Simple Summary
The wild scale insect Dactylopius opuntiae is one of the major insect pests of prickly pear Opuntia ficus-indica (L.) in Morocco. The present study investigated the insecticidal potential of the aqueous and hydroalcoholic extracts of Atriplex halimus (leaves), Salvia rosmarinus (leaves), and Cuminum cyminum (seeds) to control nymphs and adult females of D. opuntiae under laboratory and greenhouse conditions. Among the tested samples, the aqueous extract of A. halimus showed the highest activity on nymphs and adult females of D. opuntiae in the laboratory and when combined with black soap under greenhouse conditions. The difference in toxicity of the plant species in the study was correlated with their saponin content. These results provide evidence that the aqueous extract of A. halimus leaves could be incorporated into the management of the wild scale insect as an alternative to chemical insecticides.
Abstract
The wild cochineal Dactylopius opuntiae (Hemiptera: Dactylopiidae) is one of the major insect pests of the prickly pear Opuntia ficus-indica (L.) in Morocco, a well-known fruit and vegetable crop of arid and semi-arid regions around the world. The present study investigated the insecticidal potential of six extracts (three aqueous and three hydroalcoholic (MeOH/H2O, 20/80 (v/v)) from Atriplex halimus (leaves), Salvia rosmarinus (leaves) and Cuminum cyminum (seeds) to control nymphs and adult females of D. opuntiae under laboratory and greenhouse conditions. Out of the tested samples, A. halimus aqueous extract showed the highest activity, inducing mortality rates of 67.04% (after 4 days) and 85% (after 8 days) on nymphs and adult females of D. opuntiae, respectively, at a concentration of 5% under laboratory conditions. It also showed the highest mortality rate of nymphs with 100% (4 days after application) and 83.75% of adult females (7 days after the second application) at a concentration of 5% when combined with black soap at 10 g/L under greenhouse conditions. The difference in the toxicity of plant species of the study was correlated with their saponin content. A total of 36 of these triterpene glucosides were suggested after a comprehensive LC-MSn profiling of the most active extract, A. halimus, in addition to phytoecdysones and glycosylated phenolic acids and flavonoids. These findings provided evidence that the aqueous leaf extract of A. halimus could be incorporated in the management of the wild cochineal as an alternative to chemical insecticides.
1. Introduction
The Cactus pear, Opuntia ficus-indica (L.) Mill. (Caryophyllales: Cactaceae), is native to Mexico’s arid and semi-arid regions [1]. It was introduced to Morocco in 1770 as a good crop for years of low levels of rainfall, and for its multiple uses and applications [2]. The Cactus pear is widely represented in the Moroccan rural landscape because it requires low maintenance, and it is a natural boundary of crop fields. The cactus has experienced a resurgence of interest in several countries during the last decade because of its ecological, environmental, and socio-economic role. In most Mediterranean countries, cactus cultivation has become an intensive crop [3]. In Morocco, prickly pear plantations have been strongly encouraged by the Green Morocco Plan strategy (PMV-2008) as an alternative crop in less economically and culturally advantaged regions. The area of cacti has increased from 45,000 ha in the early 1990s to more than 150,000 ha in 2017 [4]. Several companies and cooperatives have been created to process the prickly pear, producing various products and by-products for different industrial, medicinal, pharmaceutical, and cosmetic sectors. This crop has suffered from the attack of the wild cochineal Dactylopius opuntiae (Cockerell) (Hemiptera: Dactylopiidae) in many countries, especially in the Mediterranean basin (Spain, Portugal, Morocco, Tunisia, Egypt, Israel, Lebanon, Turkey, and Greece), where it has become a serious pest of prickly pear crops [5]. This wild cochineal was first detected in the Sidi Bennour area, 70 km from El Jadida (western central Morocco) in September 2014 [6]. Due to the Mediterranean climate, D. opuntiae has spread rapidly, causing severe damage in most of the cactus-growing areas of Morocco, where farmers and cooperatives lost thousands of hectares of cactus, causing substantial socio-economic and environmental losses [6,7]. The females of this insect develop a white waxy layer that protects them from predators and parasitoids and provides them with perfect humidity; when they are crushed, they produce carminic acid, a natural dye used in the food, textile, and cosmetic industries [8]. Nymphs and adult females of D. opuntiae feed on plants by sucking sap from the cladode, leading to the destruction of the plant, and eventually killing it in the case of severe infestation.
In Morocco, significant progress has been made in developing integrated pest management (IPM) options to control this devastating insect [4]. Morocco’s National Office of Food Safety (ONSSA) has approved several pesticides to protect cactus crops and limit new infestations, including chlorpyrifos, pyriproxyfen, and mineral oils [9]. In addition to the resistance problem, these insecticides can severely affect human health and contaminate the environment, mainly plants and soil, and negatively impact natural enemies [6]. Therefore, the use of alternative approaches to control this pest (host plant resistance, natural enemies, plant extracts, vegetable and essential oils, and mycoinsecticides) has become a priority in the research field, as it is economically feasible and environmentally friendly [2].
Different botanical extracts are used as eco-friendly insecticides to control and manage different crop pests and plant diseases, due to the secondary compounds they contain, which have insecticidal properties against different insect pests [10]. In this context, Vigueras et al., (2009) [11] evaluated the efficacy of different plant extracts against D. opuntiae under laboratory conditions. They reported a high insecticidal effect of Chenopodium ambrosioides L., Mentha piperita L, Mentha viridis L., Tagetes erecta L., and Tagetes florida L. against second instars of D. opuntiae with 82, 92, 95, 98, and 99% of mortality at 72 h after treatments, respectively. Recently, Ramdani et al., (2021) [4] found that Capsicum annuum L. fruit extract at 200 g/L in combination with black soap at 60 g/L could be used as one IPM component to control D. opuntiae. Goncalves Diniz et al., (2020) [12] evaluated the effectiveness of the combinations of Fusarium catingaense L. isolates with extracts of Nicotiana tabacum L. and the mortality of the wild cochineal, and recorded 98.7% mortality in the greenhouse using 10% of aqueous extract of N. tabacum (w/v).
