Monitoring Trends in Distribution and Seasonality of Medically Important Ticks in North America Using Online Crowdsourced Records from iNaturalist
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Region
2.2. Data Analysis
3. Results
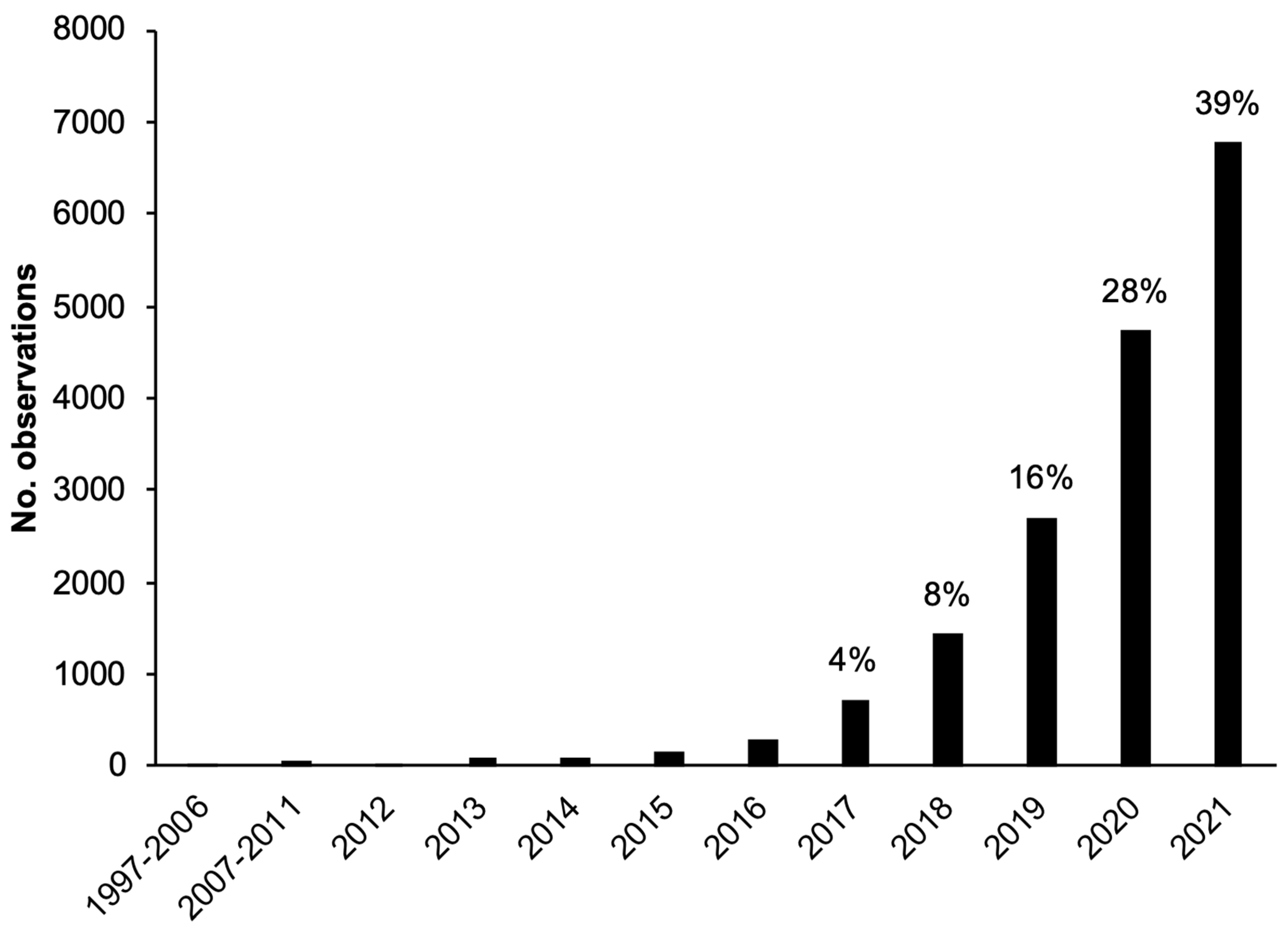
3.1. Overview of Tick Observations in Study Region
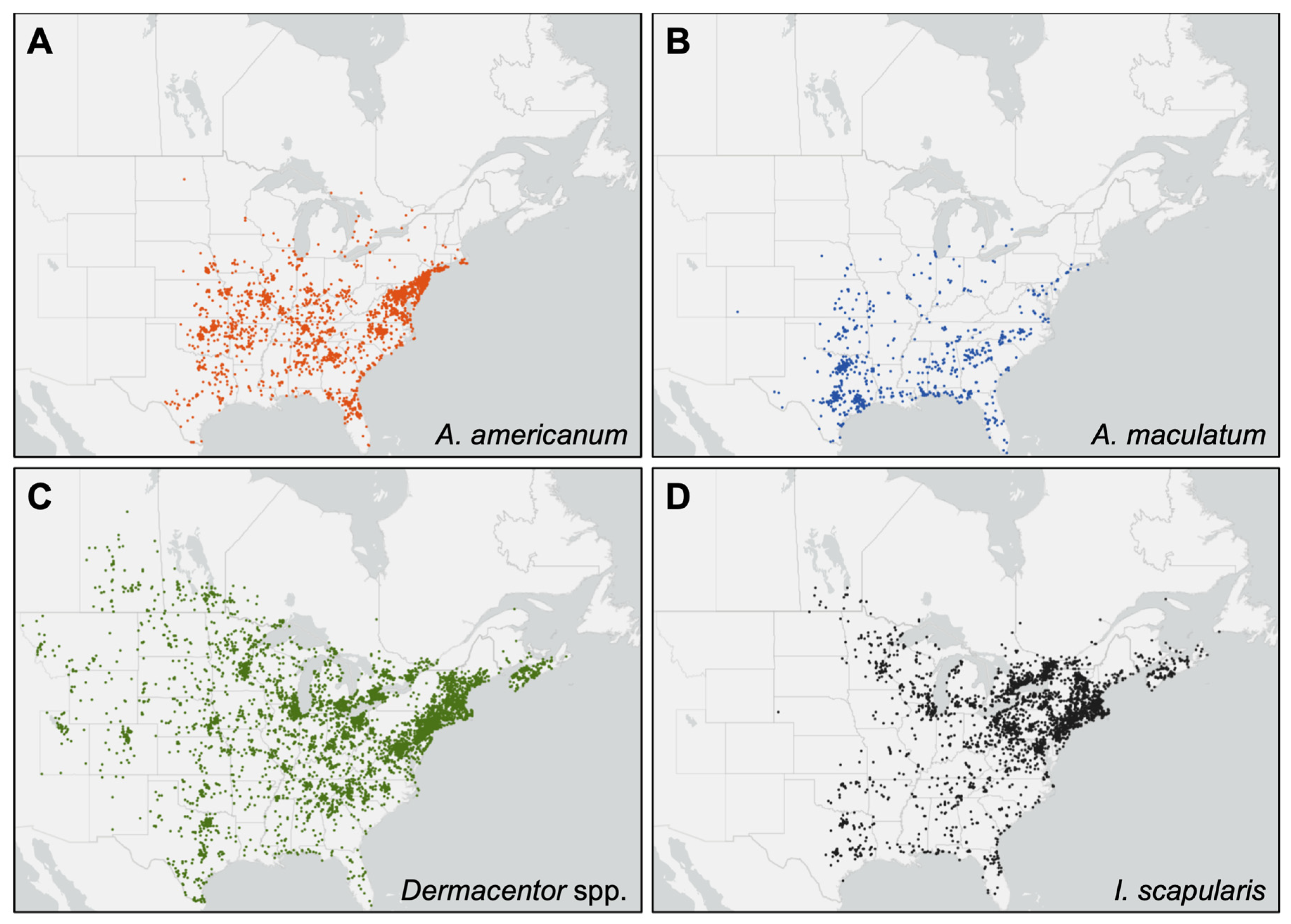
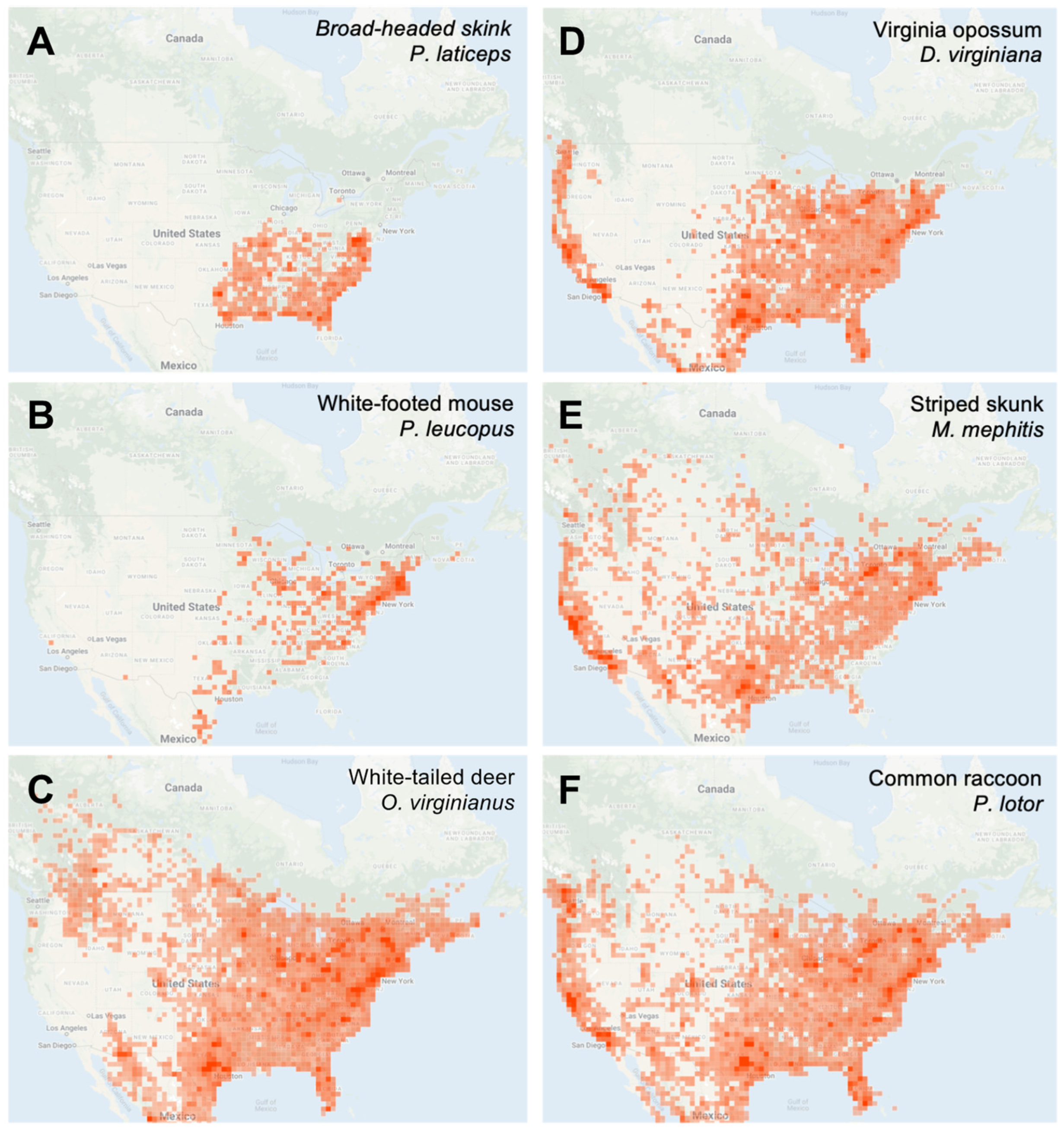
3.2. Distribution of iNaturalist Observations of Target Tick Species
