Differences in Life History Traits in Rural vs. Urban Populations of a Specialist Ground Beetle, Carabus convexus
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling Design
2.2. Test Organism
2.3. Evaluating and Measuring Traits
2.4. Statistical Analyses
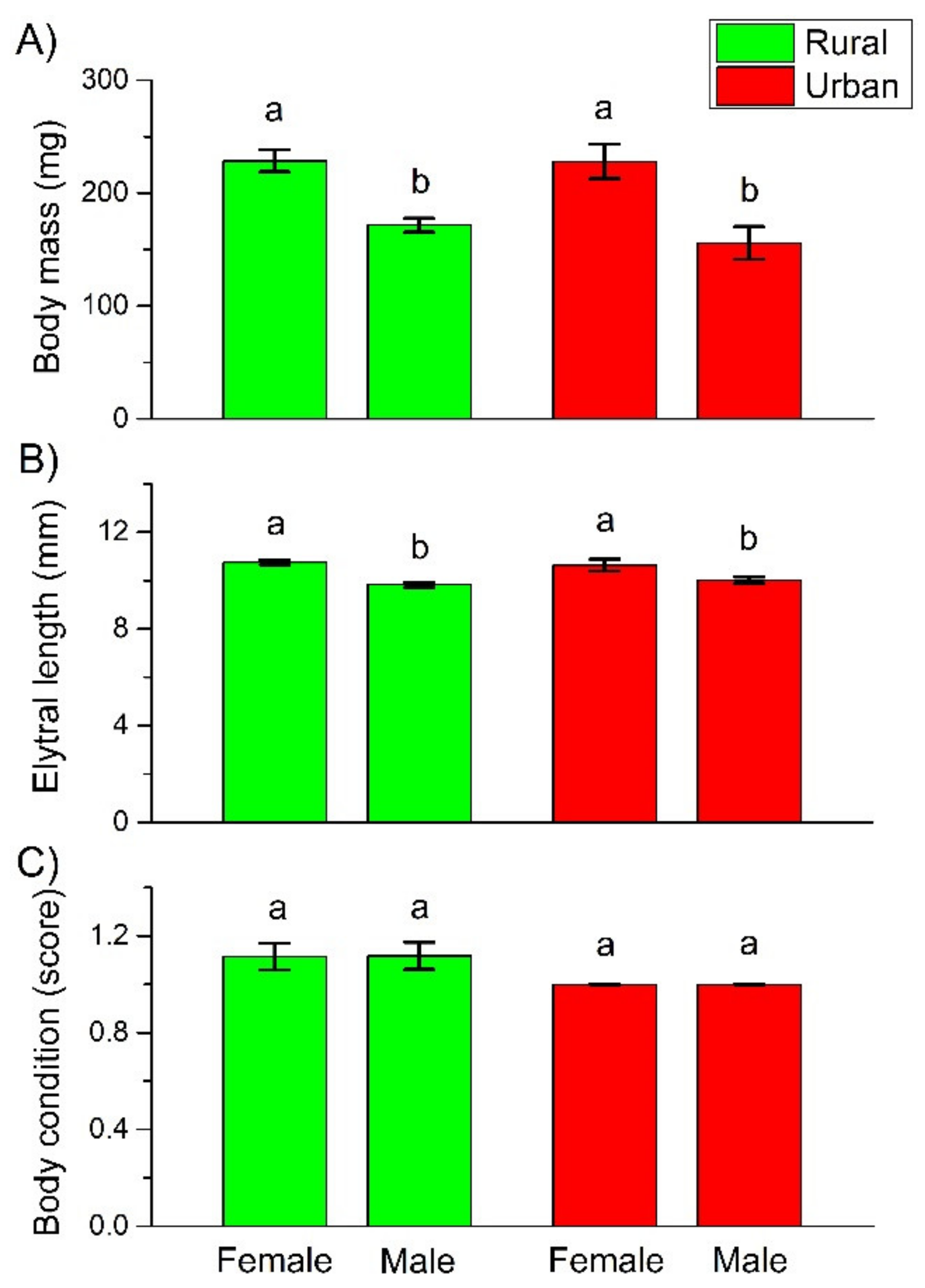
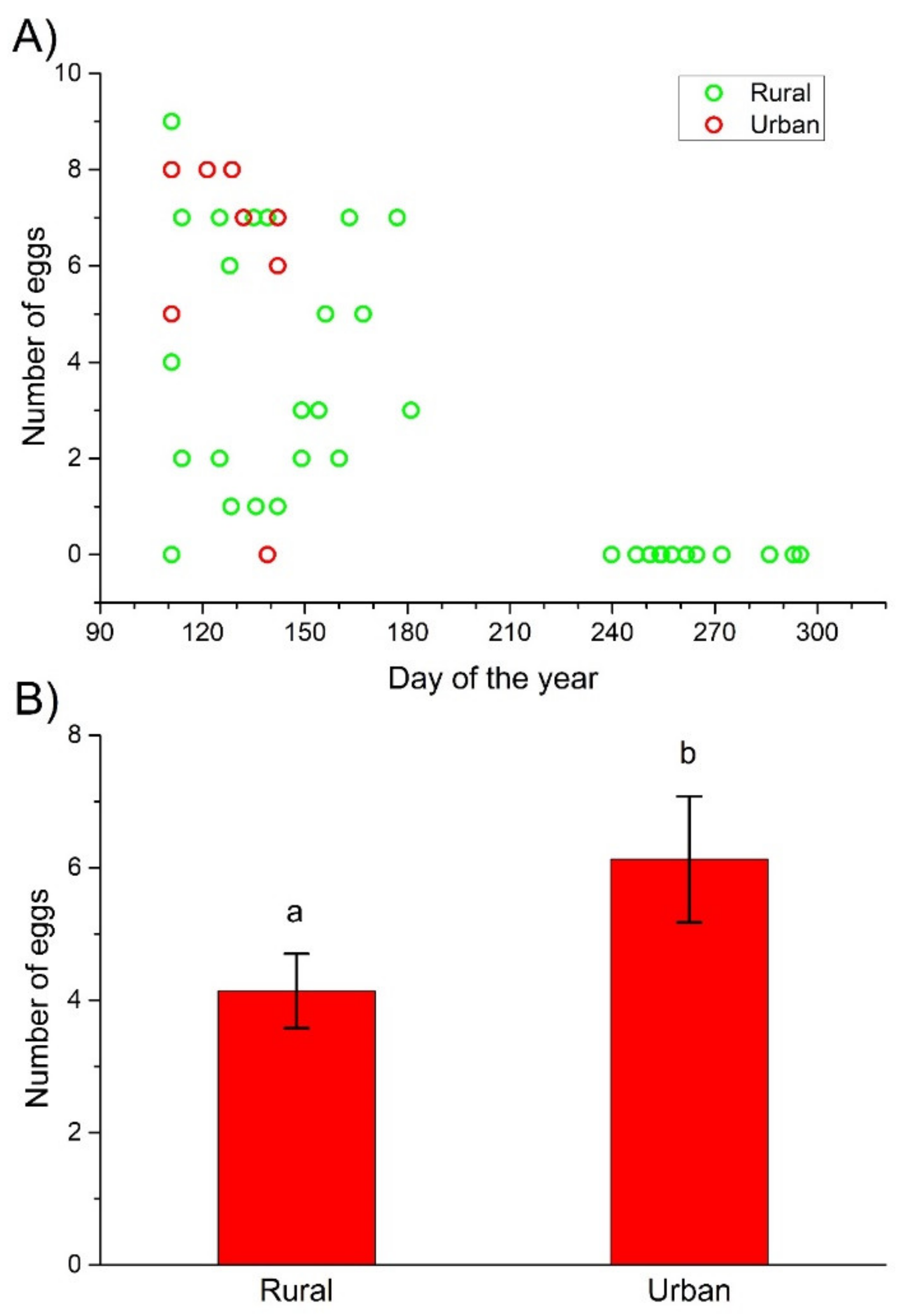
3. Results
4. Discussion
4.1. Beetle Abundance
4.2. Body Mass, Length, and Condition
4.3. Fecundity
4.4. Conserving Ground Beetles in Urban Areas
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hunter, P. The human impact on biological diversity. How species adapt to urban challenges sheds light on evolution and provides clues about conservation. EMBO Rep. 2007, 8, 316–318. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, N.E. Urban ecology—Definitions and goals. In The Routledge Handbook of Urban Ecology; Douglas, I., Goode, D., Houck, M., Wang, R., Eds.; Routledge: London, UK, 2011; pp. 7–16. [Google Scholar]
- Antrop, M. Changing patterns in the urbanized countryside of Western Europe. Landsc. Ecol. 2000, 15, 257–270. [Google Scholar] [CrossRef]
- United Nations World Urbanization Prospects: The 2018 Revision; United Nations, Department of Economic and Social Affairs, Population Division: New York, NY, USA, 2019.
