3.3. Neosuidasia sjorsvandenbergi Fan & Faraji, sp. nov.
(
Figure 1,
Figure 2,
Figure 3,
Figure 4,
Figure 5,
Figure 6,
Figure 7,
Figure 8,
Figure 9,
Figure 10,
Figure 11,
Figure 12,
Figure 13,
Figure 14,
Figure 15,
Figure 16,
Figure 17,
Figure 18,
Figure 19,
Figure 20,
Figure 21,
Figure 22,
Figure 23,
Figure 24,
Figure 25,
Figure 26 and
Figure 27,
Table 1)
urn:lsid:zoobank.org:pub:8DEEE2FB-FDAE-446E-B43E-8E23C3019F01
Material examined
Holotype female, paratypes 19 females, 12 males, 10 tritonymphs, 2 protonymphs, ex rearing culture, originally collected from a home poultry (open cage) in February 2022 in Monster, The Netherlands (52°02′18.6″ N, 4°12′27.4″ E), 20 January 2025, Sjors Van den Berg coll.; 26 females, 8 males, 5 tritonymphs, 2 protonymphs, 2 larvae, same collection data as the holotype but collected on 19 January 2024.
The holotype, 10 paratype females, 10 paratype males, 10 tritonymphs, 4 protonymphs and 2 larvae will be deposited in the New Zealand Arthropod Collection (NZAC), Landcare Research, Auckland. An additional 35 paratype females, 10 paratype males, and 5 tritonymphs are housed at the Plant Health and Environment Laboratory, Auckland, New Zealand (PANZ).
Diagnosis
This new species can be readily distinguished from the only previously known species, N. faini Ranganath & ChannaBasavanna, by the following characteristics: in adult females, setae ps2 are 2.5 (1.7–2.5)× length of ad3 (compared to slightly longer in N. faini) and about as long as anal opening (versus half its length in N. faini). In males, ps2 is more than four times the length of ps3, while in N. faini, ps2 is less than twice the length of ps1.
