Pharmacological Inhibition of Inward Rectifier Potassium Channels Induces Lethality in Larval Aedes aegypti
Abstract
1. Introduction
2. Materials and Methods
2.1. Mosquito Colony
2.2. Chemicals
2.3. Larval Toxicity Bioassays
2.4. Statistics
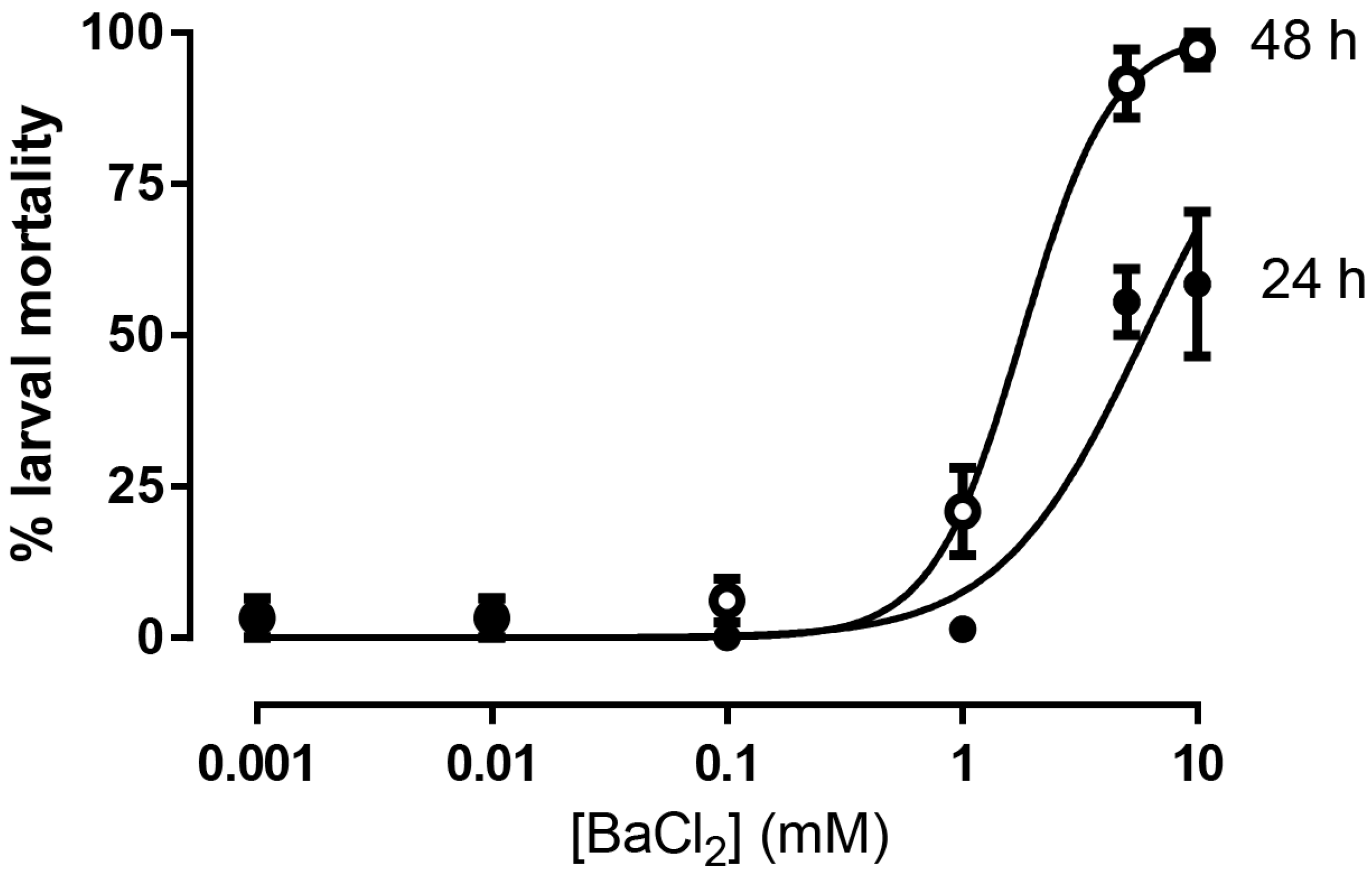
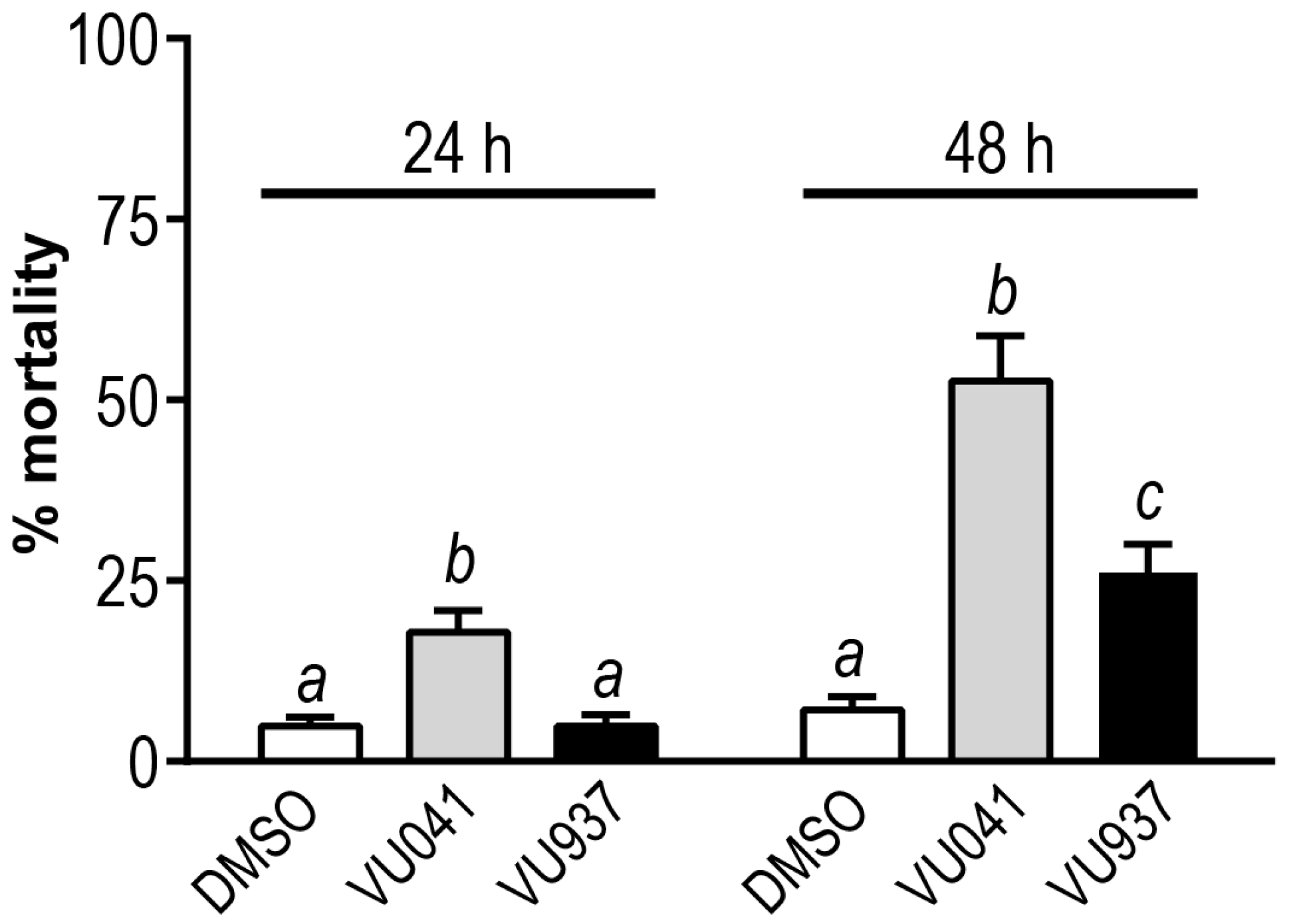
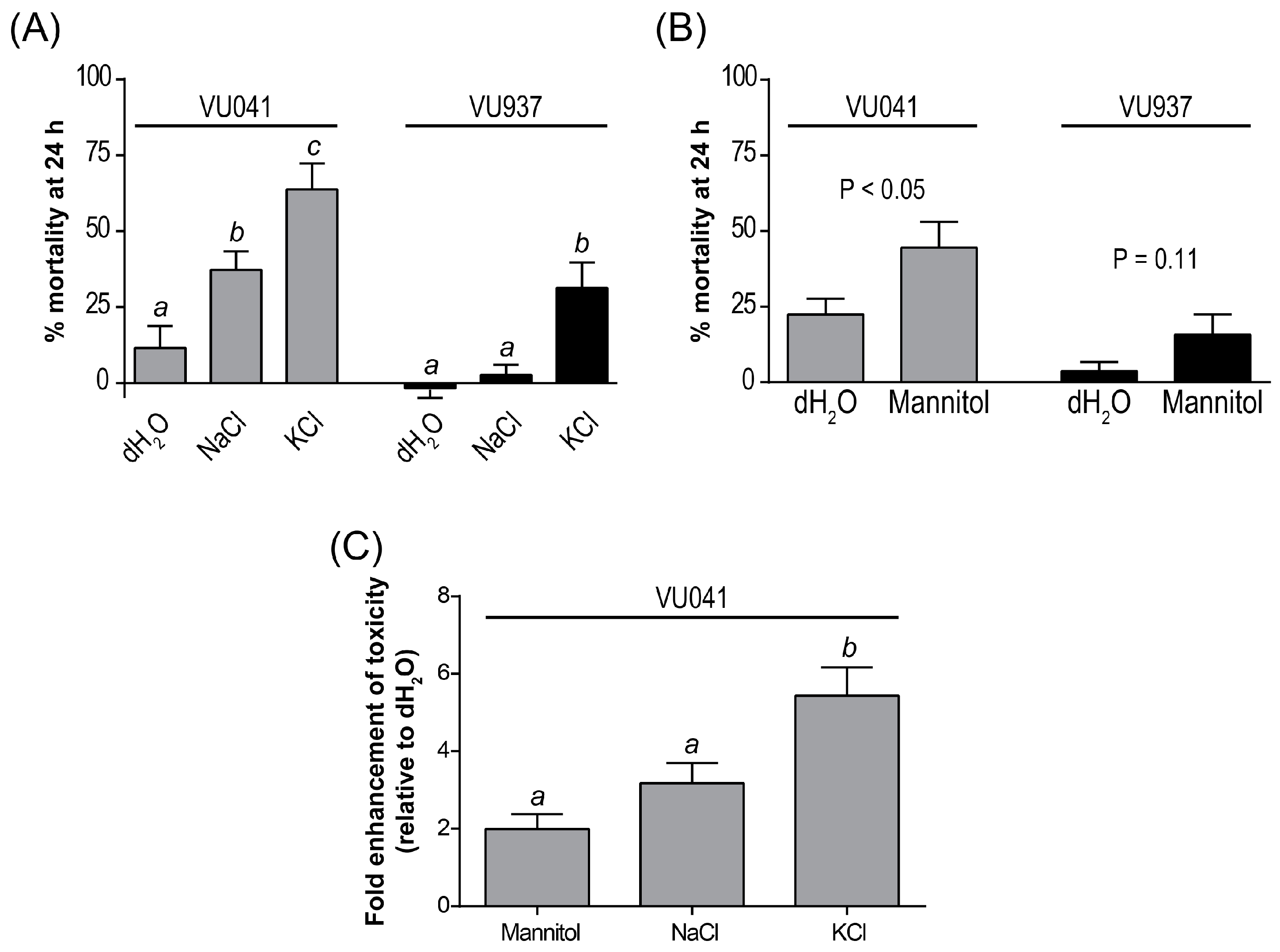
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. A Global Brief on Vector-Borne Diseases; Organization, W.H., Ed.; World Health Organization: Geneva, Switzerland, 2014; p. 54. [Google Scholar]
- Kupferschmidt, K. Pick your poison. Science 2016, 354, 171–173. [Google Scholar] [CrossRef] [PubMed]
- Paine, M.J.I.; Brooke, B. Insecticide resistance and its impact on vector control. In Advances in Insect Control and Resistance Management; Horowitz, A.R., Ishaaya, I., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 287–312. [Google Scholar]
- Beyenbach, K.W.; Yu, Y.; Piermarini, P.M.; Denton, J. Targeting renal epithelial channels for the control of insect vectors. Tissue Barriers 2015, 3, e1081861. [Google Scholar] [CrossRef] [PubMed]
- Piermarini, P.M.; Esquivel, C.J.; Denton, J.S. Malpighian Tubules as novel targets for mosquito control. Int. J. Environ. Res. Public Health 2017, 14, 111. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.M.; Allan, A.K.; Davies, S.A.; Dow, J.A. Sulphonylurea sensitivity and enriched expression implicate inward rectifier K+ channels in Drosophila melanogaster renal function. J. Exp. Biol. 2005, 208, 3771–3783. [Google Scholar] [CrossRef] [PubMed]
