Effect of the Amount of Ephestia kuehniella Eggs for Rearing on Development, Survival, and Reproduction of Orius laevigatus
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sanchez, J.A.; Alcazar, A.; Lacasa, A.; Llamas, A.; Bielza, P. Integrated pest management strategies in sweet pepper plastic houses in the Southeast of Spain. IOBC/WPRS Bull. 2000, 23, 21–30. [Google Scholar]
- Calvo, F.J.; Bolckmans, K.; Belda, J.E. Biological control based IPM in sweet pepper greenhouses using Amblyseius swirskii (Acari, Phytoseiidae). Biocontrol. Sci. Technol. 2012, 22, 1398–1416. [Google Scholar] [CrossRef]
- Balanza, V.; Mendoza, J.E.; Bielza, P. Variation in susceptibility and selection for resistance to imidacloprid and thiamethoxam in Mediterranean populations of Orius laevigatus. Entomol. Exp. Appl. 2019, 167, 626–635. [Google Scholar] [CrossRef] [Green Version]
- Van Lenteren, J.C.; Alomar, O.; Ravensberg, W.J.; Urbaneja, A. Integrated Pest and Disease Management in Greenhouse Crops. In Integrated Pest and Disease Management in Greenhouse Crops, Plant Pathology in the 21st Century 9; Gullino, M.L., Albajes, R., Nicot, P.C., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 409–439. [Google Scholar]
- Balanza, V.; Mendoza, J.E.; Cifuentes, D.; Bielza, P. Selection for resistance to pyrethroids in the predator Orius laevigatus. Pest Manag. Sci. 2021, 77, 2539–2546. [Google Scholar] [CrossRef] [PubMed]
- Balanza, V.; Mendoza, J.E.; Cifuentes, D.; Bielza, P. Genetic improvement of spinosad resistance in the biocontrol agent Orius laevigatus. BioControl 2021, 66, 673–685. [Google Scholar] [CrossRef]
