Elucidating the Common Generalist Predators of Conotrachelus nenuphar (Herbst) (Coleoptera: Curculionidae) in an Organic Apple Orchard Using Molecular Gut-Content Analysis
Abstract
:1. Introduction
2. Experimental Section
2.1. Description of Field Site
2.2. Pest Activity Sampling
2.3. Predator Population Sampling
2.4. Molecular Gut-Content Analysis
2.5. Data Analysis
3. Results
3.1. Pest Activity
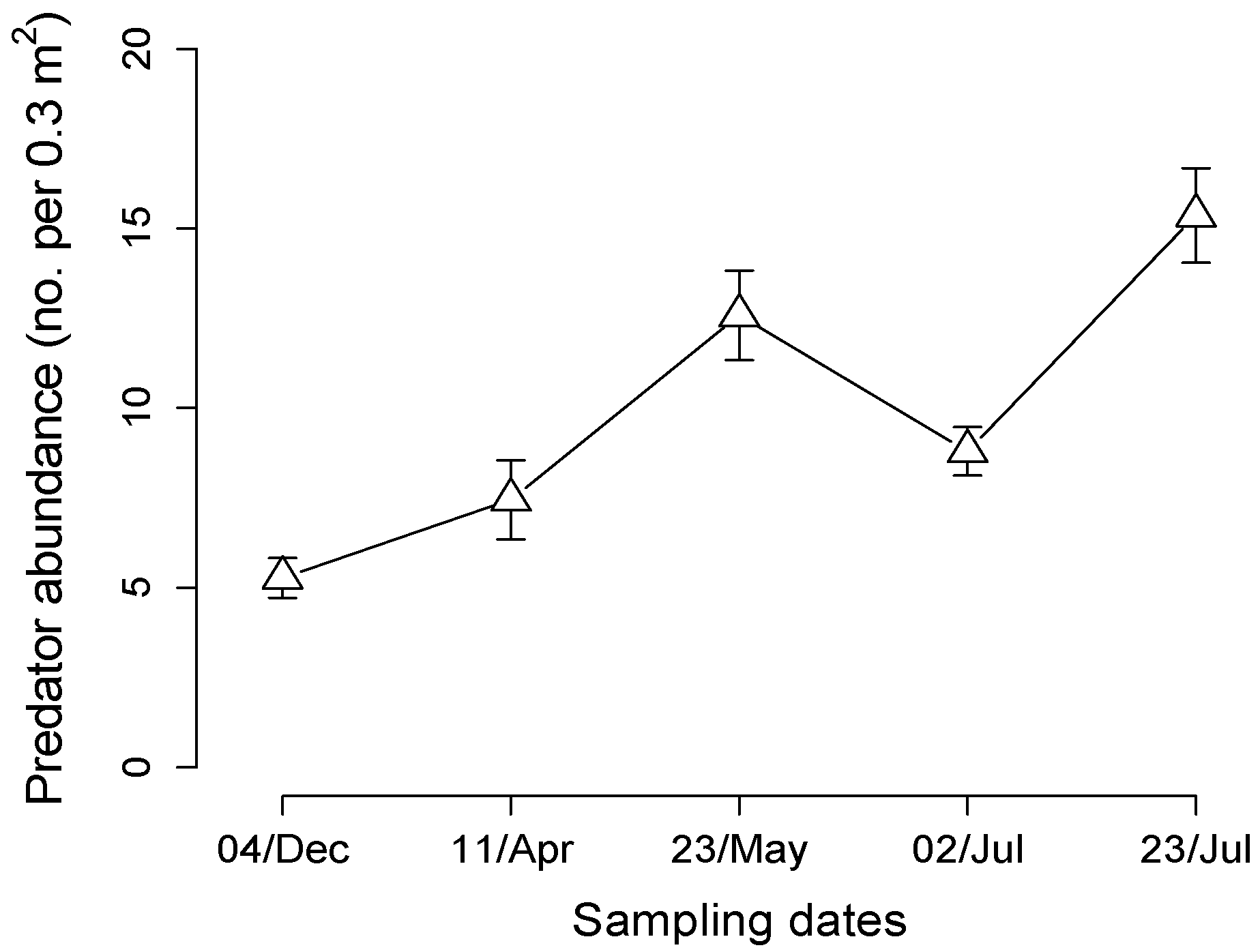
3.2. Predator Populations
3.3. Molecular Gut-Content Analysis
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Quaintance, A.L.; Jenne, E.L. The Plum Curculio; US Department of Agriculture, Bureau of Entomology: Washington, DC, USA, 1912.
