Impact of the Transboundary Interference Inhibitor on RNAi and the Baculovirus Expression System in Insect Cells
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains, Plasmids, MultiBacmid, Reagents, and Genes
2.2. Construction of Transient Expression Plasmids and Transfection into Sf9 Cells
2.3. Extraction and Reverse Transcription of RNA and Quantitative PCR
2.4. Construction of Recombinant Plasmids for Early and Late Stable Expression of VSRs
2.5. Construction of Recombinant MultiBac and Generation of Recombinant Baculovirus
2.6. The Expression of Target Genes Was Determined Using Western Blotting
2.7. Flow Cytometry to Determine Apoptosis in Insect Sf9 Cells
2.8. Evaluation of the Recombinant Baculovirus Titers Using TCID50
2.9. Statistical Analysis
3. Results
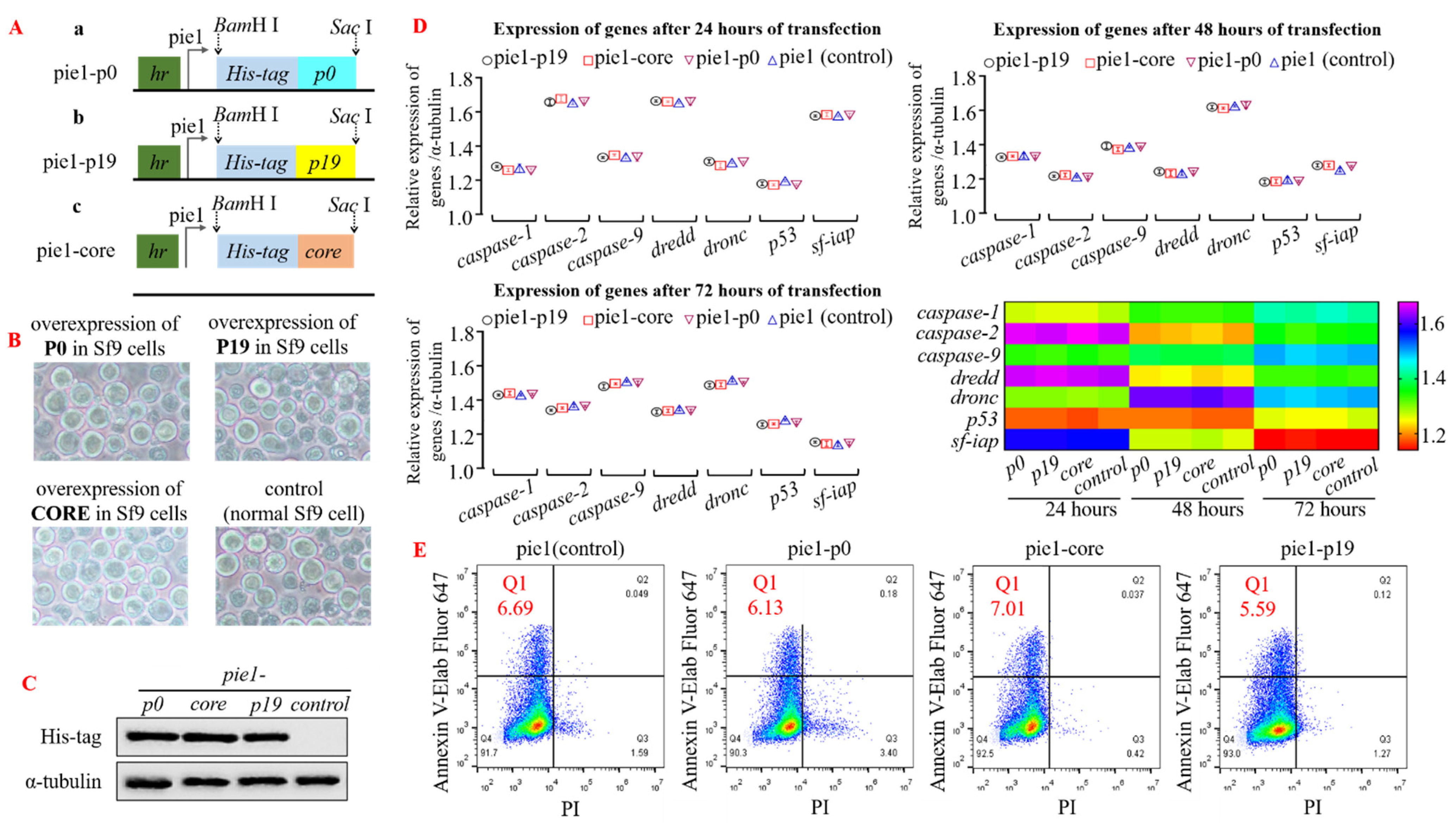
3.1. The Normal Cellular Functions Are Maintained in Insect Cells That Overexpress VSRs
3.2. The Presence of VSRs Markedly Attenuated the Impact of Intracellular RNA Interference
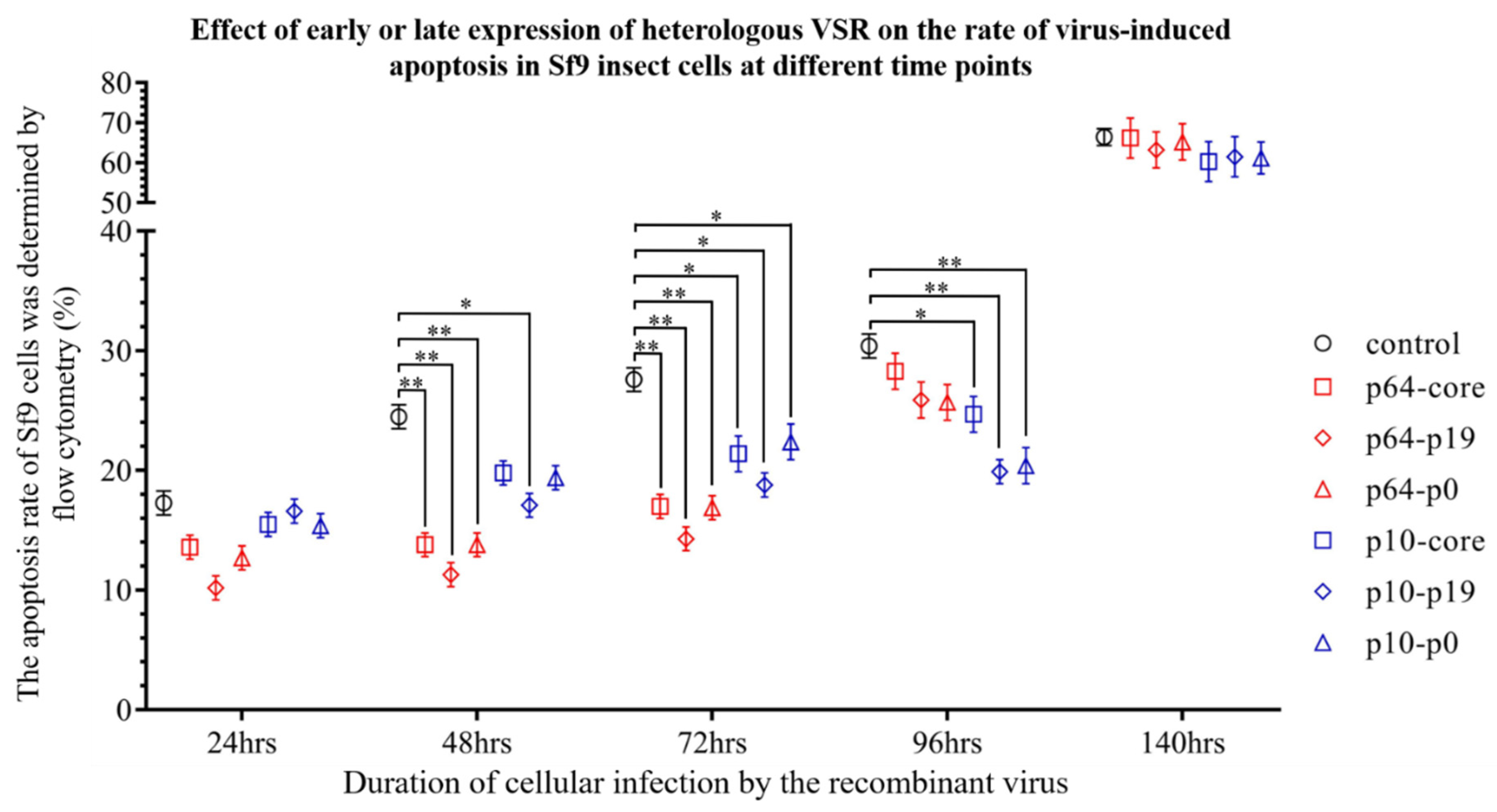
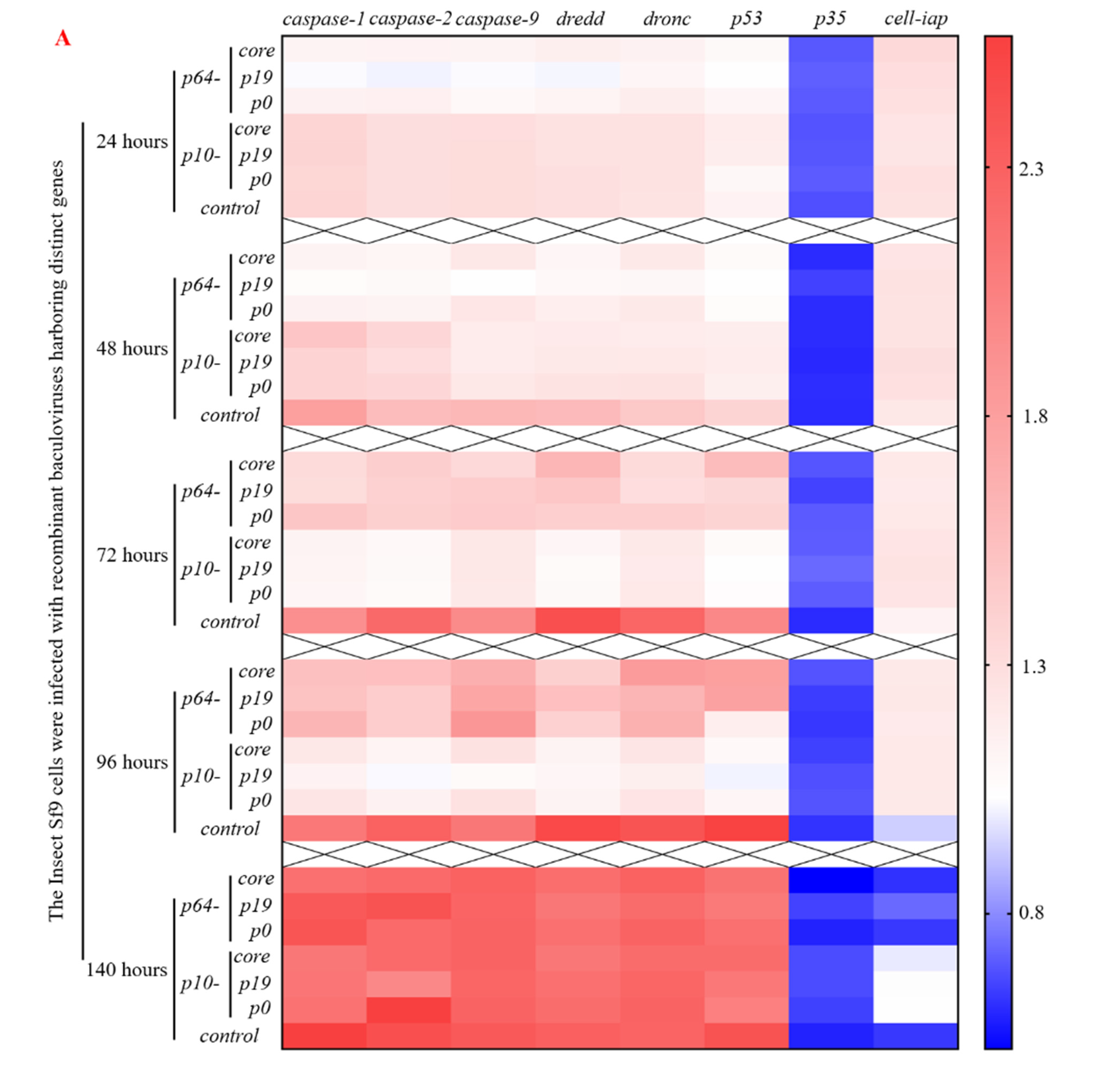
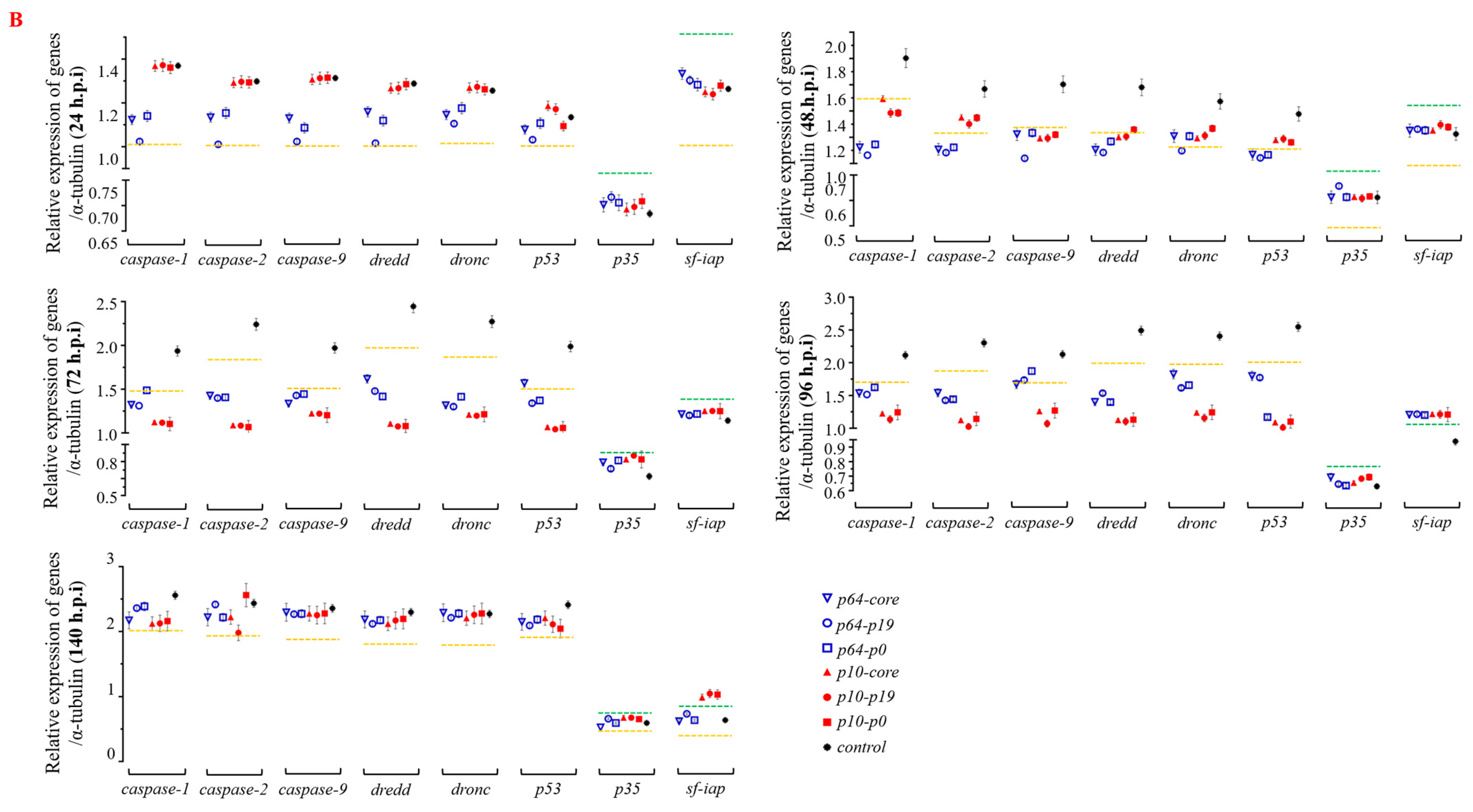
3.3. The Presence of VSR Significantly Enhanced the Expression Efficiency of the Recombinant Protein in the MultiBac System
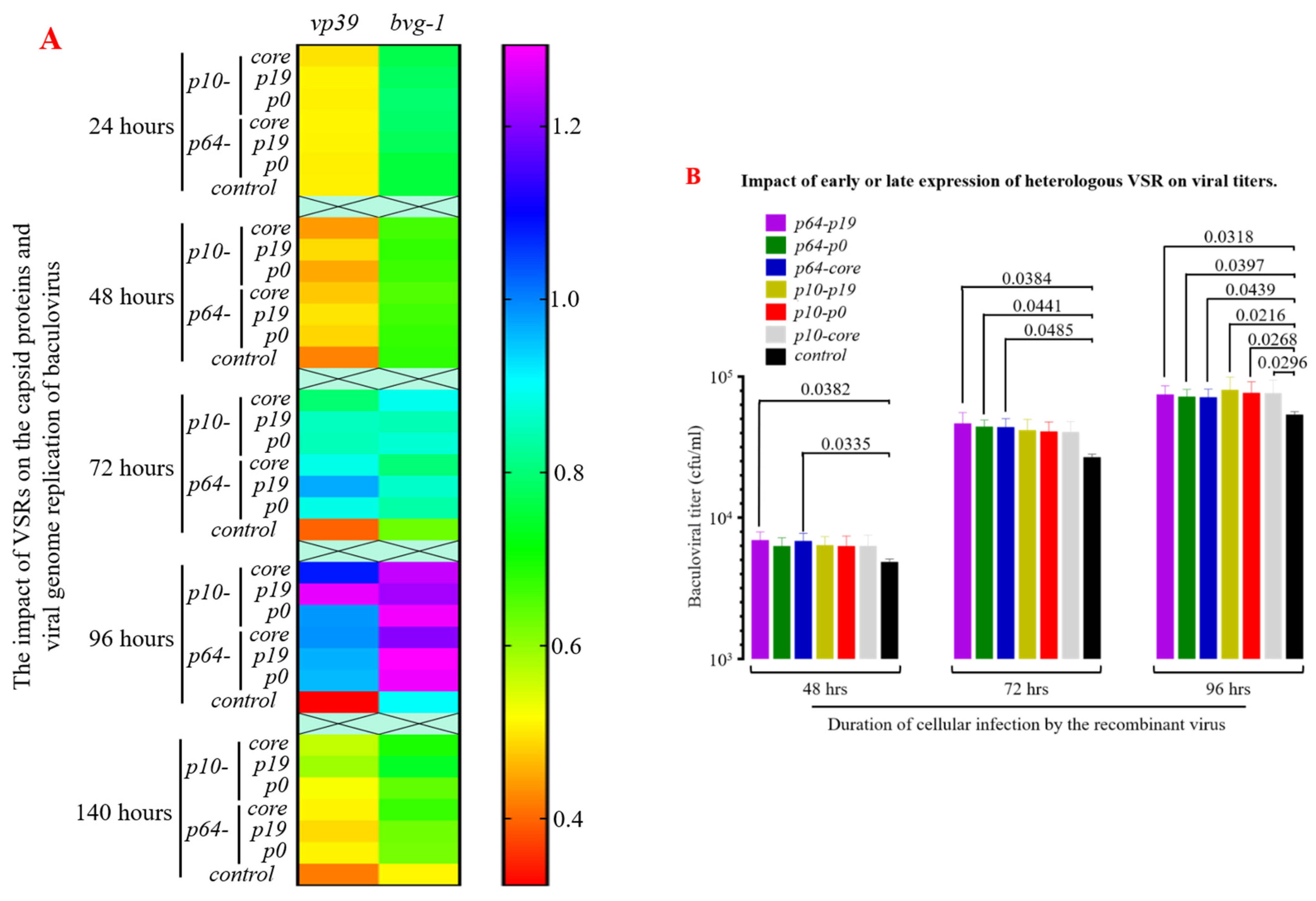
3.4. The Presence of VSRs Enhance the Production of Recombinant Viruses with Higher Titers in the MultiBac System
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Niu, J.Z.; Chen, R.Y.; Wang, J.J. RNA interference in insects: The link between antiviral defense and pest control. Insect Sci. 2023, 3, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Ha, M.; Kim, V.N. Regulation of microRNA biogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.Z.; Taning, C.N.T.; Christiaens, O.; Smagghe, G.; Wang, J.J. Rethink RNAi in insect pest control: Challenges and perspectives. Adv. Insect Physiol. 2018, 55, 1–17. [Google Scholar]
- Jain, R.G.; Robinson, K.E.; Fletcher, S.J.; Mitter, N. RNAi-based functional genomics in hemiptera. Insects 2022, 11, 557. [Google Scholar] [CrossRef] [PubMed]
- Hammond, S.M. An overview of microRNAs. Adv. Drug Deliv. Rev. 2015, 87, 3–14. [Google Scholar] [CrossRef]
