Are There Personality Differences between Rural vs. Urban-Living Individuals of a Specialist Ground Beetle, Carabus convexus?
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling Design
2.2. Test Organism
2.3. Testing, Evaluating and Measuring Behavioral Parameters
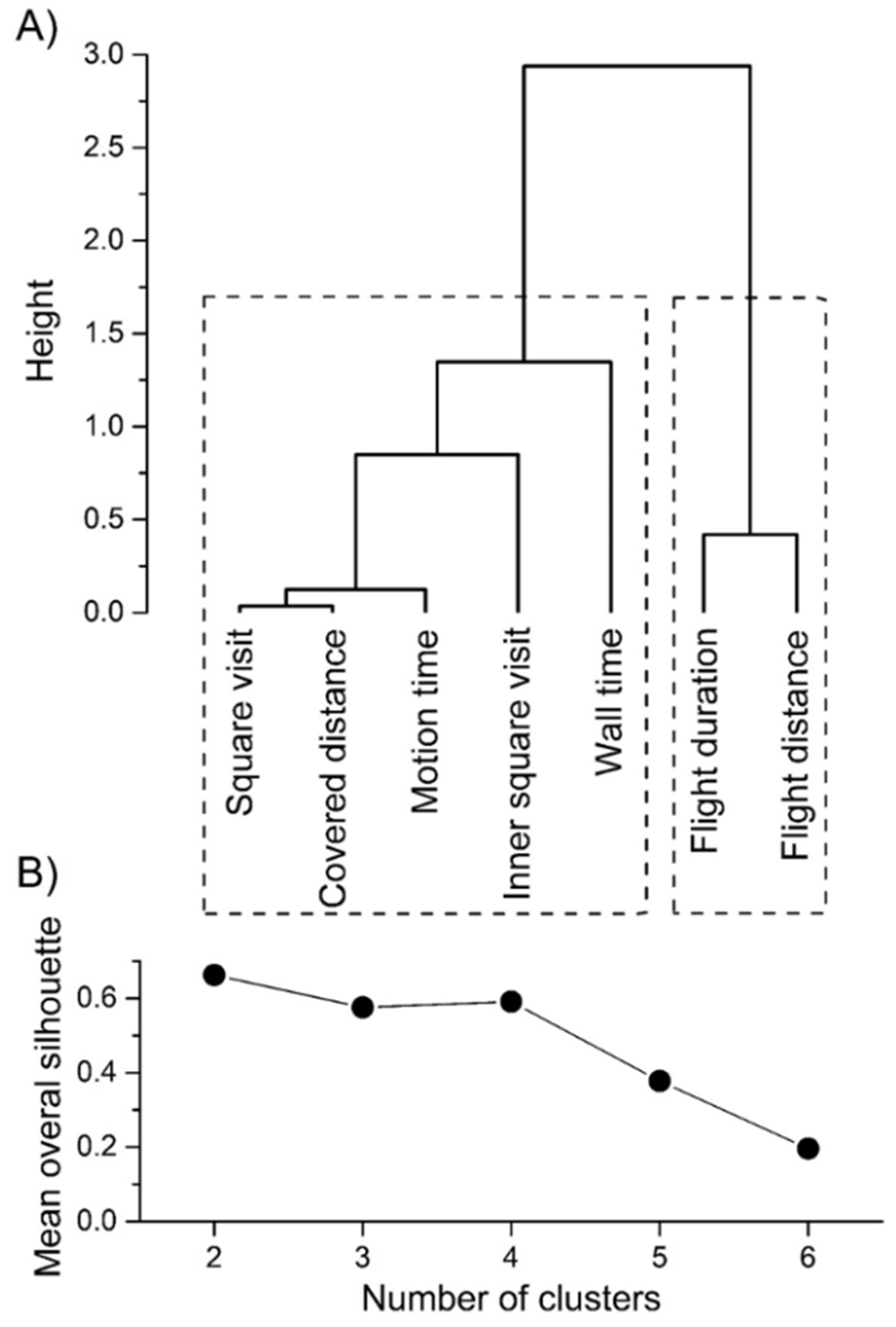
2.4. Statistical Analyses
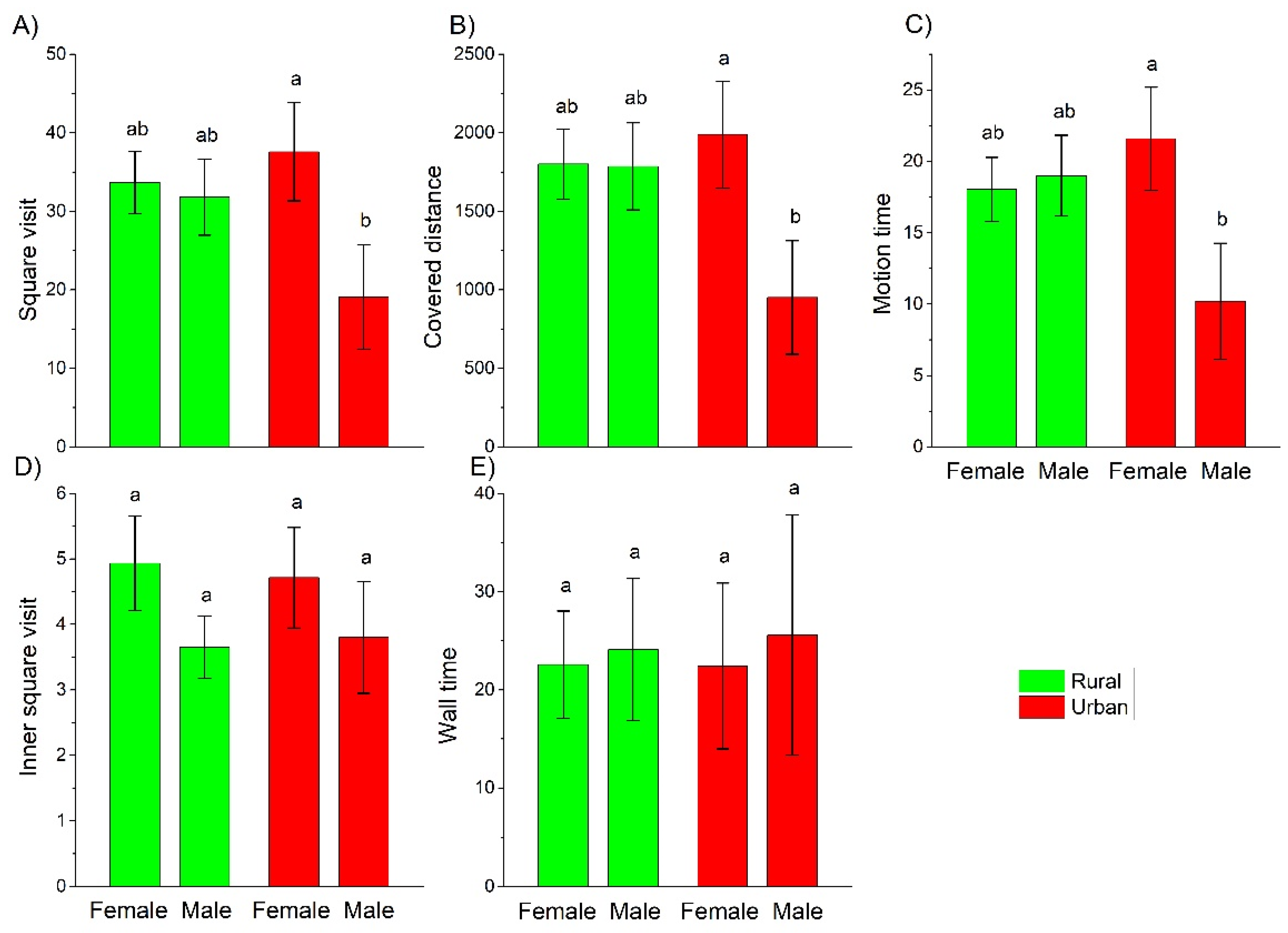
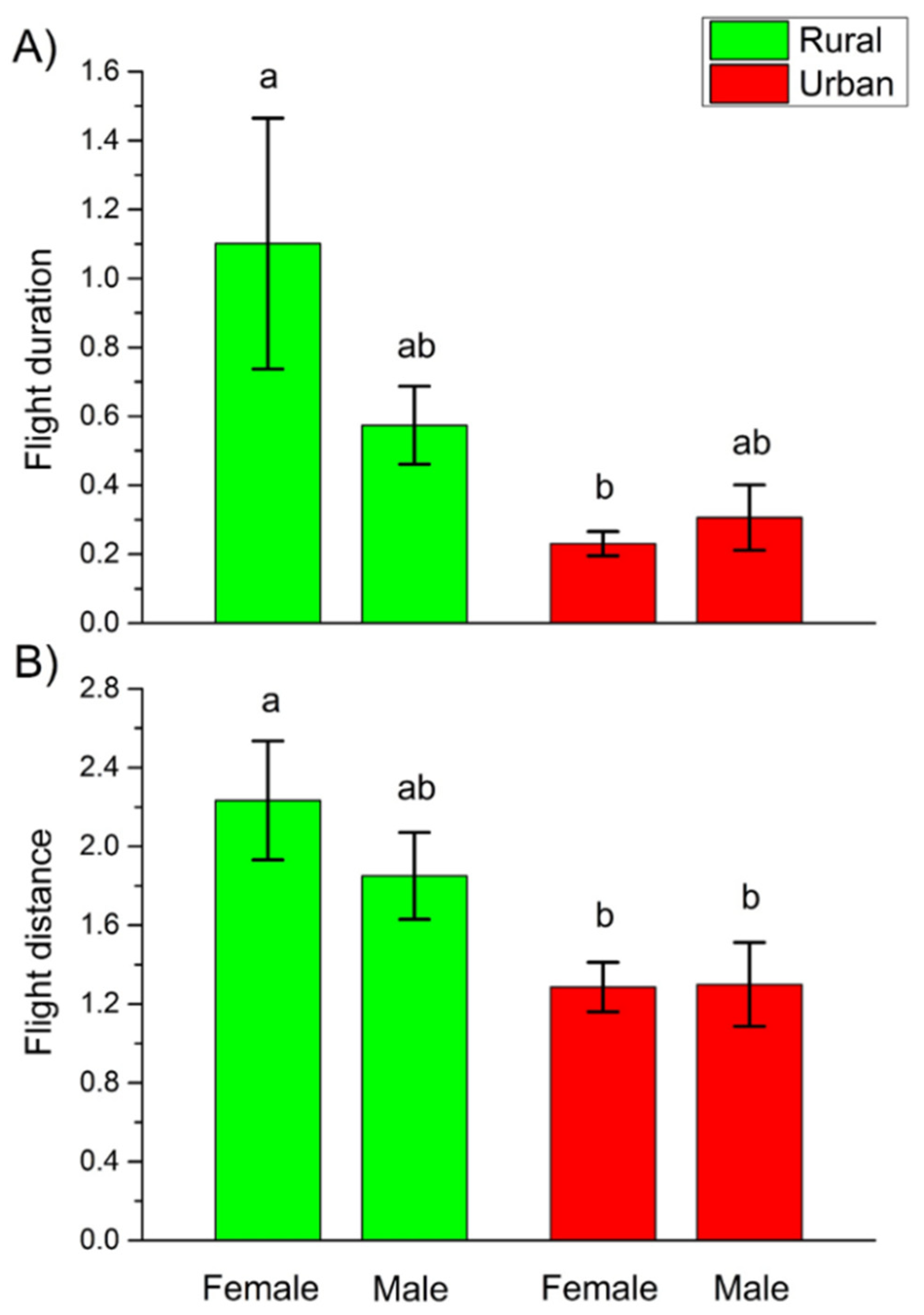
3. Results
4. Discussion
4.1. Personality
4.2. Behavioral Differences
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McIntyre, N.E. Urban Ecology—Definitions and Goals. In The Routledge Handbook of Urban Ecology; Douglas, I., Goode, D., Houck, M., Wang, R., Eds.; Routledge: London, UK, 2011; pp. 7–16. [Google Scholar]
- McKinney, M.L. Urbanization as a major cause of biotic homogenization. Biol. Conserv. 2006, 127, 247–260. [Google Scholar] [CrossRef]
- Forman, R.T.T. Urban Regions: Ecology and Planning beyond the City; Cambridge University Press: Cambridge, UK, 2008; ISBN 9780511754982. [Google Scholar]
- Niemelä, J. Ecology and urban planning. Biodivers. Conserv. 1999, 8, 119–131. [Google Scholar] [CrossRef]
- Simon, E.; Harangi, S.; Baranyai, E.; Braun, M.; Fábián, I.; Mizser, S.; Nagy, L.; Tóthmérész, B. Distribution of toxic elements between biotic and abiotic components of terrestrial ecosystem along an urbanization gradient: Soil, leaf litter and ground beetles. Ecol. Indic. 2016, 60, 258–264. [Google Scholar] [CrossRef]
- Kalnay, E.; Cai, M. Impact of urbanization and land-use change on climate. Nature 2003, 423, 528–531. [Google Scholar] [CrossRef]
- Liu, W.; You, H.; Dou, J. Urban-rural humidity and temperature differences in the Beijing area. Theor. Appl. Climatol. 2009, 96, 201–207. [Google Scholar] [CrossRef]
- Pouyat, R.V.; McDonnell, M.J.; Pickett, S.T.A. Litter decomposition and nitrogen mineralization in oak stands along an urban-rural land use gradient. Urban Ecosyst. 1997, 1, 117–131. [Google Scholar] [CrossRef]
