Elucidating the Role of Honey Bees as Biomonitors in Environmental Health Research
Abstract
:Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Search Strategy
2.3. Literature Analysis
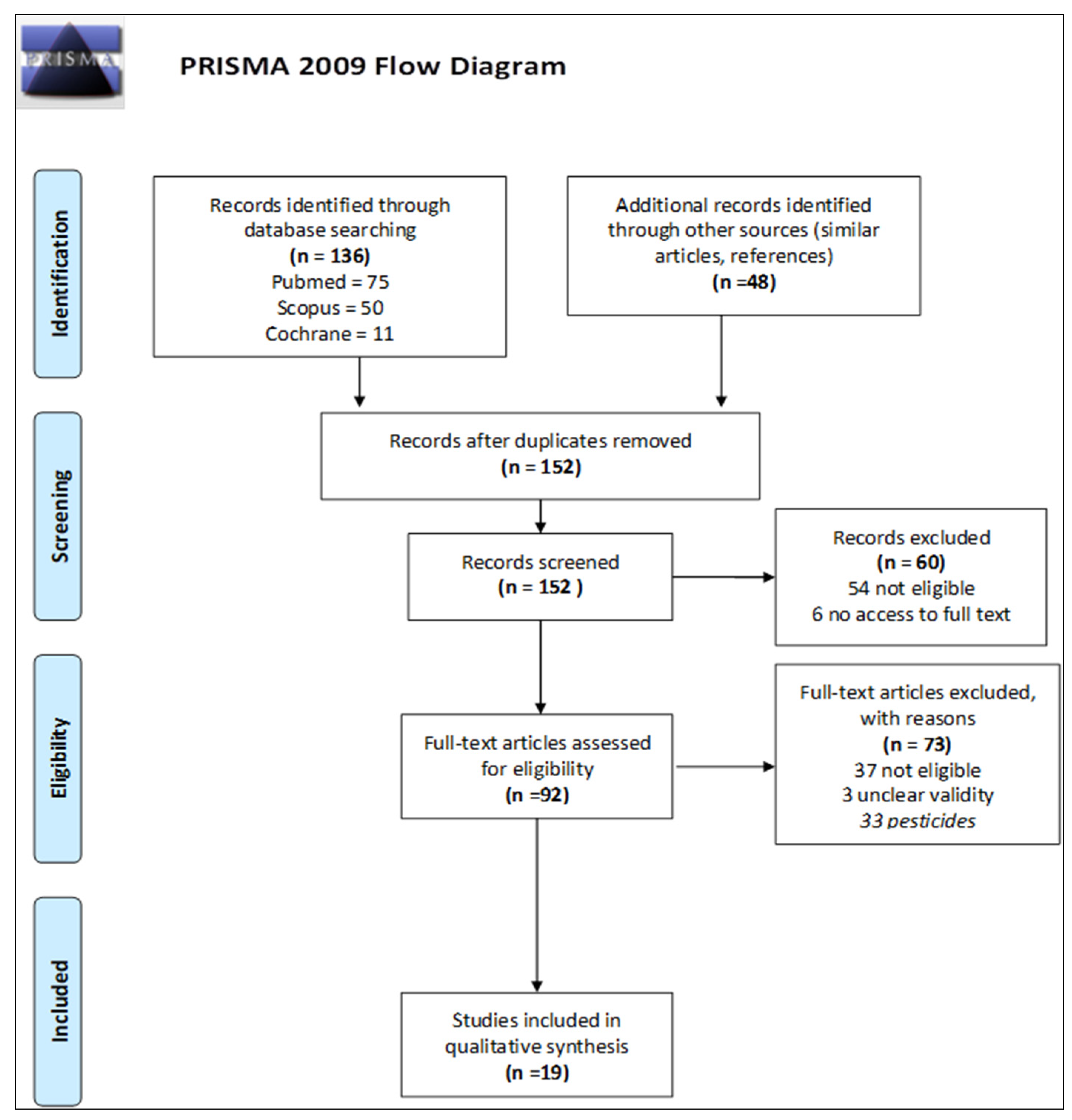
3. Results
3.1. Results of Systematic Literature Review
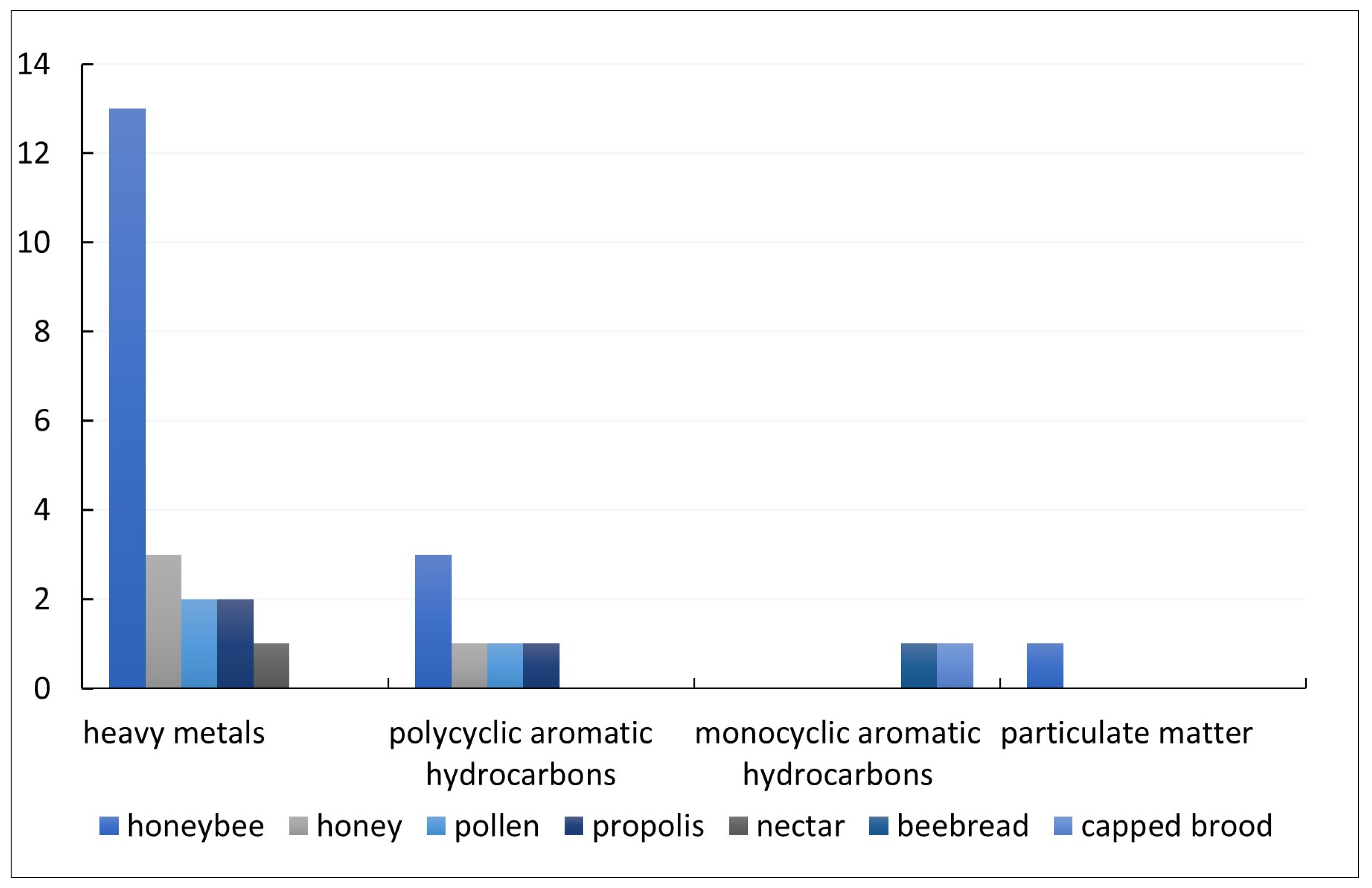
3.2. Characteristics of Reviewed Studies
3.3. Implications of Using Different Matrices
3.3.1. Foraging Honey Bees
3.3.2. Honey
3.3.3. Pollen
3.3.4. Propolis
3.3.5. Beebread
3.3.6. Capped Brood and Nectar
4. Discussion
Limitations and Perspectives for Further Research
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cohen, A.J.; Brauer, M.; Burnett, R.; Anderson, H.R.; Frostad, J.; Estep, K.; Balakrishnan, K.; Brunekreef, B.; Dandona, L.; Dandona, R.; et al. Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: An analysis of data from the Global Burden of Diseases Study 2015. Lancet 2017, 389, 1907–1918. [Google Scholar] [CrossRef] [PubMed]
- Filippelli, G.; Anenberg, S.; Taylor, M.; van Geen, A.; Khreis, H. New Approaches to Identifying and Reducing the Global Burden of Disease From Pollution. Geohealth 2020, 4, e2018GH000167. [Google Scholar] [CrossRef] [PubMed]
- Al-Alam, J.; Chbani, A.; Faljoun, Z.; Millet, M. The use of vegetation, bees, and snails as important tools for the biomonitoring of atmospheric pollution-a review. Environ. Sci. Pollut. Res. Int. 2019, 26, 9391–9408. [Google Scholar] [CrossRef] [PubMed]
- Badiou-Beneteau, A.; Carvalho, S.M.; Brunet, J.L.; Carvalho, G.A.; Bulete, A.; Giroud, B.; Belzunces, L.P. Development of biomarkers of exposure to xenobiotics in the honey bee Apis mellifera: Application to the systemic insecticide thiamethoxam. Ecotoxicol. Environ. Saf. 2012, 82, 22–31. [Google Scholar] [CrossRef]