Description
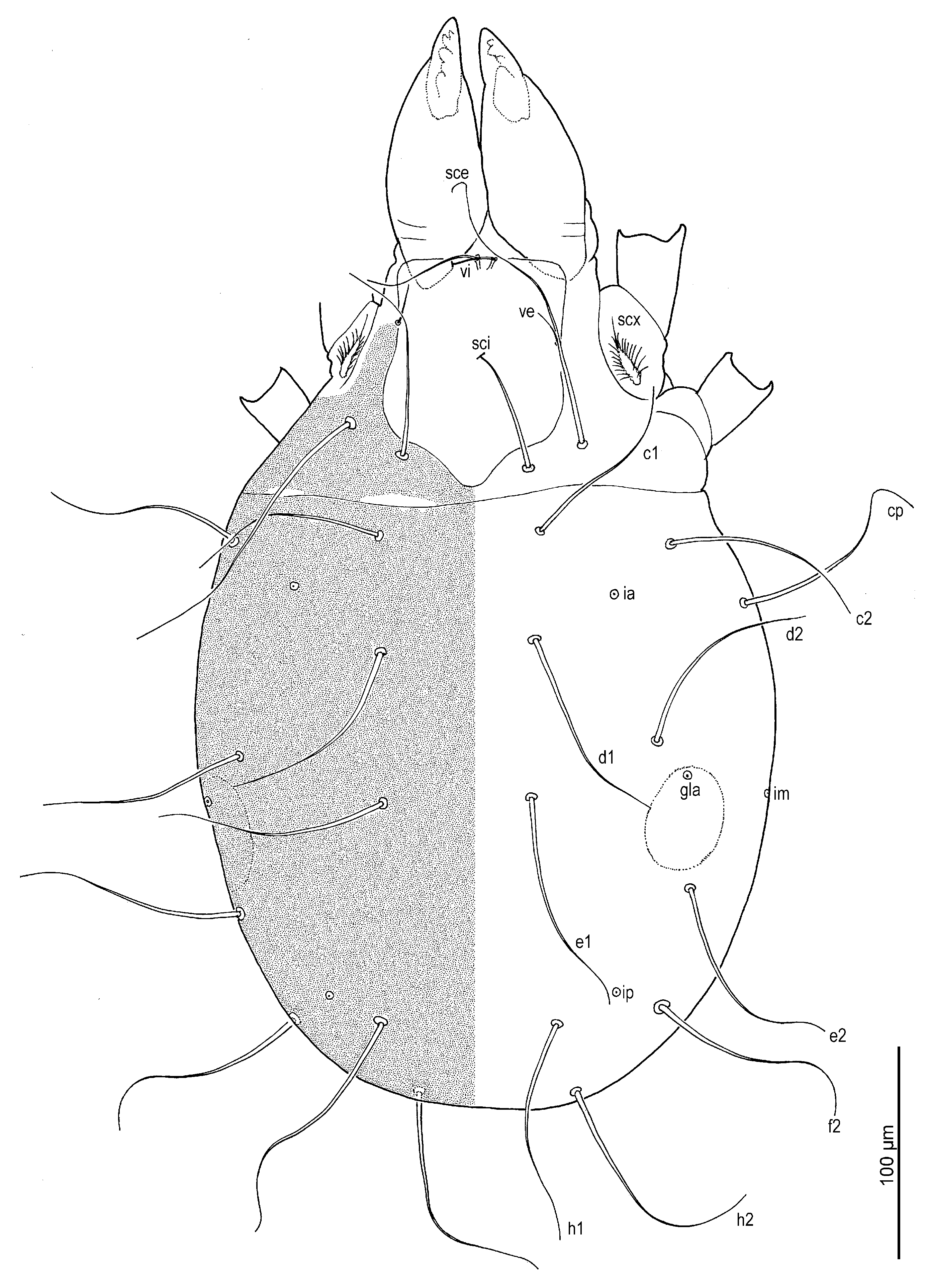
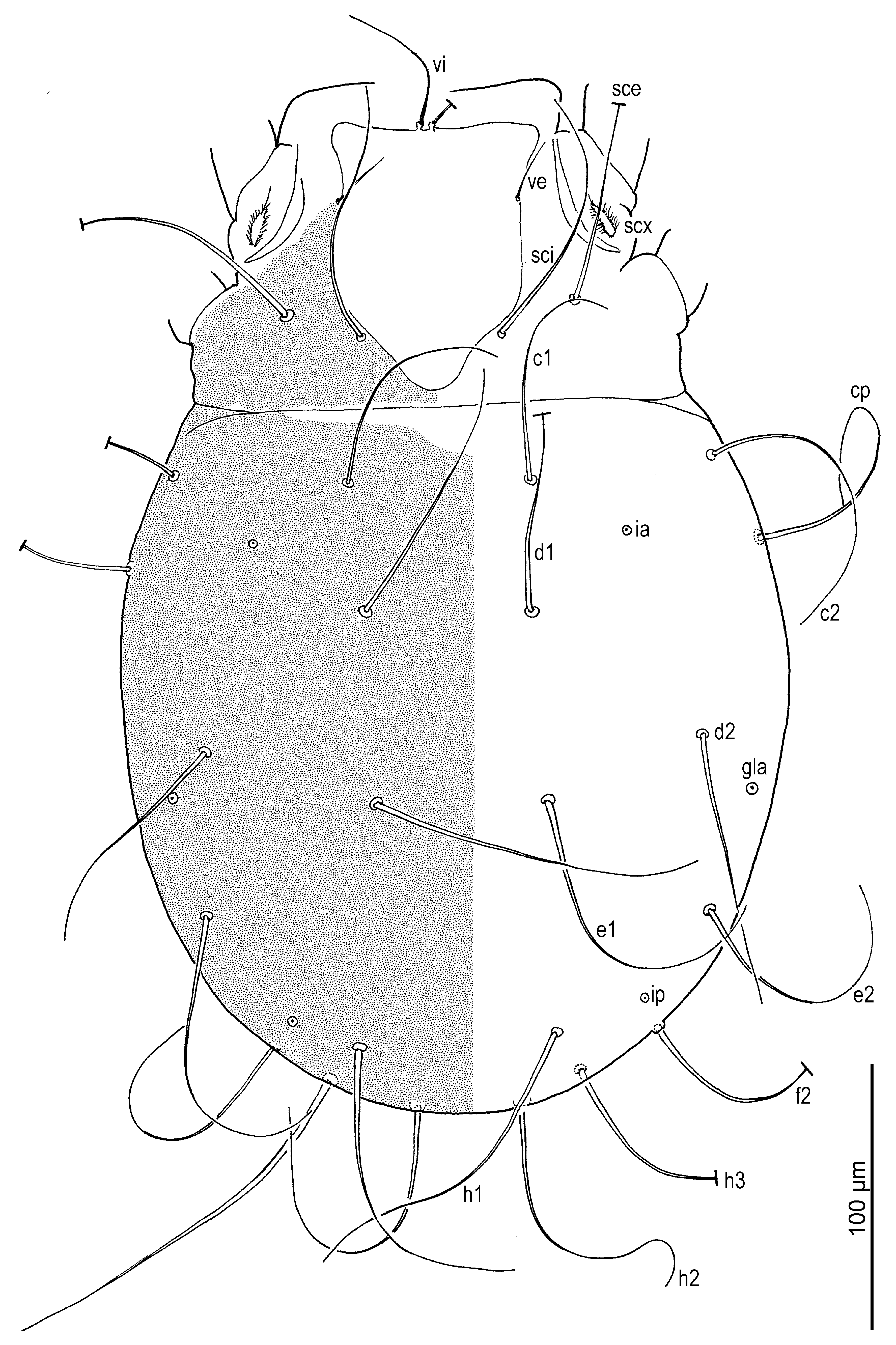
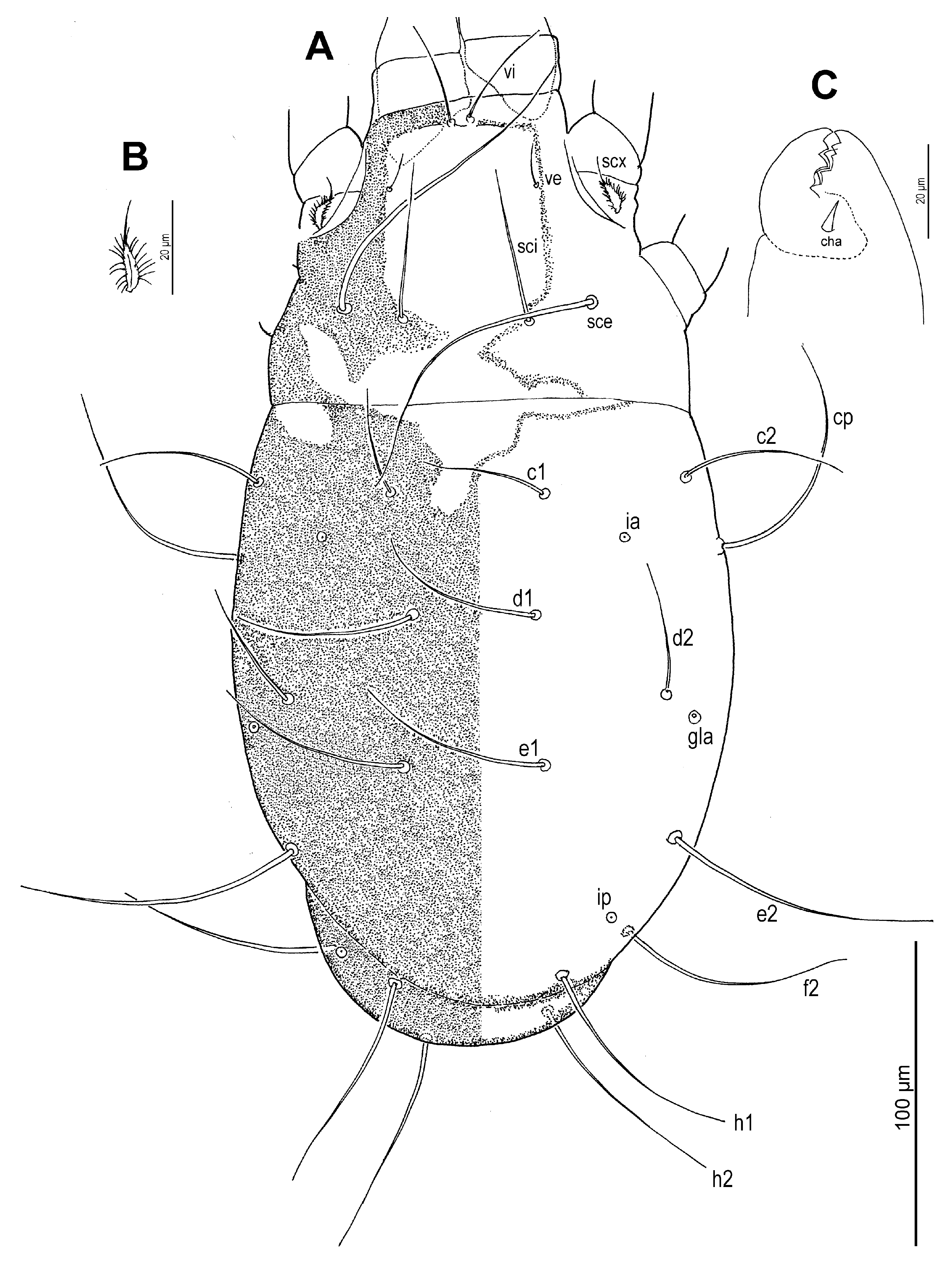
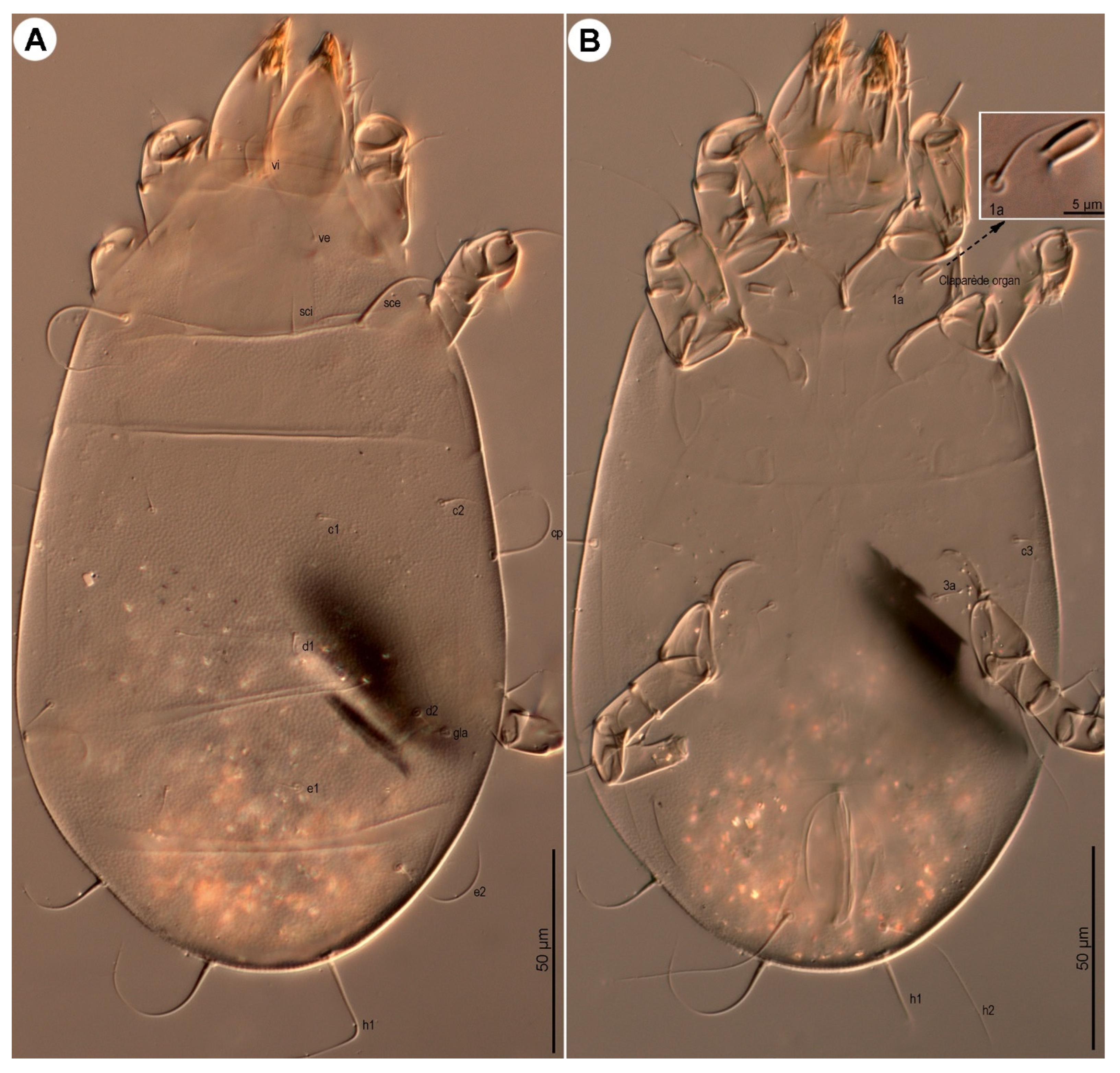
Dorsum (
Figure 1 and
Figure 3A) broadly pyriform; cuticle finely mammillated over almost entire dorsal surface, except for two narrow transverse areas anterior to sejugal furrow (
Figure 1). Prodorsal shield anterior to scapular setae pentagonal; posteromedial margin convex and projecting beyond the level of setae
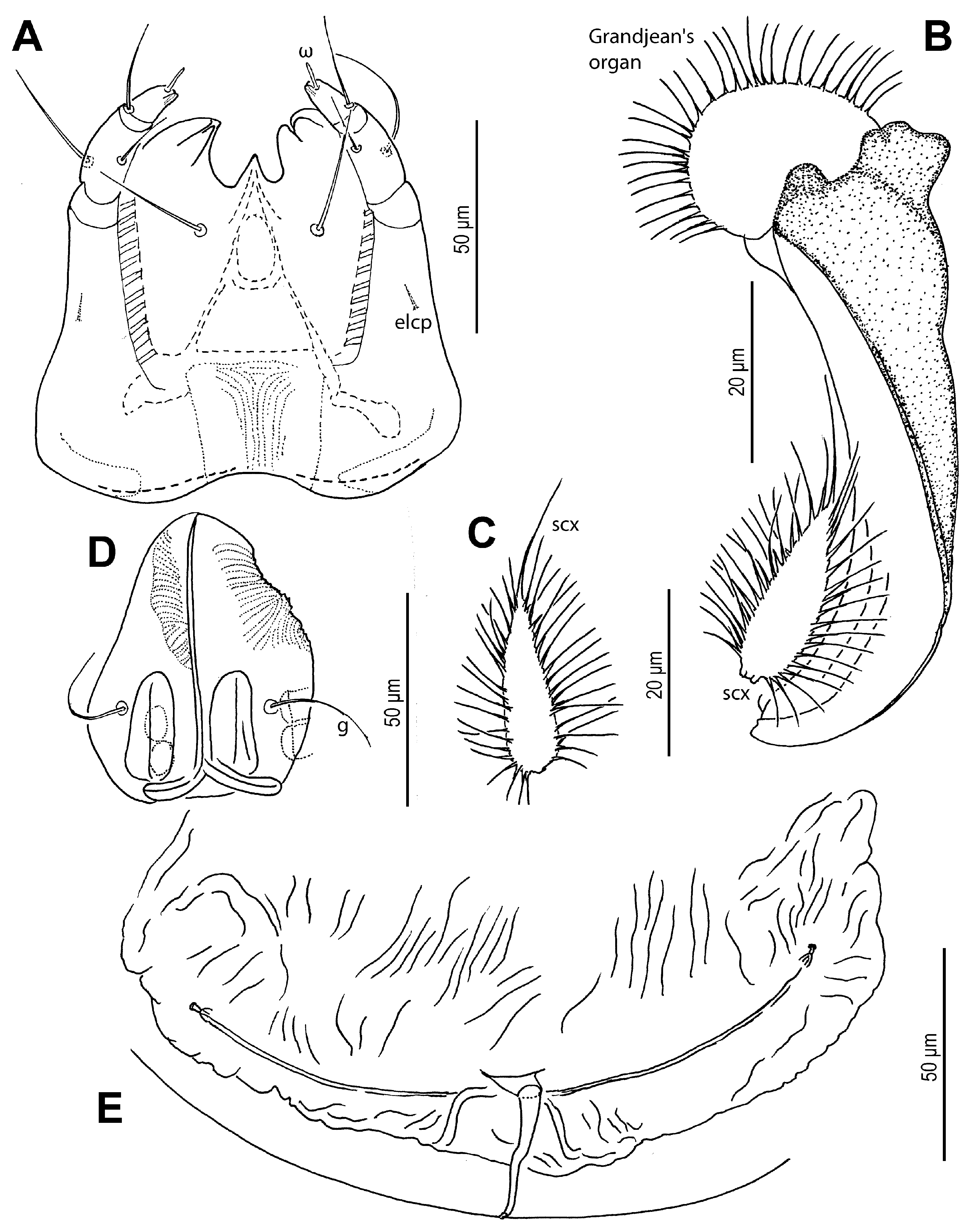
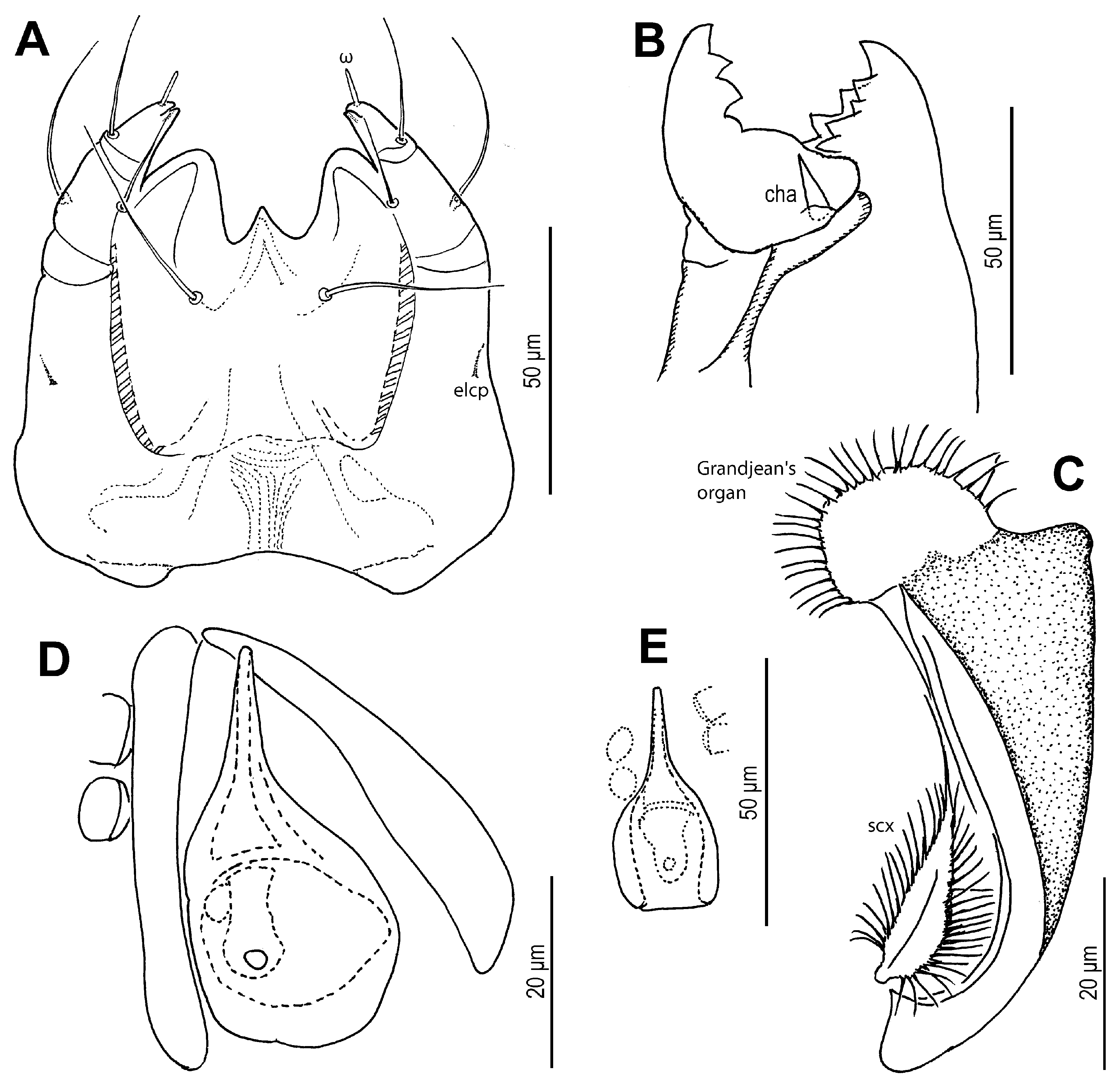
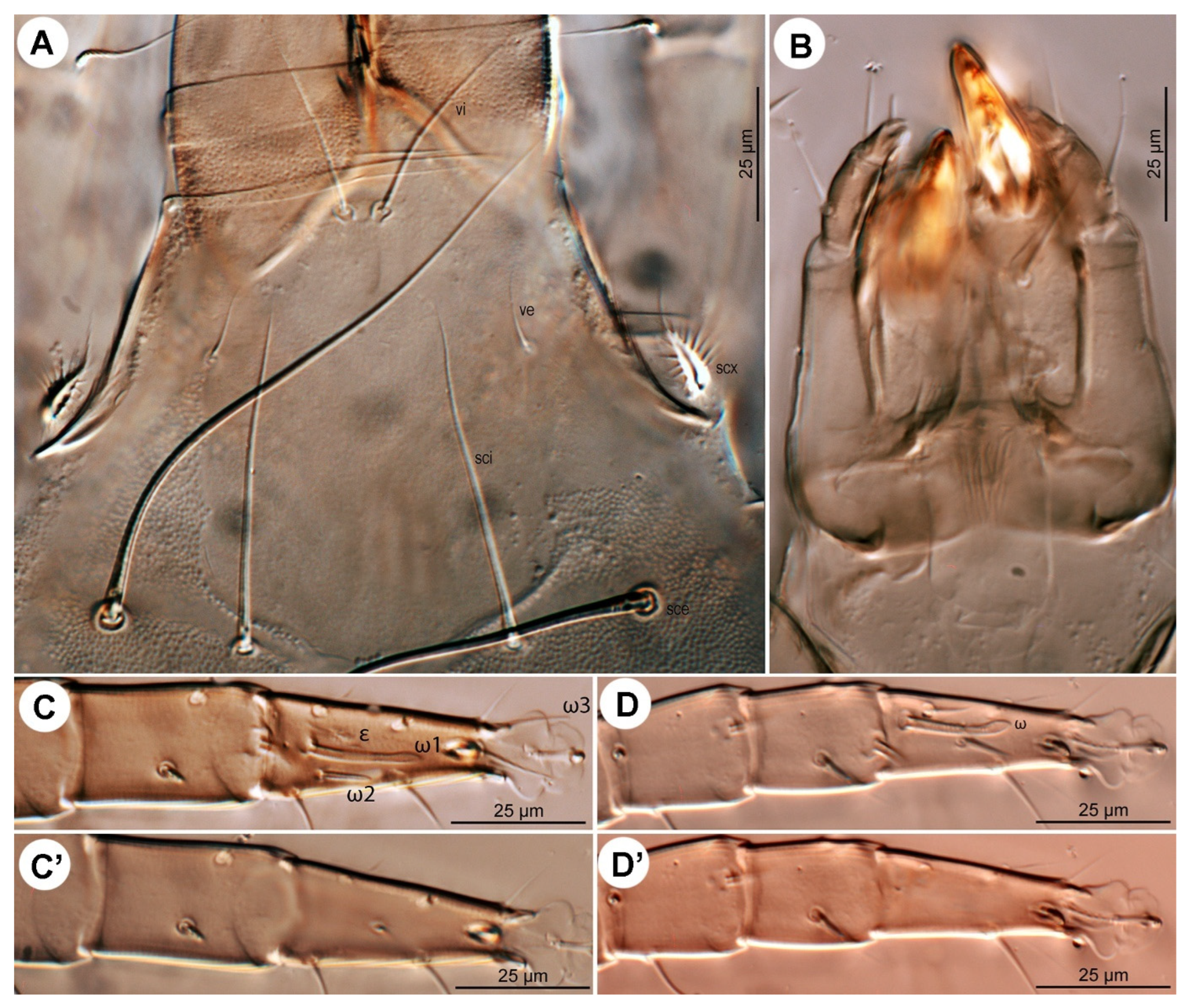
sci; surface finely punctate. Supracoxal sclerite (
Figure 4C and
Figure 5B) elongate. Grandjean’s organ (
Figure 4C and
Figure 5B) semicircular in shape, with long marginal pectinations. Supracoxal setae
scx (
Figure 4C and
Figure 5B,C) lanceolate, with 24–28 long marginal pectinations. Idiosomal setae filiform,
vi slightly shorter than prodorsal shield width,
ve much shorter than
vi; located laterally, at anterior 1/4–1/3 of prodorsal shield;
sci more than 4/5 length of
sce; distance
sci–
sci 2.1 (1.8–2.1)×
sci–
sce. Opisthonotal gland openings
gla positioned closer to
d2 than
e2. Setae
c3 shortest, on ventral surface; remaining setae similar in length, extending beyond bases of next row setae. Cupules
ia situated posterolateral to setae
c1 and posteromedial to
c2;
im located lateral to gland opening
gla;
ip positioned anterolateral to
h1 and anteromedial to
f2.
