Meta-Analysis of Transcriptomes in Insects Showing Density-Dependent Polyphenism
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Curation of Public Gene Expression Data
2.2. RNA-Seq Data Retrieval, Processing, and Quantification
2.3. Quantification of Gene Expression Level
2.4. The Detection of Differentially Expressed Genes
2.5. Gene Annotation and Gene Set Enrichment Analysis
3. Results
3.1. Data Collection of Transcriptomes in Density-Dependent Polyphenism
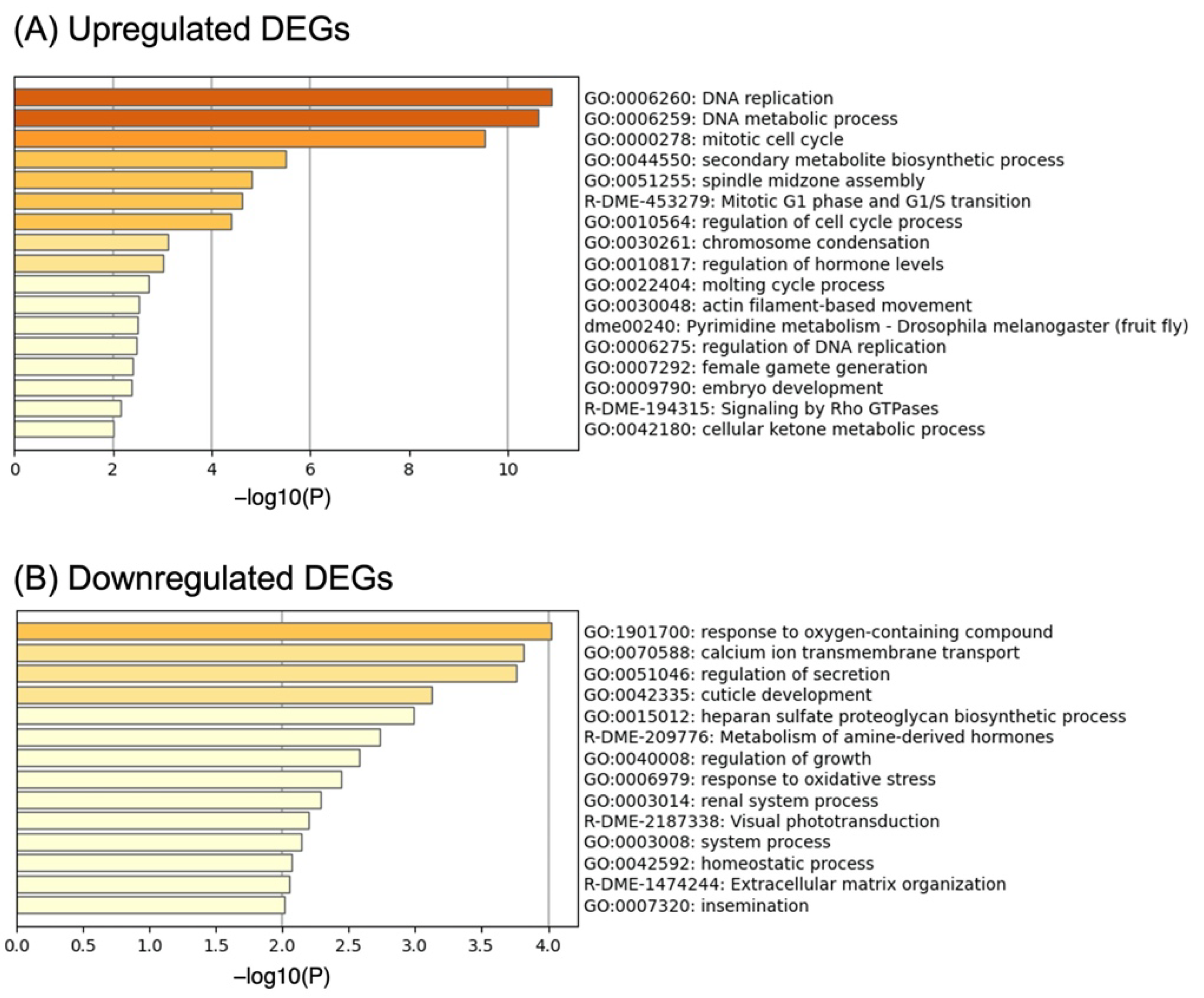
3.2. Gene Set Enrichment Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Westeberhard, M.J. Phenotypic Plasticity and the Origins of Diversity. Annu. Rev. Ecol. Syst. 1989, 20, 249–278. [Google Scholar] [CrossRef]
- West-Eberhard, M.J. Developmental Plasticity and Evolution; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Simpson, S.J.; Sword, G.A.; Lo, N. Polyphenism in Insects. Curr. Biol. 2011, 21, R738–R749. [Google Scholar] [CrossRef]
- Scott, F.; Gilbert, D. Epel Ecological Developmental Biology: Integrating Epigenetics, Medicine, And Evolution; Sinauer Associates Inc.: Sunderland, MA, USA, 2009. [Google Scholar]
- Toga, K.; Homma, Y.; Togawa, T. Control of the ecdysteroid level plays a crucial role in density-dependent metamorphosis in the giant mealworm beetle Zophobas atratus. Dev. Biol. 2021, 473, 71–79. [Google Scholar] [CrossRef]
- Ozawa, T.; Ohta, K.; Shimada, M.; Okada, K.; Okada, Y. Environmental Factors Affecting Pupation Decision in the Horned Flour Beetle Gnatocerus cornutus. Zool. Sci. 2015, 32, 183–187. [Google Scholar] [CrossRef]
- Eogawa, K.; Emiura, T. Aphid polyphenisms: Trans-generational developmental regulation through viviparity. Front. Physiol. 2014, 5, 1. [Google Scholar] [CrossRef]
- Wang, X.; Kang, L. Molecular Mechanisms of Phase Change in Locusts. Annu. Rev. Èntomol. 2014, 59, 225–244. [Google Scholar] [CrossRef]
- Breuer, M.; Hoste, B.; De Loof, A. The endocrine control of phase transition: Some new aspects. Physiol. Èntomol. 2003, 28, 3–10. [Google Scholar] [CrossRef]
- Parker, B.J.; Brisson, J.A. A Laterally Transferred Viral Gene Modifies Aphid Wing Plasticity. Curr. Biol. 2019, 29, 2098–2103. [Google Scholar] [CrossRef]
