Global Trends in Climate Suitability of Bees: Ups and Downs in a Warming World
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Occurrence Data
2.2. Data Cleaning
2.3. Environmental Variables
2.4. Model Fitting
2.5. Model Assessment
3. Results
3.1. Data Summary
3.2. Model Assessment
3.3. Potential Effects of Climate Change
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Porto, R.G.; de Almeida, R.F.; Cruz-Neto, O.; Tabarelli, M.; Viana, B.F.; Peres, C.A.; Lopes, A.V. Pollination ecosystem services: A comprehensive review of economic values, research funding and policy actions. Food Secur. 2020, 12, 1425–1442. [Google Scholar] [CrossRef]
- Eilers, E.J.; Kremen, C.; Smith Greenleaf, S.; Garber, A.K.; Klein, A.-M. Contribution of pollinator-mediated crops to nutrients in the human food supply. PLoS ONE 2011, 6, e21363. [Google Scholar] [CrossRef] [PubMed]
- Gallai, N.; Salles, J.-M.; Settele, J.; Vaissière, B.E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 2009, 68, 810–821. [Google Scholar] [CrossRef]
- Murphy, J.T.; Breeze, T.D.; Willcox, B.; Kavanagh, S.; Stout, J.C. Globalisation and pollinators: Pollinator declines are an economic threat to global food systems. People Nat. 2022, 4, 773–785. [Google Scholar] [CrossRef]
- Porto, R.G.; Cruz-Neto, O.; Tabarelli, M.; Viana, B.F.; Peres, C.A.; Lopes, A.V. Pollinator-dependent crops in Brazil yield nearly half of nutrients for humans and livestock feed. Glob. Food Secur. 2021, 31, 100587. [Google Scholar] [CrossRef]
- Jaworski, T.; Hilszczański, J. The effect of temperature and humidity changes on insects development their impact on forest ecosystems in the context of expected climate change. For. Res. Pap. 2013, 74, 345–355. [Google Scholar]
- Bale, J.S.; Masters, G.J.; Hodkinson, I.D.; Awmack, C.; Bezemer, T.M.; Brown, V.K.; Butterfield, J.; Buse, A.; Coulson, J.C.; Farrar, J. Herbivory in global climate change research: Direct effects of rising temperature on insect herbivores. Glob. Chang. Biol. 2002, 8, 1–16. [Google Scholar] [CrossRef]
- Allen, M.; Antwi-Agyei, P.; Aragon-Durand, F.; Babiker, M.; Bertoldi, P.; Bind, M.; Brown, S.; Buckeridge, M.; Camilloni, I.; Cartwright, A. Technical Summary: Global Warming of 1.5 °C. An IPCC Special Report on the Impacts of Global Warming of 1.5 °C above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty. 2019. Available online: https://www.ipcc.ch/site/assets/uploads/sites/2/2018/12/SR15_TS_High_Res.pdf (accessed on 20 October 2023).