- McDonnell, M.J.; Pickett, S.T.A.; Groffman, P.; Bohlen, P.; Pouyat, R.V.; Zipperer, W.C.; Parmelee, R.W.; Carreiro, M.M.; Medley, K. Ecosystem processes along an urban-to-rural gradient. Urban Ecosyst. 1997, 1, 21–36. [Google Scholar] [CrossRef]
- Magura, T.; Lövei, G.L.; Tóthmérész, B. Conversion from environmental filtering to randomness as assembly rule of ground beetle assemblages along an urbanization gradient. Sci. Rep. 2018, 8, 16992. [Google Scholar] [CrossRef] [Green Version]
- Magura, T.; Lövei, G.L. Consequences of urban living: Urbanization and ground beetles. Curr. Landsc. Ecol. Rep. 2021, 6, 9–21. [Google Scholar] [CrossRef]
- Eötvös, C.B.; Magura, T.; Lövei, G.L. A meta-analysis indicates reduced predation pressure with increasing urbanization. Landsc. Urban Plan. 2018, 180, 54–59. [Google Scholar] [CrossRef]
- Grimm, N.B.; Faeth, S.H.; Golubiewski, N.E.; Redman, C.L.; Wu, J.; Bai, X.; Briggs, J.M. Global change and the ecology of cities. Science 2008, 319, 756–760. [Google Scholar] [CrossRef] [Green Version]
- Kowarik, I. Novel urban ecosystems, biodiversity, and conservation. Environ. Pollut. 2011, 159, 1974–1983. [Google Scholar] [CrossRef] [PubMed]
- Magura, T.; Lövei, G.L.; Tóthmérész, B. Does urbanization decrease diversity in ground beetle (Carabidae) assemblages? Glob. Ecol. Biogeogr. 2010, 19, 16–26. [Google Scholar] [CrossRef] [Green Version]
- McKinney, M.L. Effects of urbanization on species richness: A review of plants and animals. Urban Ecosyst. 2008, 11, 161–176. [Google Scholar] [CrossRef]
- Magura, T.; Kiss, E.; Lövei, G.L. No consistent diversity patterns in terrestrial mammal assemblages along rural-urban forest gradients. Basic Appl. Ecol. 2021, 52, 38–45. [Google Scholar] [CrossRef]
- Chace, J.F.; Walsh, J.J. Urban effects on native avifauna: A review. Landsc. Urban Plan. 2006, 74, 46–69. [Google Scholar] [CrossRef]