- Vandekerkhove, B.; De Clercq, P. Pollen as an alternative or supplementary food for the mirid predator Macrolophus pygmaeus. Biol. Control. 2010, 53, 238–242. [Google Scholar] [CrossRef] [Green Version]
- Richards, P.C.; Schmidt, J.M. The effects of selected dietary supplements on survival and reproduction of Orius insidiosus (Say) (Hemiptera, Anthocoridae). Can. Entomol. 1996, 128, 171–176. [Google Scholar] [CrossRef]
- Cocuzza, G.E.; De Clercq, P.; van de Veire, M.; de Cock, A.; Degheele, D.; Vacante, V. Reproduction of Orius laevigatus and Orius albidipennis on pollen and Ephestia kuehniella eggs. Entomol. Exp. Appl. 1997, 82, 101–104. [Google Scholar] [CrossRef]
- Vacante, V.; Cocuzza, G.E.; De Clercq, P.; van de Veire, M.; Tirry, L. Development and survival of Orius albidipennis and O. laevigatus (Het., Anthocoridae) on various diets. Entomophaga 1997, 42, 493. [Google Scholar]
- Arijs, Y.; De Clercq, P. Rearing Orius laevigatus on cysts of the brine shrimp Artemia franciscana. Biol. Control. 2001, 21, 79–83. [Google Scholar] [CrossRef]
- Arijs, Y.; De Clercq, P. Liver-based artificial diets for the production of Orius laevigatus. BioControl 2004, 49, 505–516. [Google Scholar] [CrossRef]
- De Clercq, P.; Arijs, Y.; Van Meir, T.; Van Stappen, G.; Sorgeloos, P.; Dewettinck, K.; Rey, M.; Grenier, S.; Febvay, G. Nutritional value of brine shrimp cysts as a factitious food for Orius laevigatus (Heteroptera, Anthocoridae). Biocontrol Sci. Technol. 2005, 15, 467–479. [Google Scholar] [CrossRef]
