Isolation and Quantification of L-Tryptophan from Protaetia brevitarsis seulensis Larvae as a Marker for the Quality Control of an Edible Insect Extract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. General Experimental Procedures
2.2. Edible Insect Material
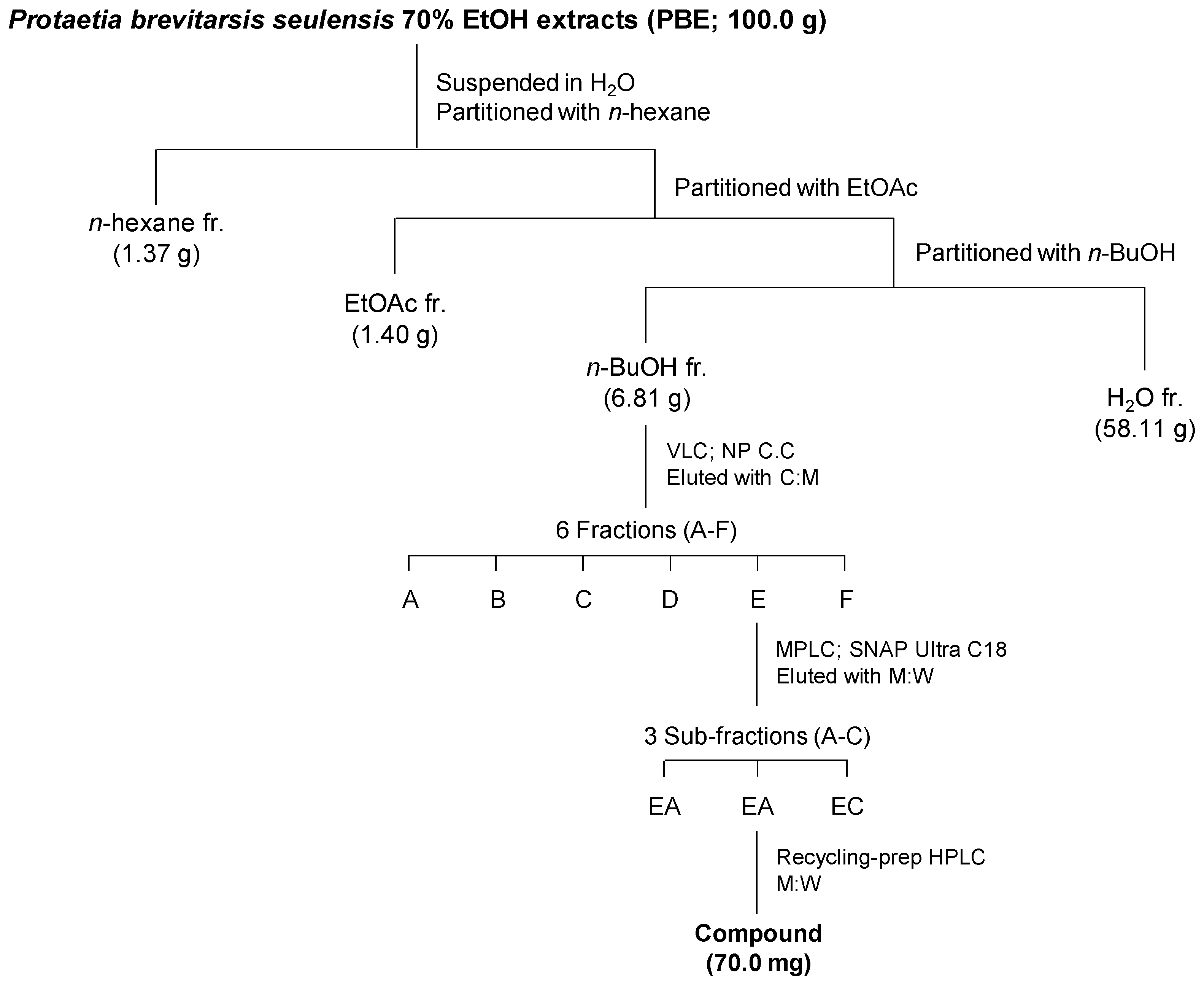
2.3. Isolation of the Major Compound
2.4. Preparation of Standard Solutions and Samples
2.5. Chromatographic Conditions
3. Results and Discussion
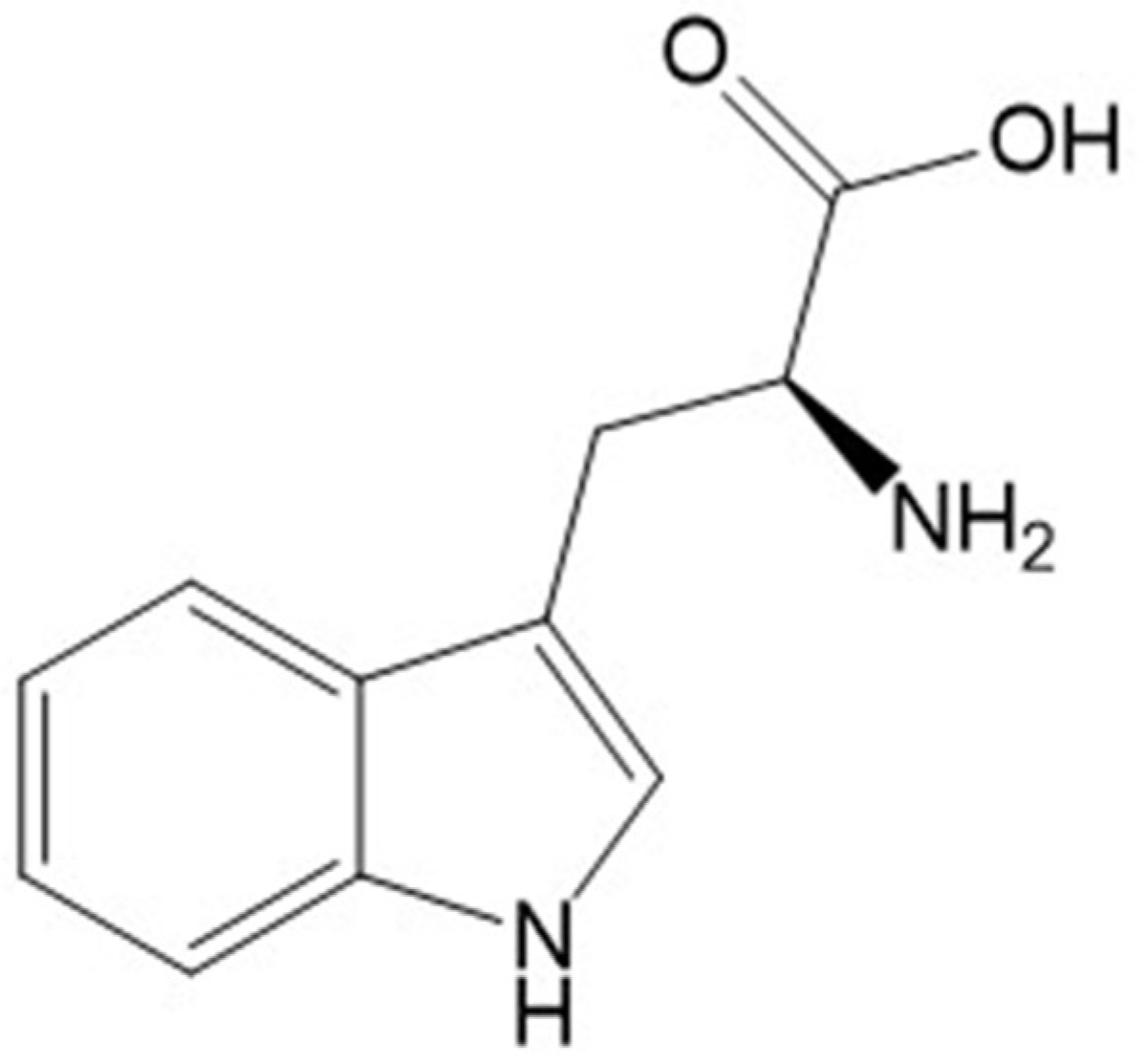
3.1. Analytical Characterization of a Major Compound from PBE
3.2. Isolation and Structural Characterization
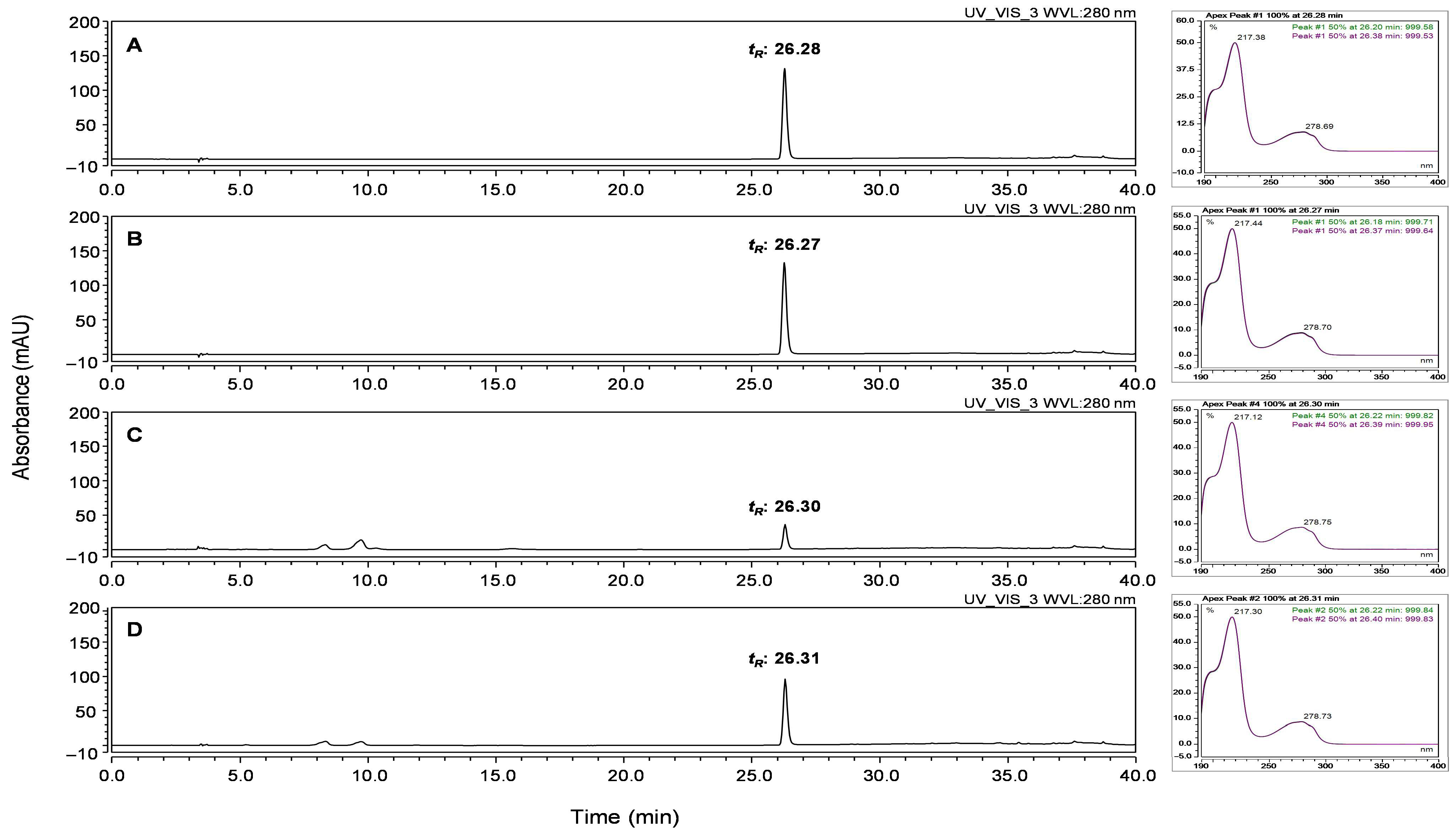
3.3. Optimization of HPLC Conditions for Quantitative Analysis
3.4. Quantitative Determination of L-Tryptophan in PBE
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- van Huis, A. Potential of insects as food and feed in assuring food security. Annu. Rev. Entomol. 2013, 58, 563–583. [Google Scholar] [CrossRef] [PubMed]
- Halloran, A.; Roos, N.; Eilenberg, J.; Cerutti, A.; Bruun, S. Life cycle assessment of edible insects for food protein: A review. Agron. Sustain. Dev. 2016, 36, 57. [Google Scholar] [CrossRef] [PubMed]
- Rumpold, B.A.; Schlüter, O.K. Nutritional composition and safety aspects of edible insects. Mol. Nutr. Food Res. 2013, 57, 802–823. [Google Scholar] [CrossRef] [PubMed]
- Payne, C.L.; Scarborough, P.; Rayner, M.; Nonaka, K. Are edible insects more or less ‘healthy’ than commonly consumed meats? A comparison using two nutrient profiling models developed to combat over- and undernutrition. Eur. J. Clin. Nutr. 2016, 70, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Dobermann, D.; Swift, J.A.; Field, L.M. Opportunities and hurdles of edible insects for food and feed. Nutr. Bull. 2017, 42, 293–308. [Google Scholar] [CrossRef]
