Utilization of Pomelo (Citrus maxima) Peel Waste into Bioactive Essential Oils: Chemical Composition and Insecticidal Properties
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection of Citrus Peel and Essential Oil Extraction
2.2. Test Insects and Larval Culture
2.3. Analaysis of Chemical Configuration Using GC-MS
2.4. Contact Toxicity
2.5. Fumigant Toxicity
2.6. Repellent Activity Assay
2.7. Effect of CMEO on Wheat Germination
2.8. Screening of Mosquito Larvicidal Activity by CMEO
2.9. Toxicity on Non-Targeted Organism (Poecilia reticulata)
2.10. Data Analysis
3. Results
3.1. Chemical Configuration of CMEO by GC-MS
3.2. Contact Toxicity
3.3. Fumigant Toxicity
3.4. Repellent Activity
3.5. Larvicidal Potential of CMEO
3.6. Phytotoxicity Analysis of the CMEO
3.7. Non-Targeted Organism Toxicity of CMEO
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gomes-Araújo, R.; Martínez-Vázquez, D.G.; Charles-Rodríguez, A.V.; Rangel-Ortega, S.; Robledo-Olivo, A. Bioactive Compounds from Agricultural Residues, Their Obtaining Techniques, and the Antimicrobial Effect as Postharvest Additives. Int. J. Food Sci. 2021, 2021, 9936722. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, N.G.; Silva, K.E.; Croda, J.; Simionatto, S. Antibacterial activity of Cinnamomum cassia L. essential oil in a carbapenem- and polymyxin-resistant Klebsiella aerogenes strain. Rev. Soc. Bras. Med. Trop. 2020, 53, e20200032. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.K.R.D.; Figueiredo, P.L.B.; Byler, K.G.; Setzer, W.N. Essential Oils as Antiviral Agents. Potential of Essential Oils to Treat SARS-CoV-2 Infection: An In-Silico Investigation. Int. J. Mol. Sci. 2020, 21, 3426. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, A.K.; Singh, V.K.; Dwivedy, A.K.; Das, S.; Upadhyay, N.; Singh, A.; Dkhar, M.S.; Kayang, H.; Prakash, B.; Dubey, N.K. Chemically characterised Pimenta dioica (L.) Merr. essential oil as a novel plant based antimicrobial against fungal and aflatoxin B1 contamination of stored maize and its possible mode of action. Nat. Prod. Res. 2020, 34, 745–749. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, K.; Zhang, J.; Fu, J.; Li, J.; Wang, G.; Qiu, Z.; Wang, X. Antibacterial Activity of Cinnamomum camphora Essential Oil on Escherichia coli During Planktonic Growth and Biofilm Formation. Front. Microbiol. 2020, 11, 561002. [Google Scholar] [CrossRef]
- Wijesinghe, G.K.; Maia, F.C.; de Oliveira, T.R.; de Feiria, S.N.B.; Joia, F.; Barbosa, J.P.; Boni, G.C.; Sardi, J.C.O.; Rosalen, P.L.; Hofling, J.F. Effect of Cinnamomum verum leaf essential oil on virulence factors of Candida species and determination of the in-vivo toxicity with Galleria mellonella model. Mem. Do Inst. Oswaldo Cruz 2020, 115, e200349. [Google Scholar] [CrossRef]
- Yap, P.S.; Lim, S.H.; Hu, C.P.; Yiap, B.C. Combination of essential oils and antibiotics reduce antibiotic resistance in plasmid-conferred multidrug resistant bacteria. Phytomed. Int. J. Phytother. Phytopharm. 2013, 20, 710–713. [Google Scholar] [CrossRef]
- Gaire, S.; O’Connell, M.; Holguin, F.O.; Amatya, A.; Bundy, S.; Romero, A. Insecticidal Properties of Essential Oils and Some of Their Constituents on the Turkestan Cockroach (Blattodea: Blattidae). J. Econ. Entomol. 2017, 110, 584–592. [Google Scholar] [CrossRef]
