Demonstration of RNAi Yeast Insecticide Activity in Semi-Field Larvicide and Attractive Targeted Sugar Bait Trials Conducted on Aedes and Culex Mosquitoes
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Rearing
2.2. Yeast Culturing and Preparation
2.3. Yeast Preparation
2.4. Yeast Shipment
2.5. Semi-Field Larvicide Trials
2.6. RNAi Yeast ATSB Sedimentation Studies
2.7. Semi-Field Yeast ATSB Trials
3. Results and Discussion
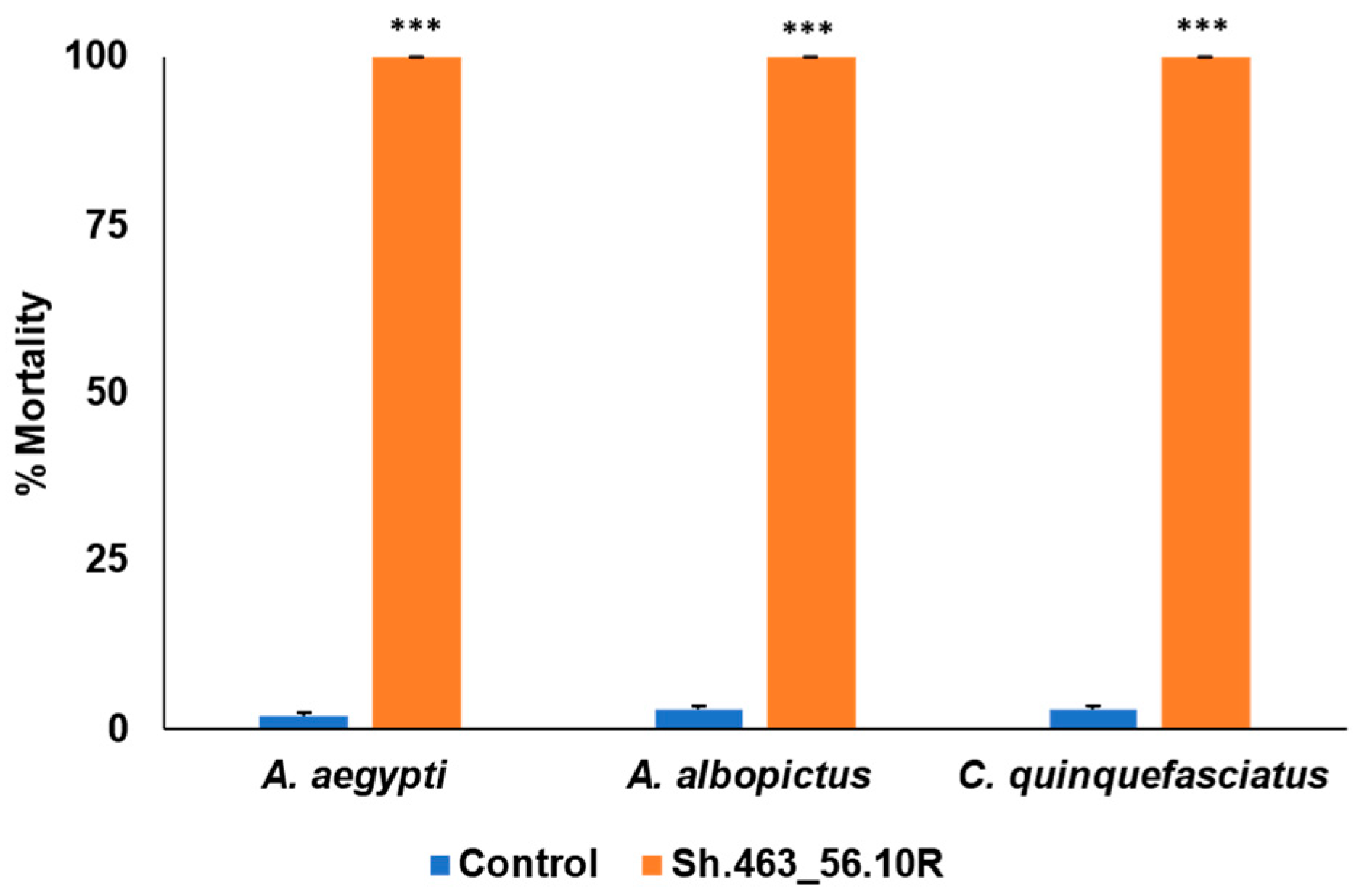
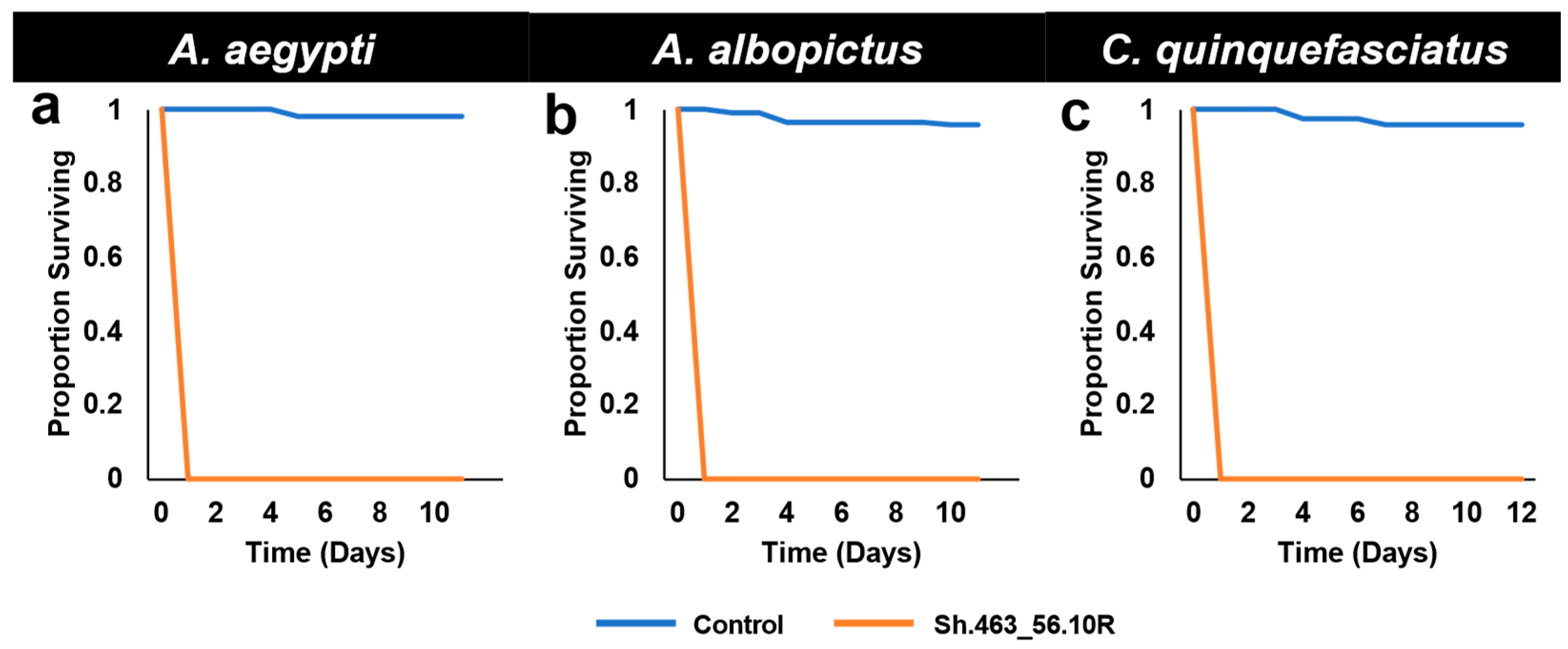
3.1. Semi-Field Larvicide Activity of Sh.463_56.10R Yeast
3.2. Development of a Stable Yeast RNAi ATSB Suspension
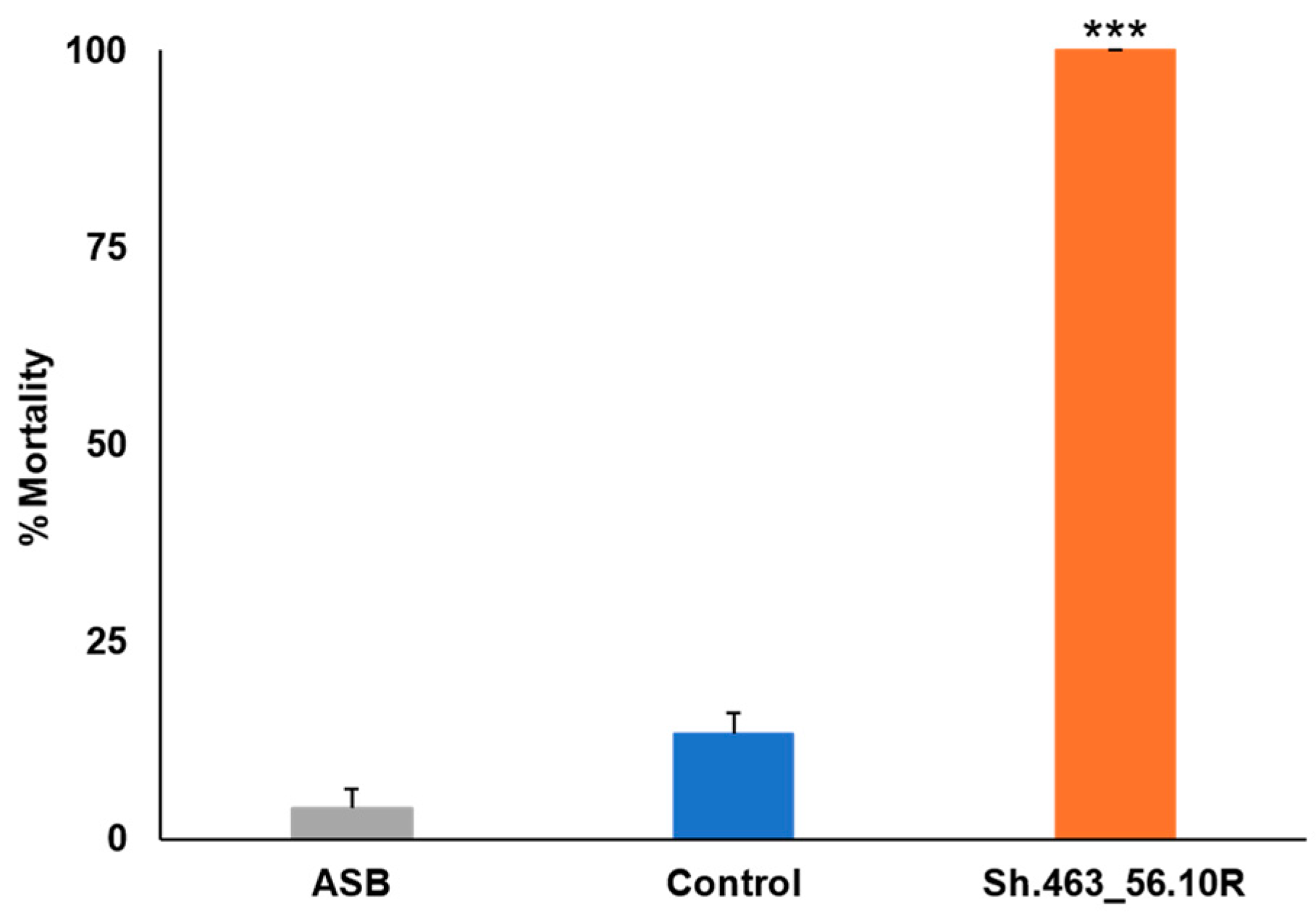
3.3. Semi-Field Trial Confirmation of Yeast RNAi ATSB Activity
3.4. Conclusions and Future Work
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hemingway, J.; Ranson, H.; Magill, A.; Kolaczinski, J.; Fornadel, C.; Gimnig, J.; Coetzee, M.; Simard, F.; Roch, D.K.; Hinzoumbe, C.K.; et al. Averting a malaria disaster: Will insecticide resistance derail malaria control? Lancet 2016, 387, 1785–1788. [Google Scholar] [CrossRef]
- Airs, P.M.; Bartholomay, L.C. RNA Interference for Mosquito and Mosquito-Borne Disease Control. Insects 2017, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Wiltshire, R.M.; Duman-Scheel, M. Advances in oral RNAi for disease vector mosquito research and control. Curr. Opin. Insect Sci. 2020, 40, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Mysore, K.; Hapairai, L.K.; Sun, L.; Li, P.; Wang, C.W.; Scheel, N.D.; Lesnik, A.; Igiede, J.; Scheel, M.P.; Wei, N.; et al. Characterization of a dual-action adulticidal and larvicidal interfering RNA pesticide targeting the Shaker gene of multiple disease vector mosquitoes. PLoS Negl. Trop. Dis. 2020, 14, e0008479. [Google Scholar] [CrossRef] [PubMed]
