Spatial Distribution of Bactrocera dorsalis and Thaumatotibia leucotreta in Smallholder Avocado Orchards along Altitudinal Gradient of Taita Hills and Mount Kilimanjaro
Abstract
1. Introduction
2. Materials and Methods
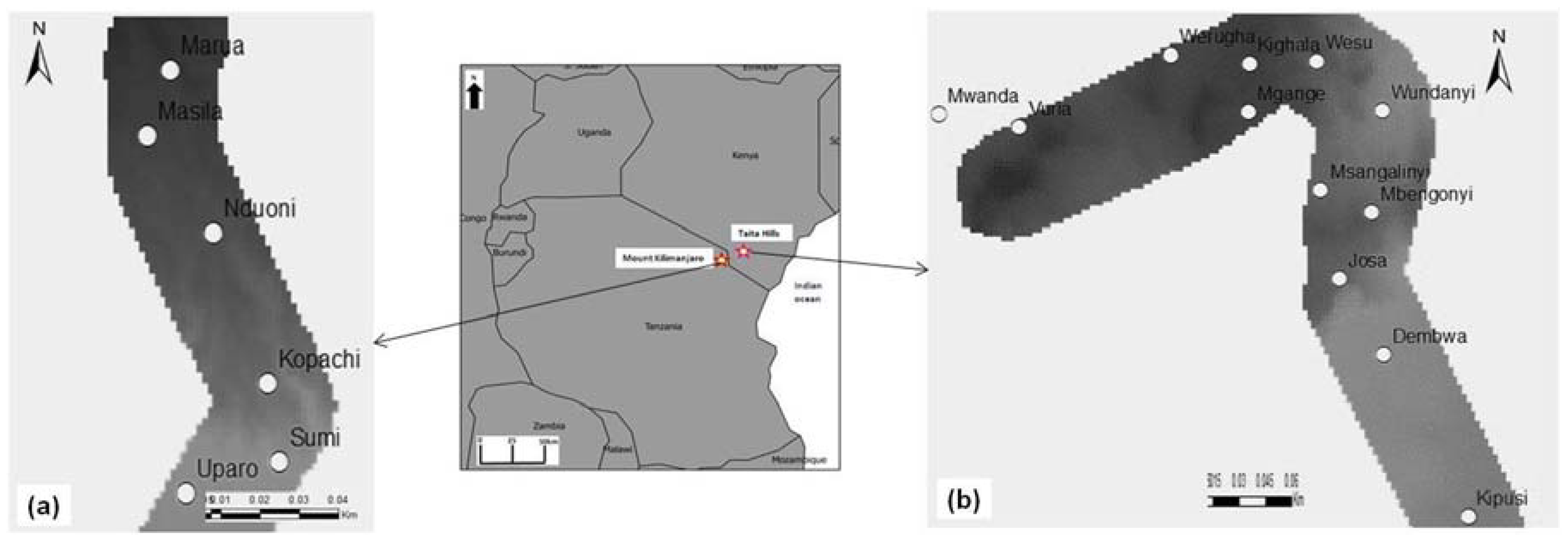
2.1. Study Areas and Agro-Ecological Conditions
2.2. Insect Monitoring Using Lures
2.3. Fruits Damage Assessment
2.4. Statistical Analysis
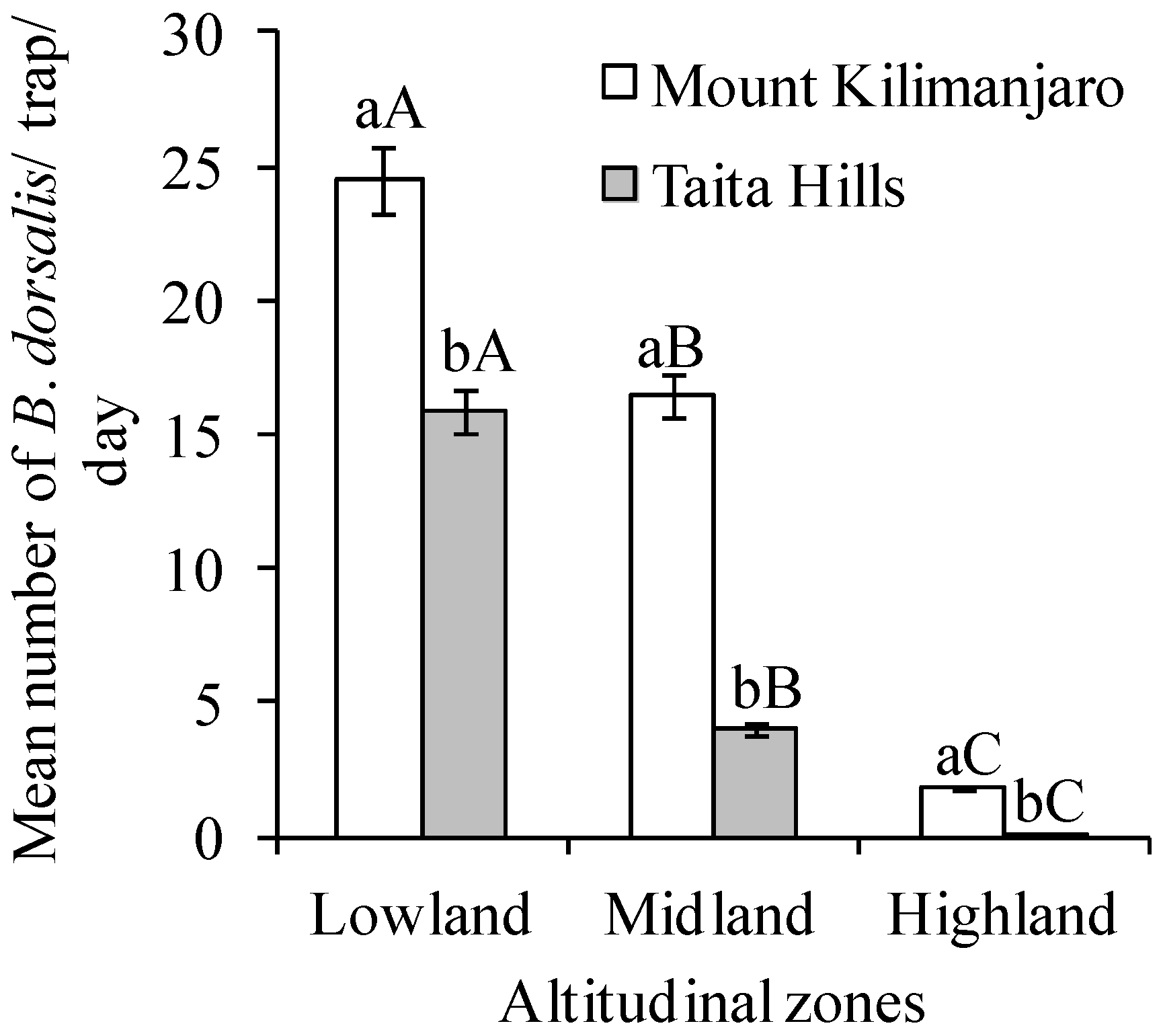
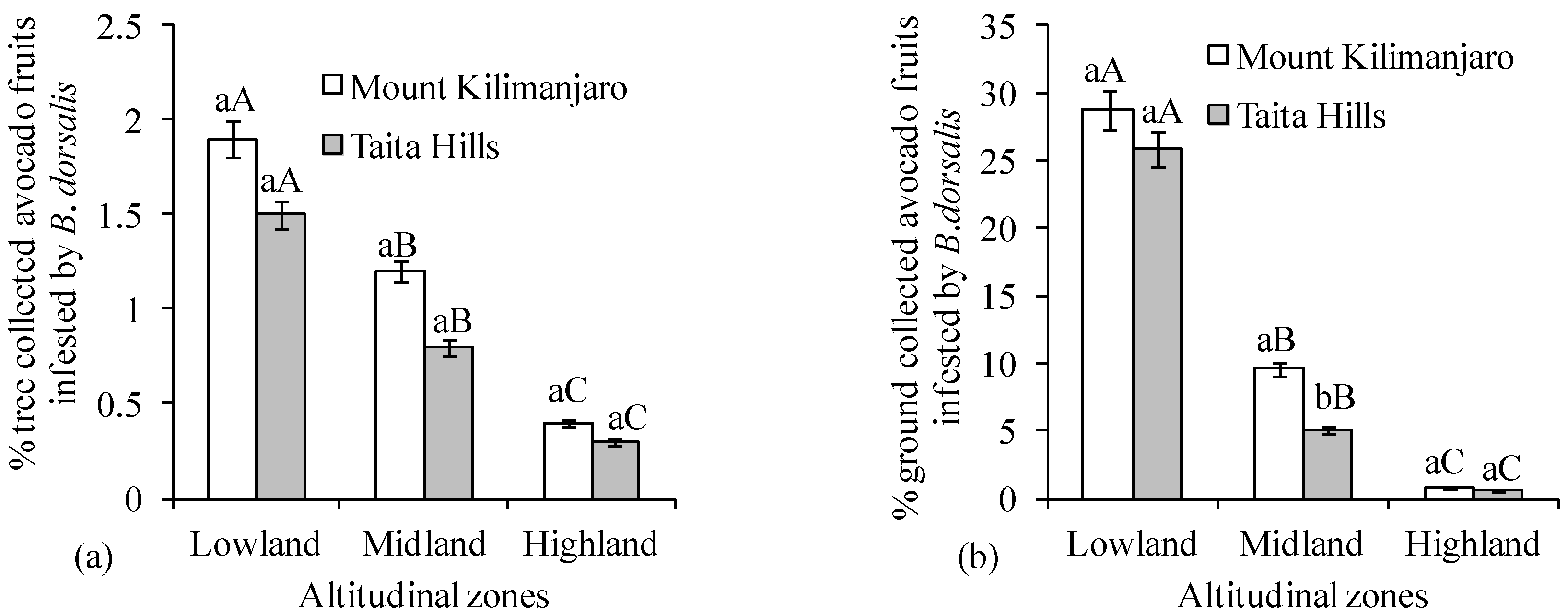
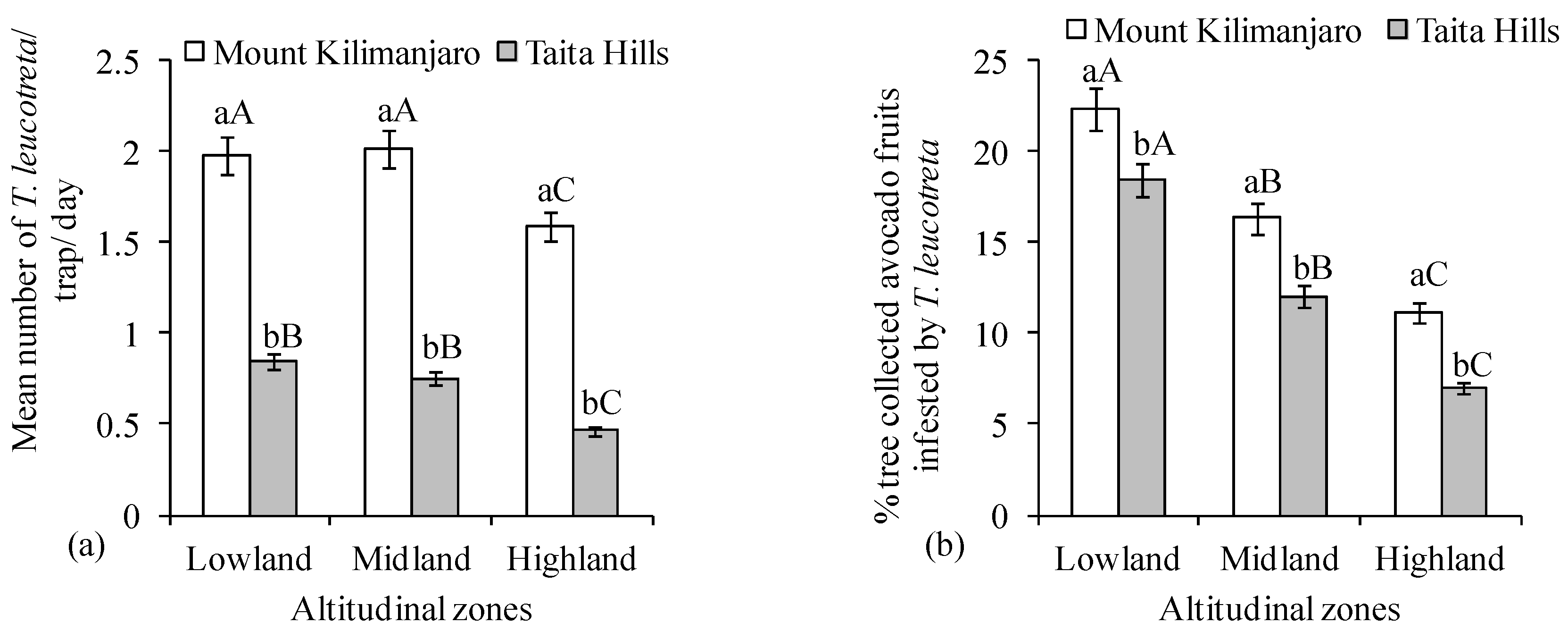
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bergh, B.O. The origin, nature and genetic improvement of avocado. Calif. Avocado Soc. Yearb. 1992, 76, 61–75. [Google Scholar]
- Knight, R.J. History, distribution, and uses. In The Avocado, Botany, Production, and Uses; Whiley, A.W., Schaffer, B., Wolstenholme, B.N., Eds.; CABI Publishing: Wallingford, UK, 2002. [Google Scholar]
- Wasilwa, L.A.; Njuguna, J.K.; Okoko, E.N.; Watani, G.W. Status of Avocado Production in Kenya; Kenya Agricultural Research Institute: Nairobi, Kenya, 2004. [Google Scholar]