- Francikowski, J.; Baran, B.; Cup, M.; Janiec, J.; Krzyżowski, M. Commercially Available Essential Oil Formulas as Repellents Against the Stored-Product Pest Alphitobius diaperinus. Insects 2019, 10, 96. [Google Scholar] [CrossRef] [Green Version]
- Aburjai, T.; Mansi, K.; Azzam, H.; Alqudah, D.A.; Alshaer, W.; Abuirjei, M. Chemical Compositions and Anticancer Potential ofEssential Oil from Greenhouse-cultivated Ocimum basilicum Leaves. Indian J. Pharm. Sci. 2020, 82, 179–184. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.C.; Xu, W.X.; Lin, L.Y.; Yang, J.J.; Liu, C.T. Chemical composition and hypoglycemic and pancreas-protective effect of leaf essential oil from indigenous cinnamon (Cinnamomum osmophloeum Kanehira). J. Agric. Food Chem. 2013, 61, 4905–4913. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.Y.; Chuang, C.H.; Chen, H.C.; Yang, K.M. Lime (Citrus aurantifolia (Christm.) Swingle) Essential Oils: Volatile Compounds, Antioxidant Capacity, and Hypolipidemic Effect. Foods 2019, 8, 398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Regnault-Roger, C.; Vincent, C.; Arnason, J.T. Essential oils in insect control: Low-risk products in a high-stakes world. Annu. Rev. Entomol. 2012, 57, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Jia, X.; Dong, L.; Liu, L.; Huang, F.; Chi, J.; Ma, Q.; Zhao, D.; Zhang, M.; Zhang, R. Structural elucidation of flavonoids from Shatianyu (Citrus grandis L. Osbeck) pulp and screening of key antioxidant components. Food Chem. 2022, 366, 130605. [Google Scholar] [CrossRef]
- Somanathan Karthiga, R.; Sukhdeo, S.V.; Madhugiri Lakshminarayan, S.; Mysuru Nanjarajurs, S. Efficacy of Citrus maxima fruit segment supplemented paranthas in STZ induced diabetic rats. J. Food Sci. 2021, 86, 2091–2102. [Google Scholar] [CrossRef]
- Tsai, M.L.; Lin, C.D.; Khoo, K.A.; Wang, M.Y.; Kuan, T.K.; Lin, W.C.; Zhang, Y.N.; Wang, Y.Y. Composition and Bioactivity of Essential Oil from Citrus grandis (L.) Osbeck ‘Mato Peiyu’ Leaf. Molecules 2017, 22, 2154. [Google Scholar] [CrossRef] [Green Version]
- Oboh, G.; Ademosun, A.O.; Olumuyiwa, T.A.; Olasehinde, T.A.; Ademiluyi, A.O.; Adeyemo, A.C. Insecticidal activity of essential oil from orange peels (Citrus sinensis) against Tribolium confusum, Callosobruchus maculatus and Sitophilus oryzae and its inhibitory effects on acetylcholinesterase and Na+/K+-ATPase activities. Phytoparasitica 2017, 45, 501–508. [Google Scholar] [CrossRef]
- Campolo, O.; Malacrinò, A.; Zappalà, L.; Laudani, F.; Chiera, E.; Serra, D.; Russo, M.; Palmeri, V. Fumigant bioactivity of fiveCitrusessential oils againstTribolium confusum. Phytoparasitica 2014, 42, 223–233. [Google Scholar] [CrossRef]
- Lou, H.; Huang, Y.; Ouyang, Y.; Zhang, Y.; Xi, L.; Chu, X.; Wang, Y.; Wang, C.; Zhang, L. Artemisia annua-sublingual immunotherapy for seasonal allergic rhinitis: A randomized controlled trial. Allergy 2020, 75, 2026–2036. [Google Scholar] [CrossRef]
- Giatropoulos, A.; Papachristos, D.P.; Kimbaris, A.; Koliopoulos, G.; Polissiou, M.G.; Emmanouel, N.; Michaelakis, A. Evaluation of bioefficacy of three Citrus essential oils against the dengue vector Aedes albopictus (Diptera: Culicidae) in correlation to their components enantiomeric distribution. Parasitol. Res. 2012, 111, 2253–2263. [Google Scholar] [CrossRef]
