Distribution of Canthon rutilans rutilans and Canthon rutilans cyanescens Along Spatio-Temporal and Temperature Gradients
Abstract
1. Introduction
2. Material and Methods
2.1. Study Area
2.2. Beetle Sampling
2.3. Temperature Measurements
2.4. Data Analysis
3. Results
3.1. Spatial and Temporal Segregation
3.2. The Explanatory Capacity of Temperature
3.3. Derived Thermal Responses
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ramsey, J.; Bradshaw, H.D.; Schemske, D.W. Components of reproductive isolation between the monkeyflowers Mimulus lewisii and M. cardinalis (Phrymaceae). Evolution 2003, 57, 1520–1534. [Google Scholar] [CrossRef] [PubMed]
- Doebeli, M.; Dieckmann, U. Speciation along environmental gradients. Nature 2003, 421, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Angert, A.L.; Schemske, D.W. The evolution of species’ distributions: Reciprocal transplants across the elevation ranges of Mimulus cardinalis and M. lewisii. Evolution 2005, 59, 1671–1684. [Google Scholar] [CrossRef] [PubMed]
- Zink, R.M. The geography of speciation: Case studies from birds. Evolution 2012, 5, 541–546. [Google Scholar] [CrossRef]
- Sobel, J.M.; Chen, G.F.; Watt, L.R.; Schemske, D.W. The biology of speciation. Evolution 2010, 64, 295–315. [Google Scholar] [CrossRef] [PubMed]
- Rahbek, C. The elevational gradient of species richness: A uniform pattern? Ecography 1995, 18, 200–205. [Google Scholar] [CrossRef]
- Lomolino, M.V. Elevation gradients of species–density historical and prospective views. Glob. Ecol. Biogeogr. 2001, 10, 3–13. [Google Scholar] [CrossRef]
- Chen, I.C.; Shiu, H.J.; Benedick, S.; Holloway, J.D.; Cheye, V.K.; Barlow, H.S.; Hill, J.K.; Thomas, C.D. Elevation increases in moth assemblages over 42 years on a tropical mountain. Proc. Natl. Acad. Sci. USA 2009, 106, 1479–1483. [Google Scholar] [CrossRef] [PubMed]
- Keller, I.; Seehausen, O. Thermal adaptation and ecological speciation. Mol. Ecol. 2012, 21, 782–799. [Google Scholar] [CrossRef] [PubMed]
- Svensson, E.I. Non-ecological speciation, niche conservatism and thermal adaptation: How are they connected? Org. Divers. Evol. 2012, 12, 229–240. [Google Scholar] [CrossRef]
- Phillimore, A.B.; Owens, I.P. Are subspecies useful in evolutionary and conservation biology? Proc. Biol. Sci. 2006, 273, 1049–1053. [Google Scholar] [CrossRef] [PubMed]
- De Queiroz, K. Ernst Mayr and the modern concept of species. Proc. Natl. Acad. Sci. USA 2005, 102, 6600–6607. [Google Scholar] [CrossRef] [PubMed]
- Chown, S.L.; Nicholson, S.W. Insect Physiological Ecology: Mechanisms and Patterns; Oxford University Press: Oxford, UK, 2004. [Google Scholar]
- Vaz-de-Mello, F.; Larsen, T.; Silva, F.; Gill, B.; Spector, S.; Favila, M. The IUCN Red List of Threatened Species. Canthon rutilans, 2014. Available online: http://www.iucnredlist.org/details/137210/0 (accessed on 20 September 2018).
