Mosquito-Textile Physics: A Mathematical Roadmap to Insecticide-Free, Bite-Proof Clothing for Everyday Life
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Mosquitoes
2.2. In Vitro Feeding/Bioassay System
2.3. In Vivo Bioassay for Bite Resistance
2.4. Walk-in-Cage Studies of Whole Garments
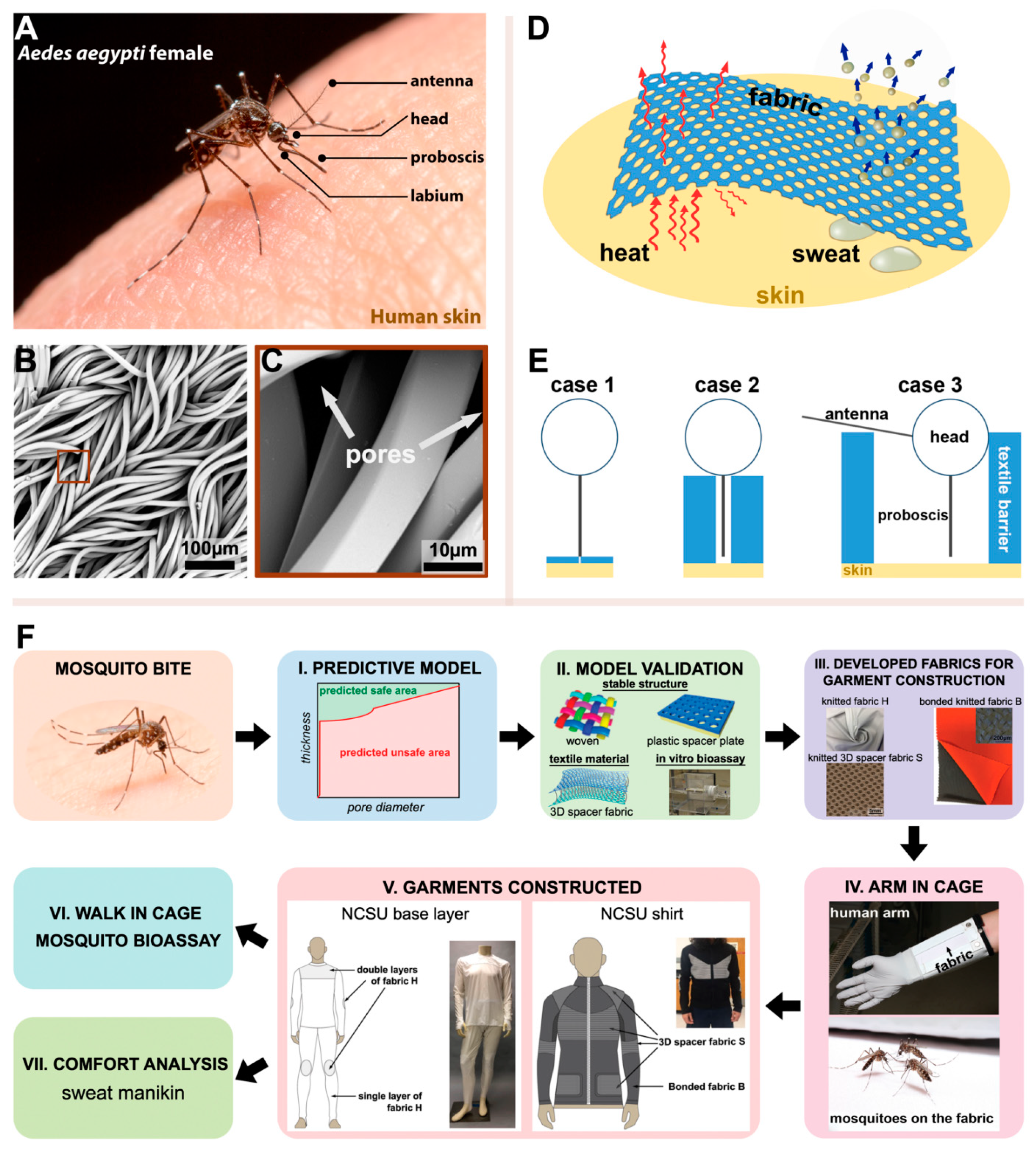
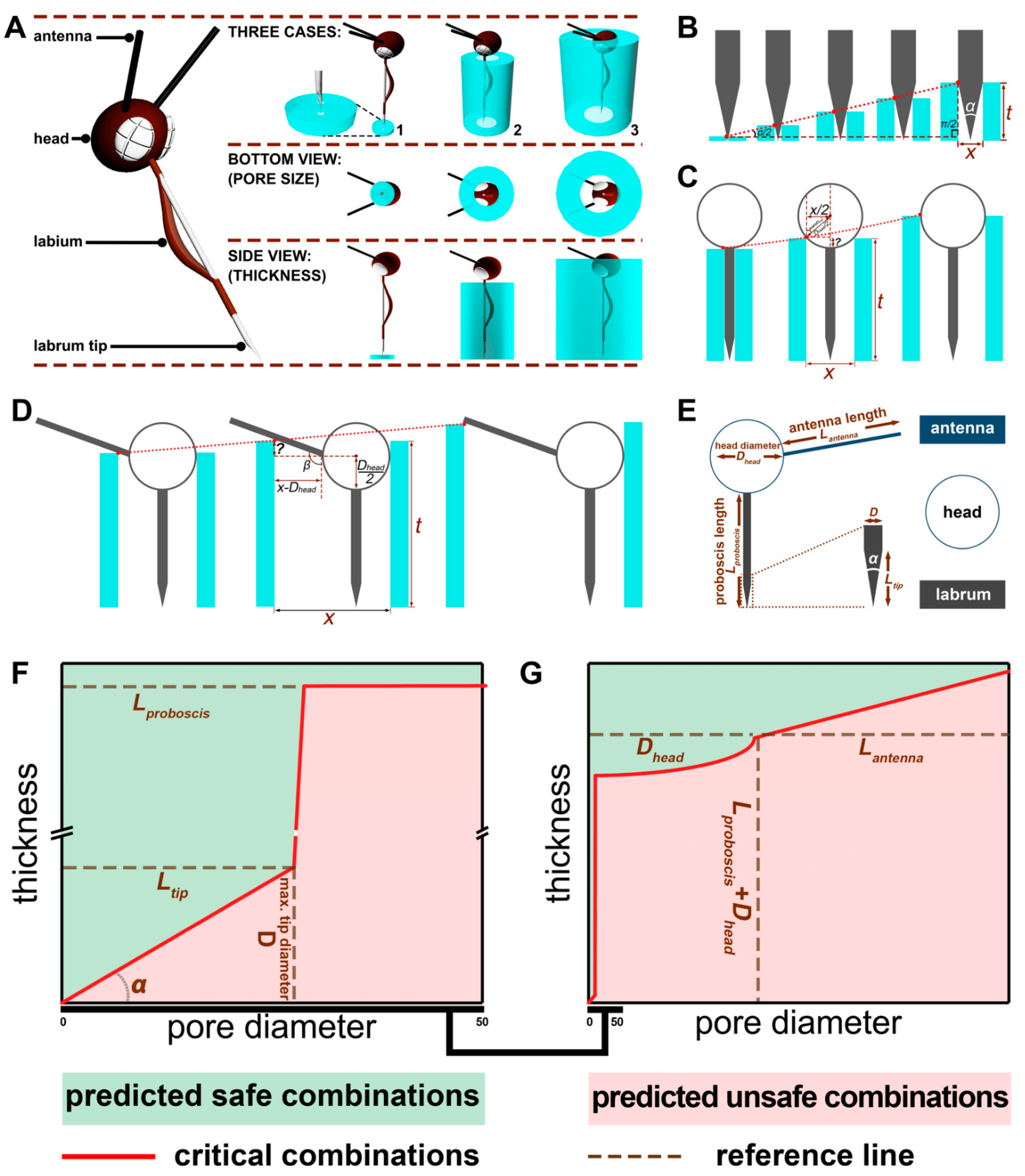
2.5. Model Rationale and Mosquito Morphometrics
2.6. Model Development
2.7. Materials for Model Validation
2.7.1. Stable Structures
2.7.2. Knitted Textile Structures
2.8. Finite Element Model for Proboscis Penetration
2.9. Prototype Bite-Resistant Fabrics Tested for Garment Construction
2.10. Textile Structural Analysis
2.11. Comparison of the Non-Insecticide and Insecticide-Treated Textiles
2.12. Construction of Protective Garments
2.13. Sweat Manikin Test for Comfort Evaluation of Garments
2.14. Data Analysis
3. Results and Discussions
3.1. Mosquito-Bite-Resistant Textile Model
3.2. Mosquito Morphometrics Used to Predict Safe Fabrics
3.3. Finite Element Analysis
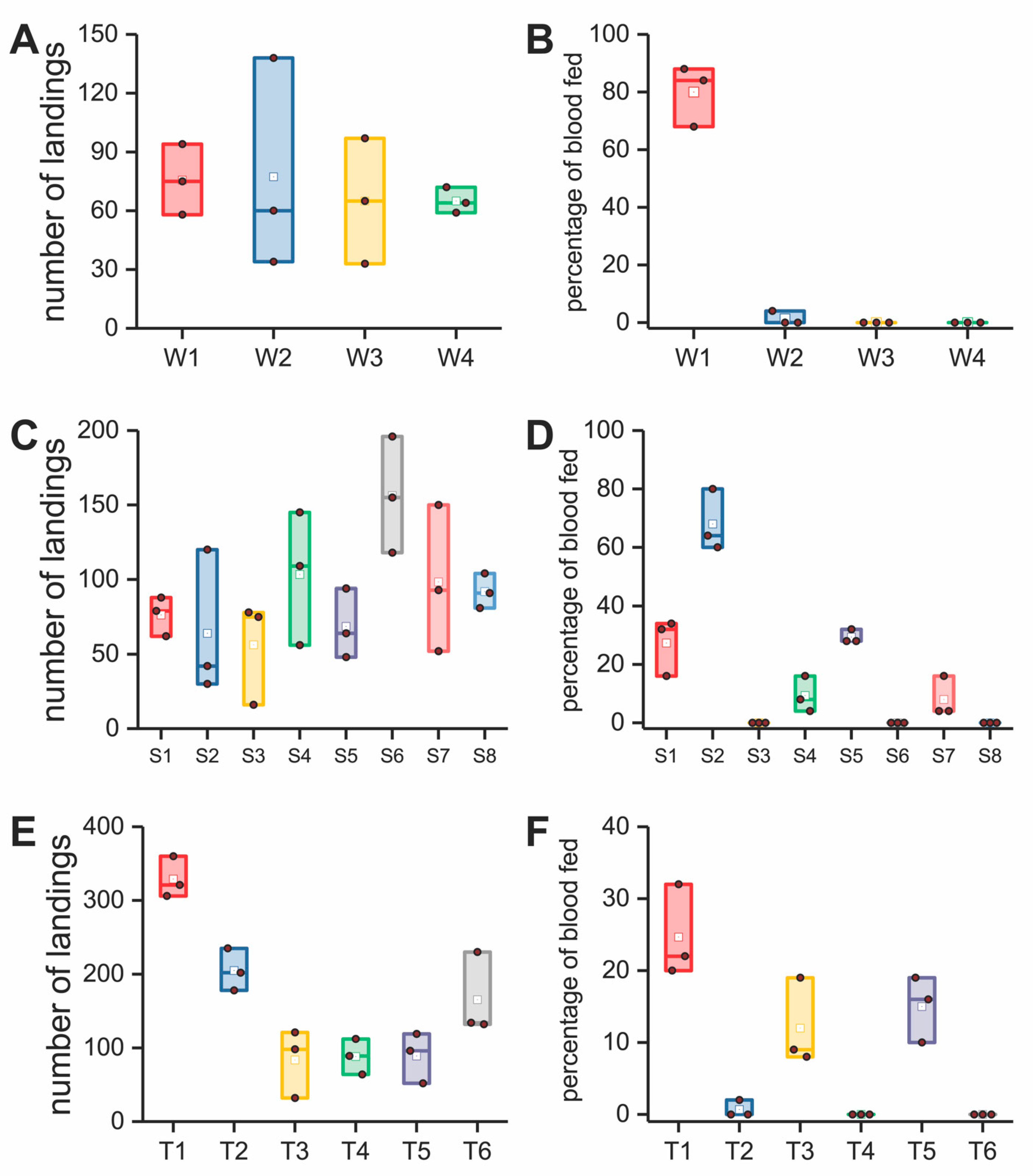
3.4. Development of Fabrics for Garment Construction
3.5. Bite Resistance of an Insecticide-Treated versus Non-Insecticidal Textile
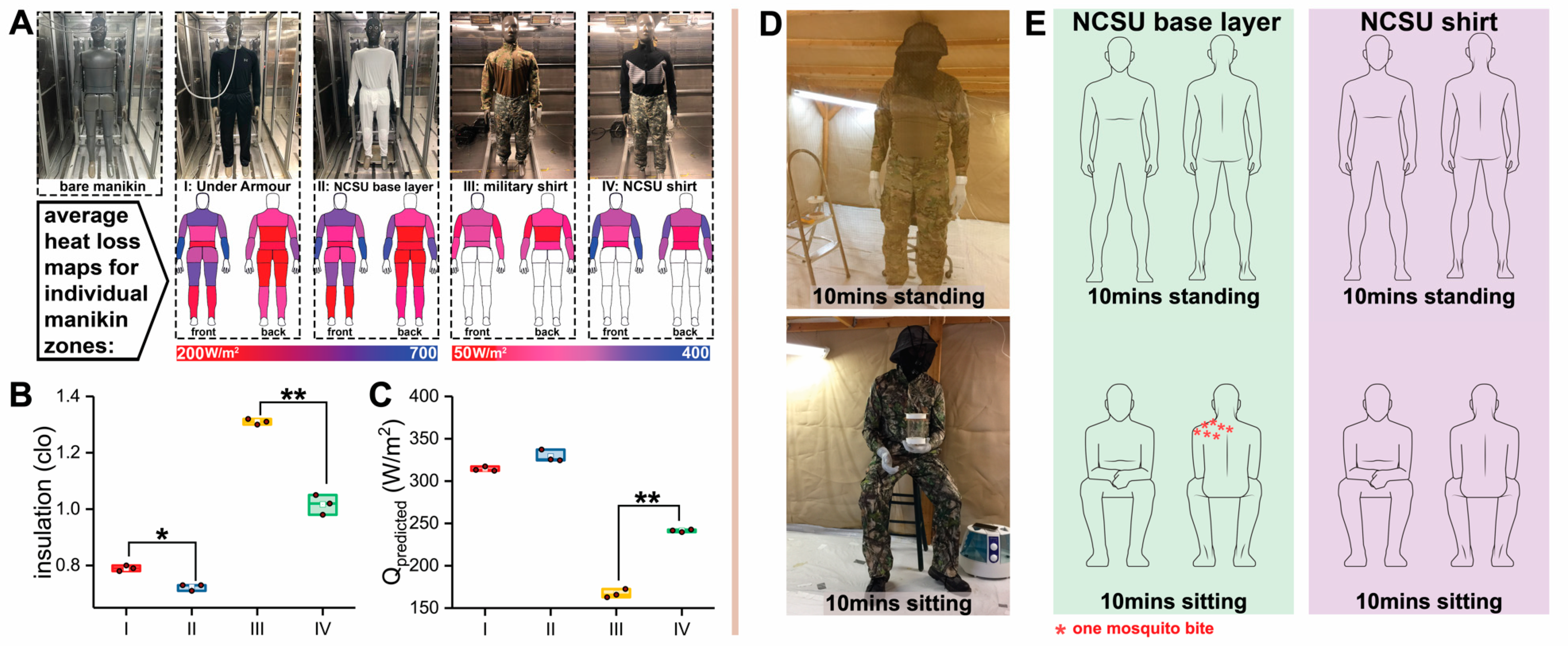
