1. Introduction
The rate of biological invasions globally has increased dramatically in the past 500 years due to an increase in human activities such as transportation, migration, and commerce [
1]. Invasive species cause direct and indirect effects on organisms living in the environment they invade, and therefore threaten biodiversity, agriculture, and human health. Besides the environmental impacts, invasive species cause major economic losses in different sectors of the U.S. economy [
2]. The practice of classical biological control (CBC), as the intentional transfer of natural enemies from one place to another, has traditionally been used as a tool to fight invasive species in agricultural settings and is now also being used to control invasive species in natural areas [
3,
4].
The enemy release hypothesis states that organisms become invasive in a new area because they have escaped the natural enemies that suppress their populations in their area of origin. Exotic species thus have an advantage over competitors in areas of introduction where indigenous species are still suppressed by their indigenous natural enemies [
5]. Therefore, CBC works under the premise that the reestablishment of top-down control by introduction of natural enemies will reduce the populations of invasive species and therefore restore balance [
3].
The history of biological control provides many examples of remarkable successes [
6]. Prime examples include the introduction of the Australian lady beetle
Rodolia cardinalis (Mulsant) (Coleoptera: Coccinellidae) to control the cottony cushion scale,
Icerya purchasi Maskell, an introduction that is credited with saving the California citrus industry, and the introduction of
Anagyrus lopezi (DeSantis) (Hymenoptera: Encyrtidae) from South America to control the cassava mealybug
Phenacoccus manihoti Matile-Ferrero, in Africa, recognized to have saved many people from starvation. Those are just two of many other remarkable examples; Van Driesche et al. [
7] provide a comprehensive review of the benefits of biological control of pests. Besides the economic benefits of this practice, the use of biological control has also led to a reduction in the use and dependence on pesticides, with coincidental benefits in terms of human and environmental health.
The potential and realized positive effects of biological control have been recognized for over a century. Many practitioners have long considered this practice environmentally safe, benign, risk-free, and a natural phenomenon [
8,
9,
10,
11]. Even though awareness of potential negative effects was also expressed over a century ago [
12], it was only in the 1980s that classical biological control was first severely criticized as posing serious environmental risks [
13,
14]. At the center of this criticism was the issue of host specificity, or the lack thereof [
15]. Biological control agents were implicated in the reduction of populations of native and desirable species, and were blamed in some cases to be agents of extinction [
13,
14]. Soon after, regulators and researchers were calling for more rigorous screening methods in the USA [
15] and revisiting means of predicting positive or negative impacts of biocontrol agents. Some authors went so far as to call for the cessation of biological control [
16].
Throughout the history of biological control, the most adverse effects in terms of non-target attacks have been due to the release of highly polyphagous species [
17,
18]. Generalist biological control agents were at one point considered superior not only because they could potentially control several pests but they could also persist on native insects at times when the target pest was rare. Such releases of generalist species were made before the concerns about environmental impacts were appreciated. In the specific case of Hawaii, after concerns were raised the state experienced an overall reduction of biological control introductions due to the implementation of rigid regulations [
19].
At the time the potential for environmental impacts of introduced biological control agents was recognized in the USA, the need for regulations and guidelines for introductions as well as for comprehensive risk assessment (RA) frameworks became apparent.
Several protocols have been developed for the selection of non-target species for screening and host range determination [
20,
21,
22,
23,
24,
25]. Some countries such as New Zealand, Australia and South Africa as well as countries within the European Union have developed their own regulations and RA frameworks. For the most part they have similar criteria, but they involve different procedures and work under different guidelines.
The United States has no comprehensive RA methodology adopted for insect biological control introductions, and this situation is further complicated by a range of regulations at the state level. Hawaii represents a unique case, with a long history of biological control introductions, and has been the center of controversy regarding non-target effects. This has resulted in overly-restrictive regulations being implemented there [
19]. The state of Hawaii and the USA in general should greatly benefit from the adoption of a general quantitative RA framework to characterize the potential risks of proposed biological control introductions.
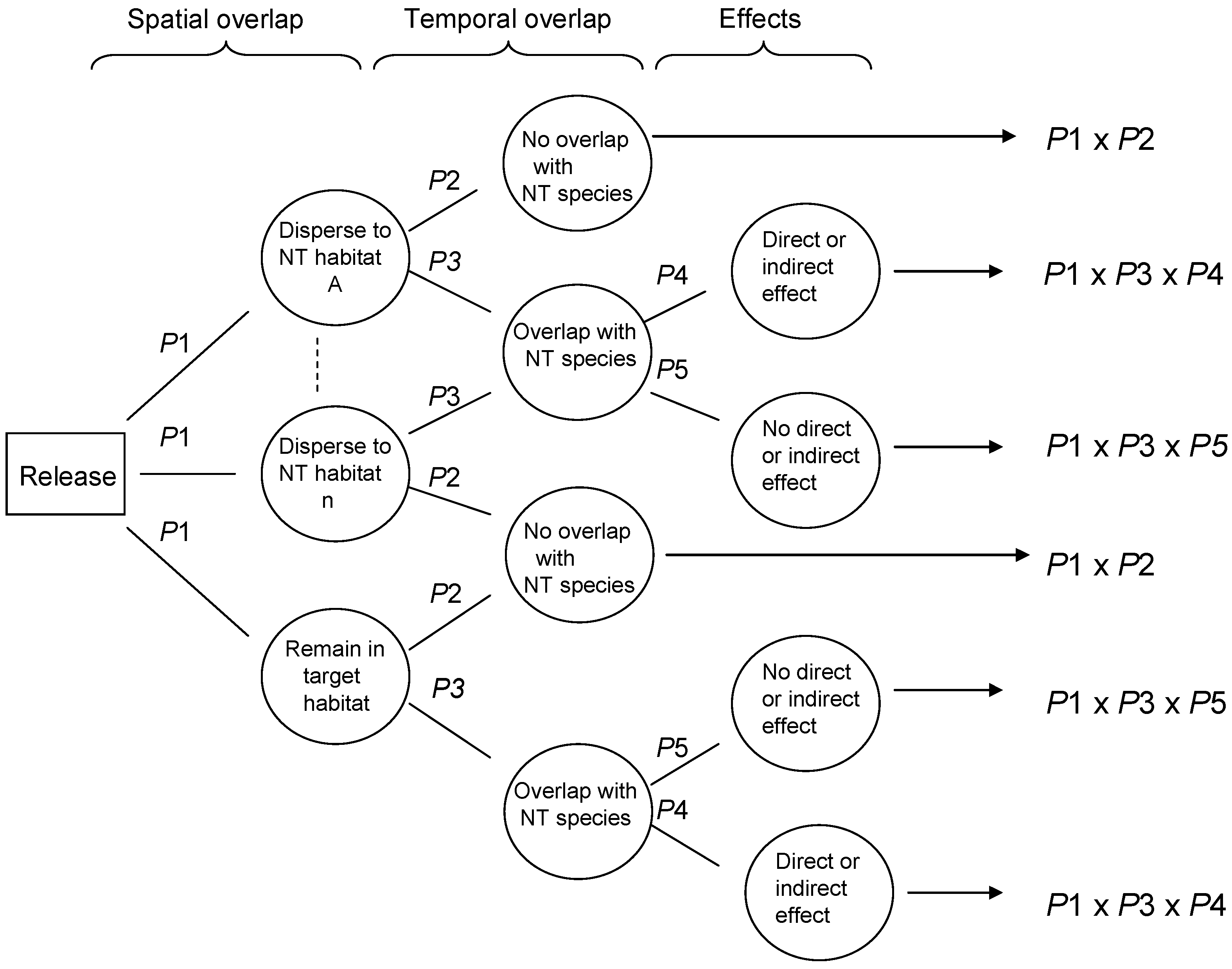
Risk assessment in the field of biological control evaluates the likelihood that adverse ecological effects may occur, and the expected magnitude of any adverse effects, as a result of a release of a purposely introduced biological control agent. Two general RA frameworks have been proposed with great potential to be widely adopted. van Lenteren et al. [
24] proposed a semi-quantitative environmental RA approach for inundative biological control agents, which was later improved and expanded to address classical biological control agents, in a stepwise procedure that identifies biological control agents with high potential non-target risk early in the process, therefore avoiding unnecessary research and use of resources [
25,
26]. Wright et al. [
27] proposed a probabilistic RA (PRA) approach for either classical or inundative biocontrol agents, using conditional probabilities in a Bayesian approach to estimating risk, illustrated conceptually in
Figure 1.
