Identification and Distribution of Human-Biting Ticks in Northwestern Spain
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
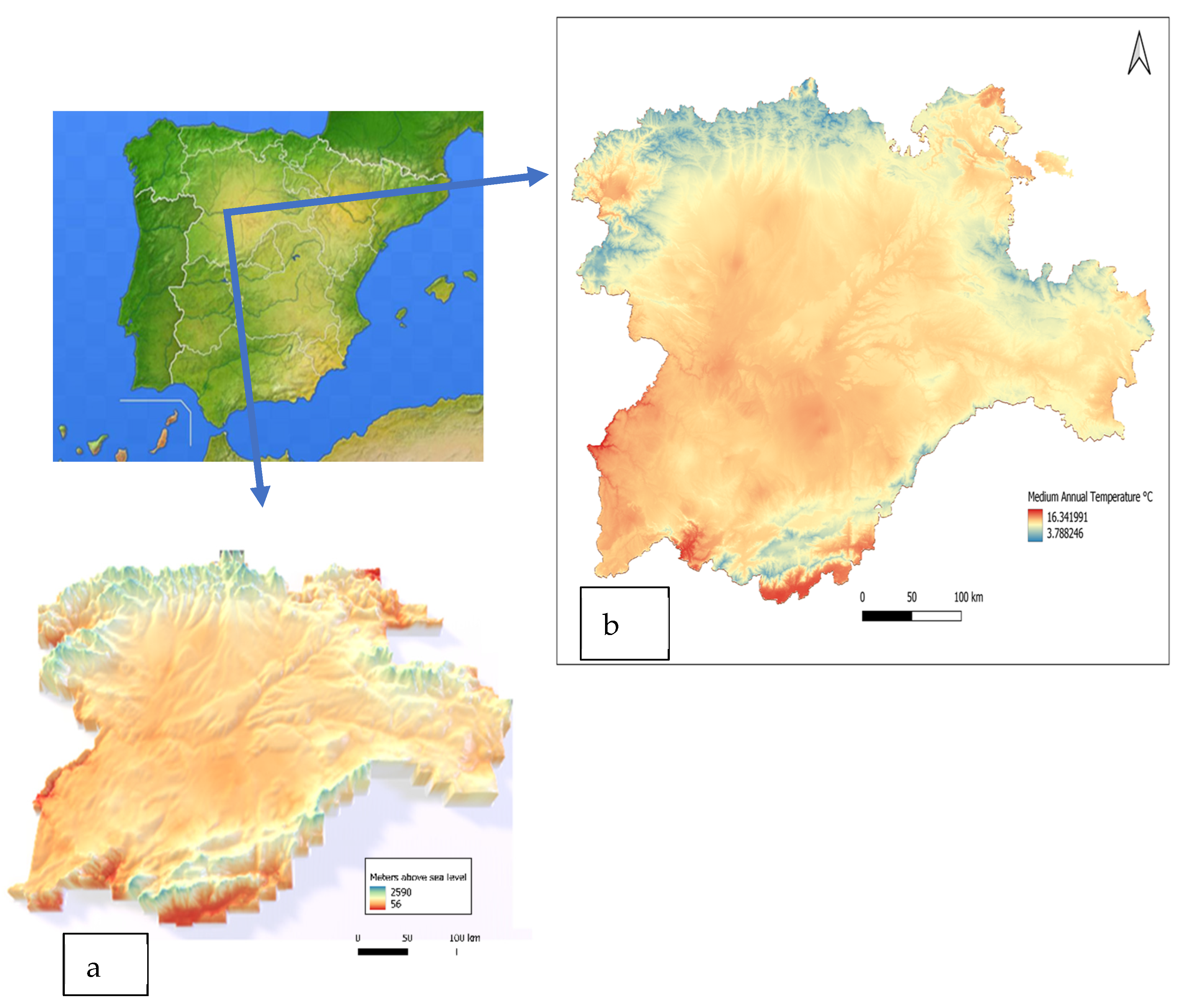
2.1. Study Site/Site Selection
2.2. Tick Collection and Identification
2.3. Geopositioning and Data Analysis
3. Results
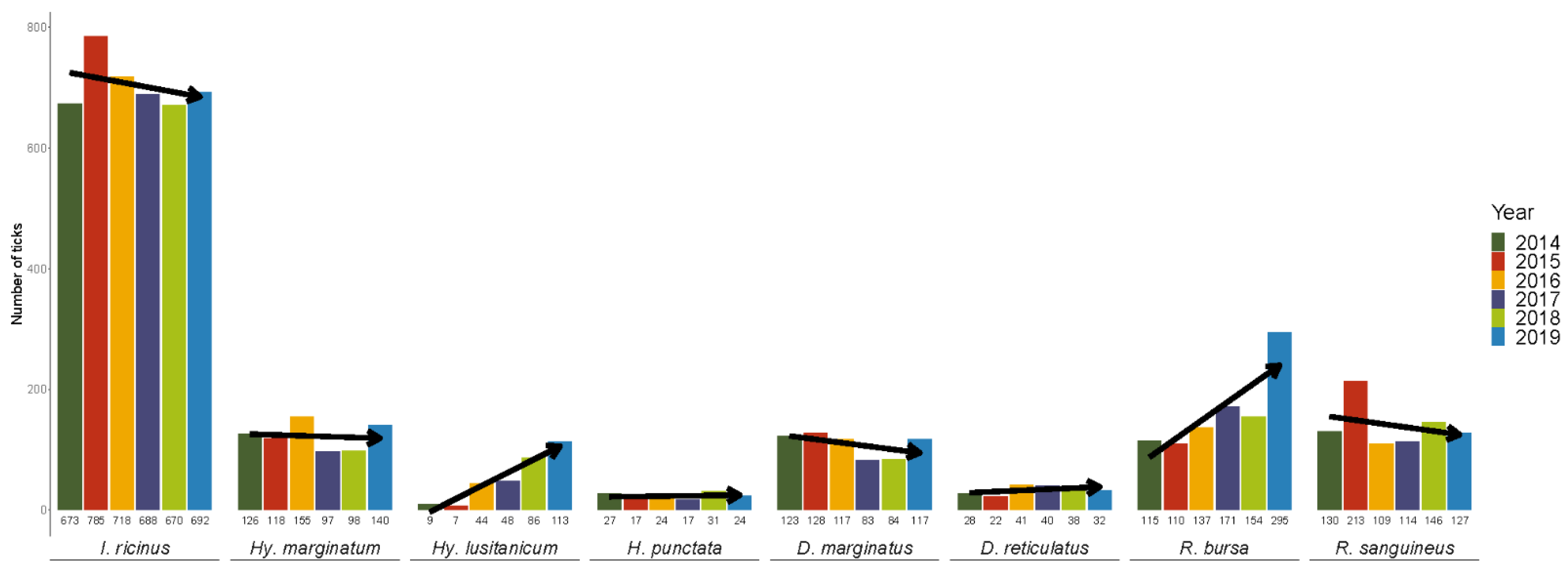
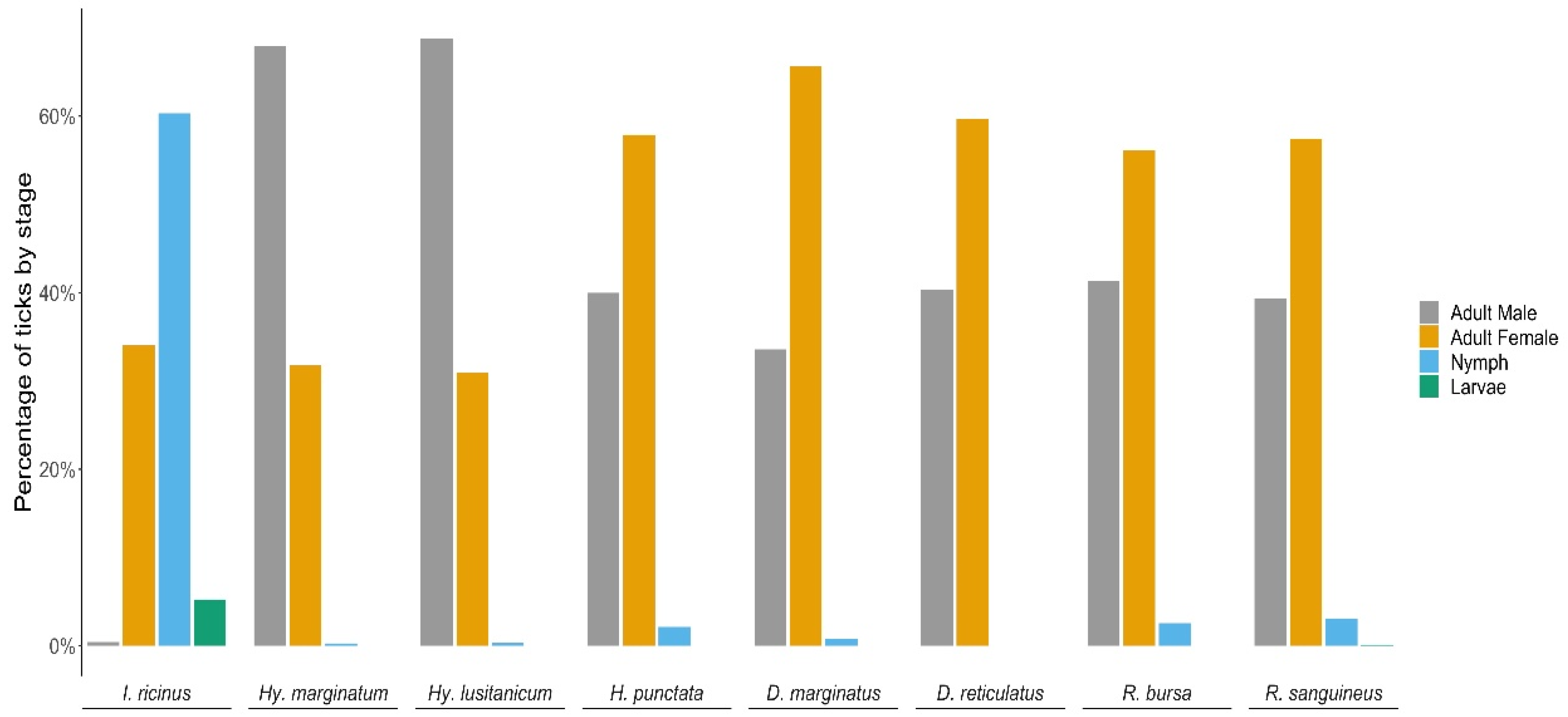
3.1. Tick Numbers, Species, Development Stages and Timeline
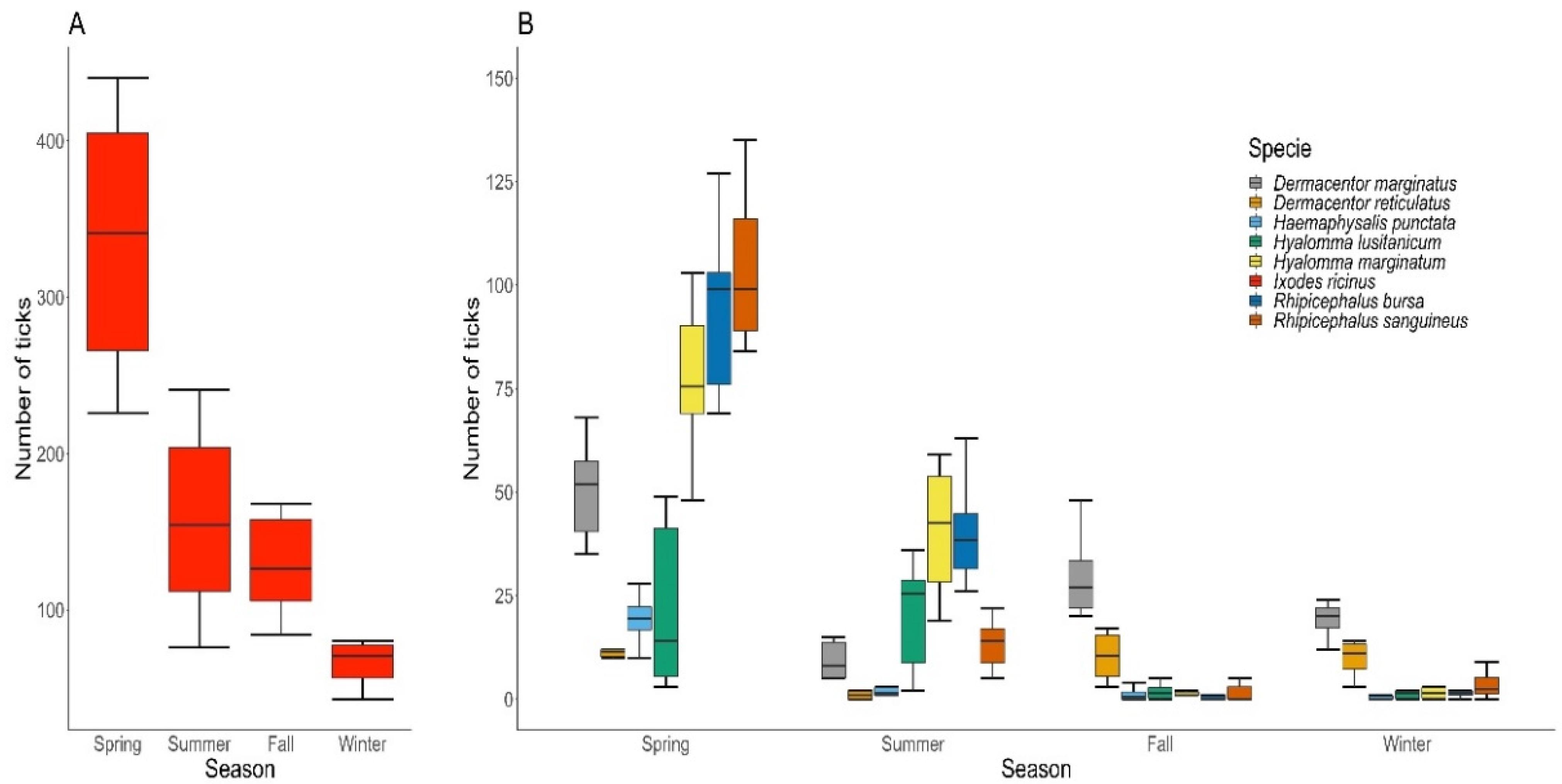
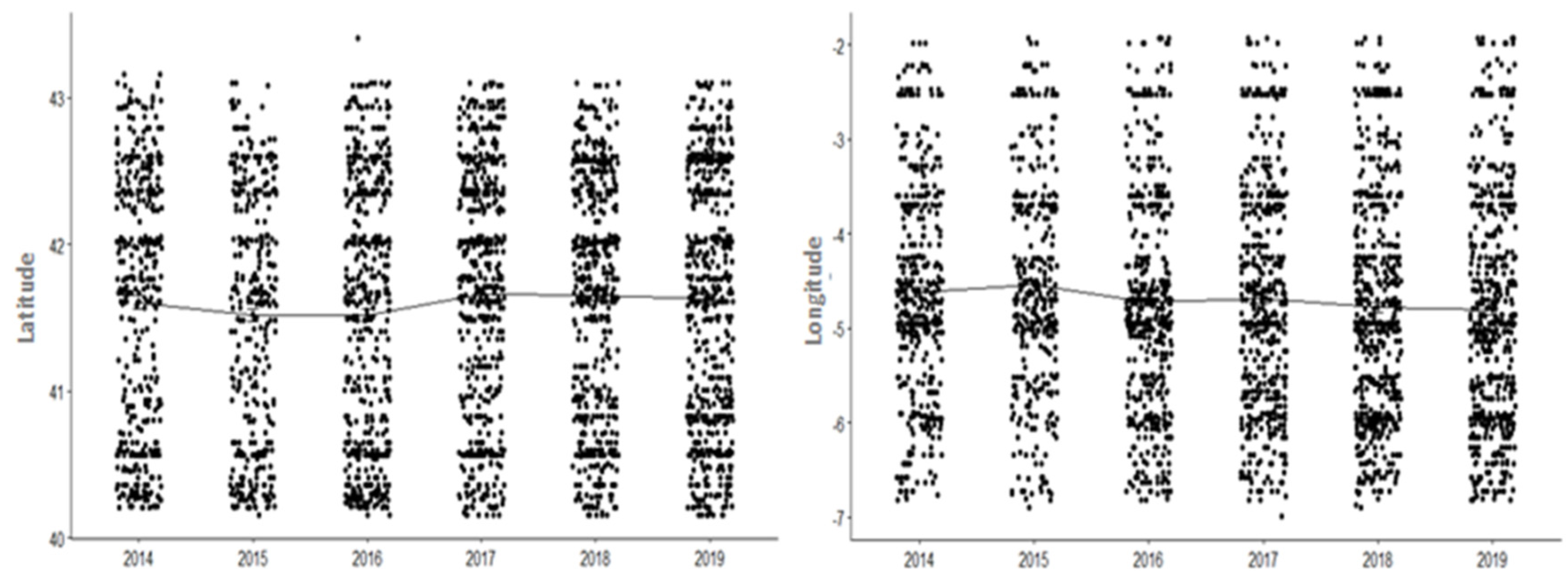
3.2. Seasonal and Geographical Distribution of Ticks
3.3. Epidemiological Characteristics of People Bitten by Ticks
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Estrada-Peña, A.; Jongejan, F. Ticks feeding on humans: A review of records on human-biting Ixodoidea with special reference to pathogen transmission. Exp. Appl. Acarol. 1999, 23, 685–715. [Google Scholar] [CrossRef] [PubMed]
- Yitayew, D.; Samuel, D. Tick borne hemoparasitic diseases of ruminants: A review. Adv. Biol. Res. 2015, 9, 210–224. [Google Scholar] [CrossRef]
- Ghosh, S.; Azhahianambi, P.; Yadav, M.P. Upcoming and future strategies of tick control: A review. J. Vector Borne Dis. 2007, 44, 79–89. [Google Scholar] [PubMed]
- Rochlin, I.; Toledo, A. Emerging tick-borne pathogens of public health importance: A mini-review. J. Med. Microbiol. 2020, 69, 781–791. [Google Scholar] [CrossRef]
- Dantas-Torres, F.; Chomel, B.B.; Otranto, D. Ticks and tick-borne diseases: A One Health perspective. Trends Parasitol. 2012, 28, 437–446. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; de la Fuente, J.; Cabezas-Cruz, A. Functional redundancy and ecological innovation shape the circulation of tick-transmitted pathogens. Front. Cell. Infect. Microbiol. 2017, 7, 234. [Google Scholar] [CrossRef]
- Gortazar, C.; Reperant, L.A.; Kuiken, T.; de la Fuente, J.; Boadella, M.; Martínez-Lopez, B.; Ruiz-Fons, F.; Estrada-Peña, A.; Drosten, C.; Medley, G.; et al. Crossing the Interspecies Barrier: Opening the Door to Zoonotic Pathogens. PLoS Pathog. 2014, 10, e1004129. [Google Scholar] [CrossRef] [Green Version]
