High Survivorship of First-Generation Monarch Butterfly Eggs to Third Instar Associated with a Diverse Arthropod Community
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
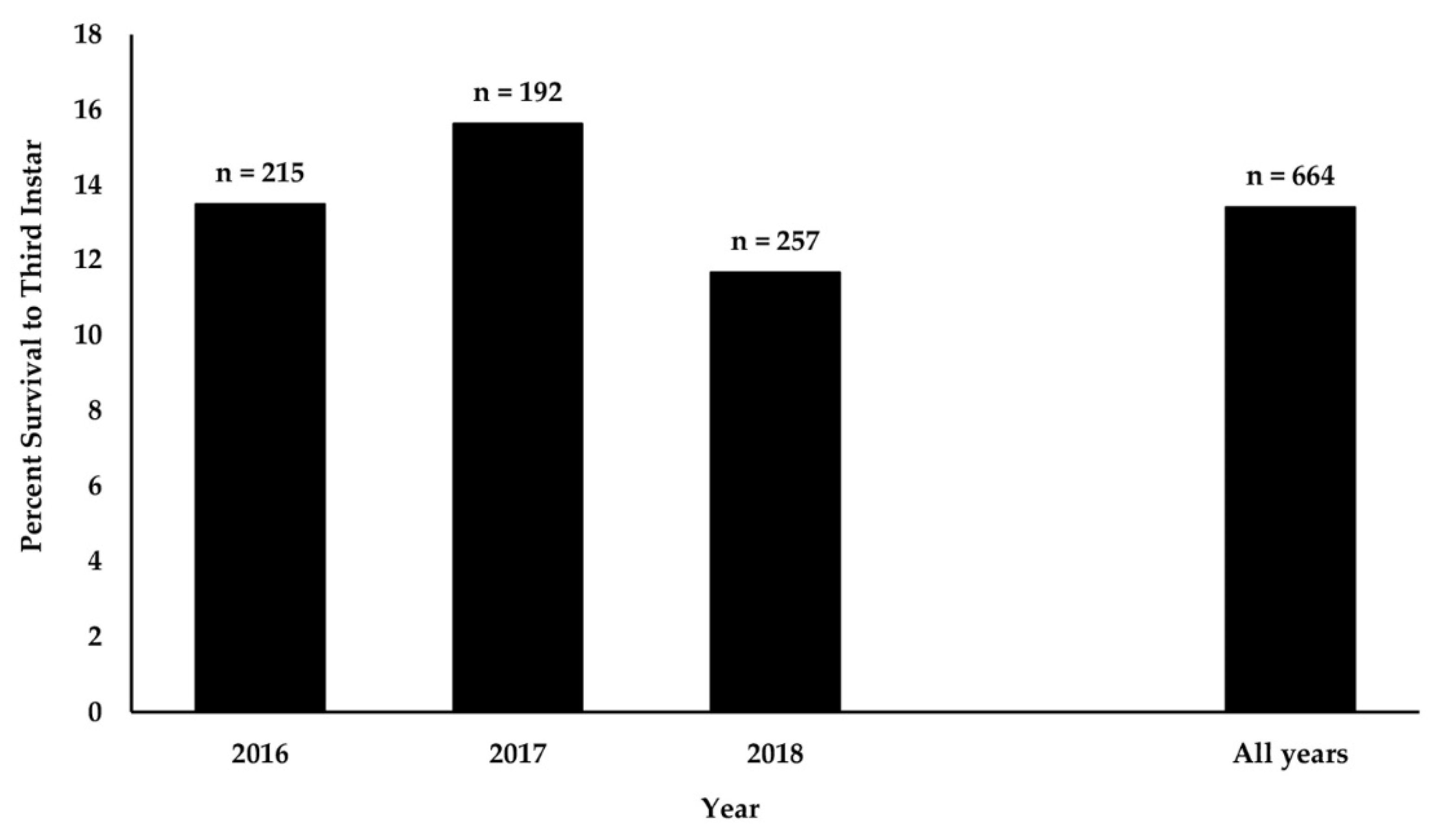
3.1. Survival of Monarch Eggs and Larvae
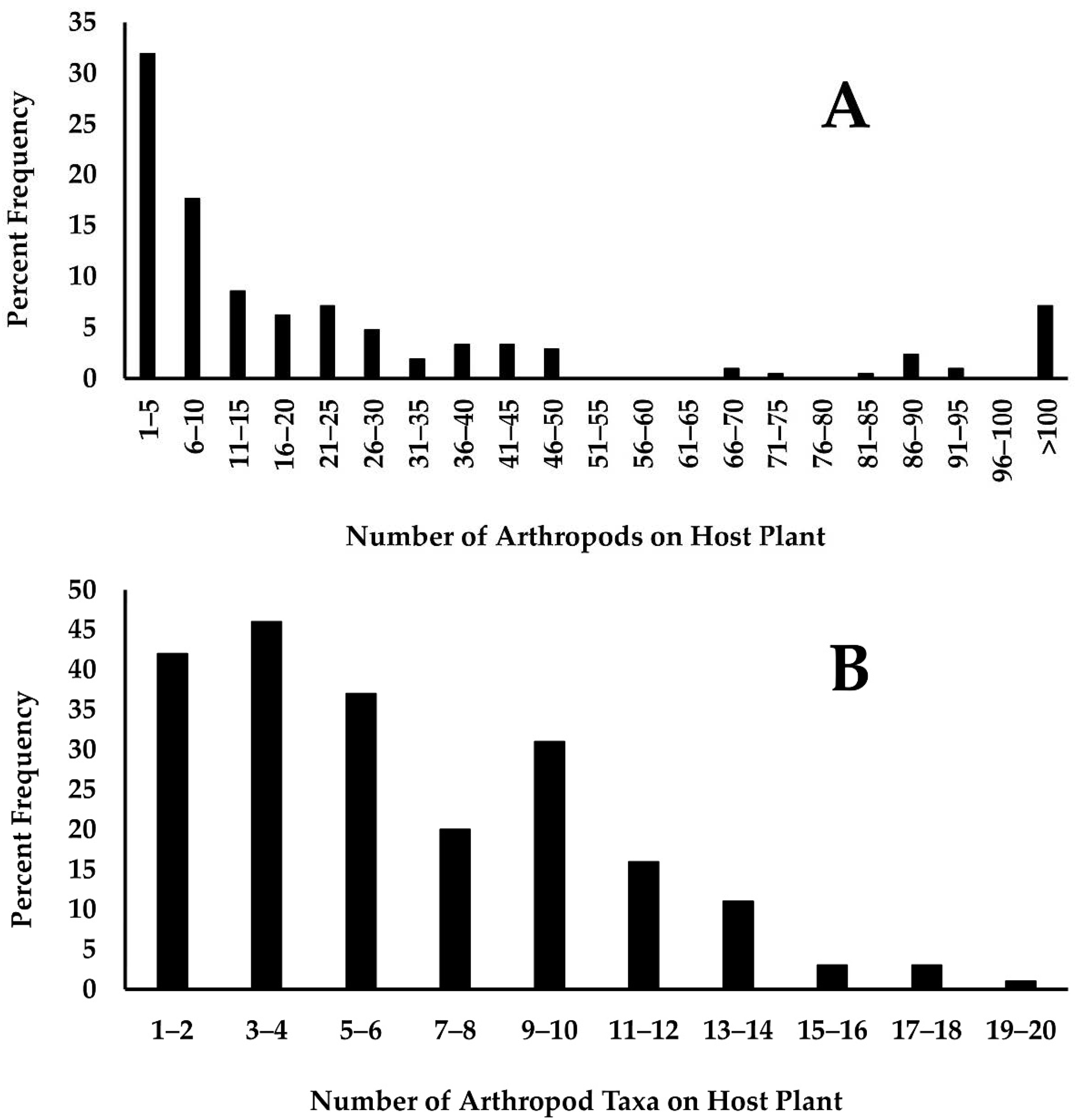
3.2. Host Plant Arthropods and the Survival of Monarch Eggs and Larvae
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Total | Percent | |||
|---|---|---|---|---|
| Taxon | Common Name | Abundance | Frequency | Frequency |
| Hemiptera, Aphidoidea | Aphid | 10,792 | 80 | 35.71 |
| Hymenoptera, Formicidae, Others | Other Ants | 907 | 37 | 16.52 |
| Hymenoptera, Formicidae, Monomorium minimum | Little Black Ant | 855 | 74 | 33.04 |
| Hymenoptera, Formicidae, Solenopsis invicta | Red Imported Fire Ant | 633 | 69 | 30.80 |
| Coleoptera, Curculionidae, Baridinae | Flower Weevil | 272 | 40 | 17.86 |
| Arachnida, Acari, Mites | Mite | 268 | 62 | 27.68 |
| Araneae, Salticidae | Jumping Spider | 246 | 116 | 51.79 |
| Coleoptera, Curculionidae, Molytinae | Stem Weevil | 167 | 42 | 18.75 |
| Coleoptera, Dermestidae | Dermestid Beetle | 139 | 28 | 12.50 |
| Hemiptera, Cicadomorpha | Leafhopper | 137 | 77 | 34.38 |
| Coleoptera, Chrysomelidae, Alticini | Flea Beetle | 128 | 48 | 21.43 |
| Diptera, Unknown | Other Flies | 116 | 72 | 32.14 |
| Hemiptera, Lygaeidae, Oncopeltus fasciatus | Large Milkweed Bug | 108 | 38 | 16.96 |
| Aranea, Unknown | Other Spider | 59 | 41 | 18.30 |
| Orthoptera, Caelifera | Grasshopper | 50 | 34 | 15.18 |
| Coleoptera, Chrysomelidae | Other Leaf Beetle | 43 | 29 | 12.95 |
| Coleoptera, Unknown | Other Beetles | 33 | 22 | 9.82 |
| Coleoptera, Curculionidae, Entiminae | Broad-Nosed Weevil | 31 | 20 | 8.93 |
| Thysanoptera | Thrip | 29 | 18 | 8.04 |
| Hemiptera, Heteroptera | Other True Bugs | 28 | 24 | 10.71 |
| Hymenoptera, Apocrita, Unknown Wasps | Wasp | 25 | 20 | 8.93 |
| Arachnida, Opiliones | Harvestman | 24 | 24 | 10.71 |
| Araneae, Araneidae | Orb-weaver Spider | 22 | 19 | 8.48 |