- Batáry, P.; Kurucz, K.; Suarez-Rubio, M.; Chamberlain, D.E. Non-linearities in bird responses across urbanization gradients: A meta-analysis. Glob. Chang. Biol. 2018, 24, 1046–1054. [Google Scholar] [CrossRef] [PubMed]
- Williams, N.S.G.; Hahs, A.K.; Vesk, P.A. Urbanisation, plant traits and the composition of urban floras. Perspect. Plant. Ecol. Evol. Syst. 2015, 17, 78–86. [Google Scholar] [CrossRef]
- Singh, A.K.; Singh, H.; Singh, J.S. Plant diversity in cities: Call for assessment and conservation. Curr. Sci. 2018, 115, 428–435. [Google Scholar] [CrossRef]
- Fenoglio, M.S.; Rossetti, M.R.; Videla, M. Negative effects of urbanization on terrestrial arthropod communities: A meta-analysis. Glob. Ecol. Biogeogr. 2020, 29, 1412–1429. [Google Scholar] [CrossRef]
- Wagner, D.L.; Grames, E.M.; Forister, M.L.; Berenbaum, M.R.; Stopak, D. Insect decline in the Anthropocene: Death by a thousand cuts. Proc. Natl. Acad. Sci. USA 2021, 118. [Google Scholar] [CrossRef]
- Niemelä, J.; Kotze, D.J. Carabid beetle assemblages along urban to rural gradients: A review. Landsc. Urban Plan. 2009, 92, 65–71. [Google Scholar] [CrossRef]
- Martinson, H.M.; Raupp, M.J. A meta-analysis of the effects of urbanization on ground beetle communities. Ecosphere 2013, 4, 60. [Google Scholar] [CrossRef]
- Magura, T.; Ferrante, M.; Lövei, G.L. Only habitat specialists become smaller with advancing urbanisation. Glob. Ecol. Biogeogr. 2020, 29, 1978–1987. [Google Scholar] [CrossRef]
- Lövei, G.L.; Sunderland, K.D. Ecology and behavior of ground beetles (Coleoptera: Carabidae). Annu. Rev. Entomol. 1996, 41, 231–256. [Google Scholar] [CrossRef] [PubMed]
- Gosling, S.D. From mice to men: What can we learn about personality from animal research? Psychol. Bull. 2001, 127, 45–86. [Google Scholar] [CrossRef] [Green Version]
- Bell, A.M.; Hankison, S.J.; Laskowski, K.L. The repeatability of behaviour: A meta-analysis. Anim. Behav. 2009, 77, 771–783. [Google Scholar] [CrossRef] [Green Version]
