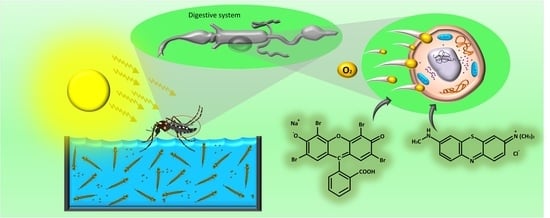
Evaluation of Eosin-Methylene Blue as a Photosensitizer for Larval Control of Aedes aegypti by a Photodynamic Process
Abstract
1. Introduction
2. Materials and Methods
2.1. Maintenance of Larvae
2.2. Photosensitizer
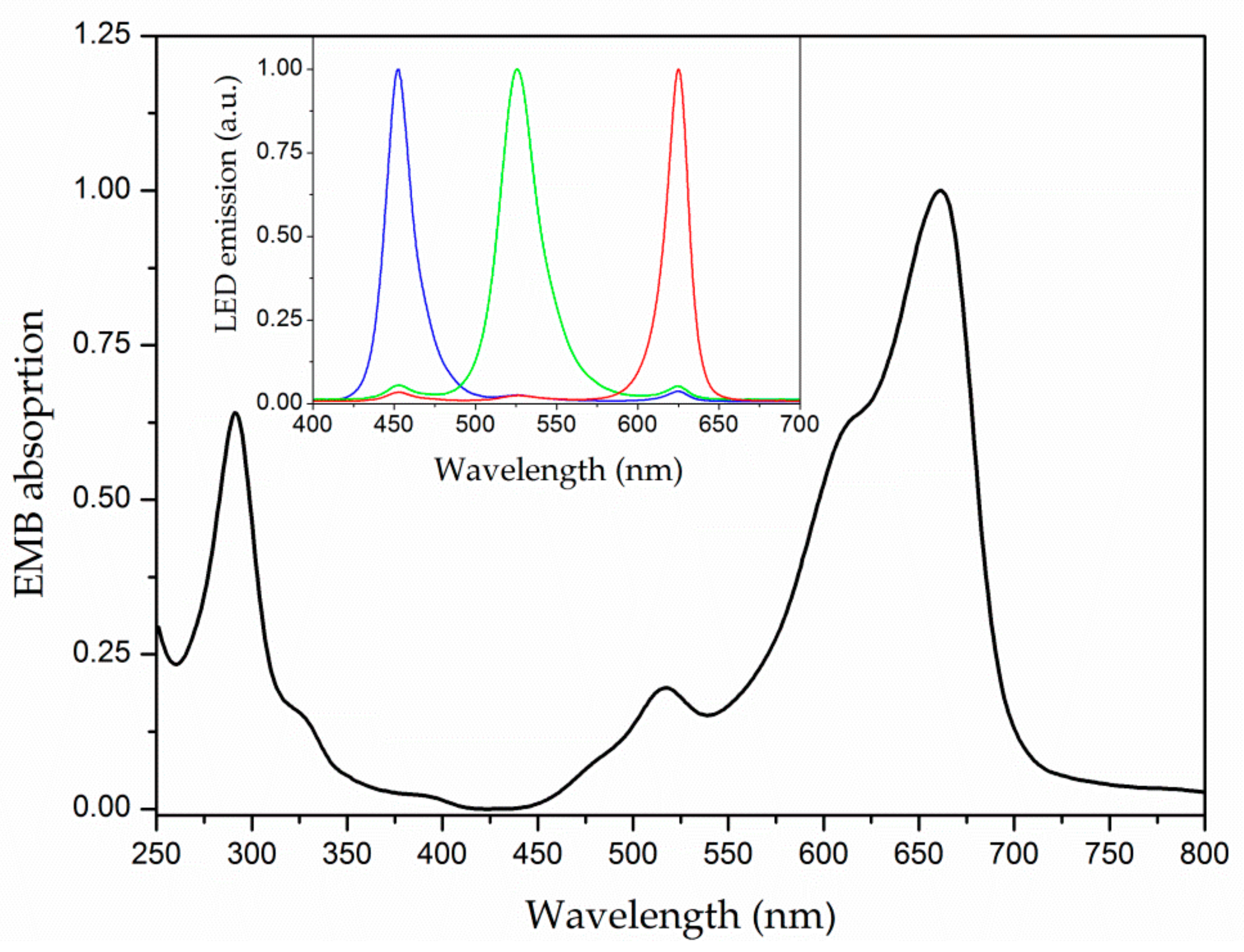
2.3. Optical Characterization
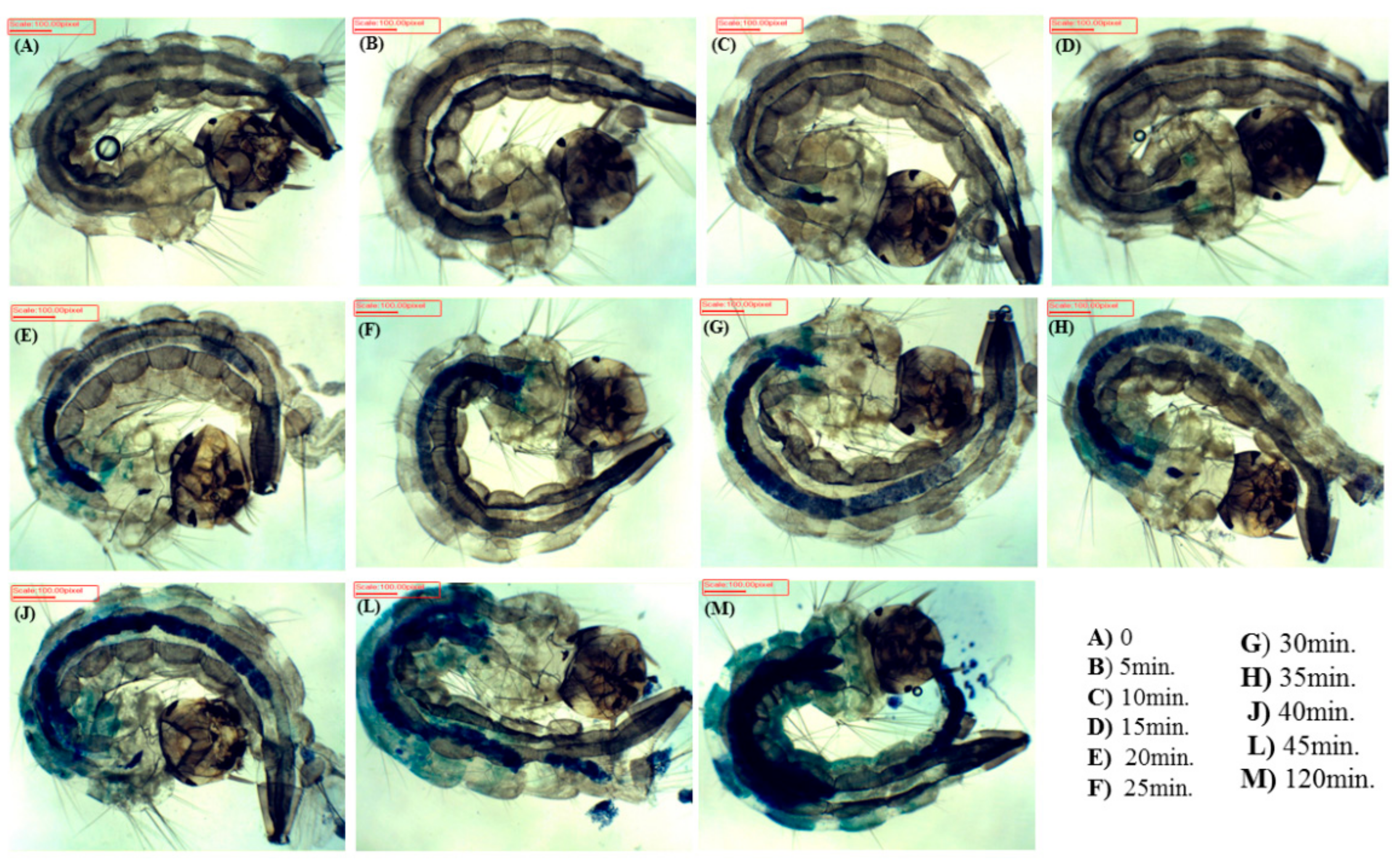
2.4. Internalization Time
2.5. Bioassays with Ae. aegypti
3. Results and Discussion
3.1. Optical Characterization
3.2. Internalization Time
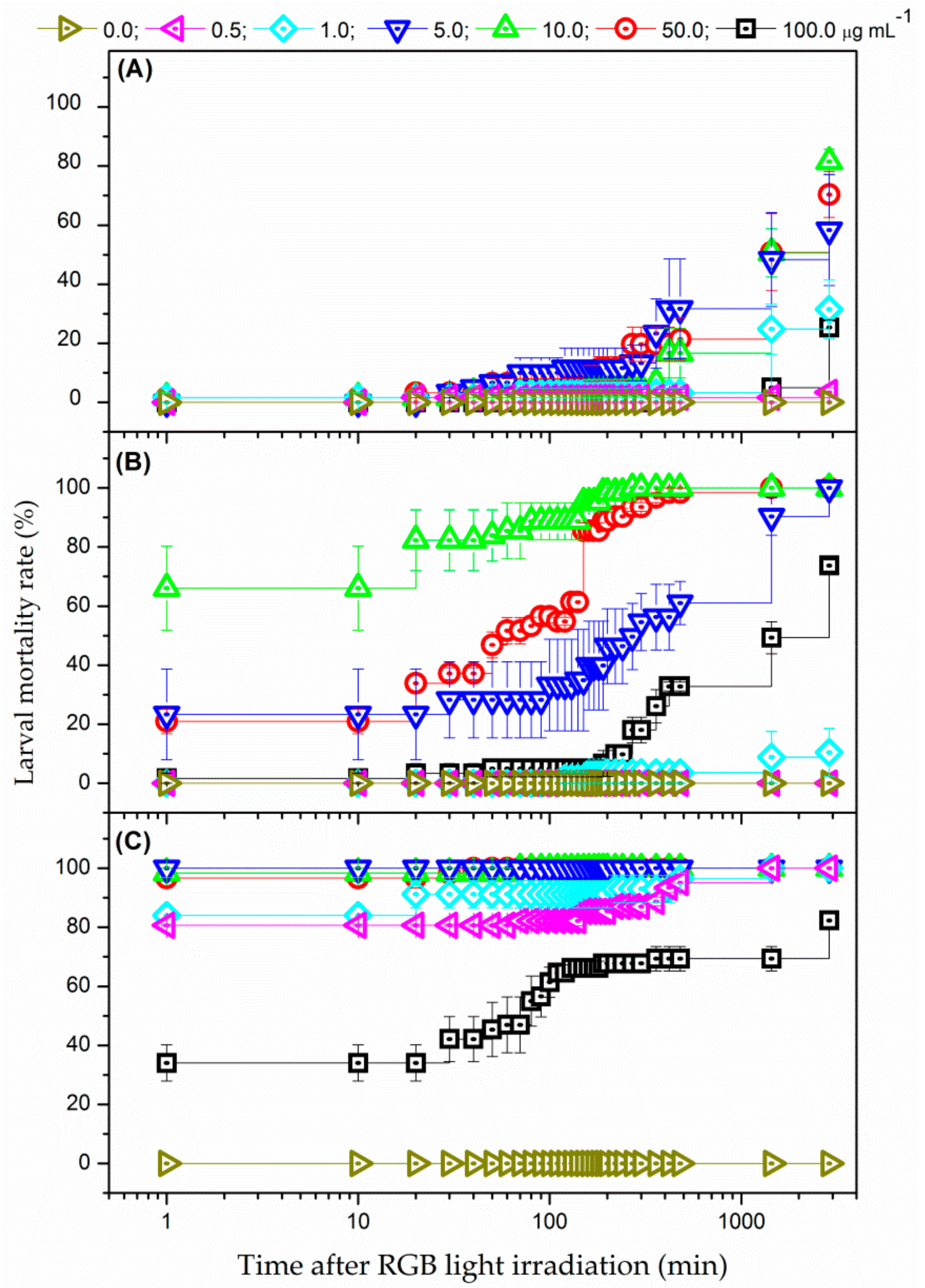
3.3. Photodynamic Control of Ae. aegypti Larvae
3.3.1. White-Light Radiation from RGB LEDs
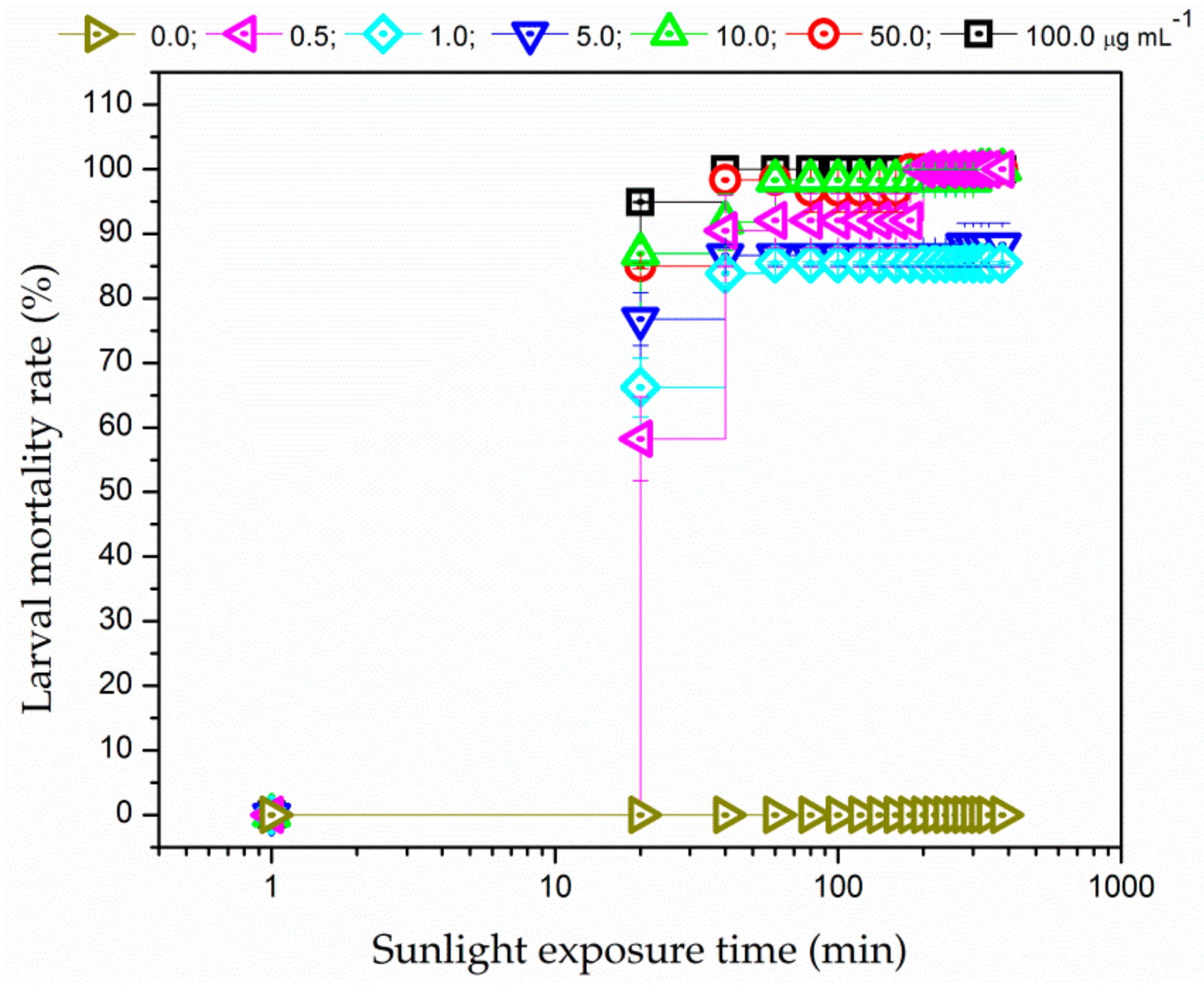
3.3.2. Sunlight Irradiation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization, Regional Office for South-East Asia. Dengue: Guidelines for Diagnosis, Treatment, Prevention, and Control, New ed.; WHO Press: Paris, France, 2009; ISBN 978-9-241-54787-1. [Google Scholar]
- World Health Organization. Zika virus outbreaks in the Americas. Wkly. Epidemiol. Rec. 2015, 90, 609–610. [Google Scholar]
- World Health Organization. Global Strategy for Dengue Prevention and Control. 2012–2020; WHO Press: Geneva, Switzerland, 2012; ISBN 978-9-241-50403-4. [Google Scholar]
- Ministério da Saúde. Diretrizes Nacionais Para a Prevenção e Controle de Epidemias de Dengue; Ministério da Saúde: Brasília, Brazil, 2009; ISBN 978-85-334-1602-4. [Google Scholar]