- Bonte, M.; De Clercq, P. Developmental and reproductive fitness of Orius laevigatus (Hemiptera, Anthocoridae) reared on factitious and artificial diets. J. Econ. Entomol. 2008, 101, 1127–1133. [Google Scholar] [CrossRef]
- Bonte, M.; De Clercq, P. Influence of diet on the predation rate of Orius laevigatus on Frankliniella occidentalis. BioControl 2010, 55, 625–629. [Google Scholar] [CrossRef]
- Bonte, M.; De Clercq, P. Influence of predator density, diet and living substrate on developmental fitness of Orius laevigatus. J Appl. Entomol. 2011, 135, 343–350. [Google Scholar] [CrossRef]
- Bonte, J.; Vangansbeke, D.; Maes, S.; Bonte, M.; Conlong, D.; De Clercq, P. Moisture source and diet affect development and reproduction of Orius thripoborus and Orius naivashae, two predatory anthocorids from southern Africa. J. Insect Sci. 2012, 12, 1. [Google Scholar] [CrossRef]
- Calixto, A.; Bueno, V.; Montes, F.; Silva, A.; van Lenteren, J. Effect of different diets on reproduction, longevity and predation capacity of Orius insidiosus (Say) (Hemiptera, Anthocoridae). Biocontrol Sci. Techn. 2013, 23, 1245–1255. [Google Scholar] [CrossRef]
- Bernardo, A.M.G.; de Oliveira, C.M.; Oliveira, R.A.; Vacacela, H.E.; Venzon, M.; Pallini, A.; Janssen, A. Performance of Orius insidiosus on alternative foods. J. Appl. Entomol. 2017, 141, 702–707. [Google Scholar] [CrossRef]
- Montoro, M.; De Clercq, P.; Overgaard, J.; Sigsgaard, L. Fitness consequences of artificial diets with different macronutrient composition for the predatory bug Orius majusculus. Entomol. Exp. Appl. 2020, 168, 492–501. [Google Scholar] [CrossRef]