- McCarthy, M.P.; Best, M.J.; Betts, R.A. Climate change in cities due to global warming and urban effects. Geophys. Res. Lett. 2010, 37, L09705. [Google Scholar] [CrossRef]
- Jacobson, C.R. Identification and quantification of the hydrological impacts of imperviousness in urban catchments: A review. J. Environ. Manag. 2011, 92, 1438–1448. [Google Scholar] [CrossRef] [PubMed]
- Fenoglio, M.S.; Calviño, A.; González, E.; Salvo, A.; Videla, M. Urbanisation drivers and underlying mechanisms of terrestrial insect diversity loss in cities. Ecol. Entomol. 2021, 46. in press. [Google Scholar] [CrossRef]
- Simon, E.; Harangi, S.; Baranyai, E.; Braun, M.; Fábián, I.; Mizser, S.; Nagy, L.; Tóthmérész, B. Distribution of toxic elements between biotic and abiotic components of terrestrial ecosystem along an urbanization gradient: Soil, leaf litter and ground beetles. Ecol. Indic. 2016, 60, 258–264. [Google Scholar] [CrossRef]
- McKinney, M.L. Urbanization, biodiversity, and conservation. Bioscience 2002, 52, 883–890. [Google Scholar] [CrossRef]
- McDonnell, M.J.; Pickett, S.T.A.; Groffman, P.; Bohlen, P.; Pouyat, R.V.; Zipperer, W.C.; Parmelee, R.W.; Carreiro, M.M.; Medley, K. Ecosystem processes along an urban-to-rural gradient. Urban Ecosyst. 1997, 1, 21–36. [Google Scholar] [CrossRef]
- Niemelä, J. Ecology and urban planning. Biodivers. Conserv. 1999, 8, 119–131. [Google Scholar] [CrossRef]
- Lowry, H.; Lill, A.; Wong, B.B.M. Behavioural responses of wildlife to urban environments. Biol. Rev. 2013, 88, 537–549. [Google Scholar] [CrossRef]
- Magura, T.; Lövei, G.L. Consequences of urban living: Urbanization and ground beetles. Curr. Landsc. Ecol. Rep. 2021, 6, 9–21. [Google Scholar] [CrossRef]
- Eötvös, C.B.; Magura, T.; Lövei, G.L. A meta-analysis indicates reduced predation pressure with increasing urbanization. Landsc. Urban Plan. 2018, 180, 54–59. [Google Scholar] [CrossRef]
- Zuñiga-Palacios, J.; Zuria, I.; Castellanos, I.; Lara, C.; Sánchez-Rojas, G. What do we know (and need to know) about the role of urban habitats as ecological traps? Systematic review and meta-analysis. Sci. Total Environ. 2021, 780, 146559. [Google Scholar] [CrossRef]
- Sih, A.; Ferrari, M.C.O.; Harris, D.J. Evolution and behavioural responses to human-induced rapid environmental change. Evol. Appl. 2011, 4, 367–387. [Google Scholar] [CrossRef]
- Seto, K.C.; Parnell, S.; Elmqvist, T. A Global Outlook on Urbanization. In Urbanization, Biodiversity and Ecosystem Services: Challenges and Opportunities: A Global Assessment; Elmqvist, T., Fragkias, M., Goodness, J., Güneralp, B., Marcotullio, P.J., McDonald, R.I., Parnell, S., Schewenius, M., Sendstad, M., Seto, K.C., et al., Eds.; Springer: Dordrecht, The Netherland, 2013; pp. 1–12. ISBN 978-94-007-7088-1. [Google Scholar]
- Aronson, M.F.J.; La Sorte, F.A.; Nilon, C.H.; Katti, M.; Goddard, M.A.; Lepczyk, C.A.; Warren, P.S.; Williams, N.S.G.; Cilliers, S.; Clarkson, B.; et al. A global analysis of the impacts of urbanization on bird and plant diversity reveals key anthropogenic drivers. Proc. R. Soc. B Biol. Sci. 2014, 281, 20133330. [Google Scholar] [CrossRef]
