Insecticide Resistance Mutations, Enzymatic Activity, and Pathogen Infection in Culex quinquefasciatus from Haiti
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Arthropod Surveillance Collection Procedures
2.2. Control Strain
2.3. Sample Homogenization, Pooling, and RNA Purification
2.4. Speciation, Knockdown Resistance, and Acetylcholinesterase Mutation Detection Assays
2.5. Metabolic Resistance Assays
2.5.1. Bradford Protein Assay
2.5.2. Cytochrome P450 Assay
2.5.3. Glutathione S-Transferase (GST) Activity Assay
2.5.4. α-Carboxylesterase Activity Assay
2.5.5. β-Carboxylesterase Activity Assay
2.6. Nanopore Sequencing and Bioinformatics
3. Results
3.1. Assessment of Knockdown and Acetylcholinesterase Target Site Mutations
3.2. Metabolic Resistance Assays
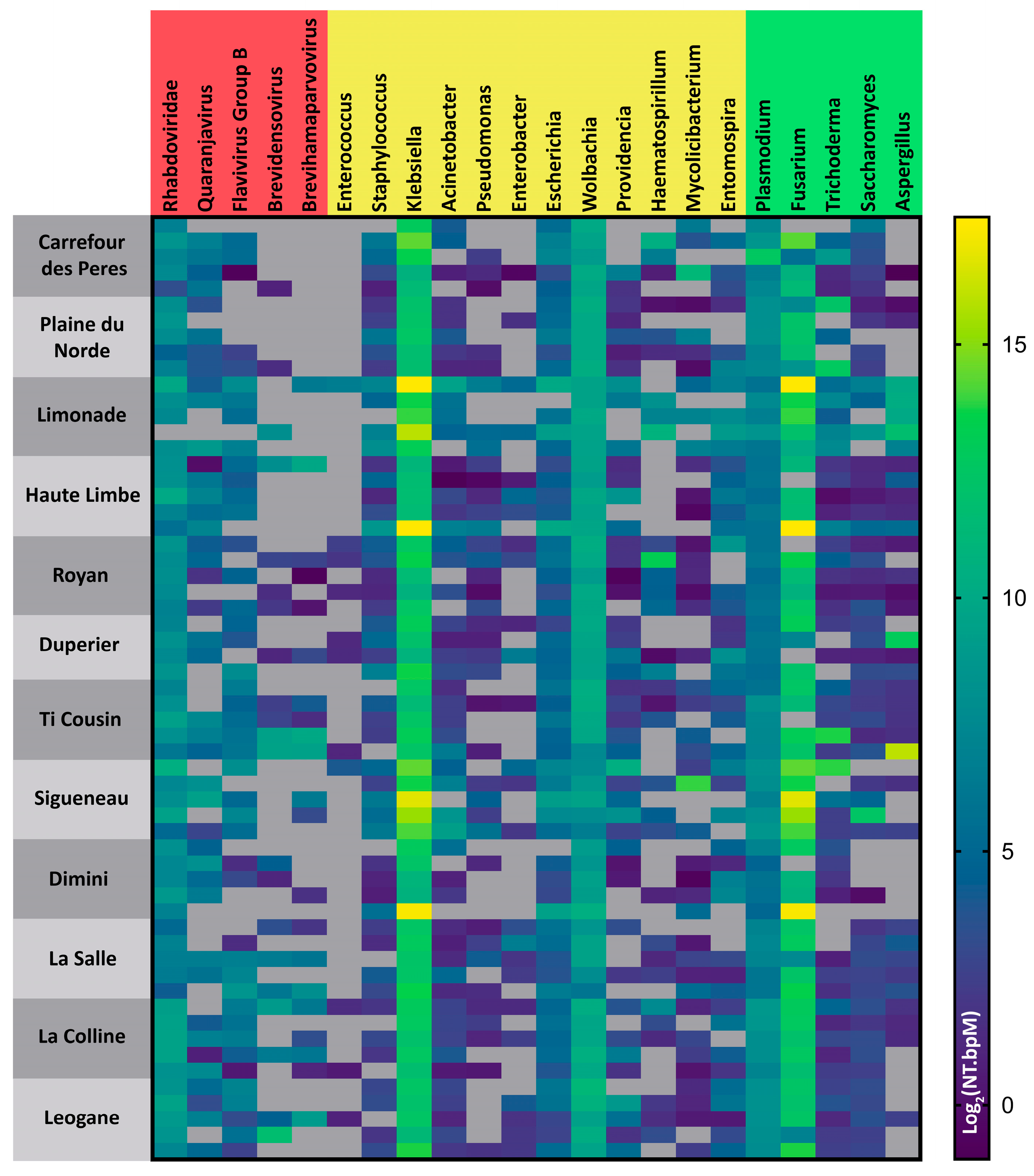
3.3. Microbiome Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| WNV | West Nile virus |
| kdr | knock down resistance |
| AchE | acetylcholinesterase |
| NFW | nuclease-free water |
| MCA | melt curve assay |
| Tm | melting temperature |
| dscDNA | double-stranded cDNA |
| Ct | cycle threshold |
| GST | glutathione S transferase |
| CytC | cytochrome C oxidase |
| NT.bpm | NT/NR database matching bases per million |
References
- Beau de Rochars, M.V.; Milord, M.D.; St Jean, Y.; Desormeaux, A.M.; Dorvil, J.J.; Lafontant, J.G.; Addiss, D.G.; Streit, T.G. Geographic distribution of lymphatic filariasis in Haiti. Am. J. Trop. Med. Hyg. 2004, 71, 598–601. [Google Scholar] [CrossRef]
- Drexler, N.; Washington, C.H.; Lovegrove, M.; Grady, C.; Milord, M.D.; Streit, T.; Lammie, P. Secondary mapping of lymphatic filariasis in Haiti-definition of transmission foci in low-prevalence settings. PLoS Negl. Trop. Dis. 2012, 6, e1807. [Google Scholar] [CrossRef]
- Kent, R.J.; Crabtree, M.B.; Miller, B.R. Transmission of West Nile virus by Culex quinquefasciatus Say infected with Culex Flavivirus Izabal. PLoS Negl. Trop. Dis. 2010, 4, e671. [Google Scholar] [CrossRef]
- Crockett, R.K.; Burkhalter, K.; Mead, D.; Kelly, R.; Brown, J.; Varnado, W.; Roy, A.; Horiuchi, K.; Biggerstaff, B.J.; Miller, B.; et al. Culex flavivirus and West Nile virus in Culex quinquefasciatus populations in the southeastern United States. J. Med. Entomol. 2012, 49, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Diaz, L.A.; Flores, F.S.; Beranek, M.; Rivarola, M.E.; Almiron, W.R.; Contigiani, M.S. Transmission of endemic St Louis encephalitis virus strains by local Culex quinquefasciatus populations in Cordoba, Argentina. Trans. R. Soc. Trop. Med. Hyg. 2013, 107, 332–334. [Google Scholar] [CrossRef]
