Stress Resistance Traits under Different Thermal Conditions in Drosophila subobscura from Two Altitudes
Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Desiccation Resistance (DR)
2.2. Heath Knock-Down Resistance (HKDR)
2.3. Starvation Resistance (SR)
2.4. Chill Coma Recovery Time (CCRT)
2.5. Statistics
3. Results
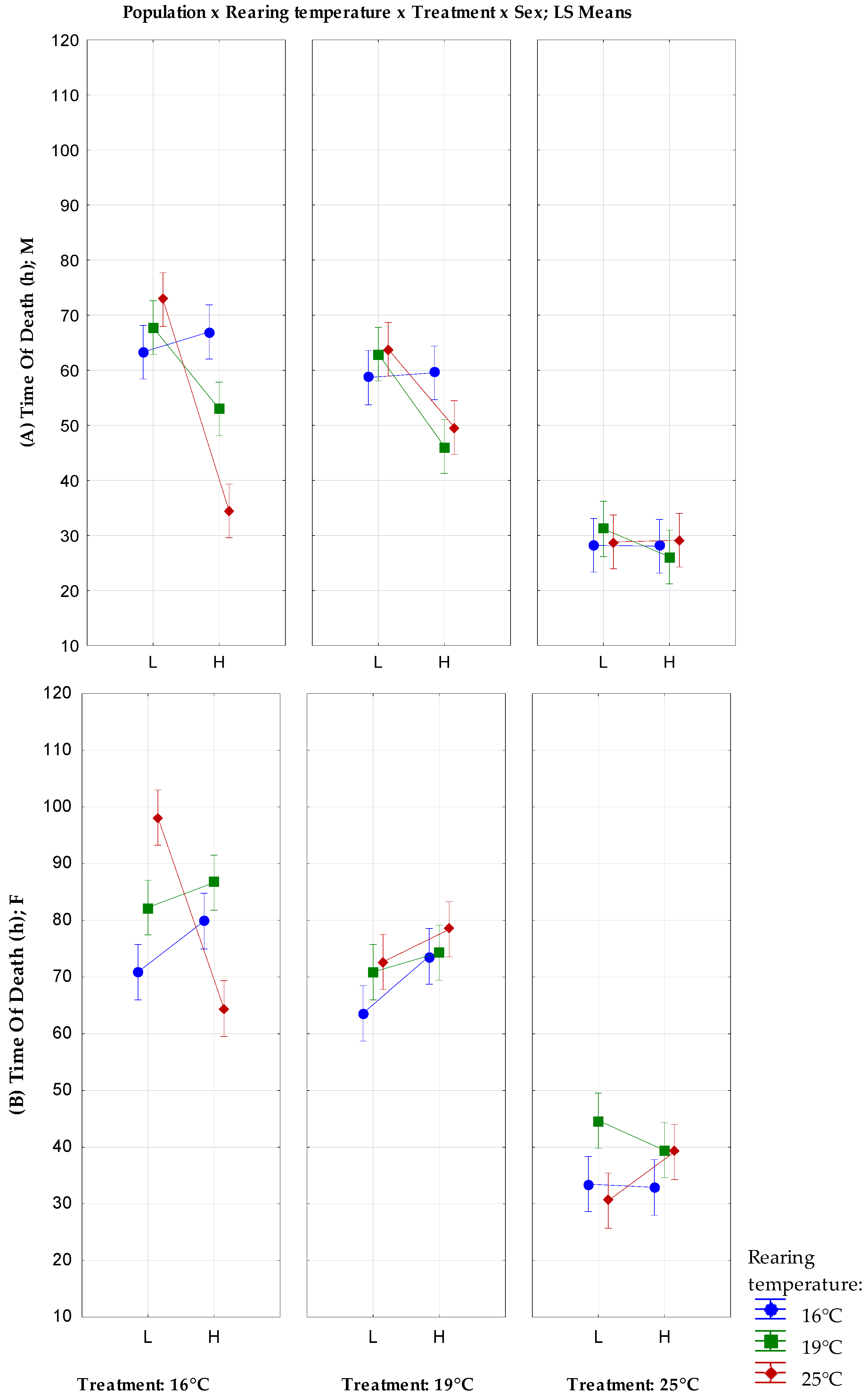
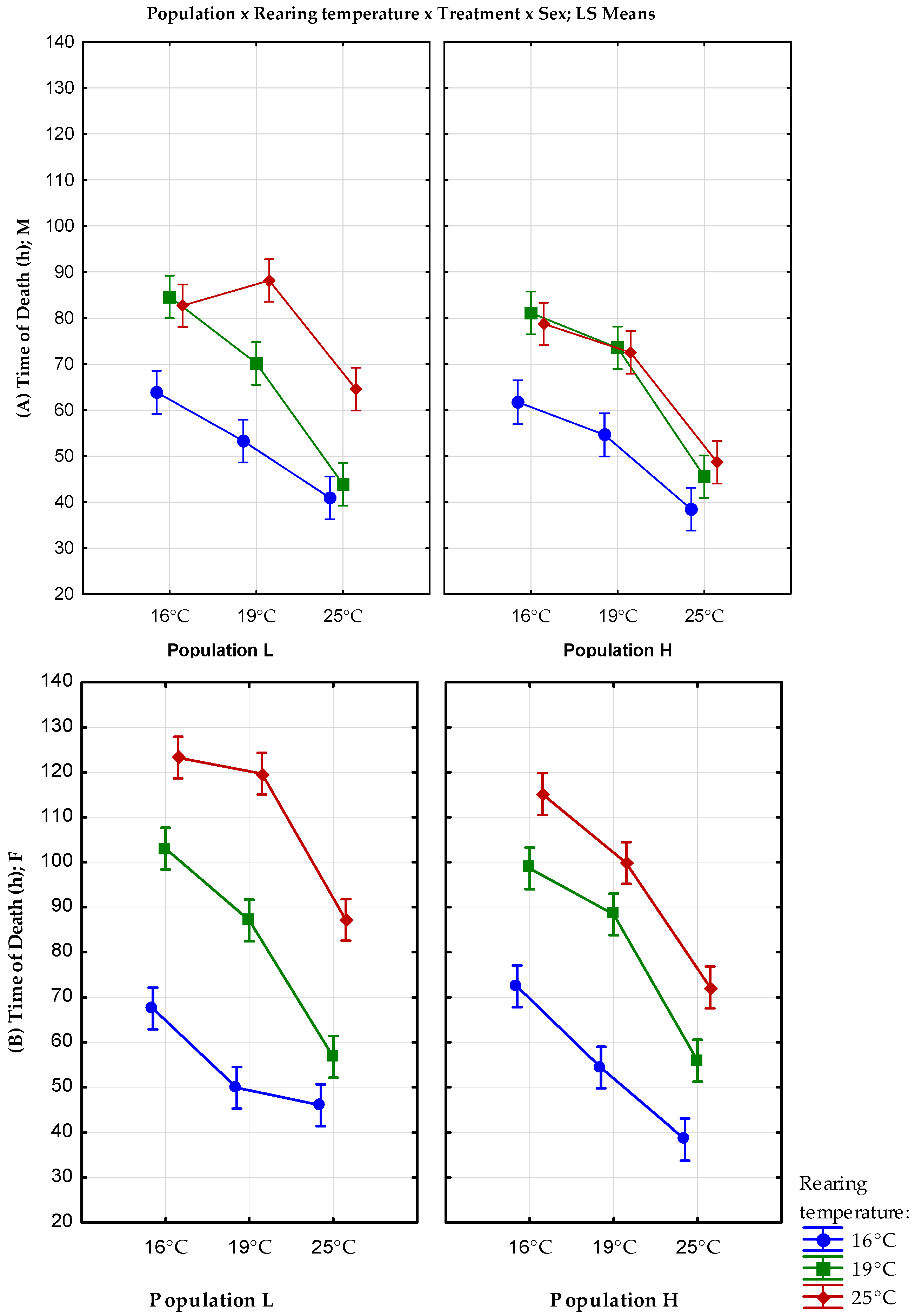
3.1. Desiccation Resistance
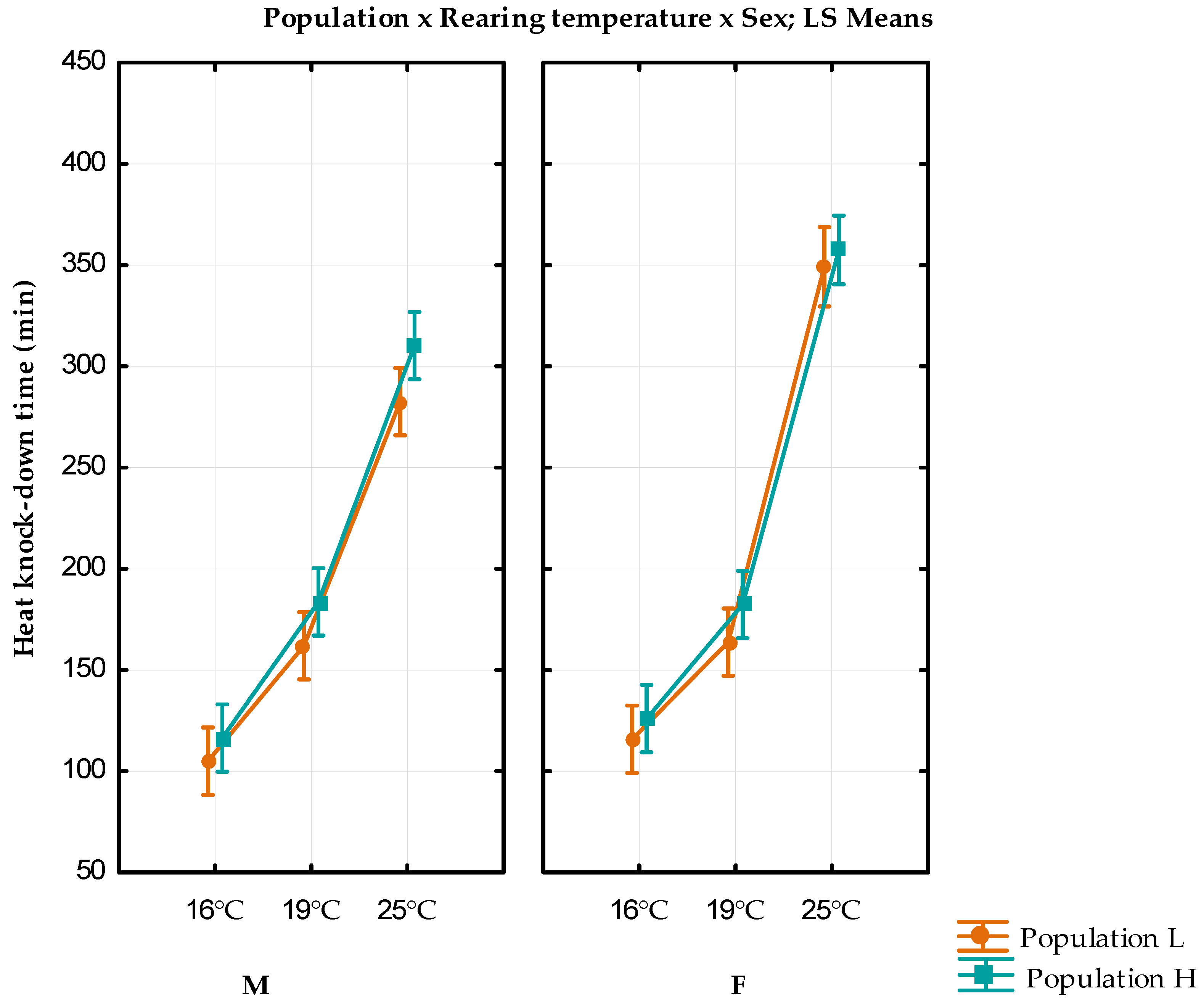
3.2. Heat Knock-Down Resistance
3.3. Starvation Resistance
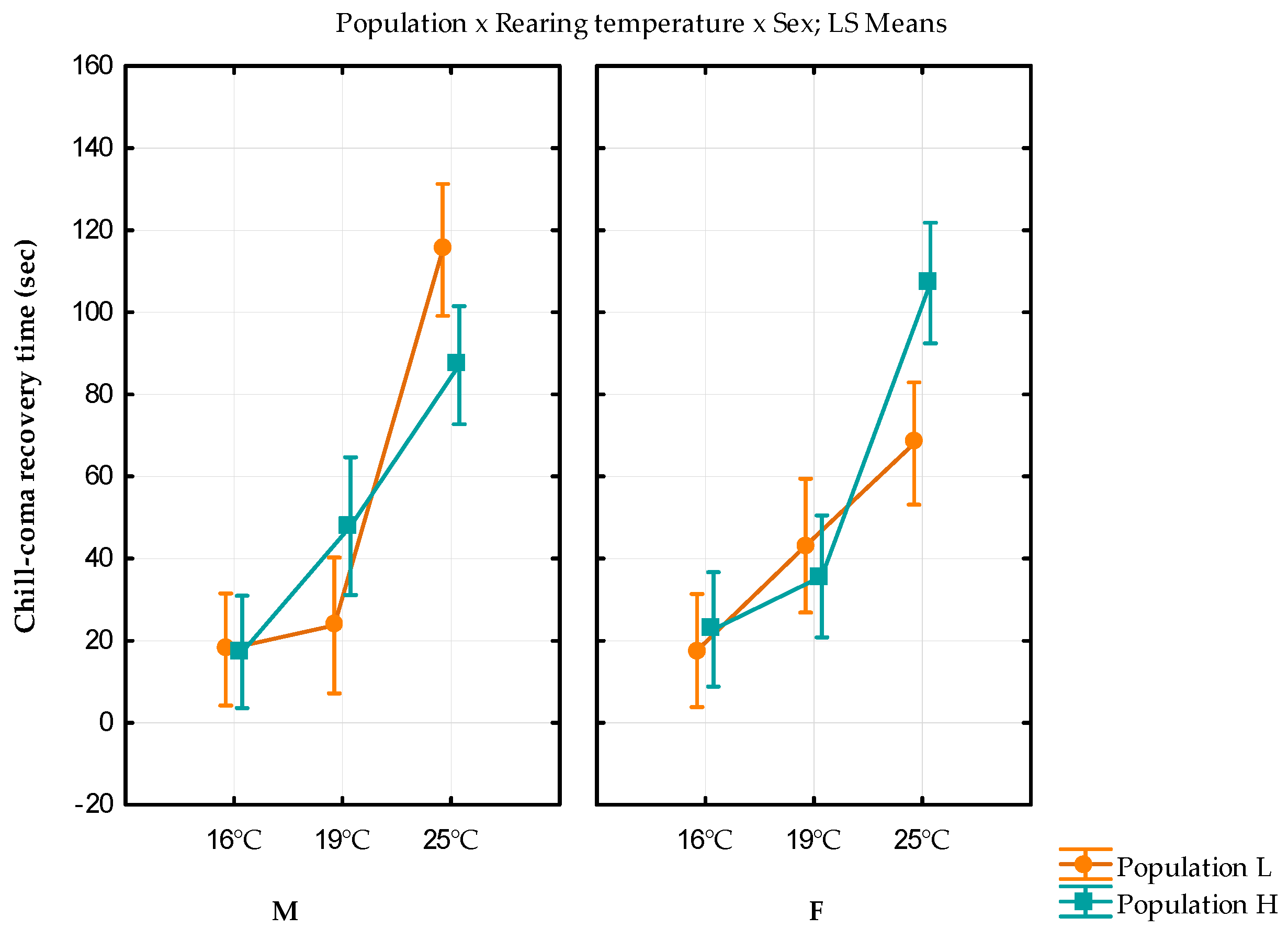
3.4. Chill Coma Recovery Time
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fischer, K.; Dierks, A.; Franke, K.; Geister, T.L.; Liszka, M.; Winter, S.; Pflicke, C. Environmental Effects on Temperature Stress Resistance in the Tropical Butterfly Bicyclus Anynana. PLoS ONE 2010, 5, e15284. [Google Scholar] [CrossRef]
- Karl, I.; Janowitz, S.A.; Fischer, K. Altitudinal life-history variation and thermal adaptation in the copper butterfly Lycaena tityrus. Oikos 2008, 117, 778–788. [Google Scholar] [CrossRef]
- Walsh, B.S.; Parratt, S.R.; Hoffmann, A.A.; Atkinson, D.; Snook, R.R.; Bretman, A.; Price, T.A.R. The Impact of Climate Change on Fertility. Trends Ecol. Evol. 2019, 34, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Buckley, L.B.; Kingsolver, J.G. Evolution of Thermal Sensitivity in Changing and Variable Climates. Annu. Rev. Ecol. Evol. Syst. 2021, 52, 563–586. [Google Scholar] [CrossRef]
- IPCC. The Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2018. [Google Scholar]
- Kumar, N.H.; Murali, M.; Girish, H.; Chandrashekar, S.; Amruthesh, K.; Sreenivasa, M.; Jagannath, S. Impact of climate change on biodiversity and shift in major biomes. In Global Climate Change; Elsevier: Amsterdam, The Netherlands, 2021; pp. 33–44. [Google Scholar]
- David, J.R.; Gibert, P.; Moreteau, B.; Gilchrist, G.W.; Huey, R.B. The Fly That Came in from the Cold: Geographic Variation of Recovery Time from Low-Temperature Exposure in Drosophila subobscura. Funct. Ecol. 2003, 17, 425–430. [Google Scholar] [CrossRef]
- Warren, R.; Price, J.; Jenkins, R. Chapter 4—Climate change and terrestrial biodiversity. In The Impacts of Climate Change; Letcher, T.M., Ed.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 85–114. [Google Scholar]
- Markow, T.A. The secret lives of Drosophila flies. eLife 2015, 4, e06793. [Google Scholar] [CrossRef] [PubMed]
- Balanyà, J.; Huey, R.B.; Gilchrist, G.W.; Serra, L. The chromosomal polymorphism of Drosophila subobscura: A microevolutionary weapon to monitor global change. Heredity 2009, 103, 364–367. [Google Scholar] [CrossRef] [PubMed]