- Zhang, X.; Rossmann, M.G. Arbovirus-mosquito interactions: RNAi pathway Reply. Curr. Opin. Virol. 2015, 15, 127–128. [Google Scholar] [CrossRef] [PubMed]
- Fire, A.; Xu, S.Q.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E.; Mello, C.C. Potent and specific genetic interference by double-stranded RNA in. Nature 1998, 391, 806–811. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, K.T.; Li, L.; Chu, Y.; Janowski, B.A.; Corey, D.R. RNAi factors are present and active in human cell nuclei. Cell Rep. 2014, 6, 211–221. [Google Scholar] [CrossRef]
- Holoch, D.; Moazed, D. RNA-mediated epigenetic regulation of gene expression. Nat. Rev. Genet. 2015, 16, 71–84. [Google Scholar] [CrossRef]
- Piombo, E.; Vetukuri, R.R.; Broberg, A.; Kalyandurg, P.B.; Kushwaha, S.; Jensen, D.F.; Karlsson, M.; Dubey, M. The role of Dicer-dependent RNA interference in regulating cross-species communication during fungus-fungus interactions. Microbiol. Spectr. 2021, 9, e01099-21. [Google Scholar] [CrossRef]
- Li, B.B.; Fan, J.Q.; Lu, K.C.; Chen, G.L.; Chen, Y.H. Identification and functional characterization of a systemic RNA interference defective 1 gene in Litopenaeus vannamei. Fish Shellfish Immunol. Rep. 2021, 2, 100033. [Google Scholar] [CrossRef] [PubMed]
- Darrington, M.; Dalmay, T.; Morrison, N.I.; Chapman, T. Implementing the sterile insect technique with RNA interference—A review. Entomol. Exp. Appl. 2017, 164, 155–175. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Li, Y.; Ding, S.W. Small RNA-based antimicrobial immunity. Nat. Rev. Immunol. 2019, 19, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Lamiable, O.; Arnold, J.; Faria, I.; Olmo, R.P.; Bergami, F.; Meignin, C.; Hoffmann, J.A.; Marques, J.T.; Imler, J.L. Analysis of the contribution of hemocytes and autophagy to Drosophila antiviral immunity. J. Virol. 2016, 90, 5415–5426. [Google Scholar] [CrossRef] [PubMed]
