Identification and Biocontrol Potential of Entomopathogenic Nematodes and Their Endosymbiotic Bacteria in Apple Orchards against the Codling Moth, Cydia pomonella (L.) (Lepidoptera: Tortricidae)
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
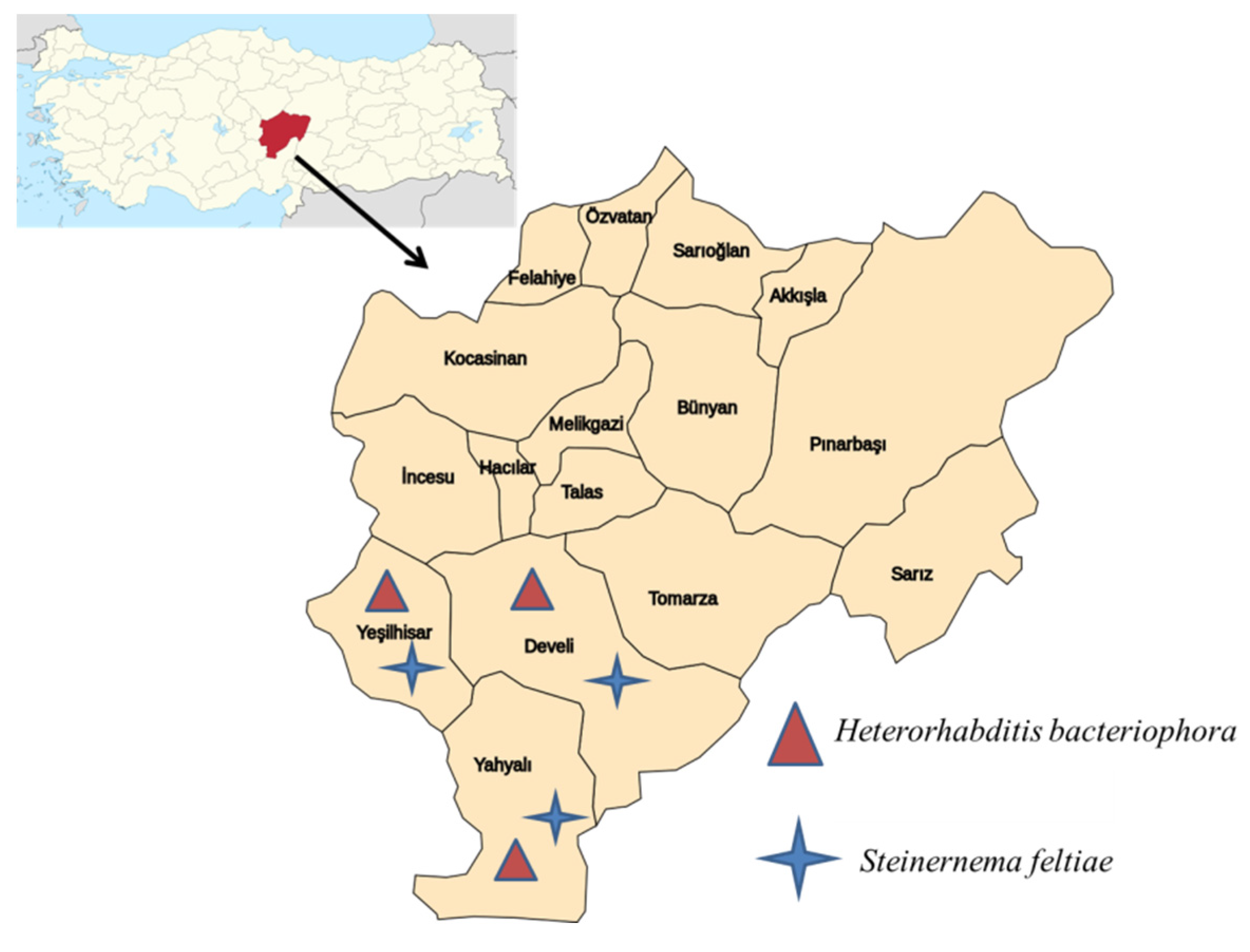
2.1. Soil Sampling
2.2. Isolation of Entomopathogenic Nematodes
2.3. Identification of Nematodes
2.4. Pathogenicity Screening on Galleria mellonella Larvae
2.5. Cydia pomonella
2.6. Isolation and Identification of Symbiotic Bacteria
2.7. Preparation of Cell-Free Bacterial Supernatants
2.8. Virulence Tests on Cydia pomonella Larvae
2.9. Evaluation of Pathogenicity of Symbiotic Bacteria
2.10. Statistical Analysis
3. Results
3.1. Survey and Identification of EPNs
3.2. Pathogenicity Screening on Galleria mellonella Larvae
3.3. Susceptibility of Cydia pomonella Larvae to Selected Isolates
3.4. Identification and Evaluation of the Symbiotic Bacteria of Selected Isolates
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Crops and Livestock Products. 2022. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 9 August 2022).
