Fumigant Toxicity of Essential Oils against Frankliniella occidentalis and F. insularis (Thysanoptera: Thripidae) as Affected by Polymer Release and Adjuvants
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Test Organisms
2.2. Chemical Preparation
2.3. Thrips Bioassays
2.4. Phytotoxicity Assays
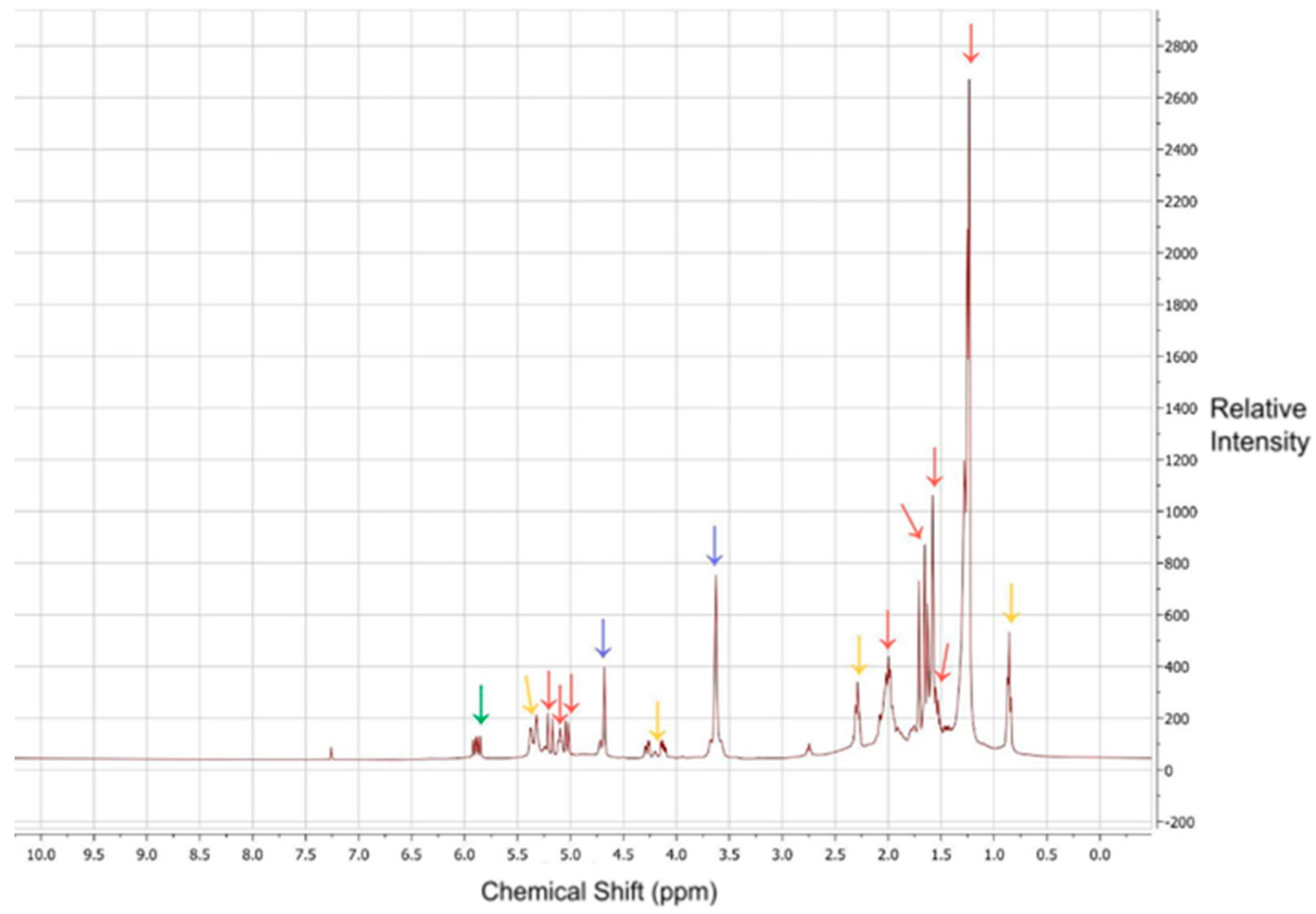
2.5. Nuclear Magnetic Resonance (NMR) Sample Preparation and Analysis
2.6. Statistical Analysis
3. Results
3.1. Hydrogel Saturation Tests
3.2. Adjuvant Tests
3.3. Phytotoxicity Assays
3.4. NMR Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Extension Entomology & UH-CTAHR Integrated Pest Management Program. Available online: http://www.extento.hawaii.edu/Kbase/crop/Type/f_occide.htm (accessed on 23 February 2022).
