Effect of Horticultural Mineral Oil on Huanglongbing Transmission by Diaphorina citri Kuwayama (Hemiptera: Psyllidae) Population in a Commercial Citrus Orchard in Sarawak, Malaysia, Northern Borneo
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Sites
- A control, unsprayed,
- nC24 HMO (Ampol D-C) Tron plus: Ampol Rural, Sydney, Australia, applied at concentrations of 0.33–0.5% (v/v), mostly 0.5%.
2.2. Sampling
2.3. Incidence and Transmission of HBL Disease Relating to D. citri Population
2.3.1. Diagnostic Protocol for the Causal Agent of Huanglongbing Disease ‘Candidatus Liberibacter asiaticus’ (CLas)
2.3.2. PCR Assays
2.4. Statistical Analysis
3. Results and Discussion
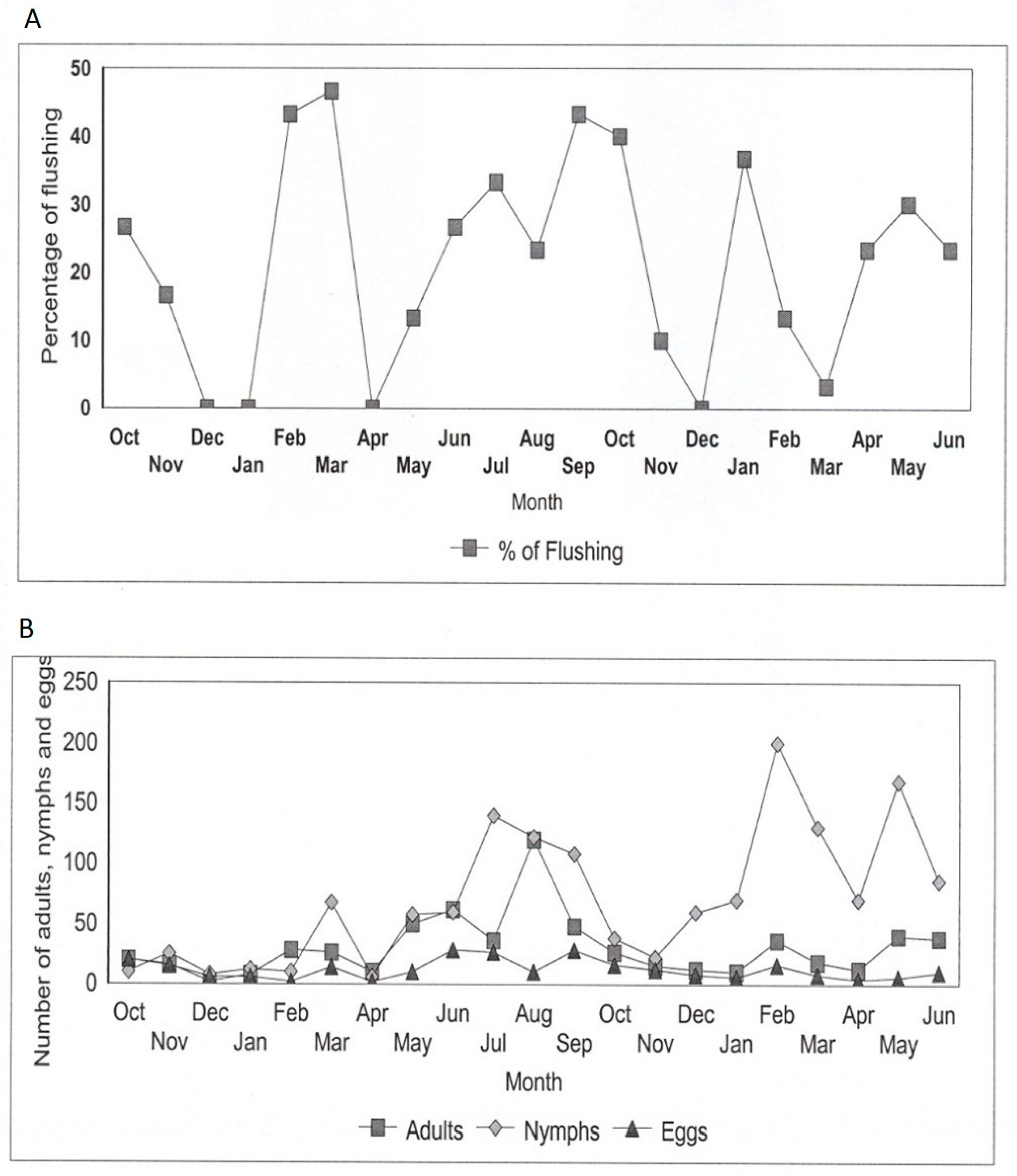
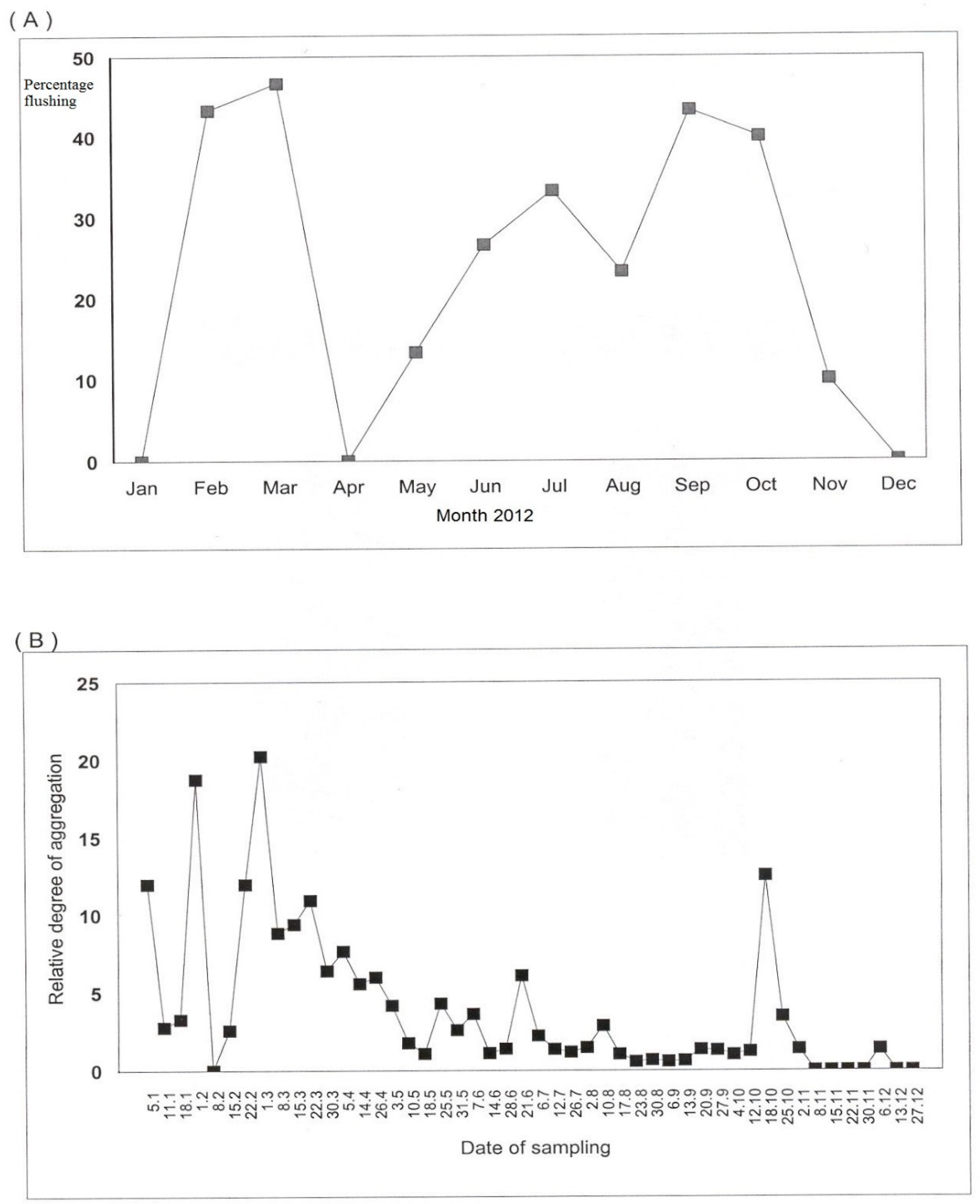
3.1. Seasonal Variation in D. citri Population Abundance Relating to Flushing
3.2. Influence of Flushing Cycle on the Spatial Dispersion of D. citri Adults
3.3. Effect of Horticultural Mineral Oil (HMO) on Oviposition, Nymphs, and Adult Mortality
3.4. Impact of Horticultural Mineral Oil (HMO) on Disease Incidence and Spread of HBL by D. citri
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aubert, B. Epidermiological aspects of the greening (Huanglungbing) disease in Asia. In Proceedings of the FAO Regional Workshop on Citrus Greening Huanglungbing Disease, Fuzhou, China, 1–13 December 1987. [Google Scholar]
- Halbert, S.E.; Manjunath, K.L. Asian citrus psyllids (Sternorrhyncha: Psyllidae) and greening disease of citrus: A literature review and assessment of risk in Florida. Florida Entomol. 2004, 87, 330–353. [Google Scholar] [CrossRef]
- Leong, S.S.; Leong, S.C.T.; Beattie, G.A.C. Dispersion pattern and sampling plan for Asian citrus psyllid, Diaphorina citri Kuwayama (Hemiptera: Psyllidae) in a citrus orchard. Serangga 2019, 24, 25–40. [Google Scholar]
- Bove, J.M. Huanglongbing: A destructive, newly-emerging, century-old disease of citrus. J. Plant Pathol. 2006, 88, 7–37. [Google Scholar] [CrossRef]
- Abdullah, T.L.; Shokrollah, H.; Sijam, K.; Abdullah, S.N.A. Control of Huanglongbing (HLB) disease with reference to its occurrence in Malaysia. Afr. J. Biotechnol. 2009, 8, 4007–4015. [Google Scholar]
- Aubert, B. Integrated activities for the control of huanglongbing-greening and its vector Diaphorina citri Kuwayama in Asia. In Proceedings of the Fourth International Asia-Pacific Conference on Citrus Rehabilitation, Chiang Mai, Thailand, 12 September 1990; Aubert, B., Tontyaporn, S., Buangsuwon, D., Eds.; NDP-FAO Press: Chiang Mai, Thailand, 1990; pp. 133–144. [Google Scholar]
- Tiwari, S.; Mann, R.S.; Rogers, M.E.; Stelinski, L.L. Insecticide resistance in field populations of Asian citrus psyllid in Florida. Pest Manag. Sci. 2011, 67, 1258–1268. [Google Scholar] [CrossRef]
- Lim, W.H.; Mohd Shamsudin, O.; Ko, W.W. Citrus greening disease in Malaysia: Status report. In Proceedings of the Fourth International Asia-Pacific Conference on Citrus Rehabilitation, Chiang Mai, Thailand, 12 September 1990; Aubert, B., Tontyaporn, S., Buangsuwon, D., Eds.; NDP-FAO Press: Chiang Mai, Thailand, 1990; pp. 100–105. [Google Scholar]
