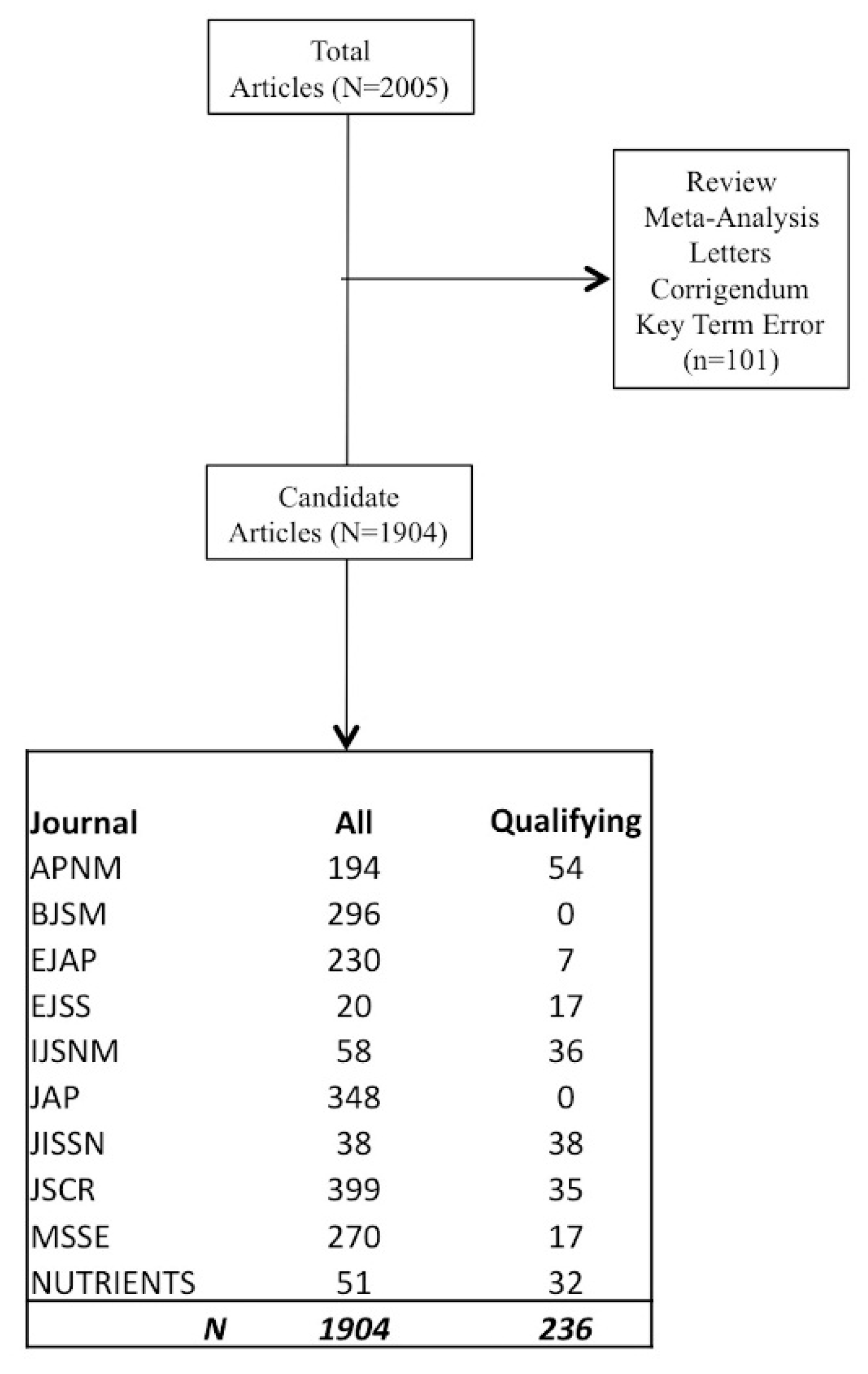
2. Methods
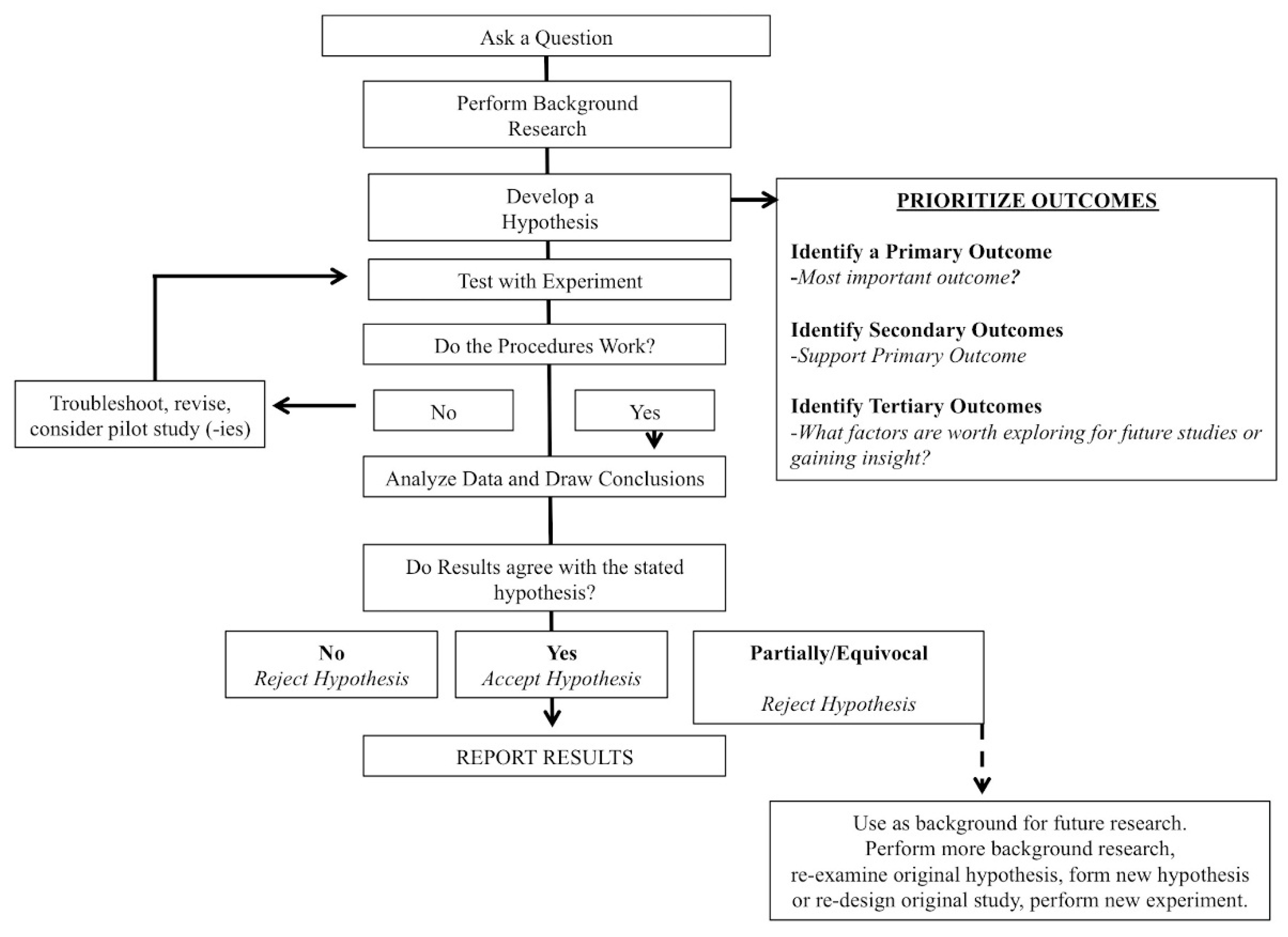
2.1. Primary Outcome
2.2. Secondary Outcomes
- (1)
- Did the authors report an effect size for their analysis? This was categorized as Yes or No.
- (2)
- Did the authors stratify or prioritize their analysis as a primary (i.e., most important), secondary, tertiary (exploratory), etc. analyses? This was categorized as Yes or No.
- (3)
- If reporting change scores (mean or percentage), did the authors use confidence intervals or did they report SD or SEM.
- (4)
- Did the authors state a directed, a priori hypothesis for their paper? Observations were categorized as: (a) directed hypotheses; (b) non-specific/general hypothesis or (c) no hypothesis stated.
