Transcriptome Analysis Revealed Genes Related to γ-Irradiation Induced Emergence Failure in Third-Instar Larvae of Bactrocera dorsalis
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Insect Rearing and Sample Preparation
2.2. RNA Isolation and Sequencing
2.3. De Novo Assembly
2.4. Expression Analysis
2.5. GO and KEGG Enrichment Analysis
2.6. Heterozygous SNP Analysis
2.7. Validation of Gene Expression by qRT-PCR
2.8. SNP Validation
3. Results
3.1. Sequencing and Transcriptome Assembly
3.2. Functional Annotation
3.3. Differentially Expressed Genes in Response to Irradiation and Pathway Enrichment Analysis of DEGs
3.4. Verification of Differentially Expressed Genes
3.5. SNP Validation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mutamiswa, R.; Nyamukondiwa, C.; Chikowore, G.; Chidawanyika, F. Overview of oriental fruit fly, Bactrocera dorsalis (Hendel) (Diptera: Tephritidae) in Africa: From invasion, bio-ecology to sustainable management. Crop Prot. 2021, 141, 105492. [Google Scholar] [CrossRef]
- Lux, S.A.; Copeland, R.S.; White, I.M.; Manrakhan, A.; Billah, M.K. A new invasive fruit fly species from the Bactrocera dorsalis (Hendel) group detected in East Africa. Int. J. Trop. Insect Sci. 2003, 23, 355–361. [Google Scholar] [CrossRef]
- Leblanc, L.; Vueti, E.T.; Drew, R.A.; Allwood, A.J. Host Plant Records for Fruit Flies (Diptera: Tephritidae: Dacini) in the Pacific Islands. Proc. Hawaiian Entomol. Soc. 2012, 44, 11–53. [Google Scholar]
- Allwood, A.J.; Chinajariyawong, A.; Kritsaneepaiboon, S.; Drew, R.A.I.; Hamacek, E.L.; Hancock, D.L.; Hengsawad, C.; Jipanin, J.C.; Jirasurat, M.; Krong, C.K.; et al. Host plant records for fruit flies (Diptera: Tephritidae) in Southeast Asia. Raffles Bull. Zool. 1999, 47, 1–92. [Google Scholar]
- Fletcher, B.S. The Biology of Dacine Fruit Flies. Annu. Rev. Entomol. 1987, 32, 115–144. [Google Scholar] [CrossRef]
- Salmah, M.; Nurul Fatihah, M.; Hailmi, M.; Norhayati, N. Growth and Development of oriental fruit fly, Bactrocera dorsalis Hendel (Diptera: Tephritidae) reared on sweet potatoes (Ipomoea batatas L.) based artificial diet. Serangga 2019, 25, 96–107. [Google Scholar]
- Ocitti, P.; Ndlela, S.; Akol, A.; Muyinza, H.; Mohamed, S. Non-chemical post-harvest disinfestation of Bactrocera dorsalis (Hendel)(Diptera: Tephritidae) in Tommy Atkins mango using hot-water immersion treatment. Afr. Entomol. 2021, 29, 238–247. [Google Scholar] [CrossRef]
- Arthur, V. Disinfestation of mangoes haden in-fested by Anastrepha fraterculus (WIED.; 1830) (Diptera, Tephritidae), with gamma radiation. Braz. J. Radiat. Sci. 2021, 9. [Google Scholar] [CrossRef]
- Anonymous. Ethylene dibromide. Fed. Regist. 1983, 48, 4263. [Google Scholar]
- Ladaniya, M. Citrus Fruit: Biology, Technology and Evaluation; Academic Press: Cambridge, MA, USA, 2010. [Google Scholar]
- Hallman, G.J. Expanding radiation quarantine treatments beyond fruit flies. Agric. For. Entomol. 2000, 2, 85–95. [Google Scholar] [CrossRef]
- Zhao, J.; Ma, J.; Wu, M.; Jiao, X.; Wang, Z.; Liang, F.; Zhan, G. Gamma radiation as a phytosanitary treatment against larvae and pupae of Bactrocera dorsalis (Diptera: Tephritidae) in guava fruits. Food Control 2017, 72, 360–366. [Google Scholar] [CrossRef]
- Follett, P.A.; Armstrong, J.W. Revised irradiation doses to control melon fly, Mediterranean fruit fly, and oriental fruit fly (Diptera: Tephritidae) and a generic dose for tephritid fruit flies. J. Econ. Entomol. 2004, 97, 1254–1262. [Google Scholar] [CrossRef] [PubMed]