In this study, we selected three aromatic and medicinal plants, namely Atriplex halimus L. (leaves), Salvia rosmarinus Spenn. (leaves), and Cuminum cyminum L. (seeds), that grow or are cultivated in the region of Rhamna for their socio-economic importance for the habitats and their richness in different types and classes of secondary metabolites that can have a toxic impact on insect pests and scale insects. The major phytochemicals in R. officinalis and C. cyminum are phenolic metabolites, which are categorized into phenolic acids, flavonoids, and non-flavonoids [13,14,15,16], which include the major acids that characterize S. rosmarinus (syn. R. officinalis), rosmarinic, chlorogenic, and caffeic acids [13,14] and those of C. cyminum, chlorogenic, neochlorogenic, salicylic, cinnamic, and coumaric acids and coumarins [15,16]. However, the presence of these constituents depends on environmental conditions and extraction methods [17]. Likewise, in flavonoids, phenolic acids, and saponins, especially syringetin derivatives and atriplexoside A and B are the main constituent of A. halimus [18,19,20]. In this context, the objective of this work was to evaluate the toxicity effect of the selected botanical extracts applied alone or in combination with detergent as alternative biopesticides to control nymphs and adult females of D. opuntiae under laboratory and greenhouse conditions.
2. Material and Methods
2.1. Collection and Preparation of Plant Extracts
Atriplex halimus (Leaves), S. rosmarinus Spenn (leaves), and C. cyminum (seeds) were collected in Benguerir-Rhamna, Morocco (32°14′34.82″ N, 7°57′34.54″ W, Altitude 449 m). The materials were sorted, dried in a well-ventilated and dark place at room temperature, and ground using a laboratory mill (Retsch GM 200). The ground material was extracted by maceration at 50 °C for 30 min in 20/80 (v/v) MeOH/H2O and 100% water. Next, the extractive solutions were filtered twice on Whatman filter sheets, evaporated under reduced pressure till dryness and then lyophilized (Labconco FreeZone Legacy 2.5 L) to afford respective crude extracts. The extraction yields were 26.94, 14.89, and 15.92% (for 100 g of dry plant) for the aqueous extracts and 24.58, 10.12, and 11.36% (for 100 g of dry plant) for the 20/80 (v/v) MeOH/H2O extracts of A. halimus, S. rosmarinus, and C. cyminum, respectively.
2.2. Determination of Secondary Metabolites
2.2.1. Total Polyphenol
The total phenol contents (TPC) of the extracts were estimated following the Folin–Ciocalteu (FC) method with slight modification [21,22]. A total of 200 µL of each extract (1 mg/mL) was mixed with 1 mL of FC reagent (Fresh diluted 1:10 with distilled water), allowed to react for 5 min at room temperature in the dark, and then 800 µL of 7.5% (w/v) of (Na2CO3) was added. After 30 min standing in the dark, the absorbance was measured at 760 nm against a blank. TPC were calculated based on the calibration curve of gallic acid and expressed as gallic acid equivalents (GAE) in micrograms per gram of the dried extract.
2.2.2. Total Flavonoids
The total flavonoid contents (TFC) of extracts were determined using the aluminum chloride (AlCl3) method [23]. A total of 1 mL of extract at 1 mg/mL in methanol was added to 0.5 mL of AlCl3 (1.2%) and 0.5 mL of potassium acetate (120 mM) and allowed to react for 30 min at room temperature. The absorbance of each reaction mixture was measured at 415 nm to express the TFC as micrograms of quercetin equivalent per g of dry weight extract.
2.2.3. Total Saponins
Saponins analysis was performed according to the method applied by Irigoyen et al., (2018) [24] by adding 250 µL of the tested extract (2.5 mg/mL) to 875 µL of Lieberman–Buchard reagent (16.7% acetic acid in 83.3% concentrated sulfuric acid). The absorbance of the vortexed solution was determined at 528 min after 30 min. The total saponin content of the extracts was evaluated as mg of Quillaija saponin per g of dry extract.
2.2.4. UHPLC-MS Characterizations
The chemical profiling of extracts was conducted at Rothamsted Research (Harpenden, UK) on an Ultra-High-Performance Liquid Chromatography-Mass Spectrometry (UHPLC-MS) system equipped with a Dionex UltiMate 3000 RS UHPLC system coupled to a LTQ-Orbitrap Elite mass spectrometer (Thermo Fisher Scientific, Bremen, Germany). Samples were injected (10 μL) onto a reversed-phase Hypersil GOLD C18 selectivity HPLC column (3 μm, 30 × 2.1 mm i.d. Thermo Fisher Scientific) maintained at 35 °C. The elution consisted of a gradient of water/0.1% formic acid (A) and acetonitrile/0.1% formic acid (B) (0 min, 5% B; 0.1–20 min, 5–100% B; 20–25 min, 100% B; 25–26 min, 100–5% B; 26–31 min, 5% B) using a flow rate of 0.3 mL/min. Mass spectra were collected using an LTQ Orbitrap Elite with a heated ESI source (Thermo Scientific, Bremen, Germany), acquired in negative mode with a resolution of 120,000 over m/z 50–1500. The source voltage, sheath gas, auxiliary gas, sweep gas and capillary temperature were set to 2.5 kV, 35 (arbitrary units), 10 (arbitrary units), 0.0 (arbitrary units), and 350 °C, respectively. Default values were used for other acquisition parameters. Automatic MS–MS fragmentation was performed on the top four ions using an isolation width of m/z. Ions were fragmented using high-energy C-trap dissociation with a normalized collision energy of 65 and an activation time of 0.1 ms. Data were collected and inspected using Xcalibur v. 2.2 (Thermo Fisher Scientific). UV spectra were derived from PDA in LC-MS analyzes.
