Simple Summary
There is increasing evidence of declining bee populations due to anthropogenic factors. We assessed the abundance of nine wild bee species over a four-year study period, estimating changes through monthly captures. Apis mellifera, the Lasioglossum (Dialictus spp.) complex, and Macrotera sinaloana had the largest population sizes and densities. Seasonal fluctuations were seen in most species in spring (March–May) coinciding with the flowering period of the main plant species and a low abundance during the winter months (December–January). Moreover, 77.7% of the populations showed a tendency to remain constant over time, with yellow tray traps capturing a higher number of individuals.
Abstract
Recognizing how populations fluctuate over time is a crucial factor in determining the environmental elements affecting population persistence. However, the limited information on wild bee populations complicates the estimation of the impact of anthropogenic threats leading to changes in population size. To address this, we conducted a study capturing and monitoring nine species of wild bees through monthly samplings over four years. Tray traps were placed in permanent plots, and capture records were used to determine population size (N) and density (D). A generalized linear model (GLM) was employed to determine how the use of traps affected bee species captures. The families Apidae and Halictidae represented the most captures. Apis mellifera, the Lasioglossum (Dialictus spp.) complex, and Macrotera sinaloana exhibited the largest number of captures and highest population density. Most species (77.7%) showed a tendency to remain constant over the years and to have a higher number of captures in the spring months. Moreover, yellow traps were the most effective in capturing bee individuals. We suggest that the availability of essential resources and the reduction in environmental stressors positively affected the capture of wild bee populations.
1. Introduction
Bees are central for terrestrial ecosystems, as eighty-five to ninety percent of angiosperms depend on pollinator services [1,2]. There are close to 20,000 bee species worldwide, and the Mexican apifauna is regarded as one of the most diverse, encompassing approximately 144 genera and 1908 species across six families, namely Andrenidae, Apidae, Colletidae, Halictidae, Megachilidae, and Melittidae [3,4]. The highest bee richness is situated in the arid and semi-arid regions of North America, as well as those with a Mediterranean climate [3,5].
There is an internationally recognized pollinator crisis [6,7,8], with 40 percent of invertebrates having a threatened status and, on the IUCN, [9] red list; of these, 9% are bees, and bee populations have decreased by 37% [6]. This decrease in bee populations could have negative consequences for vegetation in terrestrial ecosystems and economic loss [10]. About 35% of global food crops are pollinated by these insects [6,7,10,11]. For example, the strong interrelationship between the cactus pear (Opuntia spp.) and solitary bees is critical for this staple crop in North America [12], and even species that are self-compatible, such as canola agroecosystems, require pollen exchange by Apis mellifera (Linnaeus, 1758) for optimal reproductive success [6,10]. The deterioration of natural environments, mostly due to anthropogenic activities such as agriculture, livestock husbandry, deforestation, and land use change, has led to a decrease in bee populations [13,14,15]. These activities have reduced patches of wild vegetation, leading to a loss of food (pollen and nectar), as well as resting, copulation, and nesting places, which are essential for the survival of bee populations [13,14,16] For example, Cane et al. [17] reported lower numbers of bee species and individuals in urbanized patches and surrounding vegetation than in wild vegetation (predominantly Larrea tridentata ((DC.) Coville, 1893), with species such as Eucera venusta (Timberlake, 1961), Megandrena enceliae (Cockerell, 1927), and Perdita lateralis (Timberlake, 1962) (ground-nesting or L. tridentata specialist bees) being nonexistent in urbanized patches.
The introduction of exotic species has further compounded the loss of bee populations [13]. Exotic species can be vectors of pathogens (e.g., Varroa destructor (Anderson & Trueman, 2000)), affecting bee populations in a relatively short period of time (between 2 and 4 years) [18]. They can also compete with native species, which results in a loss of native species (>10%) [19]. At the same time, the use of pesticides containing neonicotinoids causes physiological alterations to the nervous and reproductive system as well as immunosuppression, with negative impacts on bee populations [20]. While the decline in bee populations is acknowledged, studies assessing the decline in wild bee populations are scarce [21]. Most studies focus on economically important bees such as Apis mellifera and Bombus spp. [6,7], while few efforts have been made to assess the wild bees on which the vegetation of natural ecosystems depends.