- Srimartpirom, M.; Burikam, I.; Limohpasmanee, W.; Kongratarporn, T.; Thannarin, T.; Bunsiri, A.; Follett, P.A. Low-dose irradiation with modified atmosphere packaging for mango against the oriental fruit fly (Diptera: Tephritidae). J. Econ. Entomol. 2018, 111, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Dias, V.S.; Hallman, G.J.; Martínez-Barrera, O.Y.; Hurtado, N.V.; Cardoso, A.A.; Parker, A.G.; Caravantes, L.A.; Rivera, C.; Araújo, A.S.; Maxwell, F.; et al. Modified atmosphere does not reduce the efficacy of phytosanitary irradiation doses recommended for tephritid fruit flies. Insects 2020, 11, 371. [Google Scholar] [CrossRef] [PubMed]
- Follett, P.A.; Wall, M.; Bailey, W. Influence of modified atmosphere packaging on radiation tolerance in the phytosanitary pest melon fly (Diptera: Tephritidae). J. Econ. Entomol. 2013, 106, 2020–2026. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guoping, Z.; Lili, R.; Ying, S.; Qiaoling, W.; Daojian, Y.; Yuejin, W.; Tianxiu, L. Gamma irradiation as a phytosanitary treatment of Bactrocera tau (Diptera: Tephritidae) in pumpkin fruits. J. Econ. Entomol. 2015, 108, 88–94. [Google Scholar] [CrossRef]
- IIPP. List of Topics for IPPC Standards; 2020. Available online: https://www.ippc.int/en/core-activities/standards-setting/list-topics-ippc-standards/ (accessed on 15 August 2022).
- Follett, P.A. Generic radiation quarantine treatments: The next steps. J. Econ. Entomol. 2009, 102, 1399–1406. [Google Scholar] [CrossRef]
- Follett, P.A.; Weinert, E.D. Phytosanitary irradiation of fresh tropical commodities in Hawaii: Generic treatments, commercial adoption, and current issues. Radiat. Phys. Chem. 2012, 81, 1064–1067. [Google Scholar] [CrossRef]
- UAUSDoAAaPHI. Treatments for Fruits and Vegetables. Fed. Regist. 2006. Available online: https://www.federalregister.gov/documents/2006/01/27/06-746/treatments-for-fruits-and-vegetables (accessed on 15 August 2022).
- IPP. International Standards for Phytosanitary Measures (ISPM) No. 28, Phytosanitary Treatments for Regulated Pests; Food and Agricultural Organization Rome: Rome, Italy, 2009. [Google Scholar]
- Fang, Y.; Kang, F.; Zhan, G.; Ma, C.; Li, Y.; Wang, L.; Wei, Y.; Gao, X.; Li, Z.; Wang, Y. The effects of a cold disinfestation on Bactrocera dorsalis survival and navel orange quality. Insects 2019, 10, 452. [Google Scholar] [CrossRef]
- Jiang, F.; Liang, L.; Wang, J.; Zhu, S. Chromosome-level genome assembly of Bactrocera dorsalis reveals its adaptation and invasion mechanisms. Commun. Biol. 2022, 5, 25. [Google Scholar] [CrossRef]
- Chen, X.; Lei, Y.; Li, H.; Xu, L.; Yang, H.; Wang, J.; Jiang, H. CRISPR/Cas9 mutagenesis abolishes odorant-binding protein BdorOBP56f-2 and impairs the perception of methyl eugenol in Bactrocera dorsalis (Hendel). Insect Biochem. Mol. Biol. 2021, 139, 103656. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Song, Z.-W.; Zhang, Y.-P.; Li, D.-S. Ability of Spalangia endius (Hymenoptera: Pteromalidae) to Parasitize Bactrocera dorsalis (Diptera: Tephritidae) after Switching Hosts. Insects 2021, 12, 613. [Google Scholar] [CrossRef] [PubMed]
- 51026 IAE; Practice for Using the Frick Dosimetry System. ASTM International: West Conshohocken, PA, USA, 2015.
- ISO/ASTM 51707:2002; Standard Guide For Estimating Uncertainties in Dosimetry For Radiation Processing. Available online: https://standards.iteh.ai/catalog/standards/iso/f7a12209-e259-4e42-9245-9e15d7d39daa/iso-astm-51707-2002 (accessed on 15 August 2022).