- Cadotte, M.W.; Yasui, S.L.E.; Livingstone, S.; MacIvor, J.S. Are urban systems beneficial, detrimental, or indifferent for biological invasion? Biol. Invasions 2017, 19, 3489–3503. [Google Scholar] [CrossRef]
- Ducatez, S.; Sayol, F.; Sol, D.; Lefebvre, L. Are urban vertebrates city specialists, artificial habitat exploiters, or environmental generalists? Integr. Comp. Biol. Biol. 2018, 58, 929–938. [Google Scholar] [CrossRef] [PubMed]
- Mogi, M.; Armbruster, P.A.; Tuno, N. Differences in responses to urbanization between invasive mosquitoes, Aedes japonicus japonicus (Diptera: Culicidae) and Aedes albopictus, in their native range, Japan. J. Med. Entomol. 2019, 57, 104–112. [Google Scholar] [CrossRef]
- Schuett, W.; Delfs, B.; Haller, R.; Kruber, S.; Roolfs, S.; Timm, D.; Willmann, M.; Drees, C. Ground beetles in city forests: Does urbanization predict a personality trait? PeerJ 2018, 6, e4360. [Google Scholar] [CrossRef] [Green Version]
- Breck, S.W.; Poessel, S.A.; Mahoney, P.; Young, J.K. The intrepid urban coyote: A comparison of bold and exploratory behavior in coyotes from urban and rural environments. Sci. Rep. 2019, 9, 2104. [Google Scholar] [CrossRef] [PubMed]
- Dammhahn, M.; Mazza, V.; Schirmer, A.; Göttsche, C.; Eccard, J.A. Of city and village mice: Behavioural adjustments of striped field mice to urban environments. Sci. Rep. 2020, 10, 13056. [Google Scholar] [CrossRef]
- Magura, T.; Tóthmérész, B.; Molnár, T. Changes in carabid beetle assemblages along an urbanisation gradient in the city of Debrecen, Hungary. Landsc. Ecol. 2004, 19, 747–759. [Google Scholar] [CrossRef]
- Molnár, T.; Magura, T.; Tóthmérész, B.; Elek, Z. Ground beetles (Carabidae) and edge effect in oak-hornbeam forest and grassland transects. Eur. J. Soil Biol. 2001, 37, 297–300. [Google Scholar] [CrossRef]
- Turin, H.; Penev, L.; Casale, A. The Genus Carabus in Europe—A Synthesis; Pensoft: Sofia-Moscow, Russia, 2003. [Google Scholar]