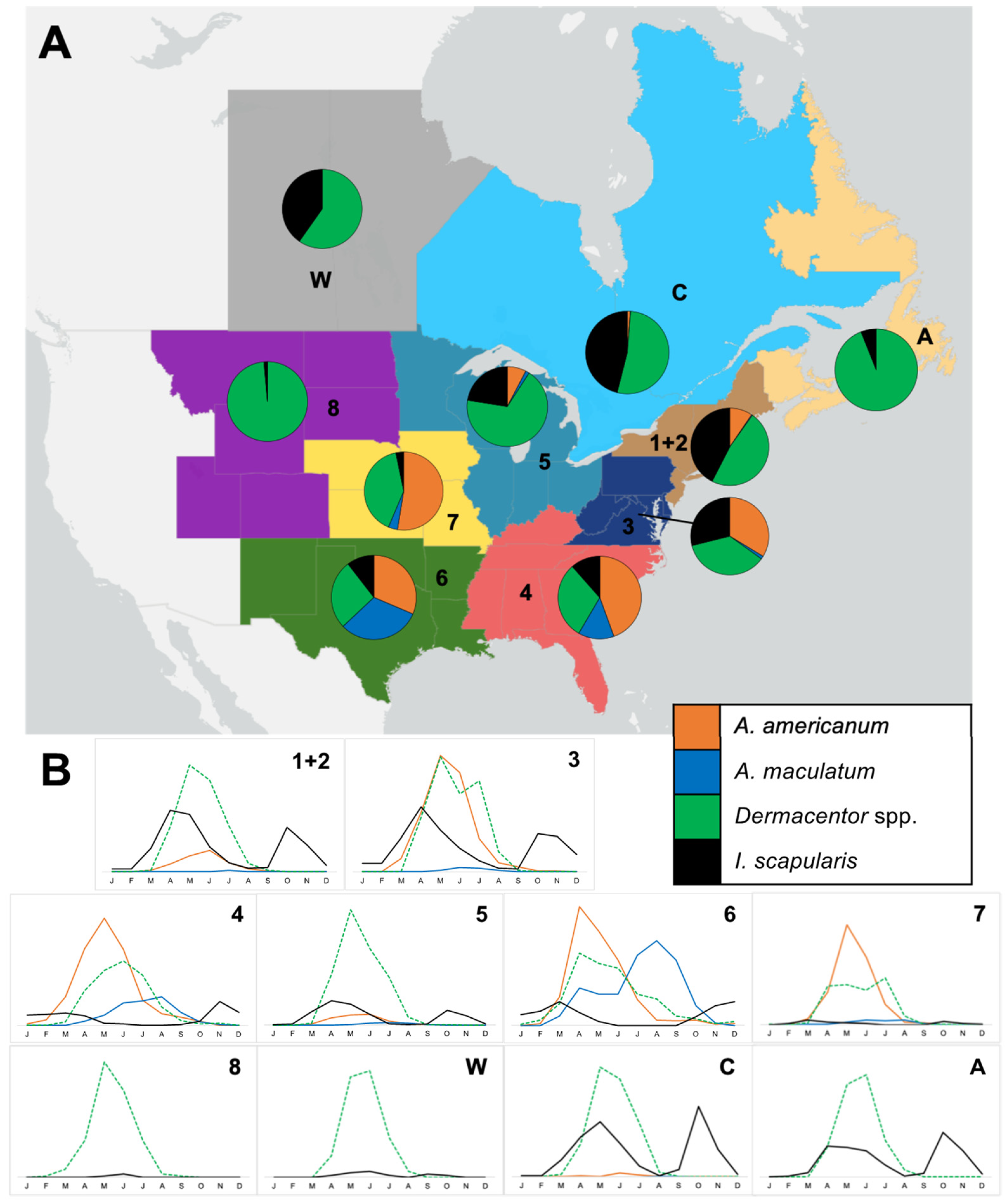
3.3. Seasonality of Tick Observations
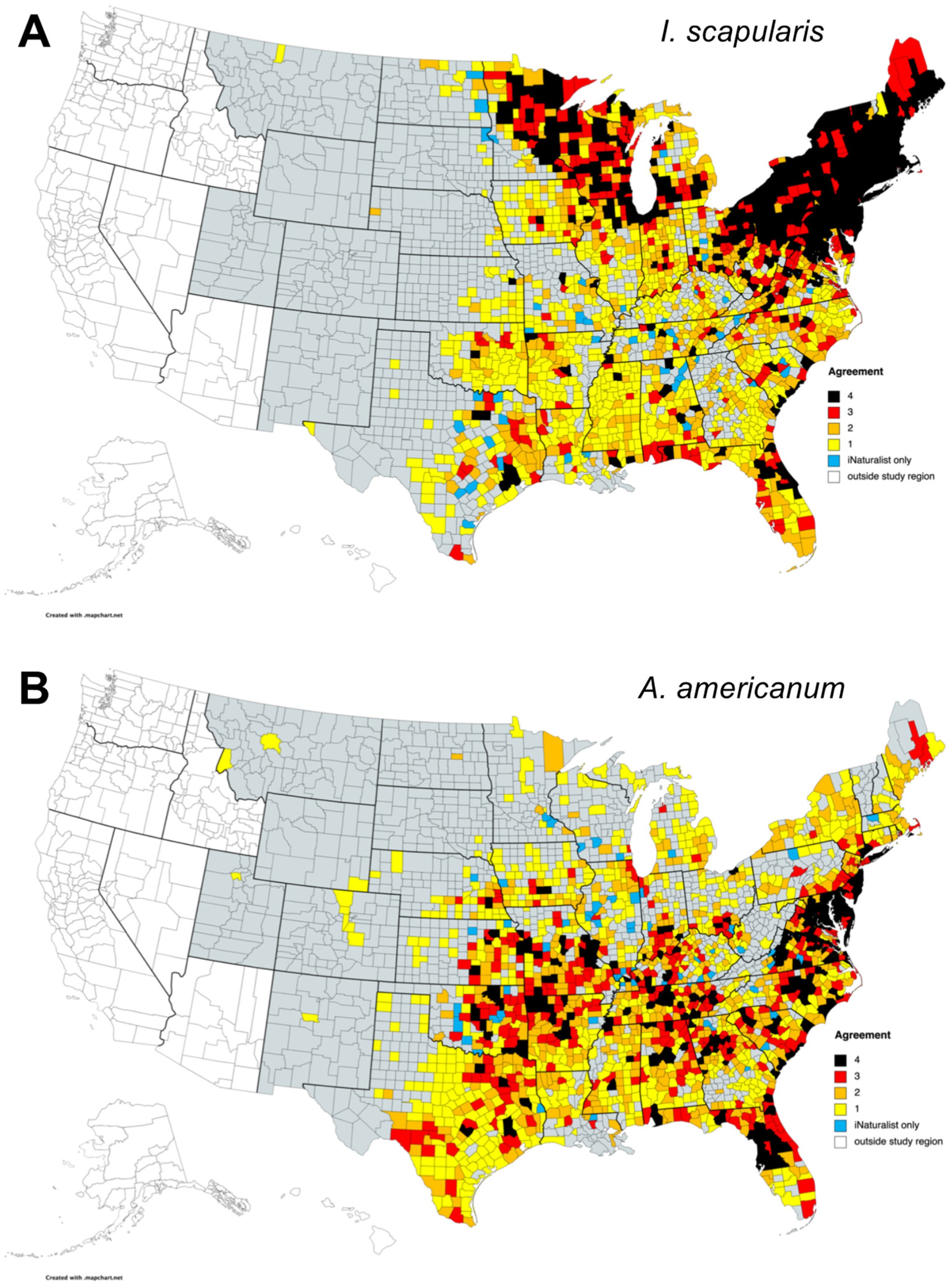
3.4. Comparison of County-Level Distributions of I. Scapularis and A. Americanum
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eisen, R.J.; Kugeler, K.J.; Eisen, L.; Beard, C.B.; Paddock, C.D. Tick-Borne Zoonoses in the United States: Persistent and Emerging Threats to Human Health. ILAR J. 2017, 58, 319–335. [Google Scholar] [CrossRef] [PubMed]
- Nelder, M.; Wijayasri, S.; Russell, C.; Johnson, K.; Marchand-Austin, A.; Cronin, K.; Johnson, S.; Badiani, T.; Patel, S.; Sider, D. The Continued Rise of Lyme Disease in Ontario, Canada: 2017. Can. Commun. Dis. Rep. 2018, 44, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Public Health Canada Lyme Disease: Monitoring. Available online: https://www.canada.ca/en/public-health/services/diseases/lyme-disease/surveillance-lyme-disease.html (accessed on 25 March 2022).