- Griesbach, J. Avocado Growing in Kenya; World Agroforestry Centre (ICRAF), Kul Graphics Ltd.: Nairobi, Kenya, 2005. [Google Scholar]
- Chen, H.; Morrell, P.L.; Ashworth, V.E.T.M.; De La Cruz, M.; Clegg, M.T. Tracing the Geographic Origins of Major Avocado Cultivars. J. Hered. 2008, 100, 56–65. [Google Scholar] [CrossRef] [PubMed]
- FAO Statistical Yearbook. World Food and Agriculture; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013.
- Dreher, M.L.; Davenport, A.J. Hass Avocado Composition and Potential Health Effects. Clin. Rev. Food Sci. Nutr. 2013, 53, 738–750. [Google Scholar] [CrossRef] [PubMed]
- Weschenfelder, C.; dos Santos, J.L.; de Souza, P.A.L.; de Campos, V.P.; Marcadenti, A. Avocado and Cardiovascular Health. Open J. Endocr. Metab. Dis. 2015, 5, 77–83. [Google Scholar] [CrossRef]
- Wang, L.; Bordi, P.L.; Fleming, J.A.; Hill, A.M.; Kris-Etherton, P.M. Effect of a Moderate Fat Diet with and without Avocados on Lipoprotein Particle Number, Size and Subclasses in Overweight and Obese Adults: A Randomized, Controlled Trial. J. Am. Heart Assoc. 2015, 4, e001355. [Google Scholar] [CrossRef] [PubMed]