It has already been shown that the risk posed by biological control agents can vary spatially and temporally within the area of introduction [
28,
29,
30,
31,
32]. Nevertheless, current RA procedures lack comprehensive incorporation of spatial and temporal components to characterize the risk. In attempting to predict how a biological control agent will respond to potential new hosts in a new area of introduction, it is important to elucidate its behavior in the area of origin and other areas of distribution to predict the nature of potential relationships with target species and potential non-target species.
The aim of this paper is to validate the probabilistic risk assessment methodology proposed by Wright et al. [
27] using retrospective and current data from a study system in Hawaii. Wright et al. [
27] proposed that non-target risk could be assessed using decision trees, which estimate the conditional probabilities of certain dependent events occurring, in this case non-target exploitation. The proposed approach included probabilities associated with various parasitoid behaviors, to conduct risk assessment for biological control agents, incorporating host range, ecological and behavioral aspects of parasitoid ecology. The probability of parasitism occurring at various levels may be modeled and incorporated into assessments that predict the ‘worst case’ (high probability of high parasitism) vs. the ‘best case’ (low probability of any non-target parasitism), through constructing probability distributions for each ecological aspect, using field collected data. The validation process addresses the three questions below using data from recently published work addressing the impacts of purposefully introduced and accidentally introduced parasitoids on an endemic Hawaiian moth,
Udea stellata (Crambidae) [
33,
34,
35,
36].
Udea stellata is a moderately abundant species specific to an endemic plant, which occurs across a fairly broad ecological gradient in Hawaii, under a range of environmental conditions. It is considered to be a significant source of food for certain Hawaiian birds. We selected
U. stellata as a study species as a representative of a typical Hawaiian insect, rather than one that was threatened, so that the impacts of introduced insects could be assessed, rather than other impacts such as habitat loss. Citations 33–36 report the results of studies on parasitoids attacking
U. stellata in Hawaii, quantifying the parasitoid assemblage, levels of mortality (both apparent mortality, measuring proportion of hosts parasitized by a parasitoid in random samples; and marginal mortality, estimated generational mortality inflicted by a parasitoid, in the presence of other mortality factors), and ecological correlates of parasitoid distributions.
Key questions we addressed were: Would it have been possible to predict that Cotesia marginiventris (Braconidae), Meteorus laphygmae (Braconidae), and Trathala flavoorbitalis (Ichneumonidae) would attack U. stellata based on earlier published data, and would it have been possible to predict their level of impact on this non-target species using PRA? How useful are estimates of marginal vs. apparent mortality (and measures of uncertainty in both) in conducting PRA? Are there any key ecological variables that would be worth considering in the hypothetical case that any of the species addressed will be considered for introduction in another place?
1.1. Validation of the Probabilistic Risk Assessment Methodology
This section presents the procedures used to answer the four main questions stated in the introduction, regarding the validation of a PRA approach applied to biological control agents. A summary overview of risk assessment methods is provided in
Supplementary Materials.
Question 1: Would it have been possible to predict that any of the three introduced parasitoid species would attack U. stellata based on published historical data, and would it have been possible to predict their level of impact on this non-target species using data gleaned from the published literature using PRA?
To address the first part of the question, whether older published records would have given an indication that any of the three introduced parasitoid species would attack
U. stellata, reports on parasitism by
C. marginiventris,
M. laphygmae, and
T. flavoorbitalis were sought in older published records (from 1913 to date of introduction or first record) by consulting printed copies of the abstract journal
Review of Applied Entomology. The Thompson Catalogues of Host-parasitoid Associations [
37,
38] were also consulted (similar procedure described by [
39,
40]).
The second part of the question, whether it would have been possible to predict their level of impact on
U. stellata based on historical data using PRA, validates a probabilistic risk assessment approach [
27] with precision trees. As far as possible, probabilistic methods were applied by deriving probabilities of parasitism under different circumstances, from available data that could be gathered on the biology and ecology of the species being examined, from the literature as if it would have been available prior to the ‘introduction’ of the species of interest. These data form the basis for predictions (hypotheses) available at the ‘time of introduction’ and provide the first step for the validation. As actual non-target impacts were measured during the course of a number of studies supporting this work [
33,
34,
35,
36], it should be possible to corroborate the probabilistic model “predictions” with contemporary quantitative field data (presented as probability distributions). The validation was carried out by developing precision trees based on published data from earlier studies, compared to current actual impacts measured in the field.
The parasitoid species used for the validation section were C. marginiventris, M. laphygmae, and T. flavoorbitalis, with U. stellata as the non-target organism. Validation was not performed for the other species that parasitize U. stellata, Diadegma blackburnii, Pristomerus hawaiiensis, Casinaria infesta and Triclistus nr. Aitkeni, since little information was available in the literature on the latter four.
1.2. Problem Formulation
1.2.1. The Non-Target Host Udea stellata (Butler)
The genus
Udea (Lepidoptera: Crambidae) is a very large group that occurs in the Americas, Eurasia, and into the Pacific [
41]. Hawaii has 44 endemic species in this genus [
42].
Udea stellata was first described by Butler in 1883.
Udea stellata (Lepidoptera: Crambidae) is a multivoltine species that undergoes six larval stages. The larvae feed on endemic host plants in the genus
Pipturus (Urticacea, common name “mamaki”). All six larval stages are susceptible to parasitism by at least one of seven introduced parasitoid species associated with them [
35].
1.2.2. Habitats Supporting the Non-Target Species, U. stellata
The endemic host plants of
U. stellata,
Pipturus spp. are typically found in mesic forests. They occur across a moderate elevation gradient (from sea level to 1400 m). Most
Pipturus populations in Hawaii currently occur in habitats with some level of disturbance by alien plant species. For this study,
Pipturus habitats in Hawaii were classified based on landuse/landcover (LULC, State of Hawaii Office of Planning) and fall into two categories: shrub and rangeland; and evergreen forests. Even though
Pipturus spp. do not occur in grassland habitats per se, they do occur in shrubland areas at the margins of grasslands. Elevation was also used to classify habitats where
Pipturus spp. occur, since it is one of the ecological factors found to significantly influence the parasitoid assemblage associated with
U. stellata [
36]. Non-target habitats below 500 m were considered low elevation, habitats between 500 and 900 m were considered medium elevation, and habitats above 900 m were considered high elevation.
1.2.3. Introduced Parasitoids Exploiting U. stellata
Cotesia marginiventris (Cresson) (Hymenoptera: Braconidae): a solitary endoparasitoid native to the West Indies [
43]. Boling and Pitre [
44] studied the biology of this species in the hosts
Tricoplusia ni (Hübner),
Pseudoplusia includes (Walker), and
Heliothis virescens (Fabricius).