- Michaelakis, A.; Papachristos, D.; Kimbaris, A.; Koliopoulos, G.; Giatropoulos, A.; Polissiou, M.G. Citrus essential oils and four enantiomeric pinenes against Culex pipiens (Diptera: Culicidae). Parasitol. Res. 2009, 105, 769–773. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, K.; Sarma, R.; Rabha, B.; Khanikor, B. Repellent Activity of Citrus Essential Oils and Two Constituent Compounds Against Aedes aegypti. Proc. Natl. Acad. Sci. USA 2022, in press. [Google Scholar] [CrossRef]
- Jiang, J.; Shan, L.; Chen, Z.; Xu, H.; Wang, J.; Liu, Y.; Xiong, Y. Evaluation of antioxidant-associated efficacy of flavonoid extracts from a traditional Chinese medicine Hua Ju Hong (peels of Citrus grandis (L.) Osbeck). J. Ethnopharmacol. 2014, 158 Pt A, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Oboh, G.; Ademosun, A.O. Shaddock peels (Citrus maxima) phenolic extracts inhibit alpha-amylase, alpha-glucosidase and angiotensin I-converting enzyme activities: A nutraceutical approach to diabetes management. Diabetes Metab. Syndr. 2011, 5, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Ademosun, A.O.; Adebayo, A.A.; Popoola, T.V.; Oboh, G. Shaddock (Citrus maxima) peels extract restores cognitive function, cholinergic and purinergic enzyme systems in scopolamine-induced amnesic rats. Drug Chem. Toxicol. 2020, 45, 1073–1080. [Google Scholar] [CrossRef]
- Kuo, P.C.; Liao, Y.R.; Hung, H.Y.; Chuang, C.W.; Hwang, T.L.; Huang, S.C.; Shiao, Y.J.; Kuo, D.H.; Wu, T.S. Anti-Inflammatory and Neuroprotective Constituents from the Peels of Citrus grandis. Molecules 2017, 22, 967. [Google Scholar] [CrossRef] [Green Version]
- Ou, M.C.; Liu, Y.H.; Sun, Y.W.; Chan, C.F. The Composition, Antioxidant and Antibacterial Activities of Cold-Pressed and Distilled Essential Oils of Citrus paradisi and Citrus grandis (L.) Osbeck. Evid.-Based Complement. Altern. Med. Ecam 2015, 2015, 804091. [Google Scholar] [CrossRef] [Green Version]
- Singh, P.; Shukla, R.; Prakash, B.; Kumar, A.; Singh, S.; Mishra, P.K.; Dubey, N.K. Chemical profile, antifungal, antiaflatoxigenic and antioxidant activity of Citrus maxima Burm. and Citrus sinensis (L.) Osbeck essential oils and their cyclic monoterpene, DL-limonene. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2010, 48, 1734–1740. [Google Scholar] [CrossRef]
- Clevenger, J.F. Apparatus for the Determination of Volatile Oil. J. Am. Pharm. Assoc. 1928, 17, 345–349. [Google Scholar] [CrossRef]
- Patiño-Bayona, W.R.; Nagles Galeano, L.J.; Bustos Cortes, J.J.; Delgado Ávila, W.A.; Herrera Daza, E.; Suárez, L.E.C.; Prieto-Rodríguez, J.A.; Patiño-Ladino, O.J. Effects of Essential Oils from 24 Plant Species on Sitophilus zeamais Motsch (Coleoptera, Curculionidae). Insects 2021, 12, 532. [Google Scholar] [CrossRef]
- Bagade, R.P.; Jadhav, A.D.; Chavan, R.V. Toxicity and repellency of four plant essential oils against Tribolium castaneum (Herbst)(Coleoptera: Tenebrionidae). Int. J. Trop. Insect Sci. 2021, 41, 1505–1512. [Google Scholar] [CrossRef]
- Culver, J.J. The Value of the Dry Substitutes for Liquid Lime Sulphur as a Control for San Jose Scale. J. Econ. Entomol. 1925, 18, 265. [Google Scholar] [CrossRef]