- Mwatawala, M.W.; De Meyer, M.; Makundi, R.H.; Maerere, A.P. Host range and distribution of fruit-infesting pestiferous fruit flies (Diptera, Tephritidae) in selected areas of Central Tanzania. Bull. Entomol. Res. 2009, 99, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Ware, A.R.; du Toit, C.L.N.; Mohamed, S.A.; Nderitu, P.W.; Ekesi, S. Cold tolerance and disinfestation of Bactrocera dorsalis (Diptera:Tephritidae) in ‘Hass’ avocado. J. Econ. Entomol. 2012, 105, 1963–1970. [Google Scholar] [CrossRef] [PubMed]
- Ekesi, S.; De Meyer, M.; Mohamed, S.A.; Virgilio, M.; Borgemeister, C. Taxonomy, ecology, and management of native and exotic fruit fly species in Africa. Annu. Rev. Entomol. 2016, 61, 219–238. [Google Scholar] [CrossRef] [PubMed]
- Schutze, M.K.; Aketarawong, N.; Amornsak, W.; Armstrong, K.F.; Augustinos, A.A.; Barr, N.; Bo, W.; Bourtzis, K.; Boykin, L.M.; Cáceres, C.; et al. Synonymization of key pest species within the Bactrocera dorsalis species complex (Diptera: Tephritidae): Taxonomic changes based on a review of 20 years of integrative morphological, molecular, cytogenetic, behavioural and chemoecological data. Syst. Entomol. 2015, 40, 456–471. [Google Scholar] [CrossRef]