Cotesia marginiventris undergoes three larval stages. Soon after molting to the third instar, the parasitoid larva exits its host to spin a white cocoon and pupates inside it. The host dies shortly after parasitoid emergence. Females have short ovipositors and have a preference to oviposit in early instar larvae.
Cotesia marginiventris has been purposely introduced to Cape Verde (Africa), Karnakata (India), Trinidad and Tobago (Caribbean), Australia (Oceania), and Hawaii (North America). Fred Bianchi, assistant entomologist of the Hawaiian Sugar Planters’ Association (H.S.P.A) Experimental Station, introduced this parasitoid to Hawaii from Brownsville, Texas to control the sugar cane pest
Spodoptera exempta [
45,
46,
47]. The introduction was made during the last half of 1942. A total of 4277 adults were distributed to Kauai, Oahu, Maui, and Hawaii. It became quickly established in the islands. At the time of the introduction there was no concern about possible non-target effects. Funasaki et al. [
48] and Henneman and Memmott [
49] report this species attacking other exotic species as well as native insects in Hawaii (
Table 1). On the non-target host
U. stellate, C. marginiventris can parasitize second, third, and fourth instars and emerges from the host when it is either fifth or sixth instar [
35].
Meteorus laphygmae (Viereck) (Hymenoptera: Braconidae) is a nocturnal solitary koinobiont endoparasitoid [
50,
51]. Fernández and Terán [
50] studied the biology of
M. laphygmae in the host
Spodoptera frugiperda (J. E. Smith).
Meteorus laphygmae undergoes three larval stages; the first two develop inside the host and soon after molting to the third instar the larva emerges from the host to immediately start spinning a cocoon, inside which it metamorphoses to the pupal stage. The pupal cocoon is suspended from a thread anchored on foliage, as a protection from natural enemies [
45].
This parasitoid is currently distributed in many states in the continental United States as well as Hawaii, Mexico, Central America (Trinidad and Tobago), and South America (Colombia and Venezuela) [
52]. This parasitoid was purposely introduced to Hawaii by Bianchi, from Brownsville, Texas, to control the sugarcane pest
S. exempta. The introduction process was done from June to November of 1942, and resulted in establishment of the species. A total of 3900 wasps were distributed on Oahu, Maui, Kauai, and Hawaii [
45,
47].
Funasaki et al. [
48] and Henneman and Memmott [
49] reported
M. laphygmae parasitizing other exotic species as well as non-target native species in Hawaii (
Table 2).
M. laphygmae can parasitize second to fifth instar larvae of
U. stellate, and emerges from the host’s sixth instar [
35].
Trathala flavoorbitalis (Cameron) (Hymenoptera: Ichneumonidae) is a solitary, parthenogenetic endoparasitoid [
35]. The literature reports that this parasitoid is a larval-pupal parasitoid [
53] but it was never observed emerging from a pupa of
U. stellata in Hawaii [
35].
Trathala flavoorbitalis occurs in Asia (China, India, Indonesia, Japan, Myanmar, Philippines, Singapore, and Sri Lanka), Europe (Russian Federation), Hawaii, and Australia. This parasitoid has not been purposely introduced anywhere in the world. It is considered an important indigenous biological control agent of many Lepidopteran pests in Asia [
36].
Trathala flavoorbitalisis, an adventive species to Hawaii, was first detected in 1910 [
54] but for the purpose of this risk assessment validation is considered intentionally introduced for the sake of risk modeling. This species has been reared from many adventive and endemic species in Hawaii (
Table 3).
T. flavoorbitalis can parasitize first to fifth instar larvae of
U. stellata, and emerges from the host at the sixth instar [
35].
1.3. Analysis
For the purpose of the validation, U. stellata is considered to be the only non-target species of concern for each of the three parasitoids. In order to explore ways to express uncertainty created by the limited available data, two different scenarios were modeled: the average outcome, and the worst-case scenario. Average-outcome scenarios were modeled based strictly on the information found in the literature, whereas the worst-case scenario was based on the assumption that the published data were not comprehensive, and that hypothetically the agents could successfully overlap with U. stellata geographically, and locate and parasitize susceptible stages of U. stellata at similar rates as the target host.
1.4. Exposure Assessment
Published reports on parasitism were gathered for the three different parasitoids. Information on elevation and habitat type (land use and land cover: LULC) reported in the literature was used to predict potential spatial overlap with
U. stellata. In the risk models, the best-case outcome scenario for the three parasitoids assumed no spatial overlap (probability,
p = 0) with habitats that maintain populations of
U. stellata (evergreen forests and scrubland/shrubland sites) at low (below 500 m), medium (between 500 and 900 m) and high elevation (above 900 m) sites, based on the fact that the published data only report parasitism in agricultural and grassland areas in the provenance of the parasitoids [
56,
57,
58,
59]. The modeled worst-case scenario assumed that published data were biased by habitat type and sampling, and that the three parasitoids could occur in other habitats such as forest and shrub land areas at the three elevation ranges, and therefore overlap spatially with
U. stellata populations (
p = 1.0).
Temporal overlap: Larvae of the non-target species,
U. stellata, are perennially present [
33]. Because of the seasonally equable environmental conditions in Hawaii, it was assumed that the three parasitoids are also perennially active. Therefore full synchronization with susceptible stages of
U. stellata is considered to be the most likely scenario for all three cases (probability of temporal overlap,
p = 1.0).