- Ives, C.D.; Lentini, P.E.; Threlfall, C.G.; Ikin, K.; Shanahan, D.F.; Garrard, G.E.; Bekessy, S.A.; Fuller, R.A.; Mumaw, L.; Rayner, L.; et al. Cities are hotspots for threatened species. Glob. Ecol. Biogeogr. 2016, 25, 117–126. [Google Scholar] [CrossRef]
- McKinney, M.L. Effects of urbanization on species richness: A review of plants and animals. Urban Ecosyst. 2008, 11, 161–176. [Google Scholar] [CrossRef]
- Magura, T.; Kiss, E.; Lövei, G.L. No consistent diversity patterns in terrestrial mammal assemblages along rural-urban forest gradients. Basic Appl. Ecol. 2021, 52, 38–45. [Google Scholar] [CrossRef]
- Chace, J.F.; Walsh, J.J. Urban effects on native avifauna: A review. Landsc. Urban Plan. 2006, 74, 46–69. [Google Scholar] [CrossRef]
- Batáry, P.; Kurucz, K.; Suarez-Rubio, M.; Chamberlain, D.E. Non-linearities in bird responses across urbanization gradients: A meta-analysis. Glob. Chang. Biol. 2018, 24, 1046–1054. [Google Scholar] [CrossRef]
- Williams, N.S.G.; Hahs, A.K.; Vesk, P.A. Urbanisation, plant traits and the composition of urban floras. Perspect. Plant Ecol. Evol. Syst. 2015, 17, 78–86. [Google Scholar] [CrossRef]
- Singh, A.K.; Singh, H.; Singh, J.S. Plant diversity in cities: Call for assessment and conservation. Curr. Sci. 2018, 115, 428–435. [Google Scholar] [CrossRef]
- Fenoglio, M.S.; Rossetti, M.R.; Videla, M. Negative effects of urbanization on terrestrial arthropod communities: A meta-analysis. Glob. Ecol. Biogeogr. 2020, 29, 1412–1429. [Google Scholar] [CrossRef]
- Wagner, D.L.; Grames, E.M.; Forister, M.L.; Berenbaum, M.R.; Stopak, D. Insect decline in the Anthropocene: Death by a thousand cuts. Proc. Natl. Acad. Sci. USA 2021, 118. [Google Scholar] [CrossRef]
- Niemelä, J.; Kotze, D.J. Carabid beetle assemblages along urban to rural gradients: A review. Landsc. Urban Plan. 2009, 92, 65–71. [Google Scholar] [CrossRef]
- Martinson, H.M.; Raupp, M.J. A meta-analysis of the effects of urbanization on ground beetle communities. Ecosphere 2013, 4, 60. [Google Scholar] [CrossRef]
- Magura, T.; Ferrante, M.; Lövei, G.L. Only habitat specialists become smaller with advancing urbanisation. Glob. Ecol. Biogeogr. 2020, 29, 1978–1987. [Google Scholar] [CrossRef]
- Lövei, G.L.; Sunderland, K.D. Ecology and behavior of ground beetles (Coleoptera: Carabidae). Annu. Rev. Entomol. 1996, 41, 231–256. [Google Scholar] [CrossRef]
- Skłodowski, J.; Bajor, P.; Trynkos, M. Carabids benefit more from pine stands with added understory or second story of broad-leaved trees favored by climate change than from one-storied pine stands. Eur. J. For. Res. 2018, 137, 745–757. [Google Scholar] [CrossRef]
- de Bello, F.; Carmona, C.P.; Dias, A.T.C.; Götzenberger, L.; Moretti, M.; Berg, M.P. Handbook of Trait-Based Ecology: From Theory to R Tools; Cambridge University Press: Cambridge, UK, 2021; ISBN 9781108472913. [Google Scholar]
