Functional Investigation of the Receptor to the Major Pheromone Component in the C-Strain and the R-Strain of the Fall Armyworm Spodoptera frugiperda
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
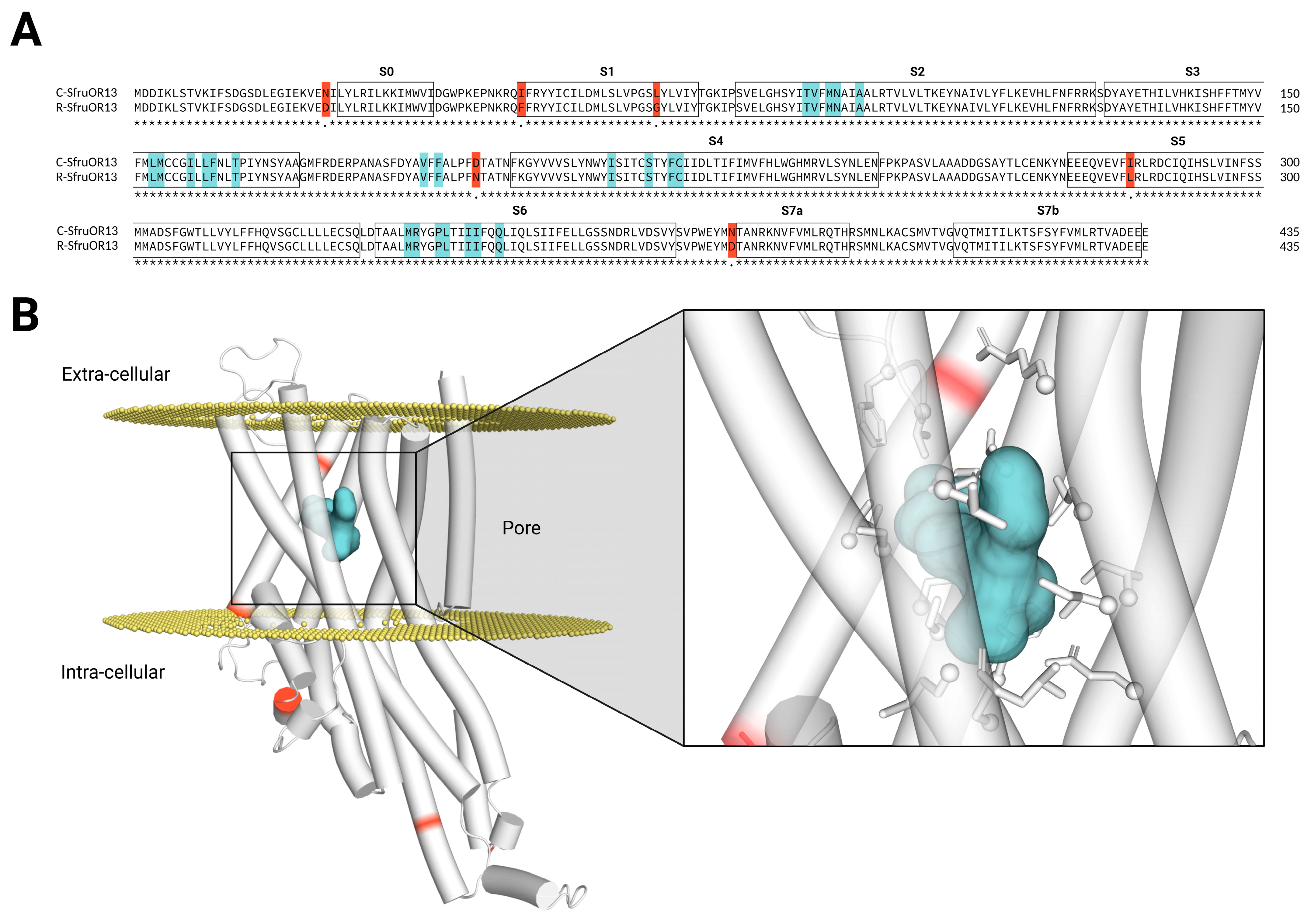
2.1. Comparative Sequence Alignment and Structural Mutation Mapping of C- and R-Strain SfruOR13
2.2. Heterologous Expression of C- and R-Strain SfruOR13 in Drosophila melanogaster
2.3. Single-Sensillum Recordings and Pheromone Compounds
3. Results
3.1. The Binding Regions of SfruOR13s Are Conserved Between the C- and the R-Strains
3.2. C- and R-SfruOR13 Exhibit the Same Specificity and Sensitivity to Pheromone Compounds
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jacquin-Joly, E.; Groot, A.T. Pheromones, Insects. In Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2024. [Google Scholar]
- De Bruyne, M.; Baker, T.C. Odor detection in insects: Volatile codes. J. Chem. Ecol. 2008, 34, 882–897. [Google Scholar] [CrossRef] [PubMed]
- Kaissling, K.-E. Peripheral mechanisms of pheromone reception in moths. Chem. Senses 1996, 21, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Montagné, N.; Wanner, K.; Jacquin-Joly, E. Olfactory genomics within the Lepidoptera. In Insect Pheromone, Biochemistry and Molecular Biology; Blomquist, G., Vogt, R.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Larsson, M.C.; Domingos, A.I.; Jones, W.D.; Chiappe, M.E.; Amrein, H.; Vosshall, L.B. Or83b encodes a broadly expressed odorant receptor essential for Drosophila olfaction. Neuron 2004, 43, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Qiu, L.; Wang, B.; Guan, Z.; Dong, Z.; Zhang, J.; Cao, S.; Yang, L.; Wang, B.; Gong, Z.; et al. Structural basis for odorant recognition of the insect odorant receptor OR-Orco heterocomplex. Science 2024, 384, 1453–1460. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, A.Q.; Ryu, J.; Del Marmol, J. Structural basis of odor sensing by insect heteromeric odorant receptors. Science 2024, 384, 1460–1467. [Google Scholar] [CrossRef]
- Zhang, D.D.; Löfstedt, C. Moth pheromone receptors: Gene sequences, fonction, and evolution. Front. Ecol. Evol. 2015, 3, 105. [Google Scholar] [CrossRef]
- Guo, J.M.; Liu, X.L.; Liu, S.R.; Wei, Z.Q.; Han, W.K.; Guo, Y.; Dong, S.L. Functional Characterization of Sex Pheromone Receptors in the Fall Armyworm (Spodoptera frugiperda). Insects 2020, 11, 193. [Google Scholar] [CrossRef]
- Guo, H.; Gong, X.L.; Li, G.C.; Mo, B.T.; Jiang, N.J.; Huang, L.Q.; Wang, C.Z. Functional analysis of pheromone receptor repertoire in the fall armyworm, Spodoptera frugiperda. Pest Manag. Sci. 2022, 78, 2052–2064. [Google Scholar] [CrossRef]
- Zhang, S.; Jacquin-Joly, E.; Montagne, N.; Liu, F.; Liu, Y.; Wang, G. Identification of an odorant receptor responding to sex pheromones in Spodoptera frugiperda extends the novel type-I PR lineage in moths. Insect Sci. 2024, 31, 489–502. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, F.; Yang, B.; Liu, Y.; Wang, G.R. Functional characterization of sex pheromone receptors in Spodoptera frugiperda, S. exigua, and S. litura moths. Insect Sci. 2023, 30, 305–320. [Google Scholar] [CrossRef]