3.6. Comfort and Bioassay Evaluation of Prototype Garments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Franklinos, L.H.V.; Jones, K.E.; Redding, D.W.; Abubakar, I. The effect of global change on mosquito-borne disease. Lancet Infect. Dis. 2019, 19, e302–e312. [Google Scholar] [CrossRef]
- Greppi, C.; Laursen, W.J.; Budelli, G.; Chang, E.C.; Daniels, A.M.; van Giesen, L.; Smidler, A.L.; Catteruccia, F.; Garrity, P.A. Mosquito heat seeking is driven by an ancestral cooling receptor. Science 2020, 367, 681–684. [Google Scholar] [CrossRef]
- World Health Organization. World Malaria Report; WHO: Geneva, Switzerland, 2019; pp. 1–232. [Google Scholar]
- Bhatt, S.; Weiss, D.J.; Cameron, E.; Bisanzio, D.; Mappin, B.; Dalrymple, U.; Battle, K.E.; Moyes, C.L.; Henry, A.; Eckhoff, P.A.; et al. The effect of malaria control on Plasmodium falciparum in Africa between 2000 and 2015. Nature 2015, 526, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Kroeger, A.; Avila, E.V.; Morison, L. Insecticide impregnated curtains to control domestic transmission of cutaneous leishmaniasis in Venezuela: Cluster randomized trial. BMJ 2002, 325, 810–813. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kroeger, A.; Lenhart, A.; Ochoa, M.; Villegas, E.; Levy, M.; Alexander, N.; McCall, P.J. Effective control of dengue vectors with curtains and water container covers treated with insecticide in Mexico and Venezuela: Cluster randomized trials. BMJ 2006, 332, 1247–1252. [Google Scholar] [CrossRef] [PubMed]