- Yano, E.; Watanabe, K.; Yara, K. Life history parameters of Orius sauteri (Poppius) (Heteroptera, Anthocoridae) reared on Ephestia kuehniella eggs and the minimum amount of the diet for rearing individuals. J. Appl. Entomol. 2002, 126, 389–394. [Google Scholar] [CrossRef]
- Van den Meiracker, R.A.F. Biocontrol of Western Flower Thrips by Heteropteran Bugs. Ph.D. Thesis, University of Amsterdam, Amsterdam, The Netherlands, June 1999. [Google Scholar]
- Mendoza, J.E.; Balanza, V.; Cifuentes, D.; Bielza, P. Genetic improvement of Orius laevigatus for better fitness feeding on pollen. J. Pest Sci. 2021, 94, 729–742. [Google Scholar] [CrossRef]
- Blümel, S. Effect of selected mass-rearing parameters on Orius majusculus (Reuter) and Orius laevigatus (Fieber). Bull. OILB SROP 1996, 19, 15–18. [Google Scholar]
- Tommasini, M.G.; van Lenteren, J.C.; Burgio, G. Biological traits and predation capacity of four Orius species on two prey species. Bull. Insectology 2004, 57, 79–93. [Google Scholar]
- Bielza, P.; Balanza, V.; Cifuentes, D.; Mendoza, J.E. Challenges facing arthropod biological control, Identifying traits for genetic improvement of predators in protected crops. Pest Manag. Sci. 2020, 76, 3517–3526. [Google Scholar] [CrossRef] [PubMed]
- Kingsolver, J.G.; Huey, R.B. Size, temperature, and fitness, three rules. Evo. Ecol. Res. 2008, 10, 251–268. [Google Scholar]
- Chown, S.L.; Gaston, K.J. Body size variation in insects, a macroecological perspective. Biol. Rev. 2010, 85, 139–169. [Google Scholar] [CrossRef]
- Kalinkat, G.; Jochum, M.; Brose, U.; Dell, A.I. Body size and the behavioral ecology of insects, linking individuals to ecological communities. Curr. Opin. Insect Sci. 2015, 9, 24–30. [Google Scholar] [CrossRef]