- Brizzee, C.; Mysore, K.; Njoroge, T.M.; McConnell, S.; Hamid-Adiamoh, M.; Stewart, A.T.M.; Kinder, J.T.; Crawford, J.; Duman-Scheel, M. Targeting mosquitoes through generation of an insecticidal RNAi yeast strain using Cas-CLOVER and super piggyBac engineering in Saccharomyces cerevisiae. J. Fungi 2023, 9, 1056. [Google Scholar] [CrossRef]
- Fiorenzano, J.M.; Koehler, P.G.; Xue, R.D. Attractive toxic sugar bait (ATSB) for control of mosquitoes and its impact on non-target organisms: A Review. Int. J. Environ. Res. Public Health 2017, 14, 398. [Google Scholar] [CrossRef] [PubMed]
- Traore, M.M.; Junnila, A.; Traore, S.F.; Doumbia, S.; Revay, E.E.; Kravchenko, V.D.; Schlein, Y.; Arheart, K.L.; Gergely, P.; Xue, R.D.; et al. Large-scale field trial of attractive toxic sugar baits (ATSB) for the control of malaria vector mosquitoes in Mali, West Africa. Malar. J. 2020, 19, 72. [Google Scholar] [CrossRef] [PubMed]
- Attractive Targeted Sugar Bait Phase III Trial Group. Attractive targeted sugar bait phase III trials in Kenya, Mali, and Zambia. Trials 2022, 23, 640. [Google Scholar] [CrossRef]
- Mysore, K.; Hapairai, L.K.; Wei, N.; Realey, J.S.; Scheel, N.D.; Severson, D.W.; Duman-Scheel, M. Preparation and use of a yeast shRNA delivery system for gene silencing in mosquito larvae. Methods Mol. Biol. 2019, 1858, 213–231. [Google Scholar] [CrossRef]
- WHO. Guidelines for Laboratory and Field Testing of Mosquito Larvicides; WHO: Geneva, Switzerland, 2005. [Google Scholar]
- US EPA, Pesticide Product Label, Attractive Toxic Sugar Bait. Available online: https://www3.epa.gov/pesticides/chem_search/ppls/085125-00002-20120319.pdf (accessed on 1 December 2023).