- Peters, R.H. The Ecological Implications of Body Size; Cambridge University Press: Cambridge, UK, 1983. [Google Scholar]
- Weller, B.; Ganzhorn, J.U. Carabid beetle community composition, body size, and fluctuating asymmetry along an urban-rural gradient. Basic Appl. Ecol. 2004, 5, 193–201. [Google Scholar] [CrossRef]
- Langraf, V.; David, S.; Babosová, R.; Petrovičová, K.; Schlarmannová, J. Change of ellipsoid biovolume (Ev) of ground beetles (coleoptera, carabidae) along an urban–suburban–rural gradient of central slovakia. Diversity 2020, 12, 475. [Google Scholar] [CrossRef]
- Sukhodolskaya, R.A.; Saveliev, A.A. Effects of ecological factors on size-related traits in the ground beetle Carabus granulatus L. (Coleoptera, Carabidae). Russ. J. Ecol. 2014, 45, 414–420. [Google Scholar] [CrossRef]
- Sadler, J.P.; Small, E.C.; Fiszpan, H.; Telfer, M.G.; Niemelä, J. Investigating environmental variation and landscape characteristics of an urban–rural gradient using woodland carabid assemblages. J. Biogeogr. 2006, 33, 1126–1138. [Google Scholar] [CrossRef]
- Elek, Z.; Lövei, G.L.; Bátki, M. Sex-specific interaction of body condition and asymmetry in carabids in distinct urbanisation stages. Community Ecol. 2017, 18, 253–259. [Google Scholar] [CrossRef]
- Magura, T.; Lövei, G.L.; Tóthmérész, B. Does urbanization decrease diversity in ground beetle (Carabidae) assemblages? Glob. Ecol. Biogeogr. 2010, 19, 16–26. [Google Scholar] [CrossRef]
- Liker, A.; Papp, Z.; Bókony, V.; Lendvai, Á.Z. Lean birds in the city: Body size and condition of house sparrows along the urbanization gradient. J. Anim. Ecol. 2008, 77, 789–795. [Google Scholar] [CrossRef]
- Bommarco, R. Reproduction and energy reserves of of a predatory carabid beetle relative to agroecosystem complexity. Ecol. Appl. 1998, 8, 846–853. [Google Scholar] [CrossRef]
- Magura, T.; Lövei, G.L.; Tóthmérész, B. Time-consistent rearrangement of carabid beetle assemblages by an urbanisation gradient in Hungary. Acta Oecol. 2008, 34, 233–243. [Google Scholar] [CrossRef]
- Magura, T.; Tóthmérész, B.; Molnár, T. Changes in carabid beetle assemblages along an urbanisation gradient in the city of Debrecen, Hungary. Landsc. Ecol. 2004, 19, 747–759. [Google Scholar] [CrossRef]
- Molnár, T.; Magura, T.; Tóthmérész, B.; Elek, Z. Ground beetles (Carabidae) and edge effect in oak-hornbeam forest and grassland transects. Eur. J. Soil Biol. 2001, 37, 297–300. [Google Scholar] [CrossRef]
- Turin, H.; Penev, L.; Casale, A. The Genus Carabus in Europe—A Synthesis; Pensoft: Moscow, Russia, 2003. [Google Scholar]
- Magura, T.; Tóthmérész, B.; Molnár, T. A species-level comparison of occurrence patterns in carabids along an urbanisation gradient. Landsc. Urban Plan. 2008, 86, 134–140. [Google Scholar] [CrossRef]
- Niemelä, J.; Kotze, D.J.; Venn, S.; Penev, L.; Stoyanov, I.; Spence, J.; Hartley, D.; de Oca, E.M. Carabid beetle assemblages (Coleoptera, Carabidae) across urban-rural gradients: An international comparison. Landsc. Ecol. 2002, 17, 387–401. [Google Scholar] [CrossRef]