- Estrada-Peña, A.; De La Fuente, J. The ecology of ticks and epidemiology of tick-borne viral diseases. Antivir. Res. 2014, 108, 104–128. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Martinez, J.M.; Sanchez Acedo, C.; Quilez, J.; Del Cacho, E. Phenology of the tick, Ixodes ricinus, in its southern distribution range (central Spain). Med. Vet. Entomol. 2004, 18, 387–397. [Google Scholar] [CrossRef]
- Gilbert, L. Altitudinal patterns of tick and host abundance: A potential role for climate change in regulating tick-borne diseases? Oecologia 2010, 162, 217–225. [Google Scholar] [CrossRef]
- Rizzoli, A.; Silaghi, C.; Obiegala, A.; Rudolf, I.; Hubálek, Z.; Földvári, G.; Plantard, O.; Vayssier-Taussat, M.; Bonnet, S.; Špitalská, E.; et al. Ixodes ricinus and its transmitted pathogens in urban and peri-urban areas in Europe: New hazards and relevance for public health. Front. Public Health 2014, 2, 251. [Google Scholar] [CrossRef] [PubMed]
- Boulanger, N.; Boyer, P.; Talagrand-Reboul, E.; Hansmann, Y. Ticks and tick-borne diseases. Med. Mal. Infect. 2019, 49, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Cull, B.; Pietzsch, M.E.; Gillingham, E.L.; McGinley, L.; Medlock, J.M.; Hansford, K.M. Seasonality and anatomical location of human tick bites in the United Kingdom. Zoonoses Public Health 2019, 67, 112–121. [Google Scholar] [CrossRef]
- Fernández-Soto, P. Garrapatas que Parasitan a Personas en Castilla y León, Determinación por Serología de su Parasitismo y Detección Molecular de los Patógenos que Albergan; Universidad de Salamanca: Salamanca, Spain, 2003. [Google Scholar]
- El-Sayed, A.; Kamel, M. Climatic changes and their role in emergence and re-emergence of diseases. Environ. Sci. Pollut. Res. 2020, 27, 22336–22352. [Google Scholar] [CrossRef] [PubMed]