- Williams, J.P.; Williams, J.R.; Kirabo, A.; Chester, D.; Peterson, M. Chapter 3—Nutrient Content and Health Benefits of Insects. In Insects as Sustainable Food Ingredients; Dossey, A.T., Morales-Ramos, J.A., Rojas, M.G., Eds.; Academic Press: San Diego, CA, USA, 2016; pp. 61–84. [Google Scholar] [CrossRef]
- Jantzen da Silva Lucas, A.; Menegon de Oliveira, L.; da Rocha, M.; Prentice, C. Edible insects: An alternative of nutritional, functional and bioactive compounds. Food Chem. 2020, 311, 126022. [Google Scholar] [CrossRef] [PubMed]
- Xavier, J.R.; Sanjay, B.S.; Gupta, D.; Mehta, S.; Chauhan, O.P. Bioactive compounds of foods: Phytochemicals and peptides. Food Humanit. 2024, 3, 100354. [Google Scholar] [CrossRef]
- Ferrazzano, G.F.; D’Ambrosio, F.; Caruso, S.; Gatto, R.; Caruso, S. Bioactive Peptides Derived from Edible Insects: Effects on Human Health and Possible Applications in Dentistry. Nutrients 2023, 15, 4611. [Google Scholar] [CrossRef] [PubMed]
- Yeo, H.; Youn, K.; Kim, M.; Yun, E.Y.; Hwang, J.S.; Jeong, W.S.; Jun, M. Fatty Acid Composition and Volatile Constituents of Protaetia brevitarsis Larvae. Prev. Nutr. Food Sci. 2013, 18, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Suh, H.J.; Kang, S.C. Antioxidant activity of aqueous methanol extracts of Protaetia brevitarsis Lewis (Coleoptera: Scarabaedia) at different growth