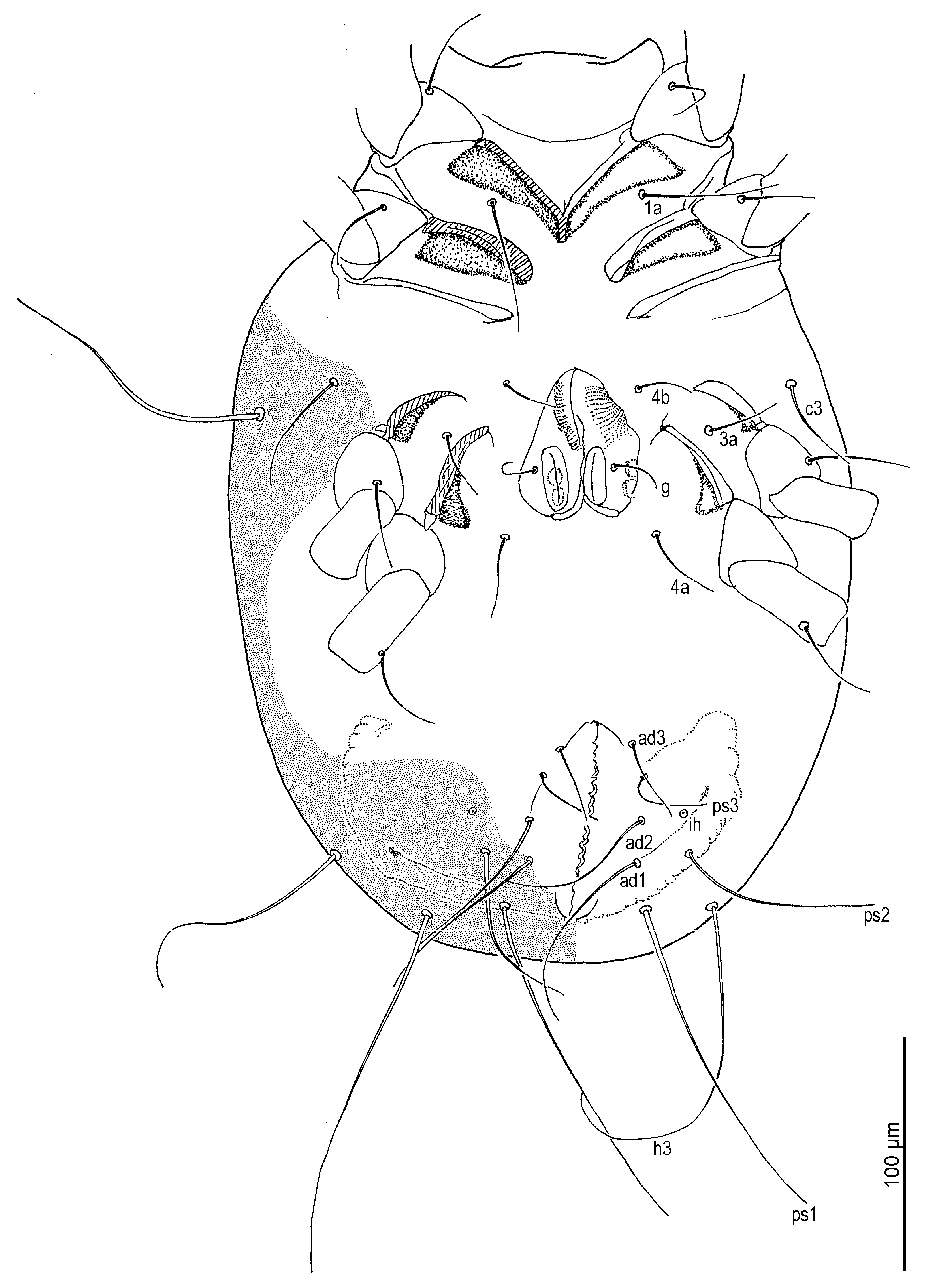
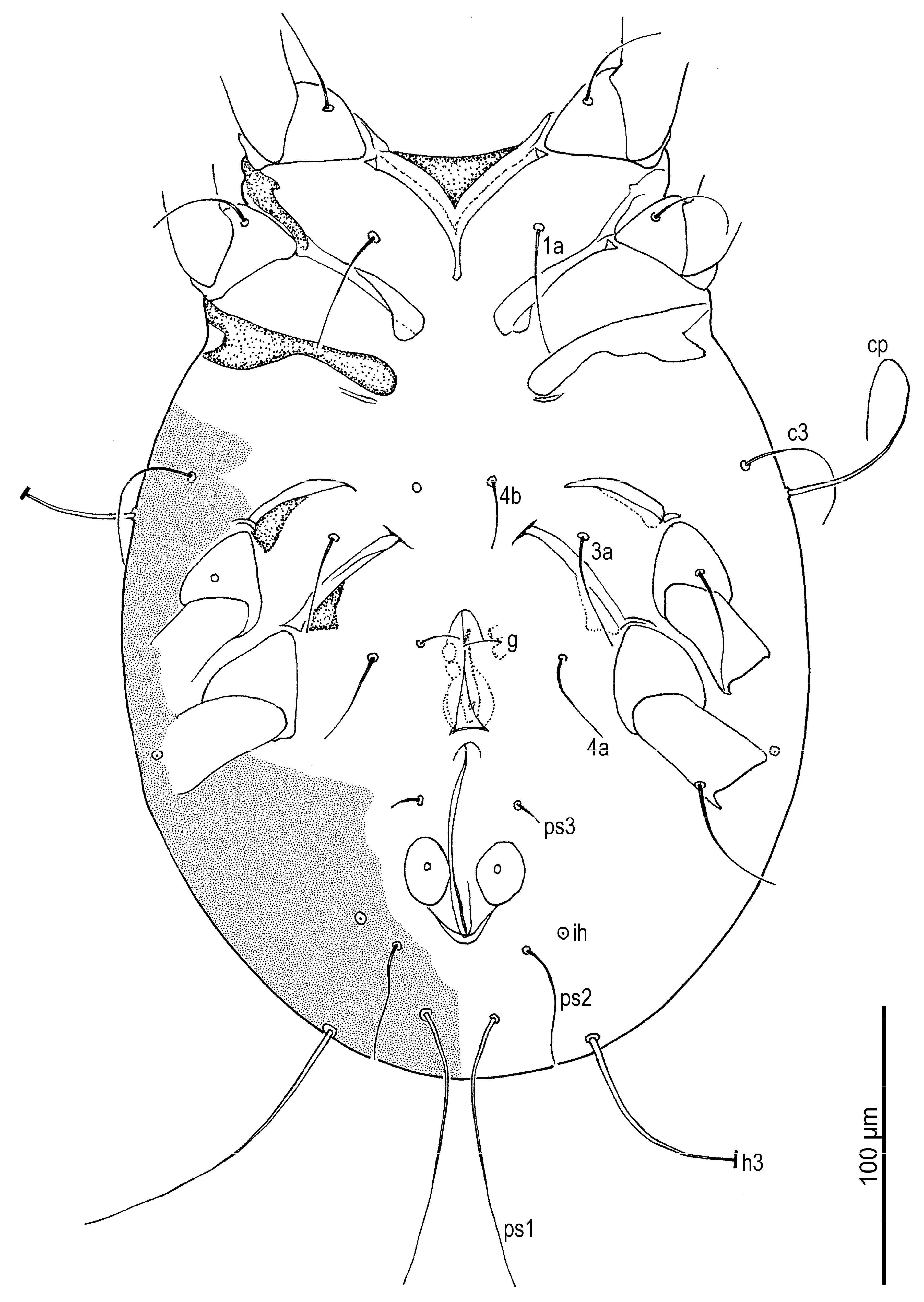
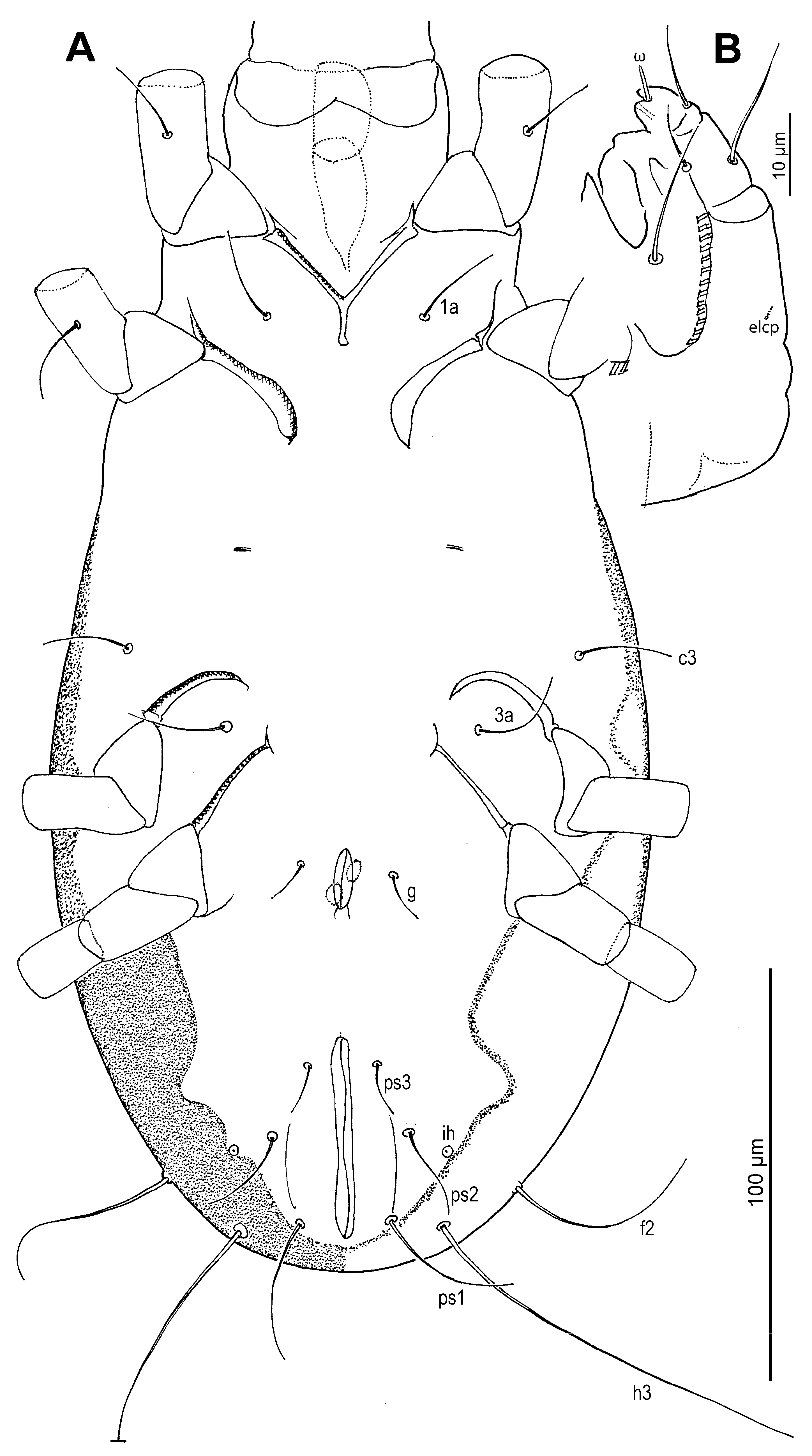
Venter (
Figure 2,
Figure 3B,
Figure 4D,F and
Figure 5D,E): Cuticle finely mammillated in anterolateral region, lateral surfaces of coxae III–IV, and extending along lateral margin toward region near to anus. Coxal apodemes I fused medially, forming short prosternal apodeme directed posteromedially. Coxal plates I reach apex of prosternal apodeme; posterior margins slightly concave. Setae
1a lateral to prosternal apodeme. Coxal apodemes II directed posteromedially; coxal plates II approximately twice as wide as apodemes II, extending to their apex; posterior margins nearly straight. Posterior apodemes of coxae II distinct, appearing as ridges, narrower than apodemes II. Genital opening (
Figure 2,
Figure 3B,
Figure 4D and
Figure 5D) inverted V-shaped, situated between coxae III and IV. Apodemes III and IV directed anteromedially. Setae
4b lateral to anterior margin of genital opening;
g lateral to first pair of genital papillae;
4a posterolateral to lateral genital arms. Genital and anal openings separated by more than 1.3 (1.3–2.2)× genital opening length. Anal opening distinctly longer than genital opening; surrounded by 3 pairs of adanal setae (
ad1–3) and 3 pairs of pseudanal setae (
ps1–3). Setae
ad3 and
ps3 obviously shorter;
ps1 longest, 1.2 (1.1–1.3)×
ps2, 2.8 (2.1–2.8)×
ps3;
ps2 2.5 (1.7–2.5)× length of
ad3 and nearly equal to anal opening length. Cupule
ih situated lateral to setae
ad2. Copulatory opening (
Figure 4F and
Figure 5E) near posterior margin of idiosoma. Inseminatory canal (spermathecal duct) (37 (36–48)) slender, cylindrical, slightly expanded at base of receptaculum seminis (spermathecal sac). Sclerites of efferent ducts (sclerites of oviducts) (4 (3–4)) bell-shaped, widely separated (112 (112–176)), approximately as wide as distance between trochanter IV bases.