- Piermarini, P.M.; Rouhier, M.F.; Schepel, M.; Kosse, C.; Beyenbach, K.W. Cloning and functional characterization of inward-rectifying potassium (Kir) channels from Malpighian tubules of the mosquito Aedes aegypti. Insect Biochem. Mol. Biol. 2013, 43, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Raphemot, R.; Rouhier, M.F.; Hopkins, C.R.; Gogliotti, R.D.; Lovell, K.M.; Hine, R.M.; Ghosalkar, D.; Longo, A.; Beyenbach, K.W.; Denton, J.S.; et al. Eliciting renal failure in mosquitoes with a small-molecule inhibitor of inward-rectifying potassium channels. PLoS ONE 2013, 8, e64905. [Google Scholar] [CrossRef] [PubMed]
- Raphemot, R.; Estevez-Lao, T.Y.; Rouhier, M.F.; Piermarini, P.M.; Denton, J.S.; Hillyer, J.F. Molecular and functional characterization of Anopheles gambiae inward rectifier potassium (Kir1) channels: A novel role in egg production. Insect Biochem. Mol. Biol. 2014, 51C, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Rouhier, M.F.; Hine, R.M.; Park, S.T.; Raphemot, R.; Denton, J.S.; Piermarini, P.M.; Beyenbach, K.W. The excretion of NaCl and KCl loads in mosquitoes: 2. Effects of the small molecule Kir channel modulator VU573 and its inactive analog VU342. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 307, R850–R861. [Google Scholar] [CrossRef] [PubMed]
- Piermarini, P.M.; Dunemann, S.M.; Rouhier, M.F.; Calkins, T.L.; Raphemot, R.; Denton, J.S.; Hine, R.M.; Beyenbach, K.W. Localization and role of inward rectifier K channels in Malpighian tubules of the yellow fever mosquito Aedes aegypti. Insect Biochem. Mol. Biol. 2015, 67, 59–73. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Baum, M.; Huang, C.-L.; Rodan, A.R. Two inwardly rectifying potassium channels, Irk1 and Irk2, play redundant roles in Drosophila renal tubule function. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 309, R747–R756. [Google Scholar] [CrossRef] [PubMed]