- Loeschcke, V.; Sørensen, J.G.; Kristensen, T.N. Ecologically relevant stress resistance: From microarrays and quantitative trait loci to candidate genes—A research plan and preliminary results using Drosophila as a model organism and climatic and genetic stress as model stresses. J. Biosci. 2004, 29, 503–511. [Google Scholar] [CrossRef]
- Kristensen, T.N.; Dahlgaard, J.; Loeschcke, V. Effects of inbreeding and environmental stress on fitness—Using Drosophila buzzatii as a model organism. Conserv. Genet. 2003, 4, 453–465. [Google Scholar] [CrossRef]
- Xue, Q.; Majeed, M.Z.; Zhang, W.; Ma, C.-S. Adaptation of Drosophila species to climate change—A literature review since 2003. J. Integr. Agric. 2019, 18, 805–814. [Google Scholar] [CrossRef]
- Collinge, J.E.; Hoffmann, A.A.; McKechnie, S.W. Altitudinal patterns for latitudinally varying traits and polymorphic markers in Drosophila melanogaster from eastern Australia. J. Evol. Biol. 2006, 19, 473–482. [Google Scholar] [CrossRef]
- Kenig, B.; Jelić, M.; Kurbalija, Z.; Stamenković-Radak, M.; Anđelković, M. Inversion polymorphism in populations of Drosophila subobscura from urban and non-urban environment. Arch. Biol. Sci. 2010, 62, 565–574. [Google Scholar] [CrossRef]
- Rodríguez-Trelles, F.; Rodríguez, M.A. Rapid micro-evolution and loss of chromosomal diversity in Drosophila in response to climate warming. Evol. Ecol. 1998, 12, 829–838. [Google Scholar] [CrossRef]
- Rodríguez–Trelles, F.; Rodríguez, M.A.; Scheiner, S.M. Tracking the genetic effects of global warming: Drosophila and other model systems. Conserv. Ecol. 1998, 2, 2. Available online: http://www.consecol.org/vol2/iss2/art2/ (accessed on 16 December 2021). [CrossRef]
- Davis, A.J.; Lawton, J.H.; Shorrocks, B.; Jenkinson, L.S. Individualistic species responses invalidate simple physiological models of community dynamics under global environmental change. J. Anim. Ecol. 1998, 67, 600–612. [Google Scholar] [CrossRef]
- Stamenkovic-Radak, M.; Kenig, B.; Djurakic, M.R.; Jelj, M.; Eric, K.; Andjelkovj, M. Associations between environmental variability and inversion polymorphism of Drosophila subobscura: Meta-analysis of populations from the Central Balkans. Clim. Res. 2019, 77, 205–217. [Google Scholar] [CrossRef]
- Rezende, E.L.; Balanyà, J.; Rodríguez-Trelles, F.; Rego, C.; Fragata, I.; Matos, M.; Serra, L.; Santos, M. Climate change and chromosomal inversions in Drosophila subobscura. Clim. Res. 2010, 43, 103–114. [Google Scholar] [CrossRef]
- Vuković, A.J.; Vujadinović, M.P.; Rendulić, S.M.; Đurđević, V.S.; Ruml, M.M.; Babić, V.P.; Popović, D.P. Global warming impact on climate change in Serbia for the period 1961-2100. Therm. Sci. 2018, 22, 2267–2280. [Google Scholar] [CrossRef]
- Stamenkovic-Radak, M.; Rasic, G.; Savic, T.; Kalajdzic, P.; Kurbalija, Z.; Kenig, B.; Andjelkovic, M. Monitoring of the genetic structure of natural populations: Change of the effective population size and inversion polymorphism in Drosophila subobscura. Genetica 2008, 133, 57–63. [Google Scholar] [CrossRef]