- Yudin, L.S.; Cho, J.J.; Mitchell, W.C. Host range of western flower thrips, Frankliniella occidentalis (Thysanoptera: Thripidae), with special reference to Leucaena glauca. Environ. Entomol. 1986, 15, 1292–1295. [Google Scholar] [CrossRef]
- Robb, K.L.; Newman, J.; Virzi, J.K.; Parrella, M.P. Insecticide resistance in western flower thrips. In Thrips Biology and Management; Springer: Boston, MA, USA, 1995; Volume 276, pp. 341–346. [Google Scholar] [CrossRef]
- Wijkamp, I.; van Lent, J.; Kormelink, R.; Goldbach, R.; Peters, D. Multiplication of tomato spotted wilt virus in its insect vector, Frankliniella occidentalis. J. Gen. Virol. 1993, 74, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Nagata, T.; Inoue-Nagata, A.K.; van Lent, J.; Goldbach, R.; Peters, D. Factors determining vector competence and specificity for transmission of tomato spotted wilt virus. J. Gen. Virol. 2002, 83, 663–671. [Google Scholar] [CrossRef]
- Goldbach, R.; Peters, D. Possible causes of the emergence of tospovirus diseases. Semin. Virol. 1994, 5, 113–120. [Google Scholar] [CrossRef]
- Stuart, R.R.; Gao, Y.; Lei, Z. Thrips: Pests of concern to China and the United States. Agric. Sci. China 2011, 10, 867–892. [Google Scholar] [CrossRef]
- Kirk, W.D.J.; Terry, L.I. The spread of the western flower thrips Frankliniella occidentalis (Pergande). Agric. For. Entomol. 2003, 5, 301–310. [Google Scholar] [CrossRef]
- Lima, É.F.B.; Monteiro, R.C.; Zucchi, R.A. Thrips species (Insecta: Thysanoptera) associated to Fabaceae of agricultural importance in Cerrado and Amazon-Caatinga ecotone from Brazilian mid-north. Biota Neotrop. 2013, 13, 283–289. [Google Scholar] [CrossRef][Green Version]
- Hoddle, M.S.; Mound, L.A. Thrips of California. Available online: https://keys.lucidcentral.org/keys/v3/thrips_of_california/authors/authors.html (accessed on 22 December 2021).
- Kaushik, P.; Kaushik, G. An assessment of structure and toxicity correlation in organochlorine pesticides. J. Hazard. Mater. 2007, 143, 102–111. [Google Scholar] [CrossRef]
- Sobti, R.C.; Krishan, A.; Pfaffenberger, C.D. Cytokinetic and cytogenetic effects of some agricultural chemicals on human lymphoid cells in vitro: Organophosphates. Mutat. Res. Genet. Toxicol. 1982, 102, 89–102. [Google Scholar] [CrossRef]
- Sandal, S.; Yilmaz, B. Genotoxic effects of chlorpyrifos, cypermethrin, endosulfan and 2,4-D on human peripheral lymphocytes cultured from smokers and nonsmokers. Environ. Toxicol. 2010, 26, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Lal, R.; Saxena, D.M. Accumulation, metabolism, and effects of organochlorine insecticides on microorganisms. Microbiol. Rev. 1982, 46, 95–127. [Google Scholar] [CrossRef] [PubMed]
- Riley, D.G.; Pappu, H.R. Evaluation of tactics for management of thrips-vectored tomato spotted wilt virus in tomato. Plant Dis. 2000, 84, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Fu, B.; Li, Q.; Qiu, H.; Tang, L.; Zeng, D.; Liu, K.; Gao, Y. Resistance development, stability, cross-resistance potential, biological fitness and biochemical mechanisms of spinetoram resistance in Thrips hawaiiensis (Thysanoptera: Thripidae). Pest Manag. Sci. 2018, 74, 1564–1574. [Google Scholar] [CrossRef]
- Kamatou, G.P.P.; Viljoen, A.M. Linalool—A review of a biologically active compound of commercial importance. Nat. Prod. Commun. 2008, 3, 1183–1187. [Google Scholar] [CrossRef]
- Stepanycheva, E.; Petrova, M.; Chermenskaya, T.; Pavela, R. Fumigant effect of essential oils on mortality and fertility of thrips Frankliniella occidentalis Perg. Environ. Sci. Pollut. Res. 2019, 26, 30885–30892. [Google Scholar] [CrossRef]