| Diptera, Chironomidae | Midge Fly | 22 | 18 | 8.04 |
| Hemiptera, Lygaeidae, Lygaeus kalmii | Small Milkweed Bug | 19 | 16 | 7.14 |
| Araneae, Oxyopidae | Lynx Spider | 18 | 17 | 7.59 |
| Araneae, Thomisidae | Other Crab Spider | 18 | 15 | 6.70 |
| Hemiptera, Lygaeidae, Unknown | Other Seed Bug | 17 | 9 | 4.02 |
| Araneae, Lycosidae | Wolf Spider | 14 | 12 | 5.36 |
| Othoptera, Tettigoniidae | Katydid | 13 | 12 | 5.36 |
| Myriapoda, Diplopoda | Millipede | 13 | 8 | 3.57 |
| Collembola | Springtail | 12 | 8 | 3.57 |
| Diptera, Muscidae | House Fly | 11 | 11 | 4.91 |
| Araneae, Thomisidae, Misumena vatia | Goldenrod Crab Spider | 11 | 8 | 3.57 |
| Phasmatodea | Stick Insect | 10 | 10 | 4.46 |
| Coleoptera, Coccinellidae, Coccinella septempunctata | Seven-Spotted Ladybeetle | 10 | 10 | 4.46 |
| Coleoptera, Cerambycidae | Longhorn Beetle | 10 | 7 | 3.13 |
| Insecta, Unknown Egg | Insect Egg | 10 | 1 | 0.45 |
| Araneae, Tetragnathidae | Long-jawed Orb Weaver | 8 | 7 | 3.13 |
| Coleoptera, Coccinellidae, Harmonia axyridis | Asian Ladybeetle | 8 | 7 | 3.13 |
| Diptera, Calyptratae | Other Calyptrate Fly | 8 | 5 | 2.23 |
| Hymenoptera, Apidae, Xylocopa sp. | Carpenter Bee | 6 | 6 | 2.68 |
| Hemiptera, Reduviidae | Assassin Bug | 6 | 4 | 1.79 |
| Arachnida, Acari | Tick | 5 | 3 | 1.34 |
| Hemiptera, Coreidae | Leaf-Footed Bug | 5 | 3 | 1.34 |
| Othoptera, Grylidae | Field Cricket | 4 | 4 | 1.79 |
| Coleoptera, Carabidae | Ground Beetle | 4 | 4 | 1.79 |
| Hymenoptera, Anthophila, Unknown | Other Bee | 4 | 4 | 1.79 |
| Blattodea, Isoptera | Termite | 4 | 4 | 1.79 |
| Isopoda | Isopod | 4 | 3 | 1.34 |
| Neuroptera, Adult | Lacewing | 4 | 3 | 1.34 |
| Neuroptera, Larvae | Lacewing Larva | 4 | 2 | 0.89 |
| Araneae, Agelenidae | Grass Spider | 3 | 3 | 1.34 |
| Hemiptera, Miridae | Plant Bug | 3 | 3 | 1.34 |
| Coleoptera, Cantharidae | Soldier Beetle | 3 | 3 | 1.34 |
| Hymenoptera, Apidae, Bombus sp. | Bumblebee | 3 | 3 | 1.34 |
| Hymenoptera, Apidae, Apis sp. | Honey Bee | 3 | 2 | 0.89 |