- Cean, M.; Taddei, A.; Gottsberger, R.A.; Reisenzein, H.; Georgescu, E.I.V. First report of the fall armyworm Spodoptera frugiperda (J.E. Smith, 1797) in Romania. EPPO Bull. 2024, 54, 212–216. [Google Scholar] [CrossRef]
- Montezano, D.; Specht, A.; Sosa-Gómez, D.; Roque-Specht, V.; Sousa-Silva, J.; Paula-Moraes, S.; Peterson, J.; Hunt, T. Host plants of Spodoptera frugiperda (Lepidoptera: Noctuidae) in the Americas. Afr. Entomol. 2018, 26, 286–300. [Google Scholar] [CrossRef]
- Tay, W.T.; Meagher, R.L., Jr.; Czepak, C.; Groot, A.T. Spodoptera frugiperda: Ecology, Evolution, and Management Options of an Invasive Species. Annu. Rev. Entomol. 2023, 68, 299–317. [Google Scholar] [CrossRef] [PubMed]
- Pashley, D.P. Host-associated genetic differentiation in fall armyworm (Lepidoptera, Noctuidae)—A sibling species complex. Ann. Entomolical Soc. Am. 1986, 79, 898–904. [Google Scholar] [CrossRef]
- Tessnow, A.E.; Nagoshi, R.N.; Meagher, R.L.; Gilligan, T.M.; Sadd, B.M.; Carriere, Y.; Davis, H.N.; Fleischer, S.J.; Richers, K.; Palumbo, J.C.; et al. Genomic patterns of strain-specific genetic structure, linkage, and selection across fall armyworm populations. BMC Genom. 2025, 26, 116. [Google Scholar] [CrossRef]
- Groot, A.T.; Marr, M.; Schofl, G.; Lorenz, S.; Svatos, A.; Heckel, D.G. Host strain specific sex pheromone variation in Spodoptera frugiperda. Front. Zool. 2008, 5, 20. [Google Scholar] [CrossRef]
- Unbehend, M.; Hanniger, S.; Meagher, R.L.; Heckel, D.G.; Groot, A.T. Pheromonal divergence between two strains of Spodoptera frugiperda. J. Chem. Ecol. 2013, 39, 364–376. [Google Scholar] [CrossRef]
- Karpati, Z.; Dekker, T.; Hansson, B.S. Reversed functional topology in the antennal lobe of the male European corn borer. J. Exp. Biol. 2008, 211, 2841–2848. [Google Scholar] [CrossRef]
- Koutroumpa, F.A.; Groot, A.T.; Dekker, T.; Heckel, D.G. Genetic mapping of male pheromone response in the European corn borer identifies candidate genes regulating neurogenesis. Proc. Natl. Acad. Sci. USA 2016, 113, E6401–E6408. [Google Scholar] [CrossRef]
- Malo, E.A.; Cruz-Lopez, L.; Valle-Mora, J.; Virgen, A.; Sanchez, J.A.; Rojas, J.C. Evaluation of Commercial Pheromone Lures and Traps for Monitoring Male Fall Armyworm (Lepidoptera: Noctuidae) in the Coastal Region of Chiapas, Mexico. Fla. Entomol. 2001, 84, 659–664. [Google Scholar] [CrossRef]
- Andrade, R.; Rodriguez, C.; Oehlschlager, A.C. Optimization of a Pheromone Lure for Spodoptera frugiperda (Smith) in Central America. J. Braz. Chem. Soc. 2000, 11, 609–613. [Google Scholar] [CrossRef]
- Mitchell, E.R.; McLaughlin, J.R. Suppression of mating and oviposition by fall armyworm (Lepidoptera, Noctuidae) and mating by corn-earworm (Lepidoptera, Noctuidae) in corn, using the air permeation technique. J. Econ. Entomol. 1982, 75, 270–274. [Google Scholar] [CrossRef]