- Mysore, K.; Sun, L.; Hapairai, L.K.; Wang, C.W.; Roethele, J.B.; Igiede, J.; Scheel, M.P.; Scheel, N.D.; Li, P.; Wei, N.; et al. A Broad-Based Mosquito Yeast Interfering RNA Pesticide Targeting Rbfox1 Represses Notch Signaling and Kills Both Larvae and Adult Mosquitoes. Pathogens 2021, 10, 1251. [Google Scholar] [CrossRef]
- Mysore, K.; Sun, L.; Hapairai, L.K.; Wang, C.W.; Igiede, J.; Roethele, J.B.; Scheel, N.D.; Scheel, M.P.; Li, P.; Wei, N.; et al. A Yeast RNA-Interference Pesticide Targeting the Irx Gene Functions as a Broad-Based Mosquito Larvicide and Adulticide. Insects 2021, 12, 986. [Google Scholar] [CrossRef] [PubMed]
- Maine, E.M. Studying gene function in Caenorhabditis elegans using RNA-mediated interference. Brief. Funct. Genom. 2008, 7, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Wudarski, J.; Ustyantsev, K.; Glazenburg, L.; Berezikov, E. Influence of temperature on development, reproduction and regeneration in the flatworm model organism, Macrostomum lignano. Zoological Lett. 2019, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Adelman, Z.N.; Anderson, M.A.; Wiley, M.R.; Murreddu, M.G.; Samuel, G.H.; Morazzani, E.M.; Myles, K.M. Cooler temperatures destabilize RNA interference and increase susceptibility of disease vector mosquitoes to viral infection. PLoS Negl. Trop. Dis. 2013, 7, e2239. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.B.; Dong, W.; Li, K.X.; Wang, J.J.; Shen, J.; Moussian, B.; Zhang, J.Z. Flexible manipulation of Omb levels in the endogenous expression region of Drosophila wing by combinational overexpression and suppression strategy. Insect Sci. 2020, 27, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Paixao, E.S.; Teixeira, M.G.; Rodrigues, L.C. Zika, chikungunya and dengue: The causes and threats of new and re-emerging arboviral diseases. BMJ Glob. Health 2018, 3, e000530. [Google Scholar] [CrossRef]