- Rahimi, E.; Jung, C. Plant–pollinator metanetworks in fragmented landscapes: A simulation study. Ecol. Process. 2023, 12, 29. [Google Scholar] [CrossRef]
- Rafferty, N.E. Effects of global change on insect pollinators: Multiple drivers lead to novel communities. Curr. Opin. Insect Sci. 2017, 23, 22–27. [Google Scholar] [CrossRef]
- Schweiger, O.; Biesmeijer, J.C.; Bommarco, R.; Hickler, T.; Hulme, P.E.; Klotz, S.; Kühn, I.; Moora, M.; Nielsen, A.; Ohlemüller, R. Multiple stressors on biotic interactions: How climate change and alien species interact to affect pollination. Biol. Rev. 2010, 85, 777–795. [Google Scholar] [CrossRef]
- Hegland, S.J.; Nielsen, A.; Lázaro, A.; Bjerknes, A.L.; Totland, Ø. How does climate warming affect plant-pollinator interactions? Ecol. Lett. 2009, 12, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, E.; Barghjelveh, S.; Dong, P. Estimating potential range shift of some wild bees in response to climate change scenarios in northwestern regions of Iran. J. Ecol. Environ. 2021, 45, 14. [Google Scholar] [CrossRef]
- Rasmont, P.; Franzén, M.; Lecocq, T.; Harpke, A.; Roberts, S.P.; Biesmeijer, J.C.; Castro, L.; Cederberg, B.; Dvorak, L.; Fitzpatrick, Ú. Climatic Risk and Distribution Atlas of European Bumblebees; Pensoft Publishers: Sofia, Bulgaria, 2015; Volume 10. [Google Scholar]
- Suzuki-Ohno, Y.; Yokoyama, J.; Nakashizuka, T.; Kawata, M. Estimating possible bumblebee range shifts in response to climate and land cover changes. Sci. Rep. 2020, 10, 19622. [Google Scholar] [CrossRef] [PubMed]
- Tylianakis, J.M.; Didham, R.K.; Bascompte, J.; Wardle, D.A. Global change and species interactions in terrestrial ecosystems. Ecol. Lett. 2008, 11, 1351–1363. [Google Scholar] [CrossRef]
- Gillooly, J.F.; Brown, J.H.; West, G.B.; Savage, V.M.; Charnov, E.L. Effects of size and temperature on metabolic rate. Science 2001, 293, 2248–2251. [Google Scholar] [CrossRef] [PubMed]
- Elias, M.A.; Borges, F.J.; Bergamini, L.L.; Franceschinelli, E.V.; Sujii, E.R. Climate change threatens pollination services in tomato crops in Brazil. Agric. Ecosyst. Environ. 2017, 239, 257–264. [Google Scholar] [CrossRef]
- Doyle, T.; Hawkes, W.L.; Massy, R.; Powney, G.D.; Menz, M.H.; Wotton, K.R. Pollination by hoverflies in the Anthropocene. Proc. R. Soc. B 2020, 287, 20200508. [Google Scholar] [CrossRef] [PubMed]
- Dorey, J.B.; Fischer, E.E.; Chesshire, P.R.; Nava-Bolaños, A.; O’Reilly, R.L.; Bossert, S.; Collins, S.M.; Lichtenberg, E.M.; Tucker, E.M.; Smith-Pardo, A. A globally synthesised and flagged bee occurrence dataset and cleaning workflow. Sci. Data 2023, 10, 747. [Google Scholar] [CrossRef]
- Brown, J.L.; Carnaval, A.C. A tale of two niches: Methods, concepts, and evolution. Front. Biogeogr. 2019, 11, 1–25. [Google Scholar] [CrossRef]
- de Andrade, A.F.A.; Velazco, S.J.E.; Júnior, P.D.M. ENMTML: An R package for a straightforward construction of complex ecological niche models. Environ. Model. Softw. 2020, 125, 104615. [Google Scholar] [CrossRef]
- Velazco, S.J.E.; Rose, M.B.; de Andrade, A.F.A.; Minoli, I.; Franklin, J. flexsdm: An R package for supporting a comprehensive and flexible species distribution modelling workflow. Methods Ecol. Evol. 2022, 13, 1661–1669. [Google Scholar] [CrossRef]
- Bean, W.T.; Stafford, R.; Brashares, J.S. The effects of small sample size and sample bias on threshold selection and accuracy assessment of species distribution models. Ecography 2012, 35, 250–258. [Google Scholar] [CrossRef]
- Stockwell, D.R.; Peterson, A.T. Effects of sample size on accuracy of species distribution models. Ecol. Model. 2002, 148, 1–13. [Google Scholar] [CrossRef]
- Williams, J.N.; Seo, C.; Thorne, J.; Nelson, J.K.; Erwin, S.; O’Brien, J.M.; Schwartz, M.W. Using species distribution models to predict new occurrences for rare plants. Divers. Distrib. 2009, 15, 565–576. [Google Scholar] [CrossRef]
- Orr, M.C.; Hughes, A.C.; Chesters, D.; Pickering, J.; Zhu, C.-D.; Ascher, J.S. Global patterns and drivers of bee distribution. Curr. Biol. 2021, 31, 451–458.e4. [Google Scholar] [CrossRef]
- Paaijmans, K.P.; Heinig, R.L.; Seliga, R.A.; Blanford, J.I.; Blanford, S.; Murdock, C.C.; Thomas, M.B. Temperature variation makes ectotherms more sensitive to climate change. Glob. Chang. Biol. 2013, 19, 2373–2380. [Google Scholar] [CrossRef]
- Naimi, B. Package ‘usdm’. Uncertainty Analysis for Species Distribution Models. Available online: https://cran.r-project.org/ (accessed on 20 October 2023).