- Bellinato, D.F.; Viana-Medeiros, P.F.; Araújo, S.C.; Martins, A.J.; Lima, J.B.P.; Valle, D. Resistance Status to the Insecticides Temephos, Deltamethrin, and Diflubenzuron in Brazilian Aedes aegypti Populations. BioMed Res. Int. 2016, 2016, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.B.; Kasai, S.; Scott, J.G. Pyrethroid resistance in Aedes aegypti and Aedes albopictus: Important mosquito vectors of human diseases. Pestic. Biochem. Physiol. 2016, 133, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Haddi, K.; Tomé, H.V.V.; Du, Y.; Valbon, W.R.; Nomura, Y.; Martins, G.F.; Dong, K.; Oliveira, E.E. Detection of a new pyrethroid resistance mutation (V410L) in the sodium channel of Aedes aegypti: A potential challenge for mosquito control. Sci. Rep. 2017, 7, 46549. [Google Scholar] [CrossRef] [PubMed]
- Moyes, C.L.; Vontas, J.; Martins, A.J.; Ng, L.C.; Koou, S.Y.; Dusfour, I.; Raghavendra, K.; Pinto, J.; Corbel, V.; David, J.P.; et al. Contemporary status of insecticide resistance in the major Aedes vectors of arboviruses infecting humans. PLoS Negl. Trop. Dis. 2017, 11, e0005625. [Google Scholar] [CrossRef] [PubMed]