- Ayhan, N.; Güler, P.; Onder, B.S. Altitudinal variation in lifespan of Drosophila melanogaster populations from the Firtina Valley, northeastern Turkey. J. Therm. Biol. 2016, 61, 91–97. [Google Scholar] [CrossRef]
- Dahlgaard, J.; Hasson, E.; Loeschcke, V. Behavioral differentiation in oviposition activity in Drosophila buzzatii from highland and lowland populations in argentina: Plasticity or thermal adaptation? Evol. Int. J. Org. Evol. 2001, 55, 738–747. [Google Scholar] [CrossRef]
- Parkash, R.; Tyagi, P.K.; Sharma, I.; Rajpurohit, S. Adaptations to environmental stress in altitudinal populations of two Drosophila species. Physiol. Entomol. 2005, 30, 353–361. [Google Scholar] [CrossRef]
- Sambucetti, P.; Loeschcke, V.; Norry, F.M. Developmental time and size-related traits in Drosophila buzzatii along an altitudinal gradient from Argentina. Hereditas 2006, 143, 77–83. [Google Scholar] [CrossRef]
- Pitchers, W.; Pool, J.E.; Dworkin, I. Altitudinal clinal variation in wing size and shape in African Drosophila melanogaster: One cline or many? Evol. Int. J. Org. Evol. 2013, 67, 438–452. [Google Scholar] [CrossRef]
- Klepsatel, P.; Gáliková, M.; Huber, C.D.; Flatt, T. Similarities and differences in altitudinal versus latitudinal variation for morphological traits in Drosophila melanogaster. Evol. Int. J. Org. Evol. 2014, 68, 1385–1398. [Google Scholar] [CrossRef]
- Bijlsma, R.; Loeschcke, V. Environmental stress, adaptation and evolution: An overview. J. Evol. Biol. 2005, 18, 744–749. [Google Scholar] [CrossRef]
- Sgrò, C.M.; Overgaard, J.; Kristensen, T.N.; Mitchell, K.A.; Cockerell, F.E.; Hoffmann, A.A. A comprehensive assessment of geographic variation in heat tolerance and hardening capacity in populations of Drosophila melanogaster from eastern Australia. J. Evol. Biol. 2010, 23, 2484–2493. [Google Scholar] [CrossRef]
- Rion, S.; Kawecki, T.J. Evolutionary biology of starvation resistance: What we have learned from Drosophila. J. Evol. Biol. 2007, 20, 1655–1664. [Google Scholar] [CrossRef]
- Kellermann, V.; Hoffmann, A.A.; Kristensen, T.N.; Moghadam, N.N.; Loeschcke, V. Experimental Evolution under Fluctuating Thermal Conditions Does Not Reproduce Patterns of Adaptive Clinal Differentiation in Drosophila melanogaster. Am. Nat. 2015, 186, 582–593. [Google Scholar] [CrossRef]
- Erić, K.; Erić, P.; Davidović, S.; Veselinović, M.S.; Patenković, A.; Stamenković-Radak, M.; Tanasković, M. Thermal Adaptation and Stress Resistance in D. subobscura Populations from Two Altitudes at Stara Planina Mountain (Serbia). In Proceedings of the 1st International Electronic Conference on Entomology, Online Conference, 1 July 2021. [Google Scholar]