- Del-Rahman, A.; Dechkovskaia, A.M.; Goldstein, L.B.; Bullman, S.H.; Khan, W.; El-Masry, E.M.; Abou-Donia, M.B. Neurological Deficits Induced by Malathion, Deet, and Permethrin, Alone or in Combination in Adult Rats. J. Toxicol. Environ. Health Part A 2004, 67, 331–356. [Google Scholar] [CrossRef] [PubMed]
- Hancock, P.A.; Hendriks, C.J.M.; Tangena, J.-A.; Gibson, H.; Hemingway, J.; Coleman, M.; Gething, P.; Cameron, E.; Bhatt, S.; Moyes, C.L. Mapping trends in insecticide resistance phenotypes in African malaria vectors. PLoS Biol. 2020, 18, e3000633. [Google Scholar] [CrossRef]
- Banks, S.D.; Murray, N.; Wilder-Smith, A.; Logan, J.G. Insecticide-treated clothes for the control of vector-borne diseases: A review on effectiveness and safety. Med. Veter. Ètomol. 2014, 28, 14–25. [Google Scholar] [CrossRef]
- Ghosh, T. Stretch, wrap, and relax to smartness. Science 2015, 349, 382–383. [Google Scholar] [CrossRef]
- Luan, K.; West, A.; Denhartog, E.; McCord, M. Auxetic deformation of the weft-knitted Miura-ori fold. Text. Res. J. 2019, 90, 617–630. [Google Scholar] [CrossRef]
- Jinno, H.; Fukuda, K.; Xu, X.; Park, S.; Suzuki, Y.; Koizumi, M.; Yokota, T.; Osaka, I.; Takimiya, K.; Someya, T. Stretchable and waterproof elastomer-coated organic photovoltaics for washable electronic textile applications. Nat. Energy 2017, 2, 780–785. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, H.; Zuo, F.; Yin, X.; Yu, J.; Ding, B. A controlled design of ripple-like Polyamide-6 nanofiber/nets membrane for high-efficiency air filter. Small 2017, 13, 1603151. [Google Scholar] [CrossRef]
- Cui, Y.; Gong, H.; Wang, Y.; Li, D.; Bai, H. A thermally insulating textile inspired by polar bear hair. Adv. Mater. 2018, 30, 1706807. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Cui, Y. Advanced Textiles for Personal Thermal Management and Energy. Joule 2020, 4, 724–742. [Google Scholar] [CrossRef]
- Castilho, C.J.; Li, D.; Liu, M.; Liu, Y.; Gao, H.; Hurt, R.H. Mosquito bite prevention through graphene barrier layers. Proc. Natl. Acad. Sci. USA 2019, 116, 18304–18309. [Google Scholar] [CrossRef] [PubMed]
- Thangamani, S.; Huang, J.; Hart, C.E.; Guzman, H.; Tesh, R.B. Vertical transmission of Zika virus in Aedes aegypti mosquitoes. Am. J. Trop. Med. Hyg. 2016, 95, 1169–1173. [Google Scholar] [CrossRef] [PubMed]
- Clements, A.N. The Biology of Mosquitoes: Development, Nutrition and Reproduction; Chapman & Hall: London, UK, 1992; Volume 1. [Google Scholar]
- Takken, W.; Verhulst, N.O. Host Preferences of Blood-Feeding Mosquitoes. Annu. Rev. Èntomol. 2013, 58, 433–453. [Google Scholar] [CrossRef]