- Porrini, C.; Ghini, S.; Girotti, S.; Sabatini, A.G.; Gattavecchia, E.; Celli, G.B.; Devillers, J.; Pham-Delègue, M.-H. Use of Honey Bees as Bioindicators of Environmental Pollution in Italy; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Molzahn, D.; Assmann-Werthmüller, U. Caesium radioactivity in several selected species of honey. Sci. Total Environ. 1993, 130–131, 95–108. [Google Scholar] [CrossRef]
- Barker, R.J.; Lehner, Y.; Kunzman, M.R. Pesticides and honey bees: Nectar and pollen contamination in alfalfa treated with dimethoate. Arch. Environ. Contam. Toxicol. 1980, 9, 125–133. [Google Scholar] [CrossRef]
- Bunzl, K.; Kracke, W.; Vorwohl, G. Transfer of Chernobyl-derived 134Cs, 137Cs, 131I and 103Ru from flowers to honey and pollen. J. Environ. Radioact. 1988, 6, 261–269. [Google Scholar] [CrossRef]
- Ford, B.C.; Jester, W.A.; Griffith, S.M.; Morse, R.A.; Zall, R.R.; Burgett, D.M.; Bodyfelt, F.W.; Lisk, D.J. Cesium-134 and cesium-137 in honey bees and cheese samples collected in the U.S. after the chernobyl accident. Chemosphere 1988, 17, 1153–1157. [Google Scholar] [CrossRef]
- Perugini, M.; Manera, M.; Grotta, L.; Abete, M.C.; Tarasco, R.; Amorena, M. Heavy metal (Hg, Cr, Cd, and Pb) contamination in urban areas and wildlife reserves: Honeybees as bioindicators. Biol. Trace Elem. Res. 2011, 140, 170–176. [Google Scholar] [CrossRef]
- Martinello, M.; Manzinello, C.; Borin, A.; Avram, L.E.; Dainese, N.; Giuliato, I.; Gallina, A.; Mutinelli, F. A Survey from 2015 to 2019 to Investigate the Occurrence of Pesticide Residues in Dead Honeybees and Other Matrices Related to Honeybee Mortality Incidents in Italy. Diversity 2020, 12, 15. [Google Scholar] [CrossRef]
- Crane, E. On the scientific front. Bee World 1984, 65, 47–49. [Google Scholar] [CrossRef]