- Magura, T.; Tóthmérész, B.; Molnár, T. A species-level comparison of occurrence patterns in carabids along an urbanisation gradient. Landsc. Urban Plan. 2008, 86, 134–140. [Google Scholar] [CrossRef] [Green Version]
- Niemelä, J.; Kotze, D.J.; Venn, S.; Penev, L.; Stoyanov, I.; Spence, J.; Hartley, D.; de Oca, E.M. Carabid beetle assemblages (Coleoptera, Carabidae) across urban-rural gradients: An international comparison. Landsc. Ecol. 2002, 17, 387–401. [Google Scholar] [CrossRef]
- Magura, T.; Mizser, S.; Horváth, R.; Nagy, D.D.; Tóth, M.; Csicsek, R.; Lövei, G.L. Differences in life history traits in rural vs. urban populations of a specialist ground beetle, Carabus convexus. Insects 2021, 12, 540. [Google Scholar] [CrossRef] [PubMed]
- Réale, D.; Reader, S.M.; Sol, D.; McDougall, P.T.; Dingemanse, N.J. Integrating animal temperament within ecology and evolution. Biol. Rev. 2007, 82, 291–318. [Google Scholar] [CrossRef] [Green Version]
- Kortet, R.; Hedrick, A.N.N. A behavioural syndrome in the field cricket Gryllus integer: Intrasexual aggression is correlated with activity in a novel environment. Biol. J. Linn. Soc. 2007, 91, 475–482. [Google Scholar] [CrossRef] [Green Version]
- Jones, K.A.; Godin, J.-G.J. Are fast explorers slow reactors? Linking personality type and anti-predator behaviour. Proc. R. Soc. B Biol. Sci. 2010, 277, 625–632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Labaude, S.; O’Donnell, N.; Griffin, C.T. Description of a personality syndrome in a common and invasive ground beetle (Coleoptera: Carabidae). Sci. Rep. 2018, 8, 17479. [Google Scholar] [CrossRef] [PubMed]
- Harmer, A.M.T.; Thomas, D.B. pathtrackr: An r package for video tracking and analysing animal movement. Methods Ecol. Evol. 2019, 10, 1196–1202. [Google Scholar] [CrossRef] [Green Version]
- Tremmel, M.; Müller, C. Insect personality depends on environmental conditions. Behav. Ecol. 2013, 24, 386–392. [Google Scholar] [CrossRef] [Green Version]
