Chemical and Non-Chemical Options for Managing Twospotted Spider Mite, Western Tarnished Plant Bug and Other Arthropod Pests in Strawberries
Abstract
:1. Introduction
2. Materials and Methods
2.1. Twospotted Spider Mite
2.2. Western Tarnished Plant Bug and Other Insect Pests
3. Results
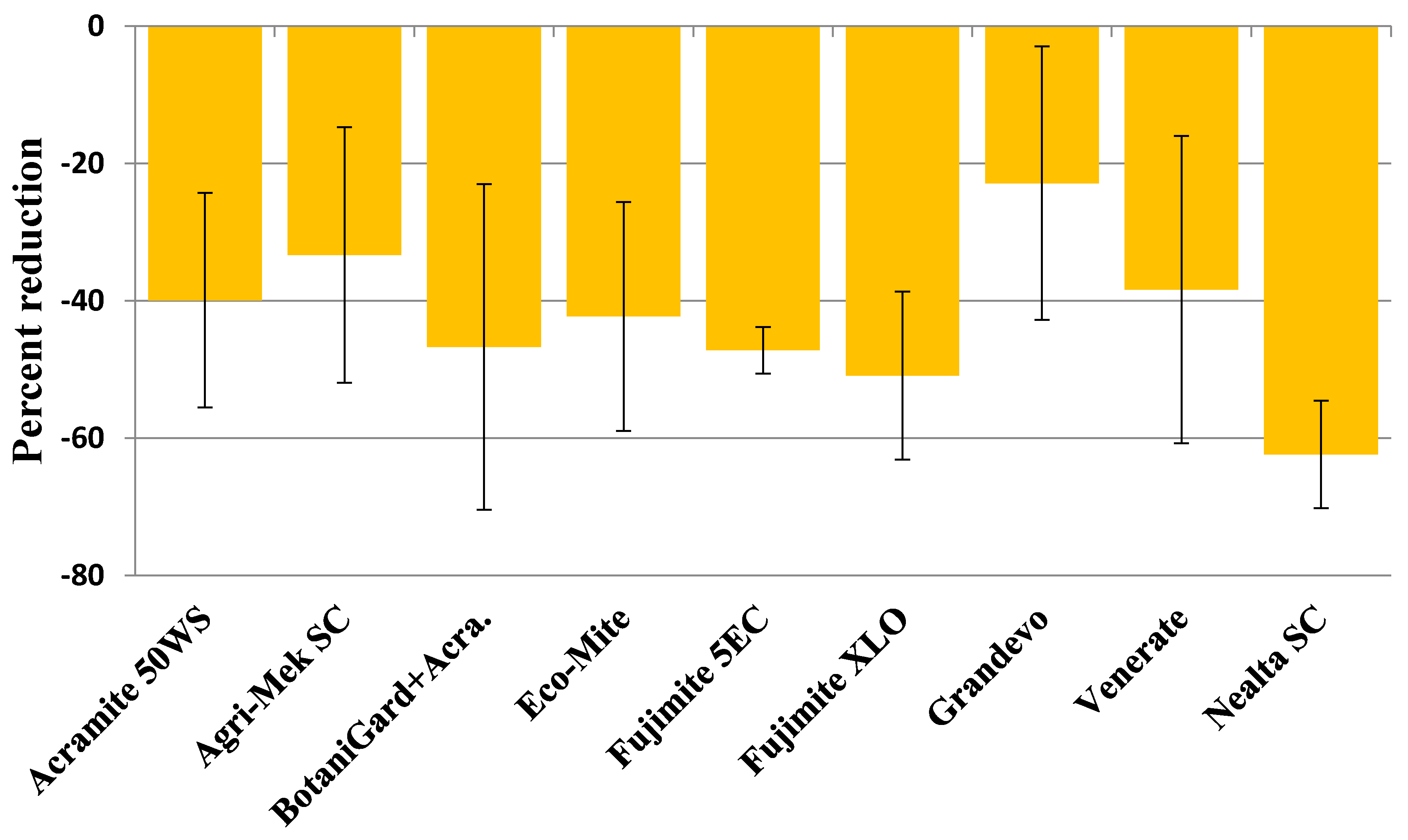
3.1. Twospotted Spider Mite
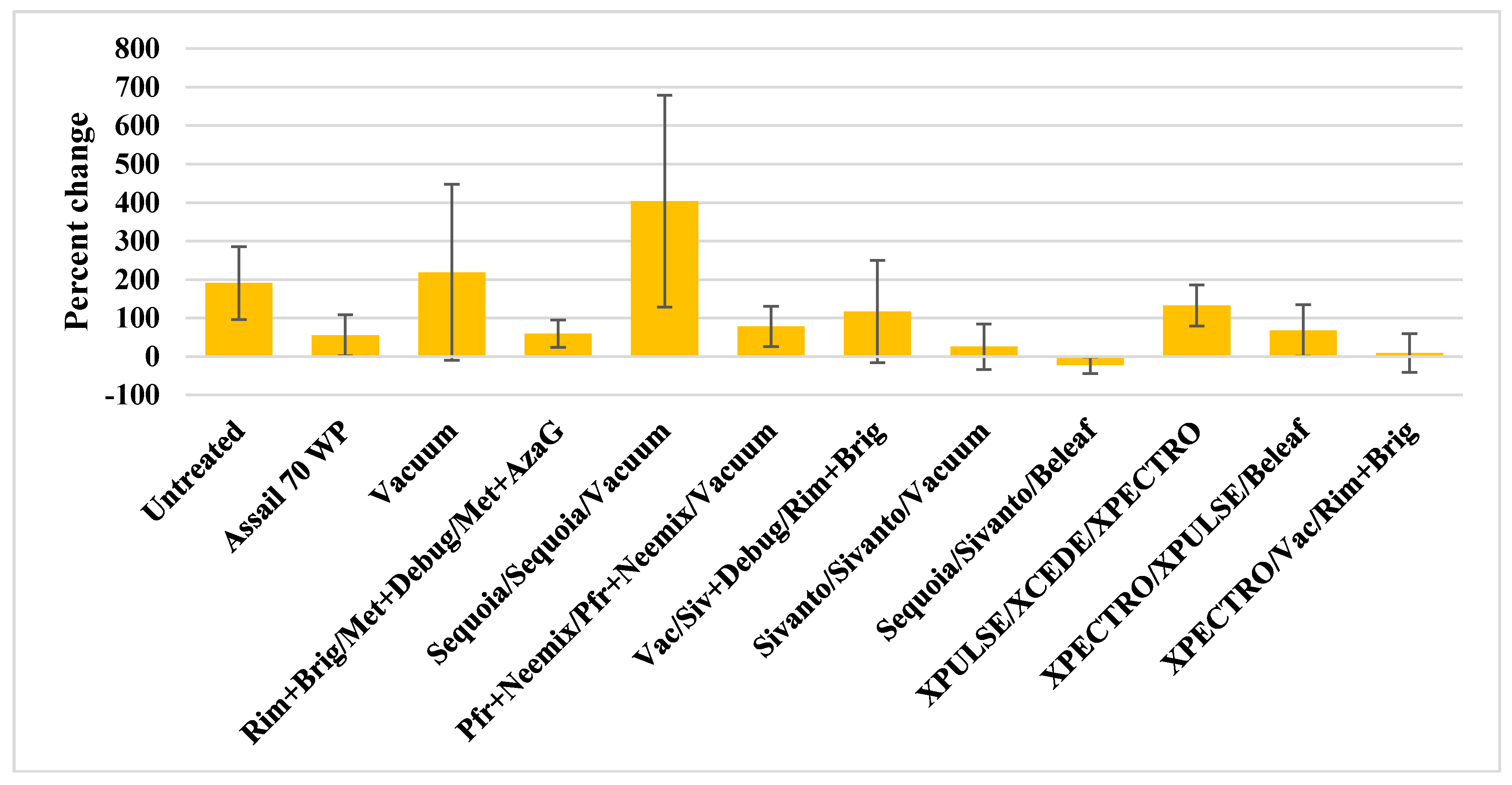
3.2. Western Tarnished Plant Bug
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- California Department of Food and Agriculture (CDFA). California Agricultural Statistics Review, 2016–2017. 2018. Available online: https://www.cdfa.ca.gov/Statistics/PDFs/2016-17AgReport.pdf (accessed on 25 September 2018).
- Zalom, F.G.; Bolda, M.P.; Dara, S.K.; Joseph, S. UC IPM Pest Management Guidelines: Strawberry. University of California Statewide Integrated Pest Management Program; UC ANR Publication: Davis, CA, USA, 2014; p. 3468. [Google Scholar]
- Dara, S.K. Virus decline of strawberry in California and the role of insect vectors and associated viruses. Plant Health Prog. 2015, 16, 211–215. [Google Scholar] [CrossRef]
- Vincent, C.; Lachance, P. Evaluation of a tractor-propelled vacuum device for management of tarnished plant bug (Heteroptera: Miridae) populations in strawberry plantations. Environ. Entomol. 1993, 22, 1103–1107. [Google Scholar] [CrossRef]
- Pickel, C.; Zalom, F.G.; Walsh, D.B.; Welch, N.C. Vacuums provide limited lygus control in strawberries. Calif. Agric. 1995, 49, 19–22. [Google Scholar] [CrossRef]
- Joseph, S.V.; Bolda, M. Evaluating the potential utility of an electrostatic sprayer and a tractor-mounted vacuum machine for Lygus hesperus (Hemiptera: Miridae) management in California’s coastal strawberry. Crop Prot. 2018, 113, 104–111. [Google Scholar] [CrossRef]
- California Department of Pesticide Regulation (CDPR). Summary of Pesticide Use Report Data 2013: Indexed by Commodity. 2015. Available online: http://www.cdpr.ca.gov/docs/pur/pur13rep/comrpt13.pdf (accessed on 25 September 2018).