- Devillers, J. The ecological importance of honey bees and their relevance to ecotoxicology. In Honey Bees: Estimating the Environmental Impact of Chemicals; Taylor and Francis: London, UK, 2002; Chapter 1; pp. 1–11. [Google Scholar]
- Negri, I.; Mavris, C.; Di Prisco, G.; Caprio, E.; Pellecchia, M. Honey Bees (Apis mellifera, L.) as Active Samplers of Airborne Particulate Matter. PLoS ONE 2015, 10, e0132491. [Google Scholar] [CrossRef]
- Celli, G.; Maccagnani, B. Honey bees as bioindicators of environmental pollution. Bull. Insectology 2003, 56, 137–139. [Google Scholar]
- Satta, A.; Verdinelli, M.; Ruiu, L.; Buffa, F.; Salis, S.; Sassu, A.; Floris, I. Combination of beehive matrices analysis and ant biodiversity to study heavy metal pollution impact in a post-mining area (Sardinia, Italy). Environ. Sci. Pollut. Res. Int. 2012, 19, 3977–3988. [Google Scholar] [CrossRef]
- Leita, L.; Muhlbachova, G.; Cesco, S.; Barbattini, R.; Mondini, C. Investigation of the use of honey bees and honey bee products to assess heavy metals contamination. Environ. Monit. Assess. 1996, 43, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lerner, H.; Berg, C. The concept of health in One Health and some practical implications for research and education: What is One Health? Infect. Ecol. Epidemiol. 2015, 5, 25300. [Google Scholar] [CrossRef] [PubMed]
- de Jongh, E.J.; Harper, S.L.; Yamamoto, S.S.; Wright, C.J.; Wilkinson, C.W.; Ghosh, S.; Otto, S.J.G. One Health, One Hive: A scoping review of honey bees, climate change, pollutants, and antimicrobial resistance. PLoS ONE 2022, 17, e0242393. [Google Scholar] [CrossRef]
- Scimago Journal & Country Rank. Available online: http://www.scimagojr.com (accessed on 7 November 2023).