- Szujecki, A.; Szyszko, J.; Mazur, S.; Perliński, S. The Process of Forest Soil Macrofauna Formation after Afforestation of Farmland; Warsaw Agricultural Press: Warsaw, Poland, 1983; ISBN 83-00-018?6-X. [Google Scholar]
- Jelaska, L.Š.; Dumbovich, V.; Kucinic, M. Carabid beetle diversity and mean individual biomass in beech forests of various ages. Zookeys 2011, 100, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Andorkó, R.; Kádár, F. Life-history characteristics of the ground beetle Carabus scheidleri (Coleoptera: Carabidae) in Hungary. Acta Zool. Acad. Sci. Hung. 2009, 55, 381–393. [Google Scholar]
- Bommarco, R. Stage sensitivity to food limitation for a generalist arthropod predator, Pterostichus cupreus (Coleoptera: Carabidae). Environ. Entomol. 1998, 27, 863–869. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. Artic. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Fox, J.; Weisberg, S. An R Companion to Applied Regression, 3rd ed.; SAGE Publications: Thousand Oaks, CA, USA, 2019; ISBN 9781544336473. [Google Scholar]
- Venables, W.; Ripley, B. Modern Applied Statistics with S, 4th ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Zuur, A.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer: New York, NY, USA, 2009. [Google Scholar]
- Weeks, R.D.; McIntyre, N.E. A comparison of live versus kill pitfall trapping techniques using various killing agents. Entomol. Exp. Appl. 1997, 82, 267–273. [Google Scholar] [CrossRef]
- Garbalińska, P.; Skłodowski, J. Body size differentiation in selected carabid species inhabiting Puszcza Piska forest stands disturbed by the hurricane. Baltic J. Coleopterol. 2008, 8, 101–114. [Google Scholar]
- Gray, J.S. Effects of environmental stress on species rich assemblages. Biol. J. Linn. Soc. 1989, 37, 19–32. [Google Scholar] [CrossRef]
- Merckx, T.; Souffreau, C.; Kaiser, A.; Baardsen, L.F.; Backeljau, T.; Bonte, D.; Brans, K.I.; Cours, M.; Dahirel, M.; Debortoli, N.; et al. Body-size shifts in aquatic and terrestrial urban communities. Nature 2018, 558, 113–116. [Google Scholar] [CrossRef]
- Atkinson, D. Temperature and organism size—A biological law for ectotherms? Adv. Ecol. Res. 1994, 25, 1–58. [Google Scholar] [CrossRef]
- Jones, L.E.; Leather, R.S. Invertebrates in urban areas: A review. Eur. J. Entomol. 2012, 109, 463–478. [Google Scholar] [CrossRef]
- Nelemans, M.N.E. Surface activity and growth of larvae of Nebria brevicollis (F.) (Coleoptera, Carabidae). Neth. J. Zool. 1988, 38, 74–95. [Google Scholar] [CrossRef]
- Lenski, R.E. Food limitation and competition: A field experiment with two Carabus species. J. Anim. Ecol. 1984, 53, 203–216. [Google Scholar] [CrossRef]
- Bilde, T.; Toft, S. Quantifying food limitation of arthropod predators in the field. Oecologia 1998, 115, 54–58. [Google Scholar] [CrossRef]
- Loreau, M. Competition in a carabid beetle community: A field experiment. Oikos 1990, 58, 25–38. [Google Scholar] [CrossRef]