- Soni, S.; Gill, V.J.S.; Anusheel; Singh, J.; Chhabra, J.; Gill, G.J.S.; Bakshi, R. Dengue, Chikungunya, and Zika: The causes and threats of emerging and re-emerging arboviral diseases. Cureus 2023, 15, e41717. [Google Scholar] [CrossRef]
- Balkew, M.; Mumba, P.; Dengela, D.; Yohannes, G.; Getachew, D.; Yared, S.; Chibsa, S.; Murphy, M.; George, K.; Lopez, K.; et al. Geographical distribution of Anopheles stephensi in eastern Ethiopia. Parasites Vectors 2020, 13, 35. [Google Scholar] [CrossRef]
- Timmons, L.; Court, D.L.; Fire, A. Ingestion of bacterially expressed dsRNAs can produce specific and potent genetic interference in Caenorhabditis elegans. Gene 2001, 263, 103–112. [Google Scholar] [CrossRef]
- Whyard, S.; Erdelyan, C.N.; Partridge, A.L.; Singh, A.D.; Beebe, N.W.; Capina, R. Silencing the buzz: A new approach to population suppression of mosquitoes by feeding larvae double-stranded RNAs. Parasites Vectors 2015, 8, 96. [Google Scholar] [CrossRef]
- Taracena, M.; Hunt, C.; Pennington, P.; Andrew, D.; Jacobs-Lorena, M.; Dotson, E.; Wells, M. Effective oral RNA interference (RNAi) administration to adult Anopheles gambiae mosquitoes. J. Vis. Exp. 2022, 181, e63266. [Google Scholar] [CrossRef]
- Asgarian, T.S.; Vatandoost, H.; Hanafi-Bojd, A.A.; Nikpoor, F. Worldwide status of insecticide resistance of Aedes aegypti and Ae. albopictus, vectors of arboviruses of Chikungunya, Dengue, Zika and Yellow Fever. J. Arthropod Borne Dis. 2023, 17, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Vereecken, S.; Vanslembrouck, A.; Kramer, I.M.; Muller, R. Phenotypic insecticide resistance status of the Culex pipiens complex: A European perspective. Parasites Vectors 2022, 15, 423. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Cha, S.J.; Jacobs-Lorena, M. New weapons to fight malaria transmission: A historical view. Entomol. Res. 2022, 52, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Environmental Protection Agency. Final Regulatory Decision for New Active Jngredients Double-Stranded Ribonucleic Acid Transcript Comprising a DvSnj7 Inverted Repeat Sequence Derived from Western Corn Rootworm (Diabrotica virgifera virgifera) and Bacillus thuringiensis cry3Bbl Protein and the Genetic, Aterial (vector PV-ZMIR10871) Necessary for Their Production in MON 87411 Corn (OECD Unique Identifier: MON-87411-9). Available online: https://www.regulations.gov/document/EPA-HQ-OPP-2014-0293-0398 (accessed on 15 October 2015).
- Erickson, B.E. RNAi Pesticide Moves Closer to US Approval. Chemical & Engineering News, 4 October 2023; p. 17. [Google Scholar]
- EPA. Pesticide Registration. Available online: https://www.epa.gov/pesticide-registration/about-pesticide-registration (accessed on 1 July 2023).
- EPA. Drinking Water and Pesticides. Available online: https://www.epa.gov/safepestcontrol/drinking-water-and-pesticides (accessed on 1 December 2023).
- Murphy, K.A.; Tabuloc, C.A.; Cervantes, K.R.; Chiu, J.C. Ingestion of genetically modified yeast symbiont reduces fitness of an insect pest via RNA interference. Sci. Rep. 2016, 6, 22587. [Google Scholar] [CrossRef] [PubMed]
- Michigan State University Extension. Spotted Wing Drosophila. Available online: https://www.canr.msu.edu/ipm/Invasive_species/spotted_wing_drosophila/index (accessed on 1 September 2022).
- Bolda, M.P.; Coates, W.W.; Grant, A.; Zalom, F.G.; Van Steenwyk, R.A.; Caprile, J.; Flint, M.L. Spotted Wing Drosophila, Drosophila suzukii: A New Pest in California. 2009. Available online: www.ipm.ucdavis.edu/EXOTIC/drosophila.html (accessed on 1 September 2022).