- Kang, L.; Chen, X.; Zhou, Y.; Liu, B.; Zheng, W.; Li, R.; Wang, J.; Yu, J. The analysis of large-scale gene expression correlated to the phase changes of the migratory locust. Proc. Natl. Acad. Sci. USA 2004, 101, 17611–17615. [Google Scholar] [CrossRef]
- Badisco, L.; Ott, S.R.; Rogers, S.M.; Matheson, T.; Knapen, D.; Vergauwen, L.; Verlinden, H.; Marchal, E.; Sheehy, M.R.J.; Burrows, M.; et al. Microarray-Based Transcriptomic Analysis of Differences between Long-Term Gregarious and Solitarious Desert Locusts. PLoS ONE 2011, 6, e28110. [Google Scholar] [CrossRef]
- Chen, S.; Yang, P.; Jiang, F.; Wei, Y.; Ma, Z.; Kang, L. De Novo Analysis of Transcriptome Dynamics in the Migratory Locust during the Development of Phase Traits. PLoS ONE 2010, 5, e15633. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Guo, W.; Guo, X.; Wang, X.; Kang, L. Modulation of behavioral phase changes of the migratory locust by the catecholamine metabolic pathway. Proc. Natl. Acad. Sci. USA 2011, 108, 3882–3887. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Fang, X.; Yang, P.; Jiang, X.; Jiang, F.; Zhao, D.; Li, B.; Cui, F.; Wei, J.; Ma, C.; et al. The locust genome provides insight into swarm formation and long-distance flight. Nat. Commun. 2014, 5, 2957. [Google Scholar] [CrossRef] [PubMed]
- Bono, H. Meta-Analysis of Oxidative Transcriptomes in Insects. Antioxidants 2021, 10, 345. [Google Scholar] [CrossRef]
- Ono, Y.; Bono, H. Multi-Omic Meta-Analysis of Transcriptomes and the Bibliome Uncovers Novel Hypoxia-Inducible Genes. Biomedicines 2021, 9, 582. [Google Scholar] [CrossRef]
- Suzuki, T.; Ono, Y.; Bono, H. Comparison of Oxidative and Hypoxic Stress Responsive Genes from Meta-Analysis of Public Transcriptomes. Biomedicines 2021, 9, 1830. [Google Scholar] [CrossRef]
- Rahman, M.R.; Petralia, M.C.; Ciurleo, R.; Bramanti, A.; Fagone, P.; Shahjaman, M.; Wu, L.; Sun, Y.; Turanli, B.; Arga, K.Y.; et al. Comprehensive Analysis of RNA-Seq Gene Expression Profiling of Brain Transcriptomes Reveals Novel Genes, Regulators, and Pathways in Autism Spectrum Disorder. Brain Sci. 2020, 10, 747. [Google Scholar] [CrossRef]