- Garman, P.; Zappe, M.P. Control Studies on the Plum Curculio in Connecticut Apple Orchards; Connecticut Agricultural Experiment Station: New Haven, CT, USA, 1929; Volume 301, pp. 309–437. [Google Scholar]
- Lan, Z.; Scherm, H.; Horton, D.L. Temperature-dependent development and prediction of emergence of the summer generation of plum curculio (Coleoptera: Curculionidae) in the southeastern United States. Environ. Entomol. 2004, 33, 174–181. [Google Scholar] [CrossRef]
- Wise, D.H. Spiders in Ecological Webs; Cambridge University Press: Cambridge, UK, 1993. [Google Scholar]
- Wise, J.C.; Coombs, A.B.; Vandervoort, C.; Gut, L.J.; Hoffmann, E.J.; Whalon, M.E. Use of residue profile analysis to identify modes of insecticide activity contributing to control of plum curculio in apples. J. Econ. Entomol. 2006, 99, 2055–2064. [Google Scholar] [CrossRef] [PubMed]
- Racette, G.; Chouinard, G.; Vincent, C.; Hill, S.B. Ecology and management of plum curculio, Conotrachelus nenuphar (Coleoptera, Curculionidae), in apple orchards. Phytoprotection 1992, 73, 85–100. [Google Scholar] [CrossRef]
- Armstrong, T. Life-history and ecology of the plum curculio, Conotrachelus nenuphar (Hbst.) (Coleoptera: Curculionidae) in the Niagara Peninsula, Ontario. Can. Entomol. 1958, 90, 8–17. [Google Scholar] [CrossRef]
- Mampe, C.D.; Neunzig, H.H. Biology parasitism and population sampling of plum curculio on blueberry in North Carolina. J. Econ. Entomol. 1967, 60, 807–812. [Google Scholar] [CrossRef]
- de Roince, C.B.; Lavigne, C.; Ricard, J.M.; Franck, P.; Bouvier, J.C.; Garcin, A.; Symondson, W.O.C. Predation by generalist predators on the codling moth versus a closely-related emerging pest the oriental fruit moth: A molecular analysis. Agric. Forest Entomol. 2012, 14, 260–269. [Google Scholar] [CrossRef]
- Epstein, D.L.; Zack, R.S.; Brunner, J.F.; Gut, L.; Brown, J.J. Ground beetle activity in apple orchards under reduced pesticide management regimes. Biol. Control 2001, 21, 97–104. [Google Scholar] [CrossRef]
- Glen, D. Predation of codling moth eggs, Cydia pomonella, the predators responsible and their alternative prey. J. Appl. Ecol. 1977, 14, 445–456. [Google Scholar] [CrossRef]
- Hagley, E.A.C.; Allen, W.R. Ground beetles (Coleoptera, Carabidae) as predators of the codling moth, Cydia pomonella (Lepidoptera, Tortricidae). Can. Entomol. 1988, 120, 917–925. [Google Scholar] [CrossRef]
- Mathews, C.R.; Bottrell, D.G.; Brown, M.W. Habitat manipulation of the apple orchard floor to increase ground-dwelling predators and predation of Cydia pomonella (L.) (Lepidoptera: Tortricidae). Biol. Control 2004, 30, 265–273. [Google Scholar] [CrossRef]
- Miñarro, M.; Dapena, E. Effects of groundcover management on ground beetles (Coleoptera: Carabidae) in an apple orchard. Appl. Soil Ecol. 2003, 23, 111–117. [Google Scholar] [CrossRef]
- Solomon, M.G.; Cross, J.V.; Fitzgerald, J.D.; Campbell, C.A.M.; Jolly, R.L.; Olszak, R.W.; Niemczyk, E.; Vogt, H. Biocontrol of pests of apples and pears in northern and central Europe—3. Predators. Biocontrol Sci. Technol. 2000, 10, 91–128. [Google Scholar] [CrossRef]