- Kugeler, K.J.; Schwartz, A.M.; Delorey, M.J.; Mead, P.S.; Hinckley, A.F. Estimating the Frequency of Lyme Disease Diagnoses, United States, 2010–2018. Emerg. Infect. Dis. 2021, 27, 616–619. [Google Scholar] [CrossRef] [PubMed]
- Eisen, R.J.; Eisen, L. The Blacklegged Tick, Ixodes scapularis: An Increasing Public Health Concern. Trends Parasitol. 2018, 34, 295–309. [Google Scholar] [CrossRef]
- Eisen, R.J.; Eisen, L.; Beard, C.B. County-Scale Distribution of Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae) in the Continental United States. J. Med. Entomol. 2016, 53, 349–386. [Google Scholar] [CrossRef]
- Ripoche, M.; Gasmi, S.; Adam-Poupart, A.; Koffi, J.K.; Lindsay, L.R.; Ludwig, A.; Milord, F.; Ogden, N.H.; Thivierge, K.; Leighton, P.A. Passive Tick Surveillance Provides an Accurate Early Signal of Emerging Lyme Disease Risk and Human Cases in Southern Canada. J. Med. Entomol. 2018, 55, 1016–1026. [Google Scholar] [CrossRef]
- Springer, Y.P.; Eisen, L.; Beati, L.; James, A.M.; Eisen, R.J. Spatial Distribution of Counties in the Continental United States with Records of Occurrence of Amblyomma americanum (Ixodida: Ixodidae). J. Med. Entomol. 2014, 51, 342–351. [Google Scholar] [CrossRef]
- Rochlin, I.; Egizi, A.; Lindström, A. The Original Scientific Description of the Lone Star Tick (Amblyomma americanum, Acari: Ixodidae) and Implications for the Species’ Past and Future Geographic Distributions. J. Med. Entomol. 2022, 59, 412–420. [Google Scholar] [CrossRef]
- Dahlgren, S.F.; Paddock, C.D.; Springer, Y.P.; Eisen, R.J.; Behravesh, C.B. Expanding Range of Amblyomma americanum and Simultaneous Changes in the Epidemiology of Spotted Fever Group Rickettsiosis in the United States. Am. J. Trop. Med. Hyg. 2016, 94, 35–42. [Google Scholar] [CrossRef]
- Billeter, S.A.; Blanton, H.L.; Little, S.E.; Levy, M.G.; Breitschwerdt, E.B. Detection of “Rickettsia amblyommii” in Association with a Tick Bite Rash. Vector-Borne Zoonotic Dis. 2007, 7, 607–610. [Google Scholar] [CrossRef]
- Yen, W.-Y.; Stern, K.; Mishra, S.; Helminiak, L.; Sanchez-Vicente, S.; Kim, H.K. Virulence Potential of Rickettsia amblyommatis for Spotted Fever Pathogenesis in Mice. Pathog. Dis. 2021, 79, 79. [Google Scholar] [CrossRef] [PubMed]
- Snellgrove, A.N.; Krapiunaya, I.; Scott, P.; Levin, M.L. Assessment of the Pathogenicity of Rickettsia amblyommatis, Rickettsia bellii, and Rickettsia montanensis in a Guinea Pig Model. Vector-Borne Zoonotic Dis. 2021, 21, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Levin, M.L.; Zemtsova, G.E.; Killmaster, L.F.; Snellgrove, A.; Schumacher, L.B.M. Vector Competence of Amblyomma americanum (Acari: Ixodidae) for Rickettsia rickettsii. Ticks Tick-Borne Dis. 2017, 8, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Breitschwerdt, E.B.; Hegarty, B.C.; Maggi, R.G.; Lantos, P.M.; Aslett, D.M.; Bradley, J.M. Rickettsia rickettsii Transmission by a Lone Star Tick, North Carolina. Emerg. Infect. Dis. 2011, 17, 873–875. [Google Scholar] [CrossRef] [PubMed]
- Goddard, J. Experimental Infection of Lone Star Ticks, Amblyomma americanum (L.), with Rickettsia parkeri and Exposure of Guinea Pigs to the Agent. J. Med. Entomol. 2003, 40, 686–689. [Google Scholar] [CrossRef] [PubMed]
- Wright, C.L.; Sonenshine, D.E.; Gaff, H.D.; Hynes, W.L. Rickettsia parkeri Transmission to Amblyomma americanum by Cofeeding with Amblyomma maculatum (Acari: Ixodidae) and Potential for Spillover. J. Med. Entomol. 2015, 52, 1090–1095. [Google Scholar] [CrossRef]
- Paddock, C.D.; Goddard, J. The Evolving Medical and Veterinary Importance of the Gulf Coast Tick (Acari: Ixodidae). J. Med. Entomol. 2015, 52, 230–252. [Google Scholar] [CrossRef]
- Molaei, G.; Little, E.A.H.; Khalil, N.; Ayres, B.N.; Nicholson, W.L.; Paddock, C.D. Established Population of the Gulf Coast Tick, Amblyomma maculatum (Acari: Ixodidae), Infected with Rickettsia parkeri (Rickettsiales: Rickettsiaceae), in Connecticut. J. Med. Entomol. 2021, 58, 1459–1462. [Google Scholar] [CrossRef]
- Phillips, V.C.; Zieman, E.A.; Kim, C.H.; Stone, C.M.; Tuten, H.C.; Jiménez, F.A. Documentation of the Expansion of the Gulf Coast Tick (Amblyomma maculatum) and Rickettsia parkeri: First Report in Illinois. J. Parasitol. 2020, 106, 9–13. [Google Scholar] [CrossRef]
- Ramírez-Garofalo, J.R.; Curley, S.R.; Field, C.E.; Hart, C.E.; Thangamani, S. Established Populations of Rickettsia parkeri- Infected Amblyomma maculatum Ticks in New York City, New York, USA. Vector-Borne Zoonotic Dis. 2022, 22, 184–187. [Google Scholar] [CrossRef]
- Wojan, C.; Thrasher, T.; Lacey, E.; Clay, K. Distribution, Dynamics, and Diversity of Questing Ticks in the Lower Midwest. J. Med. Entomol. 2022, 59, 273–282. [Google Scholar] [CrossRef] [PubMed]