- Shaar-Moshe, L.; Hübner, S.; Peleg, Z. Identification of conserved drought-adaptive genes using a cross-species meta-analysis approach. BMC Plant Biol. 2015, 15, 111. [Google Scholar] [CrossRef]
- Barrett, T.; Wilhite, S.E.; Ledoux, P.; Evangelista, C.; Kim, I.F.; Tomashevsky, M.; Marshall, K.A.; Phillippy, K.H.; Sherman, P.M.; Holko, M.; et al. NCBI GEO: Archive for functional genomics data sets—Update. Nucleic Acids Res. 2013, 41, D991–D995. [Google Scholar] [CrossRef]
- Kodama, Y.; Shumway, M.; Leinonen, R. The sequence read archive: Explosive growth of sequencing data. Nucleic Acids Res. 2011, 40, D54–D56. [Google Scholar] [CrossRef]
- The NCBI SRA (Sequence Read Archive); NCBI—National Center for Biotechnology Information/NLM/NIH: Bethesda, MD, USA, 2021.
- Krueger, F. Trim Galore. Available online: Https://Www.Bioinformatics.Babraham.Ac.Uk/Projects/Trim_galore/ (accessed on 30 April 2021).
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Kim, D.; Paggi, J.M.; Park, C.; Bennett, C.; Salzberg, S.L. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat. Biotechnol. 2019, 37, 907–915. [Google Scholar] [CrossRef] [PubMed]
- Pertea, M.; Kim, D.; Pertea, G.M.; Leek, J.T.; Salzberg, S.L. Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat. Protoc. 2016, 11, 1650–1667. [Google Scholar] [CrossRef]
- Patro, R.; Duggal, G.; Love, M.I.; Irizarry, R.A.; Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 2017, 14, 417–419. [Google Scholar] [CrossRef]
- Pertea, G.; Pertea, M. GFF Utilities: GffRead and GffCompare. F1000Research 2020, 9, 304. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, B.; Pache, L.; Chang, M.; Khodabakhshi, A.H.; Tanaseichuk, O.; Benner, C.; Chanda, S.K. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 2019, 10, 1523. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, M.; Martín-Blázquez, R. RNA-Seq reveals large quantitative differences between the transcriptomes of outbreak and non-outbreak locusts. Sci. Rep. 2018, 8, 9207. [Google Scholar] [CrossRef]