- Partridge, L.; Barrie, B.; Fowler, K.; French, V. Evolution and development of body size and cell size in Drosophila melanogaster in response to temperature. Evol. Int. J. Org. Evol. 1994, 48, 1269–1276. [Google Scholar] [CrossRef]
- Hoffmann, A.A.; Sørensen, J.G.; Loeschcke, V. Adaptation of Drosophila to temperature extremes: Bringing together quantitative and molecular approaches. J. Therm. Biol. 2003, 28, 175–216. [Google Scholar] [CrossRef]
- Zamudio, K.R.; Huey, R.B.; Crill, W.D. Bigger isn’t always better: Body size, developmental and parental temperature and male territorial success in Drosophila melanogaster. Anim. Behav. 1995, 49, 671–677. [Google Scholar] [CrossRef]
- French, V.; Feast, M.; Partridge, L. Body size and cell size in Drosophila: The developmental response to temperature. J. Insect Physiol. 1998, 44, 1081–1089. [Google Scholar] [CrossRef]
- McCabe, J.; Partridge, L. An interaction between environmental temperature and genetic variation for body size for the fitness of adult female Drosophila melanogaster. Evol. Int. J. Org. Evol. 1997, 51, 1164–1174. [Google Scholar] [CrossRef]
- Reeve, M.; Fowler, K.; Partridge, L. Increased body size confers greater fitness at lower experimental temperature in male Drosophila melanogaster. J. Evol. Biol. 2000, 13, 836–844. [Google Scholar] [CrossRef]
- Gilchrist, G.W.; Jeffers, L.M.; West, B.; Folk, D.G.; Suess, J.; Huey, R.B. Clinal patterns of desiccation and starvation resistance in ancestral and invading populations of Drosophila subobscura. Evol. Appl. 2008, 1, 513–523. [Google Scholar] [CrossRef]
- Sorensen, J.G.; Dahlgaard, J.; Loeschcke, V. Genetic Variation in Thermal Tolerance among Natural Populations of Drosophila buzzatii: Down Regulation of Hsp70 Expression and Variation in Heat Stress Resistance Traits. Funct. Ecol. 2001, 15, 289–296. [Google Scholar] [CrossRef]
- Chown, S.L.; Chown, S.; Nicolson, S. Insect Physiological Ecology: Mechanisms and Patterns; Oxford University Press: Oxford, UK, 2004. [Google Scholar]
- Krupp, J.J.; Nayal, K.; Wong, A.; Millar, J.G.; Levine, J.D. Desiccation resistance is an adaptive life-history trait dependent upon cuticular hydrocarbons, and influenced by mating status and temperature in D. melanogaster. J. Insect Physiol. 2020, 121, 103990. [Google Scholar] [CrossRef]
- Kellermann, V.; McEvey, S.F.; Sgrò, C.M.; Hoffmann, A.A. Phenotypic Plasticity for Desiccation Resistance, Climate Change, and Future Species Distributions: Will Plasticity Have Much Impact? Am. Nat. 2020, 196, 306–315. [Google Scholar] [CrossRef]
- Bowler, K. Acclimation, heat shock and hardening. J. Therm. Biol. 2005, 30, 125–130. [Google Scholar] [CrossRef]