- Riefler, J.; Koschier, E.H. Behaviour-modifying activity of eugenol on Thrips tabaci Lindeman. J. Pest Sci. 2008, 82, 115–121. [Google Scholar] [CrossRef]
- Koschier, E.H.; Sedy, K.A.; Novak, J. Influence of plant volatiles on feeding damage caused by the onion thrips Thrips tabaci. Crop Prot. 2002, 21, 419–425. [Google Scholar] [CrossRef]
- Moretti, M.D.L.; Sanna-Passino, G.; Demontis, S.; Bazzoni, E. Essential oil formulations useful as a new tool for insect pest control. AAPS Pharm. Sci. Tech. 2002, 3, 64–74. [Google Scholar] [CrossRef]
- Stejskal, V.; Vendl, T.; Aulicky, R.; Athanassiou, C. Synthetic and natural insecticides: Gas, liquid, gel and solid formulations for stored-product and food-industry pest control. Insects 2021, 12, 590. [Google Scholar] [CrossRef]
- Janmaat, A.F.; de Kogel, W.J.; Woltering, E.J. Enhanced fumigant toxicity of p-cymene against Frankliniella occidentalis by simultaneous application of elevated levels of carbon dioxide. Pest Manag. Sci. 2002, 58, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Picard, I.; Hollingsworth, R.G.; Salmieri, S.; Lacroix, M. Repellency of essential oils to Frankliniella occidentalis (Thysanoptera: Thripidae) as affected by type of oil and polymer release. J. Econ. Entomol. 2012, 105, 1238–1247. [Google Scholar] [CrossRef]
- Rudzinski, W.E.; Dave, A.M.; Vaishnav, U.H.; Kumbar, S.G.; Kulkarni, A.R.; Aminabhavi, T.M. Hydrogels as controlled release devices in agriculture. Des. Monomers Polym. 2002, 5, 39–65. [Google Scholar] [CrossRef]
- Regnault-Roger, C.; Vincent, C.; Arnason, J.T. Essential oils in insect control: Low-risk products in a high-stakes world. Annu. Rev. Entomol. 2012, 57, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Tay, J.-W.; Hoddle, M.S.; Mulchandani, A.; Choe, D.-H. Development of an alginate hydrogel to deliver aqueous bait for pest ant management: Alginate hydrogel for ant management. Pest. Manag. Sci. 2017, 73, 2028–2038. [Google Scholar] [CrossRef]
- Tay, J.-W.; Choe, D.-H.; Mulchandani, A.; Rust, M.K. Hydrogels: From controlled release to a new bait delivery for insect pest management. J. Econ. Entomol. 2020, 113, 2061–2068. [Google Scholar] [CrossRef]
- Nicholas, A.H.; Follett, P.A. Postharvest irradiation treatment for quarantine control of western flower thrips (Thysanoptera: Thripidae). J. Econ. Entomol. 2018, 111, 1185–1189. [Google Scholar] [CrossRef]
- Hartzell, A.; Wilcoxon, F. Naphthalene fumigation at controlled concentrations 1, 2. J. Econ. Entomol. 1930, 23, 608–618. [Google Scholar] [CrossRef]
- IBM Corp. IBM SPSS Statistics for Windows; Version 27.0; IBM Corp.: Armonk, NY, USA, 2020. [Google Scholar]
- Finney, D.J. Probit analysis. J. Pharm. Sci. 1971, 60, 1432. [Google Scholar] [CrossRef]
- Chadwick, P.R. A comparison of safroxan and piperonyl butoxide as pyrethrum synergists. Pyrethrum Post 1961, 6, 30–37. [Google Scholar]
- Metcalf, R.L. Mode of action of insecticide synergists. Annu. Rev. Entomol. 1967, 12, 229–256. [Google Scholar] [CrossRef] [PubMed]
- Lordelo, M.M.; Davis, A.J.; Calhoun, M.C.; Dowd, M.K.; Dale, N.M. Relative toxicity of gossypol enantiomers in broilers. Poult. Sci. 2005, 84, 1376–1382. [Google Scholar] [CrossRef] [PubMed]