- Moinina, A.; Lahlali, R.; Boulif, M. Important pests, diseases and weather conditions affecting apple production in Morocco: Current state and perspectives. Rev. Maroc. Des. Sci. Agron. Et. Vétérinaires 2019, 7, 2–25. [Google Scholar]
- Kuyulu, A.; Genç, H. Biology and laboratory rearing of codling moth, Cydia pomonella (L.) (Lepidoptera: Tortricidae) on its natural host “green immature apple” Malus domestica (Borkh) (Rosales: Rosaceae). Türk Tarım Doğa Bilim. Derg. 2019, 6, 546–556. [Google Scholar] [CrossRef]
- Hepdurgun, B.; Zumreoglu, A.N.; Demir, S.T.K.; Ibis, M.A.N. Early studies on mating disruption technique of codling moth, Cydia pomonella, in the Aegean Region, Türkiye. IOBC Wprs Bull. 2001, 24, 43–46. [Google Scholar]
- Reyes, M.; Franck, P.; Charmillot, P.J.; Ioriatti, C.; Olivares, J.; Pasqualini, E.; Sauphanor, B. Diversity of insecticide resistance mechanisms and spectrum in European populations of the codling moth, Cydia pomonella. Pest. Manag. Sci. 2007, 63, 890–902. [Google Scholar] [CrossRef] [PubMed]
- İşci, M.; Ay, R. Determination of resistance and resistance mechanisms to thiacloprid in Cydia pomonella L. (Lepidoptera: Tortricidae) populations collected from apple orchards in Isparta Province, Türkiye. Crop. Prot. 2017, 91, 82–88. [Google Scholar] [CrossRef]
- Ju, D.; Mota-Sanchez, D.; Fuentes-Contreras, E.; Zhang, Y.L.; Wang, X.Q.; Yang, X.Q. Insecticide resistance in the Cydia pomonella (L): Global status, mechanisms, and research directions. Pestic. Biochem. Phys. 2021, 178, 104925. [Google Scholar] [CrossRef] [PubMed]
- Doemoetoerova, M.; Hercegova, A.; Matisova, E. Monitoring of pesticide residues in apples from Slovakia for baby food production. Czech J. Food Sci. 2006, 24, 84. [Google Scholar] [CrossRef]
- Szpyrka, E.; Kurdziel, A.; Słowik-Borowiec, M.; Grzegorzak, M.; Matyaszek, A. Consumer exposure to pesticide residues in apples from the region of south-eastern Poland. Environ. Monit. Assess. 2013, 185, 8873–8878. [Google Scholar] [CrossRef] [PubMed]
- Toptanci, İ.; Kiralan, M.; Ramadan, M.F. Levels of pesticide residues in fruits and vegetables in the Turkish domestic markets. Environ. Sci. Pollut. Res. 2021, 28, 39451–39457. [Google Scholar] [CrossRef]
- Sánchez-Bayo, F. Insecticides mode of action in relation to their toxicity to non-target organisms. J. Environ. Anal. Toxicol. 2012, S4, S4-002. [Google Scholar]
- Lacey, L.A.; Unruh, T.R. Biological control of codling moth (Cydia pomonella, Lepidoptera: Tortricidae) and its role in integrated pest management, with emphasis on entomopathogens. Vedalia 2005, 12, 33–60. [Google Scholar]
- Jaffe, B.D.; Guédot, C.; Landolt, P.J. Mass-trapping codling moth, Cydia pomonella (Lepidopteran: Torticidae), using a kairomone lure reduces fruit damage in commercial apple orchards. J. Econ. Entomol. 2018, 111, 1983–1986. [Google Scholar] [CrossRef]
- Koppenhöfer, A.M.; Shapiro-Ilan, D.I.; Hiltpold, I. Entomopathogenic nematodes in sustainable food production. Front. Sustain. Food Syst. 2020, 4, 125. [Google Scholar] [CrossRef]
- Lacey, L.A.; Georgis, R. Entomopathogenic nematodes for control of insect pests above and below ground with comments on commercial production. J. Nematol. 2012, 44, 218. [Google Scholar]
- Boemare, N.E.; Akhurst, R.J. The genera Photorhabdus and Xenorhabdus. In The Prokaryotes: An Evolving Electronic Resource for the Microbiological Community; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.H., Stackebrandt, E., Eds.; Springer: New York, NY, USA, 2006; pp. 1–65. [Google Scholar]
- Shapiro-Ilan, D.I.; Hazir, S.; Glazer, I. Basic and applied research: Entomopathogenic nematodes. In Microbial Agents for Control of Insect Pests: From Discovery to Commercial Development and Use; Lacey, L.A., Ed.; Academic Press: San Diego, CA, USA, 2017; pp. 91–105. [Google Scholar]
