Pollination of Enclosed Avocado Trees by Blow Flies (Diptera: Calliphoridae) and a Hover Fly (Diptera: Syrphidae)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Fly Colonies for Use in Multi-Tree Enclosure Trials
2.2. Multi-Tree Enclosures
2.2.1. Busselton (2021)
2.2.2. Pemberton (2021)
2.2.3. Capel (2022)
2.2.4. Capel (2023)
2.3. Fly Releases into the Enclosures
2.4. Methods of Assessment and Data Collection
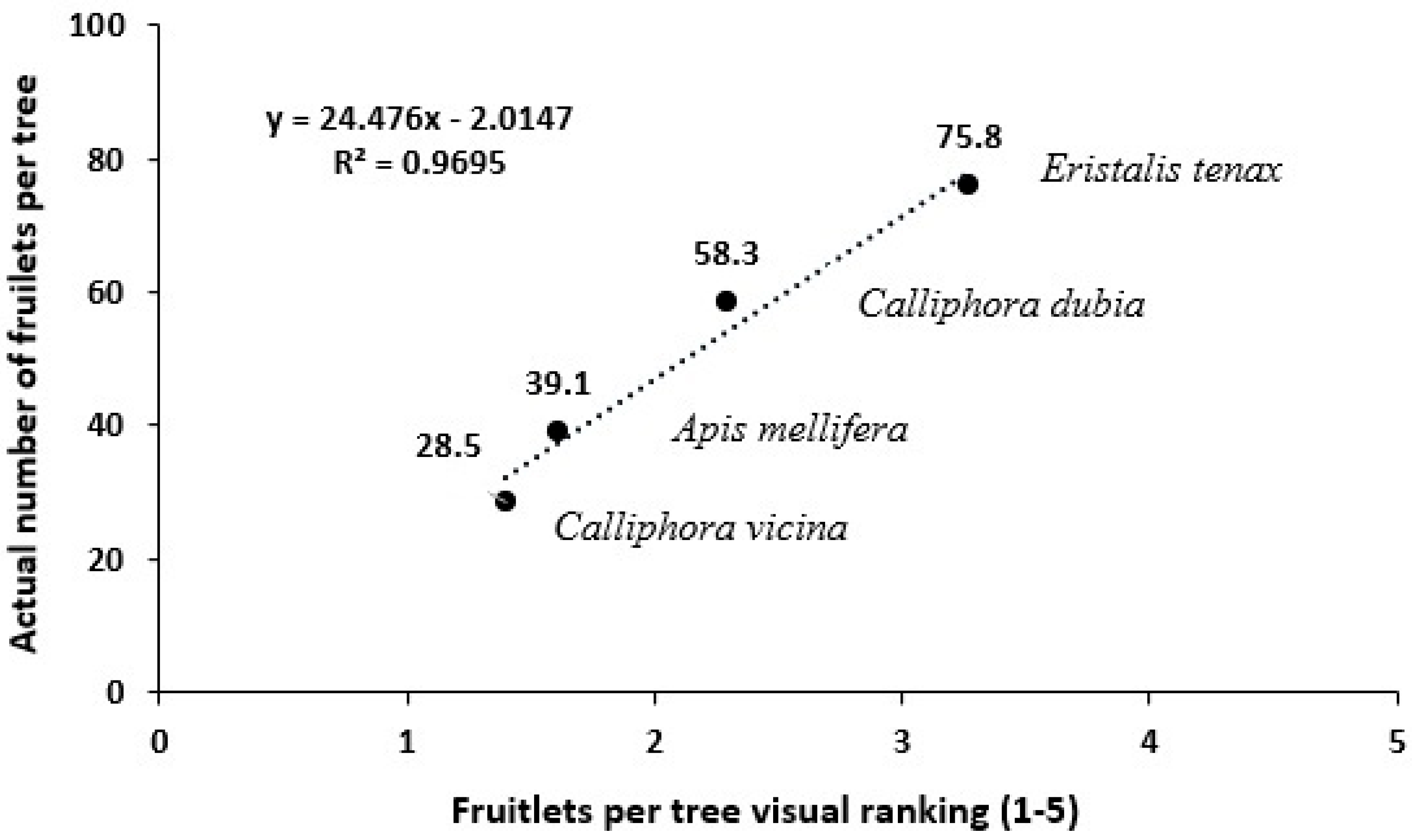
Visual Scoring System to Estimate Fruit Yield
2.5. Weather Data
2.6. Statistical Analysis
3. Results
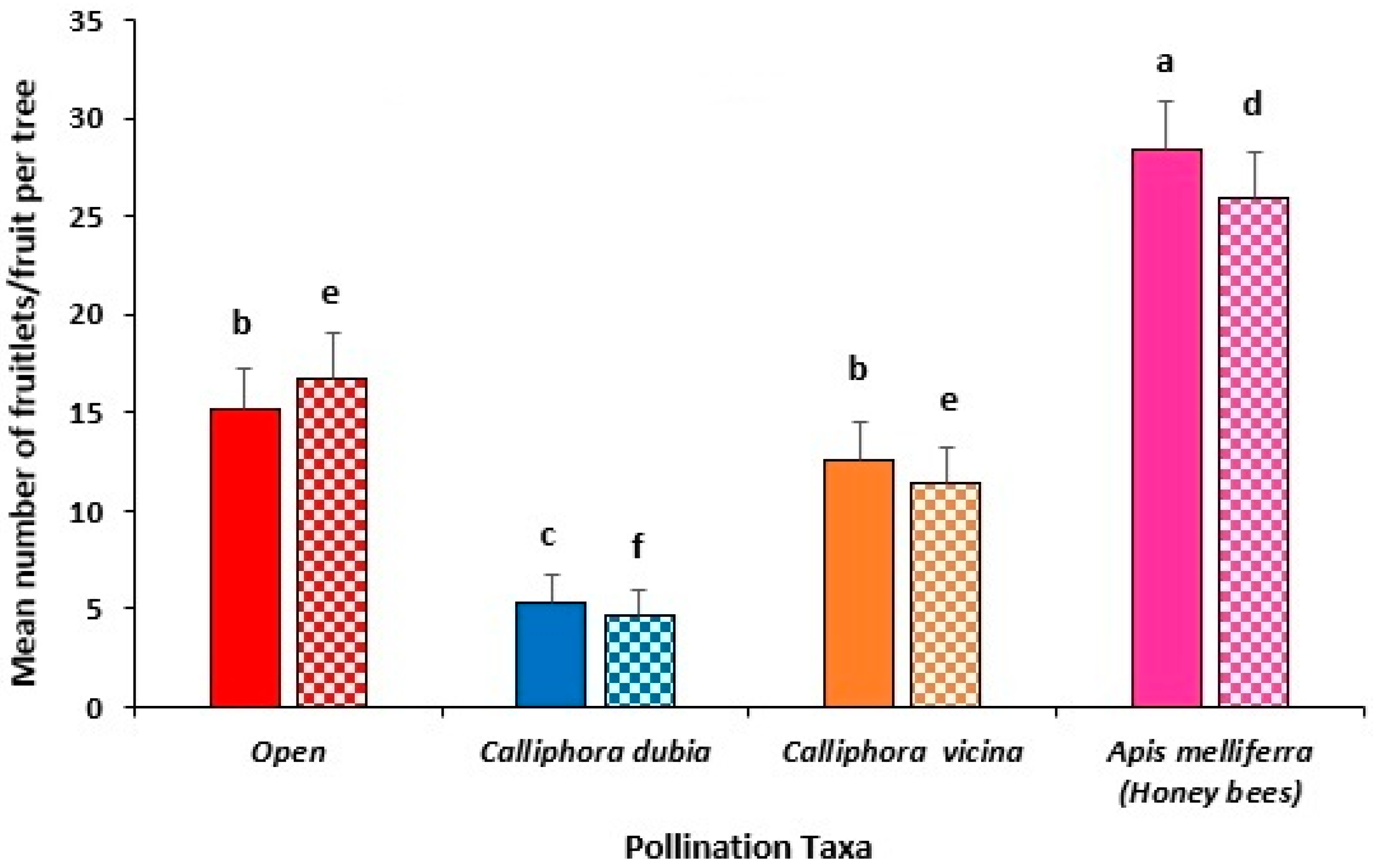
3.1. Year 1 Field Trial, Busselton (2021)
3.2. Year 1 Field Trial, Pemberton (2021)
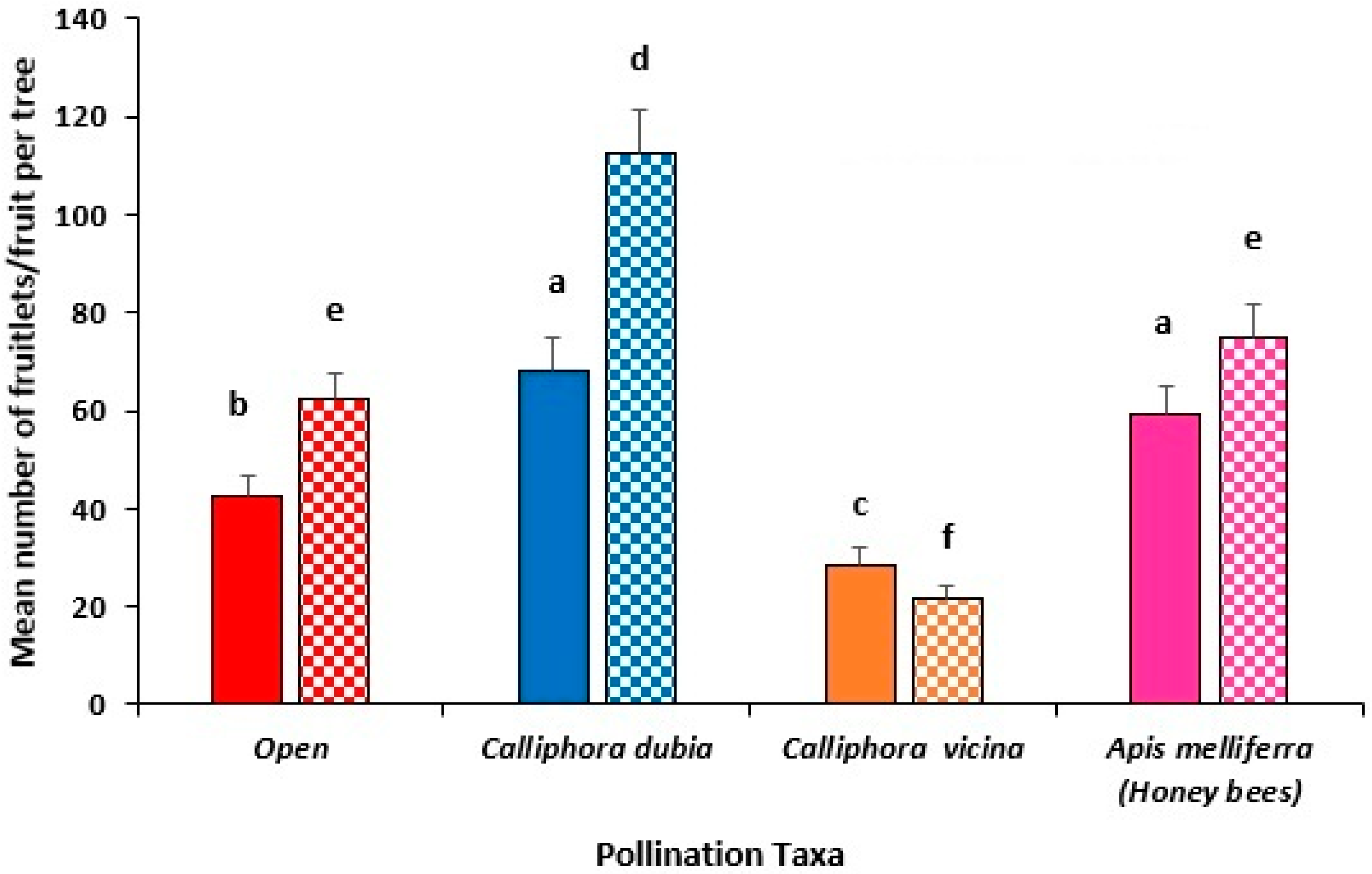
3.3. Year 2 Field Trial, Capel (2022)
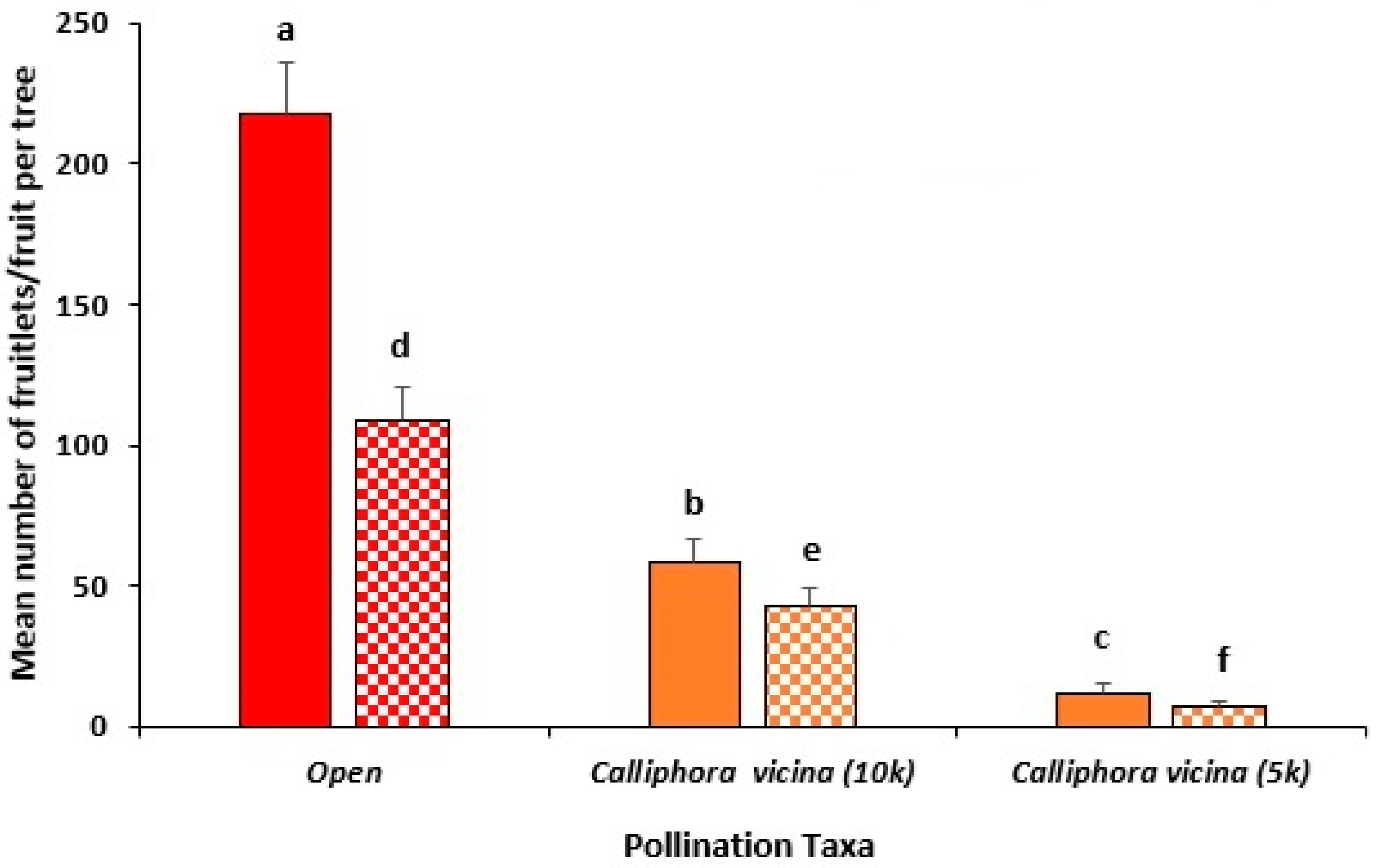
3.4. Year 3 Field Trial, Capel (2023)
Testing of Visual Scoring System to Estimate Fruit Yield
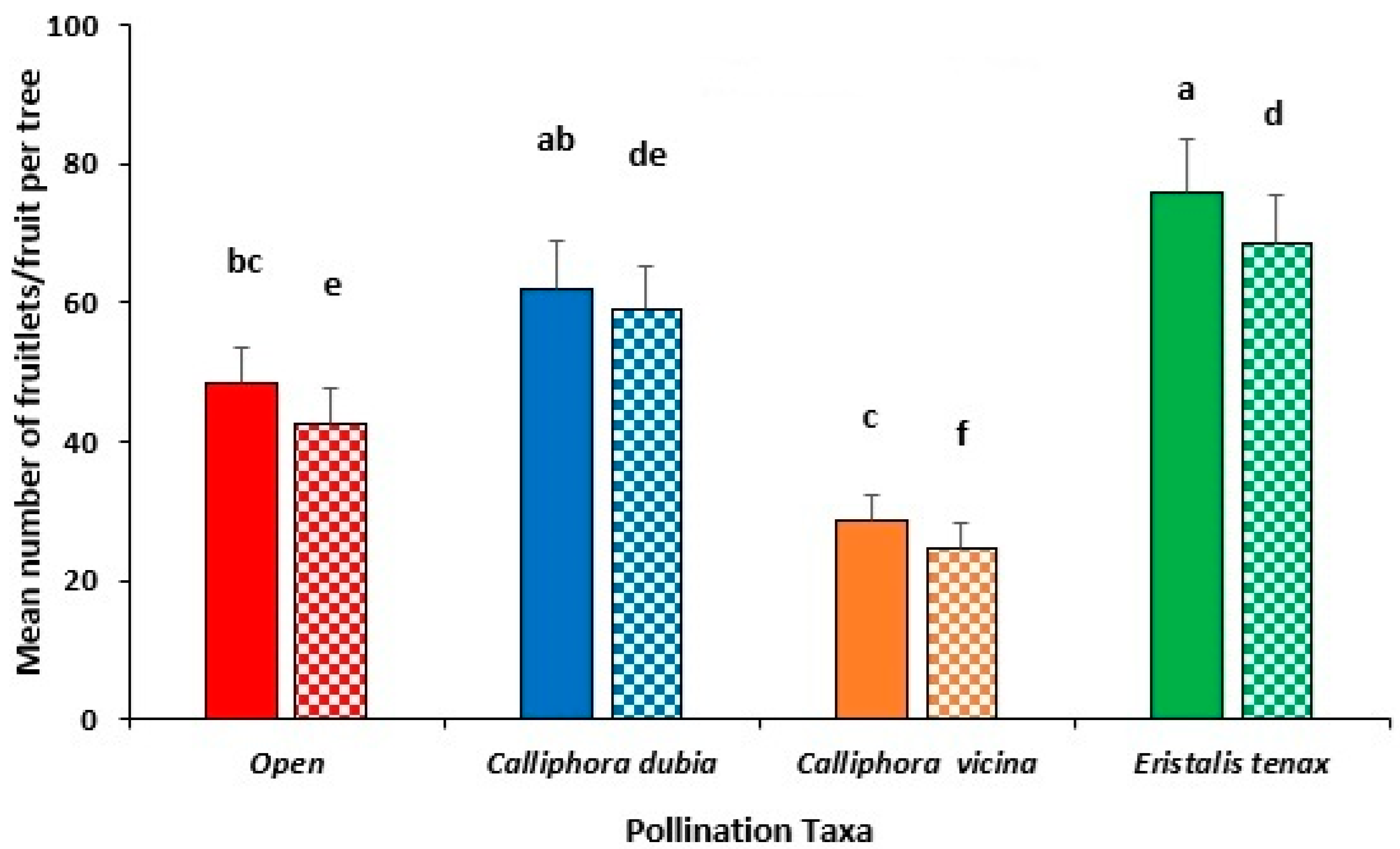
3.5. Comparative Analysis Across All Field Trials
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aizen, M.A.; Aguiar, S.; Biesmeijer, J.C.; Garibaldi, L.A.; Inouye, D.W.; Jung, C.; Martins, D.J.; Medel, R.; Morales, C.L.; Ngo, H.; et al. Global agricultural productivity is threatened by increasing pollinator dependence without a parallel increase in crop diversification. Glob. Change Biol. 2019, 25, 3516–3527. [Google Scholar] [CrossRef]