2.3. Insect Rearing
Healthy young cladodes of prickly pear cactus were planted in plastic pots (27 cm in diameter by 24 cm in height) filled with a mixture of soil, sand, and peat at greenhouse temperature of 30 ± 2 °C. They were exposed to heavily infested cladodes with D. opuntiae collected from Marchouch region (33°56′10″ N 6°69′21″ W). Each infested cladode was placed between two pots of healthy ones for 20 days to allow the development of mature pest females, which were carefully selected for further bioassays.
2.4. Laboratory Bioassays
The toxicity of plant extracts against nymphs and adult females of D. opuntiae was assessed at a temperature of 26 ± 2 °C, a relative humidity of 75% and a photoperiod of 14/10 h of light/darkness. A preliminary test was performed for all extracts at 5% against adult females of D. opuntiae by direct contact application to select the most effective extracts. Further bioassays were conducted at four concentrations (0.625, 1.25, 2.5, and 5%) of each plant extract using a 1 L hand sprayer (0.5 mL of the tested extract per petri dish) in a completely randomized design with five replications per concentration for each treatment. Ten females of the same age and ten first instar nymphs of D. opuntiae were deposited on pieces of cladodes of the same size with an entomological brush and placed in glass Petri dishes (90 mm × 15 mm). Control nymphs and female cochineal were treated with water. The mortality of adult females was evaluated every 24 h within the eight days of treatments using a binocular microscope (Motic DM-143), whereas nymphs’ mortality was recorded 1, 3, 24, 48, 72, and 96 h after exposure. Dead females lost their natural color to dark brown and desiccation of their bodies, while dead nymphs showed no movement and severe color changes.
Mortality was calculated according to Abbott’s formula [25]:
2.5. Greenhouse Trials
Healthy young cladodes of O. ficus-indica were planted in pots (27 cm in diameter by 24 cm high) filled with a mixture of soil, sand, and peat at a temperature of 30 ± 2 °C. The cladodes were infested artificially using the method described above. Treatments were applied to selected cladodes at medium infestation levels (26–50%) using a modified rating scale from Silva (1991). In general, the approximate number of colonies ranged from 41 to 80 per cladode. The ratings for the infestation level were performed using a scale of 1–6 using: (1) 1 to 5 colonies, (2) 6 to 15 colonies, (3) 16 colonies up to 25% of the cladode covered by cochineal, (4) 26 to 50%, (5) 51 to 75%, (6) 76 to 100% of the cladode covered by cochineal.
Experiments were conducted in a randomized complete block design with four replications. Cladodes were sprayed beforehand with black soap at 10 g/L, which served to remove the cuticular wax and exposed the females and nymphs to tested plant extracts, and then the 5% treatments were applied twice using a 1 L hand sprayer (5 mL per cladode) at a seven day interval [4]. Cladodes treated with water alone and water mixed with black soap served as controls. Nymphs’ mortality was recorded at 1, 2, 3, and 4 days after the treatment and females’ mortality was assessed 3, 5, and 7 days after the first and second application, each.
2.6. Data Analysis
Mortality percentages were transformed into angular values (arcsine √P) before the statistical analysis. Under laboratory conditions, the transformed percentages were subjected to two-way analysis of variance (ANOVA) (concentration and extract source). Under greenhouse conditions, the transformed percentages were subjected to one-way analysis of variance. Lethal concentration for 50% and 90% mortality (LC50 and LC90, respectively) were determined using probit analysis [26]. The means were compared by Newman–Keuls tests at p < 0.05 using XLSTAT 2021.1 by Addinsoft.
3. Results
3.1. Laboratory Bioassays
3.1.1. Insecticidal Effects of Different Plant Extracts on D. opuntiae: Preliminary Bioassays
Initially, we evaluated the insecticidal activities of the six extracts at a concentration of 5% against adult females of D. opuntiae using foliar application under lab conditions. The results revealed a significant difference in mortality of females between the six tested extracts after 5 days of application, where the aqueous extracts were more effective than the hydroalcoholic extracts irrespective of the plant species; see Table 1. The corrected mortality rates significantly increased to 89.28% for both A. halimus aqueous and hydroalcoholic extracts 8 days after application (DF = 6, p < 0.0001). However, S. rosmarinus and C. cyminum extracts possessed the lowest insecticidal activity against D. opuntiae at 5%, with a corrected mortality rate not exceeding 17.85%.

Table 1.
Mean percentage mortality ± SE of D. opuntiae adult females after exposure to six plant extracts at 5%.
3.1.2. Effect of A. halimus Extracts on D. opuntiae Nymphs and Adult Females
Based on the preliminary results, the insecticidal activities of the most toxic extracts (A. halimus, aqueous and hydroalcoholic) against the nymphs and adult females were tested using four different concentrations; see Table 2 and Table 3. The results showed a significant difference in mortality of nymphs from 24 h after treatment in a dose- and time-dependent manner; see Table 2. The ANOVA analysis showed a highly significant difference (DF = 2, p < 0.0001) in mortality of D. opuntiae nymphs 4 days after the spray. The methanolic extract of A. halimus leaves and the aqueous extract at 5% concentration recorded the highest corrected mortality rates with 77.27% and 67.04%, respectively.

Table 2.
Mean percentage mortality ± SE of D. opuntiae nymphs after exposure to aqueous and methanolic extracts of A. halimus.

Table 3.
Mean percentage mortality ± SE of D. opuntiae females after exposure to aqueous and methanolic extracts of A. halimus.
Data analysis showed a highly significant difference in the insecticidal effect of both A. halimus extracts on D. opuntiae females at different exposure times (p < 0.001) (Table 3).