- Overmyer, J.P.; Rouse, D.R.; Avants, J.K.; Garrison, A.W.; DeLorenzo, M.E.; Chung, K.W.; Key, P.B.; Wilson, W.A.; Black, M.C. Toxicity of fipronil and its enantiomers to marine and freshwater non-targets. J. Environ. Sci. Health Part B 2007, 42, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Gan, J.J.; Qin, S. Separation and aquatic toxicity of enantiomers of synthetic pyrethroid insecticides. Chirality 2005, 17, S127–S133. [Google Scholar] [CrossRef]
- Sun, J.Q.; Tao, N.E.; Zhang, A.P.; Qi, H. Effects of cyclodextrin on the stereoselectivity inhibition of acetylcholinesterase by isomalathion. J. Environ. Sci. Health Part B 2018, 53, 580–586. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, N.; Li, L.; Zhang, Q.; Zhao, M.; Liu, W. Enantioselective interaction with acetylcholinesterase of an organophosphate insecticide fenamiphos. Chirality 2009, 22, 612–617. [Google Scholar] [CrossRef]
- Shaaya, E.; Rafaeli, A. Essential oils as biorational insecticides–potency and mode of action. In Insecticides Design Using Advanced Technologies; Ishaaya, I., Horowitz, A.R., Nauen, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2007; pp. 249–261. [Google Scholar] [CrossRef]
- Vicenço, C.B.; Silvestre, W.P.; Lima, T.S.; Pauletti, G.F. Insecticidal activity of Cinnamomum camphora Ness and Eberm var. linaloolifera Fujita leaf essential oil and linalool against Anticarsia gemmatalis. J. Essent. Oil Res. 2021, 33, 601–609. [Google Scholar] [CrossRef]
- Taktak, N.E.M.; Badawy, M.E.I. Potential of hydrocarbon and oxygenated monoterpenes against Culex pipiens larvae: Toxicity, biochemical, pharmacophore modeling and molecular docking studies. Pestic. Biochem. Physiol. 2019, 158, 156–165. [Google Scholar] [CrossRef]
- Farag, M.A.; Ezzat, S.M.; Salama, M.M.; Tadros, M.G.; Serya, R.A.T. Anti-acetylcholinesterase activity of essential oils and their major constituents from four Ocimum species. Z. Nat. C. J. Biosci. 2016, 71, 393–402. [Google Scholar] [CrossRef]
- Praveena, A.; Sanjayan, K.P. Inhibition of acetylcholinesterase in three insects of economic importance by linalool, a monoterpene phytochemical. Insect Pest Manag. Curr. Scenar. 2011, 1, 340–345. [Google Scholar]
- Agostini-Costa, T.d.S.; Vieira, R.F.; Bizzo, H.R.; Silveira, D.; Gimenes, M.A. Secondary metabolites. In Chromatography and Its Applications; InTech: Rijeka, Croatia, 2012. [Google Scholar] [CrossRef]
- López, M.D.; Pascual-Villalobos, M.J. Mode of inhibition of acetylcholinesterase by monoterpenoids and implications for pest control. Ind. Crops Prod. 2010, 31, 284–288. [Google Scholar] [CrossRef]
- Heimes, K.; Hauk, F.; Verspohl, E.J. Mode of action of peppermint oil and (−)-menthol with respect to 5-HT3 receptor subtypes: Binding studies, cation uptake by receptor channels and contraction of isolated rat ileum. Phytother. Res. 2010, 25, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Liu, J.; Weng, H.; Ma, Z.; Zhang, X. Efficacy of binary combinations between methyl salicylate and carvacrol against thrips Anaphothrips obscurus: Laboratory and field trials. Pest Manag. Sci. 2019, 76, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.L.; Cho, I.K.; Li, Q.X. Insecticidal activity of basil oil, trans-anethole, estragole, and linalool to adult fruit flies of Ceratitis capitata, Bactrocera dorsalis, and Bactrocera cucurbitae. J. Econ. Entomol. 2009, 102, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Salvatella, L. The alkyl group is a –I + R substituent. Educ. Química 2017, 28, 232–237. [Google Scholar] [CrossRef]
- Organic Chemistry Online Learning Center. Available online: https://www.chem.ucalgary.ca/courses/351/Carey5th/Ch10/ch10-2.html (accessed on 22 February 2022).