- Schutze, M.K.; Mahmood, K.; Pavasovic, A.; Bo, W.; Newman, J.; Clarke, A.R.; Krosch, M.N.; Cameron, S.L. One and the same: Integrative taxonomic evidence that Bactrocera dorsalis (Diptera: Tephritidae) is the same species as the Oriental fruit fly Bactrocera dorsalis. Syst. Entomol. 2015, 40, 472–486. [Google Scholar] [CrossRef]
- Drew, R.A.I.; Tsuruta, K.; White, I.M. A new species of pest fruit fly (Diptera: Tephritidae: Dacinae) from Sri Lanka and Africa. Afr. Entomol. 2005, 13, 149–154. [Google Scholar]
- Dennill, G.B.; Erasmus, M.J. The insect pests of avocado fruits-increasing pest complex and changing pest status. J. Entomol. Soc. S. Afr. 1992, 55, 51–57. [Google Scholar]
- Whiley, A.W. Crop management. In Avocado: Botany, Production and Uses; Whiley, A.W., Schaffer, B., Wolstenholme, B.N., Eds.; CABI Publishing: Oxon, UK, 2002; pp. 231–258. [Google Scholar]
- CPC (Crop Protection Compendium). 2007. Available online: http://www.cabicompendium.org/cpc/home.asp (accessed on 24 January 2013).