- Beattie, G.A.C.; Holford, P. Current HLB situation and industry perspective in Asia. In Proceedings of the International Research Conference on Huanglongbing, Orlando, FL, USA, 1–4 December 2008; p. 94. [Google Scholar]
- Spiegel-Roy, P.; Goldschmidt, E.E. Biology of Horticultural Crops: Biology of Citrus, 1st ed.; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Sétamou, M.; Flores, D.; French, J.V.; Hall, D.G. Dispersion patterns and sampling plans for Diaphorina citri (Hemiptera: Psyllidae) in citrus. J. Econ. Entomol. 2008, 101, 1478–1487. [Google Scholar] [CrossRef]
- Boina, D.R.; Meyer, W.L.; Onagbola, E.O.; Stelinski, L.L. Quantifying dispersal of Diaphorina citri (Hemiptera: Psyllidae) by immunomarking and potential impact of unmanaged groves on commercial citrus management. Environ. Entomol. 2009, 38, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Cifuentes-Arenas, J.C.; de Goes, A.; de Miranda, M.P.; Beattie, G.A.C.; Lopes, S.A. Citrus flush shoot ontogeny modulates biotic potential of Diaphorina citri. PLoS ONE 2018, 13, e0190563. [Google Scholar] [CrossRef]
- Chavan, V.M.; Summanwar, A.S. Population dynamics and aspects of the biology of citrus psylla, Diaphorina citri Kuw., in Maharashtra. Int. Organ. Citrus Virol. Conf. Proc. 1993, 12, 286–290. [Google Scholar]
- Ammar, E.-D.; Hall, D.G.; Shatters, R.G. Stylet morphometrics and citrus leaf vein structure in relation to feeding behavior of the asian citrus psyllid Diaphorina citri, vector of citrus huanglongbing bacterium. PLoS ONE 2013, 8, e59914. [Google Scholar] [CrossRef]
- Pluke, R.; Qureshi, J.; Stansly, P. Citrus flushing patterns, Diaphorina citri (Hemiptera: Psyllidae) populations and parasitism by Tamarixia radiata (Hymenoptera: Eulophidae) in Puerto Rico. Fla. Entomol. 2008, 91, 36–42. [Google Scholar] [CrossRef]
- Leong, S.S.; Leong, S.C.T.; Fatimah, A.; Andrew, C.B.G.; Kueh, R.J.H. Distribution patterns of Diaphorina citri Kuwayama (Hemiptera: Psyllidae) eggs, nymphs and adults in a Malaysian citrus orchard. Isr. J. Entomol. 2018, 48, 49–62. [Google Scholar]
- Karley, A.J.; Douglas, A.E.; Parker, W.E. Amino acid composition and nutritional quality of potato leaf phloem sap for aphids. J. Exp. Biol. 2002, 205, 3009–3018. [Google Scholar] [CrossRef]
- Wenninger, E.J.; Stelinski, L.L.; Hall, D.G. Roles of olfactory cues, visual cues, and mating status in orientation of Diaphorina citri Kuwayama (Hemiptera: Psyllidae) to four different host plants. Environ. Entomol. 2009, 38, 225–234. [Google Scholar] [CrossRef]
- Patt, J.M.; Sétamou, M. Responses of the asian citrus psyllid to volatiles emitted by the flushing shoots of its rutaceous host plants. Environ. Entomol. 2010, 39, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Patt, J.; Stockton, D.; Meikle, W.; Sétamou, M.; Mafra-Neto, A.; Adamczyk, J. Innate and conditioned responses to chemosensory and visual cues in asian citrus psyllid, Diaphorina citri (Hemiptera: Liviidae), vector of Huanglongbing pathogens. Insects 2014, 5, 921–941. [Google Scholar] [CrossRef]