- Richards, S.L.; Lord, C.C.; Pesko, K.; Tabachnick, W.J. Environmental and biological factors influencing Culex pipiens quinquefasciatus Say (Diptera: Culicidae) vector competence for Saint Louis encephalitis virus. Am. J. Trop. Med. Hyg. 2009, 81, 264–272. [Google Scholar] [CrossRef]
- Mitchell, C.J.; Gubler, D.J.; Monath, T.P. Variation in infectivity of Saint Louis encephalitis viral strains for Culex pipiens quinquefasciatus (Diptera: Culicidae). J. Med. Entomol. 1983, 20, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Sang, R.; Kioko, E.; Lutomiah, J.; Warigia, M.; Ochieng, C.; O’Guinn, M.; Lee, J.S.; Koka, H.; Godsey, M.; Hoel, D.; et al. Rift Valley fever virus epidemic in Kenya, 2006/2007: The entomologic investigations. Am. J. Trop. Med. Hyg. 2010, 83, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Ben-Chetrit, E.; Schwartz, E. Vector-borne diseases in Haiti: A review. Travel. Med. Infect. Dis. 2015, 13, 150–158. [Google Scholar] [CrossRef]
- Beatty, M.E.; Hunsperger, E.; Long, E.; Schurch, J.; Jain, S.; Colindres, R.; Lerebours, G.; Bernard, Y.M.; Dobbins, J.G.; Brown, M.; et al. Mosquitoborne infections after Hurricane Jeanne, Haiti, 2004. Emerg. Infect. Dis. 2007, 13, 308–310. [Google Scholar] [CrossRef]
- Verma, M.; Phartyal, R.; Bhatt, A. Introduction to Flaviviruses and Their Global Prevalence. In Human Viruses: Diseases, Treatments and Vaccines: The New Insights; Ahmad, S.I., Ed.; Springer International Publishing: Cham, Switzerland, 2021; pp. 411–439. [Google Scholar]
- Scott, J.G.; Yoshimizu, M.H.; Kasai, S. Pyrethroid resistance in Culex pipiens mosquitoes. Pestic. Biochem. Physiol. 2015, 120, 68–76. [Google Scholar] [CrossRef]
- Burgess, E.R.T.; Lopez, K.; Irwin, P.; Jaeger, C.P.; Estep, A.S. Assessing pyrethroid resistance status in the Culex pipiens complex (Diptera: Culicidae) from the northwest suburbs of Chicago, Illinois using Cox regression of bottle bioassays and other detection tools. PLoS ONE 2022, 17, e0268205. [Google Scholar] [CrossRef]
- Mathews, G.; Derraik, J.G.B.; Walker, M.; Knox, R.; Barraclough, R.K. Morphological variation in invasive Culex quinquefasciatus Say (Diptera: Culicidae) larvae from an urban site in Auckland, New Zealand. N. Z. J. Zool. 2017, 44, 342–353. [Google Scholar] [CrossRef]