- Loeschcke, V.; Sørensen, J.G. Acclimation, heat shock and hardening—A response from evolutionary biology. J. Therm. Biol. 2005, 30, 255–257. [Google Scholar] [CrossRef]
- Franks, S.J.; Hoffmann, A.A. Genetics of Climate Change Adaptation. Annu. Rev. Genet. 2012, 46, 185–208. [Google Scholar] [CrossRef]
- Hoffmann, A.A.; Sgrò, C.M. Climate change and evolutionary adaptation. Nature 2011, 470, 479–485. [Google Scholar] [CrossRef]
- Stazione, L.; Norry, F.M.; Gomez, F.H.; Sambucetti, P. Heat knockdown resistance and chill-coma recovery as correlated responses to selection on mating success at high temperature in Drosophila buzzatii. Ecol. Evol. 2020, 10, 1998–2006. [Google Scholar] [CrossRef]
- McCue, M.D. Starvation physiology: Reviewing the different strategies animals use to survive a common challenge. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2010, 156, 1–18. [Google Scholar] [CrossRef]
- Schwasinger-Schmidt, T.E.; Kachman, S.D.; Harshman, L.G. Evolution of starvation resistance in Drosophila melanogaster: Measurement of direct and correlated responses to artificial selection. J. Evol. Biol. 2012, 25, 378–387. [Google Scholar] [CrossRef]
- Sørensen, J.G.; Norry, F.M.; Scannapieco, A.C.; Loeschcke, V. Altitudinal variation for stress resistance traits and thermal adaptation in adult Drosophila buzzatii from the New World. J. Evol. Biol. 2005, 18, 829–837. [Google Scholar] [CrossRef]
- Hasson, E.; Rodríguez, C.; Fanara, J.J.; Naveira, H.; Reig, O.A.; Fontdevila, A. The evolutionary history of Drosophila buzzatti. XXVI. Macrogeographic patterns of inversion polymorphism in New World populations. J. Evol. Biol. 1995, 8, 369–384. [Google Scholar] [CrossRef]
- Lee, K.P.; Jang, T. Exploring the nutritional basis of starvation resistance in Drosophila melanogaster. Funct. Ecol. 2014, 28, 1144–1155. [Google Scholar] [CrossRef]
- Skorupa, D.A.; Dervisefendic, A.; Zwiener, J.; Pletcher, S.D. Dietary composition specifies consumption, obesity, and lifespan in Drosophila melanogaster. Aging Cell 2008, 7, 478–490. [Google Scholar] [CrossRef]
- Chippindale, A.K.; Chu, T.J.; Rose, M.R. Complex trade-offs and the evolution of starvation resistance in Drosophila melanogaster. Evol. Int. J. Org. Evol. 1996, 50, 753–766. [Google Scholar] [CrossRef] [PubMed]
- Djawdan, M.; Chippindale, A.K.; Rose, M.R.; Bradley, T.J. Metabolic reserves and evolved stress resistance in Drosophila melanogaster. Physiol. Zool. 1998, 71, 584–594. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.A.; Harshman, L.G. Desiccation and starvation resistance in Drosophila: Patterns of variation at the species, population and intrapopulation levels. Heredity 1999, 83, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.A.; Hallas, R.; Anderson, A.R.; Telonis-Scott, M. Evidence for a robust sex-specific trade-off between cold resistance and starvation resistance in Drosophila melanogaster. J. Evol. Biol. 2005, 18, 804–810. [Google Scholar] [CrossRef]