- Benzon, G.L.; Apperson, C.S. An electrically heated membrane blood-feeding device for mosquito colony maintenance. J. Am. Mosq. Control. Assoc. 1987, 3, 322–324. [Google Scholar]
- Halbkat, L.; Luan, K.; Cave, G.; Mccord, M.; Roe, M.; Denhartog, E.; Travanty, N.J.; Apperson, C.S.; West, A.J. Fabric infused with a botanical repellent for protection against mosquitoes. J. Text. Inst. 2019, 110, 1468–1474. [Google Scholar] [CrossRef]
- Kočevar, T.N.; Gabrijelčič Tomc, H. Analysis of methods used for texture preparation for 3D visualization of fabric porosity. J. Text. Inst. 2018, 109, 1270–1281. [Google Scholar] [CrossRef]
- Zhou, Y.-H.; Zhang, Z.-W.; Fu, Y.-F.; Zhang, G.-C.; Yuan, S. Carbon Dioxide, Odorants, Heat and Visible Cues Affect Wild Mosquito Landing in Open Spaces. Front. Behav. Neurosci. 2018, 12, 86. [Google Scholar] [CrossRef] [PubMed]
- Luan, K.; Sun, B.; Gu, B. Ballistic impact damages of 3-D angle-interlock woven composites based on high strain rate constitutive equation of fiber tows. Int. J. Impact Eng. 2013, 57, 145–158. [Google Scholar] [CrossRef]
- Xia, Z.; Zhou, C.; Yong, Q.; Wang, X. On selection of repeated unit cell model and application of unified periodic boundary conditions in micro-mechanical analysis of composites. Int. J. Solids Struct. 2006, 43, 266–278. [Google Scholar] [CrossRef]
- Bhushan, B. Insects locomotion, piercing, sucking and stinging mechanisms. Microsyst. Technol. 2018, 24, 4703–4728. [Google Scholar] [CrossRef]