- Filazzola, A.; Matter, S.F.; Roland, J. Inclusion of trophic interactions increases the vulnerability of an alpine butterfly species to climate change. Glob. Chang. Biol. 2020, 26, 2867–2877. [Google Scholar] [CrossRef]
- Miličić, M.S.; Janković, M.A.; Milić, D.M.; Radenković, S.R.; Vujić, A.A. Strictly protected species of hoverflies (Diptera: Syrphidae) in Serbia in the face of climate change. Zb. Matice Srp. Prir. Nauk. 2018, 135, 53–62. [Google Scholar] [CrossRef]
- Aguirre-Gutiérrez, J.; Carvalheiro, L.G.; Polce, C.; van Loon, E.E.; Raes, N.; Reemer, M.; Biesmeijer, J.C. Fit-for-purpose: Species distribution model performance depends on evaluation criteria–Dutch hoverflies as a case study. PLoS ONE 2013, 8, e63708. [Google Scholar] [CrossRef]
- Tabor, J.A.; Koch, J.B. Ensemble models predict invasive bee habitat suitability will expand under future climate scenarios in Hawai’i. Insects 2021, 12, 443. [Google Scholar] [CrossRef]
- Martínez-López, O.; Koch, J.B.; Martínez-Morales, M.A.; Navarrete-Gutiérrez, D.; Enríquez, E.; Vandame, R. Reduction in the potential distribution of bumble bees (Apidae: Bombus) in Mesoamerica under different climate change scenarios: Conservation implications. Glob. Chang. Biol. 2021, 27, 1772–1787. [Google Scholar] [CrossRef]
- Abrol, D.P. Defensive behaviour of Apis cerana F. against predatory wasps. J. Apic. Sci. 2006, 50, 39. [Google Scholar]
- O’Neill, B.C.; Tebaldi, C.; Van Vuuren, D.P.; Eyring, V.; Friedlingstein, P.; Hurtt, G.; Knutti, R.; Kriegler, E.; Lamarque, J.-F.; Lowe, J. The scenario model intercomparison project (ScenarioMIP) for CMIP6. Geosci. Model Dev. 2016, 9, 3461–3482. [Google Scholar] [CrossRef]
- Wang, R.; Li, Q.; He, S.; Liu, Y.; Wang, M.; Jiang, G. Modeling and mapping the current and future distribution of Pseudomonas syringae pv. actinidiae under climate change in China. PLoS ONE 2018, 13, e0192153. [Google Scholar] [CrossRef]