- Free, J.B. Insect Pollination of Crops; Academic Press: London, UK, 1993. [Google Scholar]
- Brunet, J.; Fragoso, F.P. What are the main reasons for the worldwide decline in pollinator populations? CABI Rev. 2024, 19, 1. [Google Scholar] [CrossRef]
- Janousek, W.M.; Douglas, M.R.; Cannings, S.; Graves, T.A. Recent and future declines of a historically widespread pollinator linked to climate, land cover, and pesticides. Proc. Natl. Acad. Sci. USA 2023, 120, e2211223120. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Bayo, F.; Goka, K. Pesticide residues and bees—A risk assessment. PLoS ONE 2014, 9, e94482. [Google Scholar] [CrossRef] [PubMed]
- Tennakoon, S.; Apan, A.; Maraseni, T. Unravelling the impact of climate change on honey bees: An ensemble modelling approach to predict shifts in habitat suitability in Queensland, Australia. Ecol. Evol. 2024, 14, e11300. [Google Scholar] [CrossRef]
- Kendall, D.A. The viability and compatibility of pollen on insects visiting apple blossom. J. Appl. Ecol. 1973, 10, 847–853. [Google Scholar] [CrossRef]
- Williams, R.R.; Wilson, D. Towards Regulated Cropping: A Report of Recent Fruit-Set Experiments in British Orchards; Grower Books: London, UK, 1970. [Google Scholar]
- Cook, D.F.; Voss, S.C.; Finch, J.T.; Rader, R.C.; Cook, J.M.; Spurr, C.J. The role of flies as pollinators of horticultural crops: An Australian case study with worldwide relevance. Insects 2020, 11, 341. [Google Scholar] [CrossRef] [PubMed]
- Aizen, M.A.; Harder, L.D. The global stock of domesticated honey bees is growing slower than agricultural demand for pollination. Curr. Biol. 2009, 19, 915–918. [Google Scholar] [CrossRef]
- Larson, B.M.H.; Kevan, P.G.; Inouye, D.W. Flies and flowers: Taxonomic diversity of anthophiles and pollinators. Can. Entomol. 2001, 133, 439–465. [Google Scholar] [CrossRef]
- Ollerton, J.; Winfree, R.; Tarrant, S. How many flowering plants are pollinated by animals? Oikos 2011, 120, 321–326. [Google Scholar] [CrossRef]
- Rader, R.; Cunningham, S.A.; Howlett, B.G.; Inouye, D.W. Non-bee insects as visitors and pollinators of crops: Biology, ecology, and management. Annu. Rev. Entomol. 2020, 65, 391–407. [Google Scholar] [CrossRef]
- Ssymank, A.; Kearns, C.A.; Pape, T.; Thompson, C. Pollinating flies (Diptera): A major contribution to plant diversity and agricultural production. Biodivers 2008, 9, 86–89. [Google Scholar] [CrossRef]