- Chemistry Libretexts. Available online: https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Map%3A_Organic_Chemistry_(Wade)/14%3A_Reactions_of_Alcohols/14.04%3A_Dehydration_Reactions_of_Alcohols (accessed on 22 February 2022).
- Park, I.-K.; Lee, S.-G.; Choi, D.-H.; Park, J.-D.; Ahn, Y.-J. Insecticidal activities of constituents identified in the essential oil from leaves of Chamaecyparis obtusa against Callosobruchus chinensis (L.) and Sitophilus oryzae (L.). J. Stored Prod. Res. 2003, 39, 375–384. [Google Scholar] [CrossRef]
- Arena, J.S.; Omarini, A.B.; Zunino, M.P.; Peschiutta, M.L.; Defagó, M.T.; Zygadlo, J.A. Essential oils from Dysphania ambrosioides and Tagetes minuta enhance the toxicity of a conventional insecticide against Alphitobius diaperinus. Ind. Crops Prod. 2018, 122, 190–194. [Google Scholar] [CrossRef]
- Songkro, S.; Jenboonlap, M.; Boonprasertpon, M.; Maneenuan, D.; Bouking, K.; Kaewnopparat, N. Effects of glucam P-20, vanillin, and fixolide on mosquito repellency of citronella oil lotions. J. Med. Entomol. 2012, 49, 672–677. [Google Scholar] [CrossRef][Green Version]
- McGovern, T.P.; Beroza, M. Effect of fixatives and other chemicals in extending the activity of the insect attractant trimedlure. J. Econ. Entomol. 1967, 60, 379–383. [Google Scholar] [CrossRef]
- Hidayatulfathi, O.; Mamood, S.N.H.; Kalaivany, M.; Budin, S.B.; Ahmad Rohi, G.; Zulfakar, M.H. Fixatives increase the efficacy of gel formulations containing Piper aduncum Linnaeus (Piperales: Piperaceae) essential oil as repellency. J. Sains Kesihat. Malays. 2019, 17, 9–15. [Google Scholar] [CrossRef]
- Suzuki, H. Physical control of Thrips palmi Karny. Proc. Assoc. Plant Prot. Kyushu 1982, 28, 134–137. [Google Scholar] [CrossRef]
- Roselló, S.; Díez, M.J.; Nuez, F. Viral diseases causing the greatest economic losses to the tomato crop. I. the tomato spotted wilt virus—A review. Sci. Hortic. 1996, 67, 117–150. [Google Scholar] [CrossRef]
- Gao, Y.; Lei, Z.; Reitz, S.R. Western flower thrips resistance to insecticides: Detection, mechanisms and management strategies. Pest Manag. Sci. 2012, 68, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Reitz, S.; Funderburk, J. Management strategies for western flower thrips and the role of insecticides. In Insecticides: Pest Engineering; Khan Perveen, F., Ed.; InTech: Rijeka, Croatia, 2015; pp. 355–372. [Google Scholar]
- Immaraju, J.A.; Paine, T.D.; Bethke, J.A.; Robb, K.L.; Newman, J.P. Western flower thrips (Thysanoptera: Thripidae) resistance to insecticides in coastal California greenhouses. J. Econ. Entomol. 1992, 85, 9–14. [Google Scholar] [CrossRef]
- Denholm, I.; Jespersen, J.B. Insecticide resistance management in Europe: Recent developments and prospects. Pestic. Sci. 1998, 52, 193–195. [Google Scholar] [CrossRef]
- Hara, A.; Mau, R.F.L.; Heu, R.; Jacobsen, C.; Niino-DuPonte, R. Banana rust thrips damage to banana and ornamentals in Hawaii. Insect Pests 2002, 10, 1–4. [Google Scholar]