- Fielding, A.H.; Bell, J.F. A review of methods for the assessment of prediction errors in conservation presence/absence models. Environ. Conserv. 1997, 24, 38–49. [Google Scholar] [CrossRef]
- Hung, K.-L.J.; Kingston, J.M.; Albrecht, M.; Holway, D.A.; Kohn, J.R. The worldwide importance of honey bees as pollinators in natural habitats. Proc. R. Soc. B Biol. Sci. 2018, 285, 20172140. [Google Scholar] [CrossRef] [PubMed]
- Badenes-Pérez, F.R. Benefits of insect pollination in Brassicaceae: A meta-analysis of self-compatible and self-Incompatible crop species. Agriculture 2022, 12, 446. [Google Scholar] [CrossRef]
- Heinrich, B.; Esch, H. Thermoregulation in bees. Am. Sci. 1994, 82, 164–170. [Google Scholar]
- Scaven, V.L.; Rafferty, N.E. Physiological effects of climate warming on flowering plants and insect pollinators and potential consequences for their interactions. Curr. Zool. 2013, 59, 418–426. [Google Scholar] [CrossRef]
- Saunders, M.E.; Kendall, L.K.; Lanuza, J.B.; Hall, M.A.; Rader, R.; Stavert, J.R. Climate mediates roles of pollinator species in plant–pollinator networks. Glob. Ecol. Biogeogr. 2023, 32, 511–518. [Google Scholar] [CrossRef]
- Miller-Struttmann, N.E.; Geib, J.C.; Franklin, J.D.; Kevan, P.G.; Holdo, R.M.; Ebert-May, D.; Lynn, A.M.; Kettenbach, J.A.; Hedrick, E.; Galen, C. Functional mismatch in a bumble bee pollination mutualism under climate change. Science 2015, 349, 1541–1544. [Google Scholar] [CrossRef]
- Sponsler, D.; Iverson, A.; Steffan-Dewenter, I. Pollinator competition and the structure of floral resources. Ecography 2023, 2023, e06651. [Google Scholar] [CrossRef]
- Le Conte, Y.; Navajas, M. Climate change: Impact on honey bee populations and diseases. Rev. Sci. Tech. Off. Int. Epizoot. 2008, 27, 499–510. [Google Scholar]
- Halsch, C.A.; Shapiro, A.M.; Fordyce, J.A.; Nice, C.C.; Thorne, J.H.; Waetjen, D.P.; Forister, M.L. Insects and recent climate change. Proc. Natl. Acad. Sci. USA 2021, 118, e2002543117. [Google Scholar] [CrossRef]