- Grzybowski, A.; Tiboni, M.; Silva, M.A.; Chitolina, R.F.; Passos, M.; Fontana, J.D. Synergistic larvicidal effect and morphological alterations induced by ethanolic extracts of Annona muricata and Piper nigrum against the dengue fever vector Aedes aegypti. Pest. Manag. Sci. 2013, 69, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Arruda, E.J.; Rossi, A.P.L.; Porto, K.R.A.; Oliveira, L.C.S.; Arakaki, A.H.; Scheidt, G.N.; Soccol, C.R. Evaluation of toxic effects with transition metal ions, EDTA, SBTI and acrylic polymers on Aedes aegypti (L., 1762) (Diptera: Culicidae) and Artemia salina (Artemidae). Braz. Arch. Biol. Technol. 2010, 53, 335–341. [Google Scholar] [CrossRef]
- Boethling, R.S.; Sommer, E.; DiFiore, D. Designing Small Molecules for Biodegradability. Chem. Rev. 2007, 107, 2207–2227. [Google Scholar] [CrossRef] [PubMed]
- Coelho, J.S.; Santos, N.D.L.; Napoleão, T.H.; Gomes, F.S.; Ferreira, R.S.; Zingali, R.B.; Coelho, L.C.B.B.; Leite, S.P.; Navarro, D.M.A.F.; Paiva, P.M.G. Effect of Moringa oleifera lectin on development and mortality of Aedes aegypti larvae. Chemosphere 2009, 77, 934–938. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, A. Sensibilizadores fluorescentes como larvicidas. Accion fotodinamica de la luz. Riv. Malariol. 1928, 7, 456. [Google Scholar]
- Amor, T.B.; Jori, G. Sunlight-activated insecticides: Historical background and mechanisms of phototoxic activity. Insect Biochem. Mol. Biol. 2000, 30, 915–925. [Google Scholar] [CrossRef]