- Howlett, B. Hybrid carrot seed crop pollination by the fly Calliphora vicina (Diptera: Calliphoridae). J. Appl. Entomol. 2012, 136, 421–430. [Google Scholar] [CrossRef]
- Read, S.; Howlett, B.; Jesson, L.; Pattemore, D.E. Insect visitors to avocado flowers in the Bay of Plenty, New Zealand. N. Z. Plant Prot. 2017, 70, 38–44. [Google Scholar] [CrossRef]
- Willcox, B.K.; Howlett, B.G.; Robson, A.J. Evaluating the taxa that provide shared pollination services across multiple crops and regions. Sci. Rep. 2019, 9, 13538. [Google Scholar] [CrossRef] [PubMed]
- Jirón, L.F.; Hedstrom, I. Pollination ecology of mango (Mangifera indica L.) (Anacardiaceae) in the Neotropic region. Turrialba 1985, 35, 269–277. [Google Scholar]
- Dag, A.; Gazit, S. Mango pollinators in Israel. J. Appl. Hortic. Lucknow. 2000, 2, 39–43. [Google Scholar] [CrossRef]
- Albano, S.; Salvado, E.; Duarte, S.; Mexia, A.; Borges, P. Pollination effectiveness of different strawberry floral visitors in Ribatejo, Portugal: Selection of potential pollinators. Part 2. Adv. Hortic. Sci. 2009, 23, 246–253. Available online: https://www.jstor.org/stable/42883505 (accessed on 1 April 2025).