- Lima, V.P.; Marchioro, C.A. Brazilian stingless bees are threatened by habitat conversion and climate change. Reg. Environ. Chang. 2021, 21, 14. [Google Scholar] [CrossRef]
- Dew, R.M.; Silva, D.P.; Rehan, S.M. Range expansion of an already widespread bee under climate change. Glob. Ecol. Conserv. 2019, 17, e00584. [Google Scholar] [CrossRef]
- Giannini, T.C.; Costa, W.F.; Borges, R.C.; Miranda, L.; da Costa, C.P.W.; Saraiva, A.M.; Fonseca, V.L.I. Climate change in the Eastern Amazon: Crop-pollinator and occurrence-restricted bees are potentially more affected. Reg. Environ. Chang. 2020, 20, 9. [Google Scholar] [CrossRef]
- Kerr, J.T.; Pindar, A.; Galpern, P.; Packer, L.; Potts, S.G.; Roberts, S.M.; Rasmont, P.; Schweiger, O.; Colla, S.R.; Richardson, L.L.; et al. Climate change impacts on bumblebees converge across continents. Science 2015, 349, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Pyke, G.H.; Thomson, J.D.; Inouye, D.W.; Miller, T.J. Effects of climate change on phenologies and distributions of bumble bees and the plants they visit. Ecosphere 2016, 7, e01267. [Google Scholar] [CrossRef]
- Ploquin, E.F.; Herrera, J.M.; Obeso, J.R. Bumblebee community homogenization after uphill shifts in montane areas of northern Spain. Oecologia 2013, 173, 1649–1660. [Google Scholar] [CrossRef]
- Michener, C.D. The Bees of the World; JHU Press: Baltimore, MD, USA, 2000; Volume 1. [Google Scholar]
- Requier, F.; Pérez-Méndez, N.; Andersson, G.K.; Blareau, E.; Merle, I.; Garibaldi, L.A. Bee and non-bee pollinator importance for local food security. Trends Ecol. Evol. 2023, 38, 196–205. [Google Scholar] [CrossRef]
- Rader, R.; Cunningham, S.; Howlett, B.G.; Inouye, D. Non-bee insects as visitors and pollinators of crops: Biology, ecology, and management. Annu. Rev. Entomol. 2020, 65, 391–407. [Google Scholar] [CrossRef]