- Prinsloo, G.L.; Uys, V.M. Insects of Cultivated Plants and Natural Pastures in Southern Africa; Entomological Society of Southern Africa: Hatfield, South Africa, 2015. [Google Scholar]
- Otieno, W. EPHIS experience with market access and compliance with official standards. Acta Hortic. 2011, 911, 73–76. [Google Scholar] [CrossRef]
- Ware, A.B.; du Toit, C.L.N.; du Toit, E.; Collins, R.; Clowes, R.; Ekesi, S.; Mohamed, S. Host suitability of three avocado cultivars (Persea americana Miller: Lauraceae) to oriental fruit fly (Bactrocera (invadens) dorsalis (Hendel) (Diptera: Tephritidae). Crop Prot. 2016, 90, 84–89. [Google Scholar] [CrossRef]
- Hemp, A. Continuum or zonation? Altitudinal gradients in the forest vegetation of Mt. Kilimanjaro. Plant Ecol. 2006, 184, 27–42. [Google Scholar] [CrossRef]
- Hemp, A. Vegetation of Kilimanjaro: Hidden endemics and missing bamboo. Afr. J. Ecol. 2006, 44, 305–328. [Google Scholar] [CrossRef]
- Aerts, R.; Thijs, K.W.; Lehouck, V.; Beentje, H.; Bytebier, B.; Matthysen, E.; Gulinck, H.; Lens, L.; Muys, B. Woody plant communities of isolated Afromontane cloud forests in Taita Hills, Kenya. Plant Ecol. 2011, 212, 639–649. [Google Scholar] [CrossRef]
- Ekesi, S.; Nderitu, P.W.; Rwomushana, I. Field infestation, life history and demographic parameters of Bactrocera invadens Drew, Tsuruta & White, a new invasive fruit fly species in Africa. Bull. Entomol. Res. 2006, 96, 379–386. [Google Scholar] [PubMed]
- Du Toit, W.J.; De Villiers, E.A.; Tuffin, A. The identification of typical surface lesions on avocado fruit. S. Afr. Avocado Grow. Assoc. Yearb. 1979, 3, 52–53. [Google Scholar]
- Crawley, M. Statistics, an Introduction to R; John Wiley & Sons Ltd.: West Sussex, UK, 2005. [Google Scholar]
- Verzani, J. Using R for Introductory Statistics; Chapman & Hall/CRC: New York, NY, USA, 2005. [Google Scholar]
- Crawley, M.J. The R Book; John Wiley and Sons, Ltd.: Chichester, UK, 2007. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2012; Available online: http://www.R-project.org/ (accessed on 27 March 2018).