- Andersen, J.L.; Manenti, T.; Sørensen, J.G.; MacMillan, H.A.; Loeschcke, V.; Overgaard, J. How to assess Drosophila cold tolerance: Chill coma temperature and lower lethal temperature are the best predictors of cold distribution limits. Funct. Ecol. 2015, 29, 55–65. [Google Scholar] [CrossRef]
- Gibert, P.; Moreteau, B.; Pétavy, G.; Karan, D.; David, J.R. Chill-coma tolerance, a major climatic adaptation among drosophila species. Evol. Int. J. Org. Evol. 2001, 55, 1063–1068. [Google Scholar] [CrossRef]
- Krimbas, C.B.; Krimpas, K.V. Drosophila subobscura: Biology, Genetics and Inversion Polymorphism; Kovač: Mishawaka, Indiana, 1993. [Google Scholar]
- Rego, C.; Balanyà, J.; Fragata, I.; Matos, M.; Rezende, E.L.; Santos, M. Clinal patterns of chromosomal inversion polymorphisms in Drosophila subobscura are partly associated with thermal preferences and heat stress resistance. Evol. Int. J. Org. Evol. 2010, 64, 385–397. [Google Scholar] [CrossRef]
- Kristensen, T.N.; Ketola, T.; Kronholm, I. Adaptation to environmental stress at different timescales. Ann. N. Y. Acad. Sci. 2020, 1476, 5–22. [Google Scholar] [CrossRef]
- Hoffmann, A.A.; Anderson, A.; Hallas, R. Opposing clines for high and low temperature resistance in Drosophila melanogaster. Ecol. Lett. 2002, 5, 614–618. [Google Scholar] [CrossRef]
- Hoffmann, A.A.; Sgrò, C.M.; Kristensen, T.N. Revisiting Adaptive Potential, Population Size, and Conservation. Trends Ecol. Evol. 2017, 32, 506–517. [Google Scholar] [CrossRef]
- Davis, H.E.; Cheslock, A.; MacMillan, H.A. Chill coma onset and recovery fail to reveal true variation in thermal performance among populations of Drosophila melanogaster. Sci. Rep. 2021, 11, 10876. [Google Scholar] [CrossRef] [PubMed]
- Savic Veselinovic, M.; Kurbalija Novicic, Z.; Kenig, B.; Jelic, M.; Patenkovic, A.; Tanaskovic, M.; Pertoldi, C.; Stamenkovic-Radak, M.; Andjelkovic, M. Local adaptation at fine spatial scale through chromosomal inversions and mito-nuclear epistasis: Findings in Drosophila subobscura (Diptera: Drosophilidae). Eur. J. Entomol. 2019, 116, 492–503. [Google Scholar] [CrossRef]
- Porcelli, D.; Gaston, K.J.; Butlin, R.K.; Snook, R.R. Local adaptation of reproductive performance during thermal stress. J. Evol. Biol. 2017, 30, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M.T. Cold and heat tolerance of drosophilid flies with reference to their latitudinal distributions. Oecologia 2004, 140, 442–449. [Google Scholar] [CrossRef]
- Santos, M.; Iriarte, P.F.; Céspedes, W.; Balanyà, J.; Fontdevila, A.; Serra, L. Swift laboratory thermal evolution of wing shape (but not size) in Drosophila subobscura and its relationship with chromosomal inversion polymorphism. J. Evol. Biol. 2004, 17, 841–855. [Google Scholar] [CrossRef]