- Köppen, W. Das geographische System der Klimate. In Handbuch der Klimatologie, Gebrüder Borntraeger; Köppen, W., Geiger, R., Eds.; Gerbrüder Bornträger: Berlin, Germany, 1936; pp. 1–44. [Google Scholar]
- Halffter, G.; Edmonds, W.D. The Nesting Behavior of Dung Beetles (Scarabaeinae). An Ecological and Evolutive Approach; Instituto de Ecología: Mexico City, Mexico, 1982; pp. 1–176. [Google Scholar]
- Halffter, G.; Matthews, E.G. The natural history of dung beetles of the subfamily Scarabaeinae (Coleoptera, Scarabaeidae). Folia Entomol. Mex. 1966, 12–14, 1–312. [Google Scholar]
- Nichols, E.; Spector, S.; Louzada, J.; Larsen, T.; Amezquita, S.; Favila, M.E. Ecological functions and ecosystem services provided by Scarabaeinae dung beetles. Biol. Conserv. 2008, 141, 1461–1474. [Google Scholar] [CrossRef]
- Davis, A.L.; Brink, D.J.; Scholtz, C.H.; Prinsloo, L.C.; Deschodt, C.M. Functional implications of temperature-correlated colour polymorphism in an iridescent, scarabaeine dung beetle. Ecol. Entomol. 2008, 33, 771–779. [Google Scholar] [CrossRef]
- Alves, V.M.; Hernández, M.I.M.; Lobo, J.M. Elytra absorb ultraviolet radiation but transmit infrared radiation in Neotropical Canthon species (Coleoptera, Scarabaeinae). Photochem. Photobiol. 2018, 94, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Hernández, M.I.M. The night and day of dung beetles (Coleoptera, Scarabaeidae) in the Serra do Japi, Brazil: Elytra colour related to daily activity. Rev. Bras. Entomol. 2002, 46, 597–600. [Google Scholar] [CrossRef]
- Amore, V.; Hernández, M.I.M.; Carrascal, L.M.; Lobo, J.M. Exoskeleton may influence the internal body temperatures of Neotropical dung beetles (Col. Scarabaeinae). PeerJ 2017, 5, e3349. [Google Scholar] [CrossRef] [PubMed]
- Korasaki, V.; Lopes, J.; Gardner Brown, G.; Louzada, J. Using dung beetles to evaluate the effects of urbanization on Atlantic Forest biodiversity. Insect Sci. 2013, 20, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Culot, L.; Bovy, E.; Vaz-de-Mello, F.Z.; Guevara, R.; Galetti, M. Selective defaunation affects dung beetle communities in continuous Atlantic rainforest. Biol. Conserv. 2013, 163, 79–89. [Google Scholar] [CrossRef]