- Thomas, J.K.D.; McKenzie, J.E.; Brennan, S.E.; Bhaumik, S. Cochrane Handbook for Systematic Reviews of Interventions Version 6.2; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; Cochrane: London, UK, 2021. [Google Scholar]
- Bilotta, G.S.; Milner, A.M.; Boyd, I.L. Quality assessment tools for evidence from environmental science. Environ. Evid. 2014, 3, 14. [Google Scholar] [CrossRef]
- Macura, B.; Byström, P.; Airoldi, L.; Eriksson, B.K.; Rudstam, L.; Støttrup, J.G. Impact of structural habitat modifications in coastal temperate systems on fish recruitment: A systematic review. Environ. Evid. 2019, 8, 14. [Google Scholar] [CrossRef]
- Drucker, A.M.; Fleming, P.; Chan, A.W. Research Techniques Made Simple: Assessing Risk of Bias in Systematic Reviews. J. Invest. Dermatol. 2016, 136, 109–114. [Google Scholar] [CrossRef]
- Al-Alam, J.; Fajloun, Z.; Chbani, A.; Millet, M. Determination of 16 PAHs and 22 PCBs in honey samples originated from different region of Lebanon and used as environmental biomonitors sentinel. J. Environ. Sci. Health A Tox. Hazard. Subst. Environ. Eng. 2019, 54, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Khalifa, M.H.; Aly, G.F.; Abdelhameed, K.M.A. Heavy Metal Accumulation and the Possible Correlation with Acetylcholinesterase Levels in Honey Bees from Polluted Areas of Alexandria, Egypt. Afr. Entomol. 2020, 28, 385–393. [Google Scholar] [CrossRef]
- Taha, E.K.A.; Al-Jabr, A.M.; Al-Kahtani, S.N. Honey Bees, Bee-collected Pollen and Honey as Monitors of Environmental Pollution at an Industrial Cement Area in Saudi Arabia. J. Kans. Entomol. Soc. 2017, 90, 1–10. [Google Scholar] [CrossRef]
- Zhou, X.; Taylor, M.P.; Davies, P.J.; Prasad, S. Identifying Sources of Environmental Contamination in European Honey Bees (Apis mellifera) Using Trace Elements and Lead Isotopic Compositions. Environ. Sci. Technol. 2018, 52, 991–1001. [Google Scholar] [CrossRef]
- Morgano, M.A.; Teixeira Martins, M.C.; Rabonato, L.C.; Milani, R.F.; Yotsuyanagi, K.; Rodriguez-Amaya, D.B. Inorganic contaminants in bee pollen from southeastern Brazil. J. Agric. Food Chem. 2010, 58, 6876–6883. [Google Scholar] [CrossRef]
- Kazazic, M.; Djapo-Lavic, M.; Mehic, E.; Jesenkovic-Habul, L. Monitoring of honey contamination with polycyclic aromatic hydrocarbons in Herzegovina region. Chem. Ecol. 2020, 36, 726–732. [Google Scholar] [CrossRef]
- Calatayud-Vernich, P.; Calatayud, F.; Simo, E.; Pico, Y. Efficiency of QuEChERS approach for determining 52 pesticide residues in honey and honey bees. MethodsX 2016, 3, 452–458. [Google Scholar] [CrossRef]
- Lambert, O.; Veyrand, B.; Durand, S.; Marchand, P.; Le Bizec, B.; Piroux, M.; Puyo, S.; Thorin, C.; Delbac, F.; Pouliquen, H. Polycyclic aromatic hydrocarbons: Bees, honey and pollen as sentinels for environmental chemical contaminants. Chemosphere 2012, 86, 98–104. [Google Scholar] [CrossRef]
- Lambert, O.; Piroux, M.; Puyo, S.; Thorin, C.; Larhantec, M.; Delbac, F.; Pouliquen, H. Bees, honey and pollen as sentinels for lead environmental contamination. Environ. Pollut. 2012, 170, 254–259. [Google Scholar] [CrossRef]
- van der Steen, J.J.; de Kraker, J.; Grotenhuis, T. Spatial and temporal variation of metal concentrations in adult honeybees (Apis mellifera L.). Environ. Monit. Assess. 2012, 184, 4119–4126. [Google Scholar] [CrossRef]