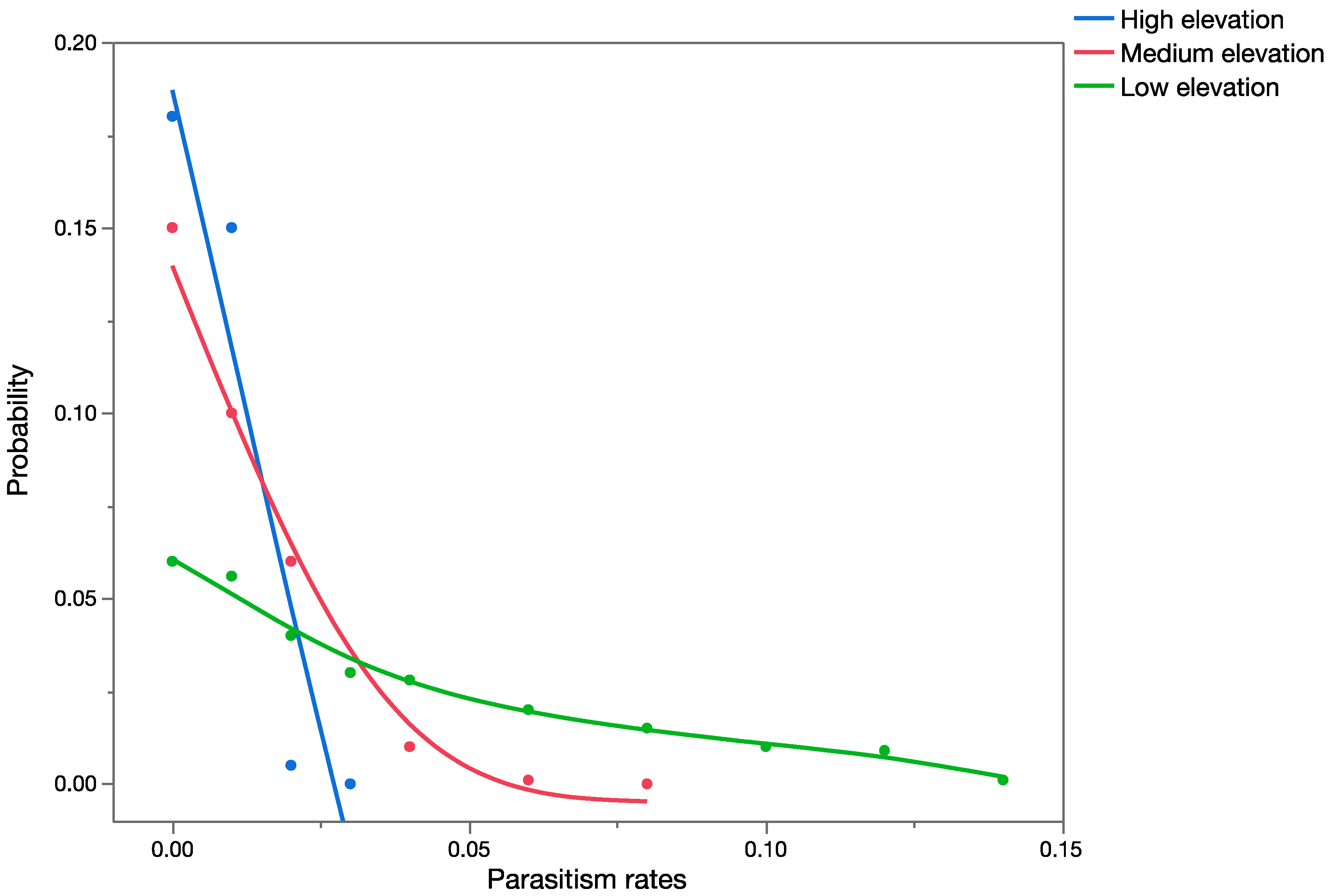
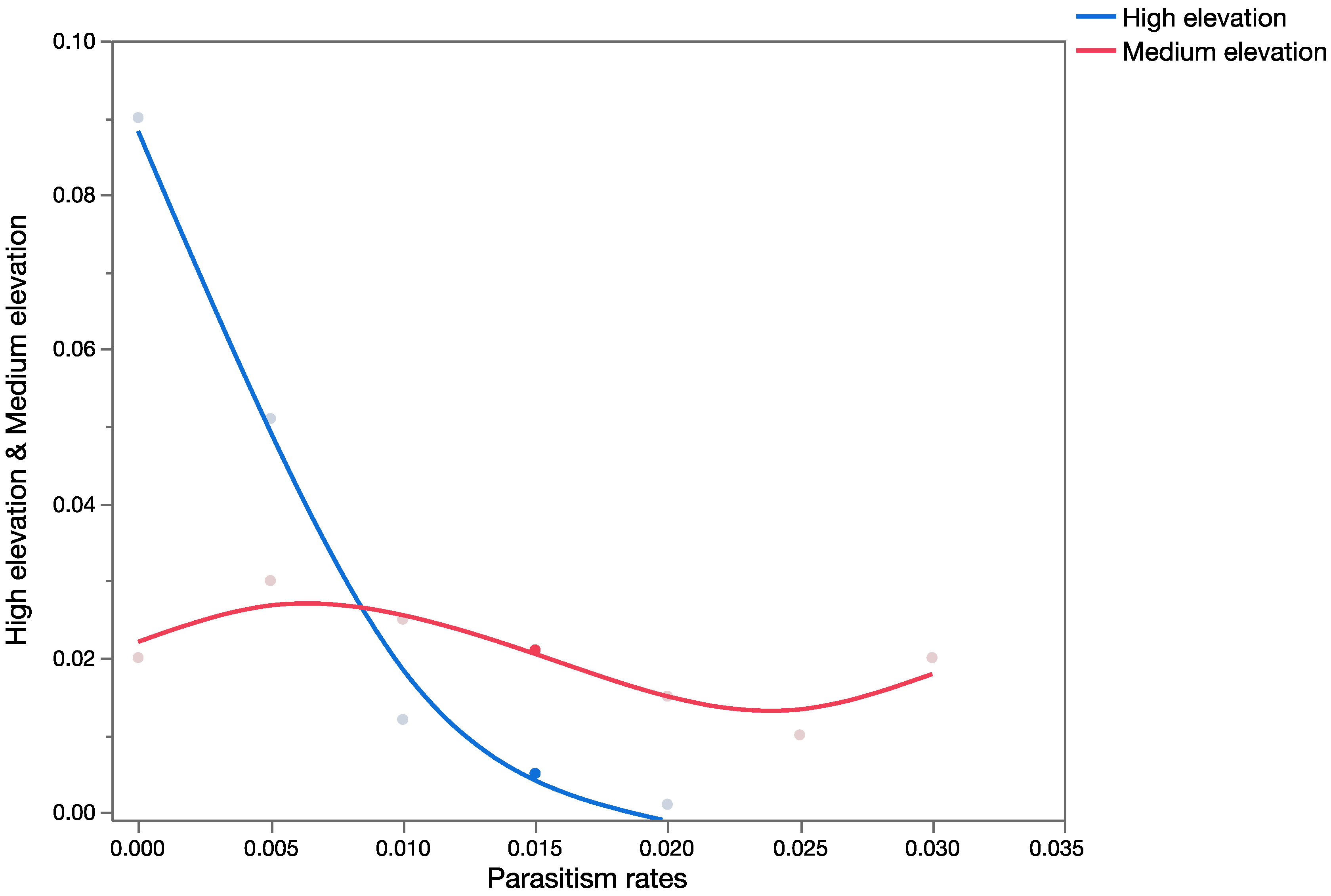
The effects assessment phase of the PRA was based on published records of parasitism for each of the three parasitoid species from outside of Hawaii. Parasitism data obtained from the literature were used to construct probability distributions for each elevation level (low, medium, and high).
The average outcome scenario assumed that each of the parasitoid species would attack only hosts in the families with records of parasitism obtained from the published data. The worst-case scenario assumed that potential hosts in the family Crambidae (specifically U. stellata) will also be subject to parasitism.
For
C. marginiventris and
M. laphygmae, parasitism data were obtained for the target hosts
S. frugiperda and
S. exigua at at low, medium, and high elevations [
56,
58] in Mexico (sites at similar latitude to Hawaii). For
T. flavoorbitalis, parasitism rates were available for the target pest
Antigastra cautalaunalis in India only at low-elevation sites (below 500 m) [
57,
59] (latitude: 29 and 23 degrees north, respectively).