- Clement, S.L.; Hellier, B.C.; Elberson, L.R.; Staska, R.T.; Evans, M.A. Flies (Diptera: Muscidae: Calliphoridae) are efficient pollinators of Allium ampeloprasum L. (Alliaceae) in field cages. J. Econ. Entomol. 2007, 100, 131–135. [Google Scholar] [CrossRef]
- Sajjad, A.; Saeed, S.; Masood, A. Pollinator community of onion (Allium cepa L.) and its role in crop reproductive success. Pak. J. Zool. 2008, 40, 451–456. [Google Scholar]
- Toivonen, M.; Karimaa, A.-E.; Herzon, I.; Kuussaari, M. Flies are important pollinators of mass-flowering caraway and respond to landscape and floral factors differently from honeybees. Agric. Ecosyst. Environ. 2022, 323, 107698. [Google Scholar] [CrossRef]
- Jauker, F.; Wolters, V. Hover flies are efficient pollinators of oilseed rape. Oecologia 2008, 156, 819. [Google Scholar] [CrossRef]
- Inouye, D.W.; Larson, B.M.H.; Ssymank, A.; Kevan, P.G. Flies and flowers III: Ecology of foraging and pollination. J. Pollinat. Ecol. 2015, 16, 115–133. [Google Scholar] [CrossRef]
- Carabali-Banguero, D.; Montoyera-Lerma, J.; Carabalí-Muñoz, A. Diptera associated with the flowering of avocado, Persea americana Mill cv. Hass in Cauca, Colombia. Colomb. Biota 2018, 19, 92–111. [Google Scholar] [CrossRef]
- Rader, R.; Howlett, B.G.; Cunningham, S.A.; Westcott, D.A.; Newstrom-Lloyd, L.E.; Walker, M.K.; Teulon, D.A.J.; Edwards, W. Alternative pollinator taxa are equally efficient but not as effective as the honeybee in a mass flowering crop. J. Appl. Ecol. 2009, 46, 1080–1087. [Google Scholar] [CrossRef]
- Howlett, B.G.; Gee, M. The potential management of the drone fly (Eristalis tenax) as a crop pollinator in New Zealand. N. Z. Plant Protect 2019, 72, 221–230. [Google Scholar] [CrossRef]
- Klecka, J.; Hadrava, J.; Biella, P.; Akter, A. Flower visitation by hoverflies (Diptera: Syrphidae) in a temperate plant-pollinator network. PeerJ 2018, 6, e6025. [Google Scholar] [CrossRef]
- Bohart, G.E.; Nye, W.P.; Hawthorn, L.R. Onion pollination as affected by different levels of pollinator activity. Utah Agric. Exp. Stn. Bull. 1970, 482. Available online: https://digitalcommons.usu.edu/piru_pubs/271/ (accessed on 1 April 2025).
- Jarlan, A.; De Oliveira, D.; Gingras, J. Pollination by Eristalis tenax (Diptera: Syrphidae) and seed set of greenhouse sweet pepper. J. Econ. Entomol. 1997, 90, 1646–1649. [Google Scholar] [CrossRef]
- Raguso, R.A. Don’t forget the flies: Dipteran diversity and its consequences for floral ecology and evolution. Appl. Entomol. Zool. 2020, 55, 1–7. [Google Scholar] [CrossRef]
- Upchurch, A.; Spurr, C.J.; Quarrell, S.R.; Rowbottom, R.M.; Allen, G.R. Toward optimising reproductive output of Eristalis tenax (Diptera: Syrphidae) for commercial mass rearing systems. Austral Entomol. 2023, 62, 360–371. [Google Scholar] [CrossRef]
- Rader, R.; Edwards, W.; Westcott, D.A.; Cunningham, S.A.; Howlett, B.G. Diurnal effectiveness of pollination by bees and flies in agricultural Brassica rapa: Implications for ecosystem resilience. Basic. Appl. Ecol. 2013, 14, 20–27. [Google Scholar] [CrossRef]
- Stavert, J.R.; Pattemore, D.E.; Bartomeus, I.; Gaskett, A.C.; Beggs, J.R. Exotic flies maintain pollination services as native pollinators decline with agricultural expansion. J. Appl. Ecol. 2018, 55, 1737–1746. [Google Scholar] [CrossRef]
- Broussard, M.A.; Howlett, B.G.; Evans, L.J.; McBrydie, H.; Cutting, B.T.; Read, S.F.J.; Pattemore, D.E. Pollinator identify and behavior affect pollination in kiwifruit (Actinidia chinensis Planch). Peer J. 2022, 10, e12963. [Google Scholar] [CrossRef]
- Gervais, A.; Chagnon, M.; Fournier, V. Diversity and pollen loads of flower flies (Diptera: Syrphidae) in cranberry crops. Ann. Entomol. Soc. Am. 2018, 111, 326–344. [Google Scholar] [CrossRef]
- Schittenhelm, S.; Gladis, T.; Rao, V.R. Efficiency of various insects in germplasm regeneration of carrot, onion and turnip rape accessions. Plant Breed. 1997, 116, 369–375. [Google Scholar] [CrossRef]
- Gaffney, A.; Allen, G.R.; Brown, P.H. Insect visitation to flowering hybrid carrot seed crops. N. Z. J. Crop Hortic. Sci. 2011, 39, 79–93. [Google Scholar] [CrossRef]
- Suvamayathipat, W. Mango Cultivation; Kasetsart University: Bangkok, Thailand, 1984. [Google Scholar]
- Windriyanti, W.; Karindah, S.; Sulistyowati, L.; Rahardjo, B.T. Insect diversity visitors to gadung 21 variety of mango flower on off season and on season. JBES 2019, 14, 63–70. Available online: https://innspub.net/insect-diversity-visitors-to-gadung-21-variety-of-mango-flower-on-off-season-and-on-season/ (accessed on 10 July 2025).