stages. Nat. Prod. Res. 2012, 26, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Kang, C.; Lee, H.; Kim, E.; Kim, J.; Kwon, O.; Lee, H.; Kang, H.; Kim, C.; Jang, H. Effects of fermented aloe vera mixed diet on larval growth of Protaetia brevitarsis seulensis (Kolbe) (coleopteran:Cetoniidae) and protective effects of its extract against CCl4-induced hepatotoxicity in Sprague–Dawley rats. Entomol. Res. 2012, 42, 111–121. [Google Scholar] [CrossRef]
- Choi, R.-Y.; Kim, I.-W.; Ji, M.; Paik, M.-J.; Ban, E.-J.; Lee, J.H.; Hwang, J.S.; Kweon, H.; Seo, M. Protaetia brevitarsis seulensis larvae ethanol extract inhibits RANKL-stimulated osteoclastogenesis and ameliorates bone loss in ovariectomized mice. Biomed. Pharmacother. 2023, 165, 115112. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Seo, Y.H.; Song, J.H.; Kim, W.J.; Lee, J.H.; Moon, B.C.; Ang, M.J.; Kim, S.H.; Moon, C.; Lee, J.; et al. Neuroprotective Effect of Protaetia brevitarsis seulensis’ Water Extract on Trimethyltin-Induced Seizures and Hippocampal Neurodegeneration. Int. J. Mol. Sci. 2021, 22, 679. [Google Scholar] [CrossRef] [PubMed]
- Seo, Y.S.; Shin, N.R.; Nam, H.H.; Song, J.H.; Cheol Moon, B.; Choi, G.; Shin, I.S.; Kim, J.S. Effects of larval extracts from identified Protaetia brevitarsis seulensis against benign prostatic hyperplasia induced by testosterone in rats. Food Sci. Nutr. 2021, 9, 5361–5369. [Google Scholar] [CrossRef] [PubMed]