The mortality of adult females increased significantly after 5 days of application (DF = 3, p < 0.0001). The increasing concentrations of the extracts significantly increased the mortality of females for different periods. At 8 days after treatment, the highest corrected mortality, 85.00% of females, was recorded for the aqueous extract of A. halimus at 5% concentration followed by hydromethanolic extract with 52.50% mortality for the same concentration (DF = 3, p < 0.0001).
Lethal concentrations LC50 and LC90 varied between the tested plant extracts. The lethal concentration values of plant extracts tested against D. opuntiae nymphs showed that A. halimus aqueous extract recorded LC50 = 3.25% and LC90 = 5.47%, and A. halimus extract 20% methanol recorded LC50 = 3.06% and LC90 = 5.20% (Table 4) four days after treatment. The lethal concentration values of plant extracts tested against D. opuntiae females indicated that the A. halimus aqueous extract (LC50 = 2.99% and LC90 = 5.29%) were more effective than A. halimus extract 20% methanol (LC50 = 3.43% and LC90 = 7.13%) seven days after treatment (Table 5).

Table 4.
Lethal concentrations (LC50 and LC90) of A. halimus extract, against D. opuntiae nymphs (at p < 0.05) after exposure to different times.

Table 5.
Lethal concentrations (LC50 and LC90) of A. halimus extracts, against D. opuntiae females (at p < 0.05) after exposure to different times.
3.2. Greenhouse Trials
According to the results mentioned above, we further evaluated the insecticidal effects of A. halimus aqueous extract against D. opuntiae nymphs and adult females under greenhouse conditions. No evidence of phytotoxicity was observed (chlorosis, change in coloration, and necrosis lesions) in treated cladodes during and after treatment. The statistical analysis showed significant differences in mortality of adult females and nymphs caused by the aqueous extract of A. halimus (5%) in combination with black soap during the different exposure periods (Table 6 and Table 7). At day 2 after treatment, A. halimus with the detergent induced high mortality of nymphs with 90.00%, which increased significantly up to 100% at day 4 after application (DF = 2, p < 0.0001) (Table 6).

Table 6.
Mean percentage ± SE of D. opuntiae nymphs after exposure to A. halimus extract and its combination with black soap under greenhouse conditions.

Table 7.
Mean percentage ± SE of D. opuntiae females after exposure to A. halimus extract using two sprays.
The ANOVA showed significant differences in mortality of adult females caused by different treatments and the control starting from day 7 after the first spray (Table 7). The mortality of adult females remained low and did not exceed 40% during the first week after application. The second application significantly increased the mortality rate of the females from 63.75% (day 10 after spraying) to 83.75%, (day 14 after application) (DF = 2, p < 0.0001), while the solution in water of black soap at 10 g/L used as a control induced a very low female mortality rate (3.50%) following the same treatment (Table 7).
3.3. Phytoconstituents Composition
3.3.1. Total Polyphenol, Flavonoid, and Saponin Contents
To correlate the observed activities with the phytoconstituents of the tested samples, we initially quantified their total phenols, flavonoids and saponins contents; see Table 8. Overall, aqueous extracts were shown to contain higher levels of phenolic compounds compared to hydroalcoholic extracts. Water extracts of S. rosmarinus featured the highest total polyphenol (170.03 ± 12.33 mg GAE/g DW) and flavonoid (33.46 ± 1.46 mg QE/g DW) contents compared to C. cyminum and A. halimus. However, both aerial part extracts of A. halimus showed high contents of saponins (24.09 ± 0.71 mg SSE/g DW) compared to the other plant extracts, which explains the foamy aspect of the extracts.

Table 8.
The total contents ± SE of polyphenols, flavonoids, and saponins in the tested extracts.
3.3.2. Phytochemical Profiling of A. halimus Extract
Annotation of the secondary metabolites of the most active aqueous extract, A. halimus, was carried out using LC-MS/MS (Figure 1). Inspection of the total ion chromatogram allowed a list of major components to be generated (Table 9) and use of the MSMS data suggested the identities of the observed metabolites. A total of 58 compounds were detected according to the following criteria: (i) the calculated mass was close to the experimental mass (error ≤ 5 ppm); (ii) fragmentation patterns (MS/MS) were in agreement with the proposed structure; (iii) Putative compounds belonged to a class of compounds previously listed to occur in the studied plant species (or genus).
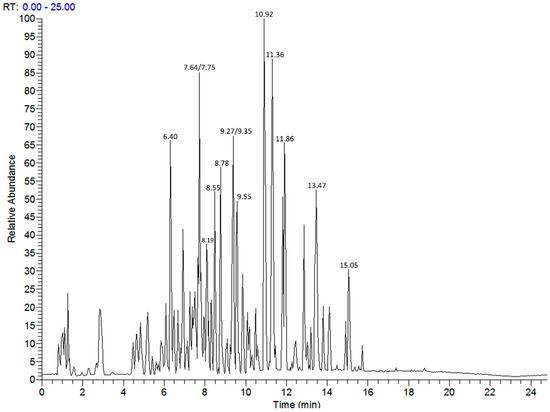
Figure 1.
LC-MS profile of A. halimus aqueous extract. Data collected in negative ion mode following reversed phase separation. Numbers relate to retention time.

Table 9.
Chemical profiling of the aqueous extract of A. halimus.
Most of them were triterpenoid saponins and hydroxylated and glycosylated compounds derived mainly from olean-12-en-28-oic acid or oleanolic acid (37 compounds), in addition to phytoecdysones (e.g., Dimorphamide A derivative). Simple organic acids such as citric acid, succinic acid, hydroxymethylglutaric acid, and phenolic acids including protocatechuic acid were also identified. Flavonoid aglycones and sulphated derivatives, including isorhamnetin and isorhamnetin sulfate, atriplexoside A, narcissin, and quercetin derivatives were also detected.