Population size is one of the attributes most used by ecologists to determine the status of biological populations [22,23]. However, in practice, the estimation of the number of individual bees poses methodological problems, is often not as precise as desirable, and is generally measured with data from captured individuals to determine their abundance, frequency, and density [24]. Short-term studies of biological populations can provide information allowing us to quickly identify emerging patterns and potential trends of populations at a specific moment [25]. However, they must be complemented with long-term monitoring programs and other evaluation techniques to ensure the persistence of populations over time [25].
The aim of this study was to compare the population abundance and density of wild bee species over a four-year period to highlight potential trends, threats, and factors that favor bee populations in the southern Chihuahuan Desert.
2. Materials and Methods
2.1. Study Area
This study was carried out at the southernmost edge of the Chihuahuan Desert in the desert scrubland of Cadereyta de Montes, Querétaro, Mexico. The climate is hot and semi-arid with summer rains (BS1kw) [26], with an annual precipitation between 400 and 450 mm and an annual temperature that ranges between 16 and 18 °C [27]. These heterogeneous environmental conditions result in a varied mosaic of microphyllous, rosetophyllous, and carissicaulous scrub as the predominant vegetation [28]. Bee populations were sampled monthly for four years (2015, 2016, 2018, and 2019) in an area managed by the Cadereyta Regional Botanical Garden, “Ing. Manuel González de Cosío”. This 7.3 ha area maintains a conserved remnant of crassicaulous desert scrub, where the vegetation is characterized by mesquite (Prosopis laevigata (Humb. & Bonpl. Ex Willd.)), prickly pears (Opuntia cantabrigiensis (Lynch, 1903), O. robusta (H.L.Wendl. ex Pfeiff., 1837), O. tomentosa (Salm-Dyck, 1822), and O. straptacantha (Lem., 1839)), agave (Agave mapisaga (Trel., 1920)), leatherstem (Jatropha dioica (Cerv.)), fishhook cactus (Mammillaria uncinata (Zucc. Ex Pfeiff., 1837)), candy barrel cactus (Ferocactus histrix ((DC.) G.E.Linds., 1955)), blue myrtle cactus (Myrtillocactus geometrizans (Mart. Ex Pfeiff., 1897) Console), and yucca (Yucca filifera (Chabaud, 1876)) [29]. The area around the conserved space of the botanical garden is bounded by a Y. filifera plantation, an urbanized area, and an area with a remnant of xerophytic scrub that has been disturbed due to the introduction of livestock alongside the establishment of pastures for livestock feeding. Environmental variables such as mean temperature and precipitation and maximum wind speed (Table 1) were taken from the meteorological station found in the Botanical Garden [30].

Table 1.
Average and standard deviation of the environmental parameters, as well as the Pearson regression coefficients and the estimated p-values between the total abundance of bees with each environmental parameter (temperature, precipitation, and wind speed). Data from monthly sampling of wild bees across four years (2015, 2016, 2018, and 2019), Cadereyta de Montes, Querétaro, Mexico. Environmental parameters are from the meteorological station at the Botanical Garden [30].
2.2. Bee Capture
A three-day-long sampling period was carried out once per month for four years (2015, 2016, 2018, and 2019). Within the study site, 3 × 5 m plots were sampled in which three pan traps (yellow, blue, and purple for a total of 30 traps per sample period) were placed in each plot. Traps were placed on the ground and near flowering plants to capture the largest number of individuals [31]. Each pan trap (16 cm in diameter and 6.5 cm deep) had a 125 mL water solution with 5% commercial liquid soap [31]. These traps were distributed throughout all areas of the conserved area to encompass both central and peripheral sites. This arrangement sought to ensure comprehensive coverage of the various areas where bees occur [32]. The selection of colors has been shown to increase the diversity of species captured as they respond differently to color [33,34]. Furthermore, these colors match those of the flowers found in most plants in the study area. Traps were placed at 0900 h and removed at 2000 h every day, encompassing the opening hours of most of the flowers on the site [35]. Each color trap was 5 m apart since traps separated by smaller distances decreased the number of bees captured [35]. Collected bees were kept in 70% alcohol for later identification carried with the help of Dr. Ismael Hinojosa Diaz and Dr. Ricardo Ayala Barajas, Institute of Biology, UNAM.