- Tassetto, M.; Kunitomi, M.; Andino, R. Circulating immune cells mediate a systemic RNAi-based adaptive antiviral response in Drosophila. Cell 2017, 169, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Nandety, R.S.; Kuo, Y.W.; Nouri, S.; Falk, B.W. Emerging strategies for RNA interference (RNAi) applications in insects. Bioengineered 2015, 6, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xu, W.; Gao, X.; Li, W.; Qi, S.; Guo, D.; Ajayi, O.E.; Ding, S.W.; Wu, Q. lncRNA sensing of a viral suppressor of RNAi activates non-canonical innate immune signaling in drosophila. Cell Host Microbe 2020, 27, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.S.; Swevers, L.; Kolliopoulou, A.; Smagghe, G. Arboviruses and the challenge to establish systemic and persistent infections in competent mosquito vectors: The interaction with the RNAi mechanism. Front. Physiol. 2019, 10, 890. [Google Scholar] [CrossRef]
- Devendran, R.; Prakash, V.; Kumar, R.V. Molecular biology of antiviral arms race between plants and viruses. In Plant Virus-Host Interaction; Academic Press: Cambridge, MA, USA, 2021; Volume 13, pp. 331–358. [Google Scholar]
- Qiu, Y.; Xu, Y.P.; Wang, M.; Miao, M.; Zhou, H.; Xu, J.; Kong, J.; Zheng, D.; Li, R.T.; Zhang, R.R.; et al. Flavivirus induces and antagonizes antiviral RNA interference in both mammals and mosquitoes. Sci. Adv. 2020, 6, eaax7989. [Google Scholar] [CrossRef]
- Chen, L.; Yan, Z.; Xi, Z.; Cheng, Y.; Jiao, Z.; Sun, B.; Zho, T.; Fa, Z. A violaxanthin deepoxidase interacts with a viral suppressor of RNA silencing to inhibit virus amplification. Plant Physiol. 2017, 175, 1774–1794. [Google Scholar] [CrossRef]
- Mehrabadi, M.; Hussain, M.; Matindoost, L.; Asgari, S. The baculovirus antiapoptotic p35 protein functions as an inhibitor of the host RNA interference antiviral response. J. Virol. 2015, 89, 8182–8192. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Lu, R. Characterization of Virus-Encoded RNA Interference Suppressors in Caenorhabditis elegans. J. Virol. 2013, 87, 5414–5423. [Google Scholar] [CrossRef] [PubMed]
- Stav, R.; Hendelman, A.; Buxdorf, K.; Arazi, T. Transgenic expression of tomato bushy stunt virus silencing suppressor P19 via the pOp/LhG4 transactivation system induces viral-like symptoms in tomato. Virus Genes 2010, 40, 119. [Google Scholar] [CrossRef] [PubMed]
- Ye, K.; Malinina, L.; Patel, D.J. Recognition of small interfering RNA by a viral suppressor of RNA silencing. Nature 2003, 426, 874–878. [Google Scholar] [CrossRef] [PubMed]