Under the collision energies employed, the fragmentation pattern of the saponins showed the molecular ion followed by low intensities of ions resulting from the successive loss of sugar moieties starting from the external unit. A loss of m/z 162 was characteristic of an ether glucosidation (rather than an ester glucosidation which releases predominantly m/z 180). Glucuronic acid ether-type conjugation was characterized by a loss of m/z 176. The mass spectrum also showed ions from the aglycone (m/z 455, 469, 471, 515, etc.) depending on the oxidation pattern of the molecule. As usual in olean-12-en-28 oic acid, ions from the retro-Diels–Alder (RDA) degradation mechanism were often evidenced with very low intensities at m/z 206 and 248 for the aglycone at m/z 455 or at m/z 207 and 262 (eventually m/z 221 and 248) for the aglycone at m/z 469. Flavonoids were also identified based on their fragmentation patterns displaying the characteristic aglycone ion in the MS2. Isorhamnetin was believed to constitute the aglycone of most of the flavonoids owing to characteristic peaks in the MS2 at m/z 152 and 163 from the RDA in ring C. The number of substitutions on the isorhamnetin backbone was determined to be identical to the number of protons lost from its corresponding mass (m/z 315) when comparing to the aglycone ion of a particular flavonoid (e.g., monodesmodic with aglycone at m/z 315 and bidesmodic with aglycone at m/z 314). Sugars were identified based on the losses of their corresponding masses (m/z 162 for glucoside, m/z 146 for rhamnose, m/z 132 for arabinose-like sugar). Finally, a compound was putatively annotated when it has been previously reported from either the studied plant or one of the species of the genus [18,20,27,28,29] and its MS characteristics matched with fragmentation patterns listed above. The other compounds were annotated by comparison of their respective MS characteristics with reported MS data in the literature [30,31,32,33].
4. Discussion
The present study demonstrated the insecticidal activity of the aqueous and hydroalcoholic extracts of A. halimus against D. opuntiae, quantified their total phenol, flavonoid and saponin contents, and annotated their secondary metabolites. Both extracts were able to kill nymphs and adult females under laboratory conditions, while only the aqueous extract preserved this activity at the greenhouse scale where the mortality rate exceeded 70%. The A. halimus aqueous extract showed substantial activity and was able to kill the nymphs and adult females by 100% and 83.75%, respectively, when tested at 5% in a combination with 10 g/L black soap. The observed activities might be attributed to the high contents of saponins that amounted to 24.09 mg SSE/g DW at A. halimus aqueous extract. These in vitro results were in line with the LC-MS studies in which we identified 58 secondary metabolites, and among them 37 suggested olean-12-en-28-oic or oleanolic acids derivatives.
A. halimus is a rich source of phenolic compounds, such as various flavonol glycosides isolated from 60% methanolic fraction, namely syringetin 3-O-beta-D-rutinoside, syringetin 3-O-beta-D-glucopyranoside, and isorhamnetin 3-O-beta-D-rutinoside or narcissin [20], which agree with our investigation. In addition, the investigation of bioactive chemical compounds conducted by Kabbash and Shoeib (2012) on aerial parts of the plant identified two flavonol glycosides, designated as atriplexoside A [3′-O-methylquercetin-4′-O-β-apiofuranoside-3-O-(6″-O-α-rhamnopyranosyl-β-glucospyranoside)] that was present in our extract and atriplexoside B [3′-O-methylquercetin-4′-O-(5″-O-β-xylopyranosyl-β-apiofuranoside)-3-O-(6″-O-α-rhamnopyranosyl-β-glucoside)], as well as two phenolic glycosides, an ecdysteroid, a megastigman, and two methoxylated flavonoid glycosides [18]. The present study’s results corroborate the findings of Donia et al., (2012) that also isolated hederagenin-3-O-β-D-glucuronopyranoside and oleanolic acid-3-O-β-D-glucuronopyranoside (calenduloside E) from Atriplex farinosa Forssk [34]. The oleanolic acid 3-O-β-D-glucopyranosyl-28-O-β-D-glucopyranoside was extracted by Kumarihamy et al., (2015) from the EtOH extract of A. canescens (Pursh) Nutt. leaves [35].
Plant secondary metabolites, such as alkaloids, tannins, saponins, lectins, terpenoids, phenolics, phenylpropanoids, and glycosides, have shown significant insecticidal activity against various crop pests including the wild cochineal, D. opuntiae [4,10,36,37,38]. Our findings are consistent with instances in the literature [39,40]. El-Gougary (1998) [39] has demonstrated the insecticidal activity of the ethanol extract of A. halimus against the cotton caterpillar Spodoptera littoralis (Boisduval) (Lepidoptera: Noctuidae) with an LD50 value of 5.6 mg/larva. The ethanolic extract and petroleum ether of A. halimus were also used in a ratio of 1:1 (w/w) with organo-phosphorus insecticide, pirimiphos-methyl (actellic) and chlorpyrifos-methyl (reldan), on the adult insect Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). The petroleum-ether extracts strongly furnished synergistic effects with chlorpyrifos-ether, while the ethanol extracts displayed synergistic effects with pirimiphos-methyl with ratios of 2.8× and l.4×, respectively [39]. Kamal et al., 2017 [40] studied the insecticidal activities of saponins and various solvent extracts of Atriplex laciniata against various pests. Crude saponins from Atriplex laciniata L. were most effective against Heterotermes indicola (Wasmann) (Isoptera: Rhinotermitidae), Rhyzopertha dominica F. (Coleoptera: Bostrichidae), and T. castaneum (Herbst), causing 90.36 ± 0.6, 80.1 ± 0.7, and 86.5 ± 0.6% mortalities, respectively. The saponin extract also showed the highest activity against Monomorium pharaonic (Linnaeus) (Hymenoptera: Formicidae), with an LD50 of 78 mg/Ml.