- Simões, P.; Fragata, I.; Santos, J.; Santos, M.A.; Santos, M.; Rose, M.R.; Matos, M. How phenotypic convergence arises in experimental evolution. Evol. Int. J. Org. Evol. 2019, 73, 1839–1849. [Google Scholar] [CrossRef]
- Moreteau, B.; Morin, J.P.; Gibert, P.; Pétavy, G.; Pla, É.; David, J.R. Evolutionary changes of non linearreaction norms according to thermal adaptation: A comparison of two Drosophila species. C. R. Acad. Sci. 1997, 320, 833–841. [Google Scholar] [CrossRef]
| Population L | Population H | |||||
|---|---|---|---|---|---|---|
| Rearing Temperature | Treatment | Sex | Time of Death | Variance | Time of Death | Variance |
| 16 °C | 16 °C | F | 70.86 ± 2.06 | 214.1229 | 79.85 ± 2.84 | 394.25 |
| M | 63.28 ± 1.83 | 167.3486 | 66.98 ± 2.76 | 373.8954 | ||
| 19 °C | F | 63.6 ± 2.35 | 134.8163 | 73.63 ± 2.35 | 269.6539 | |
| M | 58.67 ± 1.91 | 178.4328 | 59.54 ± 1.42 | 67.84354 | ||
| 25 °C | F | 33.48 ± 1.44 | 103.6016 | 32.90 ± 1.18 | 100.2943 | |
| M | 28.2 ± 0.91 | 41.71429 | 28.04 ± 0.93 | 43.26367 | ||
| 19 °C | 16 °C | F | 82.23 ± 3.16 | 510.4235 | 86.64 ± 3.10 | 481.8678 |
| M | 67.76 ± 2.22 | 246.9616 | 52.98 ± 2.33 | 249.5302 | ||
| 19 °C | F | 70.86 ± 2.37 | 280.6535 | 74.28 ± 1.66 | 138.5322 | |
| M | 62.94 ± 2.17 | 234.5065 | 46.14 ± 1.42 | 100.7759 | ||
| 25 °C | F | 44.68 ± 1.19 | 159.5282 | 39.44 ± 1.35 | 90.45551 | |
| M | 30.28 ± 1.02 | 52.36898 | 26.1 ± 1.03 | 53.43878 | ||
| 25 °C | 16 °C | F | 98.14 ± 5.00 | 1250.735 | 64.44 ± 4.06 | 823.1902 |
| M | 72.84 ± 4.21 | 886.7086 | 34.46 ± 2.18 | 237.2739 | ||
| 19 °C | F | 72.64 ± 5.01 | 1256.235 | 78.4 ± 4.18 | 872.0816 | |
| M | 63.82 ± 2.67 | 356.6404 | 49.62 ± 2.22 | 245.5873 | ||
| 25 °C | F | 30.56 ± 1.42 | 100.8229 | 39.16 ± 1.91 | 181.6065 | |
| M | 28.82 ± 1.58 | 125.0486 | 29.14 ± 1.17 | 68.57184 | ||
| Population L | Population H | ||||
|---|---|---|---|---|---|
| Rearing Temperature | Sex | Heat Knock-Down Time | Variance | Heat Knock-Down Time | Variance |
| 16 °C | F | 115.8 ± 3.93 | 771.7959 | 126 ± 3.53 | 624.4898 |
| M | 105 ± 2.74 | 376.5306 | 116.4 ± 3.16 | 501.0612 | |
| 19 °C | F | 163.8 ± 3.66 | 668.9388 | 182.4 ± 5.06 | 1279.837 |
| M | 162 ± 3.64 | 661.2245 | 183.6 ± 4.67 | 1088.816 | |
| 25 °C | F | 310.2 ± 12.99 | 8434.653 | 357.5 ± 14.06 | 9491.489 |
| M | 349.17 ± 17.77 | 11,373.57 | 282.6 ± 14.32 | 10,252.29 | |
| Population L | Population H | |||||
|---|---|---|---|---|---|---|
| Rearing Temperature | Treatment | Sex | Time of Death | Variance | Time of Death | Variance |
| 16 °C | 16 °C | F | 67.5 ± 2.26 | 256.1327 | 72.42 ± 2.45 | 300.8608 |
| M | 63.86 ± 1.58 | 121.875 | 61.72 ± 2.05 | 197.1175 | ||
| 19 °C | F | 48.88 ± 2.10 | 221.2914 | 54.36 ± 1.71 | 147.1739 | |