- Azizullah, A.; Rehman, Z.U.; Ali, I.; Murad, W.; Muhammad, N.; Ullah, W.; Häder, D.P. Chlorophyll derivatives can be an efficient weapon in the fight against dengue. Parasitol. Res. 2014, 113, 4321–4326. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.M.; Inada, N.M.; Pratavieira, S.; Corbi, J.J.; Kurachi, C.; Bagnato, V.S. Efficacy of Photogem® (Hematoporphyrin Derivative) as a Photoactivatable Larvicide against Aedes aegypti (Diptera: Culicidae) Larvae. J. Life Sci. 2017, 11, 78–81. [Google Scholar] [CrossRef]
- Lucantoni, L.; Magaraggia, M.; Lupidi, G.; Ouedraogo, R.K.; Coppellotti, O.; Esposito, F.; Fabris, C.; Jori, G.; Habluetzel, A. Novel, Meso-Substituted Cationic Porphyrin Molecule for Photo-Mediated Larval Control of the Dengue Vector Aedes aegypti. PLoS Negl. Trop. Dis. 2011, 5, e1434. [Google Scholar] [CrossRef] [PubMed]
- Grinholc, M.; Rapacka-Zdonczyk, A.; Rybak, B.; Szabados, F.; Bielawski, K.P. Multiresistant Strains Are as Susceptible to Photodynamic Inactivation as Their Naı¨ve Counterparts: Protoporphyrin IX-Mediated Photoinactivation Reveals Differences between Methicillin-Resistant and Methicillin-Sensitive Staphylococcus aureus Strains. Photomed. Laser Surg. 2014, 32, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Song, R.; Feng, Y.; Wang, D.; Xu, Z.; Li, Z.; Shao, Z. Phytoalexin Phenalenone Derivatives Inactivate Mosquito Larvae and Root-knot Nematode as Type-II Photosensitizer. Sci. Rep. 2017, 7, 42058. [Google Scholar] [CrossRef] [PubMed]