- Currah, L.; Ockendon, D.J. Pollination activity by blowflies and honeybees on onions in breeders’ cages. Ann. Appl. Biol. 1984, 105, 167–176. [Google Scholar] [CrossRef]
- Jones, H.; Emsweller, S. The use of flies as onion pollinators. Proc. Am. Soc. Hortic. Sci. 1934, 31, 160–164. [Google Scholar]
- Howlett, B.G.; Davidson, M.M.; Pattemore, D.E.; Walker, M.K.; Nelson, W.R. Seasonality of calliphorid and sarcophagid flies across Canterbury arable farms requiring pollinators. N. Z. Plant Prot. 2016, 69, 290–295. [Google Scholar] [CrossRef]
- Dymond, K.; Celis-Diez, J.L.; Potts, S.G.; Howlett, B.G.; Willcox, B.K.; Garratt, M.P.D. The role of insect pollinators in avocado production: A global review. J. Appl. Entomol. 2021, 145, 369–383. [Google Scholar] [CrossRef]
- Bushuru, E. Diversity and Pollination Activity of Flower Visiting Insect Associated with Avocado Along the Slopes of Taita Hills in Kenya. Master’s Thesis, Masinde Muliro University of Science and Technology, Kakamega, Kenya, 2015. [Google Scholar]
- Can-Alonzo, C.; Quezada-Euan, J.J.; Xiu-Ancona, P.; Moo-Valle, H.; Valdovinos-Nunez, G.; Medina-Peralta, S. Pollination of ‘criollo’ avocados (Persea americana) and the behaviour of associated bees in subtropical Mexico. J. Apic. Res. 2005, 44, 3–8. [Google Scholar] [CrossRef]
- Ish-Am, G.; Eisikowitch, D. The behaviour of honey bees (Apis mellifera) visiting avocado (Persea americana) flowers and their contribution to its pollination. J. Apic. Res. 1993, 32, 75–186. [Google Scholar] [CrossRef]
- Woodcock, B.A.; Garratt, M.P.D.; Powney, G.D.; Shaw, R.F.; Osborne, J.L.; Soroka, J.; Lindström, S.A.M.; Stanley, D.; Ouvrard, P.; Edwards, M.E.; et al. Meta-analysis reveals that pollinator functional diversity and abundance enhance crop pollination and yield. Nat. Commun. 2019, 10, 1481. [Google Scholar] [CrossRef]
- Biesmeijer, J.C.; Roberts, S.P.M.; Reemer, M.; Ohlemüller, R.; Edwards, M.; Peeters, T.; Schaffers, A.P.; Potts, S.G.; Kleukers, R.; Thomas, C.D.; et al. Parallel declines in pollinators and insect-pollinated plants in Britain and the Netherlands. Science 2006, 313, 351–354. [Google Scholar] [CrossRef]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global pollinator declines: Trends, impacts and drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef]
- Peterson, P.A. Avocado flower pollination and fruit set. In California Avocado Society Yearbook; California Avocado Society: San Juan Capistrano, CA, USA, 1955; Volume 39, pp. 163–169. [Google Scholar] [CrossRef]
- Castañeda-Vildózola, A.; Equihua-Martinez, A.; Valdéz-Carrasco, J.; Barrientos-Priego, A.F.; Ish-Am, G.; Gazit, S. Insectos polin-izadores del aguacatero en los estados de México y Michoacán, México. Rev. Chapingo Ser. Hortic. 1999, 5, 129–136. [Google Scholar]
- Pérez-Balam, J.; Quezada-Euán, J.; Alfaro-Bates, R.; Medina, S.; McKendrick, L.; Soro, A.; Paxton, R.J. The contribution of honey bees, flies and wasps to avocado (Persea americana) pollination in southern Mexico. J. Pollinat. Ecol. 2012, 8, 42–47. [Google Scholar] [CrossRef]
- Sagwe, R.N. Pollinator Diversity, Pollination Deficits, and Pollination Efficiency in Avocado (Persea americana) Production Across Different Landscapes in Murang’a County, Kenya. Ph.D. Thesis, Universitat Würzburg, Würzburg, Kenya, 2022. [Google Scholar]
- Sagwe, R.N.; Peters, M.K.; Dubois, T.; Steffan-Dewenter, I.; Lattorff, H.M.G. Pollinator efficiency of avocado (Persea americana) flower insect visitors. Ecol. Solut. Evid. 2022, 3, e12178. [Google Scholar] [CrossRef]
- Mulwa, J.M. Determining Pollinators, Floral Calendar and the EFFECT of Pollination Deficit of Avocado Persea americana Mill in Kandara, Murang’a County, Kenya. Master’s Thesis, Kenyatta University, Nairobi, Kenya, 2019. [Google Scholar]
- Australian Horticulture Statistics Handbook (2023/24). Available online: www.horticulture.com.au (accessed on 12 June 2025).
- Avocados Australia. National Avocado Tree Crop Map for the Australian Avocado Industry. 2024. Available online: https://www.avocado.org.au (accessed on 17 October 2024).
- Howlett, B. Optimising Pollination of Macadamia and Avocado in Australia; Horticulture Innovation Australia, Final Report, Project: MT13060; Horticulture Innovation Australia Limited: Sydney, NSW, Australia, 2017. [Google Scholar]
- Cook, D.F.; Voss, S.C.; Deyl, R.A.; Howse, E.T.; Foley, J.; Norrish, B.; Delroy, N.; Shivananjappa, S.L. Blow flies (Diptera: Calliphoridae) ability to pollinate Hass avocado (Persea americana) trees within paired tree enclosures. J. Appl. Entomol. 2023, 147, 577–591. [Google Scholar] [CrossRef]
- Vithanage, H.I.M.V. Insect pollination of avocado and macadamia. Acta Hortic. 1986, 175, 97–102. Available online: https://hdl.handle.net/102.100.100/272477 (accessed on 4 June 2025). [CrossRef]
- Vithanage, H.I.N.V. The role of the European honeybee (Apis mellifera L.) in avocado pollination. J. Hortic. Sci. 1990, 65, 81–86. [Google Scholar] [CrossRef]
- Cook, D.F.; Deyl, R.A.; Mickan, B.S.; Howse, E.T. Yield of southern highbush blueberry (Vaccinium corymbosum) using the fly Calliphora albifrontalis, (Diptera: Calliphoridae) as a pollinator. Austral Entomol. 2020, 59, 345–352. [Google Scholar] [CrossRef]
- Beekman, M.; Sumpter, D.J.T.; Seraphides, N.; Ratnieks, F.L.W. Comparing foraging behaviour of small and large honey-bee colonies by decoding waggle dances made by foragers. Funct. Ecol. 2004, 18, 829–835. [Google Scholar] [CrossRef]
- Huang, Z.-Y.; Robinson, G.E. Regulation of honey bee division of labor by colony age demography. Behav. Ecol. Sociobiol. 1996, 39, 147–158. [Google Scholar] [CrossRef]
- Sedgley, M. Anatomical investigation of abscised avocado flowers and fruitlets. Ann. Bot. 1980, 46, 771–777. Available online: https://hdl.handle.net/102.100.100/294142?index=1 (accessed on 16 September 2024). [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 4 August 2025).