- Akaike, H. On the likelihood of a time series model. Statistician 1978, 27, 217–219. [Google Scholar] [CrossRef]
- Akaike, H. Factor analysis and Akaike Information Criterion (AIC). Pyschometrika 1987, 52, 317–332. [Google Scholar] [CrossRef]
- Johnson, J.B.; Omland, K.S. Model selection in ecology and evolution. Trends Ecol. Evol. 2004, 19, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Burnham, K.P.; Anderson, D.R. Multimodel Inference: Understanding AIC and BIC in Model Selection. Sociol. Methods Res. 2004, 33, 261–307. [Google Scholar] [CrossRef]
- Liess, A.; Guo, J.; Lind, M.I.; Rowe, O. Cool tadpoles from Arctic environments waste fewer nutrients—High gross growth efficiencies lead to low consumer-mediated nutrient recycling in the North. J. Anim. Ecol. 2015, 84, 1744–1756. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.M.Y.; McInnis, D.O.; Mochizuki, N. Seasonal distribution and abundance of adult male oriental fruit flies (Diptera: Tephritidae) in Kula, Maui, Hawaii. J. Econ. Entomol. 1985, 78, 1267–1271. [Google Scholar] [CrossRef]
- Harris, E.J.; Takara, J.M.; Nishida, T. Distribution of the melon fly Dacus cucurbitae (Diptera: Tephritidae) and host plants on Kauai, Hawaiian Islands. Environ. Entomol. 1986, 15, 488–493. [Google Scholar] [CrossRef]
- Peña, J.E.; Mohyuddin, A.I.; Wysoki, M. A review of the pest management situation in mango agroecosystems. Phytoparasitica 1998, 26, 129–148. [Google Scholar] [CrossRef]
- Geurts, K.; Mwatawala, M.; De Meyer, M. Dominance of an invasive fruit fly species, Bactrocera invadens, along an altitudinal transect in Morogoro, Eastern Central Tanzania. Bull. Entomol. Res. 2014, 104, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Geurts, K.; Mwatawala, M.; De Meyer, M. Indigenous and invasive fruit fly diversity along an altitudinal transect in Eastern Central Tanzania. J. Insect Sci. 2012, 12, 12. [Google Scholar] [CrossRef] [PubMed]
- Mwatawala, M.W.; De Meyer, M.; Makundi, R.H.; Maerere, A.P. Seasonality and host utilization of the invasive fruit fly, Bactrocera invadens (Diptera: Tephritidae) in central Tanzania. J. Appl. Entomol. 2006, 130, 530–537. [Google Scholar] [CrossRef]
- Rwomushana, I.; Ekesi, S.; Gordon, I.; Ogol, C. Host plants and host plant preference studies for Bactrocera dorsalis (Diptera: Tephritidae) in Kenya, a new invasive fruit fly species in Africa. Ann. Entomol. Soc. Am. 2008, 101, 331–340. [Google Scholar] [CrossRef]
- Way, M.J.; Khoo, K.C. Role of ants in pest management. Annu. Rev. Entomol. 1992, 37, 479–503. [Google Scholar] [CrossRef]
- Van Mele, P.; Cuc, N.T.T. Ants as Friends: Improving Your Tree Crops with Weaver Ants; CABI Bioscience: Egham, UK, 2003. [Google Scholar]
- Van Mele, P.; Chien, H.V. Farmers, biodiversity and plant protection: Developing a learning environment for sustainable tree cropping systems. Int. J. Agric. Sustain. 2004, 2, 67–76. [Google Scholar] [CrossRef]
- Duyck, P.F.; David, P.; Junod, G.; Brunel, C.; Dupont, R.; Quilici, S. Importance of competition mechanisms in successive invasions by polyphagous tephritids in La Reunion. Ecology 2006, 87, 1770–1780. [Google Scholar] [CrossRef]
- Duyck, P.F.; David, P.; Quilici, S. Climatic niche partitioning following successive invasions by fruit flies in La Reunion. J. Anim. Ecol. 2006, 75, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Duyck, P.F.; David, P.; Quilici, S. Can more K-selected species be better invaders? A case study of fruit flies in La Reunion. Divers. Distrib. 2007, 13, 535–543. [Google Scholar] [CrossRef]
- Mohamed, S.A.; Overholt, W.A.; Wharton, R.A.; Lux, S.A. Effect of temperature on developmental time and longevity of Psyttalia cosyrae (Hymenoptera: Braconidae). Biocontrol Sci. Technol. 2006, 16, 717–726. [Google Scholar] [CrossRef]