- Li, W.X.; Li, H.; Lu, R.; Li, F.; Dus, M.; Atkinson, P.; Brydon, E.W.; Johnson, K.L.; García-Sastre, A.; Ball, L.A.; et al. Interferon antagonist proteins of influenza and vaccinia viruses are suppressors of RNA silencing. Proc. Natl. Acad. Sci. USA 2004, 101, 1350–1355. [Google Scholar] [CrossRef]
- Csorba, T.; Lózsa, R.; Hutvágner, G.; Burgyán, J. Polerovirus protein P0 prevents the assembly of small RNA-containing RISC complexes and leads to degradation of ARGONAUTE1. Plant J. 2010, 62, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wu, Q.; Ito, T.; Cillo, F.; Li, W.; Chen, X.; Yu, J.; Ding, S.W. RNAi-mediated viral immunity requires amplification of virus-derived siRNAs in Arabidopsis Thaliana. Proc. Natl. Acad. Sci. USA 2009, 107, 484–489. [Google Scholar] [CrossRef]
- Giannini, C.; Brechot, C. Hepatitis C virus biology. Cell Death Differ. 2003, 10, S27–S38. [Google Scholar] [CrossRef]
- Qian, S.; Zhong, X.; Yu, L.; Ding, B.; de Haan, P.; Boris-Lawrie, K. HIV-1 Tat RNA silencing suppressor activity is conserved across kingdoms and counteracts translational repression of HIV-1. Proc. Natl. Acad. Sci. USA 2009, 106, 605–610. [Google Scholar] [CrossRef]
- Yamada, H.; Kitaguchi, K.; Hamajima, R.; Kobayashi, M.; Ikeda, M. Novel Apoptosis Suppressor Apsup from the Baculovirus Lymantria dispar Multiple Nucleopolyhedrovirus Precludes Apoptosis by Preventing Proteolytic Processing of Initiator Caspase Dronc. J. Virol. 2013, 87, 12925–12934. [Google Scholar] [CrossRef]
- Omarov, R.T.; Bersimbay, R.I. Biological function of tombusvirus-encoded suppressor of RNA silencing in plants. J. Stress Physiol. Biochem. 2012, 8, S3. [Google Scholar]
- Zheng, H.; Pan, Y.; Wang, X.; Tian, W.B.; Yao, L.G.; Sun, J.C. Impact of Molecular Modification on the Efficiency of Recombinant Baculovirus Vector Invasion to Mammalian Cells and Its Immunogenicity in Mice. Viruses 2022, 14, 140. [Google Scholar] [CrossRef] [PubMed]