- Gurera, D.; Bhushan, B.; Kumar, N. Lessons from mosquitoes’ painless piercing. J. Mech. Behav. Biomed. Mater. 2018, 84, 178–187. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- ASTM International. D13 Committee D1777−96 (Reapproved 2019); ASTM International: West Conshohocken, PA, USA, 2019; pp. 1–5. [Google Scholar]
- Barker, R.L. From fabric hand to thermal comfort: The evolving role of objective measurements in explaining human comfort response to textiles. Int. J. Cloth. Sci. Technol. 2002, 14, 181–200. [Google Scholar] [CrossRef]
- Ramasubramanian, M.K.; Barham, O.M.; Swaminathan, V. Mechanics of a mosquito bite with applications to microneedle design. Bioinspiration Biomimetics 2008, 3, 046001. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.Q.; Wu, C. Mosquito proboscis: An elegant biomicroelectromechanical system. Phys. Rev. E 2010, 82, 011910. [Google Scholar] [CrossRef] [PubMed]
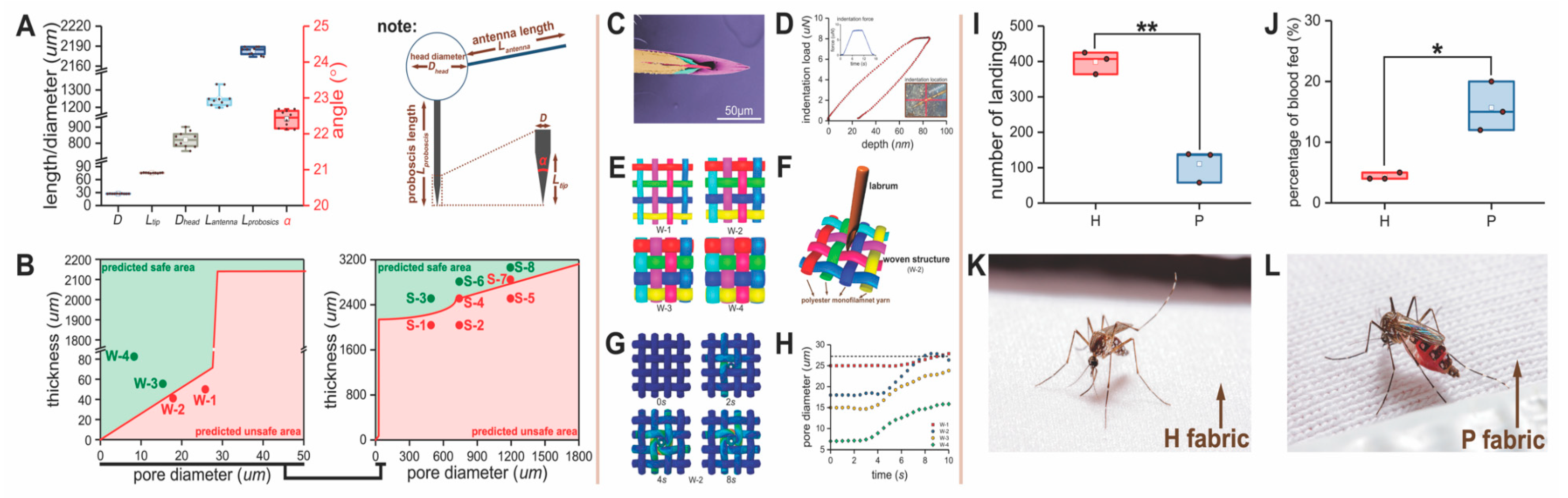
| Group | Type | Name | Abbreviation | Thickness (mm), Pore Diameter (µm) | Model Prediction † | Bioassay Result †† |
|---|---|---|---|---|---|---|
| Materials for model validation (test, in vitro) | Stable structures | Case 1 woven filtration fabrics | W1 | 0.052, 25 | unsafe | fail |
| W2 | 0.040, 18 | unsafe | fail | |||
| W3 | 0.058, 16 | safe | pass | |||