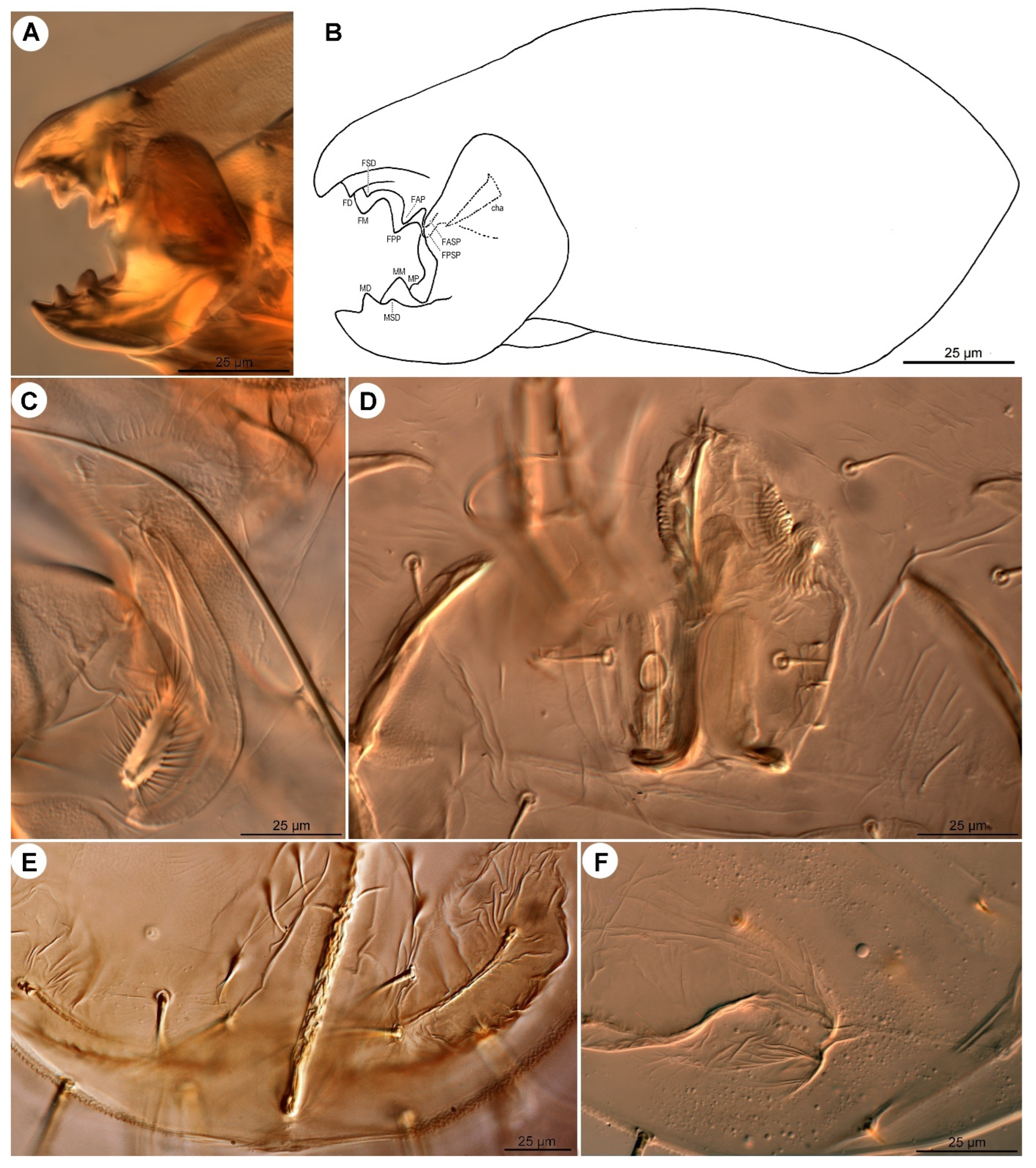
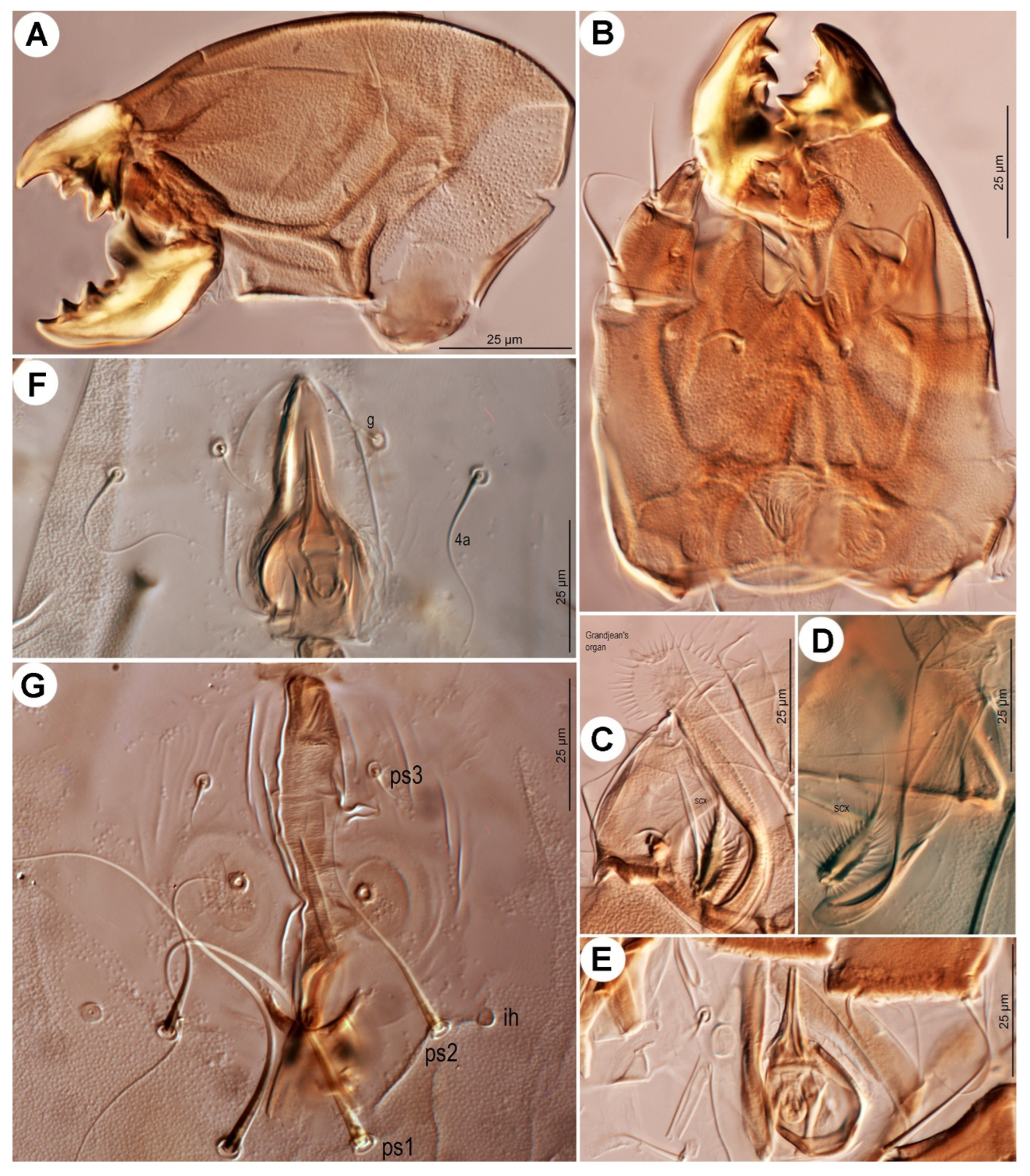
Gnathosoma (
Figure 4A,B and
Figure 5A). Chelicerae robust, chelate-dentate (
Figure 4A,B). Movable digit with 4 teeth; distal tooth (MD) nearly 3/4 of medial tooth (MM), subdistal (MSD) and proximal (MP) teeth indistinct, antiaxial and paraxial, respectively. Fixed digit with 7 teeth, 4 antiaxial and 3 paraxial, subdistal (FSD) obviously smaller than others; two most proximal teeth (FASP and FPSP) hard to observe. Cheliceral setae
cha strong, conical. Subcapitulum (
Figure 5A) with setae
h filiform. Palpal supracoxal setae
elcp minute and slender; palptibial setae (
sup,
a) and palptarsal seta (
cm) filiform; terminal palptarsal solenidion (
ω) distinct and finge-shaped; eupathidium
ul” distally rounded.
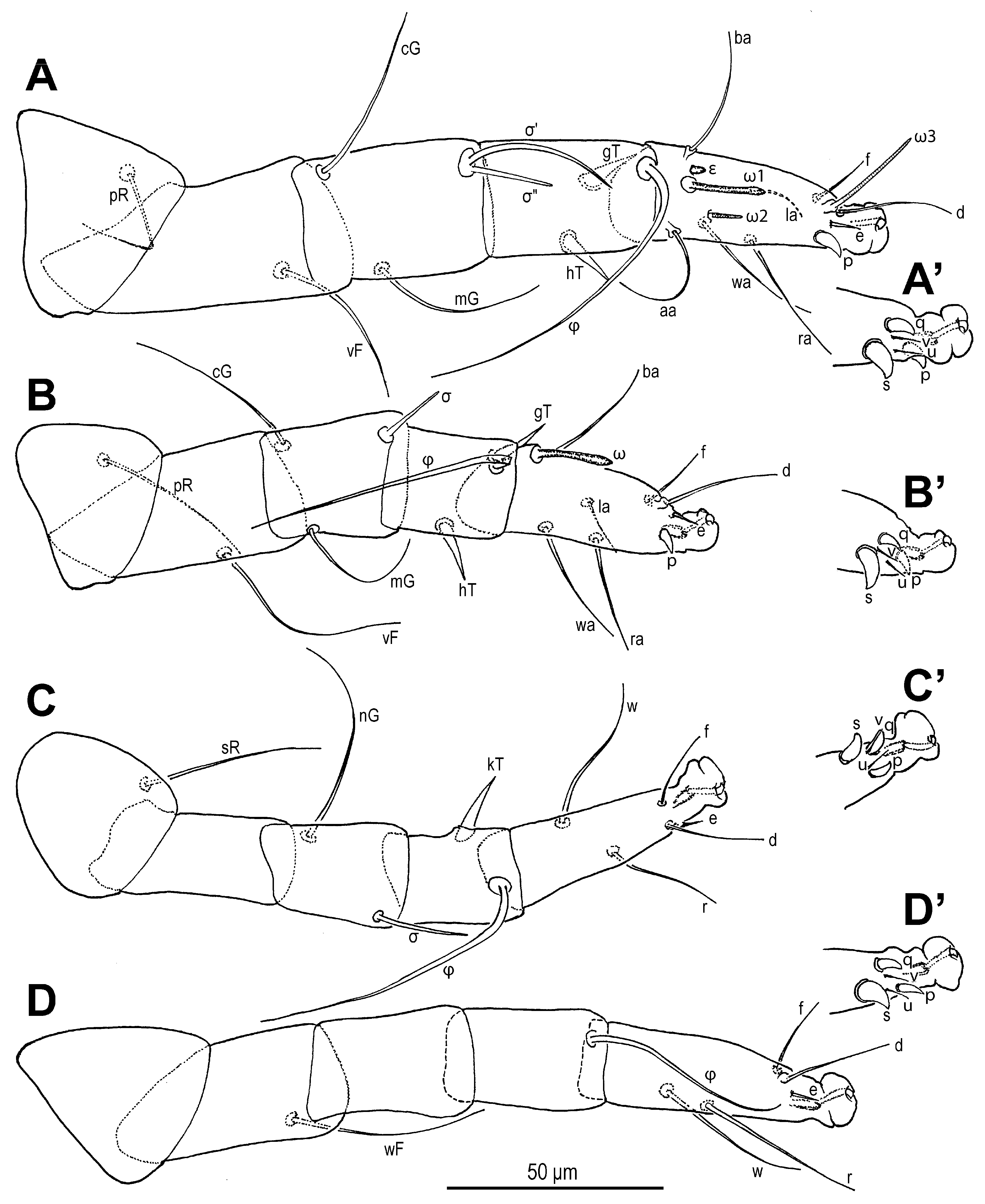
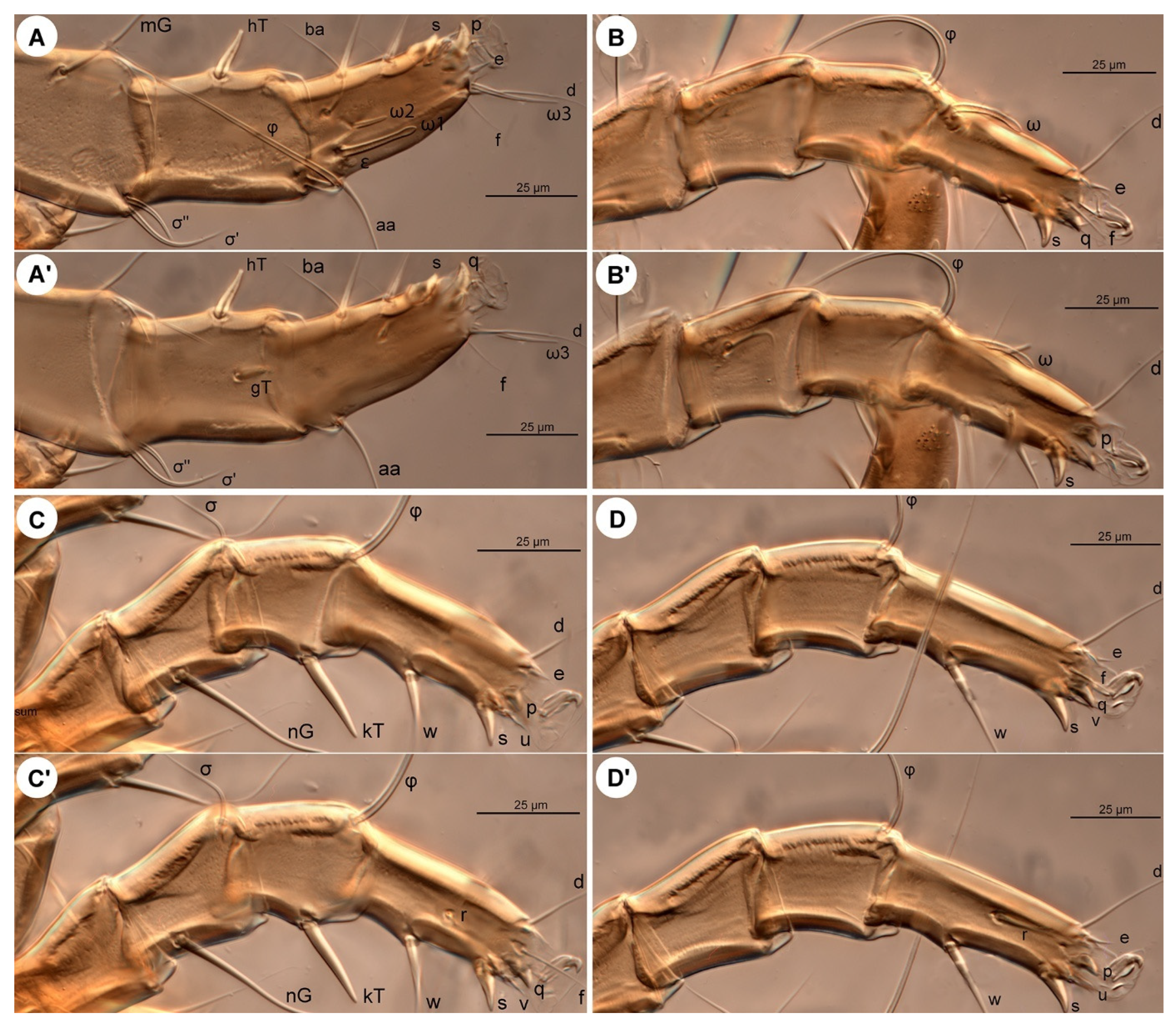
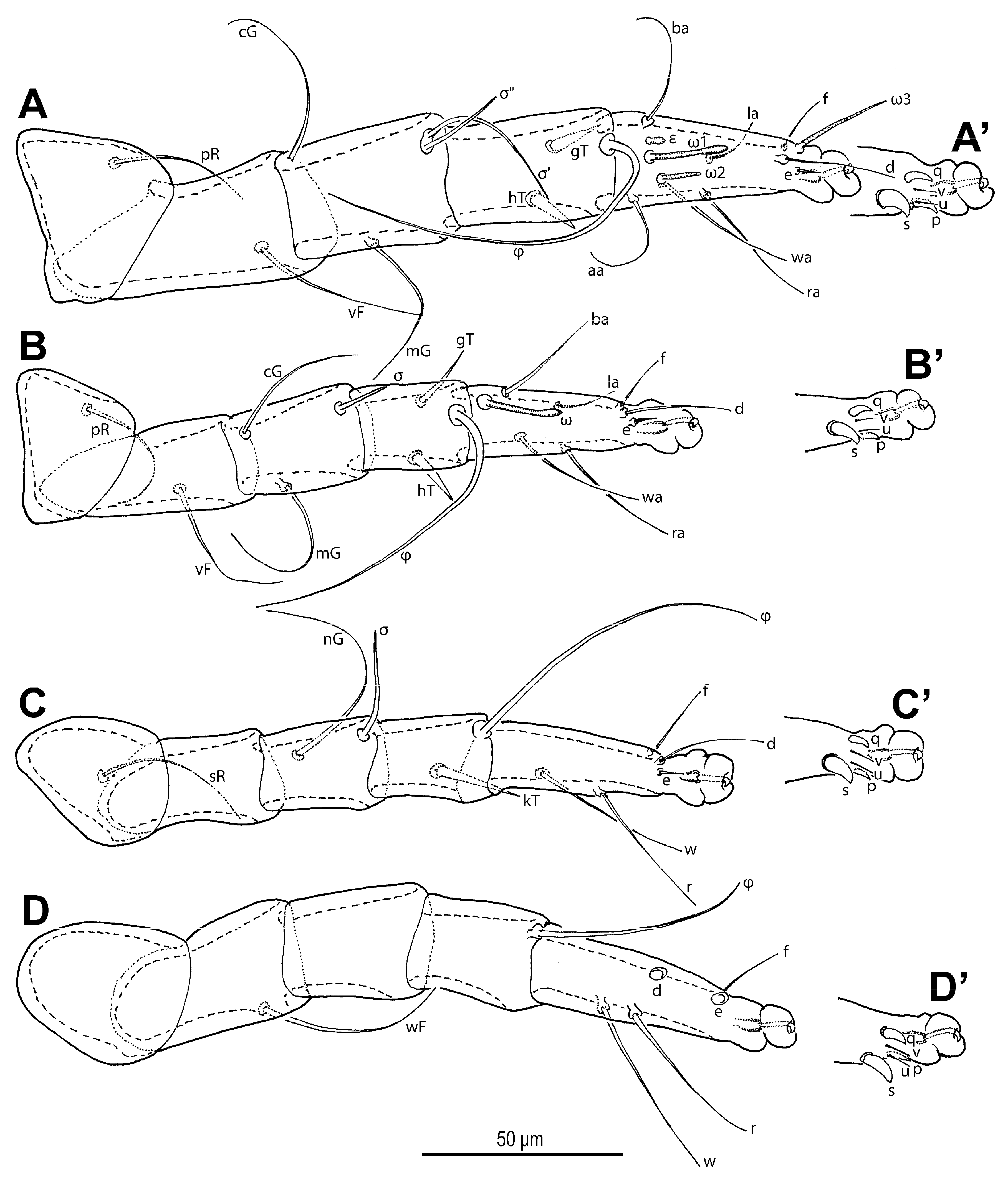
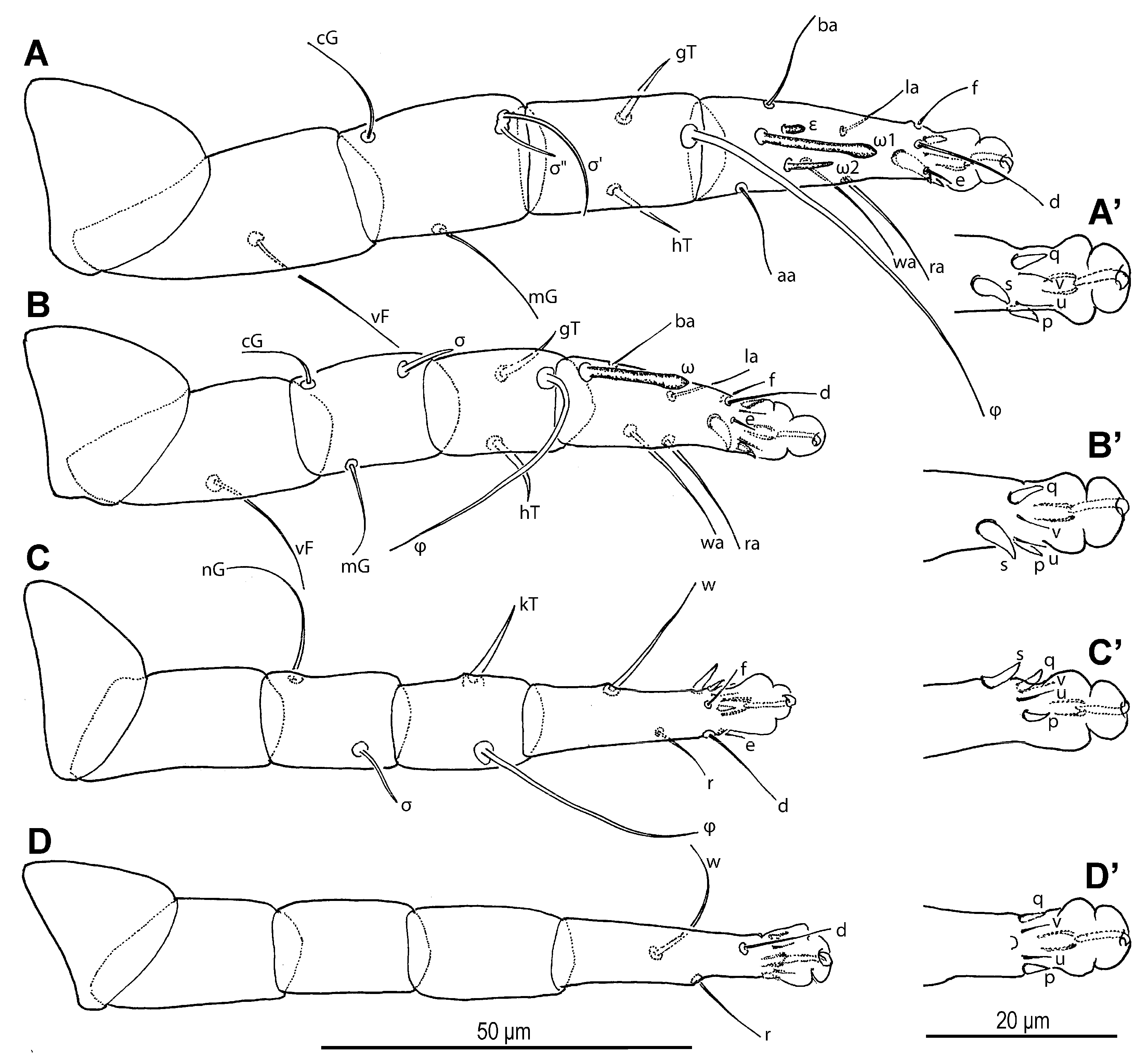
Legs (
Figure 6 and
Figure 7). Legs I and IV distinctly longer than II and III. Setae (if present) on trochanters, femora, genua smooth, filiform; tibial setae (if present) conical, spiniform.