- Lock, M.; Korn, M.; Wilson, J.; Sena-Esteves, M.; Gao, G. Measuring the Infectious Titer of Recombinant Adenovirus Using Tissue Culture Infection Dose 50% (TCID50) End-Point Dilution and Quantitative Polymerase Chain Reaction (qPCR). Cold Spring Harb. Protoc. 2019, 8, pdb-prot095562. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Han, Y.H.; Li, W.X.; Ding, S.W. Infection Defects of RNA and DNA Viruses Induced by Antiviral RNA Interference. Microbiol. Mol. Biol. Rev. 2023, 87, e00035-22. [Google Scholar] [CrossRef]
- Wang, J.X.; Li, Y. Current advances in antiviral RNA interference in mammals. FEBS J. 2023, 291, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Betting, V.; Van Rij, R.P. Countering Counter-Defense to Antiviral RNAi. Trends Microbiol. 2020, 28, 600–602. [Google Scholar] [CrossRef] [PubMed]
- Karjee, S.; Mukherjee, S.K. RNAi suppressor: The hidden weapon of SARS-CoV. J. Biosci. 2020, 45, 99. [Google Scholar] [CrossRef] [PubMed]
- Simmer, F.; Tijsterman, M.; Parrish, S.; Koushika, S.P.; Nonet, M.L.; Fire, A. Loss of the putative RNA-directed RNA polymerase RRF-3 makes C. elegans hypersensitive to RNAi. Curr. Biol. 2002, 12, 1317–1319. [Google Scholar] [CrossRef] [PubMed]
- Blagovesta, P.; Markus, K.; Andrea, H.; Fredrik, S.; Wolfgang, N. HelF, a putative RNA helicase acts as a nuclear suppressor of RNAi but not antisense mediated gene silencing. Nucleic Acids Res. 2006, 34, 773–784. [Google Scholar]
- Jin, L.Y.; Chen, M.N.; Xiang, M.Q.; Guo, Z.X. RNAi-Based Antiviral Innate Immunity in Plants. Viruses 2022, 14, 432. [Google Scholar] [CrossRef]
- Gómez, B.G.; Rosas-Díaz, T.; Shi, C.N.; Fan, P.F.; Zhang, D.; Rufian, J.S.; Lozano-Durán, R. The viral silencing suppressor P19 interacts with the receptor-like kinases BAM1 and BAM2 and suppresses the cell-to-cell movement of RNA silencing independently of its ability to bind sRNA. New Phytol. 2021, 229, 1840–1843. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Liu, Z.Z.; Qiu, Y.; Kong, J.; Fu, Y.H.; Liu, Y.; Wang, C.; Quan, J.; Wang, Q.; Xu, W.; et al. Inhibition of viral suppressor of RNAi proteins by designer peptides protects from enteroviral infection in vivo. Immunity 2021, 54, 2231–2244. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, E.; Lozano-Durán, R. On the inhibition of RNA silencing movement by the tombusvirus P19 protein: Reliance on sRNA binding and correlation with local silencing suppression. New Phytol. 2022, 235, 830–835. [Google Scholar] [CrossRef] [PubMed]