- Vangansbeke, D.; Duarte, M.V.; Gobin, B.; Tirry, L.; Wäckers, F.; De Clercq, P. Cold-born killers, exploiting temperature-size rule enhances predation capacity of a predatory mite. Pest Manag. Sci. 2019, 76, 1841–1846. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, J.E.; Balanza, V.; Cifuentes, D.; Bielza, P. Selection for larger body size in Orius laevigatus, Intraspecific variability and effects on reproductive parameters. Biol. Control 2020, 148, 104310. [Google Scholar] [CrossRef]
- Davis, R.B.; Javoiš, J.; Kaasik, A.; Õunap, E.; Tammaru, T. An ordination of life histories using morphological proxies, capital vs. income breeding in insects. Ecology 2016, 97, 2112–2124. [Google Scholar] [CrossRef]
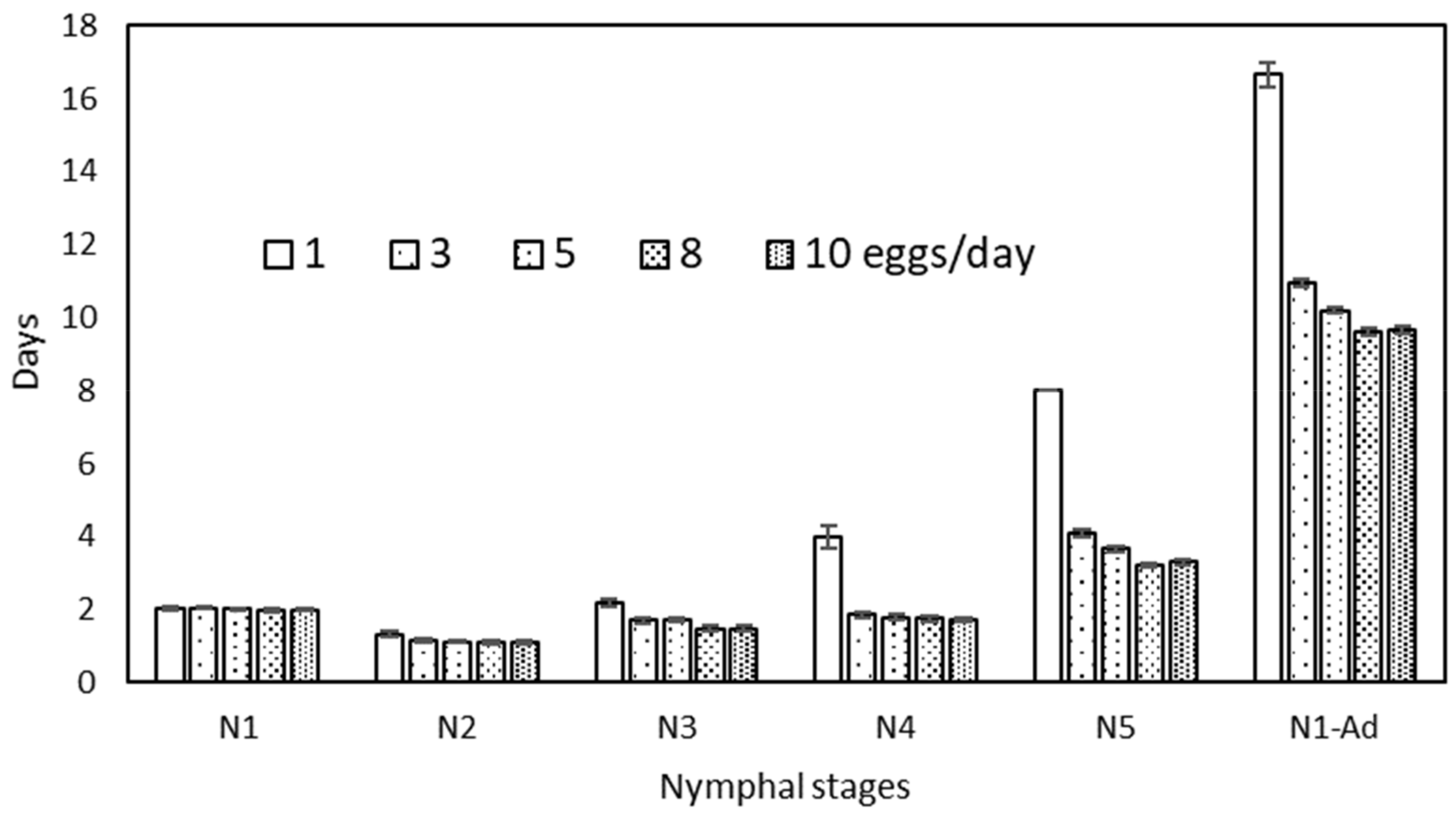
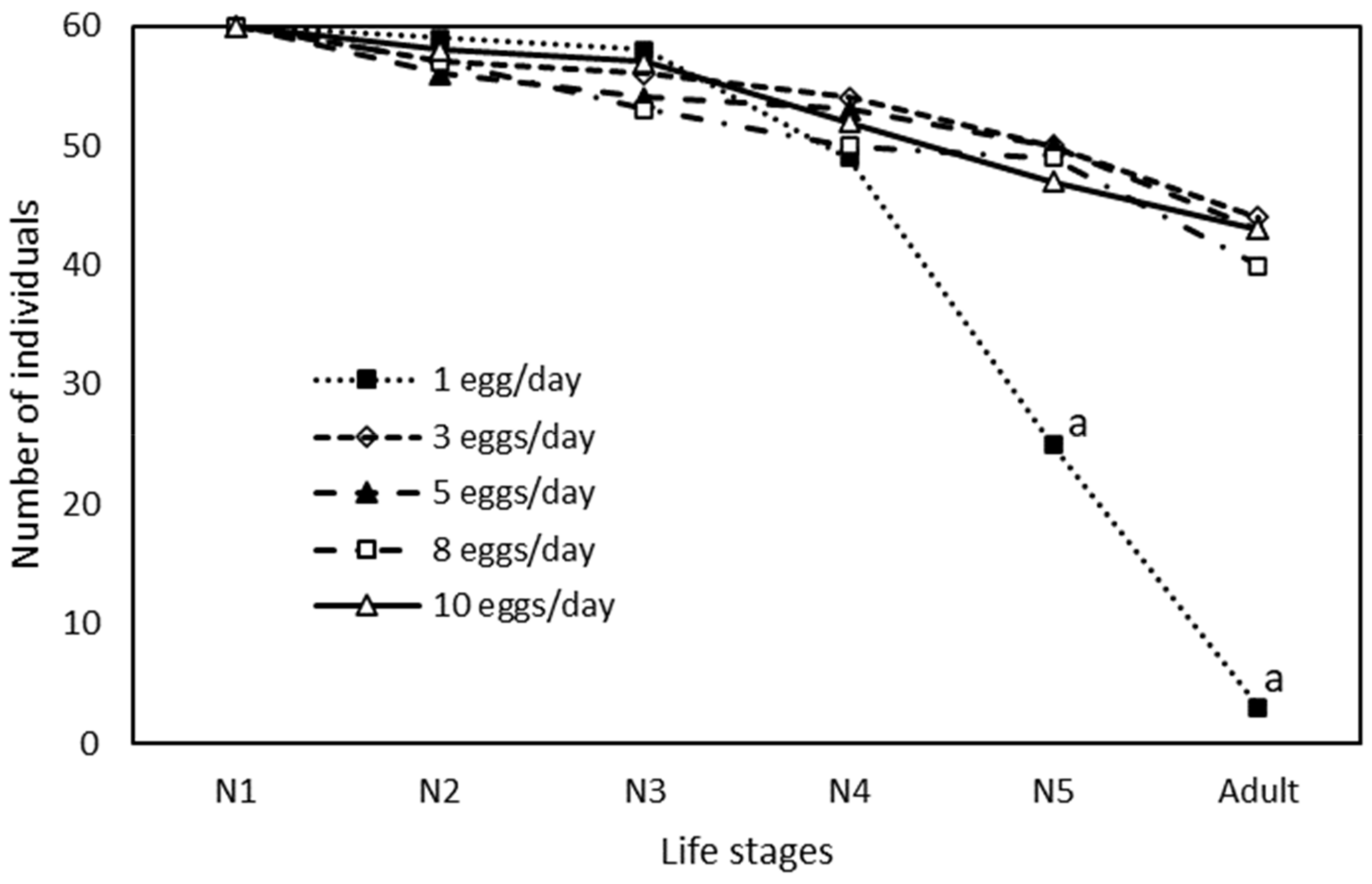
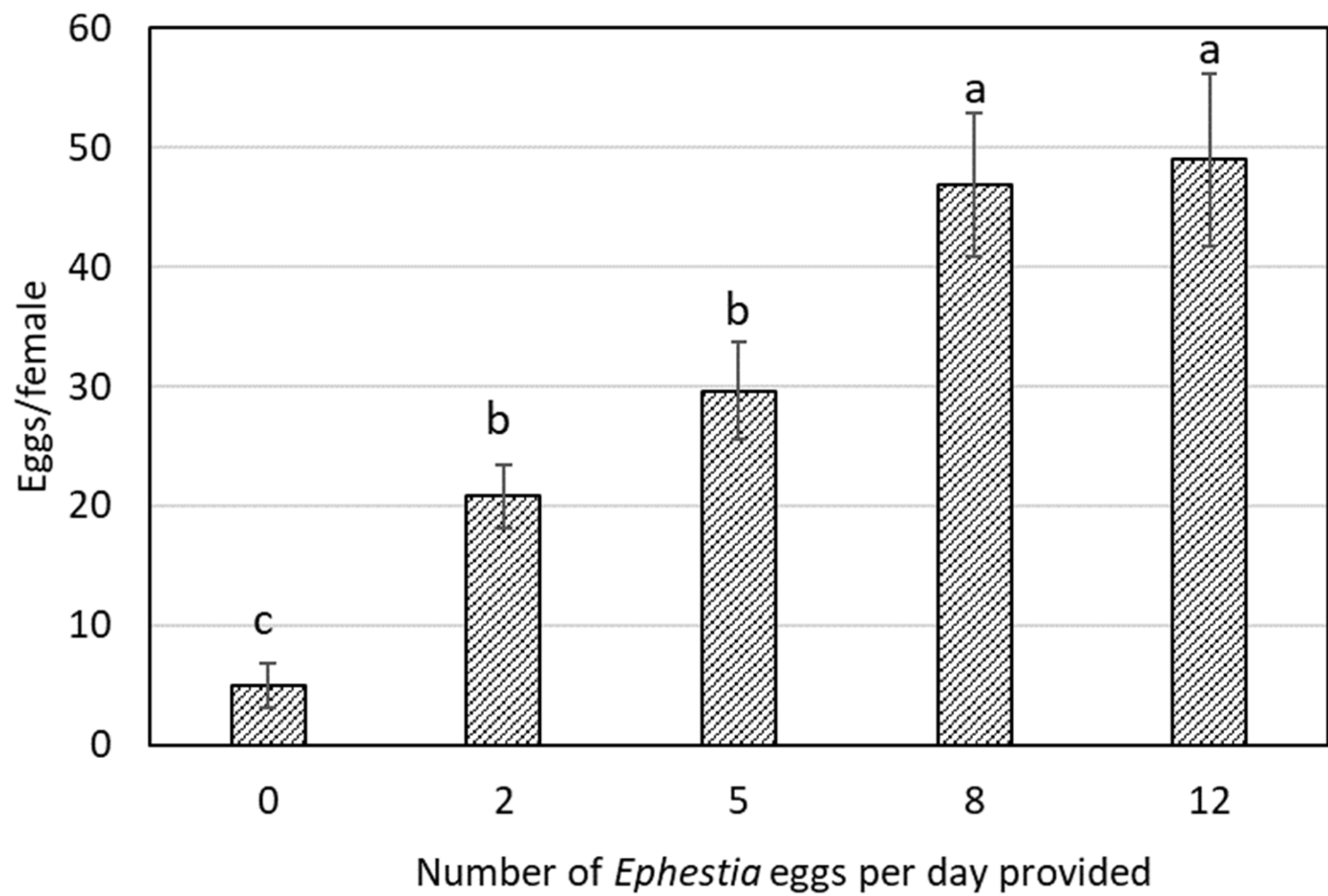
| Eggs/day | N1 | N2 | N3 | N4 | N5 | N1–N5 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2.05 ± 0.04 | a | 1.33 ± 0.07 | b | 2.18 ± 0.10 | c | 4.00 ± 0.31 | b | 8.00 ± 0.00 | d | 16.67 ± 0.33 | d |
| 3 | 2.05 ± 0.03 | a | 1.18 ± 0.05 | a | 1.72 ± 0.08 | b | 1.86 ± 0.06 | a | 4.09 ± 0.08 | c | 10.95 ± 0.11 | c |
| 5 | 2.02 ± 0.02 | a | 1.13 ± 0.05 | a | 1.72 ± 0.07 | b | 1.80 ± 0.10 | a | 3.67 ± 0.08 | b | 10.19 ± 0.08 | b |
| 8 | 1.98 ± 0.03 | a | 1.11 ± 0.05 | a | 1.48 ± 0.07 | a | 1.76 ± 0.07 | a | 3.23 ± 0.07 | a | 9.60 ± 0.10 | a |
| 10 | 2.00 ± 0.02 | a | 1.12 ± 0.04 | a | 1.48 ± 0.07 | a | 1.72 ± 0.07 | a | 3.30 ± 0.08 | a | 9.67 ± 0.10 | a |
| F4,282 = 0.99 ns | F4,273 = 2.79 * | F4,253 = 13.56 *** | F4,216 = 52.43 *** | F4,168 = 75.73 *** | F4,168 = 105.8 *** | |||||||