- Gaugler, R.; Lewis, E.; Stuart, R.J. Ecology in the service of biological control: The case of entomopathogenic nematodes. Oecologia 1997, 109, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Lacey, L.A.; Chauvin, R.L. Entomopathogenic nematodes for control of diapausing codling moth (Lepidoptera: Tortricidae) in fruit bins. J. Econ. Entomol. 1999, 92, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Lacey, L.A.; Shapiro-Ilan, D.I.; Glenn, G.M. Post-application of anti-desiccant agents improves efficacy of entomopathogenic nematodes in formulated host cadavers or aqueous suspension against diapausing codling moth larvae (Lepidoptera: Tortricidae). Biocontrol. Sci. Technol. 2010, 20, 909–921. [Google Scholar] [CrossRef]
- Ahmad, M.J.; Mohiudin, S.; Askary, T.H.; Patil, J. Efficacy of indigenous strain of entomopathogenic nematode against diapausing larvae of Codling moth, Cydia pomonella L. (Lepidoptera: Tortricidae), in apple-growing hilly areas of Ladakh Region. Egypt J. Biol. Pest Control 2020, 30, 1–6. [Google Scholar] [CrossRef]
- Yağci, M.; Özdem, A.; Erdoğuş, F.D.; Ayan, E. Efficiency of entomopathogenic nematodes (Rhabditida: Heterorhabditidae and Steinernematidae) on the codling moth (Cydia pomonella L.) (Lepidoptera: Tortricidae) under controlled conditions. Egypt J. Biol. Pest Control 2021, 31, 1–5. [Google Scholar] [CrossRef]
- Campbell, J.F.; Lewis, E.E. Entomopathogenic nematode host-search strategies. Behav. Ecol. Parasites 2002, 5, 13–38. [Google Scholar]
- Lewis, E.E.; Campbell, J.; Griffin, C.; Kaya, H.; Peters, A. Behavioral ecology of entomopathogenic nematodes. Biol. Control 2006, 38, 66–79. [Google Scholar] [CrossRef]
- Rajagopal, R.; Bhatnagar, R.K. Insecticidal toxic proteins produced by Photorhabdus luminescens akhurstii, a symbiont of Heterorhabditis indica. J. Nematol. 2002, 34, 23. [Google Scholar]
- Blackburn, M.B.; Domek, J.M.; Gelman, D.B.; Hu, J.S. The broadly insecticidal Photorhabdus luminescens toxin complex a (Tca): Activity against the Colorado potato beetle, Leptinotarsa decemlineata, and sweet potato whitefly, Bemisia tabaci. J. Insect Sci. 2005, 5, 32. [Google Scholar] [CrossRef]
- Yüksel, E.; Imren, M.; Özdemir, E.; Bozbuğa, R.; Canhilal, R. Insecticidal effect of entomopathogenic nematodes and the cell-free supernatants from their symbiotic bacteria against different larval instars of Agrotis segetum (Denis & Schiffermüller) (Lepidoptera: Noctuidae). Egypt J. Biol. Pest Control 2022, 32, 1–7. [Google Scholar]
- Wu, S.; Toews, M.D.; Cottrell, T.E.; Schmidt, J.M.; Shapiro-Ilan, D.I. Toxicity of Photorhabdus luminescens and Xenorhabdus bovienii bacterial metabolites to pecan aphids (Hemiptera: Aphididae) and the lady beetle Harmonia axyridis (Coleoptera: Coccinellidae). J. Invertebr. Pathol. 2022, 194, 107806. [Google Scholar] [CrossRef] [PubMed]
- Bode, H.B. Entomopathogenic bacteria as a source of secondary metabolites. Curr. Opin. Chem. Biol. 2009, 13, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Bedding, R.A.; Akhurst, R.J. A simple technique for the detection of insect paristic rhabditid nematodes in soil. Nematologica 1975, 21, 109–110. [Google Scholar] [CrossRef]
- Tarasco, E.; Kary, N.E.; Fanelli, E.; Mohammadi, D.; Xingyue, L.; Mehrvar, A.; De Luca, F.; Troccoli, A. Modified bait insect technique in entomopathogens’ survey from the arasbaran biosphere reserve (Iran). Redia 2020, 103, 129–132. [Google Scholar] [CrossRef]
- Metwally, H.M.; Hafez, G.A.; Hussein, M.A.; Hussein, M.A.; Salem, H.A.; Saleh, M.M.E. Low cost artificial diet for rearing the greater wax moth, Galleria mellonella L. (Lepidoptera: Pyralidae) as a host for entomopathogenic nematodes. Egypt J. Biol. Pest Control 2012, 22, 15. [Google Scholar]
- Koppenhöfer, A.M. Nematodes. In Field Manual of Techniques in Invertebrate Pathology; Springer: Dordrecht, The Netherlands, 2000; pp. 283–301. [Google Scholar]