- Baars, M.A.; van Dijk, T.S. Population dynamics of two carabid beetles at a Dutch heathland: II. Egg production and survival in relation to density. J. Anim. Ecol. 1984, 53, 389–400. [Google Scholar] [CrossRef]
- Wallin, H.; Chiverton, P.A.; Ekbom, B.S.; Borg, A. Diet, fecundity and egg size in some polyphagous predatory carabid beetles. Entomol. Exp. Appl. 1992, 65, 129–140. [Google Scholar] [CrossRef]
- Elek, Z.; Lövei, G.L. Patterns in ground beetle (Coleoptera: Carabidae) assemblages along an urbanisation gradient in Denmark. Acta Oecol. 2007, 32, 104–111. [Google Scholar] [CrossRef]
- Vergnes, A.; Le Viol, I.; Clergeau, P. Green corridors in urban landscapes affect the arthropod communities of domestic gardens. Biol. Conserv. 2012, 145, 171–178. [Google Scholar] [CrossRef]
- Norton, B.A.; Evans, K.L.; Warren, P.H. Urban biodiversity and landscape ecology: Patterns, processes and planning. Curr. Landsc. Ecol. Rep. 2016, 1, 178–192. [Google Scholar] [CrossRef]
| Response Variable | Fixed Effect | Estimate ± SE | χ2 | df | p |
|---|---|---|---|---|---|
| Body mass | Urbanization level | −0.0555 ± 0.1432 | 0.1499 | 1 | 0.6987 |
| Sex | 0.2614 ± 0.0421 | 38.4839 | 1 | <0.0001 | |
| Urbanization level × Sex | 0.0843 ± 0.1080 | 0.6081 | 1 | 0.4355 | |
| Body length | Urbanization level | 0.0164 ± 0.0328 | 0.2498 | 1 | 0.6172 |
| Sex | 0.0862 ± 0.0132 | 42.6806 | 1 | <0.0001 | |
| Urbanization level × Sex | −0.0267 ± 0.0336 | 0.6303 | 1 | 0.4272 | |
| Body condition | Urbanization level | −0.1112 ± 0.4757 | 0.0547 | 1 | 0.8151 |
| Sex | −0.0030 ± 0.2279 | 0.0002 | 1 | 0.9895 | |
| Urbanization level × Sex | 0.0030 ± 0.6140 | 0.0000 | 1 | 0.9961 | |
| Number of eggs | |||||
| All females | Urbanization level | 0.9673 ± 0.4099 | 5.5702 | 1 | 0.0183 |
| Females before oviposition | Urbanization level | 0.3926 ± 0.1772 | 4.9082 | 1 | 0.0267 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magura, T.; Mizser, S.; Horváth, R.; Nagy, D.D.; Tóth, M.; Csicsek, R.; Lövei, G.L. Differences in Life History Traits in Rural vs. Urban Populations of a Specialist Ground Beetle, Carabus convexus. Insects 2021, 12, 540. https://doi.org/10.3390/insects12060540
Magura T, Mizser S, Horváth R, Nagy DD, Tóth M, Csicsek R, Lövei GL. Differences in Life History Traits in Rural vs. Urban Populations of a Specialist Ground Beetle, Carabus convexus. Insects. 2021; 12(6):540. https://doi.org/10.3390/insects12060540
Chicago/Turabian StyleMagura, Tibor, Szabolcs Mizser, Roland Horváth, Dávid D. Nagy, Mária Tóth, Réka Csicsek, and Gábor L. Lövei. 2021. "Differences in Life History Traits in Rural vs. Urban Populations of a Specialist Ground Beetle, Carabus convexus" Insects 12, no. 6: 540. https://doi.org/10.3390/insects12060540
APA StyleMagura, T., Mizser, S., Horváth, R., Nagy, D. D., Tóth, M., Csicsek, R., & Lövei, G. L. (2021). Differences in Life History Traits in Rural vs. Urban Populations of a Specialist Ground Beetle, Carabus convexus. Insects, 12(6), 540. https://doi.org/10.3390/insects12060540