- Denloye, A.; Makanjuola, W.; Teslim, O.; Alafia, O.; Kasali, A.; Eshilokun, A. Toxicity of Chenopodium ambrosioides L.(Chenopodiaceae) products from Nigeria against three storage insects. J. Plant Prot. Res. 2010, 50, 379–384. [Google Scholar] [CrossRef]
- Bai, L.; Jiao, M.L.; Zang, H.Y.; Guo, S.S.; Wang, Y.; Sang, Y.L.; Du, S.S. Chemical composition of essential oils from four Rhododendron species and their repellent activity against three stored-product insects. Environ. Sci. Pollut. Res. Int. 2019, 26, 23198–23205. [Google Scholar] [CrossRef]
- Ebrahimifar, J.; Jamshidnia, A.; Sadeghi, R.; Ebadollahi, A. Repellency of Ferulago angulata (Schlecht.) Boiss essential oil on two major stored-product insect pests without effect on wheat germination. Int. J. Trop. Insect Sci. 2021, 41, 217–223. [Google Scholar] [CrossRef]
- Narayanankutty, A.; Kuttithodi, A.M.; Alfarhan, A.; Rajagopal, R.; Barcelo, D. Chemical Composition, Insecticidal and Mosquito Larvicidal Activities of Allspice (Pimenta dioica) Essential Oil. Molecules 2021, 26, 6698. [Google Scholar] [CrossRef]
- Gopinath, P.P.; Parsad, R.; Joseph, B.; Adarsh, V. grapesAgri1: Collection of Shiny Apps for Data Analysis in Agriculture. J. Open Source Softw. 2021, 6, 3437. [Google Scholar] [CrossRef]
- Kłyś, M.; Malejky, N.; Nowak-Chmura, M. The repellent effect of plants and their active substances against the beetle storage pests. J. Stored Prod. Res. 2017, 74, 66–77. [Google Scholar] [CrossRef]
- Rajendran, S.; Sriranjini, V. Plant products as fumigants for stored-product insect control. J. Stored Prod. Res. 2008, 44, 126–135. [Google Scholar] [CrossRef]
- Isman, M.B. Bioinsecticides based on plant essential oils: A short overview. Z. Naturforsch. C. 2020, 75, 179–182. [Google Scholar] [CrossRef]
- Gueye, M.T.; Cissokho, P.S.; Goergen, G.; Ndiaye, S.; Seck, D.; Gueye, G.; Wathelet, J.-P.; Lognay, G. Efficacy of powdered maize cobs against the maize weevil Sitophilus zeamais (Coleoptera: Curculionidae) in stored maize in Senegal. Int. J. Trop. Insect Sci. 2012, 32, 94–100. [Google Scholar] [CrossRef]
- Nerio, L.S.; Olivero-Verbel, J.; Stashenko, E. Repellent activity of essential oils: A review. Bioresour. Technol. 2010, 101, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Dao, P.T.; Tran, N.Y.T.; Tran, Q.N.; Bach, G.L.; Lam, T.V. Kinetics of pilot-scale essential oil extraction from pomelo (Citrus maxima) peels: Comparison between linear and nonlinear models. Alex. Eng. J. 2022, 61, 2564–2572. [Google Scholar] [CrossRef]
- Chen, L.; Teng, H.; Xie, Z.; Cao, H.; Cheang, W.S.; Skalicka-Woniak, K.; Georgiev, M.I.; Xiao, J. Modifications of dietary flavonoids towards improved bioactivity: An update on structure-activity relationship. Crit. Rev. Food Sci. Nutr. 2018, 58, 513–527. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.-W.; Lin, Y.-H.; Lin, C.-H.; Jen, H.-C. Antibacterial Activity of Emulsified Pomelo (Citrus grandis Osbeck) Peel Oil and Water-Soluble Chitosan on Staphylococcus aureus and Escherichia coli. Molecules 2018, 23, 840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ocheng, F.; Bwanga, F.; Joloba, M.; Softrata, A.; Azeem, M.; Pütsep, K.; Borg-Karlson, A.-K.; Obua, C.; Gustafsson, A. Essential Oils from Ugandan Aromatic Medicinal Plants: Chemical Composition and Growth Inhibitory Effects on Oral Pathogens. Evid.