- Jenkins, D.A.; Mizell, R.F.; Shapiro-Ilan, D.; Cottrell, T.; Horton, D. Invertebrate predators and parasitoids of plum curculio, Conotrachelus nenuphar (Coleoptera: Curculionidae) in Georgia and Florida. Fla. Entomol. 2006, 89, 435–440. [Google Scholar] [CrossRef]
- Dawkar, V.V.; Chikate, Y.R.; Lomate, P.R.; Dholakia, B.B.; Gupta, V.S.; Giri, A.P. Molecular insights into resistance mechanisms of lepidopteran insect pests against toxicants. J. Proteome Res. 2013, 12, 4727–4737. [Google Scholar] [CrossRef] [PubMed]
- Virant-Doberlet, M.; King, R.A.; Polajnar, J.; Symondson, W.O.C. Molecular diagnostics reveal spiders that exploit prey vibrational signals used in sexual communication. Mol. Ecol. 2011, 20, 2204–2216. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Ripoll, R.; Agusti, N.; Berruezo, R.; Gabarra, R. Conspecific and heterospecific interactions between two omnivorous predators on tomato. Biol. Control 2012, 62, 189–196. [Google Scholar] [CrossRef]
- Sheppard, S.K.; Harwood, J.D. Advances in molecular ecology: Tracking trophic links through predator-prey food-webs. Funct. Ecol. 2005, 19, 751–762. [Google Scholar] [CrossRef]
- Fournier, V.; Hagler, J.; Daane, K.; de Leon, J.; Groves, R. Identifying the predator complex of Homalodisca vitripennis (Hemiptera: Cicadellidae): A comparative study of the efficacy of an ELISA and PCR gut content assay. Oecologia 2008, 157, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Gariepy, T.D.; Messing, R.H. Development and use of molecular diagnostic tools to determine trophic links and interspecific interactions in aphid-parasitoid communities in Hawaii. Biol. Control 2012, 60, 26–38. [Google Scholar] [CrossRef]
- Greenstone, M.H.; Szendrei, Z.; Payton, M.E.; Rowley, D.L.; Coudron, T.C.; Weber, D.C. Choosing natural enemies for conservation biological control: Use of the prey detectability half-life to rank key predators of Colorado potato beetle. Entomol. Exp. Appl. 2010, 136, 97–107. [Google Scholar] [CrossRef]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar] [PubMed]
- Breton, S.; Burger, G.; Stewart, D.T.; Blier, P.U. Comparative analysis of genderassociated complete mitochondrial genomes in marine mussels (Mytilus spp.). Genetics 2006, 172, 1107–1119. [Google Scholar] [CrossRef] [PubMed]
- Chapman, E.G.; Schmidt, J.M.; Welch, K.D.; Harwood, J.D. Molecular evidence for dietary selectivity and pest suppression potential in an epigeal spider community in winter wheat. Biol. Control 2013, 65, 72–86. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: A multiple sequence alignment method with reduced time and space complexity. BMC Bioinform. 2004, 5, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Rozen, S.; Skaletsky, H. Primer3 on the WWW for general users and for biologist programmers. Methods Mol. Biol. 2000, 132, 365–386. [Google Scholar] [PubMed]
- Pinheiro, J.C.; Bates, D.M. Mixed-Effects Models in S and S-PLUS; Springer: New York, NY, USA, 2000. [Google Scholar]