- Çağatay, N.S.; Menault, P.; Riga, M.; Vontas, J.; Ay, R. Identification and characterization of Abamectin resistance in Tetranychus urticae Koch populations from greenhouses in Turkey. Crop Prot. 2018, 112, 112–117. [Google Scholar] [CrossRef]
- Parys, K.A.; Luttrell, R.G.; Snodgrass, G.L.; Portilla, M.R. Patterns of tarnished plant bug (Hemiptera: Miridae) resistance to pyrethroid insecticides in the Lower Mississippi Delta for 2008–2015: Linkage to pyrethroid use and cotton insect management. J. Insect Sci. 2018, 18, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; He, Y.; Zhang, Y.; Xie, W.; Wu, Q.; Wang, S. Status of pesticide resistance and associated mutations in the two-spotted spider mite, Tetranychus urticae, in China. Pest. Biochem. Physiol. 2018, 150, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Dara, S.K. Managing strawberry pests with chemical pesticides and non-chemical alternatives. Int. J. Fruit Sci. 2016, 16, 129–141. [Google Scholar] [CrossRef]
- Bitsadze, N.; Jaronski, S.; Khasdan, V.; Abashidze, E.; Abashidze, M.; Latchininsky, A.; Samadashvili, D.; Sokhadze, I.; Rippa, M.; Ishaaya, I.; et al. Joint action of Beauveria bassiana and the insect growth regulators diflubenzuron and novaluron, on the migratory locust, Locusta migratoria. J. Pest Sci. 2013, 86, 293–300. [Google Scholar] [CrossRef]
- Dara, S.K.; Dara, S.R.; Dara, S.S. Endophytic colonization and pest management potential of Beauveria bassiana in strawberries. J. Berry Res. 2013, 3, 203–211. [Google Scholar]
- Dara, S.K. Root aphids and their management in organic celery. CAPCA Advis. 2015, 18, 56–58. [Google Scholar]
- Tomilova, O.G.; Kryukov, V.Y.; Duisembekov, B.A.; Yaroslavtseva, O.N.; Tyurin, M.V.; Kryukova, N.A.; Skorokhod, V.; Dubovskiy, I.M.; Glupov, V.V. Immune-physiological aspects of synergy between avermectins and the entomopathogenic fungus Metarhizium robertsii in Colorado potato beetle larvae. J. Invertebr. Pathol. 2016, 140, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Tabashnik, B.E. Managing resistance with multiple pesticide tactics: Theory, evidence, and recommendations. J. Econ. Entomol. 1989, 82, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- Attique, M.N.R.; Khaliq, A.; Sayyed, A.H. Could resistance to insecticides in Plutella xylostella (Lep., Plutellidae) be overcome by insecticide mixtures? J. Appl. Entomol. 2006, 130, 122–127. [Google Scholar] [CrossRef]
- Cloyd, R.A. Pesticide mixtures and rotations: Are these viable resistance mitigating strategies. Pest Technol. 2010, 4, 14–18. [Google Scholar]
- Sudo, M.; Takahashi, D.; Andow, D.A.; Suzuki, Y.; Yamanaka, T. Optimal management strategy of insecticide resistance under various insect life histories: Heterogeneous timing of selection and interpatch dispersal. Evol. Appl. 2018, 11, 271–283. [Google Scholar] [CrossRef] [PubMed]
- Sasan, R.K.; Bidochka, M.J. The insect-pathogenic fungus Metarhizium robertsii (Clavicipitaceae) is also an endophyte that stimulates plant root development. Am. J. Bot. 2012, 99, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Dara, S.K. Entomopathogenic Fungus Beauveria bassiana Promotes Strawberry Plant Growth and Health. UC ANR eJournal Strawberries and Vegetables. 30 September 2013. Available online: https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624 (accessed on 1 September 2018).