-Based Complement. Altern. Med. 2015, 2015, 230832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinto, Z.T.; Sánchez, F.F.; Santos, A.R.D.; Amaral, A.C.F.; Ferreira, J.L.P.; Escalona-Arranz, J.C.; Queiroz, M.M.D.C. Chemical composition and insecticidal activity of Cymbopogon citratus essential oil from Cuba and Brazil against housefly. Rev. Bras. De Parasitol. Veterinária 2015, 24, 36–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akram, W.; Khan, H.A.A.; Hussain, A.; Hafeez, F. Citrus Waste-Derived Essential Oils: Alternative Larvicides for Dengue Fever Mosquito, Aedes albopictus (Skuse)(Culicidae: Diptera). Pak. J. Zool. 2011, 43, 367–372. [Google Scholar]
- de Andrade Dutra, K.; de Oliveira, J.V.; Navarro, D.M.D.A.F.; Santos, J.P.O. Control of Callosobruchus maculatus (FABR.)(Coleoptera: Chrysomelidae: Bruchinae) in Vigna unguiculata (L.) WALP. with essential oils from four Citrus spp. plants. J. Stored Prod. Res. 2016, 68, 25–32. [Google Scholar] [CrossRef]
- Ribeiro, N.C.; da Camara, C.A.G.; Melo, J.P.R.; de Moraes, M.M. Acaricidal properties of essential oils from agro-industrial waste products from citric fruit against Tetranychus urticae. J. Appl. Entomol. 2019, 143, 731–743. [Google Scholar] [CrossRef]
- Kumar, S.; Warikoo, R.; Mishra, M.; Seth, A.; Wahab, N. Larvicidal efficacy of the Citrus limetta peel extracts against Indian strains of Anopheles stephensi Liston and Aedes aegypti L. Parasitol. Res. 2012, 111, 173–178. [Google Scholar] [CrossRef]
- Sarma, R.; Adhikari, K.; Mahanta, S.; Khanikor, B. Insecticidal activities of Citrus aurantifolia essential oil against Aedes aegypti (Diptera: Culicidae). Toxicol. Rep. 2019, 6, 1091–1096. [Google Scholar] [CrossRef] [PubMed]
- Chavan, P.; Singh, A.K.; Kaur, G. Recent progress in the utilization of industrial waste and by-products of citrus fruits: A review. J. Food Process Eng. 2018, 41, e12895. [Google Scholar] [CrossRef]
- Azmy, R.M. Nanoformulated Materials from Citrus Wastes. In Waste Recycling Technologies for Nanomaterials Manufacturing; Springer: Berlin/Heidelberg, Germany, 2021; pp. 649–669. [Google Scholar]
- Baccati, C.; Gibernau, M.; Paoli, M.; Ollitrault, P.; Tomi, F.; Luro, F. Chemical Variability of Peel and Leaf Essential Oils in the Citrus Subgenus Papeda (Swingle) and Few Relatives. Plants 2021, 10, 1117. [Google Scholar] [CrossRef] [PubMed]
- Thavanapong, N.; Wetwitayaklung, P.; Charoenteeraboon, J. Comparison of Essential Oils Compositions of Citrus maxima Merr. Peel Obtained by Cold Press and Vacuum Stream Distillation Methods and of Its Peel and Flower Extract Obtained by Supercritical Carbon Dioxide Extraction Method and Their Antimicrobial Activity. J. Essent. Oil Res. 2010, 22, 71–77. [Google Scholar]
- Ngan, T.; Muoi, N.; Quan, P.; Cang, M. Evaluation of physical and chemical properties of pomelo (Citrus grandis L.) essential oil using steam distillation process. Asian J. Chem. 2020, 6, 1433–1436. [Google Scholar] [CrossRef]
- Luo, C.; Li, D.; Wang, Y.; Guo, S.; Zhang, D.; Du, S. Chemical Composition and Insecticide Efficacy of Essential Oils from Citrus medica L. var. sarcodactylis Swingle Against Tribolium castaneum Herbst in Stored Medicinal Materials. J. Essent. Oil Bear. Plants 2019, 22, 1182–1194. [Google Scholar] [CrossRef]