- Bett, J.Z.; de Farias, P.M.; da Silva, P.G.; Hernández, M.I.M. Dung beetle communities in coal mining areas in the process of recovery. Biotemas 2014, 27, 197–200. [Google Scholar]
- Da Silva, P.G.; Garcia, M.A.R.; Vidal, M.B. Besouros copro-necrófagos (Coleoptera: Scarabaeidae stricto sensu) coletados em ecótono natural de campo e mata em Bagé, RS. Ciênc. Nat. 2008, 30, 71–91. [Google Scholar]
- Da Silva, P.G.; Audino, L.D.; Nogueira, J.M.; Moraes, L.P.; Vaz-de-Mello, F.Z. Escarabeíneos (Coleoptera: Scarabaeidae: Scarabaeinae) de uma área de campo nativo no bioma Pampa, Rio Grande do Sul, Brasil. Biota Neotrop. 2012, 12, 246–253. [Google Scholar] [CrossRef]
- Da Silva, P.G. Dung beetles (Coleoptera, Scarabaeinae) from high-altitude grasslands in São Joaquim National Park, Santa Catarina, southern Brazil. Check List 2017, 13, 817–830. [Google Scholar] [CrossRef]
- Audino, L.D.; da Silva, P.G.; Nogueira, J.M.; Moraes, L.P.; Vaz-de-Mello, F.Z. Scarabaeinae (Coleoptera, Scarabaeidae) de um bosque de eucalipto introduzido em uma região originalmente campestre. Iheringia Sér. Zool. 2011, 101, 121–126. [Google Scholar] [CrossRef]
- Garcia, L.E.; Moraes, R.M.; Vianna, É.E.S. Levantamento de besouros copro-necrófagos (Coleoptera: Scarabaeidae: Scarabaeinae) do Bioma Pampa. Rev. Ciênc. Agrovet. 2016, 15, 144–154. [Google Scholar] [CrossRef]
- Campos, R.C.; Hernández, M.I.M. Dung beetle assemblages (Coleoptera, Scarabaeinae) in Atlantic forest fragments in southern Brazil. Rev. Bras. Entomol. 2013, 57, 47–54. [Google Scholar] [CrossRef]
- Campos, R.C.; Hernández, M.I.M. Changes in the dynamics of functional groups in communities of dung beetles in Atlantic forest fragments adjacent to transgenic maize crops. Ecol. Indic. 2015, 49, 216–227. [Google Scholar] [CrossRef]