2.3. Data Analysis
The abundance (N), the total number of individuals captured monthly as well as annually, their density (D = N/A) [36], and the number of individuals captured per unit of area (A = the area of the site is 7.3 ha) were recorded. We also evaluated the relationship between the total abundance of bees and environmental parameters for each sampling year using Pearson’s correlations [37]. Changes in abundance between sampled years were calculated using the cumulative abundance data for each month. The abundance between sampling periods reflects a relative change in abundance between time periods and would suggest constant captures over sampling periods or either declines or increases in the number of captured individuals. Finally, a generalized linear model (GLM) [38], suitable for abundance data, was used to analyze the relationship between abundance (dependent variable) and three factors: species, year, and color of the trap (independent variables).
3. Results
A total of 2208 individual bees were captured throughout the study period. These were classified into 12 species and the Dialictus complex, representing 12 genera and 4 families: Andrenidae (1 species), Apidae (6 species), Halictidae (5 species), and Megachilidae (3 species). Ceratina sp1 (n = 48), Ceratina sp2 (n = 33), Agapostemon sp. (n = 119), Augochlora sp. (n = 14), the Lasioglossum (Evylaeus spp.) complex (n = 63), and Lithurgus planifrons (n = 51) were only captured in 2015, an important indicator of absence. Eight bee species and the Lasioglossum (Dialictus) complex accounted for 1880 captured individuals found in at least two years (Table 2; Figure 1). The highest number of captures were for A. mellifera (n = 419), followed by Lasioglossum (Dialictus spp.) (n = 370) and Macrotera sinaloana (n = 347), while B. pensylvanicus (n = 21), Melissodes sp. (n = 63), and Megachile sp. (n = 88) had the lowest. Over time, the abundance of some captured species remained relatively constant (Figure 1), while that of others (A. mellifera and B. pensylvanicus) decreased (Figure 1). The A. mellifera population in the study site is not managed and possibly escaped from a bee farm located in the region. No captures of individuals of B. pensylvanicus were obtained after 2016, while Melissodes sp. and Lithurgus littoralis were absent in 2015 (Table 2; Figure 1). Regarding the average density over the years, the species that obtained more than 10 ind • ha−1 were M. sinaloana, A. mellifera, and the Lasioglossum (Dialictus spp.) complex (Table 2), while the rest had a low average density (<10 ind • ha−1).

Table 2.
Families and species of bees present in Cadereyta de Montes, Querétaro, Mexico. Abundance (N) and density (D, ind • ha−1) for each year of sampling, total abundances, and average density over the study period (monthly sampling across four years: 2015, 2016, 2018, and 2019).
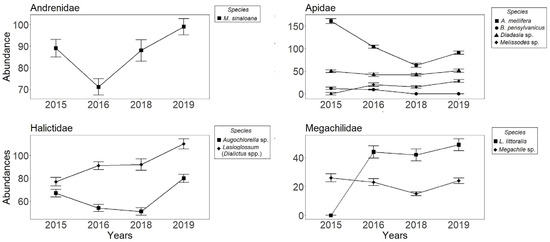
Figure 1.
Abundance of bees (±SE) collected in pan traps and grouped by family in Cadereyta de Montes, Querétaro, México. Data from monthly sampling across four years: 2015, 2016, 2018, and 2019.
The correlation between abundance and the environmental variables (Table 1) showed that over the four years, all three environmental variables had a positive relationship with abundance; however, the correlation was not always significant. Temperature had the highest correlation values during the four years (Table 1), with 2016 and 2019 having the highest correlation values. On the other hand, wind speed in 2015 had a negative correlation with bee abundance.
There was an increase in abundance within years over the course of the sampling period (Figure 2). Spring (March, April, and May) clearly reflected increased activity of solitary bee populations, as flying adults emerged from nests. Halfway through the year, there was a gradual decrease in the number of individuals for many species, such as Melissodes sp., of which we have captured no individuals as of August, or Megachile sp., which we stopped recording in the month of October. In December, only individuals of Augochlorella sp., B. pensylvanicus, the Lasioglossum (Dialictus sp.) complex, and M. sinaloana were captured. A. mellifera was present throughout the year, being especially abundant in the latter half (Figure 2). Lastly, the GLM analysis revealed that bee species (χ2 = 890.69, p < 2 × 10−16, df = 8), pan trap color (χ2 = 571.43, p < 2 × 10−16, df = 2), year (χ2 = 17.02, p < 0.0007, df = 3), and year x species interactions were significant (χ2 = 202.26, p < 2 × 10−16, df = 24). Even though the year was important, abundance was only significantly different between 2018 and 2019 (p = 0.0003). The yellow trap (n = 1049) captured the highest number of individuals, followed by the blue trap (n = 602), and, lastly, the violet trap (n = 229). Most captured individuals belonged to the Lasioglossum (Dialictus spp.) complex. This suggests that both the color of the trap and the bee species are influential factors in captures in the study area. However, the year x color (χ2 = 4.84, p < 0.56, df = 6) but not species × color interactions were significant (χ2 = 25.49, p < 0.06, df = 16).