- Yang, M.; Wang, Y.; Liu, Q.; Liu, Z.; Jiang, F.; Wang, H.; Guo, X.; Zhang, J.; Kang, L. A β-carotene-binding protein carrying a red pigment regulates body-color transition between green and black in locusts. Elife 2019, 8, e41362. [Google Scholar] [CrossRef]
- Foquet, B.; Castellanos, A.A.; Song, H. Comparative analysis of phenotypic plasticity sheds light on the evolution and molecular underpinnings of locust phase polyphenism. Sci. Rep. 2021, 11, 11925. [Google Scholar] [CrossRef]
- Yang, P.; Hou, L.; Wang, X.; Kang, L. Core transcriptional signatures of phase change in the migratory locust. Protein Cell 2019, 10, 883–901. [Google Scholar] [CrossRef]
- Guo, X.; Ma, Z.; Du, B.; Li, T.; Li, W.; Xu, L.; He, J.; Kang, L. Dop1 enhances conspecific olfactory attraction by inhibiting miR-9a maturation in locusts. Nat. Commun. 2018, 9, 1193. [Google Scholar] [CrossRef] [PubMed]
- Iga, M.; Kataoka, H. Recent Studies on Insect Hormone Metabolic Pathways Mediated by Cytochrome P450 Enzymes. Biol. Pharm. Bull. 2012, 35, 838–843. [Google Scholar] [CrossRef] [PubMed]
- Noh, M.Y.; Muthukrishnan, S.; Kramer, K.J.; Arakane, Y. Cuticle formation and pigmentation in beetles. Curr. Opin. Insect Sci. 2016, 17, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, K.; Winkler, B.; Pogodalla, N.; Mackensen, S.; Baldenius, M.; Garcia, L.; Naffin, E.; Rodrigues, S.; Klämbt, C. Redundant functions of the SLC5A transporters Rumpel, Bumpel, and Kumpel in ensheathing glial cells. Biol. Open 2022, 11, bio059128. [Google Scholar] [CrossRef] [PubMed]
- Mosca, T.J.; Luginbuhl, D.J.; E Wang, I.; Luo, L. Presynaptic LRP4 promotes synapse number and function of excitatory CNS neurons. Elife 2017, 6, e27347. [Google Scholar] [CrossRef]
- Molina-Obando, S.; Vargas-Fique, J.F.; Henning, M.; Gür, B.; Schladt, T.M.; Akhtar, J.; Berger, T.K.; Silies, M. ON selectivity in the Drosophila visual system is a multisynaptic process involving both glutamatergic and GABAergic inhibition. Elife 2019, 8, e49373. [Google Scholar] [CrossRef]
- Wu, X.; Hu, S.; Kang, X.; Wang, C. Synaptotagmins: Beyond Presynaptic Neurotransmitter Release. Neurosci. 2019, 26, 9–15. [Google Scholar] [CrossRef]
