Host-Specific Activation of Entomopathogenic Nematode Infective Juveniles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Nematode Production and Virulence Experiments
2.2. Insects
2.3. Making Homogenate from Various Insects
2.4. Activation of IJs and Quantification
2.5. Statistical Analysis
3. Results
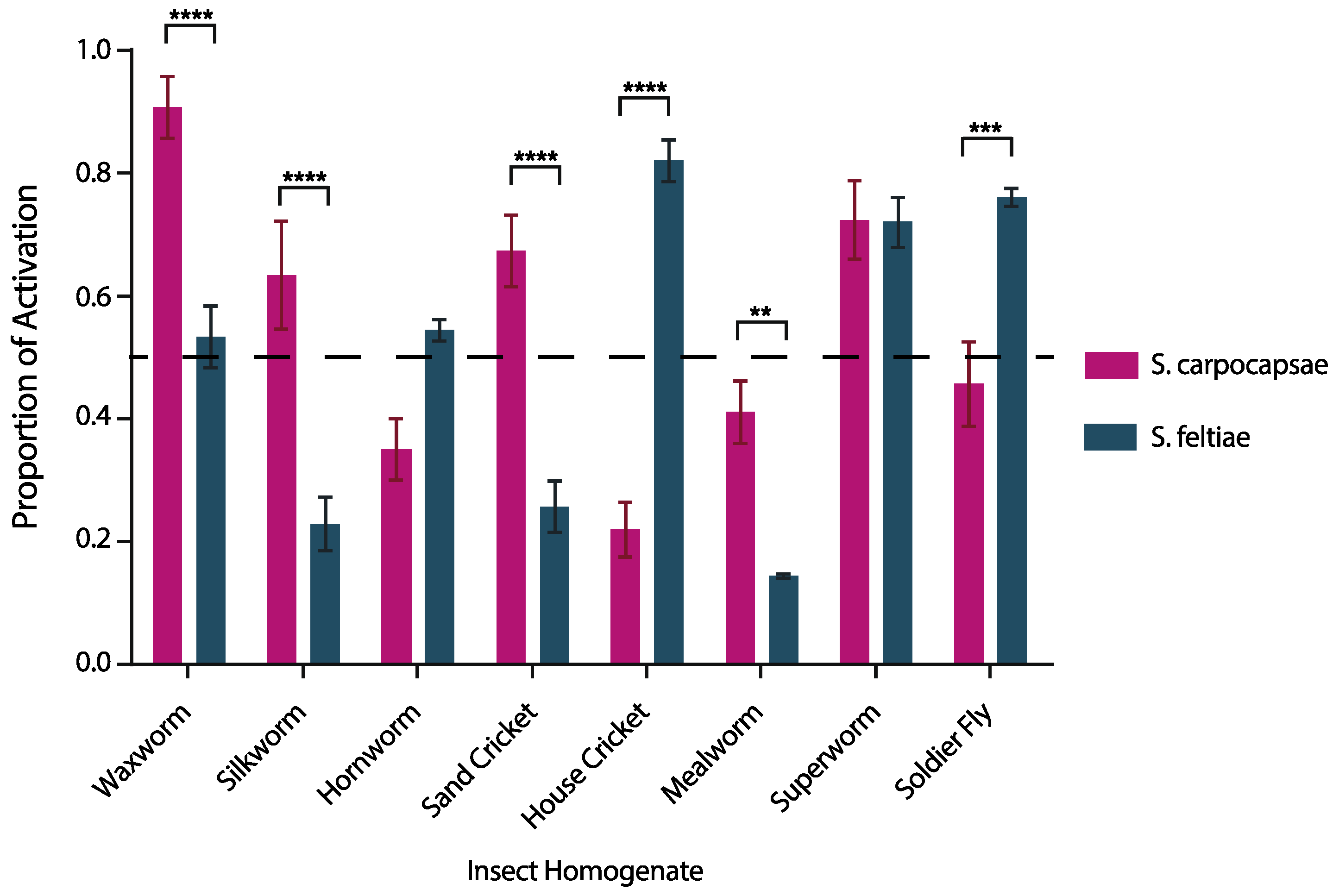
3.1. IJ Activation is Species Specific and Host Specific
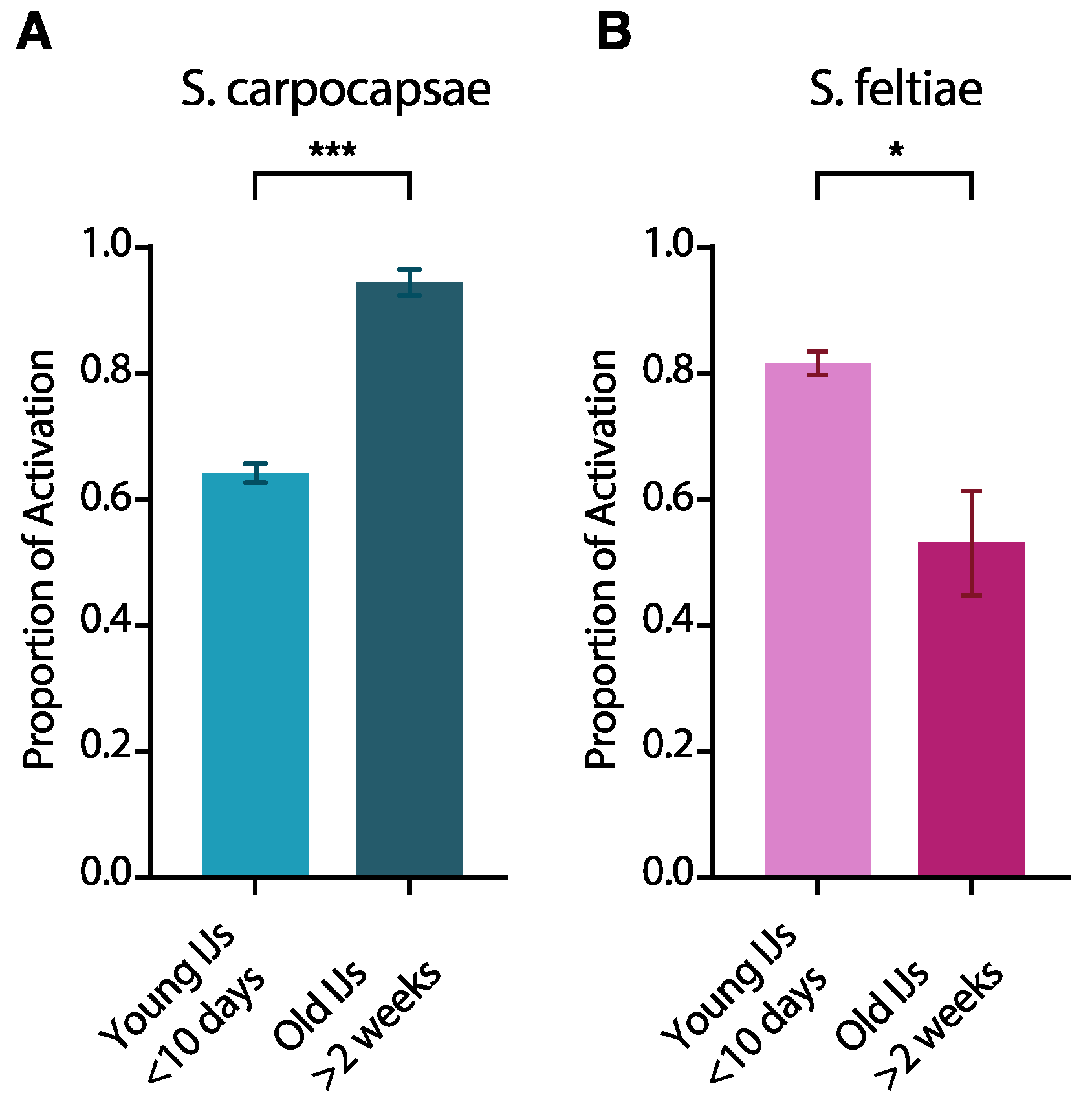
3.2. IJ Activation Is Affected by IJ Age
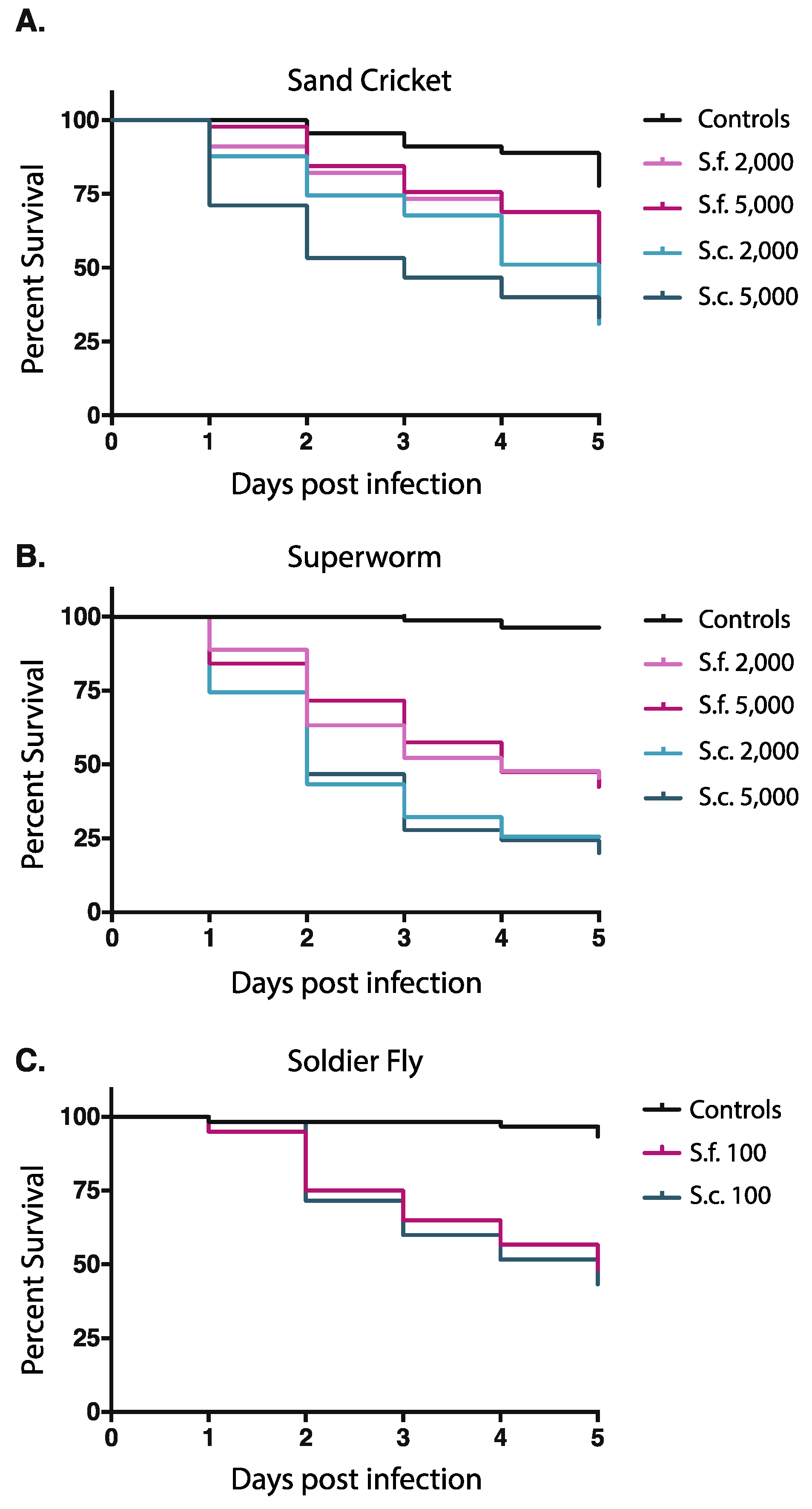
3.3. In Vitro IJ Activation Alone Is not Always Predictive of Virulence
4. Discussion
4.1. The Activation Bioassay Illustrated Species-Dependent Nematode Activation among a Range of Hosts
4.2. Age Affects IJ Activation
4.3. In Vitro Activation alone May Be Predictive of Virulence in Some Cases
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Source of Variation | % of Total Variation | pValue | Summary | ||
| Interaction | 55.57 | <0.0001 | **** | ||
| Row Factor | 35.84 | <0.001 | **** | ||
| Column Factor | 0.9130 | 0.0671 | ns | ||