- Weill, M.; Fort, P.; Berthomieu, A.; Dubois, M.P.; Pasteur, N.; Raymond, M. A novel acetylcholinesterase gene in mosquitoes codes for the insecticide target and is non-homologous to the ace gene in Drosophila. R. Soc. Lond. B Biol. Sci. 2002, 269, 2007–2016. [Google Scholar] [CrossRef] [PubMed]
- Lopes, R.P.; Lima, J.B.P.; Martins, A.J. Insecticide resistance in Culex quinquefasciatus Say, 1823 in Brazil: A review. Parasit Vectors 2019, 12, 591. [Google Scholar] [CrossRef]
- Chandrasiri, P.; Fernando, S.D.; De Silva, B. Insecticide resistance and molecular characterization of knockdown resistance (kdr) in Culex quinquefasciatus mosquitoes in Sri Lanka. J. Vector Ecol. 2020, 45, 204–210. [Google Scholar] [CrossRef] [PubMed]
- McAllister, J.C.; Godsey, M.S.; Scott, M.L. Pyrethroid resistance in Aedes aegypti and Aedes albopictus from Port-au-Prince, Haiti. J. Vector Ecol. 2012, 37, 325–332. [Google Scholar] [CrossRef]
- Estep, A.S.; Sanscrainte, N.D.; Okech, B.A. Aedes aegypti Knockdown Resistance Mutations and Dengue Virus Infection in Haiti. J. Am. Mosq. Control Assoc. 2024, 40, 102–108. [Google Scholar] [CrossRef]
- Delannay, C.; Goindin, D.; Kellaou, K.; Ramdini, C.; Gustave, J.; Vega-Rua, A. Multiple insecticide resistance in Culex quinquefasciatus populations from Guadeloupe (French West Indies) and associated mechanisms. PLoS ONE 2018, 13, e0199615. [Google Scholar] [CrossRef]
- Nchoutpouen, E.; Talipouo, A.; Djiappi-Tchamen, B.; Djamouko-Djonkam, L.; Kopya, E.; Ngadjeu, C.S.; Doumbe-Belisse, P.; Awono-Ambene, P.; Kekeunou, S.; Wondji, C.S.; et al. Culex species diversity, susceptibility to insecticides and role as potential vector of Lymphatic filariasis in the city of Yaounde, Cameroon. PLoS Negl. Trop. Dis. 2019, 13, e0007229. [Google Scholar] [CrossRef] [PubMed]
- Ukpai, O.; Ekedo, C. Insecticide susceptibility status of Culex quinquefasciatus [Diptera: Culicidae] in Umudike, Ikwuano LGA Abia State, Nigeria. Int. J. Mosq. Res. 2019, 6, 114–118. [Google Scholar]
- Davis, E.L.; Prada, J.; Reimer, L.J.; Hollingsworth, T.D. Modelling the Impact of Vector Control on Lymphatic Filariasis Programs: Current Approaches and Limitations. Clin. Infect. Dis. 2021, 72, S152–S157. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2024; Available online: https://www.R-project.org/ (accessed on 20 September 2025).