- Mau, R.F.L.; Gusukuma-Minuto, L. Insecticidal management of key thrips pests of fruiting vegetables, onions, and corn in Hawaii. In Proceedings of the Sixth International Symposium on Thysanoptera, Antalya, Turkey, 27 April–1 May 1998; pp. 107–112. [Google Scholar]
- Cloyd, R. Western flower thrips (Frankliniella occidentalis) management on ornamental crops grown in greenhouses: Have we reached an impasse? Pest Technol. 2009, 3, 1–9. [Google Scholar]
- Brattsten, L.B.; Holyoke, C.W.; Leeper, J.R.; Raffa, K.F. Insecticide resistance: Challenge to pest management and basic research. Science 1986, 231, 1255–1260. [Google Scholar] [CrossRef]
- Kim, K.-H.; Yi, C.-G.; Ahn, Y.-J.; Kim, S.I.; Lee, S.-G.; Kim, J.-R. Fumigant toxicity of basil oil compounds and related compounds to Thrips palmi and Orius strigicollis: Toxicity of basil oil compounds to palm thrips. Pest. Manag. Sci. 2015, 71, 1292–1296. [Google Scholar] [CrossRef]
- Yi, C.-G.; Choi, B.-R.; Park, H.-M.; Park, C.-G.; Ahn, Y.-J. Fumigant toxicity of plant essential oils to Thrips palmi (Thysanoptera: Thripidae) and Orius strigicollis (Heteroptera: Anthocoridae). J. Econ. Entomol. 2006, 99, 1733–1738. [Google Scholar] [CrossRef]
- Han, J.; Choi, B.-R.; Lee, S.-G.; Kim, S.I.; Ahn, Y.-J. Toxicity of plant essential oils to acaricide-susceptible and -resistant Tetranychus urticae (Acari: Tetranychidae) and Neoseiulus californicus (Acari: Phytoseiidae). J. Econ. Entomol. 2010, 103, 1293–1298. [Google Scholar] [CrossRef] [PubMed]
- De Souza Born, F.; da Camara, C.A.G.; de Melo, J.P.R.; de Moraes, M.M. Acaricidal property of the essential oil from Lippia gracilis against Tetranychus urticae and a natural enemy, Neoseiulus californicus, under greenhouse conditions. Exp. Appl. Acarol. 2018, 75, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, M.; Blázquez, M. Phytotoxicity of essential oils on selected weeds: Potential hazard on food crops. Plants 2018, 7, 79. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, M.D.; Blázquez, M.A. Phytotoxic effects of commercial essential oils on selected vegetable crops: Cucumber and tomato. Sustain. Chem. Pharm. 2020, 15, 100209. [Google Scholar] [CrossRef]
- Rolli, E.; Marieschi, M.; Maietti, S.; Sacchetti, G.; Bruni, R. Comparative phytotoxicity of 25 essential oils on pre- and post-emergence development of Solanum lycopersicum L.: A multivariate approach. Ind. Crops Prod. 2014, 60, 280–290. [Google Scholar] [CrossRef]
- Hazrati, H.; Saharkhiz, M.J.; Moein, M.; Khoshghalb, H. Phytotoxic effects of several essential oils on two weed species and tomato. Biocatal. Agric. Biotechnol. 2018, 13, 204–212. [Google Scholar] [CrossRef]
- Werrie, P.-Y.; Durenne, B.; Delaplace, P.; Fauconnier, M.-L. Phytotoxicity of essential oils: Opportunities and constraints for the development of biopesticides. A Review. Foods 2020, 9, 1291. [Google Scholar] [CrossRef]
- Schulz, M.; Kussmann, P.; Knop, M.; Kriegs, B.; Gresens, F.; Eichert, T.; Ulbrich, A.; Marx, F.; Fabricius, H.; Goldbach, H.; et al. Allelopathic monoterpenes interfere with Arabidopsis thaliana cuticular waxes and enhance transpiration. Plant Signal. Behav. 2007, 2, 231–239. [Google Scholar] [CrossRef]