| ANOVA table | SS | DF | MS | F (DFn, DFd) | pvalue |
| Interaction | 1.515 | 7 | 0.2164 | F (7, 31) = 31.31 | p < 0.001 |
| Row Factor | 0.9772 | 7 | 0.1396 | F (7, 31) = 20.20 | p < 0.001 |
| Column Factor | 0.02489 | 1 | 0.02489 | F (1, 31) = 3.601 | p = 0.0671 |
| Residual | 0.2143 | 31 | 0.006912 |
References
- Kaya, H.K.; Gaugler, R. Entomopathogenic nematodes. Annu. Rev. Entomol. 1993, 38, 181–206. [Google Scholar] [CrossRef]
- Dillman, A.R.; Chaston, J.M.; Adams, B.J.; Ciche, T.A.; Goodrich-Blair, H.; Stock, S.P.; Sternberg, P.W. An entomopathogenic nematode by any other name. PLoS Pathog. 2012, 8, e1002527. [Google Scholar] [CrossRef] [PubMed]
- Gaugler, R.; Kaya, H.K. Entomopathogenic Nematodes in Biological Control; CRC Press: Boca Raton, FL, USA, 1990; p. 365. [Google Scholar]
- Lacey, L.A.; Georgis, R. Entomopathogenic nematodes for control of insect pests above and below ground with comments on commercial production. J. Nematol. 2012, 44, 218–225. [Google Scholar] [PubMed]