- Oyedeji, A.O.; Okunowo, W.O.; Osuntoki, A.A.; Olabode, T.B.; Ayo-Folorunso, F. Insecticidal and biochemical activity of essential oil from Citrus sinensis peel and constituents on Callosobrunchus maculatus and Sitophilus zeamais. Pestic. Biochem. Physiol. 2020, 168, 24. [Google Scholar] [CrossRef]
- Abouelatta, A.M.; Keratum, A.Y.; Ahmed, S.I.; El-Zun, H.M. Repellent, contact and fumigant activities of geranium (Pelargonium graveolens L.’Hér) essential oils against Tribolium castaneum (Herbst) and Rhyzopertha dominica (F.). Int. J. Trop. Insect Sci. 2020, 40, 1021–1030. [Google Scholar] [CrossRef]
- Deb, M.; Kumar, D. Bioactivity and efficacy of essential oils extracted from Artemisia annua against Tribolium casteneum (Herbst. 1797) (Coleoptera: Tenebrionidae): An eco-friendly approach. Ecotoxicol Env. Saf 2020, 189, 22. [Google Scholar] [CrossRef]
- Upadhyay, N.; Singh, V.K.; Dwivedy, A.K.; Das, S.; Chaudhari, A.K.; Dubey, N.K. Assessment of Melissa officinalis L. essential oil as an eco-friendly approach against biodeterioration of wheat flour caused by Tribolium castaneum Herbst. Environ. Sci. Pollut. Res. Int. 2019, 26, 14036–14049. [Google Scholar] [CrossRef] [PubMed]
- Papachristos, D.P.; Kimbaris, A.C.; Papadopoulos, N.T.; Polissiou, M.G. Toxicity of citrus essential oils against Ceratitis capitata (Diptera: Tephritidae) larvae. Ann. Appl. Biol. 2009, 155, 381–389. [Google Scholar] [CrossRef]
- Rossi, Y.E.; Palacios, S.M. Fumigant toxicity of Citrus sinensis essential oil on Musca domestica L. adults in the absence and presence of a P450 inhibitor. Acta Trop. 2013, 127, 33–37. [Google Scholar] [CrossRef]
- Yoon, C.; Kang, S.-H.; Yang, J.-O.; Noh, D.-J.; Indiragandhi, P.; Kim, G.-H. Repellent activity of citrus oils against the cockroaches Blattella germanica, Periplaneta americana and P. fuliginosa. J. Pestic. Sci. 2009, 34, 77–88. [Google Scholar] [CrossRef] [Green Version]
- Devi, M.A.; Sahoo, D.; Singh, T.B.; Rajashekar, Y. Toxicity, repellency and chemical composition of essential oils from Cymbopogon species against red flour beetle Tribolium castaneum Herbst (Coleoptera: Tenebrionidae). J. Consum. Prot. Food Saf. 2020, 15, 181–191. [Google Scholar] [CrossRef]
- Tandorost, R.; Karimpour, Y. Evaluation of fumigant toxicity of orange peel Citrus sinensis (L.) essential oil against three stored product insects in laboratory condition. Munis Entomol. Zool. 2012, 7, 352–358. [Google Scholar]
- Oyedeji, A.O.; Afolayan, A.J.; Hutchings, A. Compositional variation of the essential oils of Artemisia afra Jacq. from three provinces in South Africa—A case study of its safety. Nat. Prod. Commun. 2009, 4, 849–852. [Google Scholar] [CrossRef] [Green Version]
- Hernandez-Lambraño, R.; Pajaro-Castro, N.; Caballero-Gallardo, K.; Stashenko, E.; Olivero-Verbel, J. Essential oils from plants of the genus Cymbopogon as natural insecticides to control stored product pests. J. Stored Prod. Res. 2015, 62, 81–83. [Google Scholar] [CrossRef]
- Fouad, H.A.; da Camara, C.A. Chemical composition and bioactivity of peel oils from Citrus aurantiifolia and Citrus reticulata and enantiomers of their major constituent against Sitophilus zeamais (Coleoptera: Curculionidae). J. Stored Prod. Res. 2017, 73, 30–36. [Google Scholar] [CrossRef]