3. Statistics
4. Results
5. Discussion
5.1. Primary Outcome
5.2. Secondary Outcomes
5.2.1. Effect Sizes
5.2.2. Hypotheses and Stratified Analyses
5.2.3. Change Scores
6. Limitations
7. Conclusions
Funding
Conflicts of Interest
References
- Altman, D.G.; Gore, S.M.; Gardner, M.J.; Pocock, S.J. Statistical guidelines for contributors to medical journals. Br. Med. J. 1983, 286, 1489–1493. [Google Scholar] [CrossRef]
- Gardner, M.J.; Altman, D.G.; Jones, D.R.; Machin, D. Is the statistical assessment of papers submitted to the “British Medical Journal” effective? Br. Med. J. 1983, 286, 1485–1488. [Google Scholar] [CrossRef]
- Gore, S.M.; Jones, G.; Thompson, S.G. The Lancet’s statistical review process: Areas for improvement by authors. Lancet 1992, 340, 100–102. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D.; Group, C. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. Trials 2010, 11, 32. [Google Scholar] [CrossRef] [PubMed]
- Barde, M.P.; Barde, P.J. What to use to express the variability of data: Standard deviation or standard error of mean? Perspect. Clin. Res. 2012, 3, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Nagele, P. Misuse of standard error of the mean (SEM) when reporting variability of a sample. A critical evaluation of four anaesthesia journals. Br. J. Anaesth. 2003, 90, 514–516. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. Sports Exerc. 2009, 41, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Weir, J.P. Quantifying test-retest reliability using the intraclass correlation coefficient and the SEM. J. Strength Cond. Res. 2005, 19, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Altman, D.G.; Bland, J.M. Standard deviations and standard errors. BMJ 2005, 331, 903. [Google Scholar] [CrossRef] [PubMed]
- Carlin, J.B.; Doyle, L.W. 3: Basic concepts of statistical reasoning: Standard errors and confidence intervals. J. Paediatr. Child Health 2000, 36, 502–505. [Google Scholar] [CrossRef] [PubMed]
- Webster, C.S.; Merry, A.F. The standard deviation and the standard error of the mean. Anaesthesia 1997, 52, 183. [Google Scholar] [PubMed]
- Hoffman, J.I. Standard error or standard deviation? Pediatr. Cardiol. 2015, 36, 1105–1106. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Hopewell, S.; Schulz, K.F.; Montori, V.; Gotzsche, P.C.; Devereaux, P.J.; Elbourne, D.; Egger, M.; Altman, D.G. CONSORT 2010 explanation and elaboration: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340, c869. [Google Scholar] [CrossRef] [PubMed]
- Ialongo, C. Understanding the effect size and its measures. Biochem. Med. 2016, 26, 150–163. [Google Scholar] [CrossRef] [PubMed]
- Concato, J.; Hartigan, J.A. P values: From suggestion to superstition. J. Investig. Med. 2016, 64, 1166–1171. [Google Scholar] [CrossRef] [PubMed]
- Wasserstein, R.L.; Lazar, N.A. The ASA’s Statement on p-Values: Context, Process, and Purpose. Am. Stat. 2016, 70, 129–133. [Google Scholar] [CrossRef]
- Ayala, F.J. Darwin and the scientific method. Proc. Natl. Acad. Sci. USA 2009, 106 (Suppl. S1), 10033–10039. [Google Scholar] [CrossRef]
- Supino, R. The Research Hypothesis: Role and Construction. In Principles of Research Methodology: A Guide for Clinical Investigators; Supino, P., Borer, J., Eds.; Springer: New York, NY, USA, 2012; pp. 15–30. [Google Scholar]
- Farrugia, P.; Petrisor, B.A.; Farrokhyar, F.; Bhandari, M. Practical tips for surgical research: Research questions, hypotheses and objectives. Can. J. Surg. 2010, 53, 278–281. [Google Scholar] [PubMed]
- Clark, G.T.; Mulligan, R. Fifteen common mistakes encountered in clinical research. J. Prosthodont. Res. 2011, 55, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Freemantle, N. Interpreting the results of secondary end points and subgroup analyses in clinical trials: Should we lock the crazy aunt in the attic? BMJ 2001, 322, 989–991. [Google Scholar] [CrossRef] [PubMed]
- Andrade, C. The primary outcome measure and its importance in clinical trials. J. Clin. Psychiatry 2015, 76, e1320–e1323. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.W.; Keil, L.G.; Holland, W.C.; Caughey, M.C.; Platts-Mills, T.F. Comparison of registered and published outcomes in randomized controlled trials: A systematic review. BMC Med. 2015, 13, 282. [Google Scholar] [CrossRef] [PubMed]
- Curran-Everett, D.; Williams, C.L. Explorations in statistics: The analysis of change. Adv. Physiol. Educ. 2015, 39, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, G.; Batterham, A.M. The use of ratios and percentage changes in sports medicine: Time for a rethink? Int. J. Sports Med. 2012, 33, 505–506. [Google Scholar] [CrossRef] [PubMed]
- Vickers, A.J. The use of percentage change from baseline as an outcome in a controlled trial is statistically inefficient: A simulation study. BMC Med. Res. Methodol. 2001, 1, 6. [Google Scholar] [CrossRef]
- Fu, R.; Holmer, H.K. Change Score or Followup Score? An Empirical Evaluation of the Impact of Choice of Mean Difference Estimates, Agency for Healthcare Research and Quality (US); 2015 Apr. Report No.: 15-EHC016-EF. AHRQ Methods for Effective Health Care; Agency for Healthcare Research and Quality (USA): Rockville, MD, USA, 2015.