- Dara, S.K.; Dara, S.S.R.; Dara, S.S. Impact of entomopathogenic fungi on the growth, development, and health of cabbage growing under water stress. Amer. J. Plant Sci. 2017, 8, 1224–1233. [Google Scholar] [CrossRef]
- Dara, S.S.R.; Dara, S.S.; Dara, S.K.; Anderson, T. Fighting plant pathogenic fungi with entomopathogenic fungi and other biologicals. CAPCA Advis. 2017, 20, 40–44. [Google Scholar]
- Shi, W.-B.; Feng, M.-G. Field efficacy of application of Beauveria bassiana formulation and low rate pyridaben for sustainable control of citrus red mite Panonychus citri (Acari: Tetranychidae) in orchards. Biol. Control 2006, 39, 210–217. [Google Scholar] [CrossRef]
- Hiromori, H.; Nishigaki, J. Joint action of an entomopathogenic fungus (Metarhizium anisopliae) with synthetic insecticides against the scarab beetle, Anomala cuprea (Coleoptera: Scarabaeidae) larvae. Appl. Entomol. Zool. 1998, 33, 77–84. [Google Scholar] [CrossRef]
| Treatment | 1st Application (Rate/ha) | 2nd Application (Rate/ha) | 3rd Application (Rate/ha) |
|---|---|---|---|
| 1 | Untreated | Untreated | Untreated |
| 2 | Assail 70 WP (210 g) | Assail 70 WP (210 g) | Assail 70 WP (210 g) |
| 3 | Vacuum | Vacuum | Vacuum |
| 4 | Rimon 0.83 EC (877 mL) + Brigade (1.1 kg) | Met52 EC (16 fl oz) + Debug Turbo (7.6 L) | Met52 EC (1.2 L) + AzaGuard (1.2 L) |
| 5 | Sequoia (329 mL) | Sequoia (329 mL) | Vacuum |
| 6 | Pfr-97 20% WDG (2.2 kg) + Neemix (658 mL) | Pfr-97 20% WDG (2.2 kg) + Neemix (658 mL) | Vacuum |
| 7 | Vacuum | Sivanto (1 L) + Debug Turbo (7.6 L) | Rimon 0.83 EC (877 mL) + Brigade (1.1 kg) |
| 8 | Sivanto (1 L) | Sivanto (1 L) | Vacuum |
| 9 | Sequoia (329 mL) | Sivanto (1 L) | Beleaf 50 SG (196 g) |
| 10 | XPULSE (2.3 L) | XCEDE (2.3 L) | XPECTRO (2.3 L) |
| 11 | XPECTRO (2.3 L) | XPULSE (2.3 L) | Beleaf 50 SG (196 g) |
| 12 | XPECTRO (2.3 L) | Vacuum | Rimon 0.83 EC (877 mL) + Brigade (1.1 kg) |
| Treatment | I Spray 3DAT | I Spray 7DAT | II Spray 3DAT | II Spray 7DAT | ||||
|---|---|---|---|---|---|---|---|---|
| Egg | Mobile | Egg | Mobile | Egg | Mobile | Egg | Mobile | |
| Untreated | 109.05 ± 2.73 | 23.25 ± 1.72 | 111.60 ± 15.66 | 20.40 ± 0.52 | 133.00 ± 9.61 | 22.20 ± 1.91 | 85.00 ± 5.97 | 23.8 ± 1.51 |
| Acramite 50WS | 87.60 ± 25.16 | 15.6 ± 7.42 | 75.80 ± 23.08 | 7.80 ± 2.22 | 94.00 ± 33.76 | 14.00 ± 1.97 | 57.80 ± 18.24 | 10.80 ± 3.20 |
| Agri-Mek SC | 72.45 ± 15.26 | 10.80 ± 1.78 | 69.20 ± 17.49 | 12.80 ± 3.21 | 84.20 ± 15.31 | 19.40 ± 3.58 | 56.40 ± 19.50 | 19.60 ± 3.99 |