- Barandika, J.F.; Hurtado, A.; García-Sanmartín, J.; Juste, R.A.; Anda, P.; García-Pérez, A.L. Prevalence of tick-borne zoonotic bacteria in questing adult ticks from Northern Spain. Vector-Borne Zoonotic Dis. 2008, 8, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Fernández de Mera, I.G.; Ruiz-Fons, F.; de la Fuente, G.; Mangold, A.J.; Gortázar, C.; de la Fuente, J. Spotted fever group rickettsiaein questing ticks, central Spain. Emerg. Infect. Dis. 2013, 19, 1163–1165. [Google Scholar] [CrossRef] [Green Version]
- Requena-García, F.; Cabrero-Sañudo, F.; Olmeda-García, S.; González, J.; Valcárcel, F. Influence of environmental temperature and humidity on questing ticks in central Spain. Exp. Appl. Acarol. 2017, 71, 277–290. [Google Scholar] [CrossRef]
- Remesar, S.; Fernández, P.D.; Venzal, J.M.; Pérez-Creo, A.; Prieto, A.; Estrada-Peña, A.; López, C.M.; Panadero, R.; Fernández, G.; Díez-Baños, P.; et al. Tick species diversity and population dynamics of Ixodes ricinus in Galicia (north-western Spain). Ticks Tick. Borne. Dis. 2019, 10, 132–137. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Roura, X.; Sainz, A.; Miró, G.; Solano-Gallego, L. Species of ticks and carried pathogens in owned dogs in Spain: Results of a one-year national survey. Ticks Tick. Borne. Dis. 2017, 8, 443–452. [Google Scholar] [CrossRef]
- Gil-Collado, J.; Guillén, J.L.; Zapatero, L.M. Claves para la identificación de los Ixodoidea españoles (adultos). Rev. Ibérica Parasitol. 1979, 39, 107–118. [Google Scholar]
- Estrada-Peña, A.; Mihalca, A.D.; Petney, T.N. Ticks of Europe and North Africa: A Guide to Species Identification; Springer International Publishing: Cham, Switzerland, 2017; ISBN 9783319637594. [Google Scholar]
- Apanaskevich, D.A.; Santos-Silva, M.M.; Horak, I.G. The genus Hyalomma Koch, 1844. IV. Redescription of all parasitic stages of H. (Euhyalomma) lusitanicum Koch, 1844 and the adults of H. (E.) franchinii Tonelli Rondelli, 1932 (Acari: Ixodidae) with a first description of its immature stages. Folia Parasitol. 