- Batterham, A.M.; Hopkins, W.G. The case for magnitude-based inference. Med. Sci. Sports Exerc. 2015, 47, 885. [Google Scholar] [CrossRef] [PubMed]
- Welsh, A.H.; Knight, E.J. “Magnitude-based inference”: A statistical review. Med. Sci. Sports Exerc. 2015, 47, 874–884. [Google Scholar] [CrossRef] [PubMed]
- Young, A.J. Magnitude-based inference: Progressive approach or flawed statistic? Med. Sci. Sports Exerc. 2015, 47, 873. [Google Scholar] [CrossRef] [PubMed]
- Pocock, S.J.; Stone, G.W. The Nature of the P Value. N. Engl. J. Med. 2016, 375, 2205–2206. [Google Scholar] [CrossRef] [PubMed]
- Vila, P.M.; Townsend, M.E.; Bhatt, N.K.; Kao, W.K.; Sinha, P.; Neely, J.G. The P Value Problem in Otolaryngology: Shifting to Effect Sizes and Confidence Intervals. Otolaryngol. Head Neck Surg. 2017, 156, 978–980. [Google Scholar] [CrossRef] [PubMed]
- Laber, E.B.; Shedden, K. Statistical Significance and the Dichotomization of Evidence: The Relevance of the ASA Statement on Statistical Significance and p-values for Statisticians. J. Am. Stat. Assoc. 2017, 112, 902–904. [Google Scholar] [CrossRef] [PubMed]
- Cooper, R.J.; Schriger, D.L.; Close, R.J. Graphical literacy: The quality of graphs in a large-circulation journal. Ann. Emerg. Med. 2002, 40, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Altman, D.G. Statistical reviewing for medical journals. Stat. Med. 1998, 17, 2661–2674. [Google Scholar] [CrossRef]
- Goodman, S.N.; Altman, D.G.; George, S.L. Statistical reviewing policies of medical journals: Caveat lector? J. Gen. Intern. Med. 1998, 13, 753–756. [Google Scholar] [CrossRef] [PubMed]

| Variables | Outcomes | Response (n) | Percent |
|---|---|---|---|
| Reporting of Treatment Variance | SD | 129 | 59% |
| SEM | 79 | 36% | |
| CI | 11 | 5% | |
| Effect Size Reporting | None Reported | 155 | 71% |
| Reported | 63 | 29% | |
| Outcome Stratification | No | 188 | 86% |
| Yes | 31 | 14% | |
| Directed Hypothesis | None Reported | 134 | 61% |
| Specific/Directed | 57 | 26% | |
| Non-Specific | 28 | 13% | |
| Change Score Variance (n = 99) | SD | 41 | 19% |
| SEM | 10 | 5% | |
| CI | 40 | 18% | |
| None Reported | 8 | 4% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Earnest, C.P.; Roberts, B.M.; Harnish, C.R.; Kutz, J.L.; Cholewa, J.M.; Johannsen, N.M. Reporting Characteristics in Sports Nutrition. Sports 2018, 6, 139. https://doi.org/10.3390/sports6040139
Earnest CP, Roberts BM, Harnish CR, Kutz JL, Cholewa JM, Johannsen NM. Reporting Characteristics in Sports Nutrition. Sports. 2018; 6(4):139. https://doi.org/10.3390/sports6040139
Chicago/Turabian StyleEarnest, Conrad P., Brandon M. Roberts, Christopher R. Harnish, Jessica L. Kutz, Jason M. Cholewa, and Neil M. Johannsen. 2018. "Reporting Characteristics in Sports Nutrition" Sports 6, no. 4: 139. https://doi.org/10.3390/sports6040139
APA StyleEarnest, C. P., Roberts, B. M., Harnish, C. R., Kutz, J. L., Cholewa, J. M., & Johannsen, N. M. (2018). Reporting Characteristics in Sports Nutrition. Sports, 6(4), 139. https://doi.org/10.3390/sports6040139