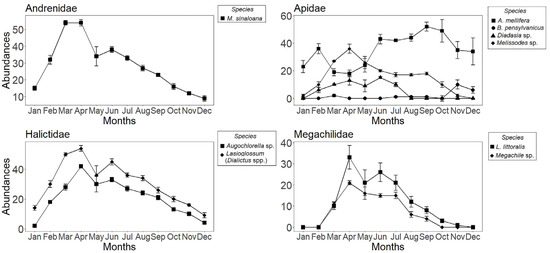
Figure 2.
Abundance of bees (±SE) per month of capture in Cadereyta de Montes, Querétaro, Mexico. Species are shown by family. Data from data monthly sampling across four years: 2015, 2016, 2018 and 2019.
4. Discussion
Our study revealed that of the eight species and one species complex caught over a span greater than one year, seven bee species experienced fluctuations in their abundance and density within each year with a tendency to remain relatively constant over time, while one exhibited a decline, and one showed an increase. This tendency to remain constant could be attributed to the availability of between-year resources and stability of environmental factors such as temperature, precipitation, and wind speed in the study area, which is consistent with previous studies [4,5,39]. Most individuals were captured during the months of March to May across the four sampled years, a time when the flowering peaks of cacti and the other dominant species are seen, suggesting that most bee populations have life cycles that are synchronized with the flowering of the plants during that period. This observation is inconsistent with Roubik [40], who found a change in the relationships between bees and the vegetation of a tropical forest due to a delay in flowering and the availability of resources. We found that bee abundance was correlated with annual temperature, with the optimal temperature ranging between 20 to 35 °C, consistent with that reported by Ortiz-Sánchez and Aguirre-Segura [39]. There was little variation between years for most species; however, changing environmental conditions over a longer period may alter these conditions. This was clearly seen with the absence of five species and the Lasioglossum (Evylaeus) complex after the 2015 sampling period.
Species were most abundant in the months of March, April, and May, declining after June. Most of these bee species are solitary, with univoltine life cycles (species that reproduce only once a year and have a diapause during the larval phase, which in this case can last for a year). The changes in seasonal abundance were related to the nature of the life cycle, with the new cohort of flying adults emerging the following year [5]. Many plants species flower between March and September in the Chihuahuan Desert, with spring being the time when there is the highest production of floral resources [41]. This synchronization with floral resources was also observed by Minckley et al. [42], who identified the spatio–temporal interaction between bees and their floral resources, especially with Larrea tridentata, in the Chihuahuan, Sonoran, and Mojave Deserts. In contrast, three species, Apis mellifera, Macrotera sinaloana, Diadasia sp., and the Lasioglossum (Dialictus sp.) complex, were found year-round and during the four-year study period. The native solitary species (Macrotera sinaloana, Lasioglossum (Dialictus sp.) complex, and Diadasia sp.) have bivoltine life cycles (two generations per year); however, these species also undergo diapause in both the larval and adult stages, which helps them maintain their populations over time [5]. Packer et al. [43] reported that several species of Halictidae, among them Lasioglossum (Dialictus sp.) laevissimum, are bivoltine, with the first individuals emerging in June and a second generation of individuals in August. Neff and Simpson [44] also mention Diadasia rinconis as having two generations per year, one that appears when there is an abundance of flowers of three species of Opuntia (O. leptocaulis, O. macrorhiza, and O. engelmannii var. lindheimeri) and the other long after the peak of flowering; this second generation generally dies without reproducing.