- Hallem, E.A.; Dillman, A.R.; Hong, A.V.; Zhang, Y.; Yano, J.M.; DeMarco, S.F.; Sternberg, P.W. A sensory code for host seeking in parasitic nematodes. Curr. Biol. 2011, 21, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Dillman, A.R.; Guillermin, M.L.; Lee, J.H.; Kim, B.; Sternberg, P.W.; Hallem, E.A. Olfaction shapes host-parasite interactions in parasitic nematodes. Proc. Natl. Acad. Sci. USA 2012, 109, E2324–E2333. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Macchietto, M.; Chang, D.; Barros, M.M.; Baldwin, J.; Mortazavi, A.; Dillman, A.R. Activated entomopathogenic nematode infective juveniles release lethal venom proteins. PLoS Pathog. 2017, 13, e1006302. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.E.; Grewal, P.S.; Gauger, R. Hierarchical order of host cues in parasite foraging strategies. Parasitology 1995, 110, 207–213. [Google Scholar] [CrossRef]
- Campbell, J.F.; Lewis, E.E. Entomopathogenic nematode host-search strategies. In The Behavioral Ecology of Parasites; Campbell, J.F., Sukhdeo, M.V.K., Eds.; CABI: New York, NY, USA, 2002; pp. 13–38. [Google Scholar]