- Xu, N.; Wei, S.-F.; Xu, H.-J. Transcriptome Analysis of the Regulatory Mechanism of FoxO on Wing Dimorphism in the Brown Planthopper, Nilaparvata lugens (Hemiptera: Delphacidae). Insects 2021, 12, 413. [Google Scholar] [CrossRef]
- Dale, J.; Tobe, S. Biosynthesis and titre of juvenile hormone during the first gonotrophic cycle in isolated and crowded Locusta migratoria females. J. Insect Physiol. 1986, 32, 763–769. [Google Scholar] [CrossRef]
- Tanaka, S.; Harano, K.-I.; Nishide, Y.; Sugahara, R. The mechanism controlling phenotypic plasticity of body color in the desert locust: Some recent progress. Curr. Opin. Insect Sci. 2016, 17, 10–15. [Google Scholar] [CrossRef]
- Du, B.; Ding, D.; Ma, C.; Guo, W.; Kang, L. Locust density shapes energy metabolism and oxidative stress resulting in divergence of flight traits. Proc. Natl. Acad. Sci. USA 2021, 119, e2115753118. [Google Scholar] [CrossRef] [PubMed]
| Species | Number of RNA Sequencing Files (under Crowded Condition) | Number of RNA Sequencing Files (under Isolated Condition) | Tissue |
|---|---|---|---|
| Schistocerca gregaria | 1 | 1 | CNS |
| Acyrthosiphon pisum | 8 | 8 | whole |
| Schistocerca americana | 10 | 10 | head, thorax |
| Schistocerca nitens | 10 | 10 | head, thorax |
| Schistocerca piceifrons | 10 | 10 | head, thorax |
| Schistocerca serialis cubense | 10 | 10 | head, thorax |
| Locusta migratoria | 17 | 17 | brain, Integument, thoracic ganglion |
| Number of Samples with Expression Patterns Responding to the Crowded Condition | ||||
|---|---|---|---|---|
| Gene_Name | Upregulated | Downregulated | Unchanged | CI Score |
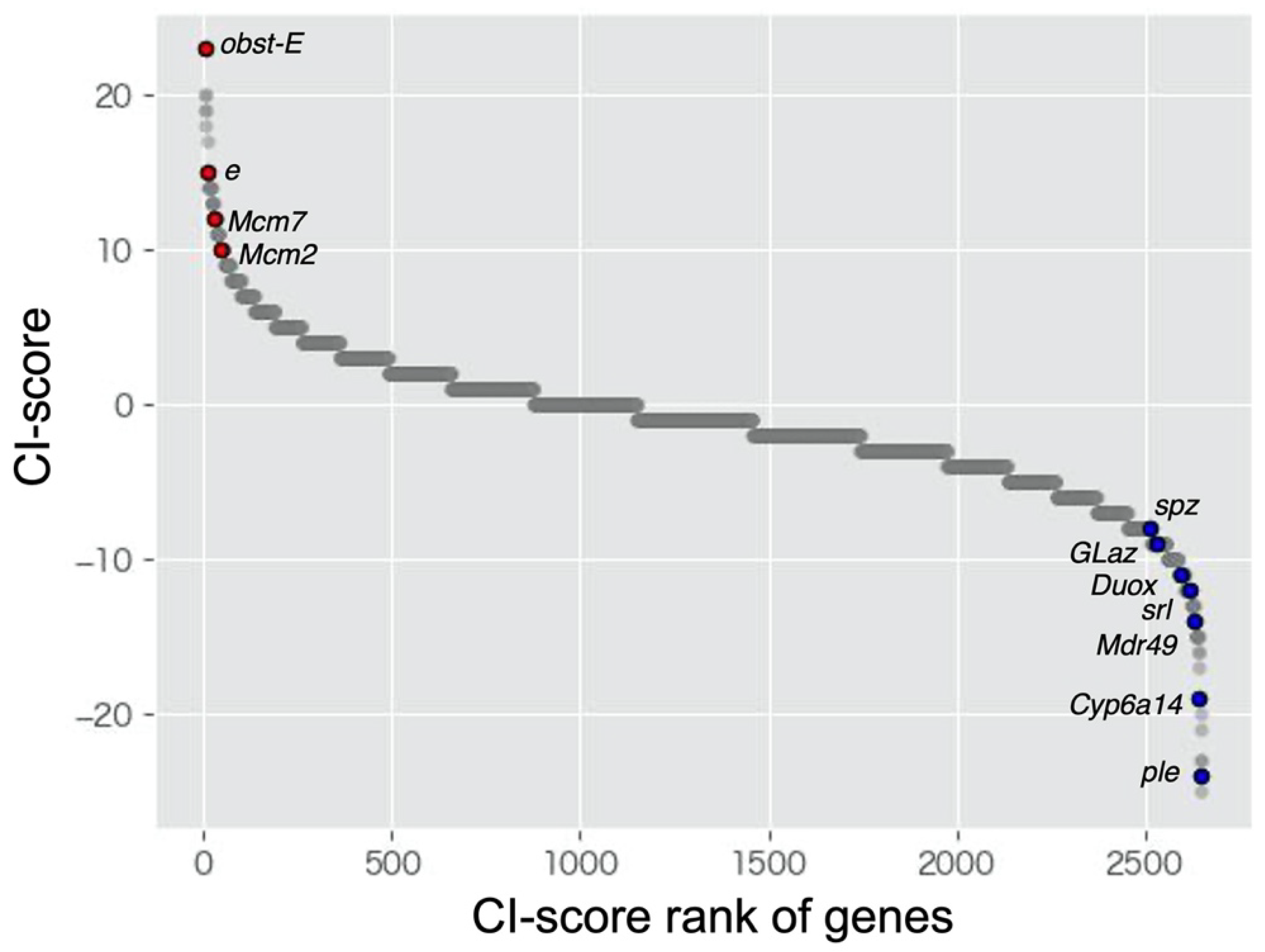
| obst-E | 31 | 8 | 27 | 23 |
| CG14803 | 23 | 3 | 40 | 20 |
| His2A:CG33835 | 30 | 10 | 26 | 20 |
| Jheh2 | 22 | 3 | 41 | 19 |
| nw | 27 | 8 | 31 | 19 |
| HDAC1 | 21 | 3 | 42 | 18 |
| Ugt36D1 | 24 | 7 | 35 | 17 |
| CG5321 | 22 | 7 | 37 | 15 |
| Cdep | 21 | 6 | 39 | 15 |
| e | 17 | 2 | 47 | 15 |
| pds5 | 19 | 4 | 43 | 15 |
| CG4572 | 21 | 7 | 38 | 14 |
| CG6765 | 16 | 2 | 48 | 14 |
| Incenp | 23 | 9 | 34 | 14 |
| Psf3 | 21 | 7 | 38 | 14 |