- Shen, G.M.; Dou, W.; Niu, J.Z.; Jiang, H.B.; Yang, W.J.; Jia, F.X.; Hu, F.; Cong, L.; Wang, J.J. Transcriptome analysis of the oriental fruit fly (Bactrocera dorsalis). PLoS ONE 2011, 6, e29127. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef] [PubMed]
- Iseli, C.; Jongeneel, C.V.; Bucher, P. ESTScan: A program for detecting, evaluating, and reconstructing potential coding regions in EST sequences. Proc. Int. Conf. Intell. Syst. Mol. Biol. 1999, 138–148. [Google Scholar]
- Li, R.; Yu, C.; Li, Y.; Lam, T.W.; Yiu, S.M.; Kristiansen, K.; Wang, J. SOAP2: An improved ultrafast tool for short read alignment. Bioinformatics 2009, 25, 1966–1967. [Google Scholar] [CrossRef]
- Mortazavi, A.; Williams, B.A.; McCue, K.; Schaeffer, L.; Wold, B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods 2008, 5, 621–628. [Google Scholar] [CrossRef]
- Audic, S.; Claverie, J.M. The significance of digital gene expression profiles. Genome Res. 1997, 7, 986–995. [Google Scholar] [CrossRef]
- Wang, L.; Feng, Z.; Wang, X.; Wang, X.; Zhang, X. DEGseq: An R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 2010, 26, 136–138. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Bioinformatics enrichment tools: Paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009, 37, 1–13. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Fan, B.; Xie, D.; Li, Y.; Wang, X.; Qi, X.; Li, S.; Meng, Z.; Chen, X.; Peng, J.; Yang, Y.; et al. A single intronic single nucleotide polymorphism in splicing site of steroidogenic enzyme hsd17b1 is associated with phenotypic sex in oyster pompano, Trachinotus anak. Proc. R. Soc. B 2021, 288, 20212245. [Google Scholar] [CrossRef] [PubMed]
- Heather, N.W.; Hallman, G.J. Pest Management and Phytosanitary Trade Barriers; CABI: Wallingford, UK, 2008. [Google Scholar]
- Joint, F. Irradiation as a phytosanitary treatment of food and agricultural commodities. In Proceedings of A Final Research Coordination Meeting; Joint FAO/IAEA Division of Nuclear Techniques in Food and Agriculture: Vienna, Austria, 2004. [Google Scholar]
- Kiran, R.; Shenoy, K.B.; Venkatesha, M.G. Effect of gamma radiation as a post-harvest disinfestation treatment against life stages of the coffee berry borer, Hypothenemus hampei (Ferrari)(Coleoptera: Curculionidae). Int. J. Radiat. Biol. 2019, 95, 1301–1308. [Google Scholar] [CrossRef] [PubMed]
- Seal, D.R.; Tilton, E.W. Effect of gamma radiation on the metamorphic stages of Dermestes maculatus DeGeer (Coleoptera: Dermestidae). Int. J. Radiat. Appl. Instrum. Part A Appl. Radiat. Isot. 1986, 37, 531–535. [Google Scholar] [CrossRef]