| BotaniGard ES+Acramite 50WS | 90.90 ± 17.67 | 12.15 ± 2.08 | 79.00 ± 14.81 | 8.20 ± 1.14 | 86.40 ± 15.31 | 15.80 ± 2.49 | 32.40 ± 6.74 | 20.40 ± 12.45 |
| Eco-Mite | 42.45 ± 15.53 | 5.25 ± 2.59 | 66.80 ± 4.99 | 11.40 ± 3.01 | 69.00 ± 7.15 | 10.80 ± 2.13 | 48.20 ± 19.36 | 14.00 ± 5.25 |
| Fujimite 5 EC | 65.10 ± 19.32 | 8.85 ± 2.79 | 68.80 ± 14.77 | 8.00 ± 1.34 | 62.80 ± 3.90 | 13.40 ± 3.15 | 45.80 ± 6.42 | 12.20 ± 1.28 |
| Fujimite XLO | 71.25 ± 18.93 | 19.50 ± 8.78 | 85.00 ± 28.52 | 11.60 ± 1.20 | 91.20 ± 15.40 | 15.00 ± 4.52 | 43.40 ± 15.78 | 10.40 ± 1.08 |
| Grandevo | 75.60 ± 10.22 | 14.40 ± 2.86 | 85.80 ± 28.15 | 12.40 ± 2.58 | 111.20 ± 15.87 | 14.80 ± 3.78 | 64.60 ± 15.69 | 16.20 ± 4.36 |
| Venerate XC | 45.60 ± 10.77 | 16.35 ± 2.86 | 54.20 ± 18.15 | 16.00 ± 5.65 | 65.60 ± 25.57 | 12.80 ± 5.66 | 48.20 ± 14.52 | 14.60 ± 5.19 |
| Nealta SC | 66.00 ± 22.99 | 18.30 ± 9.76 | 51.00 ± 11.34 | 8.80 ± 2.4 | 82.60 ± 8.95 | 8.40 ± 2.02 | 31.40 ± 7.46 | 10.40 ± 3.33 |
| p value | 0.2310 | 0.1702 | 0.4023 | 0.0490 | 0.1627 | 0.2441 | 0.2049 | 0.5918 |
| I Spray 3DAT | I Spray 7DAT | II Spray 3DAT | II Spray 7DAT | |||||
|---|---|---|---|---|---|---|---|---|
| Egg | Mobile | Egg | Mobile | Egg | Mobile | Egg | Mobile | |
| Untreated | 0.00 | 1.65 ± 1.05 | 1.00 ± 0.38 | 0.60 ± 0.20 b * | 1.80 ± 0.76 | 2.20 ± 0.68 | 2.60 ± 1.00 | 4.20 ± 1.64 |
| Acramite 50WS | 0.45 ± 0.28 | 0.60 ± 0.24 | 1.00 ± 0.60 | 0.60 ± 0.38 b | 1.00 ± 0.50 | 1.20 ± 0.40 | 1.00 ± 0.38 | 1.20 ± 0.51 |
| Agri-Mek SC | 1.20 ± 0.24 | 0.45 ± 0.28 | 1.00 ± 0.60 | 0.20 ± 0.20 b | 0.60 ± 0.20 | 1.40 ± 0.68 | 0.40 ± 0.23 | 2.60 ± 1.00 |
| BotaniGard ES+Acramite 50WS | 0.45 ± 0.28 | 0.60 ± 0.34 | 0.80 ± 0.46 | 1.60 ± 0.92 ab | 2.00 ± 1.20 | 2.60 ± 2.34 | 1.60 ± 0.65 | 4.20 ± 2.29 |
| Eco-Mite | 0.30 ± 0.17 | 0.75 ± 0.56 | 2.20 ± 0.88 | 0.60 ± 0.60 b | 0.80 ± 0.46 | 1.60 ± 0.56 | 2.00 ± 0.40 | 3.00 ± 0.88 |
| Fujimite 5 EC | 0.60 ± 0.34 | 0.45 ± 0.28 | 1.60 ± 0.86 | 4.00 ± 1.34 a | 1.00 ± 0.75 | 1.00 ± 0.20 | 2.00 ± 0.76 | 2.60 ± 1.54 |
| Fujimite XLO | 0.90 ± 0.38 | 0.60 ± 0.24 | 1.60 ± 0.65 | 1.20 ± 0.23 ab | 1.40 ± 0.50 | 0.60 ± 0.38 | 3.00 ± 1.24 | 2.60 ± 1.36 |
| Grandevo | 0.45 ± 0.28 | 0.00 | 0.60 ± 0.38 | 0.80 ± 0.32 b | 1.80 ± 0.75 | 0.60 ± 0.38 | 1.80 ± 0.68 | 2.40 ± 0.56 |
| Venerate XC | 2.40 ± 1.22 | 1.65 ± 1.26 | 1.80 ± 0.82 | 1.40 ± 0.50 ab | 1.40 ± 0.88 | 2.80 ± 1.20 | 2.00 ± 0.23 | 5.20 ± 1.51 |
| Nealta SC | 0.90 ± 0.38 | 1.35 ± 0.56 | 1.80 ± 0.68 | 0.40 ± 0.23 b | 1.60 ± 0.80 | 3.60 ± 1.74 | 2.00 ± 0.51 | 3.80 ± 0.82 |