Indirect capture methods, such as the use of pan traps, have limitations in estimating the abundance of bee populations [45] and may not reflect the actual bee diversity and population dynamics. These limitations include bias in the capture of individuals due to the lack of representativeness of the sample with respect to the total population, mortality of individuals during the study, environmental factors at the time of capture, and the color of the trap [45,46]. Pan traps have also been shown to favor the capture of Halictids [47], as found in this study. In contrast, direct capture methods, such as marking and recapturing, provide more accurate estimates of abundance since they allow for a representative sample of the total population, as well as good estimates of demographic parameters such as survival and mortality [46,48,49]. However, the latter methods are expensive and labor-intensive. Nevertheless, despite the limitations of indirect capture methods, they have certain advantages over direct methods: they allow for obtaining information on the distribution and abundance of populations in limited areas (such as solitary bees), avoiding overestimation of abundance, and underestimation of mortality [49]. To minimize capture bias, our study was carried out in a specific area where relevant aspects, such as life histories, feeding (oligolectic or polylectic), nesting, and flight ranges of bee populations were considered to properly place traps [5,50].
We found that 77% of bee abundances were rather constant over the four-year study period, while 11% increased. This stability may be due to the conditions surrounding the botanical garden, which potentially provide the necessary resources to maintain the populations [5,50]. It is important to note that short-term studies have limitations and may not provide a complete picture of wild bee populations at the study site; however, with these results, we can suggest that a management plan that includes the relationship between bee populations and plant species is needed. Plants such as prickly pears (Opuntias sp.), mesquite (Prosopis laevigata), and small shrubs and herbs (Stevia sp., Asclepias sp., Sphaeralcea sp., and Mimosa sp.) can promote the stability of bee populations and are able to conserve the necessary habitat conditions, such as soil characteristics and the presence of adequate microhabitats for the survival of the community [51,52]. Conservation policies are usually directed more towards the networks of species that interact in the ecosystem on a large scale than concentrating conservation efforts on a single species and its specific habitat [53]. However, in some cases, the management of an ecosystem can be guided by the need of a particular species or taxon, such that bees can function as a group of flagship species for plants and other animals in arid ecosystems given their importance and the richness found in these environments [50].
5. Conclusions
Although indirect sampling methods and short-term studies have their limitations, these methods can aid in providing preliminary information and are cost-effective and accessible means of assessing abundance within terrestrial ecosystems. This study highlights the importance of pan trap color, year, season, and species for bee abundance in the southern Chihuahuan Desert.
Author Contributions
Conceptualization, E.O.M.-S., J.G. and M.C.M.; methodology, E.O.M.-S. and J.G.; software, E.O.M.-S. and J.G.; validation E.O.M.-S., J.G. and M.C.M.; formal analysis, E.O.M.-S. and J.G.; investigation, E.O.M.-S.; resources, J.G. and M.C.M.; data curation, E.O.M.-S.; writing—original draft preparation, E.O.M.-S.; writing—review and editing, J.G. and M.C.M.; visualization, E.O.M.-S., J.G. and M.C.M.; supervision, J.G. and M.C.M.; project administration, J.G.; funding acquisition, J.G. and M.C.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Instituto de Ecología, UNAM, SEP-CONACyT 221362, to María C. Mandujano (MCM), the Conacyt Scholarship to Esteban Munguía-Soto (637049), and the GEF 00089333 grant for the “Enhancing National Capacities to manage Invasive Alien Species (IAS) by implementing the National Strategy on IAS” project to MCM and Dr. Jordan Golubov.
Data Availability Statement
The databases used are available in the repository https://github.com/Esteban-Mun10/Mungu-a_etal_bees (accessed on 10 October 2023).