- Mastrangelo, T.; Walder, J. Use of radiation and isotopes in insects. In Radioisotopes–Applications in Bio-Medical Science; IntechOpen: London, UK, 2011; pp. 67–92. [Google Scholar]
- Zheng, W.; Peng, T.; He, W.; Zhang, H. High-throughput sequencing to reveal genes involved in reproduction and development in Bactrocera dorsalis (Diptera: Tephritidae). PLoS ONE 2012, 7, e36463. [Google Scholar] [CrossRef]
- Kerkut, G.A.; Gilbert, L.I. Comprehensive Insect Physiology, Biochemistry, and Pharmacology; Pergamon Press: New York, NY, USA, 1985; pp. 431–472. [Google Scholar]
- Herman, W.S. Studies on the Adult Reproductive Diapause of the Monarch Butterfly, Danaus plexippus. Biol. Bull. 1981, 160, 89–106. [Google Scholar] [CrossRef]
- de Celis, J.; García-Bellido, A. Roles of the Notch gene in Drosophila wing morphogenesis. Mech. Dev. 1994, 46, 109–122. [Google Scholar] [CrossRef]
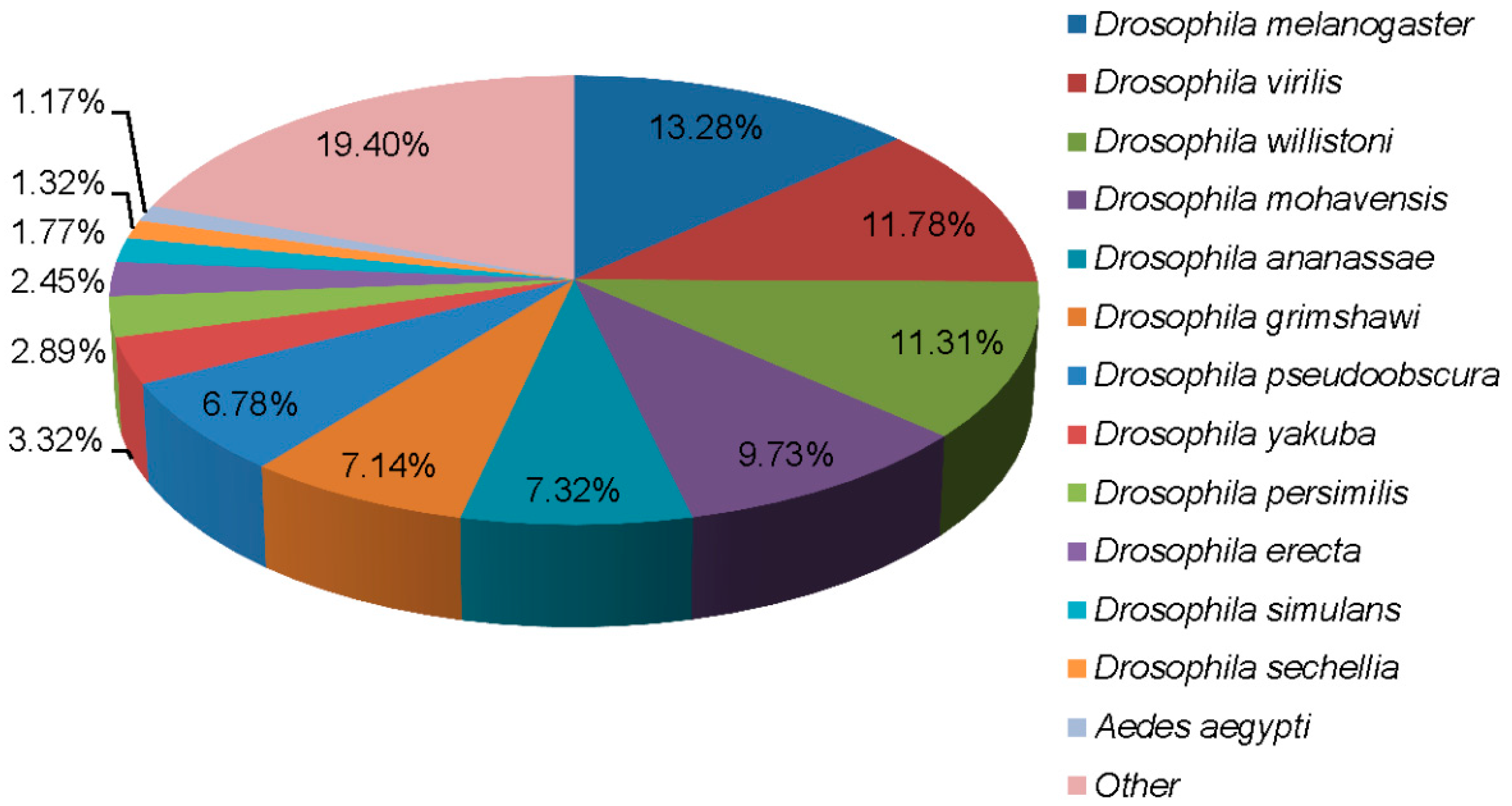



| Sample | Unigene Counts | N50 | Mean Length | All Unigene Counts | Length of All Unigene | ||||
|---|---|---|---|---|---|---|---|---|---|
| 100–500 bp | 500–1000 bp | 1000–1500 bp | 1500–2000 bp | ≥2000 bp | |||||
| CK1A | 42,012 | 7525 | 2481 | 1201 | 1419 | 668 | 466 | 54,638 | 25,475,845 |
| CK2A | 37,421 | 6482 | 2088 | 986 | 1116 | 656 | 467 | 48,093 | 22,476,031 |
| CK3A | 38,296 | 7780 | 2834 | 1370 | 1790 | 824 | 525 | 52,070 | 27,338,230 |
| R1A | 32,563 | 5640 | 1683 | 776 | 831 | 608 | 457 | 41,493 | 18,942,224 |
| R2A | 34,811 | 6803 | 2312 | 1110 | 1542 | 844 | 530 | 46,578 | 24,677,137 |
| R3A | 36,509 | 7816 | 2864 | 1630 | 2248 | 972 | 571 | 51,067 | 29,140,744 |
| All | 29,975 | 9845 | 4005 | 2365 | 4078 | 1367 | 781 | 50,268 | 39,256,184 |
| Category | Gene Ontology Term | Adjusted p-Value |
|---|---|---|
| biological process | hemolymph coagulation | 4.10× 10−25 |
| biological process | hemostasis | 4.83 × 10−22 |
| biological process | coagulation | 4.83 × 10−22 |
| biological process | regulation of body fluid levels | 1.85 × 10−21 |
| molecular function | structural constituent of cuticle | 1.30 × 10−16 |
| molecular function | structural constituent of chitin-based larval cuticle | 5.55 × 10−16 |
| cellular component | extracellular region | 1.35 × 10−15 |
| biological process | wound healing | 1.70 × 10−15 |
| molecular function | structural constituent of chitin-based cuticle | 3.22 × 10−14 |
| biological process | response to wounding | 1.42 × 10−12 |