| p value | 0.0570 | 0.3899 | 0.7879 | 0.0046 | 0.8894 | 0.5535 | 0.3962 | 0.6581 |
| Treatment | Pretreatment | Post I Spray | Post II Spray | Post III Spray | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1–3 Instar | 4–5 Instar | Adult | All Stages | 1–3 Instar | 4–5 Instar | Adult | All Stages | 1–3 Instar | 4–5 Instar | Adult | All Stages | 1–3 Instar | 4–5 Instar | Adult | All Stages | |
| Untreated | 18.50 ± 6.70 | 2.50 ± 1.32 | 2.00 ± 0.57 | 23.00 ± 8.25 | 25.50 ± 5.08 | 5.75 ± 2.80 | 2.50 ± 1.50 | 33.75 ± 8.37 | 24.00 ± 6.82 | 6.75 ± 2.39 a * | 6.75 ± 1.37 | 37.50 ± 10.51 | 33.50 ± 14.38 | 6.25 ± 1.11 a | 12.00 ± 3.89 | 51.75 ± 18.25 |
| Assail 70 WP | 20.75 ± 8.09 | 3.75 ± 2.17 | 2.50 ± 1.55 | 27.00 ± 11.27 | 21.00 ± 4.77 | 4.00 ± 2.48 | 1.50 ± 0.95 | 26.50 ± 7.35 | 22.00 ± 8.12 | 5.50 ± 1.85 ab | 4.75 ± 1.31 | 32.25 ± 10.34 | 20.00 ± 9.37 | 2.25 ± 0.63 ab | 6.50 ± 3.66 | 28.75 ± 12.75 |
| Vacuum | 21.25 ± 7.39 | 2.50 ± 0.64 | 2.25 ± 1.10 | 26.00 ± 8.45 | 15.50 ± 2.53 | 3.75 ± 1.18 | 1.50 ± 0.95 | 20.75 ± 3.27 | 22.75 ± 6.96 | 6.25 ± 2.65 ab | 4.25 ± 1.49 | 33.25 ± 10.72 | 25.25 ± 7.34 | 3.25 ± 1.25 ab | 4.50 ± 1.19 | 33.00 ± 9.60 |
| Rim+Brig/Met+Debug/Met+AzaG | 20.75 ± 5.50 | 2.50 ± 1.32 | 1.25 ± 0.94 | 24.50 ± 6.66 | 14.25 ± 5.15 | 2.50 ± 0.86 | 2.25 ± 1.10 | 19.00 ± 6.41 | 16.25 ± 5.53 | 1.25 ± 0.25 b | 5.25 ± 1.75 | 22.75 ± 7.38 | 30.25 ± 6.83 | 1.00 ± 0.70 b | 6.25 ± 2.53 | 37.50 ± 8.70 |
| Sequoia/Sequoia/Vacuum | 11.50 ± 4.57 | 1.75 ± 0.85 | 0.75 ± 0.48 | 14.00 ± 5.80 | 11.50 ± 2.72 | 2.25 ± 1.31 | 3.25 ± 1.25 | 17.00 ± 4.81 | 13.25 ± 4.95 | 2.75 ± 1.11 ab | 4.00 ± 1.78 | 20.00 ± 6.99 | 26.50 ± 10.63 | 0.50 ± 0.28 b | 7.75 ± 3.12 | 34.7513.36 |
| Pfr+Neemix/Pfr+Neemix/Vacuum | 21.25 ± 3.92 | 2.00 ± 0.91 | 1.50 ± 0.95 | 24.75 ± 4.02 | 19.75 ± 5.36 | 4.75 ± 2.75 | 1.75 ± 0.75 | 26.25 ± 6.77 | 21.00 ± 6.82 | 3.50 ± 1.44 ab | 6.00 ± 2.16 | 30.50 ± 9.57 | 32.50 ± 10.61 | 2.00 ± 1.41 ab | 7.25 ± 2.53 | 41.75 ± 12.21 |
| Vac?Siv+Debug/Rim+Brig | 17.25 ± 8.38 | 1.00 ± 0.41 | 1.00 ± 0.71 | 19.25 ± 8.87 | 21.00 ± 8.89 | 3.25 ± 1.43 | 2.50 ± 1.50 | 26.75 ± 11.76 | 18.75 ± 4.25 | 3.25 ± 1.49 ab | 2.50 ± 0.95 | 24.50 ± 6.38 | 15.25 ± 6.07 | 1.75 ± 1.18 b | 5.25 ± 2.56 | 22.25 ± 9.05 |
| Sivanto/Sivanto/Vacuum | 30.00 ± 10.97 | 2.00 ± 0.70 | 1.75 ± 0.85 | 33.75 ± 11.79 | 23.75 ± 5.90 | 4.00 ± 1.41 | 2.75 ± 0.47 | 30.50 ± 6.93 | 16.75 ± 5.43 | 6.25 ± 1.49 ab | 5.25 ± 1.75 | 28.25 ± 8.07 | 21.50 ± 6.18 | 3.50 ± 1.50 ab | 5.25 ± 1.88 | 30.25 ± 8.83 |