| Coleoptera, Elateridae | Click Beetle | 2 | 2 | 0.89 |
| Coleoptera, Tenebrionidae | Darkling Beetle | 2 | 2 | 0.89 |
| Coleoptera, Staphylinidae | Rove Beetle | 2 | 2 | 0.89 |
| Hymenoptera, Vespidae | Vespid Wasp | 2 | 2 | 0.89 |
| Diptera, Sarcophagidae | Flesh Fly | 2 | 2 | 0.89 |
| Diptera, Tachinidae | Tachinid Fly | 2 | 2 | 0.89 |
| Diptera, Syrphidae, adult | Flower Fly, adult | 2 | 2 | 0.89 |
| Coleoptera, Coccinellidae, Larva | Ladybeetle Larva | 2 | 2 | 0.89 |
| Araneae, Philodromidae | Running Crab Spider | 2 | 2 | 0.89 |
| Diptera, Tipulidae | Cranefly | 2 | 2 | 0.89 |
| Hemiptera, Pseudococcidae | Mealybug | 2 | 2 | 0.89 |
| Hemiptera, Pentatomidae, Asopinae | Predatory Stink Bug | 2 | 1 | 0.45 |
| Coleoptera, Scarabaeidae | Scarab Beetle | 1 | 1 | 0.45 |
| Hemiptera, Pentatomoidea | Stink bug, non-predatory | 1 | 1 | 0.45 |
| Araneae, Salticidae, Myrmarachne sp. | Ant-mimic Jumping Spider | 1 | 1 | 0.45 |
| Lepidoptera, larva | Caterpillar | 1 | 1 | 0.45 |
| Mecoptera | Scorpion Fly | 1 | 1 | 0.45 |
| Trichoptera | Caddisfly | 1 | 1 | 0.45 |
| Lepidoptera, Heterocera | Moth | 1 | 1 | 0.45 |
| Coleoptera, Curculionoidea, Unknown | Other Weevil | 1 | 1 | 0.45 |
References
- Gustafsson, K.M.; Agrawal, A.A.; Lewenstein, B.V.; Wolf, S.A. The Monarch Butterfly through Time and Space: The Social Construction of an Icon. BioScience 2015, 65, 612–622. [Google Scholar] [CrossRef]
- Flockhart, D.T.T.; Wassenaar, L.I.; Martin, T.G.; Hobson, K.A.; Wunder, M.B.; Norris, D.R. Tracking multi-generational colonization of the breeding grounds by monarch butterflies in eastern North America. Proc. R. Soc. B Biol. Sci. 2013, 280, 20131087. [Google Scholar] [CrossRef] [PubMed]
- Nail, K. Monarch (Danaus plexippus) Species Status Assessment Report, Version 2.1 September 2020; U.S. Fish and Wildlife Service: Washington, DC, USA, 2020; p. 126. Available online: https://www.fws.gov/savethemonarch/pdfs/Monarch-SSA-report.pdf (accessed on 19 May 2021).