| biological process | humoral immune response | 5.87 × 10−9 |
| biological process | defense response | 8.80 × 10−9 |
| biological process | innate immune response | 1.81 × 10−8 |
| molecular function | exopeptidase activity | 6.87 × 10−7 |
| molecular function | catechol oxidase activity | 5.89 × 10−6 |
| molecular function | L-DOPA monooxygenase activity | 5.89 × 10−6 |
| molecular function | dopamine monooxygenase activity | 5.89 × 10−6 |
| biological process | cellular lipid catabolic process | 1.60 × 10−5 |
| molecular function | peptidase activity | 1.73 × 10−5 |
| cellular component | intracellular ferritin complex | 4.74 × 10−5 |
| cellular component | ferritin complex | 4.74 × 10−5 |
| molecular function | ferrous iron binding | 6.24 × 10−5 |
| molecular function | peptidase activity, acting on L-amino acid peptides | 7.77 × 10−5 |
| molecular function | oxidoreductase activity, acting on diphenols and related substances as donors, oxygen as acceptor | 7.81 × 10−5 |
| biological process | regulation of mitochondrial translation | 1.10 × 10−4 |
| cellular component | extracellular space | 1.60 × 10−4 |
| biological process | triglyceride catabolic process | 2.40 × 10−4 |
| biological process | neutral lipid catabolic process | 2.40 × 10−4 |
| biological process | acylglycerol catabolic process | 2.40 × 10−4 |
| biological process | glycerolipid catabolic process | 2.40 × 10−4 |
| biological process | triglyceride metabolic process | 2.70 × 10−4 |
| molecular function | aminopeptidase activity | 3.50 × 10−4 |
| biological process | dopamine metabolic process | 5.60 × 10−4 |
| biological process | sesquiterpenoid metabolic process | 5.70 × 10−4 |
| biological process | juvenile hormone metabolic process | 5.70 × 10−4 |
| biological process | juvenile hormone catabolic process | 5.70 × 10−4 |
| biological process | isoprenoid catabolic process | 5.70 × 10−4 |
| biological process | sesquiterpenoid catabolic process | 5.70 × 10−4 |
| biological process | terpenoid catabolic process | 5.70 × 10−4 |
| biological process | negative regulation of translational initiation | 5.70 × 10−4 |
| molecular function | epoxide hydrolase activity | 9.00 × 10−4 |
| biological process | lipid catabolic process | 9.40 × 10−4 |
| biological process | immune response | 1.08 × 10−3 |
| biological process | terpenoid metabolic process | 1.71 × 10−3 |
| biological process | hormone catabolic process | 1.71 × 10−3 |
| biological process | catecholamine metabolic process | 1.90 × 10−3 |