- Engelhardt, E.K.; Biber, M.F.; Dolek, M.; Fartmann, T.; Hochkirch, A.; Leidinger, J.; Löffler, F.; Pinkert, S.; Poniatowski, D.; Voith, J. Consistent signals of a warming climate in occupancy changes of three insect taxa over 40 years in central Europe. Glob. Chang. Biol. 2022, 28, 3998–4012. [Google Scholar] [CrossRef] [PubMed]
- Kukkonen, J.M.; Mussaari, M.; Fred, M.S.; Brommer, J.E. A strong decline of the endangered Apollo butterfly over 20 years in the archipelago of southern Finland. J. Insect Conserv. 2022, 26, 673–681. [Google Scholar] [CrossRef]
- Martay, B.; Brewer, M.; Elston, D.; Bell, J.; Harrington, R.; Brereton, T.; Barlow, K.; Botham, M.; Pearce-Higgins, J. Impacts of climate change on national biodiversity population trends. Ecography 2017, 40, 1139–1151. [Google Scholar] [CrossRef]
- Fox, R.; Oliver, T.H.; Harrower, C.; Parsons, M.S.; Thomas, C.D.; Roy, D.B. Long-term changes to the frequency of occurrence of British moths are consistent with opposing and synergistic effects of climate and land-use changes. J. Appl. Ecol. 2014, 51, 949–957. [Google Scholar] [CrossRef] [PubMed]
- Radenković, S.; Schweiger, O.; Milić, D.; Harpke, A.; Vujić, A. Living on the edge: Forecasting the trends in abundance and distribution of the largest hoverfly genus (Diptera: Syrphidae) on the Balkan Peninsula under future climate change. Biol. Conserv. 2017, 212, 216–229. [Google Scholar] [CrossRef]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global pollinator declines: Trends, impacts and drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.J.; Paxton, R.J. The conservation of bees: A global perspective. Apidologie 2009, 40, 410–416. [Google Scholar] [CrossRef]
- Rahimi, E.; Barghjelveh, S.; Dong, P. Amount, distance-dependent and structural effects of forest patches on bees in agricultural landscapes. Agric. Food Secur. 2022, 11, 10. [Google Scholar] [CrossRef]
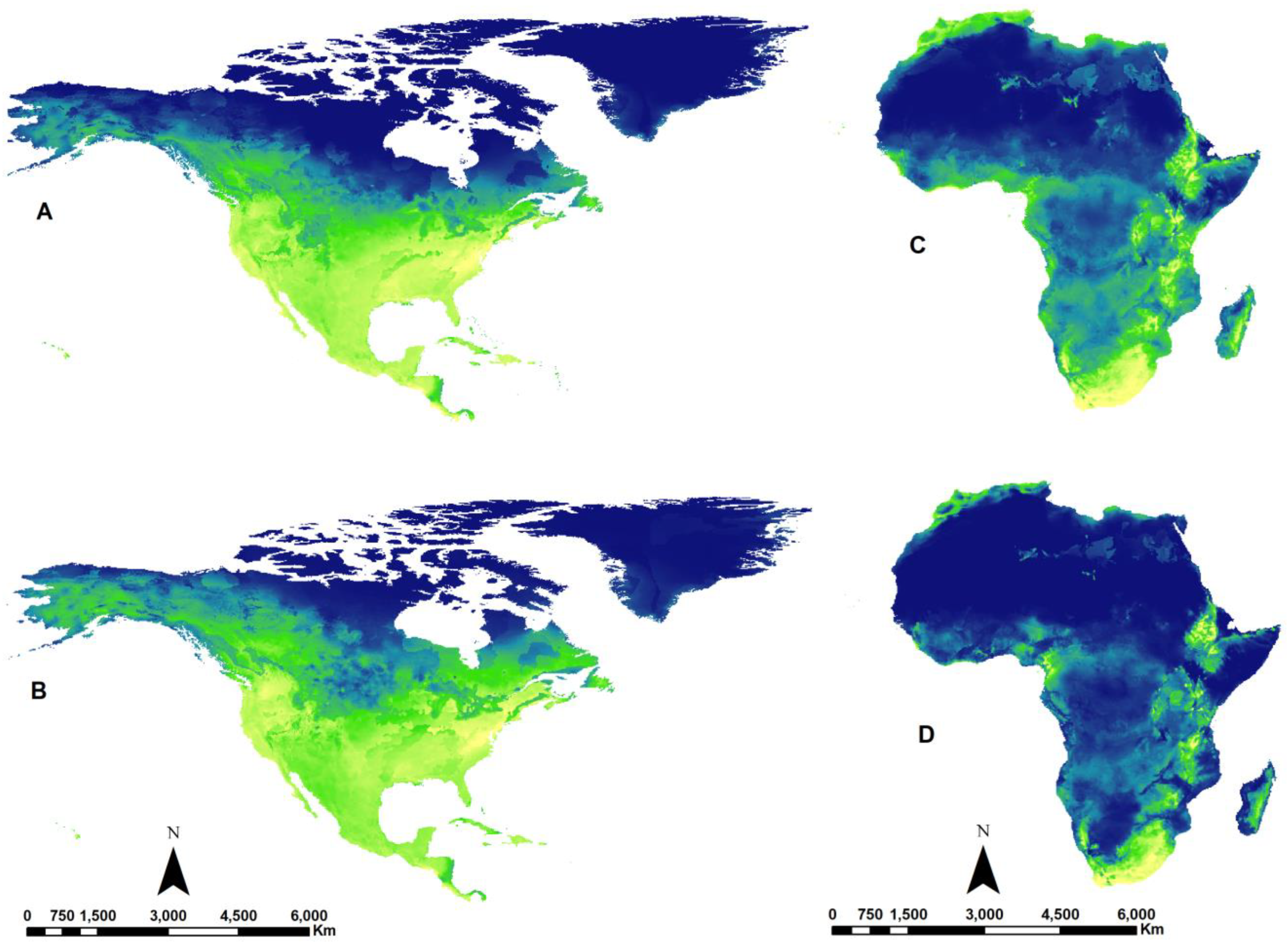
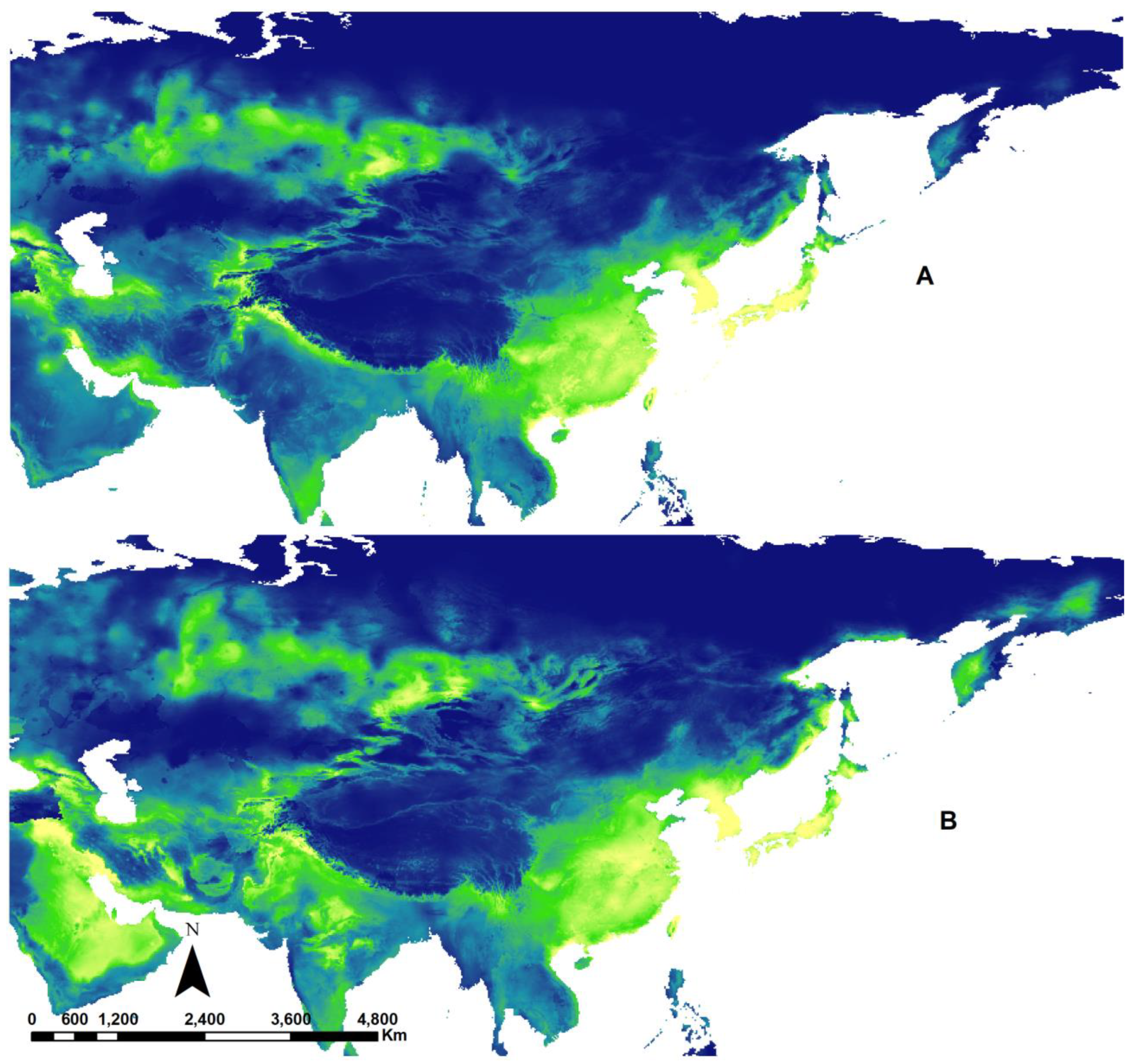
| Continent | Num. Species |