Figure 1.
Neosuidasia sjorsvandenbergi sp. nov. (adult female). Idiosoma and chelicerae, dorsal view.
Figure 1.
Neosuidasia sjorsvandenbergi sp. nov. (adult female). Idiosoma and chelicerae, dorsal view.
Figure 2.
Neosuidasia sjorsvandenbergi sp. nov. (adult female). Idiosoma, ventral view.
Figure 2.
Neosuidasia sjorsvandenbergi sp. nov. (adult female). Idiosoma, ventral view.
Figure 3.
Neosuidasia sjorsvandenbergi sp. nov. (adult female, DIC images). (A). Dorsal view; (B). Ventral view (arrow: sclerite of the efferent duct).
Figure 3.
Neosuidasia sjorsvandenbergi sp. nov. (adult female, DIC images). (A). Dorsal view; (B). Ventral view (arrow: sclerite of the efferent duct).
Figure 4.
Neosuidasia sjorsvandenbergi sp. nov. (adult female). (A). Chelicera (DIC image), antiaxial view; (B). Chelicera (line drawing), antiaxial view; (C). Supracoxal sclerite, scx and Grandjean’s organ (DIC image); (D). Genital area (DIC image); (E). Receptaculum seminis and anal area (DIC image); (F). Inseminatory canal and receptaculum seminis (DIC image). Abbreviations: cha, cheliceral seta; FAP, fixed digit, antiaxial proximal tooth; FASP, fixed digit, antiaxial subproximal tooth; FD, fixed digit, distal tooth; FM, fixed digit, medial tooth; FPP, fixed digit, paraxial proximal tooth; FPSP, fixed digit, paraxial subproximal tooth; FSD, fixed digit, subdistal tooth; MD, movable digit, distal tooth; MM, movable digit, medial tooth; MP, movable digit, proximal tooth; MSD, movable digit, subdistal tooth.
Figure 4.
Neosuidasia sjorsvandenbergi sp. nov. (adult female). (A). Chelicera (DIC image), antiaxial view; (B). Chelicera (line drawing), antiaxial view; (C). Supracoxal sclerite, scx and Grandjean’s organ (DIC image); (D). Genital area (DIC image); (E). Receptaculum seminis and anal area (DIC image); (F). Inseminatory canal and receptaculum seminis (DIC image). Abbreviations: cha, cheliceral seta; FAP, fixed digit, antiaxial proximal tooth; FASP, fixed digit, antiaxial subproximal tooth; FD, fixed digit, distal tooth; FM, fixed digit, medial tooth; FPP, fixed digit, paraxial proximal tooth; FPSP, fixed digit, paraxial subproximal tooth; FSD, fixed digit, subdistal tooth; MD, movable digit, distal tooth; MM, movable digit, medial tooth; MP, movable digit, proximal tooth; MSD, movable digit, subdistal tooth.
Figure 5.
Neosuidasia sjorsvandenbergi sp. nov. (adult female). (A). Subcapitulum; (B). Supracoxal sclerite, scx and Grandjean’s organ; (C). scx; (D). Genital opening; (E), Inseminatory canal and receptaculum seminis.
Figure 5.
Neosuidasia sjorsvandenbergi sp. nov. (adult female). (A). Subcapitulum; (B). Supracoxal sclerite, scx and Grandjean’s organ; (C). scx; (D). Genital opening; (E), Inseminatory canal and receptaculum seminis.
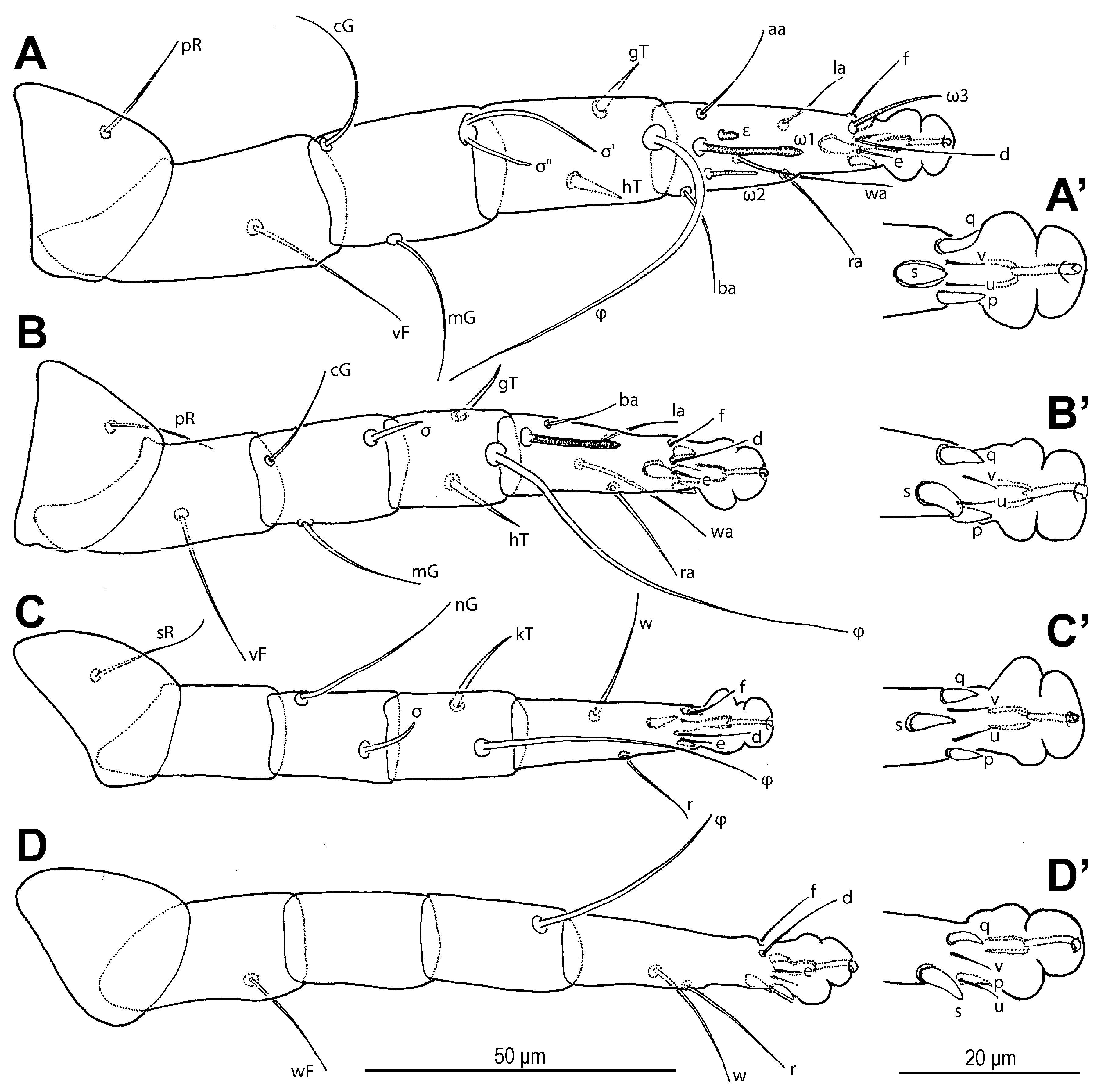
Figure 6.
Neosuidasia sjorsvandenbergi sp. nov. (adult female). (A–D). Legs I–IV, dorsal view; (A′–D′). Pretarsi I–IV, ventral view.
Figure 6.
Neosuidasia sjorsvandenbergi sp. nov. (adult female). (A–D). Legs I–IV, dorsal view; (A′–D′). Pretarsi I–IV, ventral view.
Figure 7.
Neosuidasia sjorsvandenbergi sp. nov. (adult female, DIC images). (A–D). Genu–tarsus of legs I–IV, dorsal view; (A′–D′). Genu–tarsus of legs I–IV, ventral view.
Figure 7.
Neosuidasia sjorsvandenbergi sp. nov. (adult female, DIC images). (A–D). Genu–tarsus of legs I–IV, dorsal view; (A′–D′). Genu–tarsus of legs I–IV, ventral view.
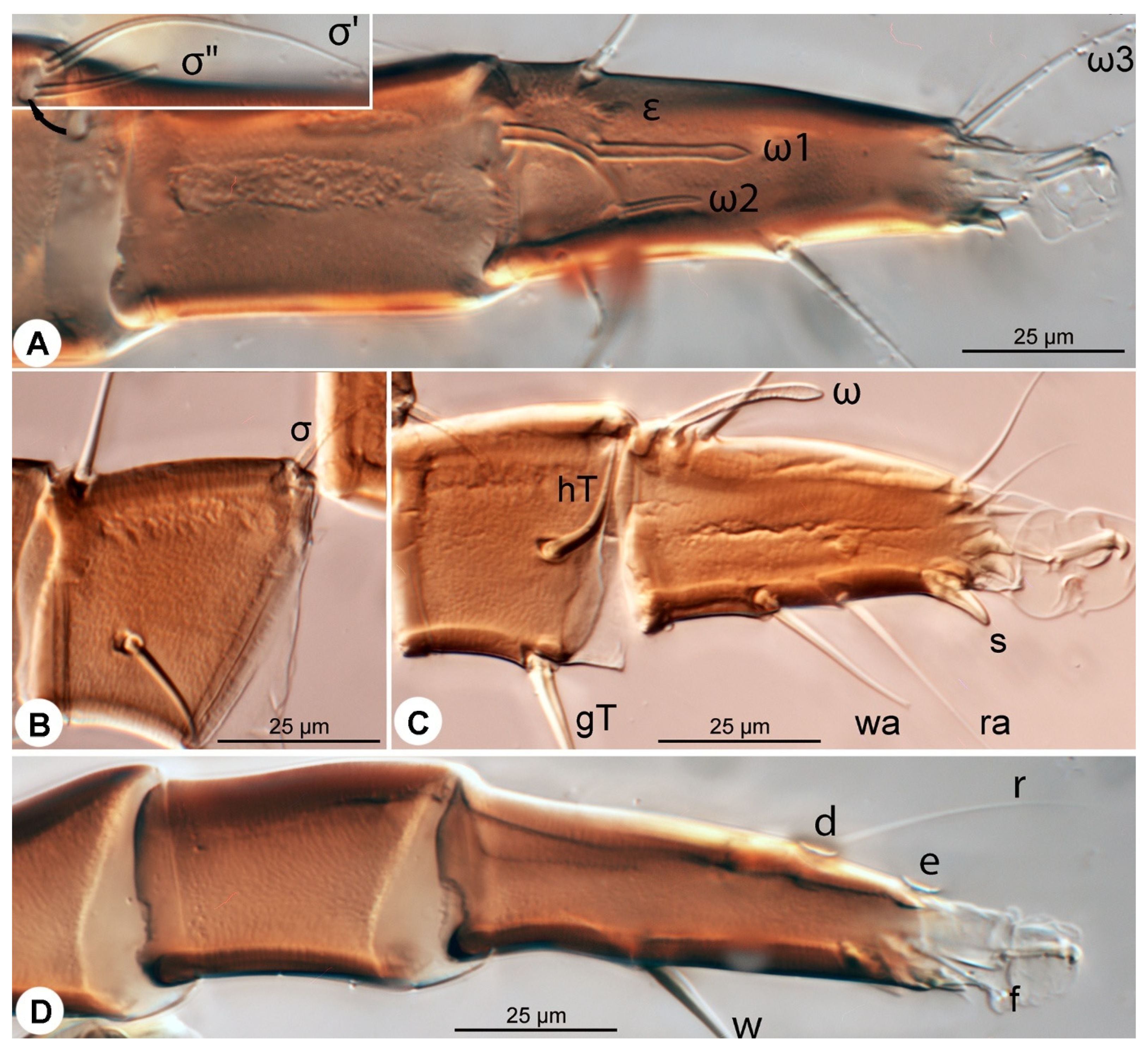
Leg I (
Figure 6A,A′ and
Figure 7A,A′): Genual solenidia ratio
σ′:
σ″ = 1.6 (1.2–1.6). Tibial solenidion
φ elongate, extending well beyond tarsal claw tip. Tarsus 2.3 (2.3–2.8)× as long as basal width. Solenidion
ω1 mostly cylindrical, tapering pre-apically into distinct terminal head;
ε conical, near base of
ω1;
ω2 cylindrical, slightly anterior to
ω1;
ω3 distal, mostly cylindrical, apically tapered, as long as
ω1. Seta
aa filiform, proximal to
ω1;
ba filiform, level with
ω1;
wa,
ra, and
la filiform,
wa and
ra similar in length to
aa and
ba, and clearly longer than
la;
d filiform, distal, projecting well beyond claw tip;
e a slender spine;
f filiform, nearly reaching claw tip. Subunguinal seta (
s) and proral setae (
p,
q) conical, ventrally curved; unguinal setae (
u,
v) slender, setiform.