- Swale, D.R.; Engers, D.W.; Bollinger, S.R.; Gross, A.; Inocente, E.A.; Days, E.; Kanga, F.; Johnson, R.M.; Yang, L.; Bloomquist, J.R.; et al. An insecticide resistance-breaking mosquitocide targeting inward rectifier potassium channels in vectors of Zika virus and malaria. Sci. Rep. 2016, 6, 36954. [Google Scholar] [CrossRef] [PubMed]
- O’Neal, S.T.; Swale, D.R.; Anderson, T.D. ATP-sensitive inwardly rectifying potassium channel regulation of viral infections in honey bees. Sci. Rep. 2017, 7, 8668. [Google Scholar] [CrossRef] [PubMed]
- O’Neal, S.T.; Swale, D.R.; Bloomquist, J.R.; Anderson, T.D. ATP-sensitive inwardly rectifying potassium channel modulators alter cardiac function in honey bees. J. Insect Physiol. 2017, 99, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Swale, D.R.; Li, Z.; Guerrero, F.; Perez De Leon, A.A.; Foil, L.D. Role of inward rectifier potassium channels in salivary gland function and sugar feeding of the fruit fly, Drosophila melanogaster. Pestic. Biochem. Physiol. 2017, 141, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Swale, D.R. Inwardly rectifying potassium (Kir) channels represent a critical ion conductance pathway in the nervous systems of insects. Sci. Rep. 2018, 8, 1617. [Google Scholar] [CrossRef] [PubMed]