- Griffin, C.T. Perspectives on the behavior of entomopathogenic nematodes from dispersal to reproduction: Traits contributing to nematode fitness and biocontrol efficacy. J. Nematol. 2012, 44, 177–184. [Google Scholar] [PubMed]
- Lu, D.; Sepulveda, C.; Dillman, A.R. Infective juveniles of the entomopathogenic nematode Steinernema scapterisci are preferentially activated by cricket tissue. PLoS ONE 2017, 12, e0169410. [Google Scholar] [CrossRef] [PubMed]
- Adams, B.J.; Peat, S.M.; Dillman, A.R. Phylogeny and evolution. In Entomopathogenic Nematodes: Systematics, Phylogeny, and Bacterial Symbionts; Nguyen, K.B., Hunt, D.J., Eds.; Brill: Boston, MA, USA, 2007; Volume 5, pp. 693–733. [Google Scholar]
- Lewis, E.E.; Clarke, D.J. Nematode parasites and entomopathogens. In Insect Pathology, 2nd ed.; Vega, F.E., Kaya, H.K., Eds.; Elsevier: New York, NY, USA, 2012; pp. 395–424. [Google Scholar]
- Nguyen, K. Species of Steinernema. Entomology and Nematology Department, University of Florida Updated March 13. , 2017. Available online: Entnem.ifas.ufl.edu/nguyen/morph/steinsp1.htm (accessed on 20 May 2018).