| ft | 21 | 7 | 38 | 14 |
| polo | 19 | 5 | 42 | 14 |
| tou | 22 | 8 | 36 | 14 |
| tum | 19 | 5 | 42 | 14 |
| CG10175 | 22 | 9 | 35 | 13 |
| CG2990 | 20 | 7 | 39 | 13 |
| CG8173 | 18 | 5 | 43 | 13 |
| DNApol-alpha60 | 22 | 9 | 35 | 13 |
| SmD3 | 18 | 5 | 43 | 13 |
| Ts | 20 | 7 | 39 | 13 |
| CG8646 | 20 | 8 | 38 | 12 |
| Number of Samples with Expression Patterns Responding to the Crowded Condition | ||||
|---|---|---|---|---|
| Gene_Name | Upregulated | Downregulated | Unchanged | CI Score |
| CG14301 | 3 | 28 | 35 | −25 |
| ple | 7 | 31 | 28 | −24 |
| CG9657 | 6 | 29 | 31 | −23 |
| Lrp4 | 3 | 26 | 37 | −23 |
| GluClalpha | 5 | 26 | 35 | −21 |
| CG34461 | 9 | 29 | 28 | −20 |
| Cyp6a14 | 9 | 28 | 29 | −19 |
| CG10211 | 10 | 27 | 29 | −17 |
| Ggt-1 | 8 | 24 | 34 | −16 |
| Syt4 | 6 | 22 | 38 | −16 |
| Cralbp | 9 | 24 | 33 | −15 |
| KaiR1D | 3 | 18 | 45 | −15 |
| RpS5a | 3 | 18 | 45 | −15 |
| Shmt | 6 | 21 | 39 | −15 |
| Tcs3 | 4 | 19 | 43 | −15 |
| tyn | 2 | 17 | 47 | −15 |
| CG13744 | 7 | 22 | 37 | −15 |
| CG15449 | 5 | 19 | 42 | −14 |
| Est-6 | 9 | 23 | 34 | −14 |
| KaiR1D | 11 | 25 | 30 | −14 |
| Mdr49 | 7 | 21 | 38 | −14 |
| dpr12 | 6 | 20 | 40 | −14 |
| CG13643 | 7 | 20 | 39 | −13 |
| CG6006 | 5 | 18 | 43 | −13 |
| CG7246 | 2 | 15 | 49 | −13 |
| Cnb | 5 | 18 | 43 | −13 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toga, K.; Yokoi, K.; Bono, H. Meta-Analysis of Transcriptomes in Insects Showing Density-Dependent Polyphenism. Insects 2022, 13, 864. https://doi.org/10.3390/insects13100864
Toga K, Yokoi K, Bono H. Meta-Analysis of Transcriptomes in Insects Showing Density-Dependent Polyphenism. Insects. 2022; 13(10):864. https://doi.org/10.3390/insects13100864
Chicago/Turabian StyleToga, Kouhei, Kakeru Yokoi, and Hidemasa Bono. 2022. "Meta-Analysis of Transcriptomes in Insects Showing Density-Dependent Polyphenism" Insects 13, no. 10: 864. https://doi.org/10.3390/insects13100864
APA StyleToga, K., Yokoi, K., & Bono, H. (2022). Meta-Analysis of Transcriptomes in Insects Showing Density-Dependent Polyphenism. Insects, 13(10), 864. https://doi.org/10.3390/insects13100864