- Van den Beek, M.; Antoniewski, C.; Carre, C. Isolation of small interfering RNAs using viral suppressors of RNA interference. Methods Mol. Biol. 2014, 1173, 147–155. [Google Scholar] [PubMed]
- Wang, Q.; Wang, J.X.; Xu, Y.; Li, Z.; Wang, B.B.; Li, Y. The Interaction of influenza A NS1 and cellular TRBP protein modulates the function of RNA interference machinery. Front. Microbiol. 2022, 13, 859420. [Google Scholar] [CrossRef]
- Asaoka, R.; Shimura, H.; Arai, M.; Masuta, C. A progeny virus from a cucumovirus pseudorecombinant evolved to gain the ability to accumulate its RNA-silencing suppressor seading to systemic infection in tobacco. Mol. Plant-Microbe Interact. 2010, 23, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Pan, Y.; Awais, M.M.; Tian, W.B.; Li, J.Y.; Sun, J.C. Impact of group II baculovirus IAPs on virus-induced apoptosis in insect cells. Genes 2022, 13, 750. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Gwak, W.S.; Bae, J.S.; Kim, T.H.; Choi, J.B.; Han, B.K.; Woo, S.D. Effects of repeated burst sequences on the p10 promoter activity of Bombyx mori nucleopolyhedrovirus. J. Asia-Pac. Entomol. 2021, 24, 7–13. [Google Scholar] [CrossRef]
- Li, Z.L.; Jiang, Z.H.; Yang, X.X.; Yue, N.; Wang, X.T.; Zhang, K.; Jackson, A.O.; Li, D.W.; Zhang, Y.L. Construction of an Infectious Poa semilatent virus cDNA Clone and Comparisons of Hordeivirus Cytopathology and Pathogenicity. Phytopathology 2020, 110, 215–227. [Google Scholar] [CrossRef]
- Radford, K.M.; Cavegn, C.; Bertrand, M.; Bernard, A.R.; Reid, S.; Greenfield, P.F. The indirect effects of multiplicity of infection on baculovirus expressed proteins in insect cells: Secreted and non-secreted products. Cytotechnology 1997, 24, 73–81. [Google Scholar] [CrossRef]
- Huynh, H.T.; Tran, T.T.B.; Chan, L.C.; Nielsen, L.K.; Reid, S. Decline in baculovirus-expressed recombinant protein production with increasing cell density is strongly correlated to impairment of virus replication and mRNA expression. Appl. Microbiol. Biotechnol. 2013, 97, 5245–5257. [Google Scholar] [CrossRef] [PubMed]