- McGregor, S.E. Insect Pollination of Cultivated Crop Plants; No. 496; Agricultural Research Service, US Department of Agriculture: Washington, DC, USA, 1976.
- Fahad, S.; Bajwa, A.A.; Nazir, U.; Anjum, S.A.; Farooq, A.; Zohaib, A.; Sadia, S.; Nasim, W.; Adkins, S.; Saud, S.; et al. Crop production under drought and heat stress: Plant responses and management options. Front. Plant Sci. 2017, 8, 1147. [Google Scholar] [CrossRef] [PubMed]
- Formisano, L.; El-Nakhel, C.; Corrado, G.; De Pascale, S.; Rouphael, Y. Biochemical, physiological, and productive response of greenhouse vegetables to suboptimal growth environment Induced by insect nets. Biology 2020, 9, 432. [Google Scholar] [CrossRef]
- Vicens, N.A.; Bosch, J. Weather-dependent pollinator activity in an apple orchard, with special reference to Osmia cornuta and Apis mellifera (Hymenoptera: Megachilidae and Apidae). Environ. Entomol. 2000, 29, 413–420. [Google Scholar] [CrossRef]
- Burrill, R.M.; Dietz, A. The response of honeybees to variations in solar radiation and temperature. Apidologie 1981, 12, 319–328. [Google Scholar] [CrossRef]
- Kevan, P.G.; Baker, H.G. Insects as flower visitors and pollinators. Annu. Rev. Entomol. 1983, 28, 40–53. [Google Scholar] [CrossRef]
- Winston, M.L. The Biology of the Honey Bee; Harvard University Press: Cambridge, MA, USA, 1987. [Google Scholar]
- Wallman, J.F.; Leys, R.; Hogendoorn, K. Molecular systematics of Australian carrion-breeding blowflies (Diptera: Calliphoridae) based on mitochondrial DNA. Invertebr. Syst. 2005, 19, 1–15. [Google Scholar] [CrossRef]
- Garner, L.C.; Lovatt, C.J. The relationship between flower and fruit abscission and alternate bearing of ‘Hass’ avocado. J. Am. Soc. Hortic. Sci. 2008, 133, 3–10. [Google Scholar] [CrossRef]
- Alcaraz, M.L.; Hormaza, J.I. Fruit set in avocado: Pollen limitation, pollen load size, and selective fruit abortion. Agronomy 2021, 11, 1603. [Google Scholar] [CrossRef]
- Degani, C.; Goldring, A.; Gazit, S. Pollen parent effect on outcrossing rate in ‘Hass’ and ‘Fuerte’ avocado plots during fruit development. J. Am. Soc. Hortic. Sci. 1989, 114, 106–111. [Google Scholar] [CrossRef]
- Hapuarachchi, N.S.; Kämper, W.; Bai, S.H.; Ogbourne, S.M.; Nichols, J.; Wallace, H.M.; Trueman, S.J. Selective retention of cross-fertilised fruitlets during premature fruit drop of Hass avocado. Horticulturae 2024, 10, 591. [Google Scholar] [CrossRef]
- Wallman, J.F. Third-instar larvae of common carrion-breeding blowflies of the genus Calliphora (Diptera: Calliphoridae) in South Australia. Invertebr. Syst. 2001, 15, 37–51. [Google Scholar] [CrossRef]
- Donovan, S.E.; Hall, M.J.R.; Turner, B.D.; Moncrieff, C.B. Larval growth rates of the blowfly, Calliphora vicina, over a range of temperatures. Med. Vet. Entomol. 2006, 20, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G. Minimum and maximum development rates of some forensically important Calliphoridae (Diptera). J. Forensic Sci. 2000, 45, 824–832. [Google Scholar] [CrossRef]
- Brown, K.; Thorne, A.; Harvey, M. Calliphora vicina (Diptera: Calliphoridae) pupae: A timeline of external morphological development and a new age and PMI estimation tool. Int. J. Legal Med. 2015, 129, 835–850. [Google Scholar] [CrossRef] [PubMed]
- Pérez, C.; Segura, N.A.; Patarroyo, M.A.; Bello, F.J. Evaluating the biological cycle and reproductive and population parameters of Calliphora vicina (Diptera: Calliphoridae) reared on three different diets. J. Med. Entomol. 2016, 53, 1268–1275. [Google Scholar] [CrossRef] [PubMed]
- Norris, K. The bionomics of blow flies. Annu. Rev. Entomol. 1965, 10, 47–68. [Google Scholar] [CrossRef]
- Brodie, B.S.; Smith, M.A.; Lawrence, J.; Gries, G. Effects of floral scent, color and pollen on foraging decisions and oocyte development of common green bottle flies. PLoS ONE 2015, 10, e0145055. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, G.J. Seed production of F1 hybrid brussel sprouts. Acta Hortic. 1978, 83, 37–42. [Google Scholar] [CrossRef]
- Faulkner, G.J.; Hinton, W.L. F1 hybrid brussels sprout seed: An assessment of production methods and their economic viability. Hortic. Res. 1980, 20, 49–59. [Google Scholar]
- Davis, A.E.; Schmidt, L.A.; Santos, K.C.B.S.; Rowbottom, R.; Lucas, A.; Harrington, S.; Spurr, C.; Rader, R. Environmental drivers, spatiotemporal dynamics, and pollination effectiveness of insect floral visitors in Australian seed carrot agroecosystems. Agric. Ecosyst. Environ. 2025, 383, 109553. [Google Scholar] [CrossRef]
- Faucherre, J.; Cherix, D.; Wyss, C. Behavior of Calliphora vicina (Diptera, Calliphoridae) under extreme conditions. J. Insect Behav. 1999, 12, 687–690. [Google Scholar] [CrossRef]
- Potts, S.G.; Imperatriz-Fonseca, V.; Ngo, H.T.; Aizen, M.A.; Biesmeijer, J.C.; Breeze, T.D.; Dicks, L.V.; Garibaldi, L.A.; Hill, R.; Settele, J.; et al. Safeguarding pollinators and their values to human well-being. Nature 2016, 540, 220–229. [Google Scholar] [CrossRef]
- Statista. Avocado Production Worldwide from 2000 to 2023. Statista. 2025. Available online: https://www.statista.com/statistics/577455/world-avocadoproduction (accessed on 10 July 2025).