- Lyons, J.I.; Pierce, A.A.; Barribeau, S.M.; Sternberg, E.D.; Mongue, A.J.; Roode, J.C.D. Lack of genetic differentiation between monarch butterflies with divergent migration destinations. Mol. Ecol. 2012, 21, 3433–3444. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.A. The Butterflies of North America: A Natural History and Field Guide; Stanford University Press: Palo Alto, CA, USA, 1992; ISBN 978-0-8047-2013-7. [Google Scholar]
- Thogmartin, W.E.; Wiederholt, R.; Oberhauser, K.; Drum, R.G.; Diffendorfer, J.E.; Altizer, S.; Taylor, O.R.; Pleasants, J.; Semmens, D.; Semmens, B.; et al. Monarch butterfly population decline in North America: Identifying the threatening processes. R. Soc. Open Sci. 2017, 4, 170760. [Google Scholar] [CrossRef]
- Brower, L.P.; Williams, E.H.; Dunford, K.S.; Dunford, J.C.; Knight, A.L.; Daniels, J.; Cohen, J.A.; Van Hook, T.; Saarinen, E.; Standridge, M.J.; et al. A long-term survey of spring monarch butterflies in north-central Florida. J. Nat. Hist. 2018, 52, 2025–2046. [Google Scholar] [CrossRef]
- Petition for Protection of the Monarch Butterfly under the Endangered Species Act|Xerces Society. Available online: https://xerces.org/publications/policy-statements/petition-for-esa-protection-monarchs (accessed on 17 February 2021).
- Ethier, D.M. Population Trends of Monarch Butterflies (Lepidoptera: Nymphalidae) Migrating from the Core of Canada’s Eastern Breeding Population. Ann. Entomol. Soc. Am. 2020, 113, 461–467. [Google Scholar]
- Badgett, G.; Davis, A.K. Population Trends of Monarchs at a Northern Monitoring Site: Analyses of 19 Years of Fall Migration Counts at Peninsula Point, MI. Ann. Entomol. Soc. Am. 2015, 108, 700–706. [Google Scholar] [CrossRef]
- Davis, A.K. Are migratory monarchs really declining in eastern North America? Examining evidence from two fall census programs. Insect Conserv. Divers. 2012, 5, 101–105. [Google Scholar] [CrossRef]
- Monarch Joint Venture. 2018 Monarch Conservation Implementation Plan; Monarch Joint Venture: St. Paul, MN, USA, 2018; Available online: https://monarchjointventure.org/our-work/2018-monarch-conservation-implementation-plan (accessed on 19 May 2021).
- Diffendorfer, J.E.; Loomis, J.B.; Ries, L.; Oberhauser, K.; Lopez-Hoffman, L.; Semmens, D.; Semmens, B.; Butterfield, B.; Bagstad, K.; Goldstein, J. National valuation of monarch butterflies indicates an untapped potential for incentive-based conservation. Conserv. Lett. 2014, 7, 253–262. [Google Scholar] [CrossRef]
- Dockx, C. Differences in phenotypic traits and migratory strategies between eastern North American monarch butterflies, Danaus plexippus (L.): Migration of Eastern North American Monarchs. Biol. J. Linn. Soc. 2012, 106, 717–736. [Google Scholar] [CrossRef][Green Version]
- Vidal, O.; Rendón-Salinas, E. Dynamics and trends of overwintering colonies of the monarch butterfly in Mexico. Biol. Conserv. 2014, 180, 165–175. [Google Scholar] [CrossRef]
- Castañeda, S.; Botello, F.; Sánchez-Cordero, V.; Sarkar, S. Spatio-Temporal Distribution of Monarch Butterflies along Their Migratory Route. Front. Ecol. Evol. 2019, 7, 7. [Google Scholar] [CrossRef]
- Malcolm, S.B.; Cockrell, B.J.; Brower, L.P. Spring recolonization of eastern North America by the monarch butterfly: Successive brood or single sweep migration. In Biology and Conservation of the Monarch Butterfly; (Science Series, No. 38); Natural History Museum of Los Angeles County: Los Angeles, CA, USA, 1993; pp. 253–267. [Google Scholar]
- Flockhart, D.T.T.; Pichancourt, J.-B.; Norris, D.R.; Martin, T.G. Unravelling the annual cycle in a migratory animal: Breeding-season habitat loss drives population declines of monarch butterflies. J. Anim. Ecol. 2015, 84, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Oberhauser, K.; Wiederholt, R.; Diffendorfer, J.E.; Semmens, D.; Ries, L.; Thogmartin, W.E.; Lopez-Hoffman, L.; Semmens, B. A trans-national monarch butterfly population model and implications for regional conservation priorities: Regional monarch conservation priorities. Ecol. Entomol. 2017, 42, 51–60. [Google Scholar] [CrossRef]
- Crewe, T.L.; Mitchell, G.W.; Larrivée, M. Size of the Canadian Breeding Population of Monarch Butterflies Is Driven by Factors Acting During Spring Migration and Recolonization. Front. Ecol. Evol. 2019, 7, 7. [Google Scholar] [CrossRef]
- Zipkin, E.F.; Ries, L.; Reeves, R.; Regetz, J.; Oberhauser, K.S. Tracking climate impacts on the migratory monarch butterfly. Glob. Change Biol. 2012, 18, 3039–3049. [Google Scholar] [CrossRef] [PubMed]
- Nail, K.R.; Stenoien, C.; Oberhauser, K.S. Immature Monarch Survival: Effects of Site Characteristics, Density, and Time. Ann. Entomol. Soc. Am. 2015, 108, 680–690. [Google Scholar] [CrossRef]