- Hamblin, M.R. Antimicrobial photodynamic inactivation: A bright new technique to kill resistant microbes. Curr. Opin. Microbiol. 2016, 33, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Dondji, B.; Duchon, S.; Diabate, A.; Herve, J.P.; Corbel, V.; Hougard, J.-M.; Santus, R.; Schrevel, J. Assessment of Laboratory and Field Assays of Sunlight-Induced Killing of Mosquito Larvae by Photosensitizers. J. Med. Entomol. 2005, 42, 652–656. [Google Scholar] [CrossRef] [PubMed]
- Fleischer, B. Editorial: 100 years ago: Giemsa’s solution for staining of plasmodia. Trop. Med. Int. Health 2004, 9, 755–756. [Google Scholar] [CrossRef] [PubMed]
- Lima, A.R.; Silva, C.M.; Caires, C.S.A.; Nascimento, V.A.; Rocha, L.R.P.; Cabrini, I.; Arruda, E.J.; Oliveira, S.L.; Caires, A.R.L. Photodynamic control of Aedes aegypti larvae (Diptera: Culicidae). Photodiagn. Photodyn. Ther. 2017, 17, A49. [Google Scholar] [CrossRef]
- Caires, C.S.A.; Leal, C.R.B.; Ramos, C.A.N.; Lima, A.R.; Caires, A.R.L.; Arruda, E.J.; Oliveira, S.L.; Nascimento, V.A. Photodynamic antimicrobial therapy on S. aureus and E. coli by using Giemsa stain as photosensitizer. Photodiagn. Photodyn. Ther. 2017, 17, A41. [Google Scholar] [CrossRef]