2008, 55, 61–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nava, S.; Beati, L.; Venzal, J.M.; Labruna, M.B.; Szabó, M.P.J.; Petney, T.; Saracho-Bottero, M.N.; Tarragona, E.L.; Dantas-Torres, F.; Silva, M.M.S.; et al. Rhipicephalus sanguineus (Latreille, 1806): Neotype designation, morphological re-description of all parasitic stages and molecular characterization. Ticks Tick. Borne. Dis. 2018, 9, 1573–1585. [Google Scholar] [CrossRef] [PubMed]
- QGIS.org. QGIS Geographic Information System; QGIS Association: Geneva, Switzerland, 2022. [Google Scholar]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Merino, F.J.; Nebreda, T.; Serrano, J.L.; Fernández-Soto, P.; Encinas, A.; Pérez-Sánchez, R. Tick species and tick-borne infections identified in population from a rural area of Spain. Epidemiol. Infect. 2005, 133, 943–949. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Soto, P.; Pérez-Sánchez, R.; Álamo-Sanz, R.; Encinas-Grandes, A. Spotted fever group rickettsiae in ticks feeding on humans in northwestern Spain: Is Rickettsia conorii vanishing? Ann. N. Y. Acad. Sci. 2006, 1078, 331–333. [Google Scholar] [CrossRef] [PubMed]
- Estrada-Peña, A.; Palomar, A.M.; Santibáñez, P.; Sánchez, N.; Habela, M.A.; Portillo, A.; Romero, L.; Oteo, J.A. Crimean-Congo Hemorrhagic Fever Virus in Ticks, Southwestern Europe, 2010. Emerg. Infect. Dis. 2012, 18, 179–180. [Google Scholar] [CrossRef] [PubMed]
- Monsalve Arteaga, L.; Muñoz Bellido, J.L.; Negredo, A.I.; García Criado, J.; Vieira Lista, M.C.; Sánchez Serrano, J.Á.; Vicente Santiago, M.B.; López Bernús, A.; de Ory Manchón, F.; Sánchez Seco, M.P.; et al. New circulation of genotype V of Crimean-Congo haemorrhagic fever virus in humans from Spain. PLoS Negl. Trop. Dis. 2021, 15, e0009197. [Google Scholar] [CrossRef]
- Monsalve-Arteaga, L.; Alonso-Sardón, M.; Muñoz Bellido, J.L.; Vicente Santiago, M.B.; Vieira Lista, M.C.; López Abán, J.; Muro, A.; Belhassen-García, M. Seroprevalence of Crimean-Congo hemorrhagic fever in humans in the World Health Organization European region: A systematic review. PLoS Negl. Trop. Dis. 2020, 14, e0008094. [Google Scholar] [CrossRef]