- James, A.M.; Freier, J.E.; Keirans, J.E.; Durden, L.A.; Mertins, J.W.; Schlater, J.L. Distribution, Seasonality, and Hosts of the Rocky Mountain Wood Tick in the United States. J. Med. Entomol. 2006, 43, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Dergousoff, S.J.; Galloway, T.D.; Lindsay, L.R.; Curry, P.S.; Chilton, N.B. Range Expansion of Dermacentor variabilis and Dermacentor andersoni (Acari: Ixodidae) near Their Northern Distributional Limits. J. Med. Entomol. 2013, 50, 510–520. [Google Scholar] [CrossRef] [PubMed]
- James, A.M.; Burdett, C.; Mccool, M.J.; Fox, A.; Riggs, P. The Geographic Distribution and Ecological Preferences of the American Dog Tick, Dermacentor variabilis (Say), in the U.S.A. Med. Vet. Entomol. 2015, 29, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Lehane, A.; Parise, C.; Evans, C.; Beati, L.; Nicholson, W.L.; Eisen, R.J.; Hamer, S. Reported County-Level Distribution of the American Dog Tick (Acari: Ixodidae) in the Contiguous United States. J. Med. Entomol. 2020, 57, 131–155. [Google Scholar] [CrossRef] [PubMed]
- Lado, P.; Glon, M.G.; Klompen, H. Integrative Taxonomy of Dermacentor variabilis (Ixodida: Ixodidae) with Description of a New Species, Dermacentor similis n. Sp. J. Med. Entomol. 2021, 58, 2216–2227. [Google Scholar] [CrossRef] [PubMed]
- Molaei, G.; Little, E.A.H.; Williams, S.C.; Stafford, K.C. First Record of Established Populations of the Invasive Pathogen Vector and Ectoparasite Haemaphysalis longicornis (Acari: Ixodidae) in Connecticut, United States. J. Med. Entomol. 2021, 58, 2508–2513. [Google Scholar] [CrossRef]
- Xu, G.; Mather, T.N.; Hollingsworth, C.S.; Rich, S.M. Passive Surveillance of Ixodes scapularis (Say), Their Biting Activity, and Associated Pathogens in Massachusetts. Vector-Borne Zoonotic Dis. 2016, 16, 520–527. [Google Scholar] [CrossRef]
- Xu, G.; Pearson, P.; Dykstra, E.; Andrews, E.S.; Rich, S.M. Human-Biting Ixodes Ticks and Pathogen Prevalence from California, Oregon, and Washington. Vector-Borne Zoonotic Dis. 2019, 19, 106–114. [Google Scholar] [CrossRef]
- Nieto, N.C.; Tanner Porter, W.; Wachara, J.C.; Lowrey, T.J.; Martin, L.; Motyka, P.J.; Salkeld, D.J. Using Citizen Science to Describe the Prevalence and Distribution of Tick Bite and Exposure to Tick-Borne Diseases in the United States. PLoS ONE 2018, 13, e0199644. [Google Scholar] [CrossRef]
- Fryxell, R.T.T.; Vann, D.N.; Butler, R.A.; Paulsen, D.J.; Chandler, J.G.; Willis, M.P.; Wyrosdick, H.M.; Schaefer, J.J.; Gerhold, R.W.; Grove, D.M.; et al. Rapid Discovery and Detection of Haemaphysalis longicornis through the Use of Passive Surveillance and Collaboration: Building a State Tick-Surveillance Network. Int. J. Environ. Res. Public Health 2021, 18, 7980. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, A.R.; Palli, S.R. Mapping Distributions of the Lyme Disease Vector, Ixodes scapularis, and Spirochete, Borrelia burgdorferi, in Kentucky Using Passive and Active Surveillance. Ticks Tick-Borne Dis. 2021, 13, 101885. [Google Scholar] [CrossRef] [PubMed]
- Lyons, L.A.; Brand, M.E.; Gronemeyer, P.; Mateus-Pinilla, N.; Ruiz, M.O.; Stone, C.M.; Tuten, H.C.; Smith, R.L. Comparing Contributions of Passive and Active Tick Collection Methods to Determine Establishment of Ticks of Public Health Concern within Illinois. J. Med. Entomol. 2021, 58, 1849–1864. [Google Scholar] [CrossRef] [PubMed]
- Morshed, M.G.; Lee, M.-K.; Boyd, E.; Mak, S.; Fraser, E.; Nguyen, J.; Dykstra, E.A.; Otterstatter, M.; Henry, B.; Galanis, E. Passive Tick Surveillance and Detection of Borrelia Species in Ticks from British Columbia, Canada: 2002–2018. Vector-Borne Zoonotic Dis. 2021, 21, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Rounsville, T.F.; Dill, G.M.; Bryant, A.M.; Desjardins, C.C.; Dill, J.F. Statewide Passive Surveillance of Ixodes scapularis and Associated Pathogens in Maine. Vector-Borne Zoonotic Dis. 2021, 21, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Chilton, N.B.; Curry, P.S.; Lindsay, L.R.; Rochon, K.; Lysyk, T.J.; Dergousoff, S.J.; Foley, J. Passive and Active Surveillance for Ixodes scapularis (Acari: Ixodidae) in Saskatchewan, Canada. J. Med. Entomol. 2020, 57, 156–163. [Google Scholar] [CrossRef]
- Kopsco, H.L.; Duhaime, R.J.; Mather, T.N. Crowdsourced Tick Image-Informed Updates to U.S. County Records of Three Medically Important Tick Species. J. Med. Entomol. 2021, 58, 2412–2424. [Google Scholar] [CrossRef]
- Little, E.A.H.; Anderson, J.F.; Stafford, K.C.; Eisen, L.; Eisen, R.J.; Molaei, G. Predicting Spatiotemporal Patterns of Lyme Disease Incidence from Passively Collected Surveillance Data for Borrelia burgdorferi Sensu Lato-Infected Ixodes scapularis Ticks. Ticks Tick-Borne Dis. 2019, 10, 970–980. [Google Scholar] [CrossRef]
- Pak, D.; Jacobs, S.B.; Sakamoto, J.M. A 117-Year Retrospective Analysis of Pennsylvania Tick Community Dynamics. Parasit. Vectors 2019, 12, 189. [Google Scholar] [CrossRef]
- Jordan, R.A.; Egizi, A. The Growing Importance of Lone Star Ticks in a Lyme Disease Endemic County: Passive Tick Surveillance in Monmouth County, NJ, 2006-2016. PLoS ONE 2019, 14, e0211778. [Google Scholar] [CrossRef]