| Sequoia/Sivanto/Beleaf | 31.50 ± 12.54 | 3.50 ± 2.02 | 0.75 ± 0.25 | 35.75 ± 14.34 | 12.50 ± 5.81 | 4.50 ± 1.19 | 1.75 ± 0.47 | 18.75 ± 7.39 | 23.25 ± 7.87 | 4.75 ± 1.49 ab | 7.50 ± 1.85 | 35.50 ± 7.22 | 15.25 ± 3.70 | 1.50 ± 0.86 b | 5.25 ± 1.31 | 22.00 ± 5.43 |
| XPULSE/XCEDE/XPECTRO | 15.00 ± 3.13 | 1.25 ± 0.94 | 1.25 ± 0.48 | 17.50 ± 4.33 | 22.00 ± 4.96 | 4.00 ± 0.70 | 2.25 ± 0.85 | 28.25 ± 5.31 | 24.00 ± 8.91 | 5.00 ± 1.47 ab | 3.00 ± 0.81 | 32.00 ± 9.83 | 29.00 ± 6.85 | 2.25 ± 0.75 ab | 6.00 ± 2.80 | 37.25 ± 9.93 |
| XPECTRO/XPULSE/Beleaf | 17.00 ± 6.01 | 1.50 ± 0.64 | 0.75 ± 0.75 | 19.25 ± 6.96 | 20.75 ± 6.70 | 3.75 ± 1.37 | 1.75 ± 0.47 | 26.25 ± 7.60 | 24.50 ± 3.38 | 5.75 ± 2.05 ab | 5.50 ± 1.70 | 35.75 ± 5.93 | 16.25 ± 5.12 | 2.00 ± 1.08 ab | 4.50 ± 2.32 | 22.75 ± 8.19 |
| XPECTRO/Vac/Rim+Brig | 32.50 ± 8.51 | 3.50 ± 1.44 | 1.50 ± 0.86 | 37.50 ± 9.34 | 31.00 ± 6.21 | 7.00 ± 2.64 | 4.00 ± 1.29 | 42.00 ± 9.12 | 26.00 ± 9.89 | 7.25 ± 1.54 a | 7.00 ± 1.58 | 40.25 ± 12.57 | 18.00 ± 6.25 | 3.00 ± 1.47 ab | 9.25 ± 3.32 | 30.25 ± 10.53 |
| p value | 0.7107 | 0.8608 | 0.8794 | 0.7562 | 0.1077 | 0.8097 | 0.7849 | 0.1493 | 0.6448 | 0.0090 | 0.2466 | 0.2422 | 0.3936 | 0.0074 | 0.4233 | 0.4619 |
| Treatment | Western Flower Thrips | Greenhouse Whiteflies | Natural Enemy Complex | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre-Treat. | Post I Treat. | Post II Treat. | Post III Treat. | Pre-Treat. | Post I Treat. | Post II Treat. | Post III Treat. | Pre-Treat. | Post I Treat. | Post II Treat. | Post III Treat. | |
| Untreated | 7.00 ± 3.00 | 15.75 ± 5.66 | 13.25 ± 4.21 | 12.25 ± 2.28 | 0.25 ± 0.25 | 1.00 ± 0.41 | 0.25 ± 0.25 | 1.75 ± 0.85 | 1.25 ± 0.63 | 2.25 ± 0.85 | 4.75 ± 3.54 | 2.25 ± 1.10 |
| Assail 70 WP | 9.00 ± 1.82 | 21.00 ± 10.32 | 13.25 ± 5.87 | 13.50 ± 2.60 | 0.25 ± 0.25 | 0.25 ± 0.25 | 1.25 ± 1.25 | 0.50 ± 0.50 | 2.25 ± 1.93 | 5.25 ± 4.60 | 3.75 ± 2.25 | 2.00 ± 1.41 |
| Vacuum | 4.75 ± 1.11 | 20.50 ± 6.20 | 15.50 ± 4.66 | 15.75 ± 4.82 | 0.75 ± 0.47 | 1.25 ± 0.94 | 0.75 ± 0.47 | 0.75 ± 0.47 | 2.00 ± 0.91 | 3.00 ± 1.78 | 3.00 ± 1.47 | 1.00 ± 0.57 |
| Rim+Brig/Met+Debug/Met+AzaG | 18.25 ± 7.69 | 12.25 ± 2.86 | 8.25 ± 2.49 | 11.50 ± 2.02 | 0.50 ± 0.50 | 0.25 ± 0.25 | 0.25 ± 0.25 | 0.50 ± 0.50 | 2.00 ± 1.35 | 3.50 ± 2.25 | 1.75 ± 0.47 | 2.00 ± 0.70 |
| Sequoia/Sequoia/Vacuum | 5.50 ± 1.04 | 17.75 ± 4.95 | 14.75 ± 4.36 | 18.25 ± 5.57 | 0.00 | 0.25 ± 0.25 | 0.00 | 0.50 ± 0.50 | 0.75 ± 0.47 | 3.00 ± 2.04 | 3.00 ± 1.73 | 2.75 ± 1.60 |