- Kaya, H.K.; Stock, S.P. Techniques in insect nematology. In Manual of 12 Techniques in Insect Pathology; Lawrence, A.L., Ed.; Academic Press: Wapato, WA, USA, 1997. [Google Scholar]
- Hominick, W.M. Biogeography. In Entomopathogenic Nematology; Gaugler, R., Ed.; CABI Publishing: Wallinford, UK, 2002; pp. 115–143. [Google Scholar]
- Nguyen, K.B.; Hunt, D.J.; Perry, R.N. Entomopathogenic Nematodes: Systematics, Phylogeny and Bacterial Symbionts. Nematology Monographs and Perspectives; Koninklijke Brill NV: Leiden, The Netherlands, 2007; pp. 569–576. [Google Scholar]
- Seinhorst, J.W. A rapid method for the transfer of nematodes from fixative to anhydrous glycerin. Nematologica 1959, 4, 67–69. [Google Scholar] [CrossRef]
- Waeyenberge, L.; Ryss, A.; Moens, M.; Pinochet, J.; Vrain, T. Molecular characterisation of 18 Pratylenchus species using rDNA restriction fragment length polymorphism. Nematology 2000, 2, 135–142. [Google Scholar] [CrossRef]
- Joyce, S.A.; Burnell, A.M.; Powers, T.O. Characterization of Heterorhabditis isolates by PCR amplification of segments of mtDNA and rDNA genes. J. Nematol. 1994, 26, 260–270. [Google Scholar]
- Ferris, V.R. Variation in spacer ribosomal DNA in some cystforming species of plant parasitic nematodes. 1993. Fundam. Appl. Nematol. 1993, 16, 177–184. [Google Scholar]
- Tailliez, P.; Pages, S.; Ginibre, N.; Boemare, N. New insights into diversity in the genus Xenorhabdus, including the description of ten novel species. IJSEM 2006, 56, 2805–2818. [Google Scholar] [CrossRef] [PubMed]
- Sergeant, M.; Baxter, L.; Jarrett, P.; Shaw, E.; Ousley, M.; Winstanley, C.; Morgan, J.A.W. Identification, typing and insecticidal activity of Xenorhabdus isolates from entomopathogenic nematodes in United Kingdom soil and characterization of the xpt toxin loci. AEM 2006, 72, 5895–5907. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547. [Google Scholar] [CrossRef]
- Cevizci, D.; Ulug, D.; Cimen, H.; Touray, M.; Hazir, S.; Cakmak, I. Mode of entry of secondary metabolites of the bacteria Xenorhabdus szentirmaii and X. nematophila into Tetranychus urticae, and their toxicity to the predatory mites Phytoseiulus persimilis and Neoseiulus californicus. J. Invertebr. Pathol. 2020, 174, 107418. [Google Scholar] [CrossRef] [PubMed]
- Yuksel, E.; Canhilal, R. Isolation, identification, and pathogenicity of entomopathogenic nematodes occurring in Cappadocia Region, Central Türkiye. Egypt J. Biol. Pest Control 2019, 29, 1–7. [Google Scholar] [CrossRef]
- Razia, M.; Padmanaban, R.; Karthik Raja, R.; Chellapandi, P.; Sivaramakrishnan, K. Monitoring entomopathogenic nematodes as ecological indicators in the cultivated lands of Karur District, Tamil Nadu: A Survey Report. J. Biol. 2011, 7, 16–19. [Google Scholar]
- Mráček, Z.; Bečvář, S.; Kindlmann, P.; Jersáková, J. Habitat preference for entomopathogenic nematodes, their insect hosts and new faunistic records for the Czech Republic. Biol. Control 2005, 34, 27–37. [Google Scholar] [CrossRef]
- Tarasco, E.; Triggiani, O.; Zamoum, M.; Oreste, M. Natural enemies emerged from Thaumetopoea pityocampa (Denis & Sciffermüller) (Lepidoptera Notodontidae) pupae in Southern Italy. Redia 2016, 98, 103–108. [Google Scholar]
- Kanga, F.N.; Waeyenberge, L.; Hauser, S.; Moens, M. Distribution of entomopathogenic nematodes in Southern Cameroon. J. Invertebr. Pathol. 2012, 109, 41–51. [Google Scholar] [CrossRef]
- Shamseldean, M.M.; Abd-Elgawad, M.M. Natural occurrence of insect pathogenic nematodes (Rhabditida: Heterorhabditidae) in Egyptian soils. Afro-Asian J. Nematol. 1994, 4, 151–154. [Google Scholar]
- Gwynn, R.L.; Richardson, P.N. Incidence of entomopathogenic nematodes in soil samples collected from Scotland, England and Wales. Fundam. Appl. Nematol. 1996, 19, 427–432. [Google Scholar]
- Kary, N.E.; Niknam, G.; Griffin, C.T.; Mohammadi, S.A.; Moghaddam, M. A survey of entomopathogenic nematodes of the families Steinernematidae and Heterorhabditidae (Nematoda: Rhabditida) in the north-west of Iran. Nematology 2009, 11, 107–116. [Google Scholar] [CrossRef]