Leg II (
Figure 6B,B′ and
Figure 7B,B′): Genual solenidion
σ extending to half-length of tibia. Tibial solenidion
φ elongate, extending beyond tarsal claw tip. Tarsus 2.1 (2.1–3.0)× as long as basal width. Solenidion
ω mostly cylindrical, tapering pre-apically into distinct terminal head. Seta
bb filiform, level with
ω;
wa,
ra, and
la filiform,
wa and
ra similar in length to
ba, and clearly longer than
la;
d filiform, distal, projecting well beyond claw tip;
e a slender spine;
f filiform, about reaching claw tip. Subunguinal seta (
s) and proral setae (
p,
q) conical, ventrally curved; unguinal setae (
u,
v) slender, setiform.
Leg III (
Figure 6C,C′ and
Figure 7C,C′): Genual solenidion
σ extending over half-length of tibia. Tibial solenidion
φ elongate, extending beyond tarsal claw tip. Tarsus 2.6 (2.6–3.1)× as long as basal width. Setae
w,
r,
d and
f filiform,
d projecting well beyond claw tip;
e a slender spine;
f about reaching claw tip. Subunguinal seta (
s) and proral setae (
p,
q) conical, ventrally curved; unguinal setae (
u,
v) slender, setiform.
Leg IV (
Figure 6D,D′ and
Figure 7D,D′): Tibial solenidion
φ reaching tarsal claw tip or nearly so. Tarsus 2.7 (2.7–3.8)× as long as basal width. Setae
w,
r,
d, and
f filiform,
d projecting well beyond claw tip;
e a slender spine;
f about reaching claw tip. Subunguinal seta (
s) and proral setae (
p,
q) conical, ventrally curved; unguinal setae (
u,
v) slender, setiform.
Table 1.
Measurements of morphological characters across life stages of Neosuidasia sjorsvandenbergi sp. nov.
Table 1.
Measurements of morphological characters across life stages of Neosuidasia sjorsvandenbergi sp. nov.
| | Females
(n = 5) | Males
(n = 5) | Tritonymphs
(n = 5) | Protonymphs
(n = 3) | Larvae
(n = 2) |
|---|
| Idiosoma-L | 398 (398–572) | 353–417 | 281–366 | 208–271 | 216 |
| Idiosoma-W | 269 (269–389) | 221–271 | 168–201 | 134–145 | 123 |
| Shield-L | 105 (103–136) | 96–117 | 66–79 | 59–74 | 43 |
| Shield-W | 86 (82–115) | 74–83 | 49–60 | 39–40 | 32–34 |
| vi | 63 (63–76) | 61–62 | 31–35 | 18–24 | 20–29 |
| vi–vi | 7 (7–10) | 6–9 | 5–6 | 5–6 | 4 |
| vi–ve | 50 (50–66) | 41–50 | 28–34 | 20–28 | 22–23 |
| ve | 28 (27–36) | 20–26 | 10–17 | 8–9 | 7–8 |
| ve–ve | 80 (80–108) | 66–79 | 43–57 | 36–40 | 32–34 |
| sci | 99 (89–167) | 103–119 | 44–57 | 18–25 | 12–16 |
| sci–sci | 58 (58–75) | 50–64 | 36–41 | 34–36 | 15–28 |
| sci–sce | 28 (28–35) | 28–31 | 20–28 | 14–18 | 15–17 |
| sce | 148 (148–197) | 139–152 | 103–109 | 68–79 | 59 |
| scx | 41 (41–49) | 29–34 | 18–26 | 17–21 | 12–15 |
| c1 | 102 (102–143) | 74–92 | 30–61 | 13–17 | 12–17 |
| c1–c1 | 77 (77–112) | 67–72 | 43–61 | 40–48 | 35–45 |
| c1–d1 | 53 (46–68) | 32–55 | 43–58 | 28–39 | 32 |
| c2 | 109 (109–151) | 78–112 | 47–67 | 24–36 | 14 |
| cp | 123 (123–169) | 129–134 | 79–82 | 58–68 | 44 |
| c3 | 62 (62–92) | 52–63 | 27–35 | 20–23 | 14 |
| d1 | 103 (103–154) | 99–133 | 31–60 | 13–18 | 12 |
| d1–d1 | 78 (76–107) | 48–67 | 38–55 | 34–41 | 31 |
| d1–e1 | 70 (57–117) | 71–95 | 50–84 | 46–52 | 43 |
| d2 | 101 (101–149) | 89–112 | 44–59 | 14–22 | 12 |
| e1 | 111 (111–152) | 122–133 | 51–74 | 13–31 | 13 |
| e1–e1 | 72 (72–105) | 56–66 | 37–57 | 37–42 | 35 |
| e2 | 116 (109–153) | 118–121 | 58–84 | 39–58 | 32 |
| f2 | 110 (110–145) | 117–142 | 67–91 | 38–67 | × |
| h1 | 125 (125–179) | 119–129 | 68–71 | 37–63 | 48 |
| h2 | 132 (132–158) | 117–131 | 67–82 | 36–54 | 49 |
| h3 | 153 (153–163) | 136–156 | 87–119 | 87–109 | × |
| ps1 | 117 (117–167) | 102–134 | 69–102 | 31–49 | × |
| ps2 | 97 (97–148) | 41–56 | 39–48 | 17–26 | × |
| ps3 | 42 (42–72) | 11–13 | 14–20 | 9–13 | × |
| ad1 | 85 (85–122) | × | × | × | × |
| ad2 | 77 (77–113) | × | × | × | × |
| ad3 | 39 (39–71) | × | × | × | × |
| Anus-L | 90 (90–128) | 66–74 | 53–64 | 39–47 | 36 |
| 1a | 64 (59–77) | 52–61 | 26–36 | 18–25 | 15 |
| 4b | 34 (34–53) | 28–35 | 15–19 | × | × |
| 3a | 37 (37–71) | 42–58 | 21–25 | 15–21 | 14 |
| g | 24 (24–33) | 18–20 | 16–17 | 8–12 | × |
| 4a | 41 (39–58) | 31–49 | × | × | × |
| Gen-L | 67 (58–85) | 44–50 | 24–29 | 14–16 | × |
| Gen–anus | 86 (86–144) | 4–22 | 25–38 | 24–41 | n/a |
| M-digit | 46 (46–57) | 42–48 | 27–30 | 17–23 | 16–17 |
| cha | 11 (10–15) | 7–10 | 6–Jul | 5–6 | 5–6 |
| h | 36 (34–44) | 34–42 | 19–24 | 19–21 | 13–14 |
| elcp | 8 (8–10) | 7–9 | 4–5 | 3–4 | 2 |
| sup | 27 (24–27) | 28–37 | 15–20 | 13–15 | 12 |
| a | 24 (20–31) | 20–36 | 13–14 | 10–12 | 9–10 |
| cm | 21 (21–27) | 21–26 | 14–15 | 12–15 | 10–12 |
| ω | 8 (7–8) | 7–9 | 4–5 | 4 | 3 |
| Leg I | 196 (196–289) | 195–238 | 134–155 | 95–126 | 75–81 |
| Fem-I-L | 71 (70–89) | 64–78 | 46–51 | 31–40 | 28–33 |
| vF I | 44 (44–54) | 41–43 | 27–32 | 17–24 | 17–19 |
| Genu-I-L | 43 (41–59) | 43–50 | 30–33 | 18–25 | 17 |
| cG I | 44 (44–77) | 41–50 | 24–30 | 20–22 | 17–18 |
| mG I | 42 (42–61) | 45–57 | 22–27 | 18–25 | 17 |
| σ′ I | 36 (36–47) | 36–50 | 23–28 | 17–24 | 18–19 |
| σ″ I | 23 (23–32) | 21–32 | 17–18 | 9–14 | 9 |
| Ti-I-L | 42 (41–55) | 42–49 | 30–32 | 20–25 | 17–18 |
| gT I | 18 (17–30) | 14–16 | 11–15 | 9–15 | 8–9 |
| hT I | 17 (17–32) | 14–18 | 10–12 | 8–9 | 7 |
| φ I | 77 (77–92) | 84–101 | 68–82 | 55–63 | 49 |
| Ta-I-L | 49 (49–72) | 49–57 | 34–38 | 24–29 | 21–22 |
| Ta-I-W | 21 (21–26) | 21–26 | 15–21 | 11–15 | 11–13 |
| ω1 I | 18 (18–22) | 19–25 | 15–18 | 14–17 | 12–13 |
| ω2 I | 8 (8–12) | 9–11 | 7–8 | 4–7 | × |
| ω3 I | 22 (22–25) | 20–28 | 14–16 | × | × |
| ε I | 4 (3–4) | 2–4 | 3–4 | 3 | 2 |
| aa | 31 (31–38) | 29–36 | 16–21 | 15–19 | 9–10 |
| ba | 32 (30–37) | 27–33 | 20–23 | 17–20 | 9–10 |
| wa I | 37 (33–41) | 37–48 | 17–26 | 20–24 | 11–13 |
| la I | 22 (22–26) | 19–25 | 11–16 | 11–15 | 7–9 |
| ra I | 31 (31–37) | 30–43 | 15–20 | 17–20 | 12–13 |
| d I | 33 (31–36) | 23–36 | 17–20 | 15–22 | 19–20 |
| e I | 6 (6–7) | 7 | 5–6 | 4–5 | 3 |
| f I | 16 (16–18) | 12–19 | 13–14 | 8–10 | 7–8 |
| s I | 12 (12–16) | 9–12 | 8–9 | 5–7 | 4–5 |
| u I | 7 (6–7) | 6–7 | 4–6 | 3–5 | 3–4 |
| v I | 7 (6–7) | 6–7 | 4–6 | 3–5 | 3–4 |
| p I | 8 (8–10) | 7–8 | 5–6 | 4–5 | 4–5 |
| q I | 8 (8–10) | 7–8 | 5–6 | 4–5 | 4–5 |
| Claw-I | 10 (10–12) | 8–11 | 7 | 6–7 | 5 |
| Leg II | 154 (154–233) | 164–201 | 113–128 | 82–102 | 55–58 |
| Fem-II-L | 57 (57–75) | 53–68 | 40–43 | 27–33 | 22–23 |
| vF II | 48 (44–65) | 41–52 | 28–32 | 20–24 | 14–16 |
| Genu-II-L | 30 (30–45) | 28–40 | 22–26 | 15–20 | 17–20 |
| cG II | 42 (42–66) | 36–39 | 28–31 | 17–21 | 15–17 |
| mG II | 33 (31–65) | 35–45 | 22–26 | 18–23 | 15–16 |
| σ II | 15 (15–19) | 13–33 | 9–12 | 7–9 | 5–6 |
| Ti-II-L | 30 (30–40) | 27–33 | 20–24 | 14–19 | 17–20 |
| gT II | 16 (16–26) | 17–20 | 9–11 | 8–10 | 8 |
| hT II | 15 (15–19) | 14–18 | 9–11 | 8–10 | 7 |
| φ II | 79 (79–98) | 79–85 | 66–71 | 36–52 | 38–42 |
| Ta-II-L | 44 (44–64) | 45–53 | 31–33 | 22–25 | 21–23 |
| Ta-II-W | 23 (18–23) | 17–19 | 12–15 | 10–11 | 10–11 |
| ω II | 18 (18–22) | 19–25 | 16–18 | 11–14 | 11–12 |
| ba II | 31 (31–36) | 21–33 | 15–18 | 11–12 | 9–11 |
| wa II | 33 (33–42) | 37–43 | 20–23 | 15–21 | 12–14 |
| la II | 20 (20–31) | 16–20 | 12–14 | 9–13 | 7–8 |
| ra II | 33 (28–39) | 27–37 | 17–22 | 15–20 | 12–13 |
| d II | 27 (24–29) | 26–34 | 15–18 | 15–20 | 11–13 |
| e II | 6 (6–9) | 6–7 | 5–7 | 4–5 | 3 |
| f II | 16 (15–17) | 12–17 | 11–12 | 9–12 | 7–8 |
| s II | 12 (10–12) | 7–9 | 7–8 | 5–7 | 4–5 |
| u II | 7 (6–7) | 6 | 5–6 | 3–5 | 3–4 |
| v II | 7 (6–7) | 6 | 5–6 | 3–5 | 3–4 |
| p II | 8 (7–8) | 6–7 | 5–6 | 4–6 | 3–4 |
| q II | 8 (7–8) | 6–7 | 5–6 | 4–6 | 3–4 |
| Claw II | 10 (10–11) | 8–10 | 7 | 5–7 | 5 |
| Leg III | 152 (152–229) | 162–188 | 115–121 | 69–94 | 79 |
| Fem-III-L | 47 (47–55) | 44–52 | 32–34 | 21–28 | 21 |
| Genu-III-L | 30 (30–35) | 29–34 | 21–23 | 15–19 | 16 |
| nG III | 46 (46–76) | 49–65 | 34–36 | 17–23 | 18 |
| σ III | 21 (21–27) | 14–25 | 14–16 | 9–13 | 7 |
| Ti-III-L | 28 (28–39) | 29–31 | 21–22 | 15–19 | 18 |
| kT III | 16 (16–26) | 19–23 | 13–15 | 10–13 | 12 |
| φ III | 79 (79–94) | 74–86 | 45–62 | 44–54 | 42 |
| Ta-III-L | 46 (46–65) | 43–50 | 27–32 | 18–28 | 42 |
| Ta-III-W | 18 (18–20) | 15–27 | 12–13 | 9–10 | 11 |
| w III | 39 (39–50) | 38–42 | 27–31 | 16–20 | 10 |
| r III | 27 (27–40) | 36–39 | 14–19 | 11–15 | 8 |
| d III | 20 (20–29) | 16–24 | 15–21 | 14–19 | 12 |
| e III | 7 (6–8) | 7–8 | 5–6 | 5 | 4 |
| f III | 15 (15–17) | 15–19 | 10–12 | 8–10 | 7 |
| s III | 9 (8–11) | 8 | 5–6 | 4–5 | 5 |
| u III | 7 (7–8) | 5–6 | 5–6 | 3 | 4 |
| v III | 7 (7–8) | 5–6 | 5–6 | 3 | 4 |
| p III | 8 (7–8) | 6–7 | 5–6 | 5 | 3 |
| q III | 8 (7–8) | 6–7 | 5–6 | 5 | 3 |
| Claw III | 10 (10–11) | 8–10 | 7 | 6–7 | 5 |
| Leg IV | 183 (183–263) | 182–229 | 126–138 | 84–103 | × |
| Fem-IV-L | 51 (51–69) | 41–58 | 34–36 | 20–26 | × |
| wF IV | 50 (50–67) | 36–56 | 24–28 | × | × |
| Gen-IV-L | 34 (34–48) | 31–41 | 22–24 | 15–19 | × |
| Ti-IV-L | 34 (33–45) | 34–40 | 24–25 | 16–18 | × |
| φ IV | 54 (54–78) | 46–66 | 28–35 | × | × |
| Ta-IV-L | 51 (51–75) | 45–62 | 35–38 | 23–28 | × |
| Ta-IV-W | 19 (19–22) | 17–22 | 11–12 | 9–10 | × |
| w IV | 35 (35–51) | 37–51 | 27–30 | 16–21 | × |
| r IV | 31 (31–45) | 34–45 | 17–24 | 13–17 | × |
| d IV | 31 (25–33) | ● | 19–22 | 14–20 | × |
| e IV | 7 (7–8) | ● | 4–6 | × | × |
| f IV | 16 (16–20) | 16–22 | 10–11 | × | × |
| s IV | 9 (9–12) | 8–10 | 6–7 | ■ | × |
| u IV | 6 (6–9) | 5–8 | 5–6 | 3 | × |
| v IV | 6 (6–9) | 5–8 | 5–6 | 5 | × |
| p IV | 7 (7–8) | 6–8 | 6 | 4–6 | × |
| q IV | 7 (7–8) | 6–8 | 6 | 4–6 | × |
| Claw IV | 10 (10–11) | 8–10 | 7–8 | 6–7 | × |
Dorsum (
Figure 8,
Figure 10C and
Figure 11C,D) broadly pyriform to oval; cuticle finely mammillated over nearly entire dorsal surface except anteromedial region between sejugal suture and level of
c1. Prodorsal shield, supracoxal sclerite, Grandjean’s organ, and supracoxal setae (
scx) as in adult female. Idiosomal setae filiform;
vi ~3/4 prodorsal shield width;
ve slightly longer than 1/3
vi;
sci 4/5 length of
sce; distance
sci–sci 1.7–2.0×
sci–sce. Distance
gla–e2 ~2×
gla–d2. Setae
c3 shortest, positioned ventrally; remaining setae subequal, long, extending beyond bases of following row. Cupules:
ia posterolateral to
c1 and posteromedial to
c2;
im on ventral surface, lateral to coxa IV;
ip anteromedial to and near
f2.