- Reinert, J.F.; Kaiser, P.E.; Seawright, J.A. Analysis of the Anopheles (Anopheles) quadrimaculatus complex of sibling species (Diptera: Culicidae) using morphological, cytological, molecular, genetic, biochemical, and ecological techniques in an integrated approach. J. Am. Mosq. Control Assoc. 1997, 13, 1–102. [Google Scholar]
- McCall, P.J.; Eaton, G. Olfactory memory in the mosquito Culex quinquefasciatus. Med. Vet. Entomol. 2001, 15, 197–203. [Google Scholar] [CrossRef]
- Pridgeon, J.W.; Pereira, R.M.; Becnel, J.J.; Allan, S.A.; Clark, G.G.; Linthicum, K.J. Susceptibility of Aedes aegypti, Culex quinquefasciatus Say, and Anopheles quadrimaculatus Say to 19 pesticides with different modes of action. J. Med. Entomol. 2008, 45, 82–87. [Google Scholar] [CrossRef]
- Estep, A.S.; Sanscrainte, N.D.; Stuck, J.; Unlu, I.; Prasauskas, A.; Mundis, S.J.; Cotter, N.; Romero-Weaver, A.L.; Fedirko, T.J.; Kendziorski, N.L.; et al. The L1014F knockdown resistance mutation is not a strong correlate of phenotypic resistance to pyrethroids in Florida populations of Culex quinquefasciatus. Insects 2024, 15, 197. [Google Scholar] [CrossRef]
- Unlu, I.; Buckner, E.A.; Medina, J.; Vasquez, C.; Cabrera, A.; Romero-Weaver, A.L.; Ramirez, D.; Kendziorski, N.L.; Kosinski, K.J.; Fedirko, T.J.; et al. Insecticide resistance of Miami-Dade Culex quinquefasciatus populations and initial field efficacy of a new resistance-breaking adulticide formulation. PLoS ONE 2024, 19, e0296046. [Google Scholar] [CrossRef] [PubMed]
- Kolde, R. Pheatmap: Pretty Heatmaps. Available online: https://CRAN.R-project.org/package=pheatmap (accessed on 20 September 2025).
- Boyd, A.; Won, K.Y.; McClintock, S.K.; Donovan, C.V.; Laney, S.J.; Williams, S.A.; Pilotte, N.; Streit, T.G.; Beau de Rochars, M.V.; Lammie, P.J. A community-based study of factors associated with continuing transmission of lymphatic filariasis in Leogane, Haiti. PLoS Negl. Trop. Dis. 2010, 4, e640. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Haiti—Traveler View. Available online: https://wwwnc.cdc.gov/travel/destinations/traveler/none/haiti (accessed on 10 March 2026).
- Edmond, B.; World Bank. Haiti Boosts Tourism in the North and Connects Artisans with Visitors. Available online: https://www.worldbank.org/en/news/feature/2015/04/30/tourisme-nord-haiti-citadelle-sans-soucis-ramiers (accessed on 10 March 2026).
- Liu, J.; Wang, Y.; Liu, P.; Yu, X.; Tan, A.; Zeng, J.; Li, L.; Qiu, X. Detection of Target Site Mutations in the Acetylcholinesterase and Voltage-Gated Sodium Channel in Field Populations of Culex quinquefasciatus and Cx. tritaeniorhynchus from Southern Sichuan Region of China. J. Am. Mosq. Control Assoc. 2023, 39, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Soto, A.; Remue, L.; Rosales Rosas, A.L.; De Coninck, L.; Verwimp, S.; Bouckaert, J.; Vanwinkel, M.; Matthijnssens, J.; Delang, L. First Report of Mutations Associated with Pyrethroid (L1014F) and Organophosphate (G119S) Resistance in Belgian Culex (Diptera: Culicidae) Mosquitoes. J. Med. Entomol. 2022, 59, 2072–2079. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Dong, Y.; Ran, X.; Wu, Z.; Guo, X.; Zhang, Y.; Xing, D.; Yan, T.; Wang, G.; Zhu, X.; et al. Point mutations associated with organophosphate and carbamate resistance in Chinese strains of Culex pipiens quinquefasciatus (Diptera: Culicidae). PLoS ONE 2014, 9, e95260. [Google Scholar] [CrossRef]
- Talipouo, A.; Mavridis, K.; Nchoutpouen, E.; Djiappi-Tchamen, B.; Fotakis, E.A.; Kopya, E.; Bamou, R.; Kekeunou, S.; Awono-Ambene, P.; Balabanidou, V.; et al. High insecticide resistance mediated by different mechanisms in Culex quinquefasciatus populations from the city of Yaounde, Cameroon. Sci. Rep. 2021, 11, 7322. [Google Scholar] [CrossRef]
- Molina-Cruz, A.; Lehmann, T.; Knockel, J. Could culicine mosquitoes transmit human malaria? Trends Parasitol. 2013, 29, 530–537. [Google Scholar] [CrossRef]
- Weppelmann, T.A.; Burne, A.; von Fricken, M.E.; Elbadry, M.A.; Beau De Rochars, M.; Boncy, J.; Okech, B.A. A Tale of Two Flaviviruses: A Seroepidemiological Study of Dengue Virus and West Nile Virus Transmission in the Ouest and Sud-Est Departments of Haiti. Am. J. Trop. Med. Hyg. 2017, 96, 135–140. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. West Nile Virus Surveillance and Control Guidelines. Available online: https://www.cdc.gov/west-nile-virus/media/pdfs/2024/08/WestNileVirus-SurveillanceControlGuidelines_508-h.pdf (accessed on 20 September 2025).