- Yang, Y.; Beattie, G.A.C.; Spooner-Hart, R.N.; Huang, M.; Barchia, I.; Holford, P. Influences of leaf age and type, non-host volatiles, and mineral oil deposits on the incidence, distribution, and form of stylet tracks of Diaphorina citri. Entomol. Exp. Appl. 2013, 147, 33–49. [Google Scholar] [CrossRef]
- Husain, M.A.; Nath, D. The citrus psylla (Diaphorina citri, Kuw.) (Psyllidae: Homoptera). Mem Dep Agric. India 1927, 10, 1–27. [Google Scholar]
- Lin, S.; Ke, Y.; Tao, C.C. Bionomics observation and integrated control of citrus psylla, Diaphorina citri Kuwayama. J. Hortic. Soc. China 1973, 19, 234–242. [Google Scholar]
- Chen, X.; Liao, C. Studies on the biology of Diaphorina citri and its relationship with HLB. China Citrus. 1982, 4, 4–17. [Google Scholar]
- Xu, C.F.; Xia, Y.H.; Ke, C. Study on the biology and control of the citrus psylla. Acta Phytophylacica Sin. 1994, 21, 53–56. [Google Scholar]
- Azam, M.; Jiang, Q.; Zhang, B.; Xu, C.; Chen, K. Citrus leaf volatiles as affected by developmental stage and genetic type. Int. J. Mol. Sci. 2013, 14, 17744–17766. [Google Scholar] [CrossRef] [PubMed]
- Leong, S.C.T.; Abang, F.; Beattie, G.A.C.; Kueh, J.H.; Wong, S.K. Seasonal population dynamics of the Asian citrus psyllid, Diaphorina citri Kuwayama in Sarawak. Am. J. Agric. Biol. Sci. 2011, 6, 536–543. [Google Scholar]
- Hall, D.G.; Wenninger, E.J.; Hentz, M.G. Temperature studies with the Asian Citrus Psyllid, Diaphorina citri: Cold hardiness and temperature thresholds for oviposition. J. Insect Sci. 2011, 11, 83. [Google Scholar] [CrossRef] [PubMed]
- Milosavljevic, I.; McCalla, K.A.; Morgan, D.J.W. The Effects of Constant and Fluctuating Temperatures on Development of Diaphorina citri (Hemiptera: Liviidae), the Asian Citrus Psyllid. J. Econ. Entomol. 2020, 113, 633–645. [Google Scholar] [CrossRef]
- Carmo-Sousa, M.; Garcia, R.B.; Wulff, N.A.; Fereres, A.; Miranda, M.P. Drench Application of Systemic Insecticides Disrupts Probing Behavior of Diaphorina citri (Hemiptera: Liviidae) and Inoculation of Candidatus Liberibacter asiaticus. Insects 2020, 11, 314. [Google Scholar] [CrossRef]
- Kistner, E.J.; Amrich, R.; Castillo, M.; Strode, V.; Hoddle, M.S. Phenology of Asian citrus psyllid (Hemiptera: Liviidae), with special reference to biological control by Tamarixia radiata, in the residential landscape of southern California. J. Econ. Entomol. 2016, 109, 1047–1057. [Google Scholar] [CrossRef]
- Milosavljevic, V.; Schall, K.; Hoddle, C.; Morgan, D.; Hoddle, M.S. Biocontrol program targets Asian citrus psyllid in California’s urban areas. Calif. Agric. 2017, 71, 169–177. [Google Scholar] [CrossRef]
- Teo, C.H.; Voon, B.H.; Leong, C.T. Integrated management of greening disease (Huanglongbing) in Sarawak. In Proceedings of the Plant Resource Management Conference, Kuching, Malaysia, 23–25 October 2000; pp. 29–31. [Google Scholar]