- Campos, R.C.; Hernández, M.I.M. The importance of maize management on dung beetle communities in Atlantic Forest fragments. PLoS ONE 2015, 10, e0145000. [Google Scholar] [CrossRef] [PubMed]
- Bogoni, J.A.; Hernández, M.I.M. Attractiveness of native mammal’s feces of different trophic guilds to dung beetles (Coleoptera: Scarabaeinae). J. Insect Sci. 2014, 14, 299. [Google Scholar] [CrossRef] [PubMed]
- Costa-Silva, V.; Cipolatto, R.P.; Abegg, A.D.; Rosa, C.M.; da Silva, P.G.; Di Mare, R.A. Escarabeídeos (Coleoptera: Scarabaeidae) de campo e floresta da Reserva Biológica de São Donato, Rio Grande do Sul, Brasil. Biotemas 2014, 27, 63–71. [Google Scholar] [CrossRef]
- Da Silva, P.G.; Hernández, M.I.M. Local and regional effects on community structure of dung beetles in a mainland-island scenario. PLoS ONE 2014, 9, e111883. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, P.G.; Hernández, M.I.M. Scale-dependence of processes structuring dung beetle metacommunities using functional diversity and community deconstruction approaches. PLoS ONE 2015, 10, e0123030. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, P.G.; Hernández, M.I.M. Spatial patterns of movement of dung beetle species in a tropical forest suggest a new trap spacing for dung beetle biodiversity studies. PLoS ONE 2015, 10, e0126112. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, P.G.; Hernández, M.I.M. Spatial variation of dung beetle assemblages associated with forest structure in remnants of southern Brazilian Atlantic Forest. Rev. Bras. Entomol. 2016, 60, 73–81. [Google Scholar] [CrossRef]
- Da Silva, P.G.; Garcia, M.A.R.; Vidal, M.B. Besouros copro-necrófagos (Coleoptera: Scarabaeidae sensu stricto) do município de Bagé, RS (Bioma Campos Sulinos). Biociências 2009, 17, 33–43. [Google Scholar]
- Hernández, M.I.M.; Vaz-de-Mello, F.Z. Seasonal and spatial species richness variation of dung beetle (Coleoptera, Scarabaeidae s. str.) in the Atlantic Forest of southeastern Brazil. Rev. Bras. Entomol. 2009, 53, 607–613. [Google Scholar] [CrossRef]