- Spies, J.M.; Liburd, O.E. Comparison of attractants, insecticides, and mass trapping for managing (Diptera: Drosophilidae) in blueberries. Fla. Entomol. 2019, 102, 315–321. [Google Scholar] [CrossRef]
- Allen, M.L. Prospects for using RNAi as control for ants. Front. Agron. 2021, 3, 591539. [Google Scholar] [CrossRef]
- Zhu, F.; Lavine, L.; O’Neal, S.; Lavine, M.; Foss, C.; Walsh, D. Insecticide resistance and management strategies in urban ecosystems. Insects 2016, 7, 2. [Google Scholar] [CrossRef]
- Lucena-Leandro, V.S.; Abreu, E.F.A.; Vidal, L.A.; Torres, C.R.; Junqueira, C.I.C.V.F.; Dantas, J.; Dantas, J.; Albuquerque, E.V.S. Current scenario of exogenously induced RNAi for Lepidopteran agricultural pest control: From dsRNA design to topical application. Int. J. Mol. Sci. 2022, 23, 15836. [Google Scholar] [CrossRef]
- Thizy, D.; Emerson, C.; Gibbs, J.; Hartley, S.; Kapiriri, L.; Lavery, J.; Lunshof, J.; Ramsey, J.; Shapiro, J.; Singh, J.A.; et al. Guidance on stakeholder engagement practices to inform the development of area-wide vector control methods. PLoS Negl. Trop. Dis. 2019, 13, e0007286. [Google Scholar] [CrossRef]
- Lavery, J.V.; Tinadana, P.O.; Scott, T.W.; Harrington, L.C.; Ramsey, J.M.; Ytuarte-Nuñez, C.; James, A. Towards a framework for community engagement in global health research. Trends Parasitol. 2010, 26, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Bartumeus, F.; Costa, G.B.; Eritja, R.; Kelly, A.H.; Finda, M.; Lezaun, J.; Okumu, F.; Quinlan, M.M.; Thizy, D.C.; Toé, L.P. Sustainable innovation in vector control requires strong partnerships with communities. PLoS Negl. Trop. Dis. 2019, 13, e0007204. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stewart, A.T.M.; Mysore, K.; Njoroge, T.M.; Winter, N.; Feng, R.S.; Singh, S.; James, L.D.; Singkhaimuk, P.; Sun, L.; Mohammed, A.; et al. Demonstration of RNAi Yeast Insecticide Activity in Semi-Field Larvicide and Attractive Targeted Sugar Bait Trials Conducted on Aedes and Culex Mosquitoes. Insects 2023, 14, 950. https://doi.org/10.3390/insects14120950
Stewart ATM, Mysore K, Njoroge TM, Winter N, Feng RS, Singh S, James LD, Singkhaimuk P, Sun L, Mohammed A, et al. Demonstration of RNAi Yeast Insecticide Activity in Semi-Field Larvicide and Attractive Targeted Sugar Bait Trials Conducted on Aedes and Culex Mosquitoes. Insects. 2023; 14(12):950. https://doi.org/10.3390/insects14120950
Chicago/Turabian StyleStewart, Akilah T. M., Keshava Mysore, Teresia M. Njoroge, Nikhella Winter, Rachel Shui Feng, Satish Singh, Lester D. James, Preeraya Singkhaimuk, Longhua Sun, Azad Mohammed, and et al. 2023. "Demonstration of RNAi Yeast Insecticide Activity in Semi-Field Larvicide and Attractive Targeted Sugar Bait Trials Conducted on Aedes and Culex Mosquitoes" Insects 14, no. 12: 950. https://doi.org/10.3390/insects14120950
APA StyleStewart, A. T. M., Mysore, K., Njoroge, T. M., Winter, N., Feng, R. S., Singh, S., James, L. D., Singkhaimuk, P., Sun, L., Mohammed, A., Oxley, J. D., Duckham, C., Ponlawat, A., Severson, D. W., & Duman-Scheel, M. (2023). Demonstration of RNAi Yeast Insecticide Activity in Semi-Field Larvicide and Attractive Targeted Sugar Bait Trials Conducted on Aedes and Culex Mosquitoes. Insects, 14(12), 950. https://doi.org/10.3390/insects14120950