- RCoreTeam. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Pereault, R.J.; Whalon, M.E.; Alston, D.G. Field efficacy of entomopathogenic fungi and nematodes targeting caged last-instar plum curculio (Coleoptera: Curculionidae) in Michigan cherry and apple orchards. Environ. Entomol. 2009, 38, 1126–1134. [Google Scholar] [CrossRef] [PubMed]
- Shapiro-Ilan, D.I.; Mizell, R.F.; Cottrell, T.E.; Horton, D.L. Measuring field efficacy of Steinernema feltiae and Steinernema riobrave for suppression of plum curculio, Conotrachelus nenuphar, larvae. Biol. Control 2004, 30, 496–503. [Google Scholar] [CrossRef]
- Symondson, W.O.C.; Sunderland, K.D.; Greenstone, M.H. Can generalist predators be effective biocontrol agents? Ann. Rev. Entomol. 2002, 47, 561–594. [Google Scholar] [CrossRef] [PubMed]
- Harwood, J.D.; Sunderland, K.D.; Symondson, W.O.C. Prey selection by linyphiid spiders: Molecular tracking of the effects of alternative prey on rates of aphid consumption in the field. Mol. Ecol. 2004, 13, 3549–3560. [Google Scholar] [CrossRef] [PubMed]
- Giorgi, J.A.; Vandenberg, N.J.; McHugh, J.V.; Forrester, J.A.; Ślipiński, S.A.; Miller, K.B.; Shapiro, L.R.; Whiting, M.F. The evolution of food preferences in Coccinellidae. Biol. Control 2009, 51, 215–231. [Google Scholar] [CrossRef]
- Koss, A.M.; Chang, G.C.; Snyder, W.E. Predation of green peach aphids by generalist predators in the presence of alternative, Colorado potato beetle egg prey. Biol. Control 2004, 31, 237–244. [Google Scholar] [CrossRef]
- Ma, J.; Li, Y.-Z.; Keller, M.; Ren, S.-X. Functional response and predation of Nabis kinbergii (Hemiptera: Nabidae) to Plutella xylostelia (Lepidoptera: Plutellidae). Insect Sci. 2005, 12, 281–286. [Google Scholar] [CrossRef]
- Riechert, S.E.; Lockley, T. Spiders as biological control agents. Ann. Rev. Entomol. 1984, 29, 299–320. [Google Scholar] [CrossRef]
- Uetz, G.W.; Halaj, J.; Cady, A.B. Guild structure of spiders in major crops. J. Arachnol. 1999, 27, 270–280. [Google Scholar]
- Stearns, L.A.; Williams, L.L.; Hadee, W.R. Control of the Plum Curculio in Delaware; University of Delaware Agricultural Experiment Station: Newark, DE, USA, 1935; pp. 1–28. [Google Scholar]
| Taxonomic Description | PCR Positives | No. Tested |
|---|---|---|
| Araneae | ||
| Agelenidae | 0 | 1 |
| Amaurobiidae: Coras sp. | 0 | 2 |
| Anyphaenidae | 0 | 4 |
| Araneidae: Mangora sp. | 0 | 9 |
| Clubionidae: Clubiona sp. | 0 | 6 |
| Corinnidae | 0 | 9 |
| Dictynidae: Emblyna sp. | 3 | 8 |
| Dictyna sp. | 0 | 3 |
| Saltonia sp. | 0 | 2 |
| Unknown-immature | 0 | 4 |
| Linyphiidae: Agyneta fabra | 2 | 11 |
| Erigone sp. | 0 | 1 |
| Semljicola sp. | 0 | 1 |
| Unknown-immature | 1 | 19 |
| Liocranidae: Agroeca pratensis | 1 | 2 |
| Lycosidae: Pardosa sp. | 1 | 8 |
| Pirata sp. | 0 | 30 |
| Schizocosa sp. | 0 | 5 |
| Trabeops sp. | 0 | 46 |
| Trochosa sp. | 0 | 1 |
| Unknown-immature | 0 | 5 |
| Mysmenidae | 0 | 1 |
| Salticidae: Pelegrina sp. | 1 | 23 |