- Kolosov, D.; Tauqir, M.; Rajaruban, S.; Piermarini, P.M.; Donini, A.; O’Donnell, M.J. Molecular mechanisms of bi-directional ion transport in the Malpighian tubules of a lepidopteran crop pest, Trichoplusia ni. J. Insect Physiol. 2018, 109, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Ren, M.; Niu, J.; Hu, B.; Wei, Q.; Zheng, C.; Tian, X.; Gao, C.; He, B.; Dong, K.; Su, J. Block of Kir channels by flonicamid disrupts salivary and renal excretion of insect pests. Insect Biochem. Mol. Biol. 2018, 99, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Piermarini, P.M.; Inocente, E.A.; Acosta, N.; Hopkins, C.R.; Denton, J.S.; Michel, A.P. Inward rectifier potassium (Kir) channels in the soybean aphid Aphis glycines: Functional characterization, pharmacology, and toxicology. J. Insect Physiol. 2018, 110, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Statler, B.-M.; Calkins, T.L.; Alfaro, E.; Esquivel, C.J.; Rouhier, M.F.; Denton, J.S.; Piermarini, P.M. Dynamic expression of genes encoding subunits of inward rectifier potassium (Kir) channels in the yellow fever mosquito Aedes aegypti. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2017, 204, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Eleftherianos, I.; Won, S.; Chtarbanova, S.; Squiban, B.; Ocorr, K.; Bodmer, R.; Beutler, B.; Hoffmann, J.A.; Imler, J.L. ATP-sensitive potassium channel (KATP)-dependent regulation of cardiotropic viral infections. Proc. Natl. Acad. Sci. USA 2011, 108, 12024–12029. [Google Scholar] [CrossRef] [PubMed]
- Dahal, G.R.; Rawson, J.; Gassaway, B.; Kwok, B.; Tong, Y.; Ptacek, L.J.; Bates, E. An inwardly rectifying K+ channel is required for patterning. Development 2012, 139, 3653–3664. [Google Scholar] [CrossRef] [PubMed]