- Magrach, A.; Sanz, A.J. Environmental and social consequences of the increase in the demand for ‘superfoods’ world-wide. People Nat. 2020, 2, 267–278. [Google Scholar] [CrossRef]
- Muñoz, A.E.; Plantegenest, M.; Amouroux, P.; Zaviezo, T. Native flower strips increase visitation by non-bee insects to avocado flowers and promote yield. Basic. Appl. Ecol. 2021, 56, 369–378. [Google Scholar] [CrossRef]
- Gilpin, A.-M.; Kobel, C.; Brettell, L.E.; O’Brien, C.; Cook, J.M.; Power, S.A. Co-flowering species richness increases pollinator visitation to apple flowers. Agriculture 2022, 12, 1246. [Google Scholar] [CrossRef]
- Barahona-Segovia, R.M.; Gatica-Barrios, P.; Durán-Sanzana, V.; Smith-Ramírez, C. No wild bees? Don’t worry! Non-bee pollinators are still hard at work: The edge effect, landscape, and local characteristics determine taxonomic and functional diversity in apple orchards. Agric. Ecosyst. Environ. 2023, 354, 108554. [Google Scholar] [CrossRef]
- Requier, F.; Garnery, L.; Kohl, P.L.; Njovu, H.K.; Pirk, C.W.W.; Crewe, R.M.; Steffan-Dewenter, I. The conservation of native honey bees is crucial. Trend Ecol. Evol. 2019, 34, 789–798. [Google Scholar] [CrossRef]
- Klein, A.-M.; Brittain, C.; Hendrix, S.D.; Thorp, R.; Williams, N.; Kremen, C. Wild pollination services to California almond rely on semi-natural habitat. J. Appl. Ecol. 2012, 49, 723–732. [Google Scholar] [CrossRef]
- Carvalheiro, L.G.; Seymour, C.L.; Nicolson, S.W.; Veldtman, R. Creating patches of native flowers facilitates crop pollination in large agricultural fields: Mango as a case study. J. Appl. Ecol. 2012, 49, 1373–1383. [Google Scholar] [CrossRef]
- Dymond, B.; Celiz-Diez, J.L.; Diaz-Siefer, P.; Rojas-Bravo, V.; Martinez-Harms, J.; Potts, S.G.; Garratt, M.P.D. Proximity to natural habitat enhances flower visitor diversity and pollination services in avocado orchards. Front Sustain. Food Syst. 2025, 9, 1560802. [Google Scholar] [CrossRef]
- Saunders, M.E.; Luck, G.W.; Mayfield, M.M. Almond orchards with living ground cover host more wild insect pollinators. J. Insect Conserv. 2013, 17, 1011–1025. [Google Scholar] [CrossRef]


| Site | Year | Insect Pollinator Species Within Enclosures | |||||
|---|---|---|---|---|---|---|---|
| Apis mellifera | Calliphora vicina | Calliphora dubia | Eristalis tenax | ||||
| Busselton | 2021 | 5k | 5k (3046) | - | 5k (2754) | - | |
| Pemberton | 2021 | 5k | 5k (3244) | - | 5k (2815) | - | - |
| Capel | 2022 | - | 5k (3899) | 10k (7899) | - | - | - |
| Capel | 2023 | - | 10k (9783) | 10k (11,050) | 5k (3532) | ||
| Year | Site | Month | Max (°C) | Min (°C) | Rainfall (mm) | Rain Days | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| FP | LTA | FP | LTA | FP | LTA | FP | LTA | |||
| 2021 | Busselton (a) * | Oct | 19.8 | 21.4 1 | 6.4 | 8.7 1 | 109 | 32 1 | 16 | 6 1 |
| Nov | 22.6 | 25.1 1 | 8.2 | 10.7 1 | 2 | 22 1 | 2 | 4 1 | ||
| 2021 | Pemberton (b) | Oct | 19.0 | 18.8 2 | 8.6 | 8.6 2 | 98 | 93 2 | 11 | 12 2 |
| Nov | 19.2 | 21.4 2 | 8.5 | 10.3 2 | 17 | 48 2 | 9 | 8 2 | ||
| 2022 | Capel (c) | Oct | 18.8 3 | 21.4 4 | 7.0 3 | 8.7 4 | 35 | 42 4 | 11 | 9 4 |
| Nov | 21.6 3 | 25.1 4 | 8.8 3 | 10.7 4 | 16 | 18 4 | 6 | 4 4 | ||
| 2023 | Capel (c) | Oct | 23.3 | 21.4 4 | 8.9 | 8.7 4 | 9 | 42 4 | 3 | 9 4 |
| Nov | 24.8 | 25.1 4 | 10.2 | 10.7 4 | 1 | 18 4 | 1 | 4 4 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cook, D.F.; Tufail, M.S.; Howse, E.T.; Voss, S.C.; Foley, J.; Norrish, B.; Delroy, N. Pollination of Enclosed Avocado Trees by Blow Flies (Diptera: Calliphoridae) and a Hover Fly (Diptera: Syrphidae). Insects 2025, 16, 899. https://doi.org/10.3390/insects16090899
Cook DF, Tufail MS, Howse ET, Voss SC, Foley J, Norrish B, Delroy N. Pollination of Enclosed Avocado Trees by Blow Flies (Diptera: Calliphoridae) and a Hover Fly (Diptera: Syrphidae). Insects. 2025; 16(9):899. https://doi.org/10.3390/insects16090899
Chicago/Turabian StyleCook, David F., Muhammad S. Tufail, Elliot T. Howse, Sasha C. Voss, Jacinta Foley, Ben Norrish, and Neil Delroy. 2025. "Pollination of Enclosed Avocado Trees by Blow Flies (Diptera: Calliphoridae) and a Hover Fly (Diptera: Syrphidae)" Insects 16, no. 9: 899. https://doi.org/10.3390/insects16090899
APA StyleCook, D. F., Tufail, M. S., Howse, E. T., Voss, S. C., Foley, J., Norrish, B., & Delroy, N. (2025). Pollination of Enclosed Avocado Trees by Blow Flies (Diptera: Calliphoridae) and a Hover Fly (Diptera: Syrphidae). Insects, 16(9), 899. https://doi.org/10.3390/insects16090899