- Espí, A.; Del Cerro, A.; Somoano, A.; García, V.; Prieto, J.M.; Barandika, J.F.; García-Pérez, A.L. Borrelia burgdorferi sensu lato prevalence and diversity in ticks and small mammals in a Lyme borreliosis endemic Nature Reserve in North-Western Spain. Incidence in surrounding human populations. Enferm. Infecc. Microbiol. Clin. (Engl. Ed.) 2017, 35, 563–568. [Google Scholar] [CrossRef]
- Barandika, J.F.; Olmeda, S.A.; Casado-Nistal, M.A.; Hurtado, A.; Juste, R.A.; Valcárcel, F.; Anda, P.; García-Pérez, A.L. Differences in questing tick species distribution between Atlantic and continental climate regions in Spain. J. Med. Entomol. 2011, 48, 13–19. [Google Scholar] [CrossRef]
- Remesar, S.; Díaz, P.; Portillo, A.; Santibáñez, S.; Prieto, A.; Díaz-Cao, J.M.; López, C.M.; Panadero, R.; Fernández, G.; Díez-Baños, P.; et al. Prevalence and molecular characterization of Rickettsia spp. in questing ticks from north-western Spain. Exp. Appl. Acarol. 2019, 79, 267–278. [Google Scholar] [CrossRef]
- Lernout, T.; De Regge, N.; Tersago, K.; Fonville, M.; Suin, V.; Sprong, H. Prevalence of pathogens in ticks collected from humans through citizen science in Belgium. Parasit. Vectors 2019, 12, 550. [Google Scholar] [CrossRef] [PubMed]
- Cull, B.; Pietzsch, M.E.; Hansford, K.M.; Gillingham, E.L.; Medlock, J.M. Surveillance of British ticks: An overview of species records, host associations, and new records of Ixodes ricinus distribution. Ticks Tick. Borne. Dis. 2018, 9, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Wilhelmsson, P.; Lindblom, P.; Fryland, L.; Nyman, D.; Jaenson, T.G.; Forsberg, P.; Lindgren, P.E. Ixodes ricinus ticks removed from humans in Northern Europe: Seasonal pattern of infestation, attachment sites and duration of feeding. Parasites Vectors 2013, 6, 362. [Google Scholar] [CrossRef] [PubMed]