- Oliver, J.D.; Bennett, S.W.; Beati, L.; Bartholomay, L.C. Range Expansion and Increasing Borrelia burgdorferi Infection of the Tick Ixodes scapularis (Acari: Ixodidae) in Iowa, 1990–2013. J. Med. Entomol. 2017, 54, 1727–1734. [Google Scholar] [CrossRef] [PubMed]
- Elias, S.P.; Maasch, K.A.; Anderson, N.T.; Rand, P.W.; Lacombe, E.H.; Robich, R.M.; Lubelczyk, C.B.; Smith, R.P. Decoupling of Blacklegged Tick Abundance and Lyme Disease Incidence in Southern Maine, USA. J. Med. Entomol. 2020, 57, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Foley-Eby, A.H.; Savidge, C.; Lloyd, V.K. Ixodes scapularis Ticks and Borrelia burgdorferi on Prince Edward Island: Passive Tick Surveillance and Canine Seroprevalence. Can. Vet. J. 2020, 61, 1107–1110. [Google Scholar] [PubMed]
- Porter, W.T.; Wachara, J.; Barrand, Z.A.; Nieto, N.C.; Salkeld, D.J. Citizen Science Provides an Efficient Method for Broad-Scale Tick-Borne Pathogen Surveillance of Ixodes pacificus and Ixodes scapularis across the United States. mSphere 2021, 6, 1–16. [Google Scholar] [CrossRef]
- Hahn, M.B.; Disler, G.; Durden, L.A.; Coburn, S.; Witmer, F.; George, W.; Beckmen, K.; Gerlach, R. Establishing a Baseline for Tick Surveillance in Alaska: Tick Collection Records from 1909–2019. Ticks Tick-Borne Dis. 2020, 11, 101495. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.; Kirby, A.M.; Harris, K.D.; Filiaggi, C.L.; Foley-Eby, A.; Mann, M.; Lieske, D.; Lloyd, V.K. Monitoring Risk: Tick and Borrelia burgdorferi Public Participatory Surveillance in the Canadian Maritimes, 2012–2020. Pathogens 2021, 10, 1284. [Google Scholar] [CrossRef]
- Nelder, M.P.; Russell, C.; Lindsay, L.R.; Dhar, B.; Patel, S.N.; Johnson, S.; Moore, S.; Kristjanson, E.; Li, Y.; Ralevski, F. Population-Based Passive Tick Surveillance and Detection of Expanding Foci of Blacklegged Ticks Ixodes scapularis and the Lyme Disease Agent Borrelia burgdorferi in Ontario, Canada. PLoS ONE 2014, 9, e105358. [Google Scholar] [CrossRef]
- Nelder, M.P.; Russell, C.B.; Clow, K.M.; Johnson, S.; Weese, J.S.; Cronin, K.; Ralevski, F.; Jardine, C.M.; Patel, S.N. Occurrence and Distribution of Ambylomma americanum as Determined by Passive Surveillance in Ontario, Canada (1999–2016). Ticks Tick-Borne Dis. 2019, 10, 146–155. [Google Scholar] [CrossRef]
- Nelder, M.P.; Russell, C.B.; Dibernardo, A.; Clow, K.M.; Johnson, S.; Cronin, K.; Patel, S.N.; Lindsay, L.R. Monitoring the Patterns of Submission and Presence of Tick-Borne Pathogens in Ixodes scapularis Collected from Humans and Companion Animals in Ontario, Canada (2011–2017). Parasites Vectors 2021, 14, 260. [Google Scholar] [CrossRef]
- Gasmi, S.; Bouchard, C.; Ogden, N.H.; Adam-Poupart, A.; Pelcat, Y.; Rees, E.E.; Milord, F.; Leighton, P.A.; Lindsay, R.L.; Koffi, J.K.; et al. Evidence for Increasing Densities and Geographic Ranges of Tick Species of Public Health Significance Other than Ixodes scapularis in Québec, Canada. PLoS ONE 2018, 13, e0201924. [Google Scholar] [CrossRef]
- Johnson, J.L.; Ginsberg, H.S.; Zhioua, E.; Whitworth, U.G.; Markowski, D.; Hyland, K.E.; Hu, R. Passive Tick Surveillance, Dog Seropositivity, and Incidence of Human Lyme Disease. Vector-Borne Zoonotic Dis. 2004, 4, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Rand, P.W.; Lacombe, E.H.; Dearborn, R.; Cahill, B.; Elias, S.; Lubelczyk, C.B.; Beckett, G.A.; Smith, R.P.J. Passive Surveillance in Maine, an Area Emergent for Tick-Borne Diseases. J. Med. Entomol. 2007, 44, 1118–1129. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gasmi, S.; Ogden, N.H.; Ripoche, M.; Leighton, P.A.; Lindsay, R.L.; Nelder, M.P.; Rees, E.; Bouchard, C.; Vrbova, L.; Rusk, R.; et al. Detection of Municipalities At-Risk of Lyme Disease Using Passive Surveillance of Ixodes scapularis as an Early Signal: A Province-Specific Indicator in Canada. PLoS ONE 2019, 14, e0212637. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.; Porter, W.T.; Salkeld, D.J.; Prusinski, M.A.; Jensen, S.T.; Brisson, D. Estimating Disease Vector Population Size from Citizen Science Data. J. R. Soc. Interface 2021, 18, 18. [Google Scholar] [CrossRef]
- Hamer, S.A.; Curtis-Robles, R.; Hamer, G.L. Contributions of Citizen Scientists to Arthropod Vector Data in the Age of Digital Epidemiology. Curr. Opin. Insect Sci. 2018, 28, 98–104. [Google Scholar] [CrossRef]
- Eisen, L.; Eisen, R.J. Benefits and Drawbacks of Citizen Science to Complement Traditional Data Gathering Approaches for Medically Important Hard Ticks (Acari: Ixodidae) in the United States. J. Med. Entomol. 2020, 58, 1–9. [Google Scholar] [CrossRef]
- Kopsco, H.L.; Xu, G.; Luo, C.-Y.; Rich, S.M.; Mather, T.N. Crowdsourced Photographs as an Effective Method for Large-Scale Passive Tick Surveillance. J. Med. Entomol. 2020, 57, 1955–1963. [Google Scholar] [CrossRef]