- Badiou-Beneteau, A.; Benneveau, A.; Geret, F.; Delatte, H.; Becker, N.; Brunet, J.L.; Reynaud, B.; Belzunces, L.P. Honeybee biomarkers as promising tools to monitor environmental quality. Environ. Int. 2013, 60, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Ruschioni, S.; Riolo, P.; Minuz, R.L.; Stefano, M.; Cannella, M.; Porrini, C.; Isidoro, N. Biomonitoring with honeybees of heavy metals and pesticides in nature reserves of the Marche Region (Italy). Biol. Trace Elem. Res. 2013, 154, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, M.; Molero, R.; Gaju, M.; van der Steen, J.J.; Porrini, C.; Ruiz, J.A. Assessment of heavy metal pollution in Cordoba (Spain) by biomonitoring foraging honeybee. Environ. Monit. Assess. 2015, 187, 651. [Google Scholar] [CrossRef] [PubMed]
- Matin, G.; Kargar, N.; Buyukisik, H.B. Bio-monitoring of cadmium, lead, arsenic and mercury in industrial districts of Izmir, Turkey by using honey bees, propolis and pine tree leaves. Ecol. Eng. 2016, 90, 331–335. [Google Scholar] [CrossRef]
- van der Steen, J.J.; Cornelissen, B.; Blacquiere, T.; Pijnenburg, J.E.; Severijnen, M. Think regionally, act locally: Metals in honeybee workers in the Netherlands (surveillance study 2008). Environ. Monit. Assess. 2016, 188, 463. [Google Scholar] [CrossRef]
- Zarić, N.M.; Ilijević, K.; Stanisavljević, L.; Gržetić, I. Metal concentrations around thermal power plants, rural and urban areas using honeybees (Apis mellifera L.) as bioindicators. Int. J. Environ. Sci. Technol. 2016, 13, 413–422. [Google Scholar] [CrossRef]
- Giglio, A.; Ammendola, A.; Battistella, S.; Naccarato, A.; Pallavicini, A.; Simeon, E.; Tagarelli, A.; Giulianini, P.G. Apis mellifera ligustica, Spinola 1806 as bioindicator for detecting environmental contamination: A preliminary study of heavy metal pollution in Trieste, Italy. Environ. Sci. Pollut. Res. Int. 2017, 24, 659–665. [Google Scholar] [CrossRef]
- Kargar, N.; Matin, G.; Matin, A.A.; Buyukisik, H.B. Biomonitoring, status and source risk assessment of polycyclic aromatic hydrocarbons (PAHs) using honeybees, pine tree leaves, and propolis. Chemosphere 2017, 186, 140–150. [Google Scholar] [CrossRef]
- Zarić, N.M.; Ilijević, K.; Stanisavljević, L.; Grzetic, I. Use of honeybees (Apis mellifera L.) as bioindicators for assessment and source appointment of metal pollution. Environ. Sci. Pollut. Res. Int. 2017, 24, 25828–25838. [Google Scholar] [CrossRef]
- Gutierrez, M.; Molero, R.; Gaju, M.; van der Steen, J.J.; Porrini, C.; Ruiz, J.A. Assessing heavy metal pollution by biomonitoring honeybee nectar in Cordoba (Spain). Environ. Sci. Pollut. Res. Int. 2020, 27, 10436–10448. [Google Scholar] [CrossRef]
- Zięba, K.; Szostak, E.; Czekońska, K.; Miśkowiec, P.; Moos-Matysik, A.; Nyczyk-Malinowska, A.; Szentgyörgyi, H. Usefulness of bee bread and capped brood for the assessment of monocyclic aromatic hydrocarbon levels in the environment. Environ. Pollut. 2020, 265 Pt A, 114882. [Google Scholar] [CrossRef]
- Cochard, P.; Laurie, M.; Veyrand, B.; Le Bizec, B.; Poirot, B.; Marchand, P. PAH7 concentration reflects anthropization: A study using environmental biomonitoring with honeybees. Sci. Total Environ. 2021, 751, 141831. [Google Scholar] [CrossRef] [PubMed]
- Ilijević, K.; Vujanovic, D.; Orcic, S.; Purac, J.; Kojic, D.; Zarić, N.M.; Gržetić, I.; Blagojevic, D.P.; Celic, T.V. Anthropogenic influence on seasonal and spatial variation in bioelements and non-essential elements in honeybees and their hemolymph. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2021, 239, 108852. [Google Scholar] [CrossRef]