- Mucciarelli, M.; Camusso, W.; Bertea, C.M.; Bossi, S.; Maffei, M. Effect of (+)-pulegone and other oil components of Mentha×piperita on cucumber respiration. Phytochemistry 2001, 57, 91–98. [Google Scholar] [CrossRef]
- Zunino, M.P.; Zygadlo, J.A. Changes in the composition of phospholipid fatty acids and sterols of maize root in response to monoterpenes. J. Chem. Ecol. 2005, 31, 1269–1283. [Google Scholar] [CrossRef]

| Treatment | F. insularis | F. occidentalis | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LC50 in mg/L Air (LCL–UCL) | LC90 in mg/L Air (LCL–UCL) | Slope ± SE | df | χ2 | p Value | LC50 in mg/L Air (LCL–UCL) | LC90 in mg/L Air (LCL–UCL) | Slope ± SE | df | χ2 | p Value | |
| 100× (R)-linalool | 11.7 (10.0–13.4) | 18.0 (16.0–21.2) | 0.20 ± 0.04 | 6 | 10.02 | 0.12 | 29.0 (27.1–30.9) | 36.5 (34.1–40.3) | 0.17 ± 0.03 | 11 | 24.20 | 0.01 |
| 200× (R)-linalool | 18.1 (14.7–21.4) | 30.2 (26.2–37.4) | 0.11 ± 0.02 | 9 | 33.40 | <0.001 | 35.1 (33.7–36.5) | 47.7 (45.6–50.3) | 0.10 ± 0.01 | 14 | 9.73 | 0.78 |
| 300× (R)-linalool | 20.6 (19.3–21.8) | 30.2 (28.4–32.4) | 0.13 ± 0.02 | 9 | 8.95 | 0.44 | 38.9 (37.4–40.4) | 52.8 (50.4–55.7) | 0.09 ± 0.01 | 14 | 6.55 | 0.95 |
| 100× (S)-linalool | 16.7 (16.0–17.3) | 19.3 (18.5–20.6) | 0.47 ± 0.10 | 6 | 0.43 | 0.10 | 34.9 (33.5–36.3) | 46.9 (44.9–49.3) | 0.12 ± 0.02 | 14 | 6.39 | 0.96 |
| 200× (S)-linalool | 17.2 (16.6–17.9) | 19.7 (18.9–21.0) | 0.52 ± 0.15 | 6 | 0.20 | 0.10 | 43.2 (41.6–44.9) | 58.9 (56.4–62.0) | 0.08 ± 0.01 | 16 | 10.41 | 0.84 |
| 300× (S)-linalool | 32.3 (31.0–33.6) | 42.4 (40.5–45.0) | 0.13 ± 0.02 | 11 | 10.60 | 0.48 | 42.1 (40.8–43.5) | 53.5 (51.6–55.9) | 0.11 ± 0.02 | 16 | 11.20 | 0.80 |
| Adjuvant | F. insularis | F. occidentalis | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LC50 in mg/L Air (LCL–UCL) | LC90 in mg/L Air (LCL–UCL) | Slope ± SE | df | χ2 | p Value | LC50 in mg/L Air (LCL-UCL) | LC90 in mg/L Air (LCL-UCL) | Slope ± SE | df | χ2 | p Value | |
| Pure (R)-linalool | 11.7 (10.0–13.4) | 18.0 (16.0–21.2) | 0.20 ± 0.04 | 6 | 10.02 | 0.12 | 29.0 (27.1–30.9) | 36.5 (34.1–40.3) | 0.17 ± 0.03 | 11 | 24.20 | 0.01 |
| Peppermint with (R)-linalool | 10.7 (7.3–12.8) | 16.2 (14.4–18.2) | 0.23 ± 0.08 | 4 | 3.36 | 0.50 | 30.0 (24.5–33.0) | 43.4 (41.0–47.4) | 0.10 ± 0.04 | 4 | 3.29 | 0.51 |
| (S)-linalool with (R)-linalool | 16.5 (11.9–18.4) | 21.5 (19.4–28.5) | 0.26 ± 0.09 | 4 | 7.28 | 0.12 | 45.8 (44.3–47.3) | 58.2 (56.1–61.0) | 0.10 ± 0.02 | 9 | 7.97 | 0.54 |
| Cedarwood with (R)-linalool | 17.7 (15.7–19.3) | 26.4 (24.6–29.0) | 0.15 ± 0.04 | 4 | 3.11 | 0.54 | 63.8 (60.3–67.5) | 80.5 (75.4–89.0) | 0.08 ± 0.01 | 12 | 36.72 | <0.001 |
| Neem with (R)-linalool | 20.7 (13.8–25.3) | 37.7 (32.4–48.1) | 0.08 ± 0.02 | 5 | 10.53 | 0.06 | 73.8 (70.8–76.7) | 105.4 (101.1–110.9) | 0.04 ± 0.01 | 15 | 15.81 | 0.40 |