| Pfr+Neemix/Pfr+Neemix/Vacuum | 13.25 ± 2.49 | 18.50 ± 4.25 | 15.25 ± 4.64 | 13.50 ± 1.19 | 0.00 | 1.00 ± 0.70 | 0.25 ± 0.25 | 1.00 ± 0.57 | 0.25 ± 0.25 | 2.50 ± 1.89 | 2.25 ± 1.60 | 2.50 ± 1.19 |
| Vac?Siv+Debug/Rim+Brig | 5.50 ± 2.46 | 14.25 ± 5.13 | 11.75 ± 3.70 | 6.25 ± 2.49 | 0.75 ± 0.75 | 0.00 | 0.00 | 0.00 | 1.00 ± 0.70 | 2.25 ± 0.85 | 2.25 ± 0.85 | 2.25 ± 1.03 |
| Sivanto/Sivanto/Vacuum | 9.75 ± 1.49 | 12.50 ± 6.58 | 11.75 ± 4.32 | 10.50 ± 3.17 | 0.25 ± 0.25 | 0.00 | 0.00 | 0.250.25 | 2.75 ± 1.11 | 3.50 ± 2.53 | 5.25 ± 3.94 | 3.00 ± 0.91 |
| Sequoia/Sivanto/Beleaf | 7.25 ± 4.62 | 14.50 ± 3.96 | 13.75 ± 2.25 | 16.75 ± 3.94 | 0.25 ± 0.25 | 1.00 ± 1.00 | 0.00 | 0.25 ± 0.25 | 1.00 ± 0.41 | 6.75 ± 3.81 | 1.25 ± 0.94 | 5.50 ± 3.57 |
| XPULSE/XCEDE/XPECTRO | 16.50 ± 7.76 | 16.50 ± 4.73 | 10.25 ± 2.75 | 11.75 ± 0.85 | 0.25 ± 0.25 | 1.25 ± 0.75 | 0.00 | 0.25 ± 0.25 | 0.75 ± 0.25 | 3.50 ± 1.55 | 2.00 ± 1.08 | 3.25 ± 1.79 |
| XPECTRO/XPULSE/Beleaf | 6.00 ± 1.91 | 17.00 ± 6.86 | 8.75 ± 2.17 | 10.25 ± 1.70 | 0.00 | 1.25 ± 0.94 | 0.50 ± 0.50 | 0.75 ± 0.47 | 2.50 ± 0.95 | 3.75 ± 2.78 | 1.75 ± 0.75 | 2.00 ± 0.41 |
| XPECTRO/Vac/Rim+Brig | 9.75 ± 5.48 | 18.00 ± 4.43 | 9.00 ± 4.02 | 14.50 ± 2.10 | 0.00 | 0.25 ± 0.25 | 0.00 | 1.00 ± 0.70 | 1.75 ± 0.75 | 5.75 ± 0.47 | 4.25 ± 1.88 | 2.75 ± 1.18 |
| p value | 0.2582 | 0.8744 | 0.6570 | 0.3309 | 0.8162 | 0.6930 | 0.6029 | 0.4671 | 0.6241 | 0.7246 | 0.4279 | 0.4619 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dara, S.K.; Peck, D.; Murray, D. Chemical and Non-Chemical Options for Managing Twospotted Spider Mite, Western Tarnished Plant Bug and Other Arthropod Pests in Strawberries. Insects 2018, 9, 156. https://doi.org/10.3390/insects9040156
Dara SK, Peck D, Murray D. Chemical and Non-Chemical Options for Managing Twospotted Spider Mite, Western Tarnished Plant Bug and Other Arthropod Pests in Strawberries. Insects. 2018; 9(4):156. https://doi.org/10.3390/insects9040156
Chicago/Turabian StyleDara, Surendra K., David Peck, and Dave Murray. 2018. "Chemical and Non-Chemical Options for Managing Twospotted Spider Mite, Western Tarnished Plant Bug and Other Arthropod Pests in Strawberries" Insects 9, no. 4: 156. https://doi.org/10.3390/insects9040156
APA StyleDara, S. K., Peck, D., & Murray, D. (2018). Chemical and Non-Chemical Options for Managing Twospotted Spider Mite, Western Tarnished Plant Bug and Other Arthropod Pests in Strawberries. Insects, 9(4), 156. https://doi.org/10.3390/insects9040156