- Unbehend, M.; Hanniger, S.; Vasquez, G.M.; Juarez, M.L.; Reisig, D.; McNeil, J.N.; Meagher, R.L.; Jenkins, D.A.; Heckel, D.G.; Groot, A.T. Geographic variation in sexual attraction of Spodoptera frugiperda corn- and rice-strain males to pheromone lures. PLoS ONE 2014, 9, e89255. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, S.; Guo, M.B.; Hou, X.Q.; Yang, B.; Wang, G.R. Optimization of a pheromone lure by analyzing the peripheral coding of sex pheromones of Spodoptera frugiperda in China. Pest Manag. Sci. 2022, 78, 2995–3004. [Google Scholar] [CrossRef]
- Leary, G.P.; Allen, J.E.; Bunger, P.L.; Luginbill, J.B.; Linn, C.E., Jr.; Macallister, I.E.; Kavanaugh, M.P.; Wanner, K.W. Single mutation to a sex pheromone receptor provides adaptive specificity between closely related moth species. Proc. Natl. Acad. Sci. USA 2012, 109, 14081–14086. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Huang, L.Q.; Ning, C.; Wang, C.Z. Two single-point mutations shift the ligand selectivity of a pheromone receptor between two closely related moth species. Elife 2017, 6, e29100. [Google Scholar] [CrossRef]
- Gouin, A.; Bretaudeau, A.; Nam, K.; Gimenez, S.; Aury, J.M.; Duvic, B.; Hilliou, F.; Durand, N.; Montagne, N.; Darboux, I.; et al. Two genomes of highly polyphagous lepidopteran pests (Spodoptera frugiperda, Noctuidae) with different host-plant ranges. Sci. Rep. 2017, 7, 11816. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform. 2019, 20, 1160–1166. [Google Scholar] [CrossRef]
- Lomize, A.L.; Todd, S.C.; Pogozheva, I.D. Spatial arrangement of proteins in planar and curved membranes by PPM 3.0. Protein Sci 2022, 31, 209–220. [Google Scholar] [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Zidek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- DeLano, W.L. Pymol: An open-source molecular graphics tool. CCP4 Newsl. Protein Crystallogr. 2002, 40, 82–92. [Google Scholar]
- Del Marmol, J.; Yedlin, M.A.; Ruta, V. The structural basis of odorant recognition in insect olfactory receptors. Nature 2021, 597, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Le Guilloux, V.; Schmidtke, P.; Tuffery, P. Fpocket: An open source platform for ligand pocket detection. BMC Bioinform. 2009, 10, 168. [Google Scholar] [CrossRef]
- Kurtovic, A.; Widmer, A.; Dickson, B.J. A single class of olfactory neurons mediates behavioural responses to a Drosophila sex pheromone. Nature 2007, 446, 542–546. [Google Scholar] [CrossRef]
- de Fouchier, A.; Sun, X.; Monsempes, C.; Mirabeau, O.; Jacquin-Joly, E.; Montagné, M. Evolution of two receptors detecting the same pheromone compound in crop pest moths of the genus Spodoptera. Front. Ecol. Evol. 2015, 3, 95. [Google Scholar] [CrossRef]
- Munch, D.; Galizia, C.G. DoOR 2.0—Comprehensive Mapping of Drosophila melanogaster Odorant Responses. Sci. Rep. 2016, 6, 21841. [Google Scholar] [CrossRef]
- Li, Z.; Capoduro, R.; Bastin-Heline, L.; Zhang, S.; Sun, D.; Lucas, P.; Dabir-Moghaddam, D.; Francois, M.C.; Liu, Y.; Wang, G.; et al. A tale of two copies: Evolutionary trajectories of moth pheromone receptors. Proc. Natl. Acad. Sci. USA 2023, 120, e2221166120. [Google Scholar] [CrossRef]