- White, G.F. A method for obtaining infective nematode larvae from cultures. Science 1927, 66, 302–303. [Google Scholar] [CrossRef] [PubMed]
- McMullen, J.G.; Stock, S.P. In vivo and in vitro rearing of entomopathogenic nematodes (Steinernematidae and Heterorhabditidae). Jove J. Vis. Exp. 2014, e52096. [Google Scholar] [CrossRef]
- Roff, D.A. The genetic basis of wing dimorphism in the sand cricket, Gryllus firmus and its relevance to the evolution of wing dimorphisms in insects. Heredity 1986, 57, 221–231. [Google Scholar] [CrossRef]
- Snyder, H.; Stock, S.P.; Kim, S.K.; Flores-Lara, Y.; Forst, S. New insights into the colonization and release processes of Xenorhabdus nematophila and the morphology and ultrastructure of the bacterial receptacle of its nematode host, Steinernema carpocapsae. Appl. Environ. Microbiol. 2007, 73, 5338–5346. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.E.J.; Campbell, C.; Griffin, H.; Kaya, A. Peters. Behavioral ecology of entomopathogenic nematodes. Biol. Control 2006, 38, 66–79. [Google Scholar] [CrossRef]
- Ogier, J.C.; Pages, S.; Bisch, G.; Chiapello, H.; Medigue, C.; Rouy, Z.; Teyssier, C.; Vincent, S.; Tailliez, P.; Givaudan, A.; et al. Attenuated virulence and genomic reductive evolution in the entomopathogenic bacterial symbiont species, Xenorhabdus poinarii. Genome Biol. Evol. 2014, 6, 1495–1513. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.H.; Aryal, S.K.; Aghai, D.T.; Casanova-Torres, A.M.; Hillman, K.; Kozuch, M.P.; Mans, E.J.; Mauer, T.J.; Ogier, J.C.; Ensign, J.C.; et al. The insect pathogenic bacterium Xenorhabdus innexi has attenuated virulence in multiple insect model hosts yet encodes a potent mosquitocidal toxin. BMC Genom. 2017, 18, 927. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, N.; Hao, Y.J.; Toubarro, D.; Nascimento, G.; Simoes, N. Purification, biochemical and molecular analysis of a chymotrypsin protease with prophenoloxidase suppression activity from the entomopathogenic nematode Steinernema carpocapsae. Int. J. Parasit. 2009, 39, 975–984. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, N.; Toubarro, D.; Simoes, N. Biochemical study and in vitro insect immune suppression by a trypsin-like secreted protease from the nematode Steinernema carpocapsae. Parasite Immunol. 2010, 32, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.J.; Montiel, R.; Nascimento, G.; Toubarro, D.; Simoes, N. Identification and expression analysis of the Steinernema carpocapsae elastase-like serine protease gene during the parasitic stage. Exp. Parasitol. 2009, 122, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Yoder, C.A.; Grewal, R.S.; Taylor, R.A.J. Rapid age-related changes in infection behavior entomopathogenic nematodes. J. Parasitol. 2004, 90, 1229–1234. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Dillman, A.R.; Hallem, E.A. Temperature-dependent changes in the host-seeking behaviors of parasitic nematodes. BMC Biol. 2016, 14, 36. [Google Scholar] [CrossRef] [PubMed]