1.5. Risk Characterization
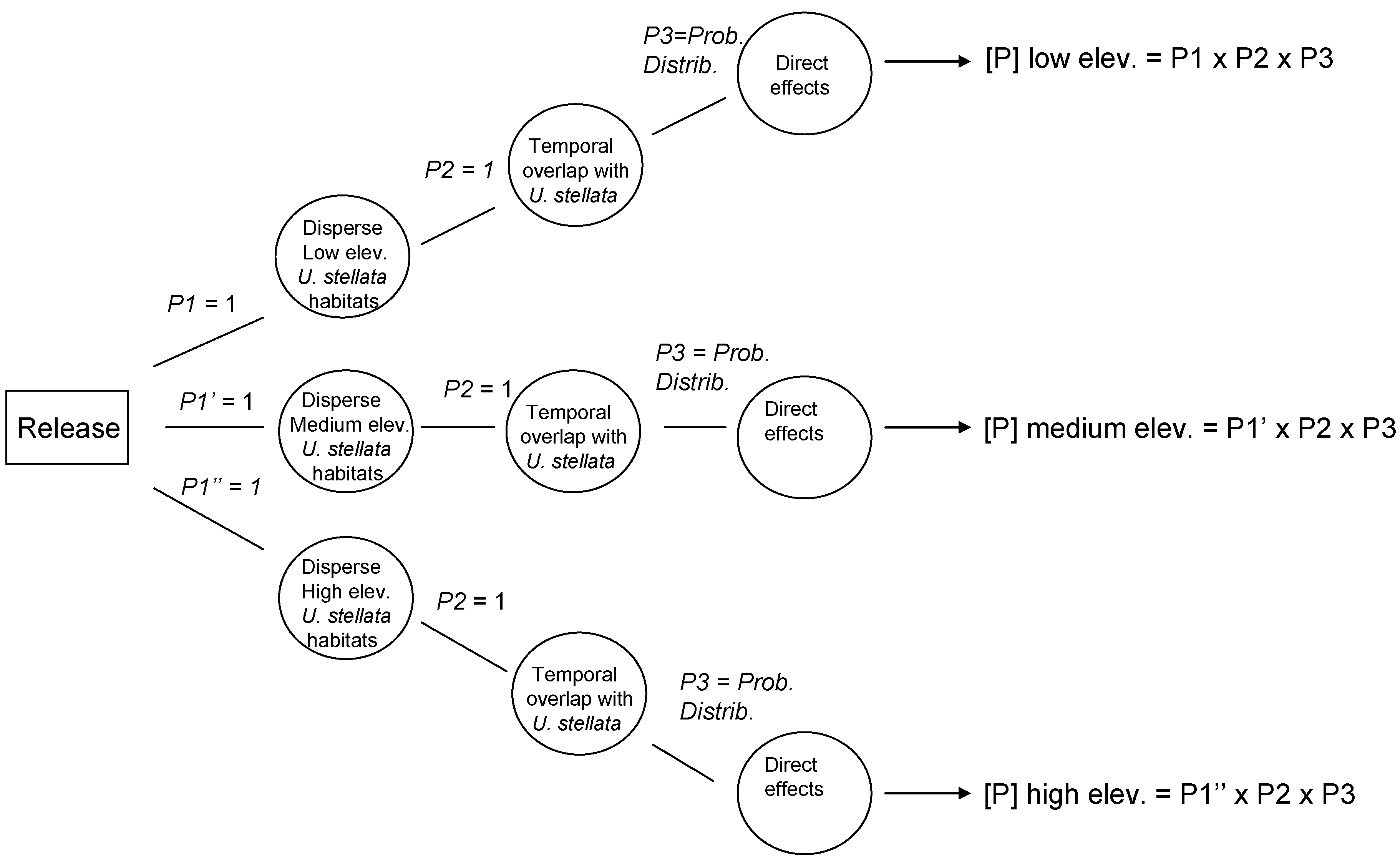
Data from the exposure assessment were used to construct overall probability distributions of parasitism rates for each of the given scenarios, and these data were used in the development of the precision-trees (
Figure 1).
Figure 2 shows the precision tree for the worst-case scenario.
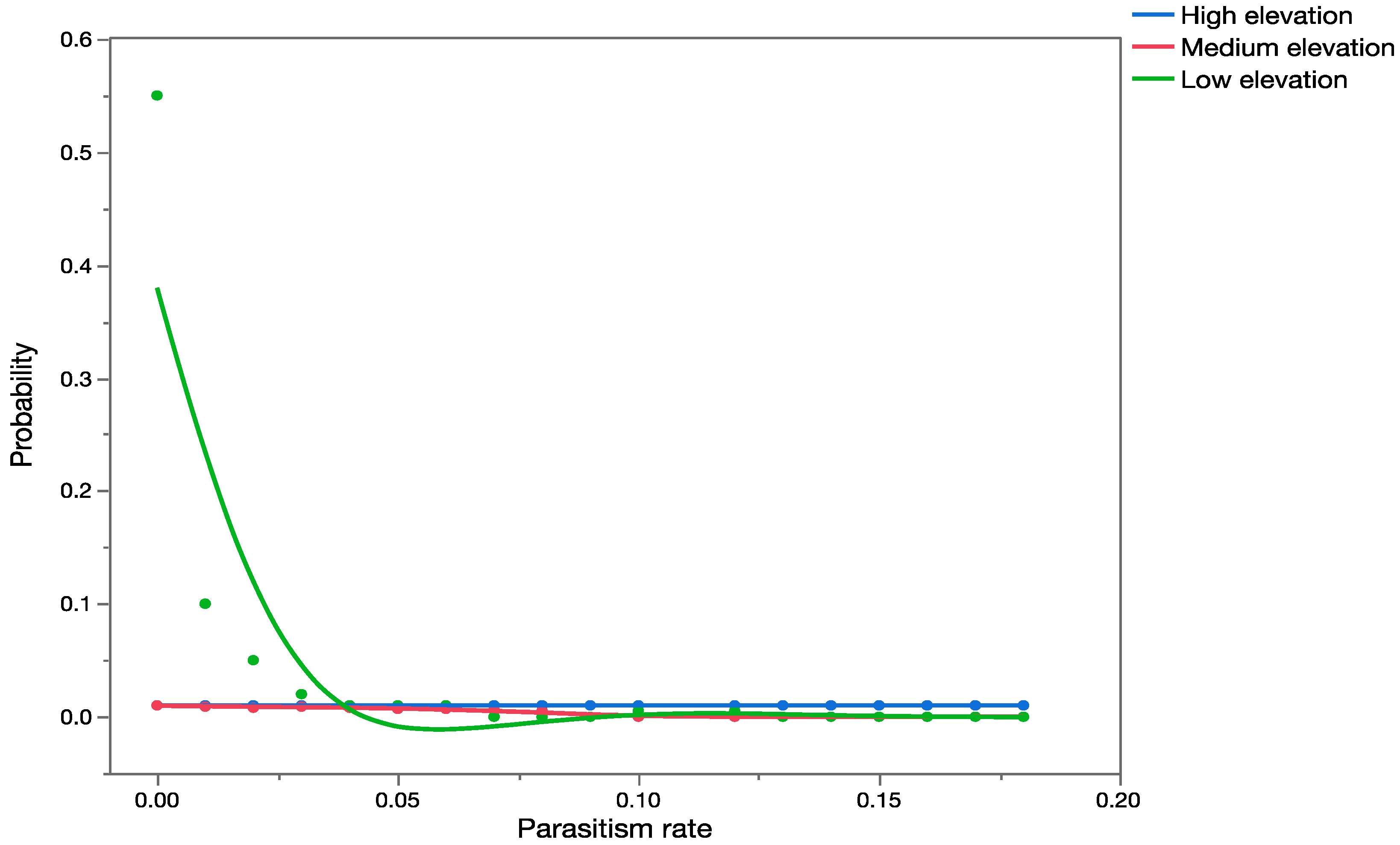
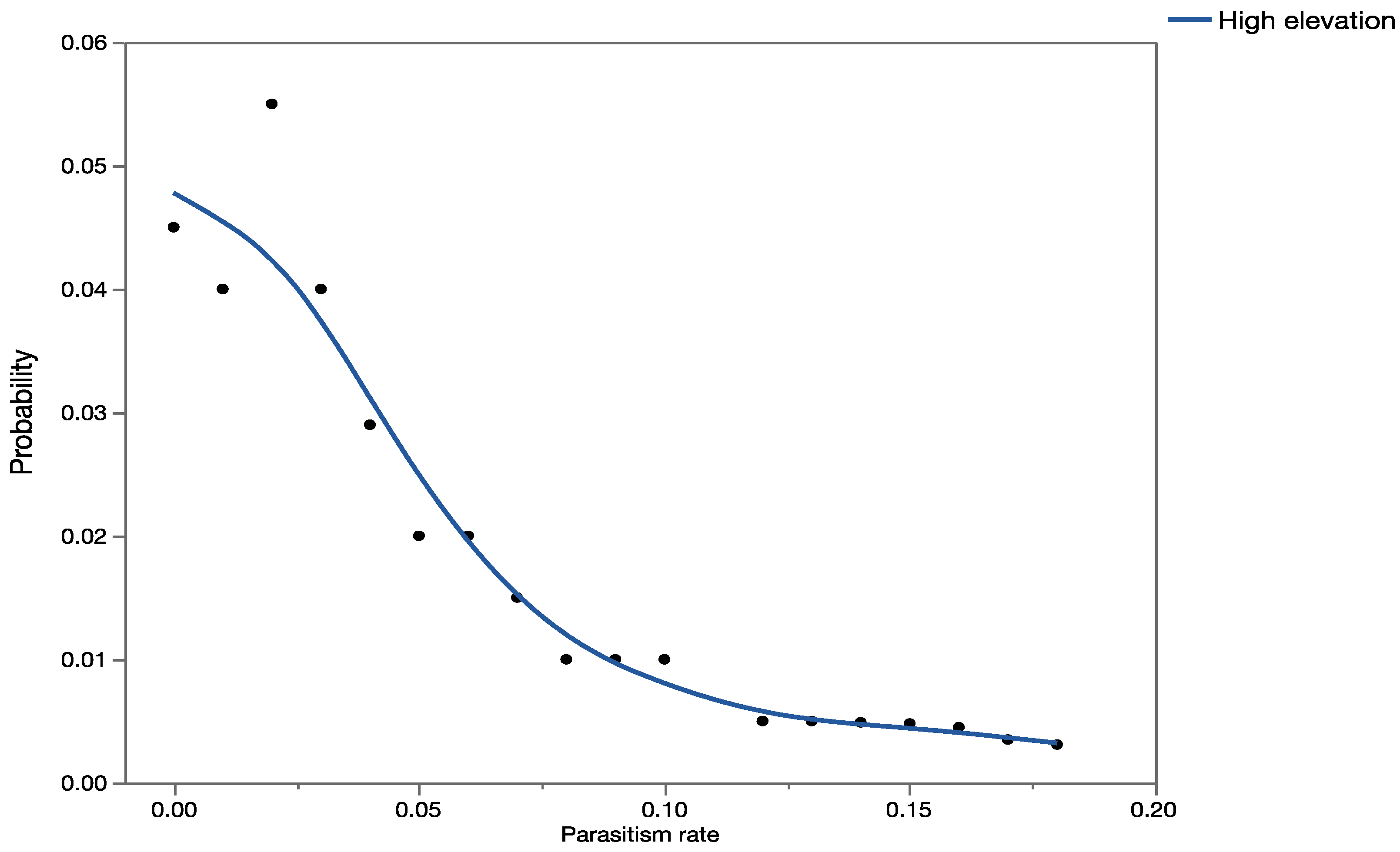
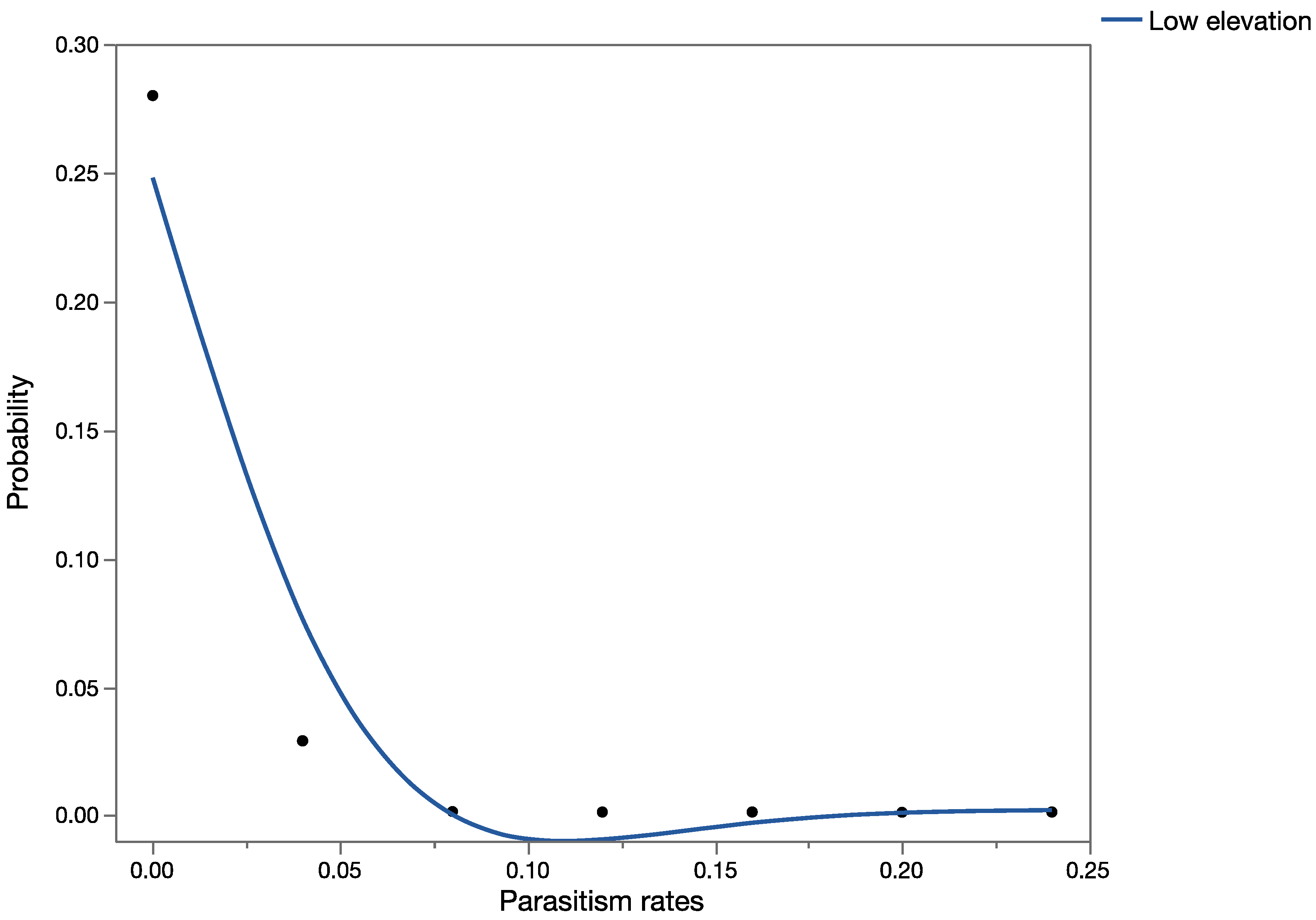
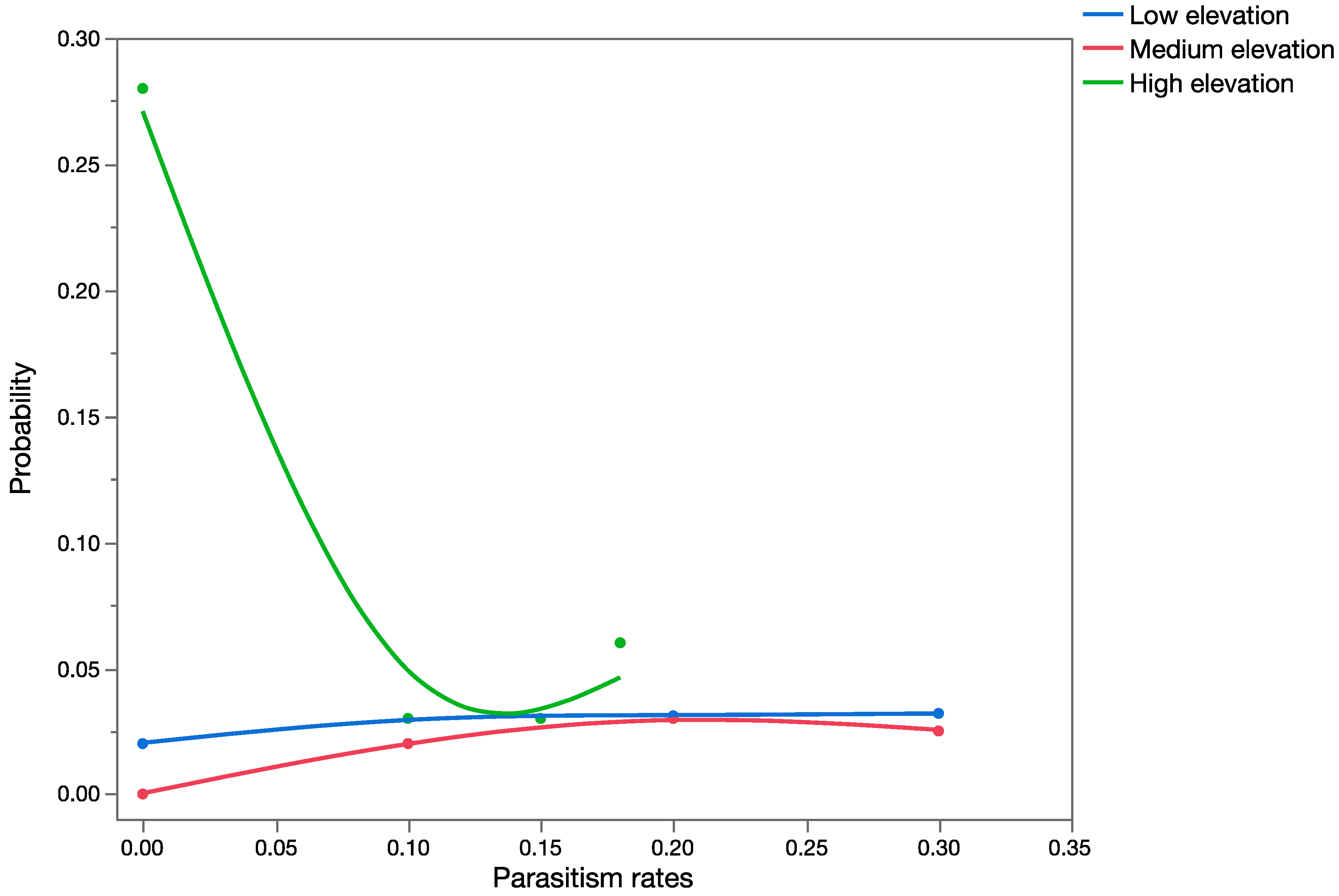