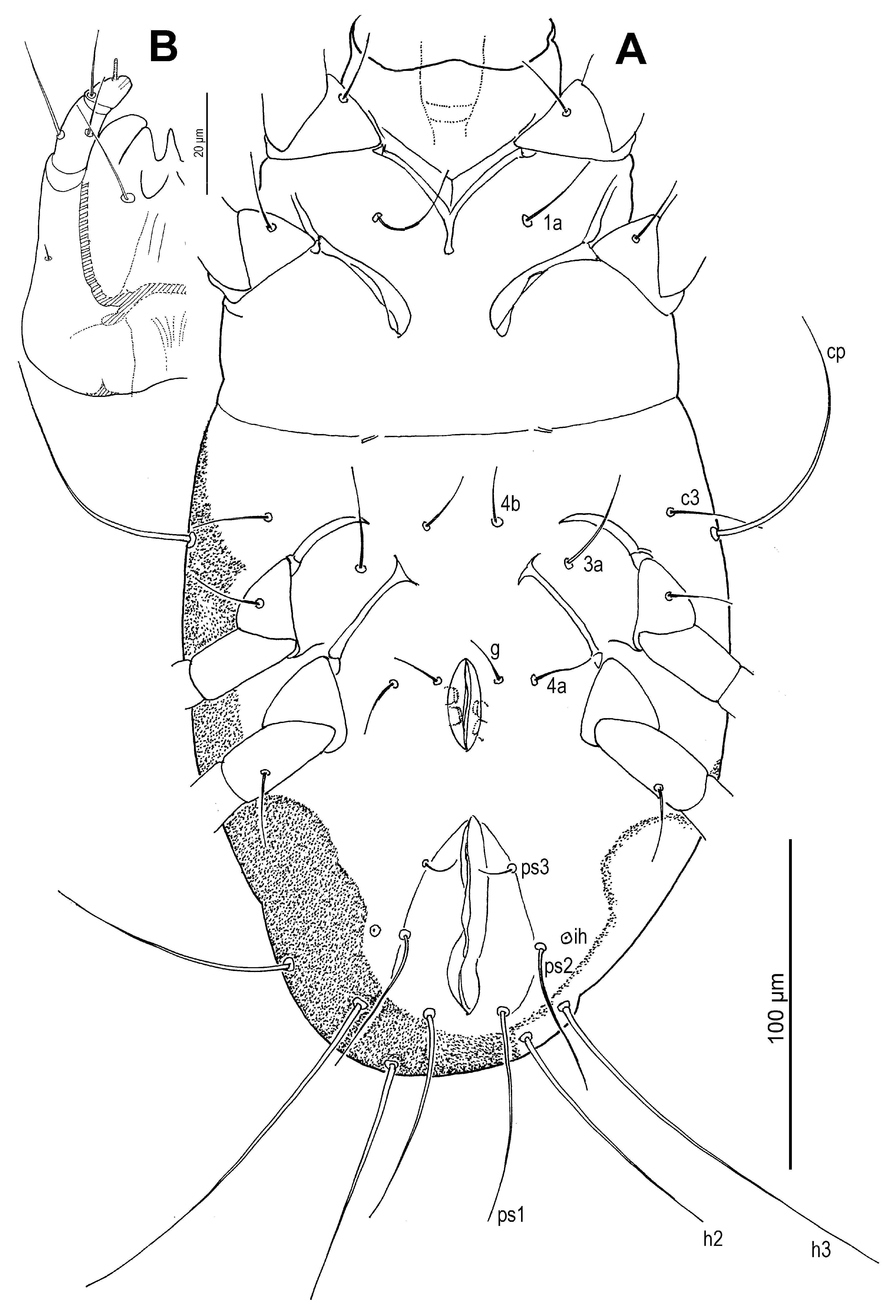
Venter (
Figure 9,
Figure 10D,E and
Figure 11E–G): Cuticle finely mammillated across anterolateral regions of coxae III, lateral surfaces of coxae III–IV, posterior to coxae IV, and along lateral areas near anus. Coxal apodemes and plates as in adult female, except posterior apodemes of coxae II broader, lateral portion approximately 2× maximum width of coxa II. Positions of coxisternal setae
1a,
4b, and
4a as in adult female. Genital opening (
Figure 9,
Figure 10D and
Figure 11E,F) inverted V-shaped, markedly narrower than in female, located between trochanters IV; setae
g at level of first pair of genital papillae. Genital and anal openings close, separated by less than half length of genital opening. Aedeagus (
Figure 10D,E and
Figure 11E,F) straight, gradually tapered, 44–46 long, extending nearly to anterior edge of supporting sclerites. Anal opening approximately 2× genital opening length. Adanal setae absent. Anal suckers pair, faintly sclerotised, posterolateral to anal opening. Pseudanal setae:
ps1 posterior to anus,
ps2 posterolateral to anus,
ps3 anterior to anal suckers;
ps1 longest, 4.6–4.7×
ps2, 10.0–11.0×
ps3;
ps2 4.2–4.7×
ps3. Cupule
ih anterolateral to
ps2.
Gnathosoma (
Figure 10A,B and
Figure 11A,B). Chelicerae teeth, seta
cha, subcapitular seta
h, palpal supracoxal seta
elcp, palptibial setae
sup and
a, palptarsal seta
cm, palptarsal solenidion
ω, eupathidium
ul″ as in adult female, except for differences in dimensions.
Legs (
Figure 12 and
Figure 13): Legs I and IV longer and wider than II and III. Setal formulae identical to female, except tarsus IV with two suckers, modified from setae
d and
e.
Leg I (
Figure 12A,A′ and
Figure 13A): Genual solenidia ratio
σ′:
σ″ = 1.5–1.9. Tibial solenidion
φ extending far beyond tarsal claw tip. Tarsus 2.2–2.6× basal width. Solenidion
ω1 cylindrical with terminal head;
ε apically tapered, near base of
ω1;
ω2 cylindrical, slightly anterior to
ω1;
ω3 dorsal distal, tapered, longer than
ω1. Setae
aa,
ba,
wa,
ra,
la,
d,
f filiform;
e slender spine. Subunguinal seta (
s), proral setae (
p,
q), and unguinal setae (
u,
v) as in adult female.
Figure 8.
Neosuidasia sjorsvandenbergi sp. nov. (adult male). Idiosoma, dorsal view.
Figure 8.
Neosuidasia sjorsvandenbergi sp. nov. (adult male). Idiosoma, dorsal view.
Figure 9.
Neosuidasia sjorsvandenbergi sp. nov. (adult male). Idiosoma, ventral view.
Figure 9.
Neosuidasia sjorsvandenbergi sp. nov. (adult male). Idiosoma, ventral view.
Figure 10.
Neosuidasia sjorsvandenbergi sp. nov. (adult male). (A). Subcapitulum; (B). Chelicera; (C). Supracoxal sclerite, scx, and Grandjean’s organ; (D). Genital opening and aedeagus; (E). Aedeagus.
Figure 10.
Neosuidasia sjorsvandenbergi sp. nov. (adult male). (A). Subcapitulum; (B). Chelicera; (C). Supracoxal sclerite, scx, and Grandjean’s organ; (D). Genital opening and aedeagus; (E). Aedeagus.
Figure 11.
Neosuidasia sjorsvandenbergi sp. nov. (adult male, DIC images). (A). Chelicera; (B). Subcapitulum and chelicera; (C,D). Supracoxal sclerite, scx, and Grandjean’s organ; (E,F). Genital opening and aedeagus; (G). Anal area.
Figure 11.
Neosuidasia sjorsvandenbergi sp. nov. (adult male, DIC images). (A). Chelicera; (B). Subcapitulum and chelicera; (C,D). Supracoxal sclerite, scx, and Grandjean’s organ; (E,F). Genital opening and aedeagus; (G). Anal area.
Figure 12.
Neosuidasia sjorsvandenbergi sp. nov. (adult male). (A–D). Legs I–IV, dorsal view; (A′–D′). Pretarsi I–IV, ventral view.
Figure 12.
Neosuidasia sjorsvandenbergi sp. nov. (adult male). (A–D). Legs I–IV, dorsal view; (A′–D′). Pretarsi I–IV, ventral view.
Figure 13.
Neosuidasia sjorsvandenbergi sp. nov. (adult male, DIC images). (A). Genu–tarsus of legs I, dorsal view; (B). Genu of legs II, dorsal view; (C). Tibia and tarsus of legs II, dorsal view; (D). Genu–tarsus of leg IV, dorsal view.
Figure 13.
Neosuidasia sjorsvandenbergi sp. nov. (adult male, DIC images). (A). Genu–tarsus of legs I, dorsal view; (B). Genu of legs II, dorsal view; (C). Tibia and tarsus of legs II, dorsal view; (D). Genu–tarsus of leg IV, dorsal view.
Leg II (
Figure 12B,B′ and
Figure 13B,C): Genual solenidion
σ extending near half-length of tibia;
φ extending far beyond tarsal claw tip. Tarsus 2.7–2.8× basal width. Solenidion
ω cylindrical with terminal head. Setae
ba,
wa,
ra,
la,
d,
f filiform; e slender spine. Subunguinal seta (
s), proral setae (
p,
q), and unguinal setae (
u,
v) as in adult female.
Leg III (
Figure 12C,C′): Genual solenidion
σ reaching base of tibial solenidion
φ;
φ extending far beyond tarsal claw tip. Tarsus 1.7–2.3× basal width. Setae
w,
r,
d,
f filiform;
e slender spine. Subunguinal seta (
s), proral setae (
p,
q), and unguinal setae (
u,
v) as in adult female.
Leg IV (
Figure 12D,D′ and
Figure 13D): Tibial solenidion
φ slightly passing
f. Tarsus 2.9–3.3× basal width. Setae
w,
r,
f filiform;
d and
e sucker-like, slightly wider than alveolus of
w or
r, distance
d–
e 11–17 Subunguinal seta (
s), proral setae (
p,
q), and unguinal setae (
u,
v) as in adult female.
Dorsum (
Figure 14A,B,
Figure 16A and
Figure 17A) elongated oval; cuticle finely mammillated across most of dorsal surface, except near sejugal suture, specifically posterior to scapular setae and anteromedial and medial to
c1. Prodorsal shield rectangular anterior to
sci, narrowing posteromedially; supracoxal sclerite and Grandjean’s organ as in adult female;
scx pectinate, bearing 20–26 long teeth. Idiosomal setae filiform;
vi about 1/2 prodorsal shield width;
ve less than 1/2
vi;
sci 1/2 length of
sce or longer; distance
sci–
sci 1.5–1.6×
sci–
sce. Distance
gla–
e2 near 3×
gla–
d2. Setae
c3 shortest, lateral to coxae III; others extending beyond or near bases of following row. Cupules:
ia posteromedial to
c2;
im marginal;
ip close to and anteromedial to
f2.
Venter (
Figure 15A and
Figure 16B): Cuticle finely mammillated across regions lateral to coxae III–IV, extending posterior beyond coxae IV and laterally to level of cupules
ih. Coxal apodemes and plates similar to those of female, except posterior apodemes of coxae II indistinct. Coxisternal setae
1a,
4b, and
4a positioned as in adult female. Genital opening a longitudinal slit between trochanters IV; setae
g at level of
4a. Distance between genital and anal openings near length of genital slit. Anal opening more than twice length of genital opening. Adanal setae absent. Pseudanal setae
ps1 at same level of posterior margin of anus;
ps2 and
ps3 located laterally;
ps1 longest, 1.6–2.0×
ps2, 3.8–6.4×
ps3;
ps2 2.3–3.3×
ps3. Cupule
ih anterolateral to
ps2.
Gnathosoma (
Figure 14C,
Figure 15B,
Figure 16B and
Figure 17B): Structure and setation of chelicerae, subcapitulum, and palps as in adult female, except for differences in dimensions.
Legs (
Figure 17 and
Figure 18): Legs I and IV longer than legs II and III. Setal formulae same as those of female.
Leg I (
Figure 17C,C′ and
Figure 18A,A′): Ratio
σ′:
σ″ = 1.3–1.7. Tibial solenidion φ extends well beyond tarsal claw tip. Tarsus length 1.7–2.3× basal width. Solenidion
ω1 cylindrical with terminal head;
ε tapered apically, located anterior to
ω1 base;
ω2 cylindrical, slightly anterior to
ω1;
ω3 positioned dorsodistally, tapered, slightly shorter than
ω1. Setae
aa,
ba,
wa,
ra,
la,
d, and
f filiform;
e setiform. Subunguinal seta (
s), proral setae (
p,
q), and unguinal setae (
u,
v) as in adult female.
Leg II (
Figure 17D,D′ and
Figure 18B,B′): Genual solenidion
σ extending to nearly one-third of tibial length; tibial solenidion
φ extends well beyond tarsal claw tip. Tarsus 2.1–2.4× basal width. Solenidion
ω cylindrical with terminal head. Setae
ba,
wa,
ra,
la,
d,
f filiform;
e setiform. Subunguinal seta (
s), proral setae (
p,
q), and unguinal setae (
u,
v) as in adult female.
Leg III (
Figure 18C,C′): Genual solenidion
σ reaching about one-third of tibial length; tibial solenidion
φ reaches tarsal claw tip. Tarsus 2.4–2.7× basal width. Setae
w,
r,
d,
f filiform;
e setiform. Subunguinal seta (
s), proral setae (
p,
q), and unguinal setae (
u,
v) as in adult female.
Leg IV (
Figure 18D,D′): Tibial solenidion
φ nearly reaching base of seta
f. Tarsus length 3.0–3.2× basal width. Setae
w,
r,
d,
f filiform;
e setiform. Subunguinal seta (
s), proral setae (
p,
q), and unguinal setae (
u,
v) as in adult female.
Figure 14.
Neosuidasia sjorsvandenbergi sp. nov. (tritonymph). (A). Idiosoma, dorsal view; (B). scx; (C). Chelicera.
Figure 14.
Neosuidasia sjorsvandenbergi sp. nov. (tritonymph). (A). Idiosoma, dorsal view; (B). scx; (C). Chelicera.