|---|---|
| Africa | 44 |
| Asia | 45 |
| Australia | 106 |
| Europe | 394 |
| North America | 697 |
| South America | 79 |
| Family | Count | Proportion% |
|---|---|---|
| Andrenidae | 67,221 | 5.25 |
| Apidae | 1,024,488 | 80.06 |
| Colletidae | 29,128 | 2.28 |
| Halictidae | 82,423 | 6.44 |
| Megachilidae | 69,143 | 5.40 |
| Melittidae | 7068 | 0.55 |
| Stenotritidae | 158 | 0.01 |
| Continent | AUC | BOYCE | IMAE |
|---|---|---|---|
| Africa | 0.94 (0.03) | 0.91 (0.05) | 0.91 (0.04) |
| Asia | 0.97 (0.02) | 0.93 (0.03) | 0.94 (0.04) |
| Australia | 0.90 (0.06) | 0.89 (0.04) | 0.85 (0.01) |
| Europe | 0.85 (0.04) | 0.91 (0.03) | 0.77 (0.07) |
| North America | 0.95 (0.02) | 0.90 (0.04) | 0.92 (0.05) |
| South America | 0.89 (0.08) | 0.91 (0.04) | 0.84 (0.11) |
| Continents | No. Species (Increase) | Increase% | No. Species (Decrease) | Decrease% |
|---|---|---|---|---|
| Africa | - | - | 44 | −51.4 (15) |
| Asia | 18 | 99 (80) | 27 | −47 (19) |
| Australia | 9 | 16.3 (18) | 97 | −28.4 (17) |
| Europe | 60 | 57.9 (66) | 334 | −56.7 (21) |
| North America | 366 | 48.2 (45) | 331 | −33 (22) |
| South America | 19 | 121.8 (195) | 60 | −45 (23) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahimi, E.; Jung, C. Global Trends in Climate Suitability of Bees: Ups and Downs in a Warming World. Insects 2024, 15, 127. https://doi.org/10.3390/insects15020127
Rahimi E, Jung C. Global Trends in Climate Suitability of Bees: Ups and Downs in a Warming World. Insects. 2024; 15(2):127. https://doi.org/10.3390/insects15020127
Chicago/Turabian StyleRahimi, Ehsan, and Chuleui Jung. 2024. "Global Trends in Climate Suitability of Bees: Ups and Downs in a Warming World" Insects 15, no. 2: 127. https://doi.org/10.3390/insects15020127
APA StyleRahimi, E., & Jung, C. (2024). Global Trends in Climate Suitability of Bees: Ups and Downs in a Warming World. Insects, 15(2), 127. https://doi.org/10.3390/insects15020127