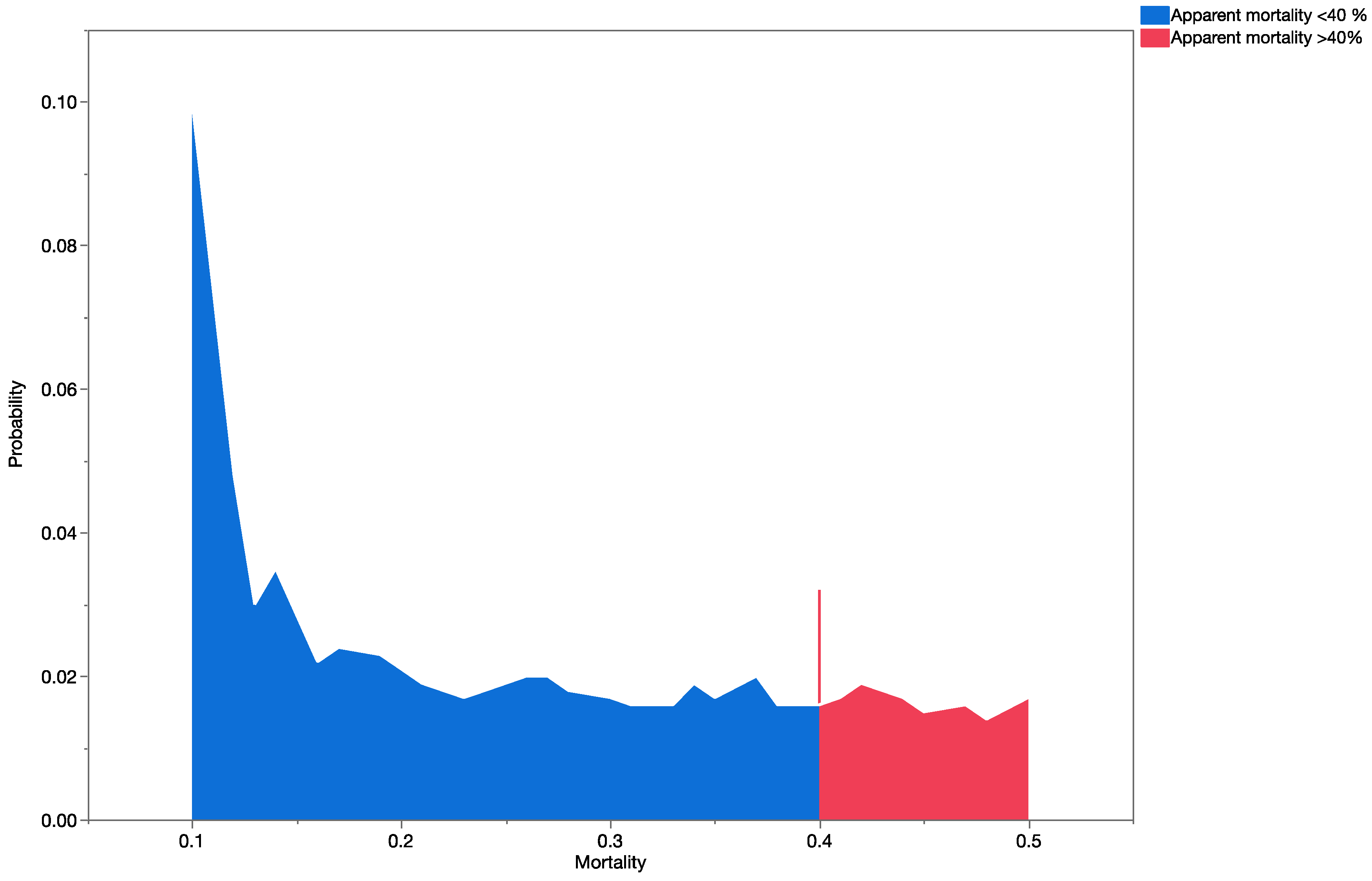
Apparent mortality data [
35] were used to construct probability distributions and validate the results obtained from the precision trees (based on information obtained from previously published data).
Uncertainty analyses (Monte Carlo, MC, simulations) were conducted using Crystal Ball
® software [
60] and each simulation was run with 2000 trials. For validation purposes, probability distributions were also generated for the observed parasitism rates in Hawaii, using recently published data [
33,
34,
35,
36]. The software presents results of the MC simulations graphically as probability/frequency distributions of all possible outcomes. Data generated during the simulations were extracted and analyzed using nonparametric statistics. The Mann–Whitney Rank Sum Test was used to test for significant differences between data when we had only two groups, and the Kruskal–Wallis ANOVA on ranks was used when we had data for multiple groups. The statistical analyses were done using SigmaStat 3.1 (Systat Software, San Jose CA, USA).