Acknowledgments
We acknowledge Consejo Nacional de Ciencia y Tecnología (CONACyT), which awarded a scholarship to Esteban O. Munguía Soto (637049). José Antonio Aranda Pineda, Isabel Briseño Sánchez, Linda Mariana Martínez Ramos, and Oscar Sandino Guerrero Eloisa are acknowledged for their help during field work. We thank Linna Kiere for English translation and M. Golubov, who thoroughly and critically read and edited the manuscript. We thank the staff of the botanical garden for their support and the opportunity to work in their facilities. The first author is a student of the PhD Program Doctorado en Ciencias Biológicas y de la Salud—Universidad Autónoma Metropolitana Xochimilco (UAM-X), and the paper is part of his dissertation in partial fulfillment of the requirements for the graduate program. Mariana Rojas Aréchiga provided logistic support for field work and processing the scientific collection permit.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Resasco, J.; Chacoff, N.P.; Vázquez, D.P. Plant–pollinator interactions between generalists persist over time and space. Ecology 2021, 102, e03359. [Google Scholar] [CrossRef]
- Hadrava, J.; Talašová, A.; Straka, J.; Benda, D.; Kazda, J.; Klečka, J. A comparison of wild bee communities in sown flower strips and semi-natural habitats: A pollination network approach. Insect Conserv. Divers. 2022, 15, 312–324. [Google Scholar] [CrossRef]
- Sagot, P.; Borrell, E.V.; Mérida-Rivas, J.A. Abejas y agricultura: Cuando la diversidad es necesidad. Ecofronteras 2021, 25, 10–13. [Google Scholar]
- Orr, M.C.; Hughes, A.C.; Chesters, D.; Pickering, J.; Zhu, C.; Ascher, J.S. Global Patterns and Drivers of Bee Distribution. Curr. Biol. 2021, 31, 451–458. [Google Scholar] [CrossRef]
- Danforth, B.N.; Minckley, R.L.; Neff, J.L.; Fawcett, F. The Solitary Bees: Biology, Evolution, Conservation; Princeton University Press: Princeton, NJ, USA, 2019. [Google Scholar]
- Potts, S.; Imperatriz-Fonseca, V.; Ngo, H.; Biesmeijer, J.; Breeze, T.; Dicks, L.; Garibaldi, L.; Hill, R.; Settele, J.; Vanbergen, A.; et al. Summary for Policymakers of the Assessment Report of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services on Pollinators, Pollination and Food Production; Secretariat of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services: Bonn, Germany, 2016; p. 36. [Google Scholar]
- FAO. Blending Tradition and Science to Protect Pollinators. Available online: http://www.fao.org/in-action/blending-tradition-and-science-toprotect-pollinators/es/ (accessed on 18 February 2017).
- Wagner, D.L.; Grames, E.M.; Forister, M.L.; Berenbaum, M.R.; Stopak, D. Insect decline in the Anthropocene: Death by a thousand cuts. Proc. Natl. Acad. Sci. USA 2021, 118, e2023989118. [Google Scholar] [CrossRef]
- IUCN. The ICN Red List of Threatened Species. Version 2022-1. Available online: https://www.iucnredlist.org. (accessed on 29 September 2022).
- Hristov, P.; Neov, B.; Shumkova, R.; Palova, N. Significance of Apoidea as main pollinators. ecological and economic impact and implications for human nutrition. Diversity 2020, 12, 280. [Google Scholar] [CrossRef]
- Vasiliev, D.; Greenwood, S. Pollinator biodiversity and crop pollination in temperate ecosystems, implications for national pollinator conservation strategies: Mini review. Sci. Total Environ. 2020, 744, 140880. [Google Scholar] [CrossRef]
- Patel, S. Reviewing the prospects of Opuntia pears as low cost functional foods. Rev. Environ. Sci. 2013, 12, 223–234. [Google Scholar] [CrossRef]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global pollinator declines: Trends, impacts and drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef]
- Winfree, R.; Bartomeus, I.; Cariveau, D.P. Native pollinators in anthropogenic habitats. Annu. Rev. Ecol. Evol. Syst. 2011, 42, 1–22. [Google Scholar] [CrossRef]
- Lasway, J.V.; Steffan-Dewenter, I.; Njovu, H.K.; Kinabo, N.R.; Eardley, C.; Pauly, A.; Peters, M.K. Positive effects of low grazing intensity on East African bee assemblages mediated by increases in floral resources. Biol. Conserv. 2022, 267, 109–490. [Google Scholar] [CrossRef]