- Calvert, W.H. Fire ant predation on monarch larvae (Nymphalidae: Danainae) in a central Texas prairie. J. Lepid. Soc. 1996, 50, 149–151. [Google Scholar]
- Lynch, S.P.; Martin, R.A. Milkweed host plant utilization and cardenolide sequestration by monarch butterflies in Louisiana and Texas. In Biology and Conservation of the Monarch Butterfly; (Science Series, No. 38); Natural History Museum of Los Angeles County: Los Angeles, CA, USA, 1993; pp. 107–124. [Google Scholar]
- Calvert, W.H. The effect of fire ants on monarchs breeding in Texas. In The Monarch Butterfly: Biology and Conservation; Cornell University Press: Ithaca, NY, USA, 2004; pp. 47–53. [Google Scholar]
- Myers, A.; Bahlai, C.A.; Landis, D.A. Habitat Type Influences Danaus plexippus (Lepidoptera: Nymphalidae) Oviposition and Egg Survival on Asclepias syriaca (Gentianales: Apocynaceae). Environ. Entomol. 2019, 48, 675–684. [Google Scholar] [CrossRef]
- Koch, R.L.; Venette, R.C.; Hutchison, W.D. Influence of Alternate Prey on Predation of Monarch Butterfly (Lepidoptera: Nymphalidae) Larvae by the Multicolored Asian Lady Beetle (Coleoptera: Coccinellidae). Environ. Entomol. 2005, 34, 410–416. [Google Scholar] [CrossRef]
- Baker, A.M.; Potter, D.A. Invasive paper wasp turns urban pollinator gardens into ecological traps for monarch butterfly larvae. Sci. Rep. 2020, 10, 9553. [Google Scholar] [CrossRef]
- Nestle, R.; Daniels, J.C.; Dale, A.G. Mixed-Species Gardens Increase Monarch Oviposition without Increasing Top-Down Predation. Insects 2020, 11, 648. [Google Scholar] [CrossRef] [PubMed]
- Prysby, M.D. Natural enemies and survival of monarch eggs and larvae. In The Monarch Butterfly: Biology and Conservation; Cornell University Press: Ithaca, NY, USA, 2004; pp. 27–37. [Google Scholar]
- McCoshum, S.M.; Andreoli, S.L.; Stenoien, C.M.; Oberhauser, K.S.; Baum, K.A. Species distribution models for natural enemies of monarch butterfly (Danaus plexippus) larvae and pupae: Distribution patterns and implications for conservation. J. Insect Conserv. 2016, 20, 223–237. [Google Scholar] [CrossRef]
- Myers, A.T.; Haan, N.L.; Landis, D.A. Video surveillance reveals a community of largely nocturnal Danaus plexippus (L.) egg predators. J. Insect Conserv. 2020, 24, 731–737. [Google Scholar] [CrossRef]
- De Anda, A.; Oberhauser, K.S. Invertebrate natural enemies and stage-specific mortality rates of monarch eggs and larvae. In Monarchs in a Changing World: Biology and Conservation of an Iconic Butterfly; Cornell University Press: Ithaca, NY, USA, 2015; pp. 60–70. ISBN 978-0-8014-5559-9. [Google Scholar]
- Hermann, S.L.; Blackledge, C.; Haan, N.L.; Myers, A.T.; Landis, D.A. Predators of monarch butterfly eggs and neonate larvae are more diverse than previously recognised. Sci. Rep. 2019, 9, 14304. [Google Scholar] [CrossRef]
- Myers, A.T. The Interacting Influences of Habitat Context and Predators on Monarch Butterfly (Danaus plexippus L.) Oviposition and Survival in Agricultural Landscapes. Ph.D. Thesis, Michigan State University, East Lansing, MI, USA, 2019. [Google Scholar]
- Oberhauser, K.S.; Anderson, M.; Anderson, S.; Caldwell, W.; De Anda, A.; Hunter, M.; Kaiser, M.C.; Solensky, M.J.; De Anda, A.; Hunter, M.D. Lacewings, wasps, and flies—oh my: Insect enemies take a bite out of monarchs. In Monarchs in a Changing World: Biology and Conservation of an Iconic Insect; Cornell University Press: Ithaca, NY, USA, 2015; pp. 71–82. [Google Scholar]
- Oberhauser, K.S.; Prysby, M.D.; Mattila, H.R.; Stanley-Horn, D.E.; Sears, M.K.; Dively, G.; Olson, E.; Pleasants, J.M.; Lam, W.-K.F.; Hellmich, R.L. Temporal and spatial overlap between monarch larvae and corn pollen. Proc. Natl. Acad. Sci. USA 2001, 98, 11913–11918. [Google Scholar] [CrossRef] [PubMed]
- Deal, K.A. Impact of Landscape Diversity and Invertebrate Community Dynamics on Monarch Butterfly (Danaus plexippus) Oviposition and Survival. Master’s Thesis, Oklahoma State University, Stillwater, OK, USA, 2017. [Google Scholar]
- Pleasants, J.M.; Zalucki, M.P.; Oberhauser, K.S.; Brower, L.P.; Taylor, O.R.; Thogmartin, W.E. Interpreting surveys to estimate the size of the monarch butterfly population: Pitfalls and prospects. PLoS ONE 2017, 12, e0181245. [Google Scholar] [CrossRef]
- Goddard, M.A.; Dougill, A.J.; Benton, T.G. Scaling up from gardens: Biodiversity conservation in urban environments. Trends Ecol. Evol. 2010, 25, 90–98. [Google Scholar] [CrossRef]
- Robertson, B.A.; Rehage, J.S.; Sih, A. Ecological novelty and the emergence of evolutionary traps. Trends Ecol. Evol. 2013, 28, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Lepczyk, C.A.; Aronson, M.F.J.; Evans, K.L.; Goddard, M.A.; Lerman, S.B.; MacIvor, J.S. Biodiversity in the City: Fundamental Questions for Understanding the Ecology of Urban Green Spaces for Biodiversity Conservation. BioScience 2017, 67, 799–807. [Google Scholar] [CrossRef]