| Eggs/day | Female size | Male size | ||
|---|---|---|---|---|
| 1 | 0.585 ± 0.015 (2) | e | 0.550 (1) | d |
| 3 | 0.684 ± 0.005 (21) | d | 0.653 ± 0.005 (23) | c |
| 5 | 0.735 ± 0.005 (21) | c | 0.685 ± 0.004 (22) | b |
| 8 | 0.752 ± 0.006 (20) | b | 0.736 ± 0.005 (20) | a |
| 10 | 0.768 ± 0.005 (26) | a | 0.735 ± 0.004 (17) | a |
| F4,85 = 57.2 *** | F4,78 = 70.8 *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallego, F.J.; Rodríguez-Gómez, A.; Reche, M.d.C.; Balanza, V.; Bielza, P. Effect of the Amount of Ephestia kuehniella Eggs for Rearing on Development, Survival, and Reproduction of Orius laevigatus. Insects 2022, 13, 250. https://doi.org/10.3390/insects13030250
Gallego FJ, Rodríguez-Gómez A, Reche MdC, Balanza V, Bielza P. Effect of the Amount of Ephestia kuehniella Eggs for Rearing on Development, Survival, and Reproduction of Orius laevigatus. Insects. 2022; 13(3):250. https://doi.org/10.3390/insects13030250
Chicago/Turabian StyleGallego, Francisco Javier, Amador Rodríguez-Gómez, María del Carmen Reche, Virginia Balanza, and Pablo Bielza. 2022. "Effect of the Amount of Ephestia kuehniella Eggs for Rearing on Development, Survival, and Reproduction of Orius laevigatus" Insects 13, no. 3: 250. https://doi.org/10.3390/insects13030250
APA StyleGallego, F. J., Rodríguez-Gómez, A., Reche, M. d. C., Balanza, V., & Bielza, P. (2022). Effect of the Amount of Ephestia kuehniella Eggs for Rearing on Development, Survival, and Reproduction of Orius laevigatus. Insects, 13(3), 250. https://doi.org/10.3390/insects13030250