- Mohamed, S.; Wharton, R.; Merey, G.V.; Schulthess, F. Acceptance and suitability of different host stages of Ceratitis capitata (Wiedemann) (Diptera: Tephritidae) and seven other tephritid fruit fly species to Tetrastichus giffardii Silvestri (Hymenoptera: Eulophidae). Biol. Control 2006, 39, 262–271. [Google Scholar] [CrossRef]
- Mohamed, S.A.; Ekesi, S.; Hanna, R. Evaluation of the impact of Diachasmimorpha longicaudata on Bactrocera dorsalis and five African fruit fly species. J. Appl. Entomol. 2008, 132, 789–797. [Google Scholar] [CrossRef]
- Mohamed, S.A.; Ekesi, S.; Hanna, R. Old and new host-parasitoid associations: Parasitism of the invasive fruit fly Bactrocera invadens (Diptera: Tephritidae) and five African fruit fly species by Fopius arisanus, an Asian opiine parasitoid. Biocontrol Sci. Technol. 2010, 20, 183–196. [Google Scholar] [CrossRef]
- Newton, P.J. False codling moth. In Citrus Pests in the Republic of South Africa, 2nd ed.; Bedford, E.C.G., van den Berg, M.A., de Villiers, E.A., Eds.; ARC—Institute for Tropical and Subtropical Crops and Outspan International: Nelspruit, South Africa, 1998; pp. 192–200. [Google Scholar]
- Stotter, R.L.; Terblanche, J.S. Low-temperature tolerance of false codling moth Thaumatotibia Leucotreta (Meyrick) (Lepidoptera: Tortricidae) in South Africa. J. Therm. Biol. 2009, 34, 320–325. [Google Scholar] [CrossRef]
- Stotter, R.L. Spatial and Temporal Distribution of False Codling Moth across Landscapes in the Citrusdal Area. Master’s Thesis, University of Stellenbosch, Western Cape Province, South Africa, 2009; 66p. [Google Scholar]
- Boardman, L.; Grout, T.G.; Terblanche, J.S. False codling moth (Thaumatotibia leucotreta) (Lepidoptera, Tortricidae) larvae are chill-susceptible. Insect Sci. 2012, 19, 315–328. [Google Scholar] [CrossRef]
- Carpenter, J.E.; Bloem, S.; Hofmeyr, J.H. Acceptability and suitability of eggs of false codling moth (Lepidoptera: Tortricidae) from irradiated parents to parasitism by Trichogrammatoidea cryptophlebiae (Hymenoptera: Trichogrammatidae). Biol. Control 2004, 30, 351–359. [Google Scholar] [CrossRef]
| Elevation Range (m.a.s.l.) | Agro-Ecological Zone | Mean Elevation (m.a.s.l.) | Mean Temperature (°C) | Relative Humidity (%) | Mean Rainfall (mm) | Peak Avocado Harvesting Months |
|---|---|---|---|---|---|---|
| 1500–1799 | Highland | 1687.0 | 17.6 | 84.4 | 218.7 | June & July |
| 1200–1499 | Midland | 1381.4 | 19.9 | 81.1 | 93.6 | May & June |
| 900–1199 | Lowland | 1085.7 | 22.0 | 75.2 | 52.2 | April & May |
| a: Models for Taita Hills | AIC | R2 | p-Value | Rank |
| Bactrocera dorsalis mean density vs. interaction term (mean temperature by agro-ecological zones) was the best model | 1176.4 | 0.588 | <0.0001 | 1 |
| Bactrocera dorsalis mean density vs. mean temperature, mean humidity, rainfall and agro-ecological zones | 1185.3 | 0.578 | <0.0001 | 2 |
| Bactrocera dorsalis mean density vs. mean temperature, mean humidity, rainfall, elevation, distance and agro-ecological zones | 1188.4 | 0.575 | <0.0001 | 3 |
| Bactrocera dorsalis mean density vs. mean temperature, mean humidity and agro-ecological zones | 1190.8 | 0.563 | <0.0001 | 4 |
| Bactrocera dorsalis mean density vs. mean temperature and habitats without interaction term (mean temperature by agro-ecological zones) | 1200.8 | 0.535 | <0.0001 | 5 |
| Bactrocera dorsalis mean density vs. mean humidity and agro-ecological zones | 1223.0 | 0.475 | <0.0001 | 6 |
| b: Models for Mount Kilimanjaro | AIC | R² | p -Value | Rank |
| Bactrocera dorsalis mean density vs. interaction term (mean temperature by agro-ecological zones) was the best model | 1455.2 | 0.534 | <0.0001 | 1 |
| Bactrocera dorsalis mean density vs. mean temperature and habitats without interaction term (mean temperature by agro-ecological zones) | 1485.8 | 0.441 | <0.0001 | 2 |
| Bactrocera dorsalis mean density vs. mean temperature, mean humidity and agro-ecological zones | 1487.8 | 0.438 | <0.0001 | 3 |