- Raphemot, R.; Rouhier, M.F.; Swale, D.R.; Days, E.; Weaver, C.D.; Lovell, K.M.; Konkel, L.C.; Engers, D.W.; Bollinger, S.F.; Hopkins, C.; et al. Discovery and characterization of a potent and selective inhibitor of Aedes aegypti inward rectifier potassium channels. PLoS ONE 2014, 9, e110772. [Google Scholar] [CrossRef] [PubMed]
- Rouhier, M.F.; Raphemot, R.; Denton, J.S.; Piermarini, P.M. Pharmacological validation of an inward-rectifier potassium (Kir) channel as an insecticide target in the yellow fever mosquito Aedes aegypti. PLoS ONE 2014, 9, e100700. [Google Scholar] [CrossRef] [PubMed]
- Swale, D.R.; Li, Z.; Kraft, J.Z.; Healy, K.; Liu, M.; David, C.M.; Liu, Z.; Foil, L.D. Development of an autodissemination strategy for the deployment of novel control agents targeting the common malaria mosquito, Anopheles quadrimaculatus say (Diptera: Culicidae). PLoS Negl. Trop. Dis. 2018, 12, e0006259. [Google Scholar] [CrossRef] [PubMed]
- Taylor-Wells, J.; Gross, A.D.; Jiang, S.; Demares, F.; Clements, J.S.; Carlier, P.R.; Bloomquist, J.R. Toxicity, mode of action, and synergist potential of flonicamid against mosquitoes. Pestic. Biochem. Physiol. 2018. [Google Scholar] [CrossRef]
- Inocente, E.A.; Shaya, M.; Acosta, N.; Rakotondraibe, L.H.; Piermarini, P.M. A natural agonist of mosquito TRPA1 from the medicinal plant Cinnamosma fragrans that is toxic, antifeedant, and repellent to the yellow fever mosquito Aedes aegypti. PLoS Negl. Trop. Dis. 2018, 12, e0006265. [Google Scholar] [CrossRef] [PubMed]