- Ke, C. The present status of Citrus Huanglungbing and its control in China. In Proceedings of the 6th Asia Pacific UNDP-FAO Conference on Integrated Citrus Health Management, Kuala Lumpur, Malaysia, 23–25 June 1991; Ke, C., Shamsudin, M.O., Eds.; FAO-UNDP-MARDI: Kuala Lumpur, Malaysia, 1991; pp. 65–76. [Google Scholar]
- Yang, Y.P.; Huang, M.D.; Beattie, G.A.C.; Xia, Y.L.; Ouyang, G.C.; Xiong, J.J. Distribution, biology, ecology and control of the psyllid Diaphorina citri Kuwayama, a major pest of citrus: A status report for China. Int. J. Pest. Manag. 2006, 52, 343–352. [Google Scholar] [CrossRef]
- Boina, D.R.; Rogers, M.E.; Wang, N.; Stelinski, L.L. Effect of pyriproxyfen, a juvenile hormone mimic, on egg hatch, nymph development, adult emergence and reproduction of the Asian citrus psyllid, Diaphorina citri Kuwayama. Pest. Manag. Sci. 2010, 66, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Mao, R. Study on control effectiveness of huanglongbing based on eradication of Asian citrus psyllid, Diaphorina citri, surrounding citrus orchard. In Proceedings of the 2016 International Congress of Entomology, Orlando, FL, USA, 25–30 September 2016; p. 123. [Google Scholar]
- Aubert, B.; Quilici, S. Biological control of the African and Asian citrus psyllids (Homoptera: Psylloidea), through eulophid and encyrtid parasites (Hymenoptera: Chalcidoidea) in Reunion Island. In Proceedings of the Ninth Conference of the International Organization of Citrus Virologists, Puerto Iguaçu, Misiones, Argentina, 9–13 May 1983; International Organization of Citrus Virologists, University of California: Riverside, CA, USA, 1984; pp. 100–108. [Google Scholar]
- Mercado, B.G.; Pableo, F.; Gavarra, M.R.; Gonzales, C.I. Population studies and biological control of Diaphorina citri Kuwayama, the insect vector of citrus greening disease in the Philippines. In Proceedings of the 6th International Asia Pacific Workshop on Integrated Citrus Health Management, Pasadena, CA, USA, 2–5 September 1991; pp. 105–117. [Google Scholar]
- Hoy, M.A.; Nguyen, R. Classical biological control of Asian citrus psylla. Citrus Ind. 2000, 81, 48–50. [Google Scholar]
- Michaud, J.P. The Asian citrus psylla, Diaphorina citri, and its natural enemies. Citrus Ind. 2000, 81, 42–44. [Google Scholar]
- Khan, A.A.; Qureshi, J.A.; Afzal, M.; Stansly, P.A. Two-spotted ladybeetle Adalia bipunctata L. (Coleoptera: Coccinellidae): A commercially available predator to control Asian Citrus Psyllid Diaphorina citri (Hemiptera: Liviidae). PLoS ONE 2016, 11, e0162843. [Google Scholar] [CrossRef]
- Catling, H.D. Distribution of the psyllid vectors of citrus greening disease with notes on the biology and bionomics of D. citri Kuw. FAO Plant Prot. Bull. 1970, 18, 8–15. [Google Scholar]
- Milosavljevic, I.; Morgan, D.J.W.; Massie, R.E.; Hoddle, M.S. Density dependent mortality, climate, and Argentine ants affect population dynamics of an invasive citrus pest, Diaphorina citri, and its specialist parasitoid, Tamarixia radiata, in Southern California, USA. Biol. Control. 2021, 159, 104627. [Google Scholar] [CrossRef]