- Goulson, D.; Lye, G.C.; Darvill, B. Decline and conservation of bumble bees. Annu. Rev. Entomol. 2008, 53, 191–208. [Google Scholar] [CrossRef] [PubMed]
- Cane, J.H.; Minckley, R.L.; Kervin, L.J.; Roulston, T.A.H.; Williams, N.M. Complex responses within a desert bee guild (Hymenoptera: Apiformes) to urban habitat fragmentation. Ecol. Appl. 2006, 16, 632–644. [Google Scholar] [CrossRef] [PubMed]
- Medina-Flores, C.A.; Guzmán-Novoa, E.; Hamiduzzaman, M.M.; Aréchiga-Flores, C.F.; López-Carlos, M.A. Africanized honey bees (Apis mellifera) have low infestation levels of the mite Varroa destructor in different ecological regions in Mexico. Genet. Mol. Res. 2014, 13, 7282–7293. [Google Scholar] [CrossRef] [PubMed]
- Valido, A.; Rodríguez-Rodríguez, M.C.; Jordano, P. Interacciones entre plantas y polinizadores en el Parque Nacional del Teide: Consecuencias ecológicas de la introducción masiva de la abeja doméstica, Apis mellifera (Apidae). In Proyectos de Investigacion en Parques Nacionales; Sanz, L.R., Nistal, B.A., Eds.; Organismo Autónomo Parques Nacionales: Madrid, Spain, 2011; pp. 205–232. [Google Scholar]
- Martin-Culma, N.Y.; Arenas-Suárez, N.E.A.-S.E. Daño colateral en abejas por la exposición a pesticidas de uso agrícola. Entramado 2018, 14, 232–240. [Google Scholar] [CrossRef]
- Drossart, M.; Gérard, M. Beyond the decline of wild bees: Optimizing conservation measures and bringing together the actors. Insects 2020, 11, 649. [Google Scholar] [CrossRef]
- Krebs, C.J. Ecological Methodology; Harper & Row: New York, NY, USA, 1989; p. 739. [Google Scholar]
- Caswell, H. Matrix Population Models: Construction, Analysis, and Interpretation, 2nd ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2001. [Google Scholar]
- Gotelli, N.J.; Colwell, R.K. Estimating Species Richness. In Biological Diversity: Frontiers in Measurement and Assessment; Oxford University Press: Oxford, UK, 2011; pp. 39–54. [Google Scholar]
- White, E.R. Minimum Time Required to Detect Population Trends: The Need for Long-Term Monitoring Programs. BioScience 2019, 69, 40–46. [Google Scholar] [CrossRef]
- García, E. Modificaciones al Sistema de Clasificación Climática de Köppen; Universidad Nacional Autónoma de México (UNAM): Mexico City, Mexico, 2004; p. 90. [Google Scholar]
- INEGI. Cuaderno Estadístico Municipal Cadereyta de Montes; Querétaro de Arteaga: Aguascalientes, Mexico, 2000. [Google Scholar]
- de la Cruz, Y.H.U.; Aguilar, B.M.; Martínez, E.S.; Carrillo-Ángeles, I.; Martínez, M.M.H.; Vázquez, H.G.A. Estrategias de Conservación in situ para Rehabilitar los Paisajes del Semidesierto Queretano-Hidalguense; Boletín Amaranto, Asociación Mexicana de Jardines Botánicos: Querétaro, Mexico, 2018; p. 33. [Google Scholar]
- Hailen, U.d.l.C.; Maruri, A.B.; Carrillo, A.I.G.; Sánchez, E. Estrategias para la restauración con un enfoque agroforestal de áreas degradadas circunscritas por zona súrbanas en la región semiárida de Querétaro. Nthe 2004, 8, 3–9. [Google Scholar]
- CEA-CONCYTEQ, W.E. Forecast for Cadereyta, Queretaro. Available online: https://www.wunderground.com/dashboard/pws/IQUERETA15 (accessed on 25 November 2020).
- Ramírez-Freire, L.; Alanís Flores, G.; Ayala Barajas, R.; Velazco Macías, C.; Favela Lara, S. El uso de platos trampa y red entomológica en la captura de abejas nativas en el estado de Nuevo León, México. Acta Zool. Mex. 2014, 30, 508–538. [Google Scholar] [CrossRef]
- Fortel, L.; Henry, M.; Guilbaud, L.; Guirao, A.L.; Kuhlmann, M.; Mouret, H.; Rollin, O.; Vaissière, B.E. Decreasing Abundance, Increasing Diversity and Changing Structure of the Wild Bee Community (Hymenoptera: Anthophila) along an Urbanization Gradient. PLoS ONE 2014, 9, e104679. [Google Scholar] [CrossRef]