- Pridgeon, J.W.; Becnel, J.J.; Clark, G.G.; Linthicum, K.J. A high-throughput screening method to identify potential pesticides for mosquito control. J. Med. Entomol. 2009, 46, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Calkins, T.L.; Piermarini, P.M. Pharmacological and genetic evidence for gap junctions as potential new insecticide targets in the yellow fever mosquito, Aedes aegypti. PLoS ONE 2015, 10, e0137084. [Google Scholar] [CrossRef] [PubMed]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Bradley, T.J. Physiology of osmoregulation in mosquitoes. Annu. Rev. Entomol. 1987, 32, 439–462. [Google Scholar] [CrossRef] [PubMed]
- Beyenbach, K.W. The plasticity of extracellular fluid homeostasis in insects. J. Exp. Biol. 2016, 219, 2596–2607. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rusconi Trigueros, R.; Hopkins, C.R.; Denton, J.S.; Piermarini, P.M. Pharmacological Inhibition of Inward Rectifier Potassium Channels Induces Lethality in Larval Aedes aegypti. Insects 2018, 9, 163. https://doi.org/10.3390/insects9040163
Rusconi Trigueros R, Hopkins CR, Denton JS, Piermarini PM. Pharmacological Inhibition of Inward Rectifier Potassium Channels Induces Lethality in Larval Aedes aegypti. Insects. 2018; 9(4):163. https://doi.org/10.3390/insects9040163
Chicago/Turabian StyleRusconi Trigueros, Renata, Corey R. Hopkins, Jerod S. Denton, and Peter M. Piermarini. 2018. "Pharmacological Inhibition of Inward Rectifier Potassium Channels Induces Lethality in Larval Aedes aegypti" Insects 9, no. 4: 163. https://doi.org/10.3390/insects9040163
APA StyleRusconi Trigueros, R., Hopkins, C. R., Denton, J. S., & Piermarini, P. M. (2018). Pharmacological Inhibition of Inward Rectifier Potassium Channels Induces Lethality in Larval Aedes aegypti. Insects, 9(4), 163. https://doi.org/10.3390/insects9040163