- Im, A.R.; Yang, W.K.; Park, Y.C.; Kim, S.H.; Chae, S. Hepatoprotective Effects of Insect Extracts in an Animal Model of Nonalcoholic Fatty Liver Disease. Nutrients 2018, 10, 735. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Hwang, I.H.; Kim, J.H.; Kim, M.A.; Hwang, J.S.; Kim, Y.H.; Na, M. Quinoxaline-, dopamine-, and amino acid-derived metabolites from the edible insect Protaetia brevitarsis seulensis. Arch. Pharmacal Res. 2017, 40, 1064–1070. [Google Scholar] [CrossRef] [PubMed]
- Yoon, C.H.; Jeon, S.H.; Ha, Y.J.; Kim, S.W.; Bang, W.Y.; Bang, K.H.; Gal, S.W.; Kim, I.S.; Cho, Y.S. Functional Chemical Components in Protaetia brevitarsis Larvae: Impact of Supplementary Feeds. Food. Sci. Anim. Resour. 2020, 40, 461–473. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, W.; Kim, M.A.; Hwang, J.S.; Na, M.; Bae, J.S. Inhibition of platelet aggregation and thrombosis by indole alkaloids isolated from the edible insect Protaetia brevitarsis seulensis (Kolbe). J. Cell Mol. Med. 2017, 21, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- la Cour, R.; Jørgensen, H.; Schjoerring, J.K. Improvement of Tryptophan Analysis by Liquid Chromatography-Single Quadrupole Mass Spectrometry Through the Evaluation of Multiple Parameters. Front. Chem. 2019, 7, 797. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Sun, S.; Chen, H.; Liu, M.; Zhang, L.; Wu, Z.; Li, J.; Wu, G. Analysis of Tryptophan and Its Metabolites by High-Performance Liquid Chromatography. In Amino Acid Analysis: Methods and Protocols; Alterman, M., Ed.; Methods in Molecular Biology; Humana Press: New York, NY, USA, 2019; Volume 2030, pp. 131–142. [Google Scholar] [CrossRef]