| Bactrocera dorsalis mean density vs. mean temperature, mean humidity, rainfall and agro-ecological zones | 1488.1 | 0.440 | <0.0001 | 4 |
| Bactrocera dorsalis mean density vs. mean temperature, mean humidity, rainfall, elevation, distance and agro-ecological zones | 1490.0 | 0.439 | <0.0001 | 5 |
| Bactrocera dorsalis mean density vs. mean humidity and agro-ecological zones | 1519.0 | 0.328 | <0.0001 | 6 |
| a: Models for Taita Hills | AIC | R2 | p-Value | Rank |
| Thaumatotibia leucotreta mean density vs. mean temperature, mean humidity, rainfall, distance and elevation was the best model | −3.98 | 0.2112 | <0.0001 | 1 |
| Thaumatotibia leucotreta mean density vs. mean temperature, mean humidity, rainfall, distance and agro-ecological zones | −2.24 | 0.1993 | <0.0001 | 2 |
| Thaumatotibia leucotreta mean density vs. mean temperature, mean humidity, rainfall and agro-ecological zones | 12.08 | 0.0899 | 0.0007 | 3 |
| Thaumatotibia leucotreta mean density vs. mean temperature | 15.65 | 0.0610 | 0.0004 | 4 |
| Thaumatotibia leucotreta mean density vs. mean humidity and agro-ecological zones | 24.92 | 0.0193 | 0.0925 | 5 |
| Thaumatotibia leucotreta mean density vs. distance and agro-ecological zones | 26.53 | 0.0234 | 0.0925 | 6 |
| b: Models for Mount Kilimanjaro | AIC | R2 | p-Value | Rank |
| Thaumatotibia leucotreta mean density vs. mean temperature, mean humidity, rainfall and elevation was the best model | 453.37 | 0.4087 | <0.0001 | 1 |
| Thaumatotibia leucotreta mean density vs. mean temperature and habitats without interaction term (mean temperature by agro-ecological zones) | 461.77 | 0.3402 | <0.0001 | 2 |
| Thaumatotibia leucotreta mean density vs. mean temperature, mean humidity and agro-ecological zones | 463.65 | 0.3389 | <0.0001 | 3 |
| Thaumatotibia leucotreta mean density vs. mean temperature, mean humidity, rainfall and agro-ecological zones | 464.46 | 0.3353 | <0.0001 | 4 |
| Thaumatotibia leucotreta mean density vs. mean temperature excluding agro-ecological zones | 468.90 | 0,2795 | <0.0001 | 5 |
| Thaumatotibia leucotreta mean density vs. mean humidity and agro-ecological zones | 503.04 | 0.2098 | <0.0001 | 6 |
| Thaumatotibia leucotreta mean density vs. distance and agro-ecological zones | 541.60 | 0.2097 | 0.0813 | 7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Odanga, J.J.; Mohamed, S.; Mwalusepo, S.; Olubayo, F.; Nyankanga, R.; Khamis, F.; Rwomushana, I.; Johansson, T.; Ekesi, S. Spatial Distribution of Bactrocera dorsalis and Thaumatotibia leucotreta in Smallholder Avocado Orchards along Altitudinal Gradient of Taita Hills and Mount Kilimanjaro. Insects 2018, 9, 71. https://doi.org/10.3390/insects9020071
Odanga JJ, Mohamed S, Mwalusepo S, Olubayo F, Nyankanga R, Khamis F, Rwomushana I, Johansson T, Ekesi S. Spatial Distribution of Bactrocera dorsalis and Thaumatotibia leucotreta in Smallholder Avocado Orchards along Altitudinal Gradient of Taita Hills and Mount Kilimanjaro. Insects. 2018; 9(2):71. https://doi.org/10.3390/insects9020071
Chicago/Turabian StyleOdanga, James J., Samira Mohamed, Sizah Mwalusepo, Florence Olubayo, Richard Nyankanga, Fathiya Khamis, Ivan Rwomushana, Tino Johansson, and Sunday Ekesi. 2018. "Spatial Distribution of Bactrocera dorsalis and Thaumatotibia leucotreta in Smallholder Avocado Orchards along Altitudinal Gradient of Taita Hills and Mount Kilimanjaro" Insects 9, no. 2: 71. https://doi.org/10.3390/insects9020071
APA StyleOdanga, J. J., Mohamed, S., Mwalusepo, S., Olubayo, F., Nyankanga, R., Khamis, F., Rwomushana, I., Johansson, T., & Ekesi, S. (2018). Spatial Distribution of Bactrocera dorsalis and Thaumatotibia leucotreta in Smallholder Avocado Orchards along Altitudinal Gradient of Taita Hills and Mount Kilimanjaro. Insects, 9(2), 71. https://doi.org/10.3390/insects9020071