- Amorena, M.; Visciano, P.; Giacomelli, A.; Marinelli, E.; Sabatini, A.G.; Medrzycki, P.; Oddo, L.P.; De Pace, F.M.; Belligoli, P.; Di Serafino, G.; et al. Monitoring of levels of polycyclic aromatic hydrocarbons in bees caught from beekeeping: Remark 1. Vet. Res. Commun. 2009, 33, 165–167. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.C. Honey as an indicator of heavy metal contamination. Water Air Soil. Pollut. 1987, 33, 179–189. [Google Scholar] [CrossRef]
- Peng, Y.S.; Marston, J.M. Filtering mechanism of the honey bee proventriculus. Physiol. Entomol. 1986, 11, 433–439. [Google Scholar] [CrossRef]
- Dżugan, M.; Zaguła, G.; Wesołowska, M.; Sowa, P.; Puchalski, C. Levels of toxic and essential metals in varietal honeys from podkarpacie. J. Elem. 2017, 22, 1039–1048. [Google Scholar] [CrossRef]
- Squadrone, S.; Brizio, P.; Stella, C.; Pederiva, S.; Brusa, F.; Mogliotti, P.; Garrone, A.; Abete, M.C. Trace and rare earth elements in monofloral and multifloral honeys from Northwestern Italy; A first attempt of characterization by a multi-elemental profile. J. Trace Elem. Med. Biol. 2020, 61, 126556. [Google Scholar] [CrossRef]
- Tuzen, M.; Silici, S.; Mendil, D.; Soylak, M. Trace element levels in honeys from different regions of Turkey. Food Chem. 2007, 103, 325–330. [Google Scholar] [CrossRef]
- Raimets, R.; Bontšutšnaja, A.; Bartkevics, V.; Pugajeva, I.; Kaart, T.; Puusepp, L.; Pihlik, P.; Keres, I.; Viinalass, H.; Mänd, M.; et al. Pesticide residues in beehive matrices are dependent on collection time and matrix type but independent of proportion of foraged oilseed rape and agricultural land in foraging territory. Chemosphere 2020, 238, 124555. [Google Scholar] [CrossRef]
- Perugini, M.; Di Serafino, G.; Giacomelli, A.; Medrzycki, P.; Sabatini, A.G.; Persano Oddo, L.; Marinelli, E.; Amorena, M. Monitoring of polycyclic aromatic hydrocarbons in bees (Apis mellifera) and honey in urban areas and wildlife reserves. J. Agric. Food Chem. 2009, 57, 7440–7444. [Google Scholar] [CrossRef] [PubMed]
- Pusceddu, M.; Annoscia, D.; Floris, I.; Frizzera, D.; Zanni, V.; Angioni, A.; Satta, A.; Nazzi, F. Honeybees use propolis as a natural pesticide against their major ectoparasite. Proc. R. Soc. B Biol. Sci. 2021, 288, 20212101. [Google Scholar] [CrossRef] [PubMed]
- Finger, D.; Filho, I.K.; Torres, Y.R.; Quinaia, S.P. Propolis as an indicator of environmental contamination by metals. Bull. Environ. Contam. Toxicol. 2014, 92, 259–264. [Google Scholar] [CrossRef] [PubMed]
- González-Martín, M.I.; Revilla, I.; Betances-Salcedo, E.V.; Vivar-Quintana, A.M. Pesticide residues and heavy metals in commercially processed propolis. Microchem. J. 2018, 143, 423–429. [Google Scholar] [CrossRef]
- Moret, S.; Purcaro, G.; Conte, L.S. Polycyclic aromatic hydrocarbons (PAHs) levels in propolis and propolis-based dietary supplements from the Italian market. Food Chem. 2010, 122, 333–338. [Google Scholar] [CrossRef]
- Mullin, C.A.; Frazier, M.; Frazier, J.L.; Ashcraft, S.; Simonds, R.; Vanengelsdorp, D.; Pettis, J.S. High levels of miticides and agrochemicals in North American apiaries: Implications for honey bee health. PLoS ONE 2010, 5, e9754. [Google Scholar] [CrossRef]
- Giroud, B.; Vauchez, A.; Vulliet, E.; Wiest, L.; Bulete, A. Trace level determination of pyrethroid and neonicotinoid insecticides in beebread using acetonitrile-based extraction followed by analysis with ultra-high-performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2013, 1316, 53–61. [Google Scholar] [CrossRef]
- Fismes, J.; Perrin-Ganier, C.; Empereur-Bissonnet, P.; Morel, J.L. Soil-to-root transfer and translocation of polycyclic aromatic hydrocarbons by vegetables grown on industrial contaminated soils. J. Environ. Qual. 2002, 31, 1649–1656. [Google Scholar] [CrossRef]