- Gyuris, E.; Feró, O.; Tartally, A.; Barta, Z. Individual behaviour in firebugs (Pyrrhocoris apterus). Proc. R. Soc. B Biol. Sci. 2011, 278, 628–633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wexler, Y.; Subach, A.; Pruitt, J.N.; Scharf, I. Behavioral repeatability of flour beetles before and after metamorphosis and throughout aging. Behav. Ecol. Sociobiol. 2016, 70, 745–753. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. Artic. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Fox, J.; Weisberg, S. An R Companion to Applied Regression, 3rd ed.; SAGE Publications: Thousand Oaks, CA, USA, 2019; ISBN 9781544336473. [Google Scholar]
- Venables, W.; Ripley, B. Modern Applied Statistics with S, 4th ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Zuur, A.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer: New York, NY, USA, 2009. [Google Scholar]
- Signorell, A.; Aho, K.; Alfons, A.; Anderegg, N.; Aragon, T.; Arachchige, C.; Arppe, A.; Baddeley, A.; Barton, K.; Bolker, B.; et al. DescTools: Tools for Descriptive Statistics 2021. Available online: https://andrisignorell.github.io/DescTools/ (accessed on 1 March 2021).
- Hervé, M. RVAideMemoire: Testing and Plotting Procedures for Biostatistics. R package version 0.9-79. 2021. Available online: https://CRAN.R-project.org/package=RVAideMemoire (accessed on 1 February 2021).
- Stoffel, M.A.; Nakagawa, S.; Schielzeth, H. rptR: Repeatability estimation and variance decomposition by generalized linear mixed-effects models. Methods Ecol. Evol. 2017, 8, 1639–1644. [Google Scholar] [CrossRef] [Green Version]
- Legendre, P. Species associations: The Kendall coefficient of concordance revisited. J. Agric. Biol. Environ. Stat. 2005, 10, 226. [Google Scholar] [CrossRef]
- Maechler, M.; Rousseeuw, P.; Struyf, A.; Hubert, M.; Hornik, K. cluster: Cluster Analysis Basics and Extensions. R package version 2.1.1. 2021. Available online: https://CRAN.R-project.org/package=cluster (accessed on 10 March 2021).