- Hügli, D.; Moret, J.; Rais, O.; Moosmann, Y.; Erard, P.; Malinverni, R.; Gern, L. Tick bites in a Lyme borreliosis highly endemic area in Switzerland. Int. J. Med. Microbiol. 2009, 299, 155–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robertson, J.N.; Gray, J.S.; Stewart, P. Tick bite and Lyme borreliosis risk at a recreational site in England. Eur. J. Epidemiol. 2000, 16, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Pańczuk, A.; Tokarska-Rodak, M.; Mikuľáková, W.; Kendrová, L.; Magurová, D. Exposure to ticks and undertaking lyme borreliosisprevention activities among students from Poland and Slovakia. Ann. Agric. Environ. Med. 2019, 26, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Briciu, V.T.; Titilincu, A.; Ţăţulescu, D.F.; Cârstina, D.; Lefkaditis, M.; Mihalca, A.D. First survey on hard ticks (Ixodidae) collected from humans in Romania: Possible risks for tick-borne diseases. Exp. Appl. Acarol. 2011, 54, 199–204. [Google Scholar] [CrossRef]
- Weisshaar, E.; Schaefer, A.; Scheidt, R.R.W.; Bruckner, T.; Apfelbacher, C.J.; Diepgen, T.L. Epidemiology of tick bites and borreliosis in children attending kindergarten or so-called “forest kindergarten” in southwest Germany. J. Investig. Dermatol. 2006, 126, 584–590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manfredi, M.T.; Dini, V.; Piacenza, S.; Genchi, C. Tick species parasitizing people in an area endemic for tick-borne diseases in north-western Italy. Parassitologia 1999, 41, 555–560. [Google Scholar]
- Lindblom, A.; Wallménius, K.; Sjöwall, J.; Fryland, L.; Wilhelmsson, P.; Lindgren, P.E.; Forsberg, P.; Nilsson, K. Prevalence of Rickettsia spp. in ticks and serological and clinical outcomes in tick-bitten individuals in Sweden and on the Åland Islands. PLoS ONE 2016, 11, e0166653. [Google Scholar] [CrossRef] [Green Version]
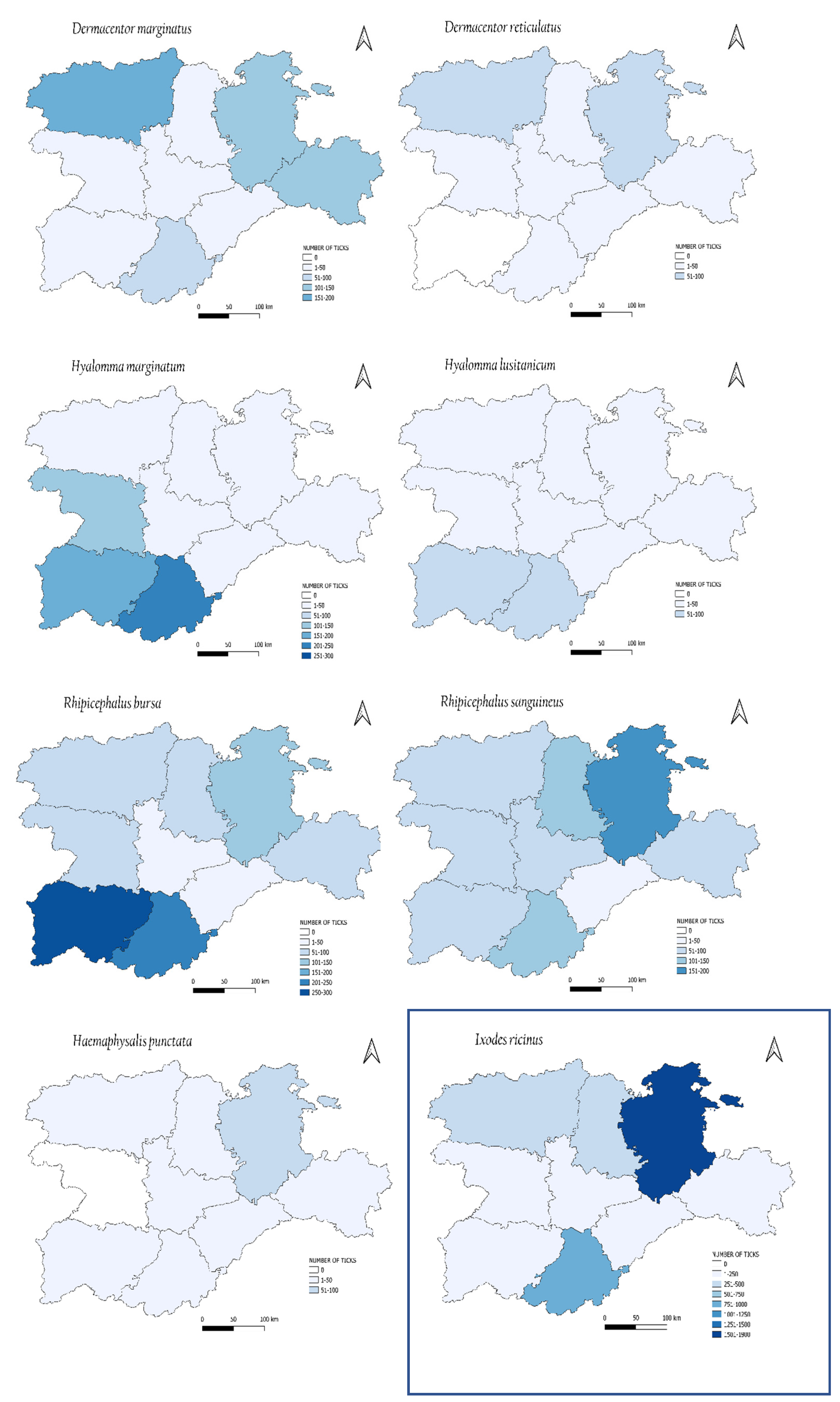
| Tick Species | Numbers of Collected Ticks | ||||
|---|---|---|---|---|---|
| Larvae | Nymphs | Male | Female | Total | |
| Ixodes ricinus | 221 | 2549 | 18 | 1438 | 4226 |
| Rhipicephalus bursa | 25 | 406 | 551 | 982 | |