Likewise, the low to moderate activity encountered for the two other plants of our study was consistent with a previous study by Idris et al., (2021) [41]. Indeed, the ethanolic extract (50%) of S. rosmarinus (syn. R. officinalis) leaves showed moderate toxicity on the adult females of the wild cochineal D. opuntiae after five days of application in both laboratory and field conditions. This mortality rate reached 38.7 ± 4.0% in laboratory conditions and 25.6 ± 5.1% in field treatments after 5 days of exposure. The wild cochineals were, however, sensitive to Virginia tobacco L. extract, which induced considerable toxicity on nymphs and adult females after 5 days of application under laboratory conditions with a mortality rate of 91.7 ± 5.1% and 83.4 ± 3.1%, respectively. In addition, the mortality rates were 87.1 ± 3.8% and 78.5 ± 3.6% for nymphs and adult females, respectively, after 5 days of treatment in the field [41].
The insecticidal activity of C. cyminum extracts has been rarely studied. However, in a recent study by Khorrami et al., (2019), it was revealed that the Fe₃O₄ methanolic extract of C. cyminum was significantly more repellent against the potato insect pest Phthorimaea operculella (Zeller) [42].
Interesting studies have been published in recent years regarding the insecticidal activities of many plant extracts against D. opuntiae. Lopes et al., (2018) showed that the extracts of leaves and pods of Libidibia ferrea (Mart. ex Tul.) L. P. Queiroz were more effective against the wild cochineal, causing 81% mortality of nymph II and 97% of adult females, ten days after treatment, while Agave sisalana (Perrine) extracts controlled only adult females, causing 51–97% mortality. According to this study, the insecticidal activity of L. ferrea extracts can be explained by the toxicity presented by their secondary compounds such as flavonoids, saponins, tannins, gallic acid coumarins, steroids, and phenolic compounds [37]. Vigueras et al. (2009) found similar results by assessing the extracts of C. ambrosioides, M. piperita, M. viridis, T. erecta, and T. florida Sweet on D. opuntiae, with mortality ranging from 35% of nymphs to 98% for adult females, suggesting that terpenoids in the extracts may be responsible for this toxicity [11].
On the other hand, Ramdani et al. (2021) reported that the application of Capsicum annuum at 200 g/L in combination with black soap at 60 g/L showed excellent control of D. opuntiae females with 87.31% at 7 days after application under field conditions. However, the use of the C. annum extract alone at 200 g/L showed less mortality of females with only 18.40% [43]. The current study showed that the insecticidal effect of plant extracts against D. opuntiae nymphs and females was enhanced by applying the detergent black soap. However, further studies are necessary to establish an adequate formulation based on aqueous A. halimus extract or its active chemical compounds, compatibility with other biopesticides, and impact on natural enemies. Laboratory and greenhouse studies should be supported by field trials to confirm the effectiveness of this bioproduct against wild cochineal and determine the most effective method of the application under field conditions.
5. Conclusions
The findings of the current study indicate that an aqueous extract of A. halimus possessed a good insecticidal activity against D. opuntiae. Nymphs and adult females of D. opuntiae were more sensitive to the aqueous extract of A. halimus leaves compared to the 20% methanolic extract from the same plant and compared to the other tested plant extracts. Phytochemical determination revealed a high concentration of total saponin. Additionally, LC-MS characterization indicated the presence of valuable biologically active compounds and a large number of saponin derivatives, especially triterpenoid saponins, which could be responsible for insect mortality. However, further studies are required on individual purified components to determine those most effective for wild cochineal mortality, and in addition, to develop formulations based on the bioactive molecules with detergent to dissolve to wax as new and more effective biopesticides.
Author Contributions
I.N. performed the extraction and the in vitro activities and drafted the manuscript. K.E.F., R.L., C.R. and R.B. performed the insecticidal activities and drafted the manuscript. G.T.M.B. analyzed the chemical composition and drafted the manuscript. W.B.B. performed the in vitro activities. I.M., A.A. and A.Y. revised the manuscript. M.E.B., J.L.W. and M.S. revised the manuscript and designed and conceived the work. All authors have read and agreed to the published version of the manuscript.
Funding
The authors extend their sincere thanks to the OCP group for funding this work. Project Acronym: FP05.
Data Availability Statement
All data are included within the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- El Kharrassi, Y.; Mazri, M.A.; Benyahia, H.; Benaouda, H.; Nasser, B.; El Mzouri, E.H. Fruit and juice characteristics of 30 accessions of two cactus pear species (Opuntia ficus indica and Opuntia megacantha) from different regions of Morocco. LWT Food Sci. Technol. 2016, 65, 610–617. [Google Scholar] [CrossRef]
- Aalaoui, M.E.; Bouharroud, R.; Sbaghi, M.; Bouhssini, M.E.; Hilali, L.; Dari, N. Natural enemies associated with Dactylopius opuntiae (Cockerell) (Hemiptera: Dactylopiidae) in Morocco and their population fluctuations. Rev. Marocaine Sci. Agron. Vét. 2019, 7. [Google Scholar]
- Portillo, L.; Burgos, A.; Vigueras, A.L. Cactus pear and cochineals: Good agricultural practice and control. Acta Hortic. 2019, 1247, 199–206. [Google Scholar] [CrossRef]
- Ramdani, C.; Bouharroud, R.; Sbaghi, M.; Mesfioui, A.; El Bouhssini, M. Field and laboratory evaluations of different botanical insecticides for the control of Dactylopius opuntiae (Cockerell) on cactus pear in Morocco. Int. J. Trop. Insect Sci. 2020, 41, 1623–1632. [Google Scholar] [CrossRef]
- Mazzeo, G.; Nucifora, S.; Russo, A.; Suma, P. Dactylopius opuntiae, a new prickly pear cactus pest in the mediterranean: An overview. Entomol. Exp. Appl. 2019, 167, 59–72. [Google Scholar] [CrossRef]
- Bouharroud, R.; Amarraque, A.; Qessaoui, R. First report of the Opuntia cochineal scale Dactylopius opuntiae (Hemiptera: Dactylopiidae) in Morocco. EPPO Bull. 2016, 46, 308–310. [Google Scholar] [CrossRef]
- El Aalaoui, M.; Bouharroud, R.; Sbaghi, M.; El Bouhssini, M.; Hilali, L. Predatory potential of eleven native moroccan adult ladybird species on different stages of Dactylopius opuntiae (Cockerell) (Hemiptera: Dactylopiidae). EPPO Bull. 2019, 49, 374–379. [Google Scholar] [CrossRef]
- Giorgi, J. Management of the false carmine cochineal Dactylopius opuntiae (Cockerell): Perspective from pernambuco state, Brazil. Phytoparasitica 2018, 46, 331–340. [Google Scholar] [CrossRef]
- Index Phytosanitaire. Available online: http://eservice.onssa.gov.ma/IndPesticide.aspx (accessed on 6 September 2022).