- Bellott, L.; Gilmer, D.; Michel, F. Hit two birds with one stone. the multiple properties of (viral) RNA silencing suppressors. Virologie 2019, 23, E38–E60. [Google Scholar]
- Pertermann, R.; Golbik, R.P.; Tamilarasan, S.; Gursinsky, T.; Gago-Zachert, S.; Pantaleo, V.; Thondorf, I.; Behrens, S.E. RNA and protein determinants mediate differential binding of miRNAs by a viral suppressor of RNA silencing thus modulating antiviral immune responses in plants. Int. J. Mol. Sci. 2022, 23, 4977. [Google Scholar] [CrossRef] [PubMed]
- Papp, I.; Mette, M.F.; Aufsatz, W.; Daxinger, L.; Schauer, S.E.; Ray, A.; van der Winden, J.; Matzke, M.; Matzke, A.J. Evidence for nuclear processing of plant microRNA and short interfering RNA precursors. Plant Physiol. 2003, 132, 1382–1390. [Google Scholar] [CrossRef] [PubMed]
- Qiu, W.; Park, J.W.; Scholthof, H.B. Tombusvirus P19-mediated suppression of virus-induced gene silencing is controlled by genetic and dosage features that influence pathogenicity. Mol. Plant-Microbe Interact. 2002, 15, 269–280. [Google Scholar] [CrossRef]
- Li, Y.; Shen, B.S.; Hu, A.L.; Deng, B.F.; Vlak, A.J.M. The functional oligomeric state of tegument protein GP41 is essential for baculovirus budded virion and occlusion-derived virion assembly. J. Virol. 2018, 92, e02083-17. [Google Scholar] [CrossRef]



| Name | Sequence | Genes |
|---|---|---|
| p0 (with His-tag) | F: 5′-cccgggatgcatcatcaccaccatcacatgattgtattgacccagt R: 5′-gcatgcttattcttgtaattccttt | Potato leafroll virus p0 Gene |
| p19 (with His-tag) | F: 5′-cccgggatgcatcatcaccaccatcacatggaacgagctatacaag R: 5′-gcatgcttactcgctttctttttcg | Tomato bushy stunt virus p19 Gene |
| core (with His-tag) | F: 5′-cccgggatgcatcatcaccaccatcacatgagcacgaatcctaaac R: 5′-gcatgcttaagccgaggcgggaacg | Hepacivirus C core Gene |
| Fluc | F: 5′-ggatccatggaagacgccaaaaacata R: 5′-tctagattacacggcgatctttccg | Firefly-luciferase Gene |
| Rluc | F: 5′-cccgggatgacttcgaaagtttatgat R: 5′-gcatgcttattgttcatttttgagaact | Renilla-luciferase Gene |
| egfp | F: 5′-ggatccatggtgagcaagggcgagga R: 5′-tctagattacttgtacagctcgtccat | Enhanced Green Fluorescent Protein Gene |
| mCherry | F: 5′-cccgggatggtgagcaagggcgagga R: 5′-gctagcttacttgtacagctcgtccatgcc | mCherry Fluorescent Protein Gene |