- Catling, H.D. The bionomics of the South African citrus psylla, Trioza erytreae (Del Guercio) (Homoptera: Psyllidae). 1. The influence of the flushing rhythm of citrus and factors which regulate flushing. J. Entomol. Soc. S. Afr. 1969, 32, 191–208. [Google Scholar]
- Ko, W.W. Citrus diseases in Malaysia. In Proceedings of the 6th International Asia Pacific Workshop on Integrated Citrus Health Management, Kuala Lumpur, Malaysia, 23–25 June 1991; Ke, C., Osman, S.B., Eds.; FAO-UNDP-MARDI: Kuala Lumpur, Malaysia, 1991; pp. 147–162. [Google Scholar]
- Lopes, S.A.; Bassanezi, J., Jr.; Belasquej, R.B.; Yamamoto, P.T. Management of citrus Huanglongbing in the State of Sao Paulo–Brazil. In Proceedings of the FFTC-PPRI-NIFTS Joint Workshop on Management of Citrus Greening and Virus Diseases for the Rehabilitation of Citrus Industry in the ASPAC, Hanoi, Vietnam, 14–18 September 2008; Ku, T.Y., Pham, T.H.H., Eds.; Pham Plant Protection Research Institute: Hanoi, Vietnam, 2008; pp. 107–117. [Google Scholar]
- Rae, D.J.; Liang, W.G.; Watson, D.M.; Beattie, G.A.C.; Huang, M.D. Evaluation of petroleum spray oils for control of the Asian citrus psyllid, Diaphorina citri (Kuwayama) (Hemiptera: Psyllidae), in China. Int. J. Pest. Mgt. 1997, 43, 71–75. [Google Scholar] [CrossRef]
- Da Graca, J.V. Citrus greening disease. Annu. Rev. Phytopathol. 1991, 29, 109–136. [Google Scholar] [CrossRef]
- Jagoueix, S.; Bové, J.M.; Garnier, M. Comparison of the 16S/23S ribosomal intergenic regions of ‘Candidatus Liberobacter asiaticum’ and ‘Candidatus Liberobacter africanum’, the two species associated with citrus Huanglongbing (greening) disease. Int. J. Syst. Bacteriol. 1997, 47, 224–227. [Google Scholar] [CrossRef]
- SAS. SAS Procedures Guide, Version 9; SAS Institute: Cary, NC, USA, 2008. [Google Scholar]
- Zar, J.H. Biostatistical Analysis, 4th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1999. [Google Scholar]
- Fung, Y.C.; Chen, C.N. Effects of temperature and host plant on population parameters of the citrus Psyllid (Diaphorina citri Kuwayama). Formos Entomol. 2006, 26, 109–123. [Google Scholar]
- Liu, Y.H.; Tsai, J.H. Effects of temperature and biology on life table parameters of the Asian citrus Psyllid, Diaphorina citri Kuwayama (Homoptera: Psyllidae). Ann. Appl. Biol. 2000, 137, 201–206. [Google Scholar] [CrossRef]
- Sétamou, M.; Bartels, D.W. Living on the edges: Spatial niche occupation of Asian Citrus Psyllid, Diaphorina citri Kuwayama (Hemiptera: Liviidae), in citrus groves. PLoS ONE 2015, 10, e0131917. [Google Scholar] [CrossRef]
- Aurambout, J.P.; Finlay, K.J.; Luck, J.; Beattie, G.A.C. A concept model to estimate the potential distribution of the Asiatic citrus psyllid (Diaphorina citri Kuwayama) in Australia under climate change—A means for assessing biosecurity risk. Ecol. Modell. 2009, 220, 2512–2524. [Google Scholar] [CrossRef]
- Wang, C.L. Ecological studies of Asiatic citrus psyllid (Diaphorina citri K.) with special reference to its spatial distribution. J. Agric. Res. China 1981, 30, 412–419. [Google Scholar]