- Wiens, J.J.; Graham, C.H. Niche Conservatism: Integrating Evolution, Ecology, and Conservation Biology. Annu. Rev. Ecol. Evol. Syst. 2005, 36, 519–539. [Google Scholar] [CrossRef]
- IBGE. Manual Técnico da Vegetação Brasileira; Instituto Brasileiro de Geografia e Estatística: Rio de Janeiro, Brazil, 1992. [Google Scholar]
- Dobson, A. An Introduction to Generalized Linear Models; Chapman and Hall/CRC: Boca Raton, FL, USA, 1999. [Google Scholar]
- Matute, D.R.; Novak, C.J.; Coyne, J.A. Temperature-based extrinsic reproductive isolation in two species of Drosophila. Evolution 2009, 63, 595–612. [Google Scholar] [CrossRef] [PubMed]
- Fuller, R.C.; McGhee, K.E.; Schrader, M. Speciation in killifish and the role of salt tolerance. J. Evol. Biol. 2007, 20, 1962–1975. [Google Scholar] [CrossRef] [PubMed]
- Lumaret, J.P.; Stiernet, N. Montane Dung Beetles. In Dung Beetle Ecology; Hanski, I., Cambefort, Y., Eds.; Princeton University Press: Princeton, NJ, USA, 1991; pp. 242–254. [Google Scholar]
- Janzen, D.H. Why mountain passes are higher in the tropics. Am. Nat. 1967, 101, 233–249. [Google Scholar] [CrossRef]
- Ghalambor, C.K.; Huey, R.B.; Martin, P.R.; Tewksbury, J.J.; Wang, G. Are mountain passes higher in the tropics? Janzen’s hypothesis revisited. Integr. Comp. Biol. 2006, 46, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Mittelbach, G.G.; Schemske, D.W.; Cornell, H.V.; Allen, A.P.; Brown, J.M.; Bush, M.B.; Harrison, S.P.; Hurlbert, A.H.; Knowlton, N.; Lessios, H.A.; et al. Evolution and the latitudinal diversity gradient: Speciation, extinction and biogeography. Ecol. Lett. 2007, 10, 315–331. [Google Scholar] [CrossRef] [PubMed]
- Sunday, J.M.; Bates, A.E.; Kearney, M.R.; Colwell, R.K.; Dulvy, N.K.; Longino, J.T.; Huey, R.B. Thermal-safety margins and the necessity of thermoregulatory behavior across latitude and elevation. Proc. Natl. Acad. Sci. USA 2014, 111, 5610–5615. [Google Scholar] [CrossRef] [PubMed]