- Rousseeuw, P.J. Silhouettes: A graphical aid to the interpretation and validation of cluster analysis. J. Comput. Appl. Math. 1987, 20, 53–65. [Google Scholar] [CrossRef] [Green Version]
- Sih, A.; Bell, A.; Johnson, J.C. Behavioral syndromes: An ecological and evolutionary overview. Trends Ecol. Evol. 2004, 19, 372–378. [Google Scholar] [CrossRef] [Green Version]
- Nakayama, S.; Nishi, Y.; Miyatake, T. Genetic correlation between behavioural traits in relation to death-feigning behaviour. Popul. Ecol. 2010, 52, 329–335. [Google Scholar] [CrossRef]
- Monceau, K.; Moreau, J.; Richet, J.; Motreuil, S.; Moret, Y.; Dechaume-Moncharmont, F. Larval personality does not predict adult personality in a holometabolous insect. Biol. J. Linn. Soc. 2017, 120, 869–878. [Google Scholar] [CrossRef] [Green Version]
- Müller, T.; Müller, C. Behavioural phenotypes over the lifetime of a holometabolous insect. Front. Zool. 2015, 12, S8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tremmel, M.; Müller, C. Diet dependent experience and physiological state shape the behavior of a generalist herbivore. Physiol. Behav. 2014, 129, 95–103. [Google Scholar] [CrossRef]
- Besson, M.; Martin, J.-R. Centrophobism/thigmotaxis, a new role for the mushroom bodies in Drosophila. J. Neurobiol. 2005, 62, 386–396. [Google Scholar] [CrossRef]
- Humphreys, R.K.; Ruxton, G.D. A review of thanatosis (death feigning) as an anti-predator behaviour. Behav. Ecol. Sociobiol. 2018, 72, 22. [Google Scholar] [CrossRef] [Green Version]
- Cardoso, J.C.F.; dos Santos Mendonça, J. The trade-off between fleeing and tonic immobility behaviors in an ectothermic animal. Acta Ethol. 2019, 22, 129–134. [Google Scholar] [CrossRef]
- Wolf, M.; van Doorn, G.S.; Leimar, O.; Weissing, F.J. Life-history trade-offs favour the evolution of animal personalities. Nature 2007, 447, 581–584. [Google Scholar] [CrossRef]
- Riyahi, S.; Björklund, M.; Mateos-Gonzalez, F.; Senar, J.C. Personality and urbanization: Behavioural traits and DRD4 SNP830 polymorphisms in great tits in Barcelona city. J. Ethol. 2017, 35, 101–108. [Google Scholar] [CrossRef]
- Biondi, L.M.; Fuentes, G.M.; Córdoba, R.S.; Bó, M.S.; Cavalli, M.; Paterlini, C.A.; Castano, M.V.; García, G.O. Variation in boldness and novelty response between rural and urban predatory birds: The Chimango Caracara, Milvago chimango as study case. Behav. Process. 2020, 173, 104064. [Google Scholar] [CrossRef] [PubMed]
- McKinney, M.L. Urbanization, biodiversity, and conservation. Bioscience 2002, 52, 883–890. [Google Scholar] [CrossRef]
- Mazza, V.; Dammhahn, M.; Lösche, E.; Eccard, J.A. Small mammals in the big city: Behavioural adjustments of non-commensal rodents to urban environments. Glob. Chang. Biol. 2020, 26, 6326–6337. [Google Scholar] [CrossRef]
- Batabyal, A.; Balakrishna, S.; Thaker, M. A multivariate approach to understanding shifts in escape strategies of urban lizards. Behav. Ecol. Sociobiol. 2017, 71, 83. [Google Scholar] [CrossRef]
- Kaiser, A.; Merckx, T.; Van Dyck, H. An experimental test of changed personality in butterflies from anthropogenic landscapes. Behav. Ecol. Sociobiol. 2020, 74, 86. [Google Scholar] [CrossRef]
- Kaiser, A.; Merckx, T.; Van Dyck, H. Urbanisation and sex affect the consistency of butterfly personality across metamorphosis. Behav. Ecol. Sociobiol. 2018, 72, 188. [Google Scholar] [CrossRef]
- den Boer, P. Facts, hypotheses and models on the part played by food in the dynamics of carabid populations. In Feeding Behaviour and Accessibility of Food for Carabid Beetles; den Boer, P., Grüm, L., Szyszko, J., Eds.; Warsaw Agricultural University Press: Warsaw, Poland, 1986; pp. 81–96. [Google Scholar]
- Bilde, T.; Toft, S. Quantifying food limitation of arthropod predators in the field. Oecologia 1998, 115, 54–58. [Google Scholar] [CrossRef]
- Heessen, H.J.L. Egg Mortality in Pterostichus oblongopunctatus (Fabricius) (Col., Carabidae). Oecologia 1981, 50, 233–235. [Google Scholar] [CrossRef]