- Schiestl, F.P.; Johnson, S.D. Pollinator-mediated evolution of floral signals. Trends Ecol. Evol. 2003, 28, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Vrdoljak, S.M.; Samways, M.J. Optimising coloured pan traps to survey flower visiting insects. J. Insect Conserv. 2012, 16, 345–354. [Google Scholar] [CrossRef]
- Shapiro, L.H.; Tepedino, V.J.; Minckley, R.L. Bowling for bees: Optimal sample number for “bee bowl” sampling transects. J. Insect Conserv. 2014, 18, 1105–1113. [Google Scholar] [CrossRef]
- Sibly, R.M.; Hone, J. Population growth rate and its determinants: An overview. Philos. Trans. R. Soc. B 2002, 357, 1153–1170. [Google Scholar] [CrossRef]
- Zar, J.H. Biostatistical Analysis; Prentis Hall: Englewood Cliffs, NJ, USA, 1999. [Google Scholar]
- Crawley, M. The R Book, 2nd ed.; John Wiley and Sons Ltd.: Sussex, UK, 2013. [Google Scholar]
- Ortiz Sánchez, F.J.; Aguirre Segura, A. Estructura y dinámica estacional de una comunidad de Apoidea (Hymenoptera) en Almería. EOS Rev. Esp. Entomol. 1991, 67, 3–22. [Google Scholar]
- Roubik, D.W. Ecological impact on native bees by the invasive Africanized honey bee. Acta Biol. Colomb. 2009, 14, 115–124. [Google Scholar]
- Granados-Sánchez, D.; Sánchez-González, A.; Granados Victorino, R.L.; Borja de la Rosa, A. Ecología de la vegetación del Desierto Chihuahuense. RCHSCFA 2011, 17, 111–130. [Google Scholar] [CrossRef]
- Minckley, R.L.; Cane, J.H.; Kervin, L.; Roulston, T. Spatial predictability and resource specialization of bees (Hymenoptera: Apoidea) at a superabundant, widespread resource. Biol. J. Linn. Soc. 1999, 67, 119–147. [Google Scholar] [CrossRef][Green Version]
- Packer, L.; Jessome, V.; Lockerbie, C.; Sampson, B. The phenology and social biology of four sweat bees in a marginal environment: Cape Breton Island. Can. J. Zool. 1989, 67, 2871–2877. [Google Scholar] [CrossRef]
- Neff, J.L.; Simpson, B.B. Partial bivoltinism in a ground-nesting bee: The biology of Diadasia rinconis in Texas (Hymenoptera, Anthophoridae). J. Kans. Entomol. Soc. 1992, 65, 377–392. [Google Scholar]
- Portman, Z.M.; Cariveau, D.P. The State of Bee Monitoring in the United States: A Call to Refocus Away From Bowl Traps and Towards More Effective Methods. Ann. Entomol. Soc. Am. 2020, 113, 337–342. [Google Scholar] [CrossRef]
- Riggs, E.L.; Baranski, C.; Schaetz, O.M.; Garrison, G.; Collazo, J.A.; Youngsteadt, E. Estimating bee abundance: Can mark-recapture methods validate common sampling protocols? Apidologie 2022, 53, 10. [Google Scholar] [CrossRef]
- Boyer, K.J.; Fragoso, F.P.; Dieterich Mabin, M.E.; Brunet, J. Netting and pan traps fail to identify the pollinator guild of an agricultural crop. Sci. Rep. 2020, 10, 13819. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, I. Population dynamics of the solitary digger bee Andrena vaga Panzer (Hymenoptera, Andrenidae) studied using mark-recapture and nest counts. Popul. Ecol. 2003, 45, 197–204. [Google Scholar] [CrossRef]
- Steffan-Dewenter, I.; Schiele, S. Nest-site fidelity, body weight and population size of the red mason bee, Osmia rufa (Hymenoptera: Megachilidae), evaluated by mark-recapture experiments. Entomol. Gen. 2004, 27, 123–131. [Google Scholar] [CrossRef]
- Michener, C.D. The Bees of the World, 2nd ed.; The Johns Hopkins University Press: Baltimore, MD, USA, 2007. [Google Scholar]
- Cano-Salgado, A.; Zavala-Hurtado, J.A.; Orozco-Segovia, A.; Valverde-Valdés, M.T.; Pérez-Rodríguez, P. Composición y abundancia del banco de semillas en una región semiárida del trópico mexicano: Patrones de variación espacial y temporal. Rev. Mex. Biodivers. 2012, 83, 437–446. [Google Scholar] [CrossRef]
- Carevic, F.S. Hacia una integración de los rasgos ecofisiológicos de las plantas para la conservación de especies en peligro en ecosistemas sometidos a estrés hídrico. Idesia (Arica) 2016, 34, 33–38. [Google Scholar] [CrossRef]
- Pickett, S.T.A.; Ostfeld, R.S.; Shachak, M.; Likens, G.E. The Ecological Basis of Conservation. Heterogeneity, Ecosystems and Biodiversity; Springer: New York, NY, USA, 1997. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).