- Alonso, V.; Nasrolahi, S.; Dillman, A.R. Host-specific activation of entomopathogenic nematode infective juveniles. Insects 2018, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.B.; Smart Jr, G.C. Steinernema scapterisci n. sp. (Rhabditida: Steinernematidae). J. Nematol. 1990, 22, 187. [Google Scholar] [PubMed]
- Canhilal, R.; Waeyenberge, L.; Toktay, H.; Bozbuga, R.; Çerintas, R.; Imren, M. Distribution of Steinernematids and Heterorhabditids (Rhabditida: Steinernematidae and Heterorhabditidae) in the Southern Anatolia Region of Türkiye. Egypt J. Biol. Pest Control 2016, 26, 6. [Google Scholar]
- Canhilal, R.; Waeyenberge, L.; Yüksel, E.; Koca, A.S.; Deniz, Y.; Imren, M. Assessment of the natural presence of entomopathogenic nematodes in Kayseri soils, Türkiye. Egypt J. Biol. Pest Control 2017, 27, 237–244. [Google Scholar]
- Hazir, S.; Keskin, N.; Stock, S.P.; Kaya, H.K.; Özcan, S. Diversity and distribution of entomopathogenic nematodes (Rhabditida: Steinernematidae and Heterorhabditidae) in Türkiye. Biodivers. Conserv. 2003, 12, 375–386. [Google Scholar] [CrossRef]
- Campos-Herrera, R.; Escuer, M.; Labrador, S.; Robertson, L.; Barrios, L.; Gutiérrez, C. Distribution of the entomopathogenic nematodes from La Rioja (Northern Spain). J. Invertebr. Pathol. 2007, 95, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Stock, S.P.; Blair, H.G. Entomopathogenic nematodes and their bacterial symbionts: The inside out of a mutualistic association. Symbiosis 2008, 46, 65–75. [Google Scholar]
- Laznik, Ž.; Tóth, T.; Lakatos, T.; Vidrih, M.; Trdan, S. First record of Steinernema feltiae (Filipjev) (Rhabditida: Steinernematidae) in Slovenia. Helminthologia 2009, 46, 135–138. [Google Scholar] [CrossRef]
- Gümüş Askar, A.; Yüksel, E.; Öcal, A.; Özer, G.; Kütük, H.; Dababat, A.; İmren, M. Identification and control potential of entomopathogenic nematodes against the black cutworm, Agrotis ipsilon (Fabricius) (Lepidoptera: Noctuidae), in potato-growing areas of Türkiye. JPDP 2022, 129, 1–12. [Google Scholar] [CrossRef]
- Kepenekci, İ. Entomopathogenic nematodes (Rhabditida) in the Mediterranean Region of Türkiye. Nematol. Mediterr. 2002, 30, 13–15. [Google Scholar]
- Yavuzaslanoglu, E.; Elekcioglu, I.H.; Nicol, J.M.; Sheedy, J.G. Resistance of Iranian landrace wheat to the cereal cyst nematode, Heterodera filipjevi. Australas. Plant Pathol. 2016, 45, 411–414. [Google Scholar] [CrossRef]
- Hazir, S.; Stock, S.P.; Kaya, H.K.; Koppenhöfer, A.M.; Keskin, N. Developmental temperature effects on five geographic isolates of the entomopathogenic nematode Steinernema feltiae (Nematoda: Steinernematidae). J. Invertebr. Pathol. 2001, 77, 243–250. [Google Scholar] [CrossRef]
- Mráček, Z.; Bečvář, S. Insect aggregations and entomopathogenic nematode occurrence. Nematology 2000, 2, 297–301. [Google Scholar]
- Stock, S.P.; Mrácek, Z.; Webster, J. Morphological variation between allopatric populations of Steinernema kraussei (Steiner, 1923) (Rhabditida: Steinernematidae). Nematology 2000, 2, 143–152. [Google Scholar] [CrossRef]
- Rolston, A.; Meade, C.; Boyle, S.; Kakouli-Duarte, T.; Downes, M. Intraspecific variation among isolates of the entomopathogenic nematode Steinernema feltiae from Bull Island, Ireland. Nematology 2009, 11, 439–451. [Google Scholar]
- Addis, T.; Mulawarman, M.; Waeyenberge, L.; Moens, M.; Viaene, N.; Ehlers, R.U. Identification and intraspecific variability of Steinernema feltiae strains from Cemoro Lawang village in Eastern Java, Indonesia. Russ. J. Nematol. 2011, 19, 21–29. [Google Scholar]
- Köhl, J.; Postma, J.; Nicot, P.; Ruocco, M.; Blum, B. Stepwise screening of microorganisms for commercial use in biological control of plant-pathogenic fungi and bacteria. Biol. Control 2011, 57, 1–12. [Google Scholar] [CrossRef]