| Rhipicephalus sanguineus | 1 | 26 | 330 | 482 | 839 |
| Hyalomma marginatum | 2 | 499 | 233 | 734 | |
| Dermacentor marginatus | 5 | 219 | 428 | 652 | |
| Hyalomma lusitanicum | 1 | 211 | 95 | 307 | |
| Dermacentor reticulatus | 81 | 120 | 201 | ||
| Haemaphysalis punctata | 3 | 56 | 81 | 140 | |
| Total | 222 | 2611 | 1820 | 3428 | 8081 |
| Characteristics of Patients | Numbers and Species of Ticks Collected from Patients | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| I.ricinus | R. bursa | R. sanguineus | D. marginatus | D. reticulatus | Hy. marginatum | Hy. lusitanicum | H. punctata | Total | |
| Sex: men (%) women (%) | 2247 (65.75%) 1170 (34.24%) | 633 (73.43%) 229 (26.56%) | 411(59.13%) 284 (40.86%) | 292 (50.78%) 283 (49.21%) | 100 (56.81%) 76 (43.18%) | 495 (75.68%) 159 (24.31%) | 218 (75.69%) 70 (24.30%) | 7 (10.76%) 58 (89.23%) | 4403 (65.40%) 2329 (34.59%) |
| Total | 3417 | 862 | 695 | 575 | 176 | 654 | 288 | 65 | 6732 |
| Age: | |||||||||
| 0–14 | 673 | 163 | 214 | 126 | 34 | 29 | 13 | 44 | 1296 (22.59%) |
| 15–35 | 505 | 140 | 108 | 51 | 40 | 81 | 35 | 12 | 972 (16.94%) |
| 36–55 | 840 | 190 | 101 | 134 | 37 | 192 | 61 | 21 | 1576 (27.47%) |
| 55+ | 882 | 228 | 147 | 185 | 26 | 269 | 124 | 31 | 1892 (32.98%) |
| Total | 2900 | 721 | 570 | 496 | 137 | 571 | 233 | 108 | 5736 |
| Adults (%) Children (%) | 2478 (75.66%) 797 (24.33%) | 615 (75.64%) 198 (24.35%) | 408 (60.80%) 263 (39.19%) | 403 (73.54%) 145 (26.45%) | 124 (75.60%) 40 (24.39%) | 580 (94.61%) 33 (5.38%) | 240 (93.75%) 16 (6.25%) | 70 (58.33%) 50 (41.66%) | 4918 (76.13%) 1542 (23.86%) |
| Total | 3275 | 813 | 671 | 548 | 164 | 613 | 256 | 120 | 6460 |
| Site of bites: | Adults/Children | Adults/Children | Adults/Children | Adults/Children | Adults/Children | Adults/Children | Adults/Children | Adults/Children | Adults/Children |
| Head | 42/132 | 54/94 | 74/151 | 182/102 | 28/19 | 17/9 | 11/5 | 16/32 | 424 (43.80%)/544 (56.19%) |
| Neck | 37/59 | 43/19 | 46/33 | 26/8 | 13/3 | 29/9 | 15/5 | 6/7 | 215 (60.05%)/143 (39.94%) |