| Hentzia sp. | 0 | 5 |
| Metacyrba sp. | 0 | 1 |
| Metaphiddipus sp. | 0 | 5 |
| Phiddipus sp. | 0 | 4 |
| Unknown-immature | 0 | 2 |
| Philodromidae: Philodromus peninsulanus | 1 | 26 |
| Tibellus sp. | 0 | 7 |
| Unknown-immature | 0 | 4 |
| Pisauridae: Pisaurina sp. | 0 | 1 |
| Theridiidae: Enoplognatha ovata | 0 | 7 |
| Neottiura sp. | 0 | 7 |
| Theridion sp. | 1 | 32 |
| Unknown-immature | 6 | 37 |
| Thomisidae: Misumena sp. | 0 | 1 |
| Misumenops sp. | 0 | 3 |
| Ozyptila sp. | 0 | 3 |
| Xysticus sp. | 1 | 2 |
| Thomisidae: Unknown-immature | 1 | 3 |
| Opiliones: Phalangiidae | 0 | 8 |
| Hemiptera | ||
| Nabidae: Nabis sp. | 7 | 32 |
| Pentatomidae: Podisus maculiventris | 2 | 5 |
| Coleoptera | ||
| Cantharidae: Podabrus sp. | 1 | 8 |
| Carabidae: Amara sp. | 1 | 4 |
| Anisodactylus sp. | 0 | 2 |
| Bradycellus sp. | 0 | 3 |
| Colliuris sp. | 0 | 1 |
| Harpalus sp. | 1 | 2 |
| Coccinellidae: Brachiacantha ursina | 2 | 3 |
| Coleomegilla maculata | 0 | 7 |
| Harmonia axyridis | 3 | 4 |
| Propylea quatuordecimpunctata | 0 | 2 |
| Staphylinidae: Stenus sp. | 2 | 6 |
| Taxonomic Description | PCR Positives | No. Tested |
|---|---|---|
| Araneae | ||
| Dictynidae: Emblyna sp. | 3 | 8 |
| Linyphiidae: Agyneta fabra | 2 | 11 |
| Liocranidae: Agroeca pratensis | 1 | 2 |
| Lycosidae: Pardosa sp. | 1 | 8 |
| Salticidae: Pelegrina sp. | 1 | 23 |
| Philodromidae: Philodromus peninsulanus | 1 | 26 |
| Theridiidae (immature) | 7 | 69 |
| Thomisidae (immature) | 2 | 5 |
| Hemiptera | ||
| Nabidae: Nabis sp. | 7 | 32 |
| Pentatomidae: Podisus maculiventris | 2 | 5 |
| Coleoptera | ||
| Cantharidae: Podabrus sp. | 1 | 8 |
| Carabidae: Amara sp. | 1 | 4 |
| Harpalus sp. | 1 | 2 |
| Coccinellidae: Brachiacantha ursina | 2 | 3 |
| Harmonia axyridis | 3 | 4 |
| Staphylinidae: Stenus sp. | 2 | 6 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmidt, J.M.; Szendrei, Z.; Grieshop, M. Elucidating the Common Generalist Predators of Conotrachelus nenuphar (Herbst) (Coleoptera: Curculionidae) in an Organic Apple Orchard Using Molecular Gut-Content Analysis. Insects 2016, 7, 29. https://doi.org/10.3390/insects7030029
Schmidt JM, Szendrei Z, Grieshop M. Elucidating the Common Generalist Predators of Conotrachelus nenuphar (Herbst) (Coleoptera: Curculionidae) in an Organic Apple Orchard Using Molecular Gut-Content Analysis. Insects. 2016; 7(3):29. https://doi.org/10.3390/insects7030029
Chicago/Turabian StyleSchmidt, Jason M., Zsofia Szendrei, and Matthew Grieshop. 2016. "Elucidating the Common Generalist Predators of Conotrachelus nenuphar (Herbst) (Coleoptera: Curculionidae) in an Organic Apple Orchard Using Molecular Gut-Content Analysis" Insects 7, no. 3: 29. https://doi.org/10.3390/insects7030029
APA StyleSchmidt, J. M., Szendrei, Z., & Grieshop, M. (2016). Elucidating the Common Generalist Predators of Conotrachelus nenuphar (Herbst) (Coleoptera: Curculionidae) in an Organic Apple Orchard Using Molecular Gut-Content Analysis. Insects, 7(3), 29. https://doi.org/10.3390/insects7030029