- Cull, B. Potential for Online Crowdsourced Biological Recording Data to Complement Surveillance for Arthropod Vectors. PLoS ONE 2021, 16, e0250382. [Google Scholar] [CrossRef]
- Saul-Gershenz, L.; Grodsky, S.M.; Hernandez, R.R. Ecology of the Western Queen Butterfly Danaus gilippus thersippus (Lepidoptera: Nymphalidae) in the Mojave and Sonoran Deserts. Insects 2020, 11, 315. [Google Scholar] [CrossRef]
- Putman, B.J.; Williams, R.; Li, E.; Pauly, G.B. The Power of Citizen Science to Quantify Ecological Risks in Cities. Sci. Rep. 2021, 11, 3069. [Google Scholar] [CrossRef]
- Snyman, P.L.; Fitchett, J.M. Phenological Advance in the South African Namaqualand Daisy First and Peak Bloom: 1935–2018. Int. J. Biometeorol. 2022, 66, 699–717. [Google Scholar] [CrossRef] [PubMed]
- Hiller, T.; Haelewaters, D. A Case of Silent Invasion: Citizen Science Confirms the Presence of Harmonia axyridis (Coleoptera, Coccinellidae) in Central America. PLoS ONE 2019, 14, e0220082. [Google Scholar] [CrossRef] [PubMed]
- Happel, A. A Volunteer-Populated Online Database Provides Evidence for a Geographic Pattern in Symptoms of Black Spot Infections. Int. J. Parasitol. Parasites Wildl. 2019, 10, 156–163. [Google Scholar] [CrossRef]
- Leong, M.; Trautwein, M. A Citizen Science Approach to Evaluating US Cities for Biotic Homogenization. PeerJ 2019, 7, e6879. [Google Scholar] [CrossRef]
- Mueller, M.A.; Drake, D.; Allen, M.L. Using Citizen Science to Inform Urban Canid Management. Landsc. Urban Plan. 2019, 189, 362–371. [Google Scholar] [CrossRef]
- Barve, V.V.; Brenskelle, L.; Li, D.; Stucky, B.J.; Barve, N.V.; Hantak, M.M.; McLean, B.S.; Paluh, D.J.; Oswald, J.A.; Belitz, M.W.; et al. Methods for Broad-Scale Plant Phenology Assessments Using Citizen Scientists’ Photographs. Appl. Plant Sci. 2020, 8, e11315. [Google Scholar] [CrossRef]
- Hochmair, H.H.; Scheffrahn, R.H.; Basille, M.; Boone, M. Evaluating the Data Quality of iNaturalist Termite Records. PLoS ONE 2020, 15, e0226534. [Google Scholar] [CrossRef]
- Nowak, K.; Berger, J.; Panikowski, A.; Reid, D.G.; Jacob, A.L.; Newman, G.; Young, N.E.; Beckmann, J.P.; Richards, S.A. Using Community Photography to Investigate Phenology: A Case Study of Coat Molt in the Mountain Goat (Oreamnos americanus) with Missing Data. Ecol. Evol. 2020, 10, 13488–13499. [Google Scholar] [CrossRef]
- Doherty, J.F.; Filion, A.; Bennett, J.; Raj Bhattarai, U.; Chai, X.; De Angeli Dutra, D.; Donlon, E.; Jorge, F.; Milotic, M.; Park, E.; et al. The People vs Science: Can Passively Crowdsourced Internet Data Shed Light on Host-Parasite Interactions? Parasitology 2021, 148, 1313–1319. [Google Scholar] [CrossRef]
- Gregson, J.D. The Ixodoidea of Canada; Science Service, Entomology Division, Canada Department of Agriculture: Ottawa, ON, Canada, 1956. [Google Scholar]
- CDC Tick Surveillance Data Sets|Ticks|CDC. Available online: https://www.cdc.gov/ticks/surveillance/TickSurveillanceData.html (accessed on 1 April 2022).
- Maestas, L.P.; Mays, S.E.; Britten, H.B.; Auckland, L.D.; Hamer, S.A. Surveillance for Ixodes scapularis (Acari Ixodidae) and Borrelia burgdorferi in Eastern South Dakota State Parks and Nature Areas. J. Med. Entomol. 2018, 55, 1549–1554. [Google Scholar] [CrossRef]
- Black, H.; Potts, R.; Fiechtner, J.; Pietri, J.E.; Britten, H.B. Establishment of Amblyomma americanum Populations and New Records of Borrelia burgdorferi-Infected Ixodes scapularis in South Dakota. J. Vector Ecol. 2021, 46, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Justen, L.; Carlsmith, D.; Paskewitz, S.M.; Bartholomay, L.C.; Bron, G.M. Identification of Public Submitted Tick Images: A Neural Network Approach. PLoS ONE 2021, 16, e0260622. [Google Scholar] [CrossRef] [PubMed]
- Laaksonen, M.; Sajanti, E.; Sormunen, J.J.; Penttinen, R.; Hänninen, J.; Ruohomäki, K.; Sääksjärvi, I.; Vesterinen, E.J.; Vuorinen, I.; Hytönen, J.; et al. Crowdsourcing-Based Nationwide Tick Collection Reveals the Distribution of Ixodes ricinus and I. persulcatus and Associated Pathogens in Finland. Emerg. Microbes Infect. 2017, 6, e31. [Google Scholar] [CrossRef] [PubMed]
- Teel, P.D.; Ketchum, H.R.; Mock, D.E.; Wright, R.E.; Strey, O.F. The Gulf Coast Tick: A Review of the Life History, Ecology, Distribution, and Emergence as an Arthropod of Medical and Veterinary Importance. J. Med. Entomol. 2010, 47, 707–722. [Google Scholar] [CrossRef] [PubMed]
- Nadolny, R.M.; Gaff, H.D. Natural History of Amblyomma maculatum in Virginia. Ticks Tick-Borne Dis. 2018, 9, 188–195. [Google Scholar] [CrossRef] [PubMed]
| Region | State/Province | Ixodidae * | Argasidae | Total | |||||
|---|---|---|---|---|---|---|---|---|---|
| Amb | Der | Hae | Ixo | Rhi | Unidentified | ||||
| US Region 1 | Connecticut | 4 | 133 | 0 | 111 | 0 | 1 | 0 | 249 |
| Maine | 0 | 167 | 0 | 48 | 0 | 7 | 0 | 222 | |
| Massachusetts | 23 | 458 | 0 | 390 | 0 | 16 | 0 | 887 | |
| New Hampshire | 0 | 240 | 0 | 83 | 0 | 1 | 0 | 324 | |
| Rhode Island | 1 | 23 | 0 | 23 | 0 | 1 | 0 | 48 | |