- Hasan, M.A.; Ahmed, S.; Mollah, M.M.I.; Lee, D.; Kim, Y. Variation in pathogenicity of different strains of Xenorhabdus nematophila; Differential immunosuppressive activities and secondary metabolite production. J. Invertebr. Pathol. 2019, 166, 107221. [Google Scholar] [CrossRef] [PubMed]
- Griffin, C.T.; Boemare, N.E.; Lewis, E.E. ‘Biology and behaviour’. CABI Books. CABI International. In Nematodes as Biocontrol Agents; Greval, P., Ehlers, R.-U., Shapiro-Ilan, D., Eds.; CABI Publishing: Wallinford, Oxon, UK, 2005; pp. 47–64. [Google Scholar] [CrossRef]
- Bastidas, B.; Portillo, E.; San-Blas, E. Size does matter: The life cycle of Steinernema spp. in micro-insect hosts. J. Invertebr. Pathol. 2014, 121, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Lacey, L.A.; Unruh, T.R. Entomopathogenic nematodes for control of codling moth, Cydia pomonella (Lepidoptera: Tortricidae): Effect of nematode species, concentration, temperature, and humidity. Biol. Control 1998, 13, 190–197. [Google Scholar] [CrossRef]
- Mahar, A.N.; Jan, N.D.; Mahar, G.M.; Mahar, A.Q. Control of insects with entomopathogenic bacterium Xenorhabdus nematophila and its toxic secretions. Int. J. Agric. Biol. 2008, 10, 52–56. [Google Scholar]
- Adithya, S.; Shivaprakash, M.; Sowmya, E. Evaluation of insecticidal activity of entomopathogenic bacteria Photorhabdus and Xenorhabdus against shoot and fruit borer Earias vittella (Lepidoptera: Noctuidae) of vegetable crops. J. Èntomol. Zoöl. Stud 2020, 8, 2343–2348. [Google Scholar] [CrossRef]
- Vicente-Díez, I.; Blanco-Pérez, R.; Chelkha, M.; Puelles, M.; Pou, A.; Campos-Herrera, R. Exploring the use of entomopathogenic nematodes and the natural products derived from their symbiotic bacteria to control the grapevine moth, Lobesia botrana (Lepidoptera: Tortricidae). Insects 2021, 12, 1033. [Google Scholar] [CrossRef] [PubMed]
- Shawer, R.; Donati, I.; Cellini, A.; Spinelli, F.; Mori, N. Insecticidal Activity of Photorhabdus luminescens against Drosophila suzukii. Insects 2018, 9, 148. [Google Scholar] [CrossRef] [PubMed]
- Bode, H.B.; Müller, R. The impact of bacterial genomics on natural product research. Angew. Chem. Int. Ed. 2005, 44, 6828–6846. [Google Scholar] [CrossRef]
- da Silva, W.J.; Pilz-Júnior, H.L.; Heermann, R.; da Silva, O.S. The great potential of entomopathogenic bacteria Xenorhabdus and Photorhabdus for mosquito control: A review. Parasites Vectors 2020, 13, 376. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.H.; Aryal, S.K.; Aghai, D.T.; Casanova-Torres, Á.M.; Hillman, K.; Kozuch, M.P.; Dillman, A.R. The insect pathogenic bacterium Xenorhabdus innexi has attenuated virulence in multiple insect model hosts yet encodes a potent mosquitocidal toxin. BMC Genom. 2017, 18, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, M.; Golubeva, E.; Bowen, D.; French-Constant, R.H. A novel insecticidal toxin from Photorhabdus luminescens, toxin complex a (Tca), and its histopathological effects on the midgut of Manduca sexta. Appl. Environ. Microbiol. 1998, 64, 3036–3041. [Google Scholar] [CrossRef]
- Iqbal, E.Y.; Nahiyoon, A.A.; Dawar, S.; Fayyaz, S. Bioremedy of cotton aphid (Aphis gossypii Glov.)(Hemiptera: Aphididae) by the application of different fractions of entomopathogenic bacteria (Xenorhabdus spp.). Pak. J. Zool. 2020, 52, 875. [Google Scholar] [CrossRef]
- Noureldeen, A.; Kumar, U.; Asad, M.; Darwish, H.; Alharthi, S.; Fawzy, M.A.; Al-Barty, A.M.; Alotaibi, S.S.; Fallatah, A.; Alghamdi, A.; et al. Aphicidal activity of five plant extracts applied singly or in combination with entomopathogenic bacteria, Xenorhabdus budapestensis against rose aphid, Macrosiphum rosae (Hemiptera: Aphididae). J. King Saud Univ. Sci. 2022, 34, 102306. [Google Scholar] [CrossRef]
- Mahmood, S.; Kumar, M.; Kumari, P.; Mahapatro, G.K.; Banerjee, N.; Sarin, N.B. Novel insecticidal chitinase from the insect pathogen Xenorhabdus nematophila. Int. J. Biol. Macromol. 2020, 159, 394–401. [Google Scholar] [CrossRef]