| biological process | catechol-containing compound metabolic process | 1.90 × 10−3 |
| biological process | phenol-containing compound metabolic process | 1.90 × 10−3 |
| biological process | diol metabolic process | 1.90 × 10−3 |
| molecular function | ether hydrolase activity | 2.07 × 10−3 |
| molecular function | oxidoreductase activity, acting on diphenols and related substances as donors | 2.90 × 10−3 |
| molecular function | carboxypeptidase activity | 3.43 × 10−3 |
| biological process | thioester biosynthetic process | 3.68 × 10−3 |
| biological process | acyl-CoA biosynthetic process | 3.68 × 10−3 |
| biological process | neutral lipid metabolic process | 4.01 × 10−3 |
| biological process | acylglycerol metabolic process | 4.01 × 10−3 |
| biological process | immune system process | 4.49 × 10−3 |
| biological process | negative regulation of protein metabolic process | 4.73 × 10−3 |
| molecular function | oxidoreductase activity, acting on paired donors, with incorporation or reduction of molecular oxygen | 7.16 × 10−3 |
| biological process | monocarboxylic acid metabolic process | 7.34 × 10−3 |
| molecular function | oxidoreductase activity | 9.03 × 10−3 |
| biological process | single-organism metabolic process | 9.08 × 10−3 |
| molecular function | 4-aminobutyrate transaminase activity | 1.11 × 10−2 |
| molecular function | juvenile hormone epoxide hydrolase activity | 1.11 × 10−2 |
| biological process | lipid metabolic process | 1.13 × 10−2 |
| molecular function | serine-type peptidase activity | 1.27 × 10−2 |
| biological process | regulation of mitochondrion organization | 1.38 × 10−2 |
| biological process | acyl-CoA metabolic process | 1.49 × 10−2 |
| biological process | thioester metabolic process | 1.49 × 10−2 |
| molecular function | serine hydrolase activity | 1.61 × 10−2 |
| molecular function | eukaryotic initiation factor 4E binding | 1.82 × 10−2 |
| biological process | dsRNA transport | 2.16 × 10−2 |
| biological process | regulation of hormone metabolic process | 2.61 × 10−2 |
| molecular function | transferase activity, transferring hexosyl groups | 2.82 × 10−2 |
| molecular function | electron carrier activity | 3.32 × 10−2 |
| biological process | fatty acid catabolic process | 3.38 × 10−2 |
| cellular component | vacuolar proton-transporting V-type ATPase, V1 domain | 4.63 × 10−2 |
| molecular function | catalytic activity | 4.79 × 10−2 |
| Pathway ID | Pathway | Adjusted p Value | Level 1 | Level 2 |
|---|---|---|---|---|