- Zalucki, M.P.; Kitching, R.L. Temporal and spatial variation of mortality in field populations of Danaus plexippus L. and D. chrysippus L. Larvae (Lepidoptera: Nymphalidae). Oecologia 1982, 53, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Zalucki, M.P.; Brower, L.P.; Malcolm, S.B. Oviposition by Danaus plexippus in relation to cardenolide content of three Asclepias species in the southeastern USA. Ecol. Entomol. 1990, 15, 231–240. [Google Scholar] [CrossRef]
- Borkin, S.S. Notes on Shifting Distribution Patterns and Survival of Immature Danaus plexippus (Lepidoptera: Danaidae) on the Food Plant Asclepias syriaca. Great Lakes Entomol. 1982, 15, 9. [Google Scholar]
- Rawlins, J.E.; Lederhouse, R.C. Developmental Influences of Thermal Behavior on Monarch Caterpillars (Danaus plexippus): An Adaptation for Migration (Lepidoptera: Nymphalidae: Danainae). J. Kans. Entomol. Soc. 1981, 54, 387–408. [Google Scholar]
- Fisher, K.E.; Hellmich, R.L.; Bradbury, S.P. Estimates of common milkweed (Asclepias syriaca) utilization by monarch larvae (Danaus plexippus) and the significance of larval movement. J. Insect Conserv. 2020, 24, 297–307. [Google Scholar] [CrossRef]
- Murtaugh, P.A. In defense of P values. Ecology 2014, 95, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Wagenmakers, E.-J.; Farrell, S. AIC model selection using Akaike weights. Psychon. Bull. Rev. 2004, 11, 192–196. [Google Scholar] [CrossRef]
- Grodsky, S.M.; Saul-Gershenz, L.S.; Moore-O’Leary, K.A.; Whitney, J.P.; Hernandez, R.R. Hare don’t care! Consumption of a rare, desert milkweed containing phytochemicals by the black-tailed jackrabbit. J. Arid Environ. 2020, 174, 103991. [Google Scholar] [CrossRef]
- Li, Y.; Liu, J.S. Robust Variable and Interaction Selection for Logistic Regression and General Index Models. J. Am. Stat. Assoc. 2019, 114, 271–286. [Google Scholar] [CrossRef]
- Greenland, S.; Mansournia, M.A.; Altman, D.G. Sparse data bias: A problem hiding in plain sight. BMJ 2016, 352, i1981. [Google Scholar] [CrossRef] [PubMed]
- Geest, E.A.; Wolfenbarger, L.L.; McCarty, J.P. Recruitment, survival, and parasitism of monarch butterflies (Danaus plexippus) in milkweed gardens and conservation areas. J. Insect Conserv. 2019, 23, 211–224. [Google Scholar] [CrossRef]
- Grant, T.J.; Flockhart, D.T.T.; Blader, T.R.; Hellmich, R.L.; Pitman, G.M.; Tyner, S.; Norris, D.R.; Bradbury, S.P. Estimating arthropod survival probability from field counts: A case study with monarch butterflies. Ecosphere 2020, 11, e03082. [Google Scholar] [CrossRef]
- Cohen, J.A.; Brower, L.P. Oviposition and Larval Success of Wild Monarch Butterflies (Lepidoptera: Danaidae) in Relation to Host Plant Size and Cardenolide Concentration. J. Kans. Entomol. Soc. 1982, 55, 343–348. [Google Scholar]
- Zalucki, M.P.; Brower, L.P. Survival of first instar larvae of Danaus plexippus (Lepidoptera: Danainae) in relation to cardiac glycoside and latex content of Asclepias humistrata (Asclepiadaceae). Chemoecology 1992, 3, 81–93. [Google Scholar] [CrossRef]
- Haan, N.L.; Landis, D.A. Grassland disturbance increases monarch butterfly oviposition and decreases arthropod predator abundance. Biol. Conserv. 2019, 233, 185–192. [Google Scholar] [CrossRef]
- Manly, B.F. Stage-structured populations. In Stage-Structured Populations; Springer: Berlin/Heidelberg, Germany, 1990; pp. 1–6. [Google Scholar]
- Johnson, E.A.; Miyanishi, K.; Kleb, H. The hazards of interpretation of static age structures as shown by stand reconstructions in a Pinus contorta–Picea engelmannii forest. J. Ecol. 1994, 82, 923–931. [Google Scholar] [CrossRef]
- Prysby, M.D.; Oberhauser, K.S. Temporal and Geographic Variation in Monarch Densities: Citizen Scientists Document Monarch Population Patterns. In The Monarch Butterfly: Biology and Conservation; Cornell University Press: Ithaca, NY, USA, 2004; pp. 9–12. ISBN 978-0-8014-4188-2. [Google Scholar]
- Cortez-Madrigal, H.; García-González, F.; Guzmán-Larralde, A.; Acuña-Soto, J.A.; Otero-Colina, G. Conserving Phytophagous Arthropods to Conserve Natural Enemies: Asclepias curassavica as the Model. Southwest. Entomol. 2016, 41, 681–692. [Google Scholar] [CrossRef]
- Cortez-Madrigal, H.; García-González, F.; Guzmán-Larralde, A. Preserving the monarch butterfly (Danaus plexippus L.), preserving natural enemies of pest. Rev. Chapingo Ser. Cienc. For. Ambiente 2014, 20, 247–253. [Google Scholar]
- Betz, R.F.; Rommel, W.R.; Dichtl, J.J. Insect herbivores of 12 milkweed (Asclepias) species. In Proceedings of the Fifteenth North American Prairie Conference; The Natural Areas Association Bend: Bend, OR, USA, 1997; pp. 7–19. Available online: http://digital.library.wisc.edu/1711.dl/EcoNatRes.NAPC15 (accessed on 19 May 2021).