- Liu, J.; Bai, H.; Song, P.; Nangong, Z.; Dong, Z.; Li, Z.; Wang, Q. Insecticidal Activity of Chitinases from Xenorhabdus nematophila HB310 and Its Relationship with the Toxin Complex. Toxins 2020, 14, 646. [Google Scholar] [CrossRef]
| Province | EPN-Positive Samples | EPN-Negative Samples | Total | Recovery Rate (%) |
|---|---|---|---|---|
| Develi | 4 | 26 | 30 | 13.3 |
| Yahyalı | 3 | 27 | 30 | 10.0 |
| Yeşilhisar | 5 | 35 | 40 | 14.2 |
| Total | 12 | 88 | 100 | 12 |
| Isolates | *L | *MBW | *EP | *ES | *TL | *a | *b | *c | *%D | *%E |
|---|---|---|---|---|---|---|---|---|---|---|
| Steinernema feltiae, Mean ± SE (Min–max) (n = 20) | ||||||||||
| A67 | 861 ± 54 (805–896) | 33 ± 2.6 (27–35) | 59 ± 3.8 (54–65) | 145 ± 4.1 (133–152) | 81 ± 2.9 (75–83) | 27 ± 3.3 (23–32) | 7.5 ± 1.6 (5.9–7.1) | 12.5 ± 2.2 (9.4–13.1) | 41 ± 2.3 (37–44) | 74 ± 3.2 (69–77) |
| A93 | 925 ± 61 (855–971) | 31 ± 1.9 (27–33) | 60 ± 4.1 (58–67) | 126 ± 7.3 (119–142) | 85 ± 4.5 (77–89) | 29 ± 3.1 (26–33) | 6.9 ± 0.9 (5.8–7.4) | 11.1 ± 1.5 (9.5–11.4) | 42.3 ± 2.9 (39–45) | 74 ± 3.9 (70–78) |
| Nguyen and Hunt (2007) | 879 ± 49 (766–928) | 29 ± 1.9 (26–32) | 63 ± 2.3 (59–67) | 136 ± 3.5 (130–143) | 86 ± 2.6 (81–89) | 30 ± 1.9 (27–34) | 6.4 ± 0.3 (5.8–6.8) | 10 ± 0.5 (9.4–11) | - | - |
| Heterorhabditis bacteriophora, Mean ± SE (Min–max) (n = 20) | ||||||||||
| A8 | 611 ± 8.1 (601–620) | 21 ± 1.9 (18–24) | 115 ± 8.2 (101–127) | 145 ± 9.1 (132–155) | 87.5 ± 3.7 (82–93) | 24.1 ± 1.9 (21–27) | 4.9 ± 0.9 (3.9–5.1) | 6.9 ± 0.8 (6.6–7.8) | 81 ± 3.1 (75–85) | 104 ± 4.1 (99–112) |
| A20 | 597 ± 8.5 (588–609) | 22 ± 2.5 (18–24) | 105 ± 7.3 (96–117) | 132 ± 4.9 (126–138) | 90.7 ± 4.5 (83–96) | 23.3 ± 2.7 (20–27) | 4.5 ± 0.6 (4.1–4.8) | 7.1 ± 0.9 (6.2–8.2) | 82 ± 3.5 (77–86) | 119 ± 6.5 (112–128) |
| Nguyen and Hunt (2007) | 588 (512–671) | 23 (18–31) | 103 (87–110) | 125 (100–139) | 98 (83–112) | 25 (17–30) | 4.5 (4.0–5.1) | 6.2 (5.5–7.0) | 84 (76–92) | 112 (103–130) |
| Code | Location | District | Species | Accession Number |
|---|---|---|---|---|
| A6 | 38°19′50 K 35°22′52 D | Develi | Steinernema feltiae | OM401705 |
| A8 | 38°21′02 K 35°23′11 D | Develi | Heterorhabditis bacteriophora | OM401700 |
| A9 | 38°24′18 K 35°26′57 D | Develi | Heterorhabditis bacteriophora | OM401701 |
| A20 | 38°24′18 K 35°26′45 D | Develi | Heterorhabditis bacteriophora | OM401702 |
| A52 | 38°20′12 K 35°27′29 D | Yahyalı | Heterorhabditis bacteriophora | OM401704 |
| A53 | 38°19′86 K 35°22′86 D | Yahyalı | Steinernema feltiae | OM401706 |
| A54 | 38°42′58 K 35°31′89 D | Yahyalı | Steinernema feltiae | OM401707 |
| A67 | 38°09′33 K 35°22′01 D | Yeşilhisar | Steinernema feltiae | OM401709 |
| A93 | 38°24′18 K 35°26′57 D | Yeşilhisar | Heterorhabditis bacteriophora | OM401703 |
| A94 | 38°21‘84 K 35°05‘64 D | Yeşilhisar | Steinernema feltiae | OM401708 |
| A97 | 38°09′52 K 35°21′56 D | Yeşilhisar | Steinernema feltiae | OM401710 |
| A100 | 38°20‘53 K 35°05‘59 D | Yeşilhisar | Steinernema feltiae | OM401711 |
| EPNs * | 24 h | 48 h | 72 h |
|---|---|---|---|
| (Mean ± S. Error) | |||
| S. feltiae A6 | 23.3 ± 6.6 BC a | 43.3 ± 3.3 BC | 60.0 ± 5.7 A |
| H. bacteriophora A8 | 16.6 ± 3.3 ABC | 56.6 ± 6.6 CD | 86.6 ± 8.8 ABC |
| H. bacteriophora A9 | 33.3 ± 3.3 C | 66.6 ± 3.3 D | 96.6 ± 3.3 BC |
| H. bacteriophora A20 | 13.3 ± 3.3 AB | 43.3 ± 3.3 BC | 86.6 ± 3.3 ABC |
| S. feltiae A52 | 16.6 ± 3.3 ABC | 43.3 ± 3.3 BC | 83.3 ± 3.3 ABC |
| S. feltiae A53 | 6.6 ± 3.3 AB | 26.6 ± 3.3 AB | 63.3 ± 6.6 A |
| S. feltiae A54 | 70.0 ± 5.7 D | 96.6 ± 3.3 E | 100.0 ± 0.0 C |
| S. feltiae A67 | 0.0 ± 0.0 A | 23.3 ± 3.3 A | 60.0 ± 10.0 A |
| H. bacteriophora A93 | 0.0 ± 0.0A | 26.6 ± 3.3 AB | 70.0 ± 5.7 AB |
| S. feltiae A94 | 60.0 ± 0.0 D | 73.3 ± 3.3 D | 86.6 ± 3.3 ABC |
| S. feltiae A97 | 13.3 ± 3.3 AB | 43.3 ± 3.3 BC | 86.6 ± 3.3 ABC |