- Lobo, J.M.; Halffter, G. Biogeographical and ecological factors affecting the altitudinal variation of mountainous communities of coprophagous beetles (Coleoptera: Scarabaeoidea): A comparative study. Ann. Entomol. Soc. Am. 2014, 93, 115–126. [Google Scholar] [CrossRef]
- Da Silva, P.G.; Lobo, J.M.; Hensen, M.C.; Vaz-de-Mello, F.Z.; Hernández, M.I.M. Turnover and nestedness in subtropical dung beetle assemblages along an elevational gradient. Divers. Distrib. 2018, 24, 1277–1290. [Google Scholar] [CrossRef]
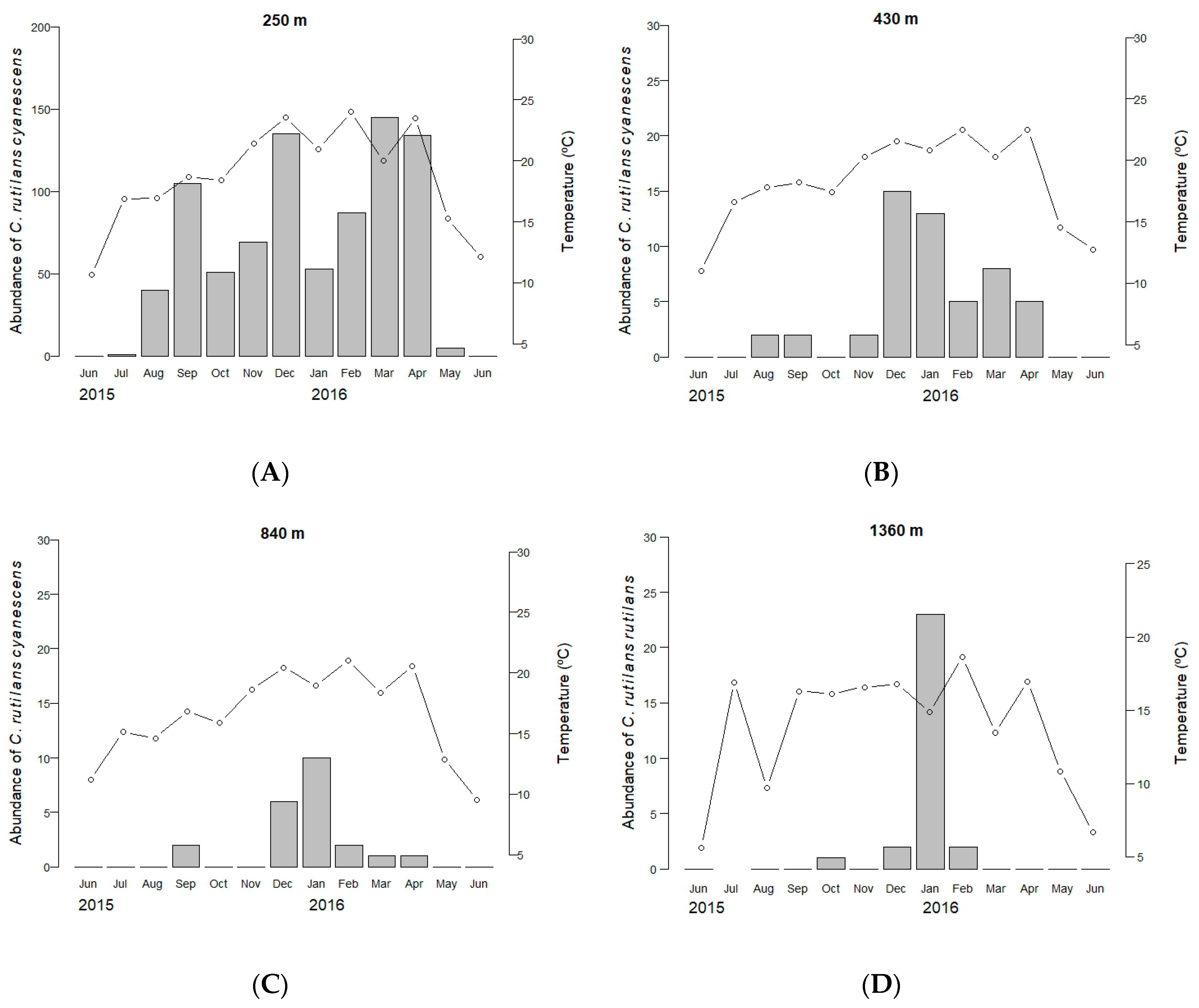
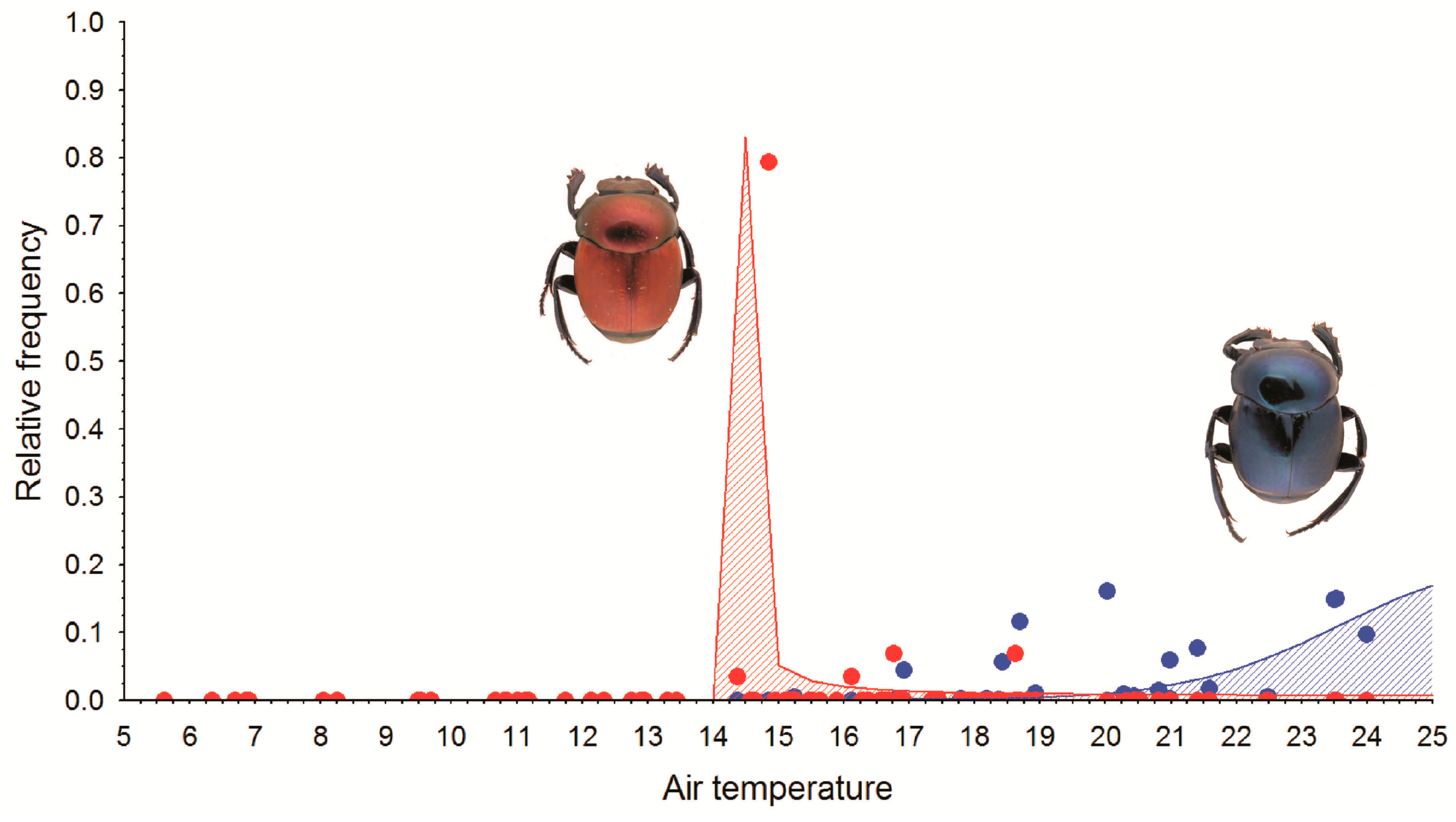
| Subspecies | Area | Data | Mean Temperature (°C) (3 d) | Mean Minimum Temperature (°C) (3 d) | Minimum Temperature (°C) (Month) |
|---|---|---|---|---|---|
| Canthon rutilans cyanescens | 250 m | VII/2015 | 16.91 | 15.47 | 6.67 |
| Canthon rutilans cyanescens | 250 m | VIII/2015 | 16.91 | 13.23 | 8.88 |
| Canthon rutilans cyanescens | 250 m | IX/2015 | 18.69 | 14.86 | 6.98 |
| Canthon rutilans cyanescens | 250 m | X/2015 | 18.42 | 15.85 | 10.26 |
| Canthon rutilans cyanescens | 250 m | XI/2015 | 21.40 | 18.62 | 12.98 |
| Canthon rutilans cyanescens | 250 m | XII/2015 | 23.54 | 19.76 | 15.57 |
| Canthon rutilans cyanescens | 250 m | I/2016 | 20.98 | 17.44 | 16.14 |
| Canthon rutilans cyanescens | 250 m | II/2016 | 24.00 | 20.49 | 14.80 |
| Canthon rutilans cyanescens | 250 m | III/2016 | 20.03 | 17.70 | 14.71 |
| Canthon rutilans cyanescens | 250 m | IV/2016 | 23.50 | 20.52 | 5.84 |
| Canthon rutilans cyanescens | 250 m | V/2016 | 15.25 | 13.36 | 7.06 |
| Canthon rutilans cyanescens | 430 m | VIII/2015 | 17.79 | 13.64 | 6.06 |
| Canthon rutilans cyanescens | 430 m | IX/2015 | 18.19 | 15.56 | 6.06 |
| Canthon rutilans cyanescens | 430 m | XI/2015 | 20.31 | 18.20 | 13.46 |
| Canthon rutilans cyanescens | 430 m | XII/2015 | 21.59 | 19.09 | 14.61 |
| Canthon rutilans cyanescens | 430 m | I/2016 | 20.82 | 17.60 | 16.24 |
| Canthon rutilans cyanescens | 430 m | II/2016 | 22.48 | 20.20 | 15.86 |
| Canthon rutilans cyanescens | 430 m | III/2016 | 20.29 | 17.57 | 14.13 |