- Davis, A.P.; Grondin, C.J.; Johnson, R.J.; Sciaky, D.; McMorran, R.; Wiegers, J.; Wiegers, T.C.; Mattingly, C.J. The Comparative Toxicogenomics Database: Update 2019. Nucleic Acids Res. 2019, 47, D948–D954. [Google Scholar] [CrossRef]
- Waters, M.D.; Fostel, J.M. Toxicogenomics and systems toxicology: Aims and prospects. Nat. Rev. Genet. 2004, 5, 936–948. [Google Scholar] [CrossRef]
- Pirmohamed, M. Pharmacogenomics: Current status and future perspectives. Nat. Rev. Genet. 2023, 24, 350–362. [Google Scholar] [CrossRef]
- Relling, M.V.; Evans, W.E. Pharmacogenomics in the clinic. Nature 2015, 526, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, M.J.; Corbel, V.; Weetman, D.; Wilding, C.S.; Williamson, M.S.; Black, W.C. Does kdr genotype predict insecticide resistance phenotype in mosquitoes? Trends Parasitol. 2009, 25, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Estep, A.S.; Sanscrainte, N.D.; Waits, C.M.; Bernard, S.J.; Lloyd, A.M.; Lucas, K.J.; Buckner, E.A.; Vaidyanathan, R.; Morreale, R.; Conti, L.A.; et al. Quantification of permethrin resistance and kdr alleles in Florida strains of Aedes aegypti (L.) and Aedes albopictus (Skuse). PLoS Negl. Trop. Dis. 2018, 12, e0006544. [Google Scholar] [CrossRef] [PubMed]
- Estep, A.S.; Sanscrainte, N.D.; Farooq, M.; Lucas, K.J.; Heinig, R.L.; Norris, E.J.; Becnel, J.J. Impact of Aedes aegypti V1016I and F1534C knockdown resistance genotypes on operational interventions. Sci. Rep. 2025, 15, 10146. [Google Scholar] [CrossRef] [PubMed]




| Primer Name | Sequence |
|---|---|
| kdr_1014F [13] | TTCACGCTGGAATACTCACGACA |
| kdr_1014L [13] | GGGCGGCGGGCAGGGCGGCGGGGGCGGGGTTCACGCTGGAATACTCACGACTA |
| kdr_1014S [13] | AGCGCGGAGCGCGGTTCACGCTGGAATACTCACGACTG |
| kdr_1014r [13] | GGATCGAATCCATGTGGGACTGCAT |
| AchE_2340S | CCGGCAGGCCGACGGCGACGACTGTGGATCTTCGGGGTTA |
| AchE_2340G | CTGTGGATCTTCGGGGGTG |
| AchE_2362_r | GTGGTCGTACACGTCCAGCG |
| Cxq/n_Cxq | GCGGGCAGGGCGGCGGGGGCGGGGGGAGCTCCAGATATGGCCTTT |
| Cxq/n_Cxn | GGAGCTCCTGATATAGCTTTC |
| Cxq/n_r | ATGAAGGAGGTAGTATTCAAAAACTTAT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tanachaiwiwat, P.; Sanscrainte, N.D.; Okech, B.A.; Estep, A.S. Insecticide Resistance Mutations, Enzymatic Activity, and Pathogen Infection in Culex quinquefasciatus from Haiti. Insects 2026, 17, 331. https://doi.org/10.3390/insects17030331
Tanachaiwiwat P, Sanscrainte ND, Okech BA, Estep AS. Insecticide Resistance Mutations, Enzymatic Activity, and Pathogen Infection in Culex quinquefasciatus from Haiti. Insects. 2026; 17(3):331. https://doi.org/10.3390/insects17030331
Chicago/Turabian StyleTanachaiwiwat, Primrose, Neil D. Sanscrainte, Bernard A. Okech, and Alden S. Estep. 2026. "Insecticide Resistance Mutations, Enzymatic Activity, and Pathogen Infection in Culex quinquefasciatus from Haiti" Insects 17, no. 3: 331. https://doi.org/10.3390/insects17030331
APA StyleTanachaiwiwat, P., Sanscrainte, N. D., Okech, B. A., & Estep, A. S. (2026). Insecticide Resistance Mutations, Enzymatic Activity, and Pathogen Infection in Culex quinquefasciatus from Haiti. Insects, 17(3), 331. https://doi.org/10.3390/insects17030331