- Caires, C.S.A.; Leal, C.R.B.; Ramos, C.A.N.; Bogo, D.; Lima, A.R.; Arruda, E.J.; Oliveira, S.L.; Caires, A.R.L.; Nascimento, V.A. Photoinactivation effect of eosin methylene blue and chlorophyllin sodium-copper against Staphylococcus aureus and Escherichia coli. Lasers Med. Sci. 2017, 32, 1081–1088. [Google Scholar] [CrossRef] [PubMed]
- Caires, C.S.A.; Leal, C.R.B.; Rodrigues, A.C.S.; Ramos, C.A.N.; Chang, M.R.; Lima, A.R.; Arruda, E.J.; Oliveira, S.L.; Nascimento, V.A.; Caires, A.R.L. Photoinactivation of mcr-1 positive Escherichia coli. Laser Phys. Lett. 2018, 15, 015601. [Google Scholar] [CrossRef]
- Arrivillaga, J.; Barrera, R. Food as a limiting factor for Aedes aegypti in water-storage containers. J. Vector Ecol. 2004, 29, 11–20. [Google Scholar] [PubMed]
- Barcia, J.J. The Giemsa Stain: Its History and Applications. Int. J. Surg. Pathol. 2007, 15, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Brammer, S.P.; Toniazzo, C.; Poersch, L.B. Corantes comumente empregados na citogenética vegetal. Arq. Inst. Biol. (Sao Paulo) 2015, 82, 1–8. [Google Scholar] [CrossRef]
- Campos, J.; Andrade, C.F.S. Insecticide resistance in Simulium populations (Diptera, Simuliidae). Cadernos Saude Publica 2002, 18, 661–671. [Google Scholar] [CrossRef]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Spring: New York, NY, USA, 2006; ISBN 978-0-387-46312-4. [Google Scholar]