- Sengottayan, S.-N. A review of biopesticides and their mode of action against insect pests. In Environmental Sustainability; Springer: Berlin/Heidelberg, Germany, 2015; pp. 49–63. [Google Scholar] [CrossRef]
- Vigueras, A.; Cibrián-Tovar, J.; Pelayo Ortiz, C. Use of botanicals extracts to control wild cochineal (Dactylopius opuntiae Cockerell) on cactus pear. Acta Hortic. 2009, 811, 229–234. [Google Scholar] [CrossRef]
- Gonçalves Diniz, A.; Barbosa, L.F.S.; da Santos, A.C.S.; de Oliveira, N.T.; da Costa, A.F.; Carneiro-Leão, M.P.; Tiago, P.V. Bio-insecticide effect of isolates of Fusarium caatingaense (Sordariomycetes: Hypocreales) combined to botanical extracts against Dactylopius opuntiae (Hemiptera: Dactylopiidae). Biocontrol Sci. Technol. 2020, 30, 384–395. [Google Scholar] [CrossRef]
- De Macedo, L.M.; dos Santos, É.M.; Militão, L.; Tundisi, L.L.; Ataide, J.A.; Souto, E.B.; Mazzola, P.G. Rosemary (Rosmarinus officinalis L., Syn Salvia rosmarinus Spenn.) and its topical applications: A review. Plants 2020, 9, 651. [Google Scholar] [CrossRef]
- Alvi, S.; Ahmad, P.; Ishrat, M.; Iqbal, D.; Khan, S. Secondary metabolites from rosemary (Rosmarinus officinalis L.): Structure, biochemistry and therapeutic implications against neurodegenerative diseases. In Natural Bio-Active Compounds: Chemistry, Pharmacology and Health Care Practices; Springer: Singapore, 2019; pp. 1–24. ISBN 9789811372049. [Google Scholar]
- Demir, S.; Korukluoglu, M. A comparative study about antioxidant activity and phenolic composition of cumin (Cuminum cyminum L.) and coriander (Coriandrum sativum L.). Indian J. Tradit. Knowl. IJTK 2020, 11, 383–393. [Google Scholar]
- Ramya, S.; Loganathan, T.; Chandran, M.; Priyanka, R.; Kavipriya, K.; Pushpalatha, G.G.L.; Aruna, D.; Ramanathan, L.; Jayakumararaj, R.; Saluja, V. Phytochemical screening, gcms, ftir profile of bioactive natural products in the methanolic extracts of Cuminum cyminum seeds and oil. J. Drug Deliv. Ther. 2022, 12, 110–118. [Google Scholar] [CrossRef]
- Kisiriko, M.; Anastasiadi, M.; Terry, L.A.; Yasri, A.; Beale, M.H.; Ward, J.L. Phenolics from medicinal and aromatic plants: Characterisation and potential as biostimulants and bioprotectants. Molecules 2021, 26, 6343. [Google Scholar] [CrossRef]
- Kabbash, A.; Shoeib, N. Chemical and biological investigation of some secondary metabolites in atriplex halimus growing in egypt. Nat. Prod. Commun. 2012, 7, 1934578X1200701114. [Google Scholar] [CrossRef]
- Walker, D.J.; Lutts, S. The tolerance of Atriplex halimus L. to environmental stresses. Emir. J. Food Agric. 2014, 26, 1081–1090. [Google Scholar] [CrossRef]
- El-Aasr, M.; Kabbash, A.; Abo El-Seoud, K.A.; Al-Madboly, L.A.; Ikeda, T. Antimicrobial and immunomodulatory activities of flavonol glycosides isolated from Atriplex halimus L. Herb. J. Pharm. Sci. Res. 2016, 8, 1159. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. In Methods in Enzymology; Oxidants and Antioxidants Part A; Academic Press: Cambridge, MA, USA, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Lee, J.; Renita, M.; Fioritto, R.J.; St Martin, S.K.; Schwartz, S.J.; Vodovotz, Y. Isoflavone characterization and antioxidant activity of ohio soybeans. J. Agric. Food Chem. 2004, 52, 2647–2651. [Google Scholar] [CrossRef]
- Brighente, I.M.C.; Dias, M.; Verdi, L.G.; Pizzolatti, M.G. Antioxidant activity and total phenolic content of some brazilian species. Pharm. Biol. 2007, 45, 156–161. [Google Scholar] [CrossRef]
- Irigoyen, R.M.T.; Giner, S.A. Extraction kinetics of saponins from quinoa seed (Chenopodium quinoa Willd). Int. J. Food Stud. 2018, 7, 76–88. [Google Scholar] [CrossRef]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Finney, D.J. Probit Analysis, 3rd ed.; Cambridge University Press: Cambridge, UK, 1971. [Google Scholar]
- Keckeis, K.; Sarker, S.D.; Dinan, L.N. Phytoecdysteroids from Atriplex nummularia. Fitoterapia 2000, 71, 456–458. [Google Scholar] [CrossRef]
- Karim, A.; Fatima, I.; Hussain, S.; Malik, A. Dimorphamides A-C, new polyphenolic amides from Atriplex dimorphostagia. Helv. Chim. Acta 2011, 94, 528–533. [Google Scholar] [CrossRef]