| W4 | 0.082, 8 | safe | pass | |||
| Case 2 plastic plates | S1 | 2.1, 500 | unsafe | fail | ||
| S2 | 2.1, 800 | unsafe | pass | |||
| S3 | 2.5, 500 | safe | pass | |||
| S4 | 2.5, 800 | unsafe | fail | |||
| S5 | 2.5, 1250 | unsafe | fail | |||
| Case 3 plastic plates | S6 | 2.72, 800 | safe | pass | ||
| S7 | 2.75, 1250 | safe | fail | |||
| S8 | 3, 1250 | safe | pass | |||
| Textile materials | Case 1 fabrics | T1 | 0.29, 36 | unsafe | fail | |
| T2 | 0.26, 16 | safe | pass | |||
| Case 2 spacer fabrics | T3 | 2, 120 | unsafe | fail | ||
| T4 | 3.2, 420 | safe | pass | |||
| Case 3 spacer fabrics | T5 | 2, 940 | unsafe | fail | ||
| T6 | 3, 770 | safe | pass | |||
| Fabrics used in garments (test, in vivo) | Textile materials | Case 1 fabric H | H | 0.3, 28 | safe | pass |
| Case 1 fabric B | B | 0.68, 0 | safe | pass | ||
| Case 2 spacer fabric S | S | 2.48, 420 | safe | pass | ||
| Permethrin-treated fabric (test, in vivo) | Chemical-treated textile materials | InsectShield® T-shirt fabric | P | 0.61, 90 | unsafe | fail |
| Garments (test, walk-in cage) | Garments | Under Armour® men’s base 1.0 crew | I | -- | -- | -- |
| NCSU base layer | II | -- | -- | pass | ||
| Winter army combat shirt | III | -- | -- | -- | ||