| Clove with (R)-linalool | 23.6 (19.4–27.0) | 30.2 (26.9–40.3) | 0.19 ± 0.05 | 4 | 13.35 | 0.01 | 63.3 (61.3–65.3) | 82.4 (79.3–86.2) | 0.07 ± 0.01 | 12 | 7.80 | 0.80 |
| 1,8-Cineole with (R)-linalool | 20.8 (15.3–24.6) | 35.9 (31.1–46.0) | 0.09 ± 0.02 | 6 | 15.44 | 0.01 | 38.8 (35.6–41.3) | 58.6 (55.6–62.6) | 0.06 ± 0.01 | 9 | 10.81 | 0.30 |
| trans-Anethole with (R)-linalool | 33.2 (31.3–35.1) | 40.8 (38.1–44.3) | 0.18 ± 0.03 | 7 | 11.84 | 0.11 | 53.2 (51.9–54.5) | 61.9 (60.2–64.2) | 0.15 ± 0.03 | 9 | 7.68 | 0.60 |
| (R)-Pulegone with (R)-linalool | 25.3 (21.7–28.4) | 35.7 (31.9–43.4) | 0.12 ± 0.02 | 7 | 22.44 | <0.01 | 47.0 (42.9–50.5) | 70.3 (65.1–78.7) | 0.06 ± 0.01 | 10 | 16.96 | 0.08 |
| Adjuvant | F. insularis | F. occidentalis |
|---|---|---|
| Peppermint and (R)-linalool | 1.093 | 0.967 |
| (S)-linalool and (R)-linalool | 0.709 | 0.633 |
| Cedarwood and (R)-linalool | 0.661 | 0.455 |
| Neem and (R)-linalool | 0.565 | 0.393 |
| Clove and (R)-linalool | 0.496 | 0.458 |
| 1,8-Cineole and (R)-linalool | 0.563 | 0.747 |
| trans-Anethole and (R)-linalool | 0.352 | 0.545 |
| (R)-Pulegone and (R)-linalool | 0.462 | 0.617 |
| α-Terpineol and (R)-linalool | N/A | 1.090 |
| Concentration (mg/L air) | Length (cm) of Seedlings (Mean ± SE) | ||||
|---|---|---|---|---|---|
| Foliar Spray | Hydrogel Fumigation | ||||
| Hypocotyl | Root | Hypocotyl | Root | ||
| Control | 5.40 ± 0.14 a | 7.45 ± 1.23 a | 6.60 ± 0.19 a | 11.10 ± 0.91 a | |
| (R)-Linalool | 21.56 | 4.55 ± 0.75 a | 7.10 ± 1.65 a | 5.90 ± 0.21 ab | 10.70 ± 0.59 ab |
| 29.00 | 3.80 ± 0.80 a | 6.95 ± 0.52 a | 5.75 ± 0.28 ab | 7.95 ± 0.67 b | |
| 36.89 | 0.00 ± 0.00 b | 0.00 ± 0.00 b | 5.65 ± 0.30 ab | 7.85 ± 0.52 b | |
| (R)-linalool with peppermint oil | 3.98 | 0.00 ± 0.00 b | 0.00 ± 0.00 b | 5.75 ± 0.21 ab | 10.95 ± 0.96 ab |
| 11.90 | 0.00 ± 0.00 b | 0.00 ± 0.00 b | 5.25 ± 0.23 b | 9.55 ± 0.85 ab | |
| 19.90 | 0.00 ± 0.00 b | 0.00 ± 0.00 b | 5.45 ± 0.31 b | 9.30 ± 0.41 ab | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gharbi, K.; Tay, J.-W. Fumigant Toxicity of Essential Oils against Frankliniella occidentalis and F. insularis (Thysanoptera: Thripidae) as Affected by Polymer Release and Adjuvants. Insects 2022, 13, 493. https://doi.org/10.3390/insects13060493
Gharbi K, Tay J-W. Fumigant Toxicity of Essential Oils against Frankliniella occidentalis and F. insularis (Thysanoptera: Thripidae) as Affected by Polymer Release and Adjuvants. Insects. 2022; 13(6):493. https://doi.org/10.3390/insects13060493
Chicago/Turabian StyleGharbi, Karim, and Jia-Wei Tay. 2022. "Fumigant Toxicity of Essential Oils against Frankliniella occidentalis and F. insularis (Thysanoptera: Thripidae) as Affected by Polymer Release and Adjuvants" Insects 13, no. 6: 493. https://doi.org/10.3390/insects13060493
APA StyleGharbi, K., & Tay, J.-W. (2022). Fumigant Toxicity of Essential Oils against Frankliniella occidentalis and F. insularis (Thysanoptera: Thripidae) as Affected by Polymer Release and Adjuvants. Insects, 13(6), 493. https://doi.org/10.3390/insects13060493