- Bohan, D.A.; Hominick, W.M. Investigations on the presence of an infectious proportion amongst populations of Steinernema feltiae (Site 76 strain) infective stages. Parasitology 1996, 112, 113–118. [Google Scholar] [CrossRef]
- Bohan, D.A.; Hominick, W.M. Long-term dynamics of infectiousness within the infective-stage pool of the entomopathogenic nematode Steinernema feltiae (site 76 strain) Filipjev. Parasitology 1997, 114, 301–308. [Google Scholar] [CrossRef]
- Fushing, H.; Zhu, L.; Shapiro-Ilan, D.I.; Campbell, J.F.; Lewis, E.E. State-space based mass event-history model I: Many decision-making agents with one target. Ann. Appl. Stat. 2008, 2, 1503–1522. [Google Scholar] [CrossRef] [PubMed]
- Bisch, G.; Pages, S.; McMullen, J.G.; Stock, S.P.; Duvic, B.; Givaudan, A.; Gaudriault, S. Xenorhabdus bovienii CS03, the bacterial symbiont of the entomopathogenic nematode Steinernema weiseri, is a non-virulent strain against lepidopteran insects. J. Invertebr. Pathol. 2015, 124, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Veesenmeyer, J.L.; Andersen, A.W.; Lu, X.J.; Hussa, E.A.; Murfin, K.E.; Chaston, J.M.; Dillman, A.R.; Wassarman, K.M.; Sternberg, P.W.; Goodrich-Blair, H. NilD CRISPR RNA contributes to Xenorhabdus nematophila colonization of symbiotic host nematodes. Mol. Microbiol. 2014, 93, 1026–1042. [Google Scholar] [CrossRef] [PubMed]
- Batalla-Carrera, L.; Morton, A.; Shapiro-Ilan, D.; Strand, M.R.; Garcia-del-Pino, F. Infectivity of Steinernema carpocapsae and S. feltiae to larvae and adults of the hazelnut weevil, Curculio nucum: Differential virulence and entry routes. J. Nematol. 2014, 46, 281–286. [Google Scholar] [PubMed]
- Ricci, M.; Glazer, I.; Campbell, J.F.; Gaugler, R. Comparison of bioassays to measure virulence of different entomopathogenic nematodes. Biocontrol. Sci. Technol. 1996, 6, 235–245. [Google Scholar] [CrossRef]
- Campos-Herrera, R.; Gutierrez, C. Steinernema feltiae intraspecific variability: Infection dynamics and sex-ratio. J. Nematol. 2014, 46, 35–43. [Google Scholar] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alonso, V.; Nasrolahi, S.; Dillman, A.R. Host-Specific Activation of Entomopathogenic Nematode Infective Juveniles. Insects 2018, 9, 59. https://doi.org/10.3390/insects9020059
Alonso V, Nasrolahi S, Dillman AR. Host-Specific Activation of Entomopathogenic Nematode Infective Juveniles. Insects. 2018; 9(2):59. https://doi.org/10.3390/insects9020059
Chicago/Turabian StyleAlonso, Valentina, Shyon Nasrolahi, and Adler R. Dillman. 2018. "Host-Specific Activation of Entomopathogenic Nematode Infective Juveniles" Insects 9, no. 2: 59. https://doi.org/10.3390/insects9020059
APA StyleAlonso, V., Nasrolahi, S., & Dillman, A. R. (2018). Host-Specific Activation of Entomopathogenic Nematode Infective Juveniles. Insects, 9(2), 59. https://doi.org/10.3390/insects9020059