| Thorax | 458/159 | 120/27 | 60/22 | 20/7 | 9/6 | 100/19 | 50/9 | 8/2 | 825 (76.67%)/251 (23.32) |
| Upper limbs | 284/76 | 41/12 | 32/14 | 20/4 | 12/8 | 36/8 | 11/2 | 6/1 | 442 (77.95%)/125 (22.04%) |
| Lower limbs | 681/188 | 98/9 | 39/9 | 19/5 | 17/8 | 125/19 | 52/8 | 6/1 | 1037 (84.65%)/188 (15.34) |
| Back | 149/47 | 46/15 | 31/11 | 14/4 | 8/3 | 44/8 | 21/2 | 4/1 | 317 (76.69%)/91 (22.30%) |
| Pelvis | 147/44 | 42/14 | 16/5 | 4/3 | 0/0 | 69/8 | 18/5 | 5/1 | 301 (79%)/80 (20.99%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vieira Lista, M.C.; Belhassen-García, M.; Vicente Santiago, M.B.; Sánchez-Montejo, J.; Pedroza Pérez, C.; Monsalve Arteaga, L.C.; Herrador, Z.; del Álamo-Sanz, R.; Benito, A.; Soto López, J.D.; et al. Identification and Distribution of Human-Biting Ticks in Northwestern Spain. Insects 2022, 13, 469. https://doi.org/10.3390/insects13050469
Vieira Lista MC, Belhassen-García M, Vicente Santiago MB, Sánchez-Montejo J, Pedroza Pérez C, Monsalve Arteaga LC, Herrador Z, del Álamo-Sanz R, Benito A, Soto López JD, et al. Identification and Distribution of Human-Biting Ticks in Northwestern Spain. Insects. 2022; 13(5):469. https://doi.org/10.3390/insects13050469
Chicago/Turabian StyleVieira Lista, María Carmen, Moncef Belhassen-García, María Belén Vicente Santiago, Javier Sánchez-Montejo, Carlos Pedroza Pérez, Lía Carolina Monsalve Arteaga, Zaida Herrador, Rufino del Álamo-Sanz, Agustin Benito, Julio David Soto López, and et al. 2022. "Identification and Distribution of Human-Biting Ticks in Northwestern Spain" Insects 13, no. 5: 469. https://doi.org/10.3390/insects13050469
APA StyleVieira Lista, M. C., Belhassen-García, M., Vicente Santiago, M. B., Sánchez-Montejo, J., Pedroza Pérez, C., Monsalve Arteaga, L. C., Herrador, Z., del Álamo-Sanz, R., Benito, A., Soto López, J. D., & Muro, A. (2022). Identification and Distribution of Human-Biting Ticks in Northwestern Spain. Insects, 13(5), 469. https://doi.org/10.3390/insects13050469