- Milosavljevic, I.; Amrich, R.; Strode, V.; Hoddle, M.S. Modeling the phenology of Asian citrus psyllid (Hemiptera: Liviidae) in urban southern California: Effects of environment, habitat, and natural enemies. Environ. Entomol. 2018, 47, 233–243. [Google Scholar] [CrossRef]
- Weerawut, P. Seasonal abundance of Diaphorina citri Kuwayama and Phyllocnistis citrella Stainton on mandarin orange in Nan. In Proceedings of the Fourth International Asia-Pacific Conference on Citrus Rehabilitation, Chiang Mai, Thailand, 12 September 1990; Aubert, B., Tontyaporn, S., Buangsuwon, D., Eds.; NDP-FAO Press: Chiang Mai, Thailand, 1990; pp. 207–211. [Google Scholar]
- Leong, S.S.; Leong, S.C.T.; Beattie, G.A.C. Incidence and spread of Huanglongbing in relation to the distribution and fluctuations of Diaphorina citri Kuwayama (Hemiptera: Psyllidae) population in a citrus orchard. Serangga 2020, 25, 24–42. [Google Scholar]
- Sétamou, M.; Simpson, C.R.; Alabi, O.J.; Nelson, S.D.; Telagamsetty, S.; Jifon, J.L. Quality matters: Influence of citrus flush physicochemical characteristics on population dynamics of the asian citrus psyllid (Hemiptera: Liviidae). PLoS ONE 2016, 11, e0168997. [Google Scholar]
- Yasuda, K.; Kuwamura, F.; Oishi, T. Location and preference of adult Asian Citrus Psyllid, Diaphorina citri (Homoptera: Psyllidae) on chinese box orange jasmine, Murraya exotica L. and flat lemon, Citrus depressa. Japanese. J. Appl. Entomol. Zool. 2005, 49, 146–149. [Google Scholar] [CrossRef][Green Version]
- Martini, X.; Stelinski, L.L. Influence of abiotic factors on flight initiation by Asian citrus psyllid (Hemiptera: Liviidae). Environ. Entomol. 2017, 46, 369–375. [Google Scholar] [CrossRef]
- Zorzenon, F.P.F.; Arthur, F.T.; Lopes, R.S.J.; Miranda, M.P. Factors associated with Diaphorina citri immigration into commercial citrus orchards in São Paulo State, Brazil. J. Appl. Entomol. 2020, 145, 326–335. [Google Scholar] [CrossRef]
- Aubert, B.; Xia, Y.H. Monitoring flight activity of Diaphorinacitri on citrus and Murraya canopies. In Proceedings of the Fourth International Asia-Pacific Conference on Citrus Rehabilitation, Chiang Mai, Thailand, 12 September 1990; Aubert, B., Tontyaporn, S., Buangsuwon, D., Eds.; NDP-FAO Press: Chiang Mai, Thailand, 1990; pp. 181–187. [Google Scholar]
- Rae, D.; Beattie, G.; Nguyen, V.; Nguyen, V.; Pham, V.; Duong, A. Use of Horticultural and Agricultural Mineral Oils in Citrus IPDM; Ha Noi Agricultural Press: Ha Noi, Vietnam, 2003. [Google Scholar]
- Huang, M.; Rae, D.; Beattie, G.; Ouyang, G.; Yang, Y.; Broadbent, P. Green Technology for Citrus Pest and Disease Control; Guangdong Science & Technology Press: Guangzhou, China, 2005. [Google Scholar]
- Rae, D.; Beattie, G.; Huang, M.; Yang, Y.; Ouyang, G. Mineral Oils and Their Use: Sustainable Pest Management and Green Agriculture; Guangdong Science & Technology Press: Guangzhou, China, 2006. [Google Scholar]