Figure 15.
Neosuidasia sjorsvandenbergi sp. nov. (tritonymph). (A). Idiosoma, ventral view; (B). Subcapitulum.
Figure 15.
Neosuidasia sjorsvandenbergi sp. nov. (tritonymph). (A). Idiosoma, ventral view; (B). Subcapitulum.
Figure 16.
Neosuidasia sjorsvandenbergi sp. nov. (tritonymph, DIC images). (A). Dorsal view; (B). Ventral view.
Figure 16.
Neosuidasia sjorsvandenbergi sp. nov. (tritonymph, DIC images). (A). Dorsal view; (B). Ventral view.
Figure 17.
Neosuidasia sjorsvandenbergi sp. nov. (tritonymph, DIC images). (A). Prodorsum; (B). Subcapitulum; (C). Tibia and tarsus of legs I, dorsal view; (C′). Tibia and tarsus of legs I, ventral view; (D). Tibia and tarsus of legs II, dorsal view; (D′). Tibia and tarsus of legs II, ventral view.
Figure 17.
Neosuidasia sjorsvandenbergi sp. nov. (tritonymph, DIC images). (A). Prodorsum; (B). Subcapitulum; (C). Tibia and tarsus of legs I, dorsal view; (C′). Tibia and tarsus of legs I, ventral view; (D). Tibia and tarsus of legs II, dorsal view; (D′). Tibia and tarsus of legs II, ventral view.
Figure 18.
Neosuidasia sjorsvandenbergi sp. nov. (tritonymph). (A–D). Legs I–IV, dorsal view; (A′–D′). Pretarsi I–IV, ventral view.
Figure 18.
Neosuidasia sjorsvandenbergi sp. nov. (tritonymph). (A–D). Legs I–IV, dorsal view; (A′–D′). Pretarsi I–IV, ventral view.
Dorsum (
Figure 19A,B and
Figure 21A) elongated oval; cuticle finely mammillated across most of dorsal surface, except along sejugal suture. Prodorsal shield rectangular, with posteromedial extension. Supracoxal sclerite and Grandjean’s organ as in adult female;
scx pectinate with 18–24 teeth. Idiosomal setae filiform;
vi approximately 0.7× width of prodorsal shield;
ve about one-quarter length of
vi. Setae
sci less than one-third length of
sce; distance
sci–
sci 2.0–2.1×
sci–
sce. Distance
gla–
e2 nearly 4×
gla–d2. Setae
c3 situated anterolateral to coxae III, subequal to
c1,
d1, and
d2. Setae
c1,
d1,
d2, and
e2 well separated from bases of following row. Cupules indistinct.
Venter (
Figure 20A and
Figure 21B): Mammillated area reduced compared to tritonymph, restricted to lateral margins lateral to coxae III–IV and anal region. Coxal apodemes and plates similar to female, without indistinct posterior apodemes of coxae II. Coxisternal setae
4a and
4b absent. Genital opening a short longitudinal slit between trochanters IV; setae
g lateral to anterior part of Genital opening. Distance between genital and anal openings approximately 2× length of genital slit. Anal opening nearly 3× longer than genital slit. Adanal setae absent. Pseudanal setae:
ps1 at level of posterior anus margin;
ps2 and
ps3 lateral. Seta
ps1 longest, 1.4–1.6×
ps2, 3.8–4.0×
ps3;
ps2 2.5–2.8×
ps3. Cupule
ih positioned posterolaterally to
ps2.
Gnathosoma (
Figure 19C,
Figure 20B and
Figure 21B): Chelicerae, subcapitulum, and palpal setation as in adult female, differing only in dimensions.
Legs (
Figure 22): Leg I distinctly longer than others. Trochanters, femur IV, and tibia IV nude.
Leg I (
Figure 22A,A′): Genual solenidia ratio
σ′:σ″ = 1.6–1.7. Tibial solenidion
φ extends well beyond tarsal claw tip. Tarsus 1.8–2.0× basal width. Solenidion
ω1 cylindrical with terminal head;
ε apically tapered, positioned anterolateral to base of
ω1;
ω2 cylindrical, slightly anterolateral to
ω1;
ω3 absent. Setae
aa,
ba,
wa,
ra,
la,
d,
f filiform;
e setiform. Subunguinal seta (
s), proral (
p,
q), and unguinal setae (
u,
v) as in adult female.
Leg II (
Figure 22B,B′): Genual solenidion
σ reaching nearly one-fourth of tibial length; tibial solenidion
φ extends to tarsal claw tip. Tarsus 2.1–2.2× basal width. Solenidion
ω cylindrical with terminal head. Setae
ba,
wa,
ra,
la,
d,
f filiform;
e setiform. Subunguinal seta (
s), proral (
p,
q), and unguinal setae (
u,
v) as in adult female.
Leg III (
Figure 22C,C′): Genual solenidion σ reaches about one-fourth of tibial length;
φ nearly reaches base of seta
d. Tarsus 2.7–2.8× basal width. Setae
w,
r,
d,
f filiform;
e setiform. Subunguinal seta (
s), proral (
p,
q), and unguinal setae (
u,
v) as in adult female.
Leg IV (
Figure 22D,D′): Tibial solenidion
φ absent. Tarsus 3.7–3.8× basal width. Setae
e and
f absent;
w,
r,
d filiform. Subunguinal seta (
s) undeveloped; proral (
p,
q), and unguinal setae (
u,
v) as in adult female.
Figure 19.
Neosuidasia sjorsvandenbergi sp. nov. (protonymph). (A). Idiosoma, dorsal view; (B). scx; (C). Chelicera.
Figure 19.
Neosuidasia sjorsvandenbergi sp. nov. (protonymph). (A). Idiosoma, dorsal view; (B). scx; (C). Chelicera.
Figure 20.
Neosuidasia sjorsvandenbergi sp. nov. (protonymph). (A). Idiosoma, ventral view; (B). Subcapitulum.
Figure 20.
Neosuidasia sjorsvandenbergi sp. nov. (protonymph). (A). Idiosoma, ventral view; (B). Subcapitulum.
Figure 21.
Neosuidasia sjorsvandenbergi sp. nov. (protonymph, DIC images). (A). Dorsal view; (B). Ventral view.
Figure 21.
Neosuidasia sjorsvandenbergi sp. nov. (protonymph, DIC images). (A). Dorsal view; (B). Ventral view.
Figure 22.
Neosuidasia sjorsvandenbergi sp. nov. (protonymph). (A–D). Legs I–IV, dorsal view; (A′–D′). Pretarsi I–IV, ventral view.
Figure 22.
Neosuidasia sjorsvandenbergi sp. nov. (protonymph). (A–D). Legs I–IV, dorsal view; (A′–D′). Pretarsi I–IV, ventral view.
Dorsum (
Figure 23 and
Figure 25A) oval; cuticle finely mammillated across most of dorsal surface, except for a narrow, smooth strip adjacent to sejugal suture. Prodorsal shield rectangular. Supracoxal sclerite and Grandjean’s organ as in the female;
scx pectinate (
Figure 23B), bearing 20–24 teeth. Idiosomal setae
f2 and
h3 absent; remaining setae filiform. Seta
vi approximately 0.7× the width of the prodorsal shield;
ve slightly more than 1/4 length of
vi;
sci roughly 1/5 length of
sce; distance between
sci–sci 1.6–1.7× that of
sci–sce. Distance
gla–
e2 nearly 4×
gla–
d2. Setae
c3 positioned anterolateral to coxae III, subequal in length to
c1,
c2,
d1,
d2, and
e1; these setae notably distant from bases of the following row. Cupules indistinct.
Venter (
Figure 24B,C and
Figure 25B) with mammillated area as in tritonymph, finely mammillated across regions lateral to coxae III–IV, extending posterior beyond coxae IV and laterally to anus. Coxal apodemes and plates indistinct. Genital opening, coxisternal setae
4a,
4b, adanal and pseudanal setae absent. Claparède’s organ (
Figure 24C) cylindrical (6.5–7.5), about twice as long as wide, located lateral to
1a and posterior to trochanter I.
Gnathosoma (
Figure 24B and
Figure 25B): Structure and setation of the chelicerae, subcapitulum, and palps as in the female, differing only in dimensions (
Table 1).
Legs (
Figure 26): Leg II shorter than legs I and III; leg IV absent.
Leg I (
Figure 26A,A′): Genual solenidia ratio
σ′:σ″ = 2.0. Tibial solenidion
φ extending well beyond tarsal claw tip. Tarsus nearly 2.0× basal width. Solenidion
ω1 cylindrical with a prominent terminal head;
ε slightly tapered apically, positioned clearly anterolateral to base of
ω1;
ω2 and
ω3 absent. Setae
aa,
ba,
wa,
ra,
la,
d,
f filiform;
e setiform. Subunguinal seta (
s), proral (
p,
q), and unguinal setae (
u,
v) as in adult female.
Leg II (
Figure 26B,B′): Genual solenidion
σ reaching one-third of tibial length; tibial solenidion
φ extending well beyond tarsal claw tip. Tarsus 2.1× basal width. Solenidion
ω cylindrical with a prominent terminal head. Setae
ba,
wa,
ra,
la,
d,
f filiform;
e setiform. Subunguinal seta (
s), proral (
p,
q), and unguinal setae (
u,
v) as in adult female.
Leg III (
Figure 26C,C′): Genual solenidion σ reaching about one-fourth of tibial length;
φ extending well beyond tarsal claw tip. Tarsus 2.1× basal width. Setae
w,
r,
d,
f filiform;
e setiform. Subunguinal seta (
s), proral (
p,
q), and unguinal setae (
u,
v) as in adult female.
Figure 23.
Neosuidasia sjorsvandenbergi sp. nov. (larva). (A). Idiosoma, dorsal view; (B). scx.
Figure 23.
Neosuidasia sjorsvandenbergi sp. nov. (larva). (A). Idiosoma, dorsal view; (B). scx.
Figure 24.
Neosuidasia sjorsvandenbergi sp. nov. (larva). (A). Idiosoma, ventral view; (B). Subcapitulum; (C). Claparède’s organ.
Figure 24.
Neosuidasia sjorsvandenbergi sp. nov. (larva). (A). Idiosoma, ventral view; (B). Subcapitulum; (C). Claparède’s organ.
Figure 25.
Neosuidasia sjorsvandenbergi sp. nov. (larva, DIC images). (A). Dorsal view; (B). Ventral view.
Figure 25.
Neosuidasia sjorsvandenbergi sp. nov. (larva, DIC images). (A). Dorsal view; (B). Ventral view.
Figure 26.
Neosuidasia sjorsvandenbergi sp. nov. (larva). (A–C). Legs I–III; (A′–C′). Pretarsi I–III, ventral view.
Figure 26.
Neosuidasia sjorsvandenbergi sp. nov. (larva). (A–C). Legs I–III; (A′–C′). Pretarsi I–III, ventral view.
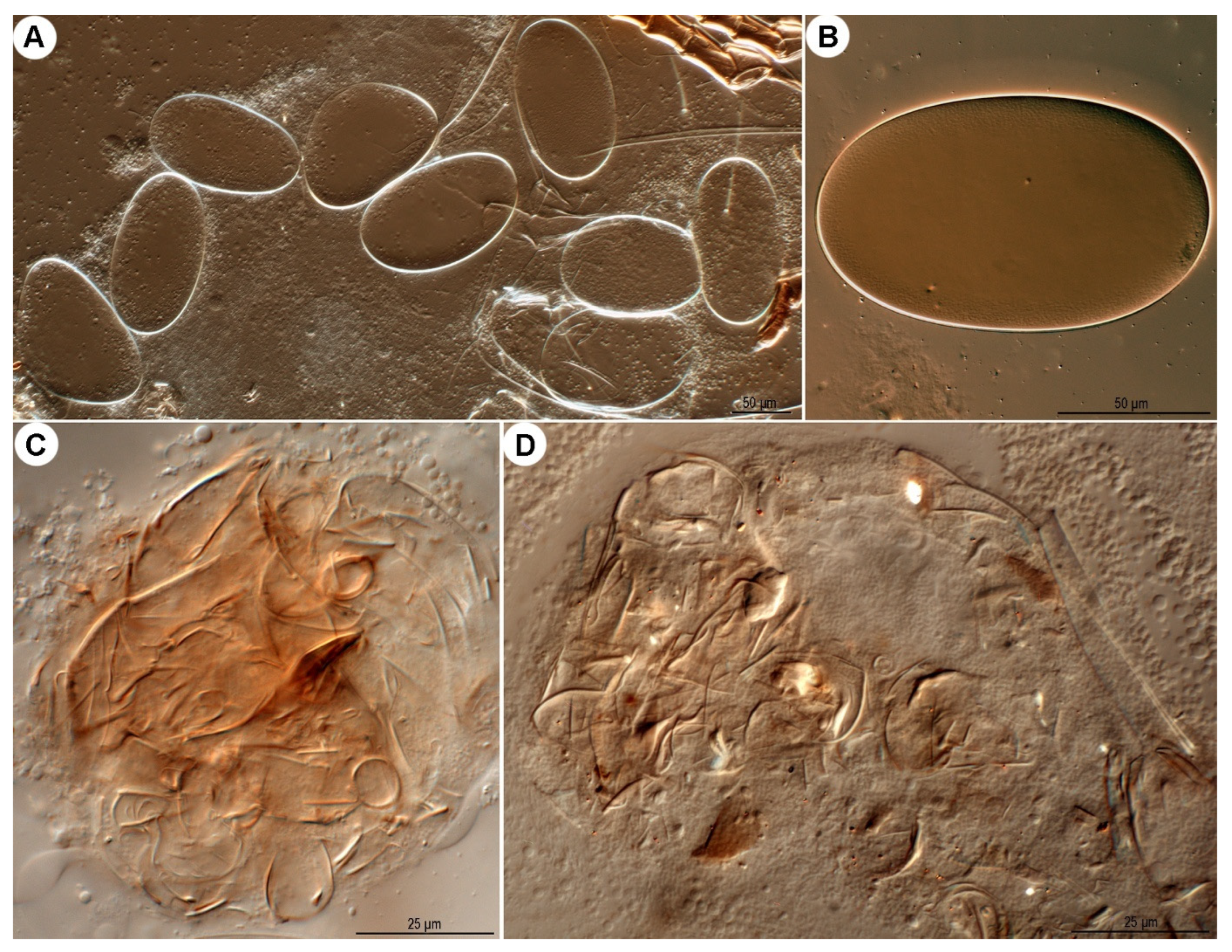
Ovate in form, approximately twice as long (134–169 μm) as wide (78–99 μm); shell surface smooth, lacking distinct ornamentation.
Deutonymph (hypopus) unknown.
Figure 27.
Neosuidasia sjorsvandenbergi sp. nov. (A,B). Eggs; (C,D). food boli.
Figure 27.
Neosuidasia sjorsvandenbergi sp. nov. (A,B). Eggs; (C,D). food boli.
Etymology
The new species N. sjorsvandenbergi is named in honour of Mr. Sjors van den Berg, (Quabio, the Netherlands) the provider of the type specimens.
Biology
Fragmented mite body parts and other unidentified materials were found in the food boli (
Figure 27C,D), suggesting that this new species is likely detritivorous. The mite fragments are likely derived from shed exoskeletons of individuals in the mite population.
Remarks
The new species is morphologically similar to N. faini in the ornamentation of the idiosomal cuticle, setal proportions, and leg setation. However, it is readily distinguishable from N. faini by the following key traits.
Key to species of Neosuidasia
- -
Adult female: pseudanal seta ps2 approximately twice as long as adanal seta ad3. Adult male: ps2 more than 4× length of ps3. Genu I with σ′ 1.2–1.6× length of σ″ in female, 1.5–1.9× in male. …………………… N. sjorsvandenbergi Fan & Faraji, sp. nov.