| M | 53.58 ± 1.30 | 85.79755 | 55.75 ± 1.79 | 154.2766 | ||
| 25 °C | F | 46.02 ± 0.99 | 48.71388 | 38.46 ± 1.35 | 90.62082 | |
| M | 40.92 ± 0.70 | 24.52408 | 38.46 ± 0.93 | 43.23306 | ||
| 19 °C | 16 °C | F | 103.02 ± 3.42 | 585.0404 | 98.64 ± 3.02 | 455.0106 |
| M | 84.6 ± 1.92 | 184.0408 | 81.12 ± 1.81 | 161.9853 | ||
| 19 °C | F | 87.06 ± 1.90 | 180.9147 | 88.44 ± 2.02 | 204.7004 | |
| M | 70.14 ± 2.29 | 262.6127 | 73.5 ± 2.18 | 237.398 | ||
| 25 °C | F | 56.76 ± 1.54 | 117.8596 | 55.92 ± 1.48 | 109.3812 | |
| M | 43.86 ± 1.00 | 50.28612 | 45.54 ± 0.92 | 42.86571 | ||
| 25 °C | 16 °C | F | 123.24 ± 3.48 | 604.5943 | 115.4 ± 2.61 | 341.5922 |
| M | 82.68 ± 2.41 | 290.6302 | 78.72 ± 2.80 | 391.4302 | ||
| 19 °C | F | 119.7 ± 3.19 | 508.1327 | 99.84 ± 3.98 | 790.5453 | |
| M | 88.14 ± 2.03 | 207.1433 | 72.54 ± 3.44 | 593.1514 | ||
| 25 °C | F | 87.18 ± 3.20 | 512.2322 | 72.18 ± 4.12 | 851.6608 | |
| M | 64.56 ± 1.99 | 197.7208 | 48.66 ± 2.82 | 396.4739 | ||
| Population L | Population H | ||||
|---|---|---|---|---|---|
| Rearing Temperature | Sex | Chill Coma Recovery Time | Variance | Chill Coma Recovery Time | Variance |
| 16 °C | F | 17.61 ± 1.72 | 143.7924 | 22.74 ± 1.75 | 146.3962 |
| M | 17.85 ± 1.77 | 156.0721 | 17.26 ± 1.83 | 167.116 | |
| 19 °C | F | 43.14 ± 5.76 | 1160.334 | 35.65 ± 2.53 | 268.8758 |
| M | 23.72 ± 2.46 | 206.3637 | 65.80 ± 12.67 | 6901.601 | |
| 25 °C | F | 68.04 ± 9.10 | 3481.358 | 107.11 ± 14.31 | 8810.542 |
| M | 115.21 ± 15.60 | 8751.539 | 87.09 ± 9.99 | 4484.564 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Erić, K.; Patenković, A.; Erić, P.; Davidović, S.; Veselinović, M.S.; Stamenković-Radak, M.; Tanasković, M. Stress Resistance Traits under Different Thermal Conditions in Drosophila subobscura from Two Altitudes. Insects 2022, 13, 138. https://doi.org/10.3390/insects13020138
Erić K, Patenković A, Erić P, Davidović S, Veselinović MS, Stamenković-Radak M, Tanasković M. Stress Resistance Traits under Different Thermal Conditions in Drosophila subobscura from Two Altitudes. Insects. 2022; 13(2):138. https://doi.org/10.3390/insects13020138
Chicago/Turabian StyleErić, Katarina, Aleksandra Patenković, Pavle Erić, Slobodan Davidović, Marija Savić Veselinović, Marina Stamenković-Radak, and Marija Tanasković. 2022. "Stress Resistance Traits under Different Thermal Conditions in Drosophila subobscura from Two Altitudes" Insects 13, no. 2: 138. https://doi.org/10.3390/insects13020138
APA StyleErić, K., Patenković, A., Erić, P., Davidović, S., Veselinović, M. S., Stamenković-Radak, M., & Tanasković, M. (2022). Stress Resistance Traits under Different Thermal Conditions in Drosophila subobscura from Two Altitudes. Insects, 13(2), 138. https://doi.org/10.3390/insects13020138