- Beattie, G.; Liu, Z.M.; Watson, D.M.; Clift, A.D.C.; Jiang, L.J. Evaluation of petroleum spray oils and polysaccharides for control of Phyllocnistis citrella Stainton (Lepidoptera: Gracillariidae). J. Aust. Ent. Soc. 1995, 34, 349–353. [Google Scholar] [CrossRef]
- Rae, D.J.; Beattie, G.A.C.; Watson, D.M.; Liu, Z.M.; Jiang, L. Effects of petroleum spray oils without with copper fungicides on the control of citrus leafminer, Phyllocnistis citrella Stainton (Lepidoptera: Gracillariidae). Aust. J. Entomol. 1996, 35, 247–251. [Google Scholar] [CrossRef]
- Aubert, B.; Garnier, M.; Guillaumin, D.; Herbagyandono, B.; Setiobudi, L.; Nurhadi, F. Greening, a serious threat for the citrus production of the Indonesia archipelago. Future prospects of integrated control. Fruits 1985, 40, 549–563. [Google Scholar]
- Xu, C.F.; Xia, Y.H.; Li, K.B.; Ke, C. Further study of the transmission of citrus huanglongbin by a psyllid, Diaphorina citri Kuwayama. In Proceedings of the 10th Conference of the International Organization of Citrus Virologists, Riverside, CA, USA, 23 November 1989; Timmer, L.W., Garnsey, S.M., Navarro, L., Eds.; IOCV: Riverside, CA, USA, 1989; pp. 243–248. [Google Scholar]
- Simons, J.N.; Zitter, T.A. Use of oils to control aphid borne viruses. Plant Dis. 1980, 64, 542–546. [Google Scholar] [CrossRef]
- MacKenzie, T.D.B.; Lavoie, J.; Nie, X.; Singh, M. Effectiveness of combined use of mineral oil and insecticide spray in reducing potato virus Y (PVY) spread under field conditions in New Brunswick, Canada. Am. J. Potato Res. 2017, 94, 70–80. [Google Scholar] [CrossRef]
- Vandenveken, J.J. Oils and other inhibitors of nonpersistent virus transmission. In Aphids as Virus Vectors; Harris, K.F., Maramorosh, K., Eds.; Acadamic Press: New York, NY, USA, 1977; pp. 435–454. [Google Scholar]
- Hall, D.G.; Moulton, K.M. Transmission rates of ‘Candidatus Liberibacter asiaticus’ to greenhouse seedlings by laboratory colonies of Asian Citrus Psyllid (Hemiptera: Liviidae). J. Econ. Entomol. 2018, 111, 2546–2552. [Google Scholar] [CrossRef] [PubMed]
- Paris, T.M.; Allan, S.A.; Udell, B.J.; Stansly, P.A. Evidence of behavior-based utilization by the Asian citrus psyllid of a combination of UV and green or yellow wavelengths. PLoS ONE 2017, 12, e0189228. [Google Scholar] [CrossRef]
- Lee, J.A.; Halbert, S.E.; Dawson, W.O.C.; Robertson, J.; Keesling, J.E.; Singer, B.H. Asymptomatic spread of huanglongbing and implications for disease control. Proc. Natl. Acad. Sci. USA 2015, 112, 7605–7610. [Google Scholar] [CrossRef]
- Hall, D.G.; Albrecht, U.; Bowman, K.D. Transmission Rates of ‘Ca. Liberibacter asiaticus’ by Asian Citrus Psyllid Are Enhanced by the Presence andevelopmental Stage of Citrus Flush. J. Econ. Entomol. 2016, 109, 558–563. [Google Scholar] [CrossRef]
- Koizumi, M.; Prommintara, M.; Linwattana, G.; Kaisuwan, T. Epidemiological aspects of citrus huanglongbing (greening) disease in thailand. Jpn. Agric. Res. Q. 1997, 31, 205–211. [Google Scholar]