- França, S.M.D.; Oliveira, J.V.D.; Esteves Filho, A.B.; Oliveira, C.M.D. Toxicity and repellency of essential oils to Zabrotes subfasciatus (Boheman)(Coleoptera, Chrysomelidae, Bruchinae) in Phaseolus vulgaris L. Acta Amaz. 2012, 42, 381–386. [Google Scholar] [CrossRef]
- Chaubey, M.K. Essential oils as green pesticides of stored grain insects. Eur. J. Biol. Res. 2019, 9, 202–244. [Google Scholar]
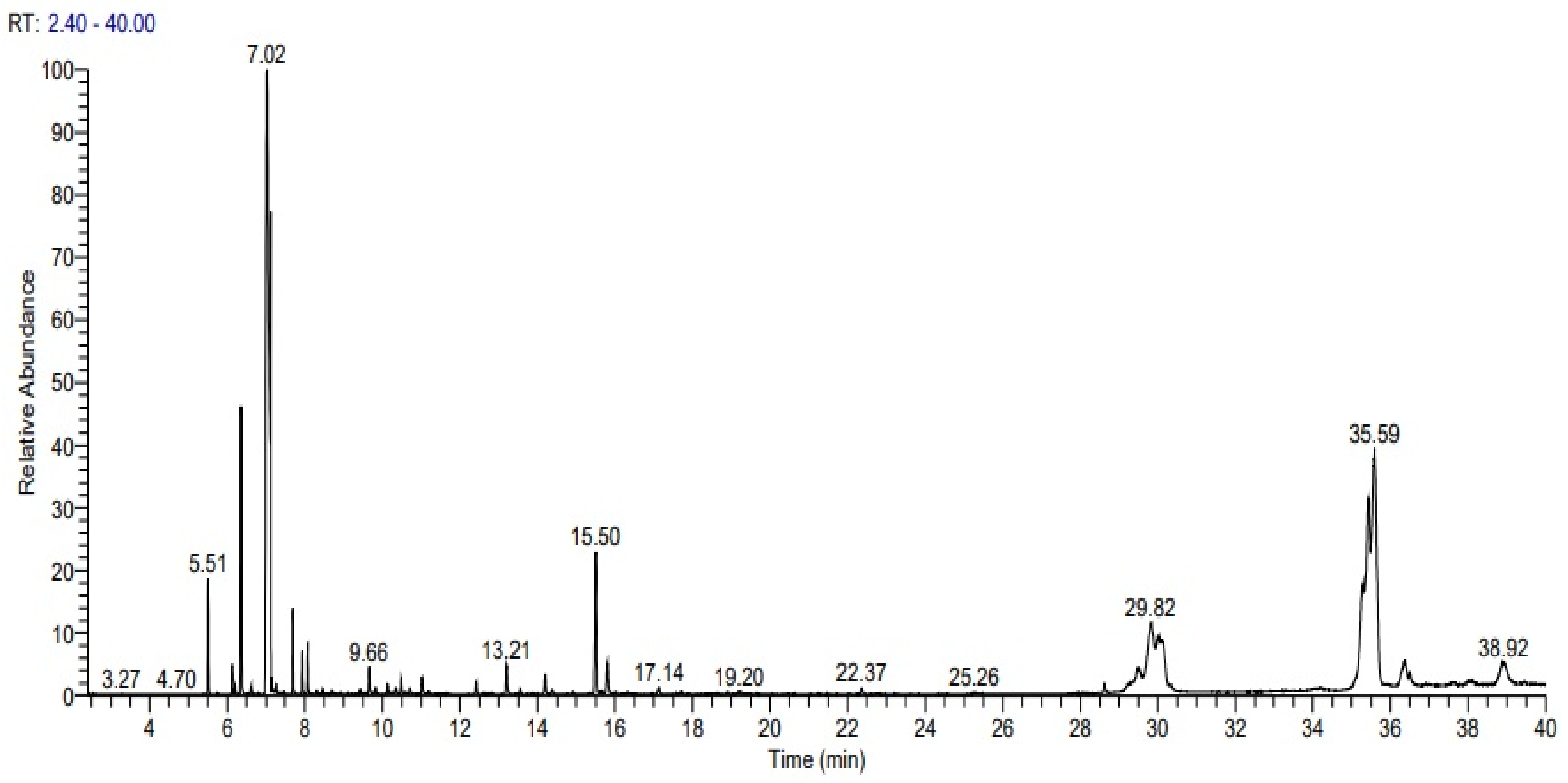
| Peak No. | Retention Time | Component | RSI a | %RA b |
|---|---|---|---|---|
| 1 | 5.51 | 3-carene | 934 | 1.49 |
| 2 | 6.12 | Cyclohexene, 4-methylene-1-(1-methylethyl) | 921 | 0.40 |
| 3 | 6.36 | α-Pinene | 895 | 4.32 |
| 4 | 7.02 | D-Limonene | 881 | 33.61 |
| 5 | 7.69 | trans-Linalool oxide (furanoid) | 878 | 1.17 |
| 6 | 7.93 | cis-Linaloloxide | 901 | 0.62 |
| 7 | 8.08 | 1,6-Octadien-3-ol, 3,7-dimethyl | 921 | 0.76 |
| 8 | 9.66 | α-Terpineol | 923 | 0.57 |
| 9 | 10.48 | 2,6-Octadienal, 3,7-dimethyl-, (Z) | 876 | 0.31 |
| 10 | 11.02 | Citral | 921 | 0.37 |
| 11 | 12.42 | 2-Carene | 883 | 0.26 |
| 12 | 13.21 | Geranyl acetate | 917 | 0.64 |
| 13 | 14.20 | Caryophyllene | 921 | 0.41 |
| 14 | 15.49 | α-Cubebene | 882 | 3.18 |
| 15 | 15.81 | α-Guaiene | 904 | 0.90 |
| 16 | 29.50 | 4,4-dimethyl Cholesta-22,24-dien-5-ol | 895 | 0.82 |
| 17 | 29.82 | Stigmasterol | 917 | 5.22 |
| 18 | 30.05 | Desmosterol | 864 | 3.79 |
| 19 | 30.12 | (3á,22E) 3-methoxy-Stigmasta-5,22-diene, | 873 | 1.95 |
| 20 | 35.29 | campesterol | 895 | 4.31 |
| 21 | 35.43 | α-Sitosterol | 838 | 12.19 |
| 22 | 35.59 | β-Sitosterol | 928 | 17.99 |
| 23 | 36.36 | 24-propylidene-, (3á) Cholest-5-en-3-ol, | 874 | 1.90 |
| 24 | 36.49 | Allopregnane-3á,7à,11à-triol-20-one | 829 | 0.39 |
| 25 | 38.91 | 9,19-Cyclolanost-24-en-3-ol, (3á) | 898 | 2.41 |
| Total (%) | 100.00 | |||
| Test Insects | Exposure (h) | LC50 a (mg/cm2) | LC90 a (mg/cm2) | Slope ± SEM b | χ2 (d.f) |
|---|---|---|---|---|---|
| T. castaneum | 24 | 63.31 (49.4–73.2) | 121.44 (106.2–151.5) | 0.022 ± 0.004 | 0.335 (3) |
| 48 | 37.15 (18.8–48.2) | 109.38 (86.6–192.8) | 2.733 ± 0.708 | 0.264 (3) | |
| C. maculatus | 24 | 7.12 (5.1–12.7) | 16.73 (10.4–153.2) | 3.458 ± 0.601 | 4.58 (3) |
| 48 | 5.06 (3.3–7.1) | 13.44 (8.8–52.2) | 3.023 ± 0.501 | 3.64 (3) |
| Test Insects | Exposure Time (h) | LC50 (mg/L Air) | LC90 (mg/L Air) | Slope ± SEM a | χ2 (d.f) |
|---|---|---|---|---|---|
| T. castaneum | 24 | 4.95 (4.1–5.9) | 12.68 (9.7–20.1) | 3.138 ± 0.503 | 1.396 (3) |
| 48 | 4.13 (3.3–5.0) | 11.82 (8.9–18.8) | 2.806 ± 0.431 | 1.262 (3) | |
| C. maculatus | 24 | 3.38 (2.1–4.8) | 29.61 (14.8–170.5) | 1.360 ± 0.318 | 1.136 (3) |
| 48 | 1.34 (0.35–2.2) | 17.80 (9.1–129.6) | 1.144 ± 0.316 | 2.535 (3) |
| Test Insects | Concentration (mg/cm2) | Repellence Percentage of Treatments After | % Repellency (Mean± bSEM) | Repellent Class | ||||
|---|---|---|---|---|---|---|---|---|
| 2 h | 4 h | 6 h | 12 h | 24 h | ||||
| T. castaneum | 0.5 | 33.3 ± 13.3 a | 46.7 ± 6.6 a | 60.0 ± 11.5 a | 66.7 ± 6.6 a | 60.0 ± 11.5 a | 53.3 ± 5.8 b | III |
| 1.5 | 33.3 ± 17.6 a | 66.7 ± 6.6 a | 60.0 ± 11.5 a | 66.6 ± 6.6 a | 66.7 ± 13.3 a | 58.7 ± 6.5 ab | III | |
| 2.5 | 60.0 ± 11.5 a | 53.3 ± 6.6 a | 66.7 ± 13.3 a | 73.3 ± 6.6 a | 73.3 ± 17.6 a | 65.3 ± 3.8 ab | IV | |