| ko04614 | Renin-angiotensin system | 5.42 × 10−10 | Organismal Systems | Endocrine system |
| ko04974 | Protein digestion and absorption | 3.60 × 10−8 | Organismal Systems | Digestive system |
| ko04972 | Pancreatic secretion | 3.60 × 10−8 | Organismal Systems | Digestive system |
| ko05146 | Amoebiasis | 3.60 × 10−8 | Human Diseases | Infectious diseases: Parasitic |
| ko00860 | Porphyrin and chlorophyll metabolism | 9.70 × 10−8 | Metabolism | Metabolism of cofactors and vitamins |
| ko00830 | Retinol metabolism | 1.60 × 10−7 | Metabolism | Metabolism of cofactors and vitamins |
| ko05110 | Vibrio cholerae infection | 3.06 × 10−6 | Human Diseases | Infectious diseases: Bacterial |
| ko00140 | Steroid hormone biosynthesis | 5.01 × 10−6 | Metabolism | Lipid metabolism |
| ko00514 | Other types of O-glycan biosynthesis | 5.78 × 10−5 | Metabolism | Glycan biosynthesis and metabolism |
| ko00980 | Metabolism of xenobiotics by cytochrome P450 | 5.78 × 10−5 | Metabolism | Xenobiotics biodegradation and metabolism |
| ko00982 | Drug metabolism—cytochrome P450 | 6.99 × 10−5 | Metabolism | Xenobiotics biodegradation and metabolism |
| ko00740 | Riboflavin metabolism | 1.09 × 10−4 | Metabolism | Metabolism of cofactors and vitamins |
| ko04976 | Bile secretion | 1.83 × 10−3 | Organismal Systems | Digestive system |
| ko00480 | Glutathione metabolism | 1.83 × 10−3 | Metabolism | Metabolism of other amino acids |
| ko00053 | Ascorbate and aldarate metabolism | 2.27 × 10−3 | Metabolism | Carbohydrate metabolism |
| ko05164 | Influenza A | 2.36 × 10−3 | Human Diseases | Infectious diseases: Viral |
| ko05221 | Acute myeloid leukemia | 2.83 × 10−3 | Human Diseases | Cancers: Specific types |
| ko04640 | Hematopoietic cell lineage | 4.31 × 10−3 | Organismal Systems | Immune system |
| ko00130 | Ubiquinone and other terpenoid-quinone biosynthesis | 8.05 × 10−3 | Metabolism | Metabolism of cofactors and vitamins |
| ko00592 | alpha-Linolenic acid metabolism | 8.05 × 10−3 | Metabolism | Lipid metabolism |
| ko00983 | Drug metabolism—other enzymes | 8.05 × 10−3 | Metabolism | Xenobiotics biodegradation and metabolism |
| ko04512 | ECM—receptor interaction | 1.21 × 10−2 | Environmental Information Processing | Signaling molecules and interaction |
| ko00040 | Pentose and glucuronate interconversions | 1.93 × 10−2 | Metabolism | Carbohydrate metabolism |
| ko01100 | Metabolic pathways | 2.07 × 10−2 | Metabolism | Global map |
| ko00350 | Tyrosine metabolism | 2.31 × 10−2 | Metabolism | Amino acid metabolism |