- Eid, S.M.; Farag, M.A.; Bawazeer, S. Underivatized Amino Acid Chromatographic Separation: Optimized Conditions for HPLC-UV Simultaneous Quantification of Isoleucine, Leucine, Lysine, Threonine, Histidine, Valine, Methionine, Phenylalanine, Tryptophan, and Tyrosine in Dietary Supplements. ACS Omega 2022, 7, 31106–31114. [Google Scholar] [CrossRef] [PubMed]
| Analyte | Range (µg/mL) | Linear Regression Equation 1 | Correlation Coefficient (n = 5) | RSD 2 (%) | LOD (μg/mL) | LOQ (μg/mL) | Content (μg/mg) |
|---|---|---|---|---|---|---|---|
| L-Tryptophan | 25–400 | y = 0.1551x + 0.6452 | 0.9999 | 1.42% | 3.28 | 9.95 | 1.93 ± 0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, H.J.; Li, W. Isolation and Quantification of L-Tryptophan from Protaetia brevitarsis seulensis Larvae as a Marker for the Quality Control of an Edible Insect Extract. Insects 2025, 16, 905. https://doi.org/10.3390/insects16090905
Yang HJ, Li W. Isolation and Quantification of L-Tryptophan from Protaetia brevitarsis seulensis Larvae as a Marker for the Quality Control of an Edible Insect Extract. Insects. 2025; 16(9):905. https://doi.org/10.3390/insects16090905
Chicago/Turabian StyleYang, Hye Jin, and Wei Li. 2025. "Isolation and Quantification of L-Tryptophan from Protaetia brevitarsis seulensis Larvae as a Marker for the Quality Control of an Edible Insect Extract" Insects 16, no. 9: 905. https://doi.org/10.3390/insects16090905
APA StyleYang, H. J., & Li, W. (2025). Isolation and Quantification of L-Tryptophan from Protaetia brevitarsis seulensis Larvae as a Marker for the Quality Control of an Edible Insect Extract. Insects, 16(9), 905. https://doi.org/10.3390/insects16090905