- Ahmed, Y.M.; Mostafa, A.M.A.; Elewa, M.A. Toxicity of certain dyes as insecticides and their joint action with some pyrethroids. J. Environ. Sci. Health Part B 1985, 20, 689–699. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lima, A.R.; Silva, C.M.; Caires, C.S.A.; Prado, E.D.; Rocha, L.R.P.; Cabrini, I.; Arruda, E.J.; Oliveira, S.L.; Caires, A.R.L. Evaluation of Eosin-Methylene Blue as a Photosensitizer for Larval Control of Aedes aegypti by a Photodynamic Process. Insects 2018, 9, 109. https://doi.org/10.3390/insects9030109
Lima AR, Silva CM, Caires CSA, Prado ED, Rocha LRP, Cabrini I, Arruda EJ, Oliveira SL, Caires ARL. Evaluation of Eosin-Methylene Blue as a Photosensitizer for Larval Control of Aedes aegypti by a Photodynamic Process. Insects. 2018; 9(3):109. https://doi.org/10.3390/insects9030109
Chicago/Turabian StyleLima, Alessandra R., Cicera M. Silva, Cynthia S. A. Caires, Esmael D. Prado, Luciana R. P. Rocha, Isaias Cabrini, Eduardo J. Arruda, Samuel L. Oliveira, and Anderson R. L. Caires. 2018. "Evaluation of Eosin-Methylene Blue as a Photosensitizer for Larval Control of Aedes aegypti by a Photodynamic Process" Insects 9, no. 3: 109. https://doi.org/10.3390/insects9030109
APA StyleLima, A. R., Silva, C. M., Caires, C. S. A., Prado, E. D., Rocha, L. R. P., Cabrini, I., Arruda, E. J., Oliveira, S. L., & Caires, A. R. L. (2018). Evaluation of Eosin-Methylene Blue as a Photosensitizer for Larval Control of Aedes aegypti by a Photodynamic Process. Insects, 9(3), 109. https://doi.org/10.3390/insects9030109