- Bove, J.M.; Garnier, M.; Ahlawat, Y.S.; Charkraborty, N.K.; Varma, A. Detection of the Asian strains of the greening BLO by DNA-DNA hybridization in Indian orchard trees and Malaysian Diaphorina citri psyllids. In Proceedings of the 12th Conference International Organization of Citrus Virologists, Riverside, CA, USA, 23–25 September 1992; Da Graca, J.V., Moreno, P., Yokimi, R.K., Eds.; IOCV: Riverside, CA, USA, 1992; pp. 258–263. [Google Scholar]
- Stelinski, L.L. Ecological aspects of the vector-borne bacterial disease, citrus greening (Huanglongbing): Dispersal and host use by Asian citrus psyllid, Diaphorina citri Kuwayama. Insects 2019, 10, 208. [Google Scholar] [CrossRef] [PubMed]
| Treatment | Mean Number 1 | Percent of Infestation 2 | Total | |||
|---|---|---|---|---|---|---|
| Adults | Eggs | Nymphs | Adults | Eggs | ||
| HMO | 3.79 ± 0.79 | 1.39 ± 0.09 | 2.66 ± 0.66 | 11.44 ± 0.14 | 8.99 ± 0.54 | 5.65 ± 4.04 A |
| Control | 7.80 ±1.47 | 4.30 ± 0.44 | 5.41 ± 0.20 | 35.75 ± 0.75 | 39.99 ± 1.89 | 18.65 ± 16.37 B |
| Total mean | 5.80 ± 2.44 a | 2.85 ± 1.62 b | 4.04 ± 1.56 b | 23.60 ± 13.32 c | 24.49 ± 17.02 c | |
| Tree | Treatment | Sampling Date | Total | ||||
|---|---|---|---|---|---|---|---|
| 7 April 2012 | 6 August 2012 | 10 April 2013 | 20 March 2014 | 8 October 2014 | |||
| SP | Control | 10.00 ± 2.00 | 38.70 ± 2.70 | 56.20 ± 1.20 | 62.30 ± 1.13 | 80.15 ± 1.23 | 49.47 ± 24.65 A |
| HMO | 6.00 ± 2.00 | 15.00 ± 1.00 | 18.60 ± 1.68 | 25.20 ± 1.11 | 40.28 ± 1.60 | 21.02 ± 11.93 B | |
| DP | Control | 2.41 ± 0.50 | 7.89 ± 0.81 | 19.19 ± 6.97 | 28.20 ± 12.61 | 42.20 ± 18.59 | 20.04 ± 14.76 a |
| HMO | 0.25 ± 0.51 | 0.51 ± 0.58 | 1.39 ± 0.96 | 5.26 ± 2.50 | 11.43 ± 3.68 | 3.77 ± 4.41 b | |
| Total | 8.00 ± 2.83 1 | 26.85 ± 13.11 2 | 37.40 ± 20.64 3 | 43.75 ± 20.35 4 | 60.22 ± 21.84 5 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leong, S.S.; Leong, S.C.T.; Beattie, G.A.C. Effect of Horticultural Mineral Oil on Huanglongbing Transmission by Diaphorina citri Kuwayama (Hemiptera: Psyllidae) Population in a Commercial Citrus Orchard in Sarawak, Malaysia, Northern Borneo. Insects 2021, 12, 772. https://doi.org/10.3390/insects12090772
Leong SS, Leong SCT, Beattie GAC. Effect of Horticultural Mineral Oil on Huanglongbing Transmission by Diaphorina citri Kuwayama (Hemiptera: Psyllidae) Population in a Commercial Citrus Orchard in Sarawak, Malaysia, Northern Borneo. Insects. 2021; 12(9):772. https://doi.org/10.3390/insects12090772
Chicago/Turabian StyleLeong, Sui S., Stephen C. T. Leong, and George Andrew Charles Beattie. 2021. "Effect of Horticultural Mineral Oil on Huanglongbing Transmission by Diaphorina citri Kuwayama (Hemiptera: Psyllidae) Population in a Commercial Citrus Orchard in Sarawak, Malaysia, Northern Borneo" Insects 12, no. 9: 772. https://doi.org/10.3390/insects12090772
APA StyleLeong, S. S., Leong, S. C. T., & Beattie, G. A. C. (2021). Effect of Horticultural Mineral Oil on Huanglongbing Transmission by Diaphorina citri Kuwayama (Hemiptera: Psyllidae) Population in a Commercial Citrus Orchard in Sarawak, Malaysia, Northern Borneo. Insects, 12(9), 772. https://doi.org/10.3390/insects12090772