- Fishbein, M.; Venable, D.L. Diversity and Temporal Change in the Effective Pollinators of Asclepias Tuberosa. Ecology 1996, 77, 1061–1073. [Google Scholar] [CrossRef]
- Liaw, S.-W. The Structure of the Gynostegium, Breeding System, and Pollination Ecology of Spider Milkweed, Asclepias viridis Walter (Apocynaceae). Okla. Native Plant Rec. 2016, 16, 10–44. [Google Scholar] [CrossRef]
- Vasconcellos-Neto, J.; Messas, Y.F.; da Silva Souza, H.; Villanueva-Bonila, G.A.; Romero, G.Q. Spider–plant interactions: An ecological approach. In Behaviour and Ecology of Spiders; Springer: Berlin/Heidelberg, Germany, 2017; pp. 165–214. [Google Scholar]
- Contreras, K. Pathology of the Green-Flowered Milkweed (Asclepias viridis) and Its Relationship to Arthropods and Monarch Butterfly (Danaus plexippus) Survivorship. Master’s Thesis, Texas A M University-Commerce, Commerce, TX, USA, 2018. [Google Scholar]
- Lehtonen, J.; Jaatinen, K. Safety in numbers: The dilution effect and other drivers of group life in the face of danger. Behav. Ecol. Sociobiol. 2016, 70, 449–458. [Google Scholar] [CrossRef]
- Smithers, C.N. A note on natural enemies of “Danaus plexippus” (L.) (Lepidoptera: Nymphalide) in Australia. Aust. Entomol. 1973, 1, 37–40. [Google Scholar]
- Finlay-Doney, M.; Walter, G.H. The conceptual and practical implications of interpreting diet breadth mechanistically in generalist predatory insects. Biol. J. Linn. Soc. 2012, 107, 737–763. [Google Scholar] [CrossRef]
- Mezőfi, L.; Markó, G.; Nagy, C.; Korányi, D.; Markó, V. Beyond polyphagy and opportunism: Natural prey of hunting spiders in the canopy of apple trees. PeerJ 2020, 8, e9334. [Google Scholar] [CrossRef] [PubMed]
- Nyffeler, M. Prey Selection of Spiders in the Field. J. Arachnol. 1999, 27, 317–324. [Google Scholar]
- Bianchi, F.J.J.A.; Schellhorn, N.A.; Werf, W.V.D. Foraging behaviour of predators in heterogeneous landscapes: The role of perceptual ability and diet breadth. Oikos 2009, 118, 1363–1372. [Google Scholar] [CrossRef]
- Isman, M.B.; Duffey, S.S.; Scudder, G.G.E. Cardenolide content of some leaf- and stem-feeding insects on temperate North American milkweeds (Asclepias spp.). Can. J. Zool. 1977, 55, 1024–1028. [Google Scholar] [CrossRef]
- Rayor, L.S. Effects of monarch larval host plant chemistry and body size on Polistes wasp predation. In The Monarch Butterfly: Biology and Conservation; Cornell University Press: Ithaca, NY, USA, 2004; pp. 39–46. [Google Scholar]
- Wootton, J.T. The nature and consequences of indirect effects in ecological communities. Annu. Rev. Ecol. Syst. 1994, 25, 443–466. [Google Scholar] [CrossRef]
- Mittelbach, G.G.; McGill, B.J. Community Ecology; Oxford University Press: Oxford, UK, 2019; ISBN 978-0-19-257286-8. [Google Scholar]
- Vidal, M.C.; Murphy, S.M. Bottom-up vs. top-down effects on terrestrial insect herbivores: A meta-analysis. Ecol. Lett. 2018, 21, 138–150. [Google Scholar] [CrossRef]
- Frago, E.; Godfray, H.C.J. Avoidance of intraguild predation leads to a long-term positive trait-mediated indirect effect in an insect community. Oecologia 2014, 174, 943–952. [Google Scholar] [CrossRef]
- Prado, S.G.; Frank, S. Optimal foraging by an aphid parasitoid affects the outcome of apparent competition. Ecol. Entomol. 2014, 39, 236–244. [Google Scholar] [CrossRef]
- Ali, J.G.; Agrawal, A.A. Asymmetry of plant-mediated interactions between specialist aphids and caterpillars on two milkweeds. Funct. Ecol. 2014, 28, 1404–1412. [Google Scholar] [CrossRef]
- Hertzog, L.R.; Meyer, S.T.; Weisser, W.W.; Ebeling, A. Experimental manipulation of grassland plant diversity induces complex shifts in aboveground arthropod diversity. PLoS ONE 2016, 11, e0148768. [Google Scholar] [CrossRef]
- Welti, E.; Helzer, C.; Joern, A. Impacts of plant diversity on arthropod communities and plant–herbivore network architecture. Ecosphere 2017, 8, e01983. [Google Scholar] [CrossRef]
- Danks, H.V. Regional Diversity of Insects in North America. Am. Entomol. 1994, 40, 50–55. [Google Scholar] [CrossRef]
- Forister, M.L.; Pelton, E.M.; Black, S.H. Declines in insect abundance and diversity: We know enough to act now. Conserv. Sci. Pract. 2019, 1, e80. [Google Scholar] [CrossRef]
- Wagner, D.L. Insect Declines in the Anthropocene. Annu. Rev. Entomol. 2020, 65, 457–480. [Google Scholar] [CrossRef]
- Van Klink, R.; Bowler, D.E.; Gongalsky, K.B.; Swengel, A.B.; Gentile, A.; Chase, J.M. Meta-analysis reveals declines in terrestrial but increases in freshwater insect abundances. Science 2020, 368, 417–420. [Google Scholar] [CrossRef]
- Hallmann, C.A.; Sorg, M.; Jongejans, E.; Siepel, H.; Hofland, N.; Schwan, H.; Stenmans, W.; Müller, A.; Sumser, H.; Hörren, T.; et al. More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 2017, 12, e0185809. [Google Scholar] [CrossRef]