- Sheppard, S.K.; Harwood, J.D. Advances in molecular ecology: Tracking trophic links through predator–prey food-webs. Funct. Ecol. 2005, 19, 751–762. [Google Scholar] [CrossRef]
- Eötvös, C.B.; Lövei, G.L.; Magura, T. Predation pressure on sentinel insect prey along a riverside urbanization gradient in Hungary. Insects 2020, 11, 97. [Google Scholar] [CrossRef] [Green Version]
- Lowry, H.; Lill, A.; Wong, B.B.M. Behavioural responses of wildlife to urban environments. Biol. Rev. 2013, 88, 537–549. [Google Scholar] [CrossRef] [PubMed]
| Behavioral Variable | Spearman Rank-Correlation RS [95% CI] * | Repeatability, r [95% CI] * |
|---|---|---|
| Square visit | 0.5201 [0.1571; 0.7516] | 0.518 [0.140; 0.687] |
| Covered distance | 0.4921 [0.1709; 0.7486] | 0.474 [0.195; 0.686] |
| Motion time | 0.4578 [0.1085; 0.7173] | 0.431 [0.116; 0.668] |
| Inner squares visited | 0.4665 [ 0.1597; 0.7094] | 0.313 [0; 0.518] |
| Wall time | 0.2382 [−0.1131; 0.5569] | 0.402 [0.088, 0.644] |
| Flight duration | 0.5723 [0.2980; 0.7831] | 0.210 [0; 0.513] |
| Flight distance | 0.3967 [0.0656; 0.6983] | 0.042 [0; 0.188] |
| Response Variable | Fixed Effect | Estimate ± SE | χ2 | df | p |
|---|---|---|---|---|---|
| Square visit | Urbanization level | −0.7512 ± 0.4271 | 3.0938 | 1 | 0.0786 |
| Sex | 0.0696 ± 0.3125 | 0.0497 | 1 | 0.8236 | |
| Urbanization level × Sex | 1.0114 ± 0.5321 | 3.9231 | 1 | 0.0476 | |
| Covered distance | Urbanization level | 11.4500 ± 0.0014 | 64,174,354 | 1 | <0.0001 |
| Sex | −0.8748 ± 0.0009 | 850,181 | 1 | <0.0001 | |
| Urbanization level × Sex | −23.1300 ± 0.0009 | 633,904,345 | 1 | <0.0001 | |
| Motion time | Urbanization level | −14.1700 ± 0.0089 | 2,539,792 | 1 | <0.0001 |
| Sex | −18.9800 ± 0.0055 | 12,037,699 | 1 | <0.0001 | |
| Urbanization level × Sex | −68.6400 ± 0.0057 | 144,057,946 | 1 | <0.0001 | |
| Inner square visit | Urbanization level | 0.0297 ± 0.2643 | 0.0126 | 1 | 0.9105 |
| Sex | 0.2687 ± 0.1928 | 1.9430 | 1 | 0.1633 | |
| Urbanization level × Sex | −0.0485 ± 0.3358 | 0.0208 | 1 | 0.8852 | |
| Wall time | Urbanization level | −0.2437 ± 1.1432 | 0.0611 | 1 | 0.8048 |
| Sex | 0.3578 ± 0.5456 | 0.4832 | 1 | 0.4870 | |
| Urbanization level × Sex | −0.1436 ± 1.3654 | 0.0135 | 1 | 0.9075 | |
| Flight duration | Urbanization level | 0.3503 ± 0.8129 | 0.1857 | 1 | 0.6666 |
| Sex | −0.6966 ± 0.5830 | 1.4279 | 1 | 0.2321 | |
| Urbanization level × Sex | −1.0001 ± 0.5537 | 3.8542 | 1 | 0.0496 | |
| Flight distance | Urbanization level | −0.3528 ± 0.3224 | 1.1975 | 1 | 0.2738 |
| Sex | 0.1883 ± 0.2048 | 0.8453 | 1 | 0.3579 | |
| Urbanization level × Sex | −1.1994 ± 0.4176 | 4.2165 | 1 | 0.0400 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magura, T.; Mizser, S.; Horváth, R.; Nagy, D.D.; Tóth, M.; Csicsek, R.; Lövei, G.L. Are There Personality Differences between Rural vs. Urban-Living Individuals of a Specialist Ground Beetle, Carabus convexus? Insects 2021, 12, 646. https://doi.org/10.3390/insects12070646
Magura T, Mizser S, Horváth R, Nagy DD, Tóth M, Csicsek R, Lövei GL. Are There Personality Differences between Rural vs. Urban-Living Individuals of a Specialist Ground Beetle, Carabus convexus? Insects. 2021; 12(7):646. https://doi.org/10.3390/insects12070646
Chicago/Turabian StyleMagura, Tibor, Szabolcs Mizser, Roland Horváth, Dávid D. Nagy, Mária Tóth, Réka Csicsek, and Gábor L. Lövei. 2021. "Are There Personality Differences between Rural vs. Urban-Living Individuals of a Specialist Ground Beetle, Carabus convexus?" Insects 12, no. 7: 646. https://doi.org/10.3390/insects12070646
APA StyleMagura, T., Mizser, S., Horváth, R., Nagy, D. D., Tóth, M., Csicsek, R., & Lövei, G. L. (2021). Are There Personality Differences between Rural vs. Urban-Living Individuals of a Specialist Ground Beetle, Carabus convexus? Insects, 12(7), 646. https://doi.org/10.3390/insects12070646