- Benhammou, N.; Bekkara, F.A.; Kadifkova Panovska, T. Antioxidant activity of methanolic extracts and some bioactive compounds of Atriplex halimus. Comptes Rendus Chim. 2009, 12, 1259–1266. [Google Scholar] [CrossRef]
- Kazzem, M.; Sun, Y.T.; Low, M.; Seto, S.W.; Chang, D.; Lee, S.; Suresh, H.; Khoo, C.S.; Bensoussan, A.; Kiat, H. Chromatographic analysis and anti-oxidative property of naoxinqing tablet, a proprietary preparation of diospyros kaki leaves. Molecules 2019, 24, 1101. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, N.; Wang, Z.; Qi, Z.; Zheng, B.; Li, P.; Liu, J. Rapid characterization of chemical constituents of platycodon grandiflorum and its adulterant Adenophora stricta by UPLC-QTOF-MS/MS. J. Mass Spectrom. 2017, 52, 643–656. [Google Scholar] [CrossRef]
- Zhumakanova, B.S.; Korona-Głowniak, I.; Skalicka-Woźniak, K.; Ludwiczuk, A.; Baj, T.; Wojtanowski, K.K.; Józefczyk, A.; Zhaparkulova, K.A.; Sakipova, Z.B.; Malm, A. Phytochemical fingerprinting and in vitro antimicrobial and antioxidant activity of the aerial parts of Thymus marschallianus Willd. And Thymus seravschanicus Klokov growing widely in southern Kazakhstan. Molecules 2021, 26, 3193. [Google Scholar] [CrossRef]
- Ismail, B.B.; Pu, Y.; Fan, L.; Dandago, M.A.; Guo, M.; Liu, D. Characterizing the phenolic constituents of baobab (Adansonia digitata) fruit shell by LC-MS/QTOF and their in vitro biological activities. Sci. Total Environ. 2019, 694, 133387. [Google Scholar] [CrossRef]
- Donia, A.E.R.; Alqasoumi, S.; Soliman, G.; Radwan, A. Secondary metabolites and antifertility potential of Atriplex farinosa Forssk. Phytopharmacology 2012, 2, 16–25. [Google Scholar]
- Kumarihamy, M.; Wang, Y.H.; Leon, F.; Wang, M.; Smesler, A.; Cutler, S.J.; Khan, I.A.; Muhammad, I. Isolation, Chemical profiling, and standardization of betaine, choline, acetylcholine, and 20-hydroxyecdysone from Atriplex species. Planta Med. 2015, 81, PX80. [Google Scholar] [CrossRef]
- Da Silva Santos, A.C.; Oliveira, R.L.S.; da Costa, A.F.; Tiago, P.V.; de Oliveira, N.T. Controlling Dactylopius opuntiae with fusarium incarnatum–equiseti species complex and extracts of Ricinus communis and Poincianella pyramidalis. J. Pest Sci. 2016, 89, 539–547. [Google Scholar] [CrossRef]
- Lopes, R.S.; Oliveira, L.G.; Costa, A.F.; Correia, M.T.S.; Lima, E.A.L.-A.; Lima, V.L.M. Efficacy of Libidibia ferrea Var. Ferrea and Agave sisalana extracts against Dactylopius opuntiae (Hemiptera: Coccoidea). J. Agric. Sci. 2018, 10, 255. [Google Scholar] [CrossRef]
- De Velez, B.A.A.; Diniz, A.G.; Barbosa, L.F.S.; da Santos, A.C.S.; da Costa, A.F.; Tiago, P.V. Potential of fusarium incarnatum-equiseti species complex isolates with Chenopodium ambrosioides and Enterolobium contortisiliquum extracts to control Dactylopius opuntiae. Int. J. Trop. Insect Sci. 2019, 39, 131–138. [Google Scholar] [CrossRef]
- El-Gougary, O.A. Insecticidal and synergistic activity of Atriplex halimus L. extracts. J. Egypt. Soc. Parasitol. 1998, 28, 191–196. [Google Scholar]
- Kamal, Z.; Ullah, F.; Ahmad, S.; Ayaz, M.; Sadiq, A.; Imran, M.; Ahmad, S.; Rahman, F.U.; Zeb, A. Saponins and solvent extracts from Atriplex laciniata L. exhibited high anthelmintic and insecticidal activities. J. Tradit. Chin. Med. 2017, 37, 599–606. [Google Scholar] [CrossRef]
- Idris, I.; Tayoub, G.; Bakri, Y. Effectiveness of some plant extracts against the different stages of the cochineal insect Dactylopius opuntiae on prickly pear in Syria. J. Agroaliment. Process. Technol. 2021, 27, 94–99. [Google Scholar]
- Khorrami, F.; Aghbash, K.O.; Soleymanzade, A.; Forouzan, M.; Ghosta, Y. Cuminum cyminum methanolic extract–Fe3O4 nanocomposite: A novel and efficient insecticide against the potato tuber moth (Lepidoptera: Gelechiidae) to protect potatoes. Acta Phytopathol. Entomol. Hung. 2019, 54, 243–251. [Google Scholar] [CrossRef]
- Ramdani, C.; El Fakhouri, K.; Sbaghi, M.; Bouharroud, R.; Boulamtat, R.; Aasfar, A.; Mesfioui, A.; El Bouhssini, M. Chemical composition and insecticidal potential of six essential oils from morocco against Dactylopius opuntiae (Cockerell) under field and laboratory conditions. Insects 2021, 12, 1007. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).