| 3.5 | 66.7 ± 13.3 a | 60.0 ± 11.5 a | 73.3 ± 17.6 a | 80.0 ± 11.5 a | 86.7 ± 13.3 a | 73.3 ± 4.7 ab | IV | |
| 5 | 73.3 ± 6.6 a | 66.7 ± 13.3 a | 73.3 ± 13.3 a | 86.7 ± 13.3 a | 93.3 ± 6.6 a | 78.7 ± 4.9 a | IV | |
| F value | 2.07 | 0.85 | 0.29 | 1.02 | 1.29 | 3.86 | ||
| p value | 0.16 | 0.53 | 0.87 | 0.44 | 0.33 | 0.02 | ||
| a d.f | 4 | 4 | 4 | 4 | 4 | 20 | ||
| C. maculatus | 0.5 | 6.7 ± 6.6 a | 13.3 ± 6.6 a | 33.3 ± 17.6 a | 33.3 ± 13.3 a | 33.3 ± 6.6 b | 23.9 ± 8.9 c | II |
| 1.5 | 20.0 ± 11.5 a | 26.7 ± 6.6 a | 40.0 ± 11.5 a | 53.3 ± 6.6 a | 53.3 ± 17.6 ab | 38.7 ± 6.7 bc | II | |
| 2.5 | 40.0 ± 11.5 a | 33.3 ± 13.3 a | 53.3 ± 17.6 a | 60.0 ± 11.5 a | 60.0 ± 11.5 ab | 49.3 ± 5.4 abc | III | |
| 3.5 | 46.7 ± 6.6 a | 53.3 ± 13.3 a | 60.0 ± 11.5 a | 80.0 ± 11.5 a | 86.7 ± 6.6 a | 65.3 ± 7.7 ab | IV | |
| 5 | 40.0 ± 11.5 a | 60.0 ± 19.9 a | 80.0 ± 11.5 a | 73.3 ± 13.3 a | 93.3 ± 6.6 a | 69.3 ± 9.1a | IV | |
| F value | 3.13 | 2.30 | 1.69 | 2.50 | 5.18 | 7.01 | ||
| p value | 0.06 | 0.12 | 0.22 | 0.10 | 0.01 | 0.001 | ||
| ad.f | 4 | 4 | 4 | 4 | 4 | 20 | ||
| Mosquito | LC50 (μg/mL) | Slope ± SEM | χ2 (d.f) |
|---|---|---|---|
| Ar. subalbatus | 76.24 ± 3.2 | 0.6566 ± 0.027 | 1.439 (3) |
| Ae. aegypti | 47.07 ± 2.4 | 1.0643 ± 0.052 | 5.855 (3) |
| Cx. tritaeniorhynchus | 58.04 ± 2.8 | 0.8630 ± 0.042 | 2.099 (3) |
| Duration (Hours) | % Germination in Untreated Grains | C. maxima Essential Oil (µg/mL) | ||
|---|---|---|---|---|
| 50 | 100 | 250 | ||
| 48 | 12.4 ± 1.0 | 13.1 ± 0.7 n | 12.2 ± 0.9 n | 11.9 ± 1.2 n |
| 72 | 30.7 ± 0.9 | 31.3 ± 0.6 n | 30.8 ± 1.2 n | 31.4 ± 1.2 n |
| 96 | 58.5 ± 1.5 | 59.2± 1.4 n | 59.9± 1.1 n | 58.4 ± 2.1 n |
| 120 | 80.6 ± 1.7 | 82.1 ± 1.6 n | 81.4 ± 1.4 n | 80.9 ± 2.1 n |
| 144 | 93.4 ± 2.1 | 92.5 ± 1.6 n | 93.1 ± 1.5 n | 92.4 ± 2.5 n |
| CMEO Dose (µg/mL) | % Mortality | Number of Fish Having Swimming Difficulty | Fishes Having Any Changes in Color | Total Time Spent on Top of Water (Seconds) |
|---|---|---|---|---|
| 0 | 0 | 0 | 0 | 27.6 ± 3.0 |
| 50 | 0 | 0 | 0 | 27.4 ± 3.0 |
| 100 | 0 | 0 | 0 | 29.7 ± 2.0 |
| 200 | 0 | 0 | 0 | 26.8 ± 3.0 |
| 250 | 0 | 0 | 0 | 29.5 ± 3.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Visakh, N.U.; Pathrose, B.; Narayanankutty, A.; Alfarhan, A.; Ramesh, V. Utilization of Pomelo (Citrus maxima) Peel Waste into Bioactive Essential Oils: Chemical Composition and Insecticidal Properties. Insects 2022, 13, 480. https://doi.org/10.3390/insects13050480
Visakh NU, Pathrose B, Narayanankutty A, Alfarhan A, Ramesh V. Utilization of Pomelo (Citrus maxima) Peel Waste into Bioactive Essential Oils: Chemical Composition and Insecticidal Properties. Insects. 2022; 13(5):480. https://doi.org/10.3390/insects13050480
Chicago/Turabian StyleVisakh, Naduvilthara U., Berin Pathrose, Arunaksharan Narayanankutty, Ahmed Alfarhan, and Varsha Ramesh. 2022. "Utilization of Pomelo (Citrus maxima) Peel Waste into Bioactive Essential Oils: Chemical Composition and Insecticidal Properties" Insects 13, no. 5: 480. https://doi.org/10.3390/insects13050480
APA StyleVisakh, N. U., Pathrose, B., Narayanankutty, A., Alfarhan, A., & Ramesh, V. (2022). Utilization of Pomelo (Citrus maxima) Peel Waste into Bioactive Essential Oils: Chemical Composition and Insecticidal Properties. Insects, 13(5), 480. https://doi.org/10.3390/insects13050480