| NCSU shirt | IV | -- | -- | pass |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luan, K.; West, A.J.; McCord, M.G.; DenHartog, E.A.; Shi, Q.; Bettermann, I.; Li, J.; Travanty, N.V.; Mitchell, R.D., III; Cave, G.L.; et al. Mosquito-Textile Physics: A Mathematical Roadmap to Insecticide-Free, Bite-Proof Clothing for Everyday Life. Insects 2021, 12, 636. https://doi.org/10.3390/insects12070636
Luan K, West AJ, McCord MG, DenHartog EA, Shi Q, Bettermann I, Li J, Travanty NV, Mitchell RD III, Cave GL, et al. Mosquito-Textile Physics: A Mathematical Roadmap to Insecticide-Free, Bite-Proof Clothing for Everyday Life. Insects. 2021; 12(7):636. https://doi.org/10.3390/insects12070636
Chicago/Turabian StyleLuan, Kun, Andre J. West, Marian G. McCord, Emiel A. DenHartog, Quan Shi, Isa Bettermann, Jiayin Li, Nicholas V. Travanty, Robert D. Mitchell, III, Grayson L. Cave, and et al. 2021. "Mosquito-Textile Physics: A Mathematical Roadmap to Insecticide-Free, Bite-Proof Clothing for Everyday Life" Insects 12, no. 7: 636. https://doi.org/10.3390/insects12070636
APA StyleLuan, K., West, A. J., McCord, M. G., DenHartog, E. A., Shi, Q., Bettermann, I., Li, J., Travanty, N. V., Mitchell, R. D., III, Cave, G. L., Strider, J. B., Wang, Y., Neumann, F., Beck, T., Apperson, C. S., & Roe, R. M. (2021). Mosquito-Textile Physics: A Mathematical Roadmap to Insecticide-Free, Bite-Proof Clothing for Everyday Life. Insects, 12(7), 636. https://doi.org/10.3390/insects12070636