| Name | Sequence | Genes |
|---|---|---|
| α-Tubulin | F: 5′-agtccagatcggtaatgc R: 5′-gctgaagaaggtgttgaag | Reference Gene |
| bvg-1 | F: 5′-cccgtaacggacctcgtactt R: 5′-ttatcgagatttatttgcatacaac | Baculovirus Genome |
| Caspase-1 | F: 5′-gattcaaagttacggtgttcccta R: 5′-ggttgtctggcttgtaatgagtat | Cysteine Aspartate-specific Gene |
| Caspase-2 | F: 5′-gtaaggttctgattggcaattagc R: 5′-cggtacttgtggttggtgtt | Cysteine Aspartate-specific Gene |
| Caspase-9 | F: 5′-acacagagtttgacaacaatatcg R: 5′-ggtctcatagtccaccaacac | Cysteine Aspartate-specific Gene |
| Dronc | F: 5′-ctggtagatacgcttggagaacta R: 5′-gcctgtttgatgtgctaagact | Apoptosis Gene |
| Dredd | F: 5′-aacaccacaaggaatggaagt R: 5′-agttacaggcatcgttggaa | Apoptosis Gene |
| Sf9-iap | F: 5′-gttggagagttgtgttgtttgttt R: 5′-aatagcgttaatgttgaggaggag | Inhibitor of Apoptosis Gene |
| vp39 | F: 5′-acccgataagaagcagtgac R: 5′-cccagagtagcgttaggatt | Baculovirus Capsid Gene |
| p35 | F: 5′-cgaacgcaacgactactac R: 5′-tgagcaaacggcacaataac | Apoptosis Gene |
| p53 | F: 5′-caccgtctcaaccgtatc R: 5′-gaggacattcttcgctattt | Apoptosis Gene |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, H.; Zhao, H.; Xiong, H.; Awais, M.M.; Zeng, S.; Sun, J. Impact of the Transboundary Interference Inhibitor on RNAi and the Baculovirus Expression System in Insect Cells. Insects 2024, 15, 375. https://doi.org/10.3390/insects15060375
Zheng H, Zhao H, Xiong H, Awais MM, Zeng S, Sun J. Impact of the Transboundary Interference Inhibitor on RNAi and the Baculovirus Expression System in Insect Cells. Insects. 2024; 15(6):375. https://doi.org/10.3390/insects15060375
Chicago/Turabian StyleZheng, Hao, Hengfeng Zhao, Haifan Xiong, Mian Muhammad Awais, Songrong Zeng, and Jingchen Sun. 2024. "Impact of the Transboundary Interference Inhibitor on RNAi and the Baculovirus Expression System in Insect Cells" Insects 15, no. 6: 375. https://doi.org/10.3390/insects15060375
APA StyleZheng, H., Zhao, H., Xiong, H., Awais, M. M., Zeng, S., & Sun, J. (2024). Impact of the Transboundary Interference Inhibitor on RNAi and the Baculovirus Expression System in Insect Cells. Insects, 15(6), 375. https://doi.org/10.3390/insects15060375