| S. feltiae A100 | 16.6 ± 3.3 ABC | 43.3 ± 3.3 BC | 83.3 ± 3.3 ABC |
| Sources * | df | F-Value | p-Value |
|---|---|---|---|
| Nematodes (N) | 3 | 4.605 | 0.007 |
| Concentrations (C) | 3 | 15.478 | 0.000 |
| N*C | 9 | 0.767 | 0.647 |
| Error 1 | 48 | ||
| Exposure time (t) | 3 | 371.153 | 0.000 |
| C*t | 9 | 5.984 | 0.000 |
| N*t | 9 | 3.987 | 0.000 |
| N*C*t | 27 | 1.071 | 0.382 |
| Error 2 | 144 |
| Concentrations | Exposure Time * (h) | Mortality Rates (%) | |||
|---|---|---|---|---|---|
| S. f. A94 | S. f. A54 | H. b. A8 | H. b. A9 | ||
| 10 IJs/larva | 24 | 15.0 ± 9.5 A a a b | 10.0 ± 5.7 Aa | 15.0 ± 5.0 Aa | 10.0 ± 5.7 Aa |
| 48 | 35.0 ± 15.0 Ba | 35.0 ± 5.0 Ba | 30.0 ± 5.7 Ba | 30.0 ± 5.7 Ba | |
| 72 | 45.0 ± 5.0 Ba | 40.0 ± 5.7 Ba | 50.0 ± 5.7 Ca | 40.0 ± 8.1 Ba | |
| 96 | 65.0 ± 12.5 Ca | 50 ± 5.7 Ba | 60.0 ± 8.1 Ca | 50.0 ± 5.7 Ba | |
| 25 IJs/larva | 24 | 50.0 ± 12.9 Ab | 35.0 ± 5.0 Aa | 55.0 ± 9.5 Ab | 50.0 ± 5.7 Ab |
| 48 | 60.0 ± 14.1 Aa | 55.0 ± 9.5 Ba | 75.0 ± 9.5 Bb | 65.0 ± 9.5 Ba | |
| 72 | 85.0 ± 9.5 Bb | 60.0 ± 5.7 Ba | 80.0 ± 8.1 Bb | 75.0 ± 9.5 Bb | |
| 96 | 90.0 ± 10.0 Bb | 70.0 ± 5.7 Ba | 85.0 ± 9.5 Bb | 85.0 ± 9.5 Bb | |
| 50 IJs/larva | 24 | 70.0 ± 10.0 Aa | 70.0 ± 5.7 Aa | 90.0 ± 10.0 Ab | 90.0 ± 10.0 Ab |
| 48 | 80.0 ± 8.1 Aa | 80.0 ± 8.1 Aa | 95.0 ± 5.0 Ab | 100.0 ± 0.0 Ab | |
| 72 | 90.0 ± 5.7 ABa | 85.0 ± 9.5 Aa | 100.0 ± 0.0 Aa | 100.0 ± 0.0 Aa | |
| 96 | 95.0 ± 5.0 Ba | 90.0 ± 5.7 Aa | 100.0 ± 0.0 Aa | 100.0 ± 0.0 Aa | |
| 100 IJs/larva | 24 | 80.0 ± 8.1 Aa | 95.0 ± 5.0 Ab | 100.0 ± 0.0 Ab | 95.0 ± 5.0 Ab |
| 48 | 90.0 ± 5.7 Aba | 100.0 ± 0.0 Aa | 100.0 ± 0.0 Aa | 100.0 ± 0.0 Aa | |
| 72 | 95.0 ± 5.0 Aba | 100.0 ± 0.0 Aa | 100.0 ± 0.0 Aa | 100.0 ± 0.0 Aa | |
| 96 | 100.0 ± 0.0 Ba | 100.0 ± 0.0 Aa | 100.0 ± 0.0 Aa | 100.0 ± 0.0 Aa | |
| Sources * | df | F-Value | p-Value |
|---|---|---|---|
| Supernatant (S) | 3 | 30.727 | 0.000 |
| Error 1 | 12 | ||
| Exposure Time (t) | 2 | 144.907 | 0.000 |
| S*t | 6 | 6.860 | 0.000 |
| Error 2 | 24 |
| Symbiotic Bacteria | Hours (h) after Treatment | ||
|---|---|---|---|
| 24 h | 48 h | 72 h | |
| X. b. A54 | 25.0 ± 2.5 A a a b | 40.0 ± 2.5 Ab | 55.0 ± 5.0 Bc |
| X. b. A94 | 12.5 ± 3.1 Ba | 50.0 ± 4.1 Ab | 62.5 ± 4.1 Bb |
| P. l. A8 | 10.0 ± 3.8 Ba | 30.0 ± 2.5 Ab | 40.0 ± 2.5 Ab |
| P. l. A9 | 5.0 ± 2.5 Ba | 15.0 ± 3.8 Bab | 25.0 ± 3.8 Ab |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gümüşsoy, A.; Yüksel, E.; Özer, G.; İmren, M.; Canhilal, R.; Amer, M.; Dababat, A.A. Identification and Biocontrol Potential of Entomopathogenic Nematodes and Their Endosymbiotic Bacteria in Apple Orchards against the Codling Moth, Cydia pomonella (L.) (Lepidoptera: Tortricidae). Insects 2022, 13, 1085. https://doi.org/10.3390/insects13121085
Gümüşsoy A, Yüksel E, Özer G, İmren M, Canhilal R, Amer M, Dababat AA. Identification and Biocontrol Potential of Entomopathogenic Nematodes and Their Endosymbiotic Bacteria in Apple Orchards against the Codling Moth, Cydia pomonella (L.) (Lepidoptera: Tortricidae). Insects. 2022; 13(12):1085. https://doi.org/10.3390/insects13121085
Chicago/Turabian StyleGümüşsoy, Asım, Ebubekir Yüksel, Göksel Özer, Mustafa İmren, Ramazan Canhilal, Mohammed Amer, and Abdelfattah A. Dababat. 2022. "Identification and Biocontrol Potential of Entomopathogenic Nematodes and Their Endosymbiotic Bacteria in Apple Orchards against the Codling Moth, Cydia pomonella (L.) (Lepidoptera: Tortricidae)" Insects 13, no. 12: 1085. https://doi.org/10.3390/insects13121085
APA StyleGümüşsoy, A., Yüksel, E., Özer, G., İmren, M., Canhilal, R., Amer, M., & Dababat, A. A. (2022). Identification and Biocontrol Potential of Entomopathogenic Nematodes and Their Endosymbiotic Bacteria in Apple Orchards against the Codling Moth, Cydia pomonella (L.) (Lepidoptera: Tortricidae). Insects, 13(12), 1085. https://doi.org/10.3390/insects13121085