| Vermont | 0 | 193 | 2 | 359 | 0 | 9 | 0 | 563 | |
| US Region 2 | New Jersey | 255 | 361 | 9 | 142 | 0 | 27 | 0 | 794 |
| New York | 123 | 348 | 22 | 589 | 0 | 29 | 0 | 1111 | |
| US Region 3 | Delaware | 63 | 33 | 0 | 5 | 0 | 4 | 0 | 105 |
| District of Columbia | 21 | 7 | 1 | 7 | 0 | 4 | 0 | 40 | |
| Maryland | 250 | 183 | 6 | 110 | 0 | 26 | 1 | 576 | |
| Pennsylvania | 33 | 297 | 9 | 424 | 0 | 15 | 0 | 778 | |
| Virginia | 501 | 290 | 5 | 118 | 0 | 29 | 0 | 943 | |
| West Virginia | 6 | 51 | 0 | 42 | 0 | 2 | 0 | 101 | |
| US Region 4 | Alabama | 176 | 66 | 0 | 29 | 0 | 14 | 0 | 285 |
| Florida | 329 | 40 | 0 | 60 | 5 | 12 | 0 | 446 | |
| Georgia | 179 | 64 | 0 | 28 | 0 | 7 | 0 | 278 | |
| Kentucky | 109 | 90 | 0 | 27 | 0 | 4 | 1 | 231 | |
| Mississippi | 79 | 27 | 0 | 16 | 0 | 5 | 0 | 127 | |
| North Carolina | 217 | 181 | 1 | 33 | 0 | 22 | 0 | 454 | |
| South Carolina | 89 | 38 | 0 | 18 | 0 | 6 | 0 | 151 | |
| Tennessee | 171 | 153 | 0 | 42 | 0 | 8 | 0 | 374 | |
| US Region 5 | Illinois | 98 | 380 | 0 | 81 | 0 | 13 | 0 | 572 |
| Indiana | 69 | 114 | 0 | 29 | 0 | 4 | 0 | 216 | |
| Michigan | 6 | 277 | 0 | 95 | 0 | 9 | 0 | 387 | |
| Minnesota | 2 | 307 | 0 | 100 | 0 | 3 | 2 | 414 | |
| Ohio | 58 | 475 | 2 | 177 | 0 | 20 | 0 | 732 | |
| Wisconsin | 5 | 171 | 0 | 100 | 0 | 2 | 0 | 278 | |
| US Region 6 | Arkansas | 125 | 25 | 0 | 10 | 0 | 9 | 0 | 169 |
| Louisiana | 61 | 16 | 0 | 14 | 0 | 1 | 0 | 92 | |
| New Mexico | 0 | 6 | 0 | 0 | 2 | 3 | 5 | 16 | |
| Oklahoma | 230 | 76 | 0 | 24 | 0 | 5 | 1 | 336 | |
| Texas | 571 | 258 | 0 | 107 | 46 | 27 | 5 | 1014 | |
| US Region 7 | Iowa | 16 | 70 | 0 | 18 | 0 | 3 | 0 | 107 |
| Kansas | 72 | 50 | 0 | 3 | 0 | 5 | 3 | 133 | |
| Missouri | 475 | 110 | 0 | 20 | 0 | 20 | 3 | 628 | |
| Nebraska | 43 | 197 | 0 | 1 | 0 | 4 | 0 | 245 | |
| US Region 8 | Colorado | 1 | 63 | 0 | 2 | 0 | 2 | 0 | 68 |
| Montana | 0 | 45 | 0 | 0 | 0 | 0 | 0 | 45 | |
| North Dakota | 1 | 53 | 0 | 3 | 0 | 1 | 0 | 58 | |
| South Dakota | 0 | 52 | 0 | 1 | 0 | 0 | 0 | 53 | |
| Utah | 1 | 44 | 0 | 1 | 1 | 5 | 0 | 52 | |
| Wyoming | 0 | 24 | 0 | 0 | 0 | 2 | 0 | 26 | |
| Atlantic | New Brunswick | 0 | 12 | 1 | 31 | 0 | 1 | 0 | 45 |
| Canada | Prince Edward Island | 0 | 0 | 0 | 5 | 0 | 0 | 0 | 5 |
| Nova Scotia | 0 | 289 | 1 | 178 | 0 | 19 | 0 | 487 | |
| Newfoundland and Labrador | 0 | 0 | 0 | 4 | 0 | 2 | 0 | 6 | |
| Central | Ontario | 21 | 829 | 0 | 739 | 0 | 25 | 0 | 1614 |
| Canada | Quebec | 0 | 20 | 1 | 60 | 0 | 0 | 0 | 81 |
| Western | Manitoba | 0 | 95 | 0 | 16 | 0 | 1 | 0 | 112 |
| Canada | Saskatchewan | 0 | 93 | 0 | 0 | 0 | 1 | 0 | 94 |
| TOTALS | 4484 | 7594 | 60 | 4523 | 54 | 436 | 21 | 17,172 | |
| Surveillance Data Source | Time Period | No. Counties with I. scapularis | No. Unique County Records | No. Counties with A. americanum | No. Unique County Records |
|---|---|---|---|---|---|
| Eisen et al., 2016 [6]; CDC, 2021 [72] | 1996–2020 | 1570 | 605 | - | - |
| Springer et al. 2014 [8] | 1898–2012 | - | - | 1292 | 454 |
| Northern Arizona University [31,45] | 2016–2019 (Is) * 2016–2017 (Aa) | 688 | 33 | 438 | 34 |
| TickSpotters [38] | 2014–2019 | 896 | 73 | 1024 | 181 |
| iNaturalist [this study] | 1997–2021 | 679 | 49 | 727 | 61 |
| CDC | TickSpotters | Northern Arizona University | iNaturalist | |
|---|---|---|---|---|
| iNaturalist | 587/1570 (37%) | 504/896 (56%) | 428/688 (62%) | - |
| Northern Arizona University | 616/1570 (39%) | 536/896 (60%) | - | 428/679 (63%) |
| TickSpotters | 762/1570 (49%) | - | 536/688 (78%) | 504/679 (74%) |
| CDC | - | 762/896 (85%) | 616/688 (90%) | 587/679 (86%) |
| CDC | TickSpotters | Northern Arizona University | iNaturalist | |
|---|---|---|---|---|
| iNaturalist | 537/1292 (42%) | 540/1024 (53%) | 285/438 (65%) | - |
| Northern Arizona University | 332/1292 (26%) | 352/1024 (34%) | - | 285/727 (39%) |
| TickSpotters | 691/1292 (53%) | - | 352/438 (80%) | 540/727 (74%) |
| CDC | - | 691/1024 (67%) | 332/438 (76%) | 537/727 (74%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cull, B. Monitoring Trends in Distribution and Seasonality of Medically Important Ticks in North America Using Online Crowdsourced Records from iNaturalist. Insects 2022, 13, 404. https://doi.org/10.3390/insects13050404
Cull B. Monitoring Trends in Distribution and Seasonality of Medically Important Ticks in North America Using Online Crowdsourced Records from iNaturalist. Insects. 2022; 13(5):404. https://doi.org/10.3390/insects13050404
Chicago/Turabian StyleCull, Benjamin. 2022. "Monitoring Trends in Distribution and Seasonality of Medically Important Ticks in North America Using Online Crowdsourced Records from iNaturalist" Insects 13, no. 5: 404. https://doi.org/10.3390/insects13050404
APA StyleCull, B. (2022). Monitoring Trends in Distribution and Seasonality of Medically Important Ticks in North America Using Online Crowdsourced Records from iNaturalist. Insects, 13(5), 404. https://doi.org/10.3390/insects13050404