- Evans, D.E.; Taylor, P.E.; Singh, M.B.; Knox, R.B. Quantitative analysis of lipids and protein from the pollen of Brassica napus L. Plant Sci. 1991, 73, 117–126. [Google Scholar] [CrossRef]
- Chen, S.J.; Wang, J.; Wang, T.; Wang, T.; Mai, B.X.; Simonich, S.L.M. Seasonal variations and source apportionment of complex polycyclic aromatic hydrocarbon mixtures in particulate matter in an electronic waste and urban area in South China. Sci. Total Environ. 2016, 573, 115–122. [Google Scholar] [CrossRef]
- Smith, K.E.C.; Thomas, G.O.; Jones, K.C. Seasonal and Species Differences in the Air−Pasture Transfer of PAHs. Environ. Sci. Technol. 2001, 35, 2156–2165. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.; Jeong, S.; Lee, D.; Kim, S.W.; Park, R.J.; Gibson, L.; Zheng, C.; Park, C.R. Foraging trip duration of honeybee increases during a poor air quality episode and the increase persists thereafter. Ecol. Evol. 2021, 11, 1492–1500. [Google Scholar] [CrossRef] [PubMed]
| Authors, Publication Year, Reference | Article Title | Journal |
|---|---|---|
| Perugini et al., 2011 [10] | Heavy Metal (Hg, Cr, Cd, and Pb) Contamination in Urban Areas and Wildlife Reserves: Honey bees as Bioindicators | Biological Trace Element Research |
| Lambert et al., 2012 [32] a | Bees, Honey and Pollen as Sentinels for Lead Environmental Contamination | Environmental Pollution |
| Lambert et al., 2012 [33] b | Polycyclic Aromatic Hydrocarbons: Bees, Honey and Pollen as Sentinels for Environmental Chemical Contaminants | Chemosphere |
| Satta et al., 2012 [16] | Combination of Beehive Matrices Analysis and Ant Biodiversity to Study Heavy Metal Pollution Impact in a Post-Mining Area (Sardinia, Italy) | Environmental Science and Pollution Research |
| van der Steen et al., 2012 [34] | Spatial and Temporal Variation of Metal Concentrations in Adult Honey Bees (Apis mellifera L.) | Environmental Monitoring and Assessment |
| Badiou-Beneteau et al., 2013 [35] | Honey Bee Biomarkers as Promising Tools to Monitor Environmental Quality | Environment International |
| Ruschioni et al., 2013 [36] | Biomonitoring with Honey bees of Heavy Metals and Pesticides in Nature Reserves of the Marche Region (Italy) | Biological Trace Element Research |
| Gutierrez et al., 2015 [37] | Assessment of Heavy Metal Pollution in Cordoba (Spain) by Biomonitoring Foraging Honey Bee | Environmental Monitoring and Assessment |
| Negri et al., 2015 [14] | Honey Bees (Apis mellifera, L.) as Active Samplers of Airborne Particulate Matter | PLOS One |
| Matin et al., 2016 [38] | Bio-Monitoring of Cadmium, Lead, Arsenic and Mercury in Industrial Districts of Izmir, Turkey by Using Honey Bees, Propolis and Pine Tree Leaves | Ecological Engineering |
| van der Steen et al., 2016 [39] | Think Regionally, Act Locally: Metals in Honey Bee Workers in The Netherlands (Surveillance Study 2008) | Environmental Monitoring and Assessment |
| Zarić et al., 2016 [40] | Metal Concentrations Around Thermal Power Plants, Rural and Urban Areas Using Honey Bees (Apis mellifera L.) as Bioindicators | International Journal of Environmental Science and Technology |
| Giglio et al., 2017 [41] | Apis mellifera ligustica, Spinola 1806 as Bioindicator for Detecting Environmental Contamination: A Preliminary Study of Heavy Metal Pollution in Trieste, Italy | Environmental Science and Pollution Research |
| Kargar et al., 2017 [42] | Biomonitoring, Status and Source Risk Assessment of Polycyclic Aromatic Hydrocarbons (Pahs) Using Honey Bees, Pine Tree Leaves, and Propolis | Chemosphere |