| ko00030 | Pentose phosphate pathway | 2.48 × 10−2 | Metabolism | Carbohydrate metabolism |
| ko04142 | Lysosome | 2.57 × 10−2 | Cellular Processes | Transport and catabolism |
| ko05323 | Rheumatoid arthritis | 2.57 × 10−2 | Human Diseases | Immune diseases |
| ko00981 | Insect hormone biosynthesis | 2.61 × 10−2 | Metabolism | Metabolism of terpenoids and polyketides |
| ko05130 | Pathogenic Escherichia coli infection | 2.61 × 10−2 | Human Diseases | Infectious diseases: Bacterial |
| ko04977 | Vitamin digestion and absorption | 2.70 × 10−2 | Organismal Systems | Digestive system |
| ko00360 | Phenylalanine metabolism | 4.50 × 10−2 | Metabolism | Amino acid metabolism |
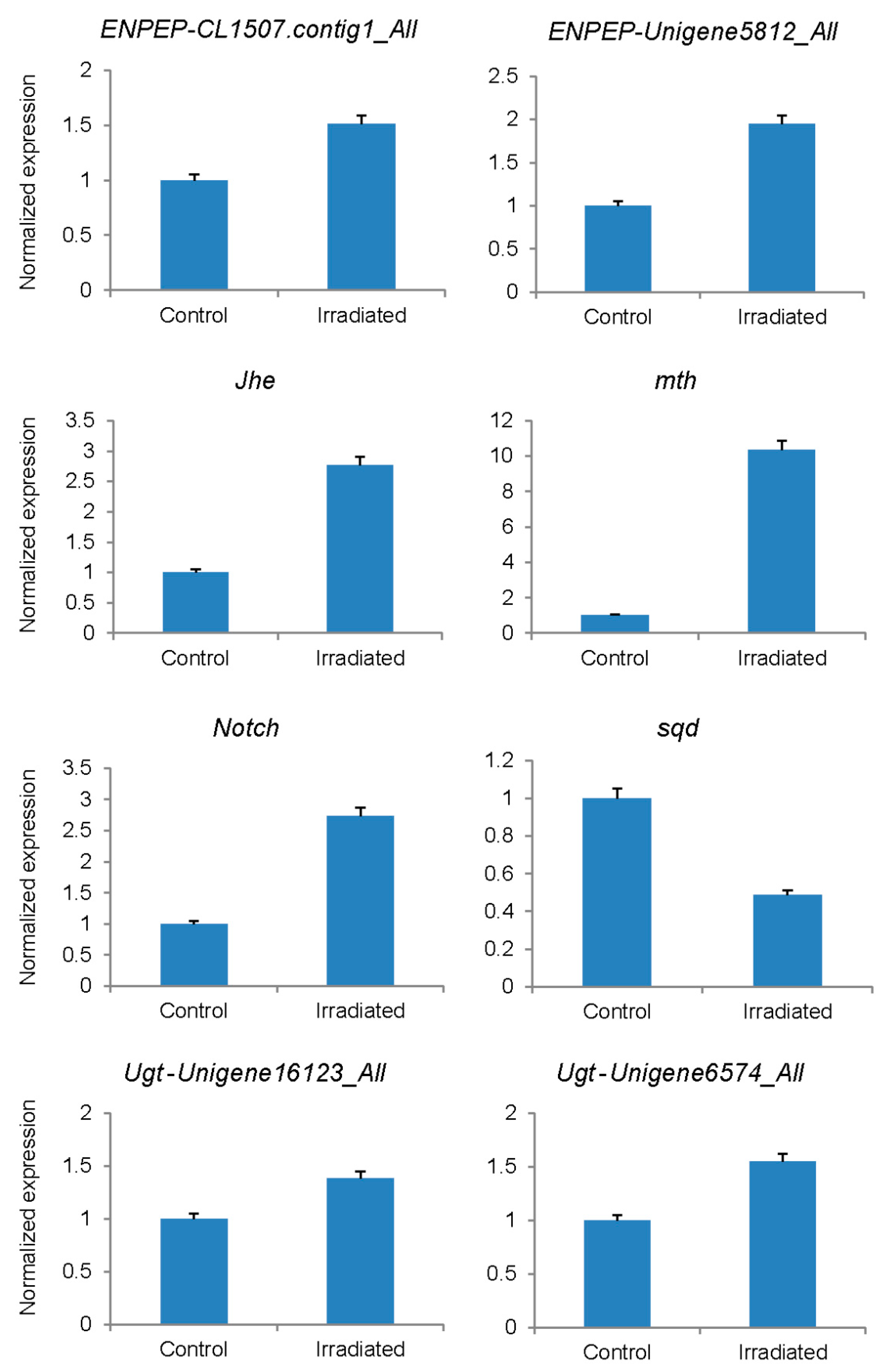
| Gene Name | Unigene | Gene Length | Fold Change by Sequencing | p Value | Fold by qPCR |
|---|---|---|---|---|---|
| ENPEP | CL1507.contig1_All | 3132 | 4.02 | 4.90 × 10−3 | 1.51 |
| ENPEP | unigene5812_All | 846 | 8.93 | 1.29 × 10−4 | 1.95 |
| Jhe | CL6471.contig1_All | 1713 | 23.44 | 3.68 × 10−2 | 2.76 |
| mth | CL6547.contig2_All | 1044 | 6.75 | 4.54 × 10−4 | 10.36 |
| Notch | CL4940.contig1_All | 9977 | 5.78 | 2.27 × 10−4 | 2.73 |
| sqd | CL816.contig2_All | 687 | −4.31 | 3.14 × 10−3 | −2.05 |
| Ugt | unigene16123_All | 1911 | 4.31 | 1.07 × 10−3 | 1.38 |
| Ugt | unigene6574_All | 1007 | 7.14 | 2.77 × 10−4 | 1.55 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, C.; Shabbir, S.; Wang, W.; Gao, Y.; Ge, C.; Lin, Q. Transcriptome Analysis Revealed Genes Related to γ-Irradiation Induced Emergence Failure in Third-Instar Larvae of Bactrocera dorsalis. Insects 2022, 13, 1017. https://doi.org/10.3390/insects13111017
Sun C, Shabbir S, Wang W, Gao Y, Ge C, Lin Q. Transcriptome Analysis Revealed Genes Related to γ-Irradiation Induced Emergence Failure in Third-Instar Larvae of Bactrocera dorsalis. Insects. 2022; 13(11):1017. https://doi.org/10.3390/insects13111017
Chicago/Turabian StyleSun, Chao, Samina Shabbir, Wenxiang Wang, Yan Gao, Cuicui Ge, and Qingsheng Lin. 2022. "Transcriptome Analysis Revealed Genes Related to γ-Irradiation Induced Emergence Failure in Third-Instar Larvae of Bactrocera dorsalis" Insects 13, no. 11: 1017. https://doi.org/10.3390/insects13111017
APA StyleSun, C., Shabbir, S., Wang, W., Gao, Y., Ge, C., & Lin, Q. (2022). Transcriptome Analysis Revealed Genes Related to γ-Irradiation Induced Emergence Failure in Third-Instar Larvae of Bactrocera dorsalis. Insects, 13(11), 1017. https://doi.org/10.3390/insects13111017