| Canthon rutilans cyanescens | 430 m | IV/2016 | 22.48 | 20.42 | 5.76 |
| Canthon rutilans cyanescens | 840 m | IX/2015 | 16.82 | 14.38 | 2.20 |
| Canthon rutilans cyanescens | 840 m | XII/2015 | 20.44 | 17.47 | 12.88 |
| Canthon rutilans cyanescens | 840 m | I/2016 | 18.93 | 14.80 | 13.17 |
| Canthon rutilans cyanescens | 840 m | II/2016 | 20.99 | 18.65 | 13.17 |
| Canthon rutilans cyanescens | 840 m | III/2016 | 18.36 | 14.99 | 12.01 |
| Canthon rutilans cyanescens | 840 m | IV/2016 | 20.53 | 18.14 | 3.89 |
| Canthon rutilans rutilans | 1360 m | X/2015 | 16.12 | 12.82 | 2.73 |
| Canthon rutilans rutilans | 1360 m | XII/2015 | 16.77 | 13.46 | 6.67 |
| Canthon rutilans rutilans | 1360 m | I/2016 | 14.85 | 8.81 | 7.18 |
| Canthon rutilans rutilans | 1360 m | II/2016 | 18.62 | 12.91 | 4.83 |
| Canthon rutilans rutilans | 1630 m | I/2016 | 14.38 | 8.61 | 7.68 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hensen, M.C.; Hernández, M.I.M.; Da Silva, P.G.; Amore, V.; Lobo, J.M. Distribution of Canthon rutilans rutilans and Canthon rutilans cyanescens Along Spatio-Temporal and Temperature Gradients. Insects 2018, 9, 124. https://doi.org/10.3390/insects9040124
Hensen MC, Hernández MIM, Da Silva PG, Amore V, Lobo JM. Distribution of Canthon rutilans rutilans and Canthon rutilans cyanescens Along Spatio-Temporal and Temperature Gradients. Insects. 2018; 9(4):124. https://doi.org/10.3390/insects9040124
Chicago/Turabian StyleHensen, Maristela Carpintero, Malva Isabel Medina Hernández, Pedro Giovâni Da Silva, Valentina Amore, and Jorge M. Lobo. 2018. "Distribution of Canthon rutilans rutilans and Canthon rutilans cyanescens Along Spatio-Temporal and Temperature Gradients" Insects 9, no. 4: 124. https://doi.org/10.3390/insects9040124
APA StyleHensen, M. C., Hernández, M. I. M., Da Silva, P. G., Amore, V., & Lobo, J. M. (2018). Distribution of Canthon rutilans rutilans and Canthon rutilans cyanescens Along Spatio-Temporal and Temperature Gradients. Insects, 9(4), 124. https://doi.org/10.3390/insects9040124