| Zarić et al., 2017 [43] | Use of Honey Bees (Apis mellifera L.) as Bioindicators for Assessment and Source Appointment of Metal Pollution | Environmental Science and Pollution Research |
| Gutierrez et al., 2020 [44] | Assessing Heavy Metal Pollution by Biomonitoring Honey Bee Nectar in Cordoba (Spain) | Environmental Science and Pollution Research |
| Zieba et al., 2020 [45] | Usefulness of Bee Bread and Capped Brood for the Assessment of Monocyclic Aromatic Hydrocarbon Levels in the Environment | Environmental Pollution |
| Cochard et al., 2021 [46] | PAH7 Concentration Reflects Anthropization: A Study Using Environmental Biomonitoring with Honey Bees | Science of the Total Environment |
| Ilijević et al., 2021 [47] | Anthropogenic Influence on Seasonal and Spatial Variation in Bioelements and Non-Essential Elements in Honey Bees and their Hemolymph | Comparative Biochemistry and Physiology C-Toxicology and Pharmacology |
| Study | Pollutant(s) | Matrices | Analytical Technique | Sampling (n) | ||
|---|---|---|---|---|---|---|
| Collection | Locations | Hives | ||||
| Badiou-Beneteau et al., 2013 [35] | HM | Honey bees | ICP-MS | 4 | 2 | 6 |
| Cochard et al., 2021 [32] | PAH | Honey bees | GC-MS/MS | 12 | 36 | 108 |
| Giglio et al., 2017 [41] | HM | Honey bees | ICP-MS | 1 | 2 | 2 |
| Gutierrez et al.2015 [37] | HM | Honey bees | ICP-OES | 20 | 5 | 10 |
| Gutierrez et al., 2020 [44] | HM | Nectar | AAS | 20 | 5 | 10 |
| Ilijević et al., 2021 [47] | HM | Honey bees (bucket collection) | ICP-OES+/−ICP-MS | 3 | 3 | 15 |
| Kargar et al., 2017 [42] | PAH | Honey bees Propolis (pine tree leaves) | GC | 1 | 5 | 5 |
| Lambert et al., 2012 a [32] | HM | Honey bees Honey Pollen | AAS | 8 | 18 | 144 |
| Lambert et al., 2012 b [33] | PAH | Honey bees Honey Pollen | GC/MS | 8 | 6 | 48 |
| Matin et al., 2016 [38] | HM | Honey bees Propolis (pine tree leaves) | EAS/FAS | 1 | 5 | 5 |
| Negri et al., 2015 [14] | PM; HM | Honey bees (sediment) | SEM-EDX | 1 | 4 | 11 |
| Perugini et al., 2011 [10] | HM | Honey bees | AAS | 6 | 8 | 24 |
| Ruschioni et al., 2013 [36] | HM | Honey bees Honey | ICP-AES/GC/MS | 15 | 11 | 22 |
| Satta et al., 2012 [16] | HM | Honey bees Honey Pollen Stream water Soil Ants | EAS | 12 | 3 | 9 |
| van der Steen et al., 2016 [39] | HM | Honey bees | ICP-MS | 1 | 9 | 750 |
| van der Steen et al., 2012 [34] | HM | Honey bees | ICP-AES | 6 | 3 | 9 |
| Zarić et al., 2017 [43] | HM | Honey bees (soil) | ICP-OES | 5 | 4 | 10 |
| Zarić et al., 2016 [40] | HM | Honey bees | ICP-OES | 2 | 6 | 13 |
| Zieba et al. 2020 [45] | BTEX | Bee bread Capped brood | GC-MS | 4 | 4 | 12 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mair, K.S.; Irrgeher, J.; Haluza, D. Elucidating the Role of Honey Bees as Biomonitors in Environmental Health Research. Insects 2023, 14, 874. https://doi.org/10.3390/insects14110874
Mair KS, Irrgeher J, Haluza D. Elucidating the Role of Honey Bees as Biomonitors in Environmental Health Research. Insects. 2023; 14(11):874. https://doi.org/10.3390/insects14110874
Chicago/Turabian StyleMair, Katharina Sophia, Johanna Irrgeher, and Daniela Haluza. 2023. "Elucidating the Role of Honey Bees as Biomonitors in Environmental Health Research" Insects 14, no. 11: 874. https://doi.org/10.3390/insects14110874
APA StyleMair, K. S., Irrgeher, J., & Haluza, D. (2023). Elucidating the Role of Honey Bees as Biomonitors in Environmental Health Research. Insects, 14(11), 874. https://doi.org/10.3390/insects14110874