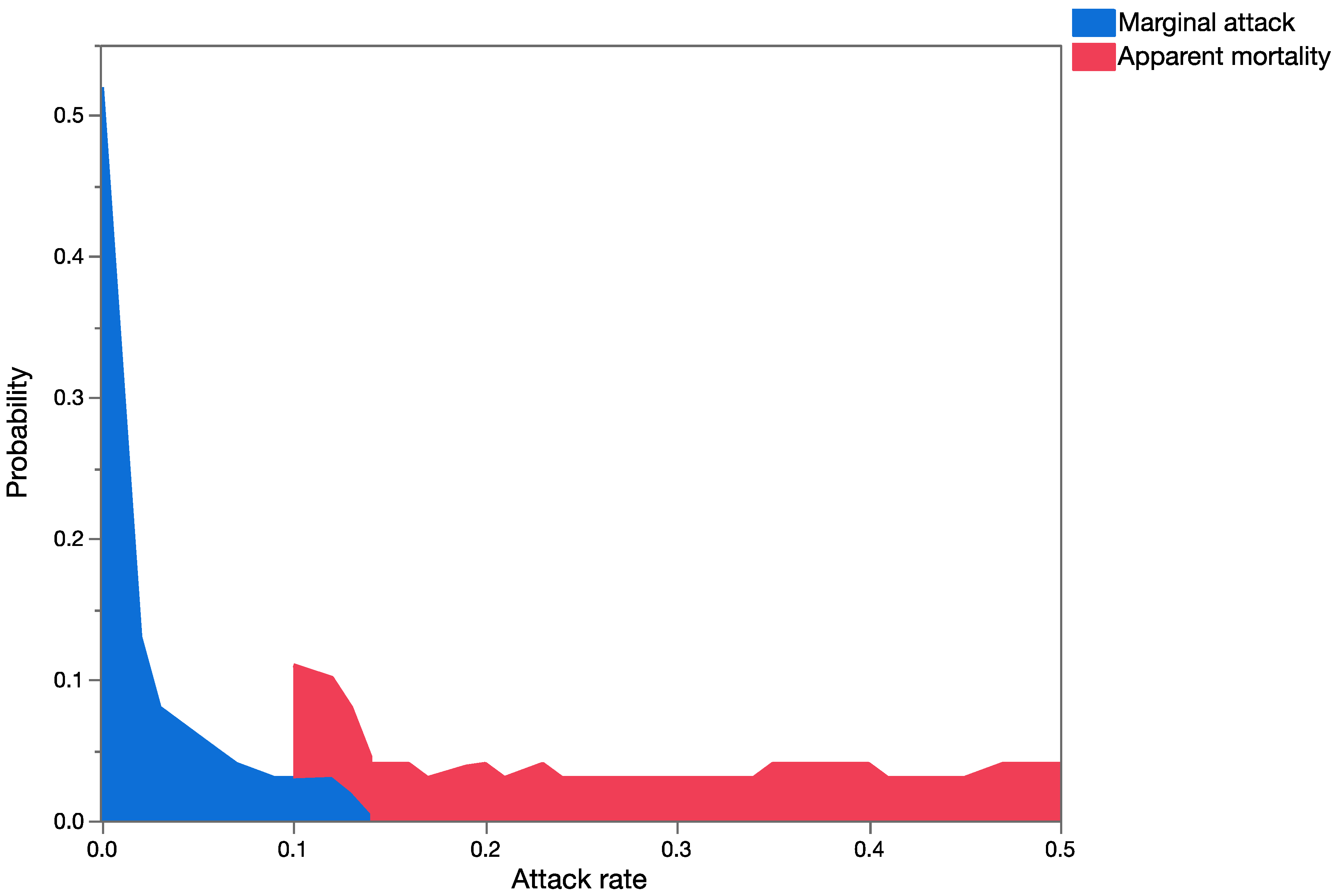
Question 3: How different are the outcomes using estimates of marginal attack rates versus apparent mortality (and measures of uncertainty in both) in conducting probabilistic risk assessment?
Apparent mortality rates (field parasitism, [
35]),
Table 4) and
U. stellata marginal attack rates (from life table studies of cohorts of larvae, [
34],
Table 5) were used for this analysis for each of the three parasitoid species to build probability distributions. Data from the life table studies reported in [
34] and from field surveys of apparent mortality ([
35],
Table 4) were pooled across exposure trials and surveys, respectively, for the development of probability distributions. Probability distributions of marginal attack rate and apparent mortality were used as input variables in the probabilistic models (run in Crystal Ball
®) keeping all other variables of the model constant (spatial and temporal overlap), to run the simulations. This provided estimates of mean mortality in the risk-analyses using measures of apparent or marginal mortality. Data generated during the simulations were extracted, and the Mann–Whitney Rank Sum Test was used to test for significant differences between the two groups.
Question 4: Are there any key ecological variables that would be worth considering in the hypothetical case that any of the three parasitoid species might be considered for introduction to another location or are generally worth considering in any risk assessment?
The results of the comparison of the prediction model and actual field parasitism in Hawaii (Question 1) were used to indicate which aspects of a potential biological control agent’s biology and ecology need to be investigated to provide a robust estimate of their non-target potential.
3. General Discussion
The major advantage of the PRA framework is that it can be designed to consider effects to non-target species at a spatial scale and takes uncertainty in datasets into consideration, and thus evaluates the risks posed by new introductions in a more ecologically meaningful way than procedures that rely on single-point (typically mean) estimates of parasitism, and that do not take spatial variability into account. Single point estimates ignore variation in a dataset, and provide a false sense of precision that leads to what is called “analysis paralysis” [
60]. More realistic and meaningful procedures are not always the simplest to apply, though. PRA will require more data than other proposed and current approaches for assessing non-target risks [
24,
25], and these data may not always be available for researchers and decision makers. It is not necessary to use probability distributions for all input variables in a PRA, but the effectiveness and robustness of risk estimates is increased if one does use them. A precise and realistic model will necessarily be limited to a few applications, whereas a very general approach applicable to many situations will almost certainly be limited in precision.
Not all non-target species are affected equally, and the same non-target species may not be affected equally under different environmental conditions [
32,
73]. Comprehensive risk assessment requires a wide-ranging understanding of the ecology of the biological control agent, as well as the ecology of the target and non-target species [
78,
79]. Observations on the ecology of the biocontrol agent in other areas of introduction and the area of origin, and how parasitism rates vary in those areas under different environmental/ecological conditions, can provide useful baseline information for predictions made for new locations.
Biological control practitioners and regulators prefer the release of agents with narrow host ranges [
77]. Nevertheless, when faced with situations where agents have great potential to control the target pest but may attack some native and beneficial species, careful characterization of the risk should be done to estimate the relative impact of the proposed introduction on populations of the non-target hosts and weighted against the potential benefits [
80,
81]. In those situations PRA could be a tool for better decision-making. Some retrospective studies on polyphagous biocontrol species have shown that they cause minimal impact on the non-target species, underscoring the importance of carefully estimating population-level impacts [
82,
83]. It would be valuable to weigh the environmental benefits attributable to those releases that pose some level of risk, to determine if the non-target parasitism on endemic species, even if minimal, was justified. Additive negative impacts such as habitat destruction and mortality from multiple sources may result in even minimal non-target mortality becoming important; therefore, it is also important to assess the possible impacts of biocontrol introductions in relation to other current sources of mortality [
73].