- Akinbuluma, M.D.; van Schaijk, R.A.H.; Roessingh, P.; Groot, A.T. Region-Specific Variation in the Electrophysiological Responses of Spodoptera frugiperda (Lepidoptera: Noctuidae) to Synthetic Sex Pheromone Compounds. J. Chem. Ecol. 2024, 50, 631–642. [Google Scholar] [CrossRef]
- Yainna, S.; Tay, W.T.; Durand, K.; Fiteni, E.; Hilliou, F.; Legeai, F.; Clamens, A.L.; Gimenez, S.; Asokan, R.; Kalleshwaraswamy, C.M.; et al. The evolutionary process of invasion in the fall armyworm (Spodoptera frugiperda). Sci. Rep. 2022, 12, 21063. [Google Scholar] [CrossRef]
- Butterwick, J.A.; Del Marmol, J.; Kim, K.H.; Kahlson, M.A.; Rogow, J.A.; Walz, T.; Ruta, V. Cryo-EM structure of the insect olfactory receptor Orco. Nature 2018, 560, 447–452. [Google Scholar] [CrossRef]
- Benton, R.; Sachse, S.; Michnick, S.W.; Vosshall, L.B. Atypical membrane topology and heteromeric function of Drosophila odorant receptors in vivo. PLoS Biol. 2006, 4, e20. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Zhang, D.D.; Yuvaraj, J.K.; Corcoran, J.A.; Andersson, M.N.; Lofstedt, C. Functional characterization of odorant receptors from the moth Eriocrania semipurpurella: A comparison of results in the Xenopus oocyte and HEK cell systems. Insect Biochem. Mol. Biol. 2020, 117, 103289. [Google Scholar] [CrossRef] [PubMed]
- Yuvaraj, J.K.; Roberts, R.E.; Sonntag, Y.; Hou, X.Q.; Grosse-Wilde, E.; Machara, A.; Zhang, D.D.; Hansson, B.S.; Johanson, U.; Lofstedt, C.; et al. Putative ligand binding sites of two functionally characterized bark beetle odorant receptors. BMC Biol. 2021, 19, 16. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Comte, A.; Delarue, A.; François, M.-C.; Monsempes, C.; Meslin, C.; Montagné, N.; Jacquin-Joly, E. Functional Investigation of the Receptor to the Major Pheromone Component in the C-Strain and the R-Strain of the Fall Armyworm Spodoptera frugiperda. Insects 2025, 16, 304. https://doi.org/10.3390/insects16030304
Comte A, Delarue A, François M-C, Monsempes C, Meslin C, Montagné N, Jacquin-Joly E. Functional Investigation of the Receptor to the Major Pheromone Component in the C-Strain and the R-Strain of the Fall Armyworm Spodoptera frugiperda. Insects. 2025; 16(3):304. https://doi.org/10.3390/insects16030304
Chicago/Turabian StyleComte, Arthur, Alizée Delarue, Marie-Christine François, Christelle Monsempes, Camille Meslin, Nicolas Montagné, and Emmanuelle Jacquin-Joly. 2025. "Functional Investigation of the Receptor to the Major Pheromone Component in the C-Strain and the R-Strain of the Fall Armyworm Spodoptera frugiperda" Insects 16, no. 3: 304. https://doi.org/10.3390/insects16030304
APA StyleComte, A., Delarue, A., François, M.-C., Monsempes, C., Meslin, C., Montagné, N., & Jacquin-Joly, E. (2025). Functional Investigation of the Receptor to the Major Pheromone Component in the C-Strain and the R-Strain of the Fall Armyworm Spodoptera frugiperda. Insects, 16(3), 304. https://doi.org/10.3390/insects16030304