| Taxon | Common Name | Total Abundance | Frequency | Percent Frequency |
|---|---|---|---|---|
| Hemiptera, Aphidoidea | Aphid | 10,792 | 80 | 35.71 |
| Hymenoptera, Formicidae, Others | Other Ants | 907 | 37 | 16.52 |
| Hymenoptera, Formicidae, Monomorium minimum | Little Black Ant | 855 | 74 | 33.04 |
| Hymenoptera, Formicidae, Solenopsis invicta | Red Imported Fire Ant | 633 | 69 | 30.80 |
| Coleoptera, Curculionidae, | Weevils | 471 | 67 | 29.91 |
| Arachnida, Acari, Mites | Mites | 268 | 62 | 27.68 |
| Arthropoda, Others | Other Non-Predatory Arthropods | 267 | 111 | 49.55 |
| Araneae, Salticidae | Jumping Spiders | 247 | 116 | 51.79 |
| Arthropoda, Others, Predatory | Other Predatory Arthropods | 227 | 123 | 54.91 |
| Coleoptera, Chrysomelidae | Other Leaf Beetle | 171 | 65 | 29.02 |
| Coleoptera, Dermestidae | Dermestid Beetle | 139 | 28 | 12.50 |
| Diptera | Flies | 138 | 81 | 36.16 |
| Hemiptera, Cicadomorpha | Leafhopper | 137 | 77 | 34.38 |
| Hemiptera, Lygaeidae, Oncopeltus fasciatus | Large Milkweed Bug | 108 | 38 | 16.96 |
| Arthropoda, Others | Other Milkweed Herbivores | 48 | 34 | 15.18 |
| Coleoptera, Others | All Other Beetles | 33 | 22 | 9.82 |
| Model | AICc | ΔAICc | wi | Likelihood Ratio X2 | Model Probability |
|---|---|---|---|---|---|
| Other Non-Predatory Arthropods, Mites, Other Ants | 251.635 | 0.000 | 0.417 | 16.8877 | 0.0007 |
| Other Non-predatory Arthropods, Mites | 251.724 | 0.089 | 0.399 | 14.7246 | 0.0006 |
| Other Non-predatory Arthropods | 253.290 | 1.656 | 0.182 | 11.1039 | 0.0009 |
| Intercept Only | 262.358 | 10.723 | 0.002 | - | - |
| Summary of the best fit model. Concordance of this model was 51.3%. | |||||
| Parameter | DF | Estimate | Standard Error | Wald Chi-Square | p-value |
| Intercept | 1 | −1.4206 | 0.2028 | 49.0425 | 0.0001 |
| Other Non-Predatory Arthropods | 1 | 0.2468 | 0.0775 | 10.1330 | 0.0015 |
| Mites | 1 | 0.0850 | 0.1133 | 0.5631 | 0.4530 |
| Other Ants | 1 | 0.0057 | 0.00772 | 0.5456 | 0.4601 |
| Model | AICc | ΔAICc | wi | Likelihood Ratio X2 | Model Probability |
|---|---|---|---|---|---|
| Number of Non-Predatory Taxa | 256.806 | 0.000 | 0.508 | 7.5880 | 0.0059 |
| Number of Non-Predatory Taxa, Number of Predatory Arthropods | 257.003 | 0.197 | 0.460 | 9.4459 | 0.0089 |
| Intercept Only | 262.358 | 5.552 | 0.032 | - | - |
| Summary of the best fit model. Concordance of this model was 55.5%. | |||||
| Parameter | DF | Estimate | Standard Error | Wald Chi-Square | p-value |
| Intercept | 1 | −1.5450 | 0.2586 | 35.7021 | 0.0001 |
| Number of Non-Predatory Taxa | 1 | 0.1390 | 0.0507 | 7.5190 | 0.0061 |
| Location | Measurement | Value | Equivalent Value in Current Study | Citation |
|---|---|---|---|---|
| Florida | Survival to 3rd instar | 9.2% | 13% | Brower et al. 2018 [7] |
| Florida | Survival to 3rd instar | About 14% | 13% | Cohen and Brower 1982 [55] |
| Florida | Survival to 3rd instar | 17–21% | 13% | Zaluki and Brower 1992 [56] |
| Texas and Louisiana | Survival to 3rd instar | 3% (0% to 40%) | 13% | Lynch and Martin 1993 [24] |
| Texas | Survival to 3rd instar | 0% | 13% | Calvert 1996 [23] |
| Texas | Survival to 3rd instar | 0.24% | 13% | Calvert 2004 [25] |
| Minnesota | Daily survival rate, survival to third instar | 0.56, 1.7% | 0.896, 13% | De Anda and Oberhauser 2015 [33] |
| Wisconsin | Survival to hatching | 35% | 63.3% | Borkin 1982 [45] |
| Wisconsin | Seven-day survival rate | 18% | 46% | Prysby 2004 [30] |
| Michigan | 48-h survival rate of first instars | 15% to 40% | 80% | Haan and Landis 2019 [57] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stevenson, M.; Hudman, K.L.; Scott, A.; Contreras, K.; Kopachena, J.G. High Survivorship of First-Generation Monarch Butterfly Eggs to Third Instar Associated with a Diverse Arthropod Community. Insects 2021, 12, 567. https://doi.org/10.3390/insects12060567
Stevenson M, Hudman KL, Scott A, Contreras K, Kopachena JG. High Survivorship of First-Generation Monarch Butterfly Eggs to Third Instar Associated with a Diverse Arthropod Community. Insects. 2021; 12(6):567. https://doi.org/10.3390/insects12060567
Chicago/Turabian StyleStevenson, Misty, Kalynn L. Hudman, Alyx Scott, Kelsey Contreras, and Jeffrey G. Kopachena. 2021. "High Survivorship of First-Generation Monarch Butterfly Eggs to Third Instar Associated with a Diverse Arthropod Community" Insects 12, no. 6: 567. https://doi.org/10.3390/insects12060567
APA StyleStevenson, M., Hudman, K. L., Scott, A., Contreras, K., & Kopachena, J. G. (2021). High Survivorship of First-Generation Monarch Butterfly Eggs to Third Instar Associated with a Diverse Arthropod Community. Insects, 12(6), 567. https://doi.org/10.3390/insects12060567






